Submitted:
26 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
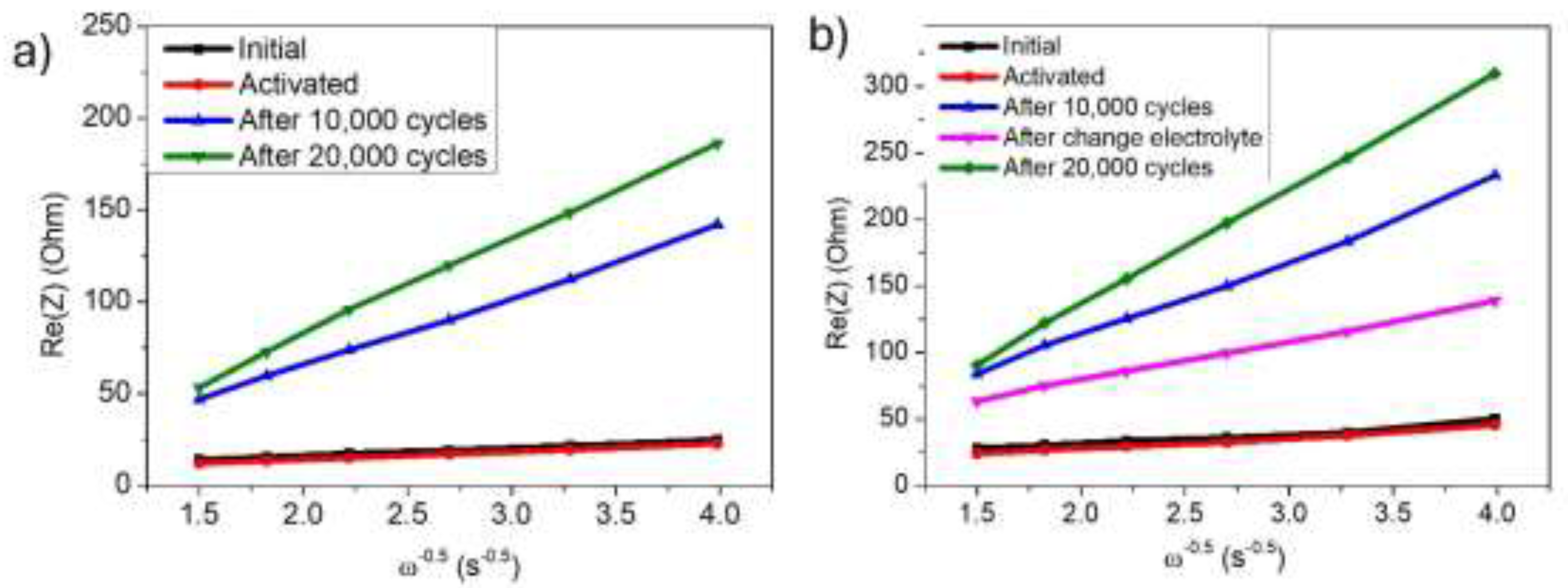
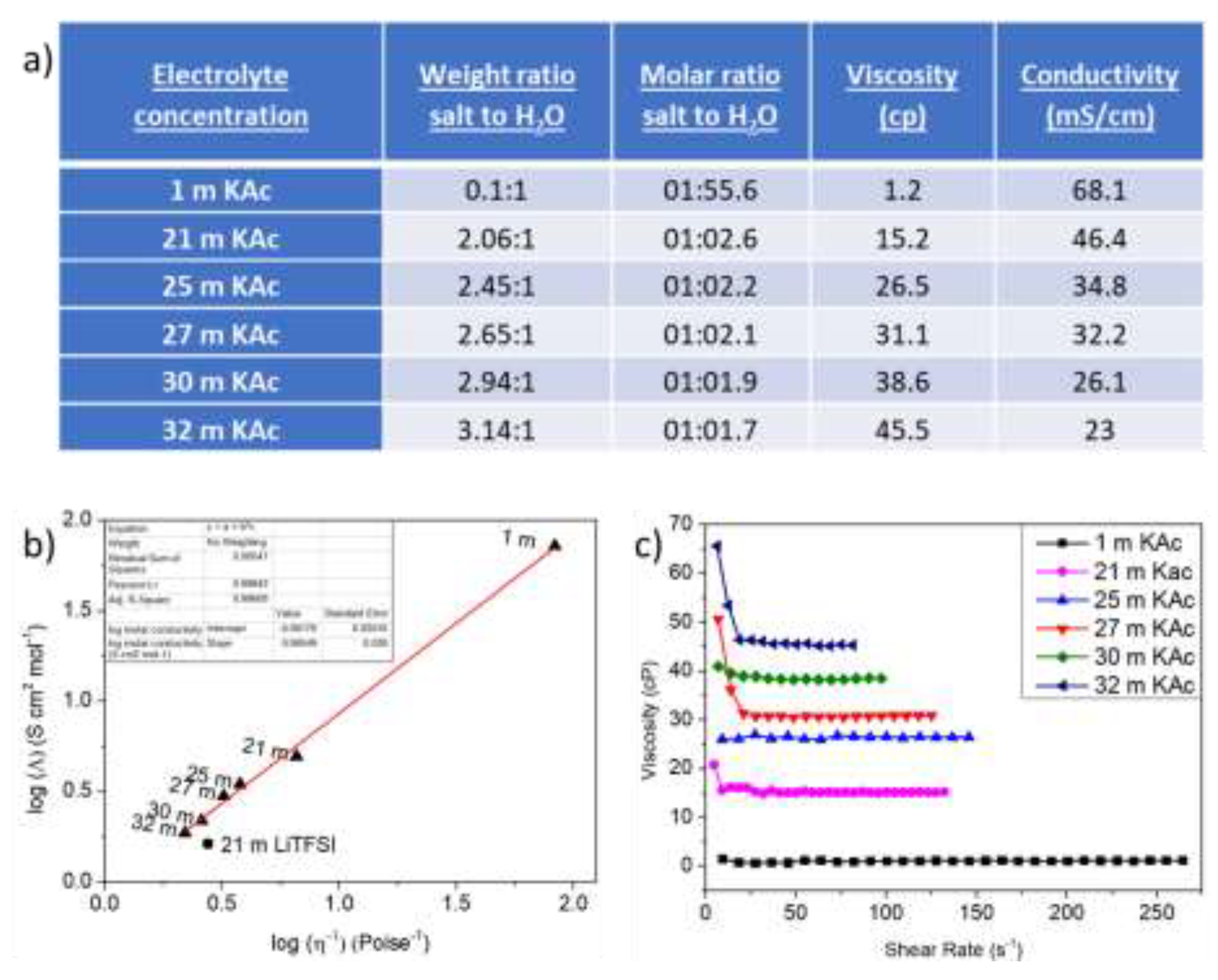
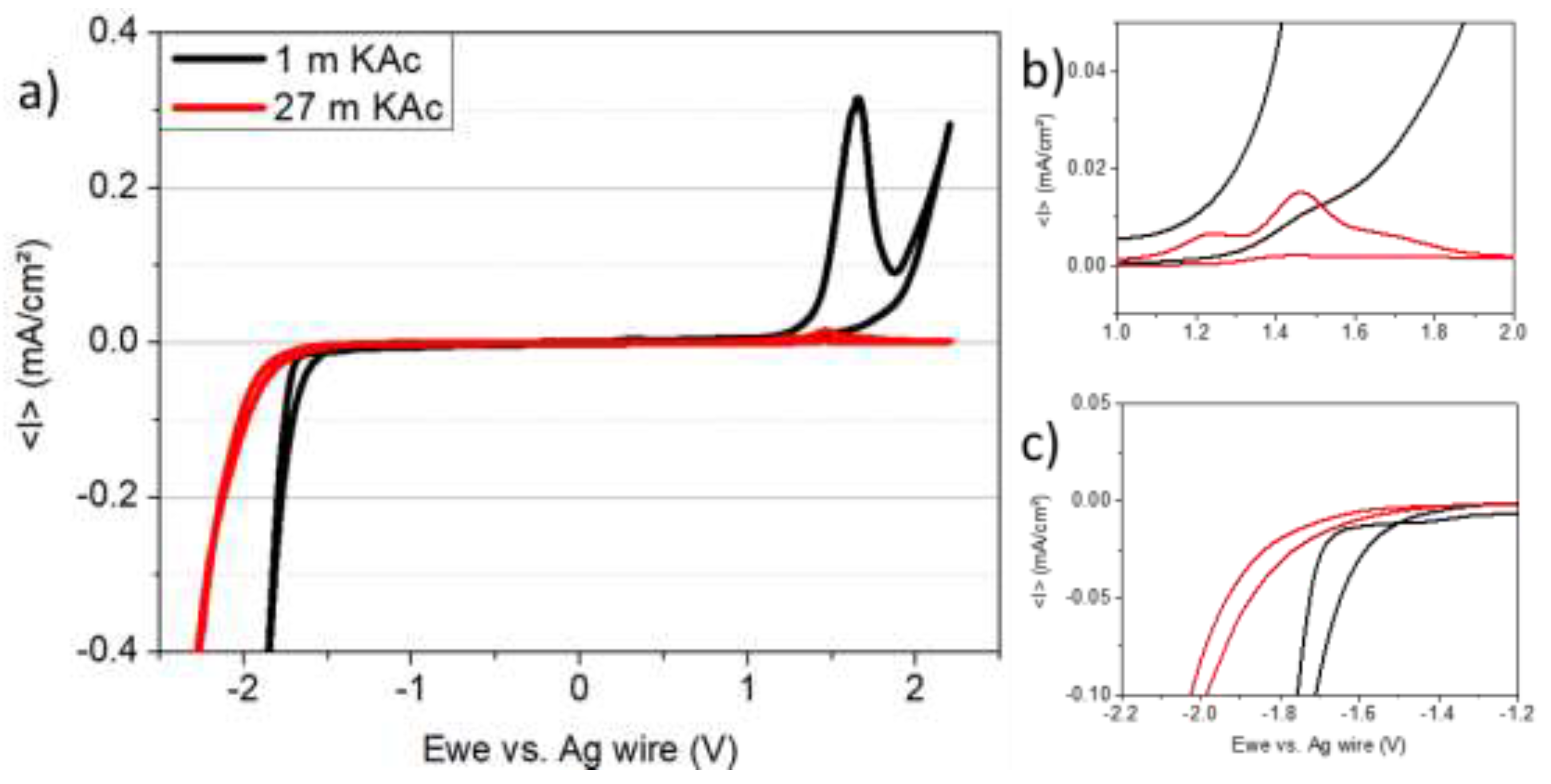
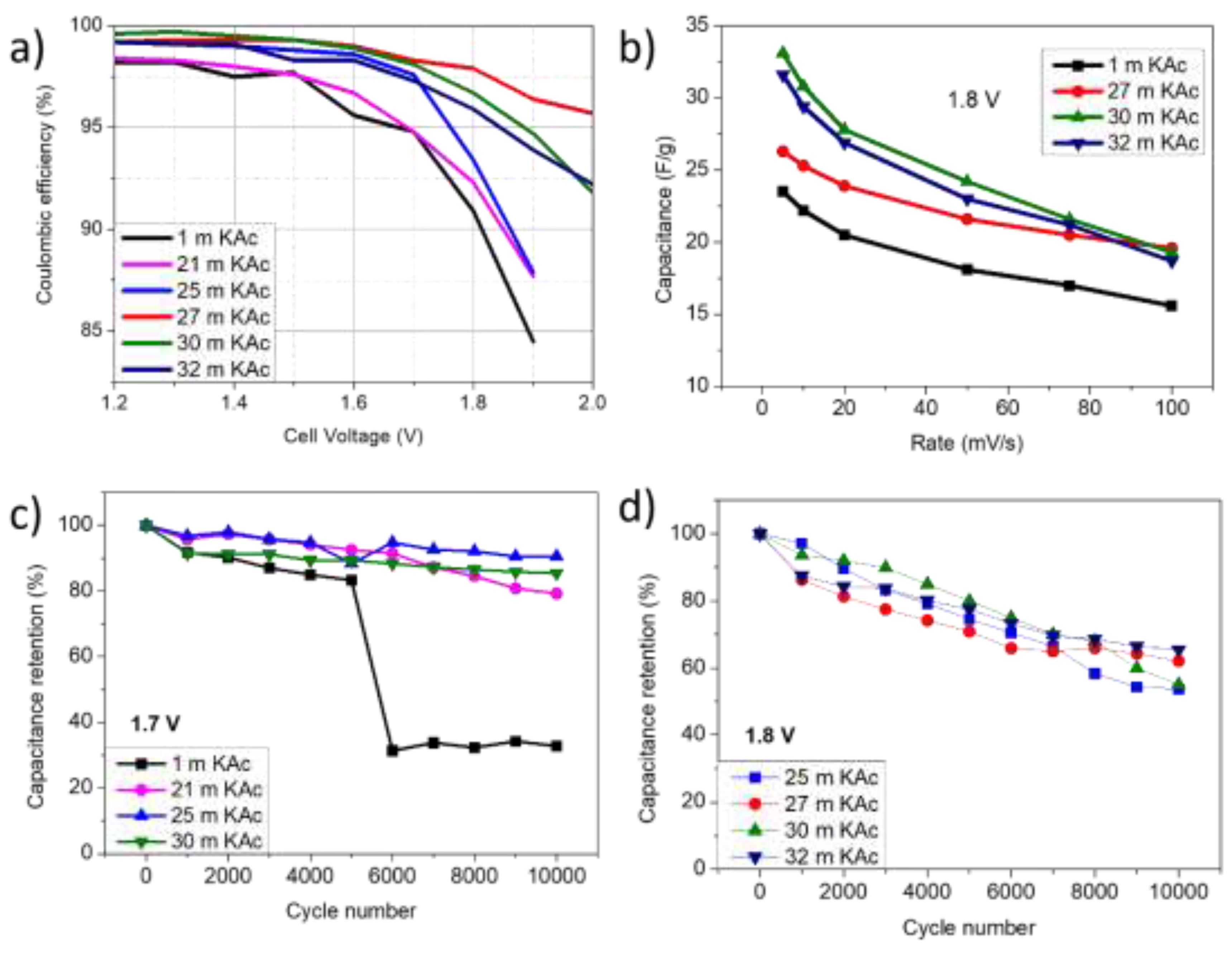
3. Results and Discussion
4. Conclusions
Supplementary Materials
References
- Zang, X., Shen, C., Sanghadasa, M., and Lin, L. (2019) High-Voltage Supercapacitors Based on Aqueous Electrolytes. ChemElectroChem, 6 (4), 976–988. [CrossRef]
- Acharjee, A., and Saha, B. (2024) Organic electrolytes in electrochemical supercapacitors: Applications and developments. J. Mol. Liq., 400 (January), 124487. [CrossRef]
- Wang, Y., Xue, K., Yan, C., Li, Y., Zhang, X., Su, K., Ma, P., Wan, S., and Lang, J. (2024) Tuning of Ionic Liquid–Solvent Electrolytes for High-Voltage Electrochemical Double Layer Capacitors: A Review. Batteries, 10 (2). [CrossRef]
- Suo, L., Borodin, O., Gao, T., Olguin, M., Ho, J., Fan, X., Luo, C., Wang, C., and Xu, K. (2015) “ Water-in-salt “ electrolyte enables high-voltage aqueous lithium-ion chemistries. 350 (6263). [CrossRef]
- Lannelongue, P., Bouchal, R., Mourad, E., Bodin, C., Olarte, M., le Vot, S., Favier, F., and Fontaine, O. (2018) “Water-in-Salt” for Supercapacitors: A Compromise between Voltage, Power Density, Energy Density and Stability. J. Electrochem. Soc., 165 (3), A657–A663. [CrossRef]
- Mishra, R.N., Madikere Raghunatha Reddy, A.K., Goulet, M.A., and Zaghib, K. (2025) Water-in-Salt Electrolytes: Advances and Chemistry for Sustainable Aqueous Monovalent-Metal-Ion Batteries. Batteries, 11 (4). [CrossRef]
- Liang, T., Hou, R., Dou, Q., Zhang, H., and Yan, X. (2021) The Applications of Water-in-Salt Electrolytes in Electrochemical Energy Storage Devices. Adv. Funct. Mater., 31 (3), 1–23. [CrossRef]
- Avireddy, H., Byles, B.W., Pinto, D., Miguel, J., Galindo, D., Jacas, J., Wang, X., Flox, C., Crosnier, O., Brousse, T., Pomerantseva, E., Ramon, J., and Gogotsi, Y. (2019) Nano Energy Stable high-voltage aqueous pseudocapacitive energy storage device with slow self-discharge. Nano Energy, 64 (August), 103961. [CrossRef]
- Deng, W., Wang, X., Liu, C., Li, C., Chen, J., Zhu, N., Li, R., and Xue, M. (2019) Li / K mixed superconcentrated aqueous electrolyte enables high-performance hybrid aqueous supercapacitors. Energy Storage Mater., 20 (July 2018), 373–379. [CrossRef]
- Leonard, D.P., Wei, Z., Chen, G., Du, F., and Ji, X. (2018) Water-in-Salt Electrolyte for Potassium-Ion Batteries. ACS Energy Lett., 3 (2), 373–374.
- Bu, X., Su, L., Dou, Q., Lei, S., and Yan, X. (2019) A low-cost “water-in-salt” electrolyte for a 2.3 V high-rate carbon-based supercapacitor. J. Mater. Chem. A Mater., 7 (13), 7541–7547. [CrossRef]
- Vedhanarayanan, B., and Seetha Lakshmi, K.C. (2024) Beyond lithium-ion: emerging frontiers in next-generation battery technologies. Frontiers in Batteries and Electrochemistry, 3 (April), 1–9. [CrossRef]
- Pameté, E., Köps, L., Kreth, F.A., Pohlmann, S., Varzi, A., Brousse, T., Balducci, A., and Presser, V. (2023) The Many Deaths of Supercapacitors: Degradation, Aging, and Performance Fading. Adv. Energy Mater., 13 (29). [CrossRef]
- Menzel, J., Slesinski, A., Galek, P., Bujewska, P., Kachmar, A., Frąckowiak, E., Washio, A., Yamamoto, H., Ishikawa, M., and Fic, K. (2022) Operando monitoring of activated carbon electrodes operating with aqueous electrolytes. Energy Storage Mater., 49, 518–528. [CrossRef]
- Gomez Vazquez, D., Ingenmey, J., Trapp, K., Ciliak, D., Salanne, M., and Lukatskaya, M.R. (2025) Extended Stability Window in Water-in-Salt Electrolytes: Understanding the Origins. J. Am. Chem. Soc., 147 (39), 35953–35961. [CrossRef]
- Phadke, S., Amara, S., and Anouti, M. (2017) Gas Evolution in Activated-Carbon-Based Supercapacitors with Protic Deep Eutectic Solvent as Electrolyte. ChemPhysChem, 18 (17), 2364–2373. [CrossRef]
- Omori, T., Nakanishi, M., and Tashima, D. (2021) High-temperature degradation tests on electric double-layer capacitors: The effect of residual voltage on degradation. Materials, 14 (6). [CrossRef]
- Mazloomian, K., Dore, T.R., Buckwell, M., Bird, L., Shearing, P.R., and Miller, T.S. (2025) Supercapacitor safety: Temperature driven instability and failure of electrochemical double layer capacitors. Energy Storage Mater., 76. [CrossRef]
- Yuan, C., Wang, L., Yin, S., and Xu, J. (2020) Generalized separator failure criteria for internal short circuit of lithium-ion battery. J. Power Sources, 467. [CrossRef]
- Borgovini, R., Pemberton, S., and Rossi, M. (1993) Failure Mode, Effects and Criticality Analysis (FMECA).
- Dai, J., and Pang, J. A risk analysis method for potential failure modes in the lithium-ion battery assembly process based on optimized FMEA and DHHFLTS. [CrossRef]
- Hendricks, C., Williard, N., Mathew, S., and Pecht, M. (2015) A failure modes, mechanisms, and effects analysis (FMMEA) of lithium-ion batteries. J. Power Sources, 297, 113–120. [CrossRef]
- Maddipatla, S., Kong, L., and Pecht, M. (2024) Safety Analysis of Lithium-Ion Cylindrical Batteries Using Design and Process Failure Mode and Effect Analysis. Batteries, 10 (3). [CrossRef]
- Held, M., and Brönnimann, R. (2016) Safe cell, safe battery? Battery fire investigation using FMEA, FTA and practical experiments. Microelectronics Reliability, 64, 705–710. [CrossRef]
- Abu-Lebdeh, Y., and Davidson, I. (2009) High-Voltage Electrolytes Based on Adiponitrile for Li-Ion Batteries. J. Electrochem. Soc., 156 (1), A60.
- Xu, W., Cooper, E.I., and Angell, C.A. (2003) Ionic liquids: Ion mobilities, glass temperatures, and fragilities. Journal of Physical Chemistry B, 107 (25), 6170–6178. [CrossRef]
- Gomez Vazquez, D., Ingenmey, J., Trapp, K., Ciliak, D., Salanne, M., and Lukatskaya, M.R. (2025) Extended Stability Window in Water-in-Salt Electrolytes: Understanding the Origins. J. Am. Chem. Soc., 147 (39), 35953–35961. [CrossRef]
- Tian, Z., Deng, W., Wang, X., Liu, C., Li, C., Chen, J., Xue, M., Li, R., and Pan, F. (2017) Superconcentrated aqueous electrolyte to enhance energy density for advanced supercapacitors. Functional Materials Letters, 10 (6), 1–5. [CrossRef]
- Park, J., Kim, J., Lee, S., Kim, J.H., Yoon, M.H., Lee, D., and Yoo, S.J. (2024) Unraveling concentration-dependent solvation structures and molecular interactions in water-in-salt electrolytes for enhanced performance of electric double-layer capacitors. Energy Storage Mater., 65. [CrossRef]
- Qiu, C., Jiang, L., Gao, Y., and Sheng, L. (2023) Effects of oxygen-containing functional groups on carbon materials in supercapacitors: A review. Mater. Des., 230. [CrossRef]
- Yang, C.H., Nguyen, Q.D., Chen, T.H., Helal, A.S., Li, J., and Chang, J.K. (2018) Functional Group-Dependent Supercapacitive and Aging Properties of Activated Carbon Electrodes in Organic Electrolyte. ACS Sustain. Chem. Eng., 6 (1), 1208–1214.
- Asrafali, S.P., Periyasamy, T., Kim, S.C., and Lee, J.W. (2024) Enhanced Wettability and Adhesive Property of PTFE through Surface Modification with Fluorinated Compounds. Materials, 17 (13). [CrossRef]
- Pletincx, S., Trotochaud, L., Fockaert, L.L., Mol, J.M.C., Head, A.R., Karslloǧlu, O., Bluhm, H., Terryn, H., and Hauffman, T. (2017) In situ characterization of the initial effect of water on molecular interactions at the interface of organic/inorganic hybrid systems. Sci. Rep., 7.
- Wijaya, O., Hartmann, P., Younesi, R., Markovits, I.I.E., Rinaldi, A., Janek, J., and Yazami, R. (2015) A gamma fluorinated ether as an additive for enhanced oxygen activity in Li-O2 batteries. J. Mater. Chem. A Mater., 3 (37), 19061–19067. [CrossRef]
- Azizpour, A., Bagovic, N., Ploumis, N., Mylonas, K., Hoxha, D., Kienberger, F., Al-Zubaidi-R-Smith, N., and Gramse, G. (2025) Electrochemical Analysis of Carbon-Based Supercapacitors Using Finite Element Modeling and Impedance Spectroscopy. Energies (Basel)., 18 (6). [CrossRef]
- Hakamy, A. (2025) Investigation of double-layer capacitance, Warburg finite-length impedance and AC conductivity of PVA/MWCNT nanocomposite films for supercapacitor applications. J. Power Sources, 656. [CrossRef]
- Chen, X., Wu, Y., and Holze, R. (2023) Ag(e)ing and Degradation of Supercapacitors: Causes, Mechanisms, Models and Countermeasures. Molecules, 28 (13). [CrossRef]
- Hahn, M., Würsig, A., Gallay, R., Novák, P., and Kötz, R. (2005) Gas evolution in activated carbon/propylene carbonate based double-layer capacitors. Electrochem. commun., 7 (9), 925–930. [CrossRef]
- Hema, R.K., and Varzi, A. (2025) Aqueous Solid Electrolyte Interphases in Water-in-Salt Electrolytes and Beyond. ChemElectroChem, 12 (15). [CrossRef]
- Gabryelczyk, A., Ivanov, S., Bund, A., and Lota, G. (2021) Corrosion of aluminium current collector in lithium-ion batteries: A review. J. Energy Storage, 43. [CrossRef]
- Chomkhuntod, P., Iamprasertkun, P., Chiochan, P., Suktha, P., and Sawangphruk, M. (2021) Scalable 18,650 aqueous-based supercapacitors using hydrophobicity concept of anti-corrosion graphite passivation layer. Sci. Rep., 11 (1). [CrossRef]
- Li, H., Li, J., Zhao, C., and Zhao, F. (2025) Research on the Preparation of Supercapacitor Separators with High Wettability and Excellent Temperature Adaptability Through In Situ Deposition of Nano-Barium Sulfate on Regenerated Cellulose. Polymers (Basel)., 17 (7). [CrossRef]
| Supercapacitor Component | Failure mode(s) | Failure mechanism | Failure cause | Failure local effect | Failure system effect | Effects analysis | Failure level |
|
Carbon electrode YP-50F: Super P: PTFE in wt.% 85:10:5 |
Electrode degradation | Mechanical stress | Long cycling, high power | PTFE degradation, particle fracture, contact loss | CTR increase, capacitance reduction, shorter cycle life | SEM did not show particle or electrode degradation | Low |
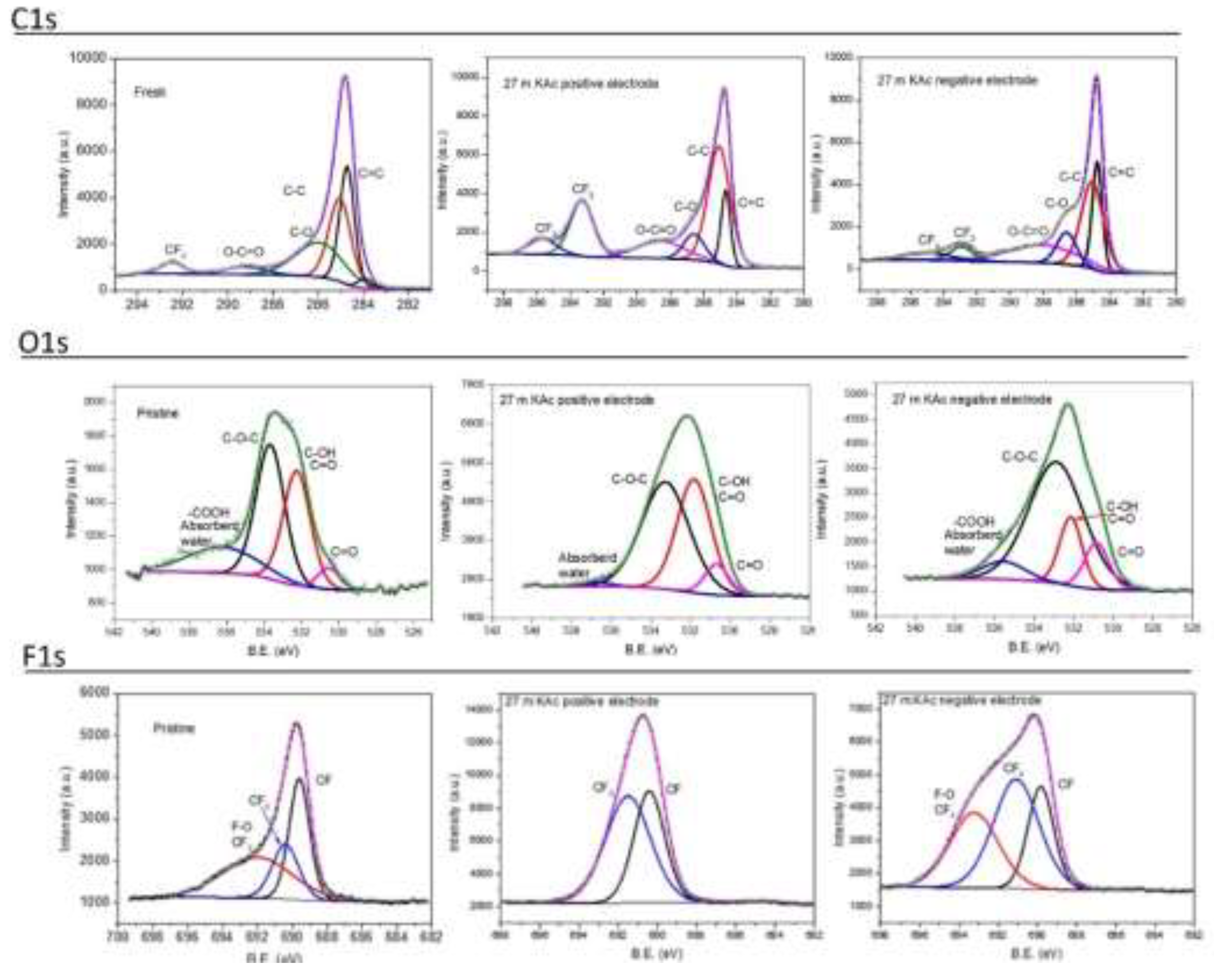
| Pore blocking | Chemical reaction and deposition | Operational voltage exceeds the stability window of the electrolyte | Gas bubbles (OER, HER) or solid decomposition products | Resistance increase, specific capacitance reduction, shorter cycle life | Increase of oxygen functional groups by XPS, increase in Warburg coefficient, and cell reassembly | High | |
|
Water-in-salt electrolyte 27 m KAc |
Electrolyte decomposition | Chemical reaction and deposition | Operational voltage exceeds the stability window of the electrolyte | OER and HER, salt precipitation | ESR increase, specific capacitance reduction, shorter cycle life | Parasitic current(s) in electrolyte stability tests, non-ideal GCD and CV profiles | High |
|
Current collectors SS304 vs GC |
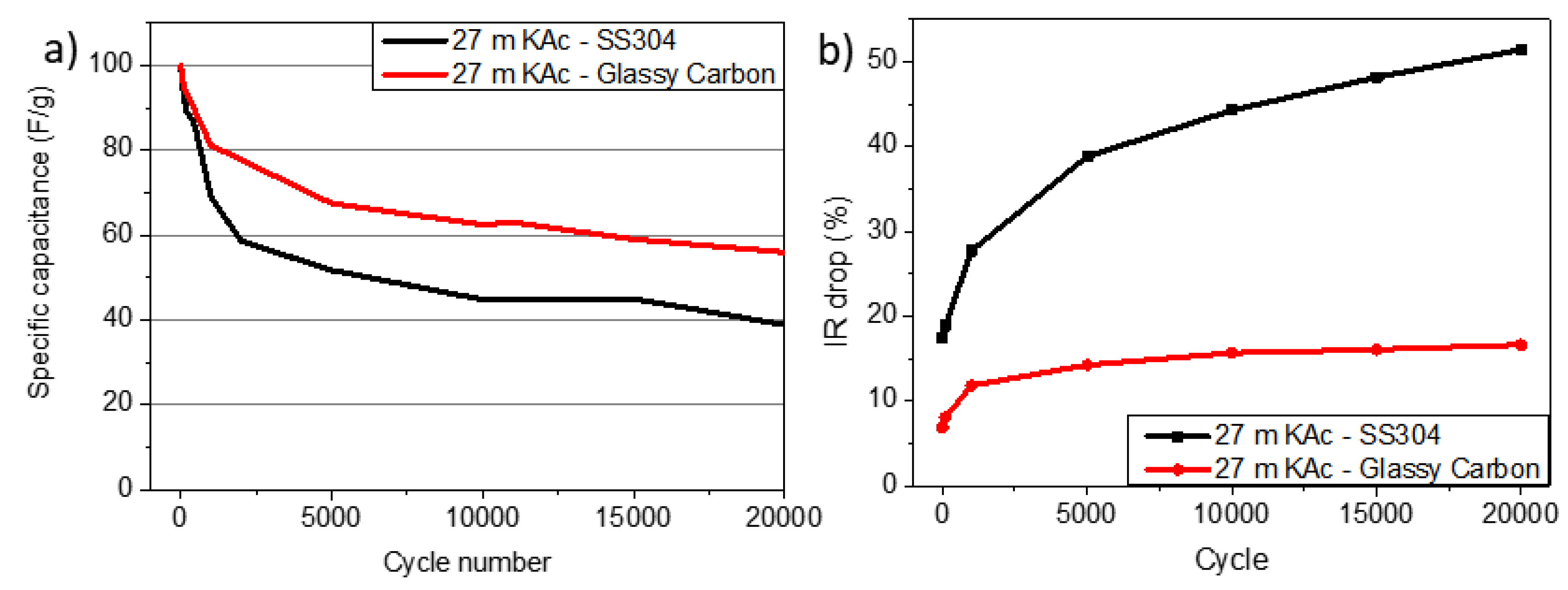
Corrosion | Chemical reaction and deposition | Non-compatibility of materials | OER and HER, current collector passivation | CTR and IR drop increase, specific capacitance reduction, shorter cycle life | Non-ideal GCD and CV profiles, replace SS by GC | Medium |
|
Separator Whatman glass microfiber GF/C |
Material shrinkage or fracture | Mechanical degradation | Temperature, long cycling, high power | Pin holes and cracks | Short circuit | Failure response not observed at room temperature | Low |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




