Stoichiometric and compact CdSe films of a zinc blende structure (JCPDS #19–0191), with a thickness of 2–3 μm, a strong (111) crystallographic texture and low roughness were obtained on Ti substrate by typical electrodeposition from an acidic selenite bath [
6]. Similar CdSe barrier films of 1–2 μm thickness with weaker texture and nodular morphology were deposited on the porous titania substrates [
4]. Depending on the plating specifications and post-treatment, the characteristic (Debye–Scherrer) size of the crystallite structure in the films varied from 40 to 130 nm. The CdSe/Ti and CdSe/(TiO
2)Ti specimens were used as electrodes in PEC, either as-prepared without any post treatment, or after thermal annealing at various temperatures. Annealing resulted in more uniform, larger grain layers, refined from any traces of elemental Se. At calcinating temperatures above 400 °C, a wurtzite CdSe structure (JCPDS #08–0459) was established from the kinetically controlled, as-deposited zinc blende phase of CdSe.
3.1. Interface energetic
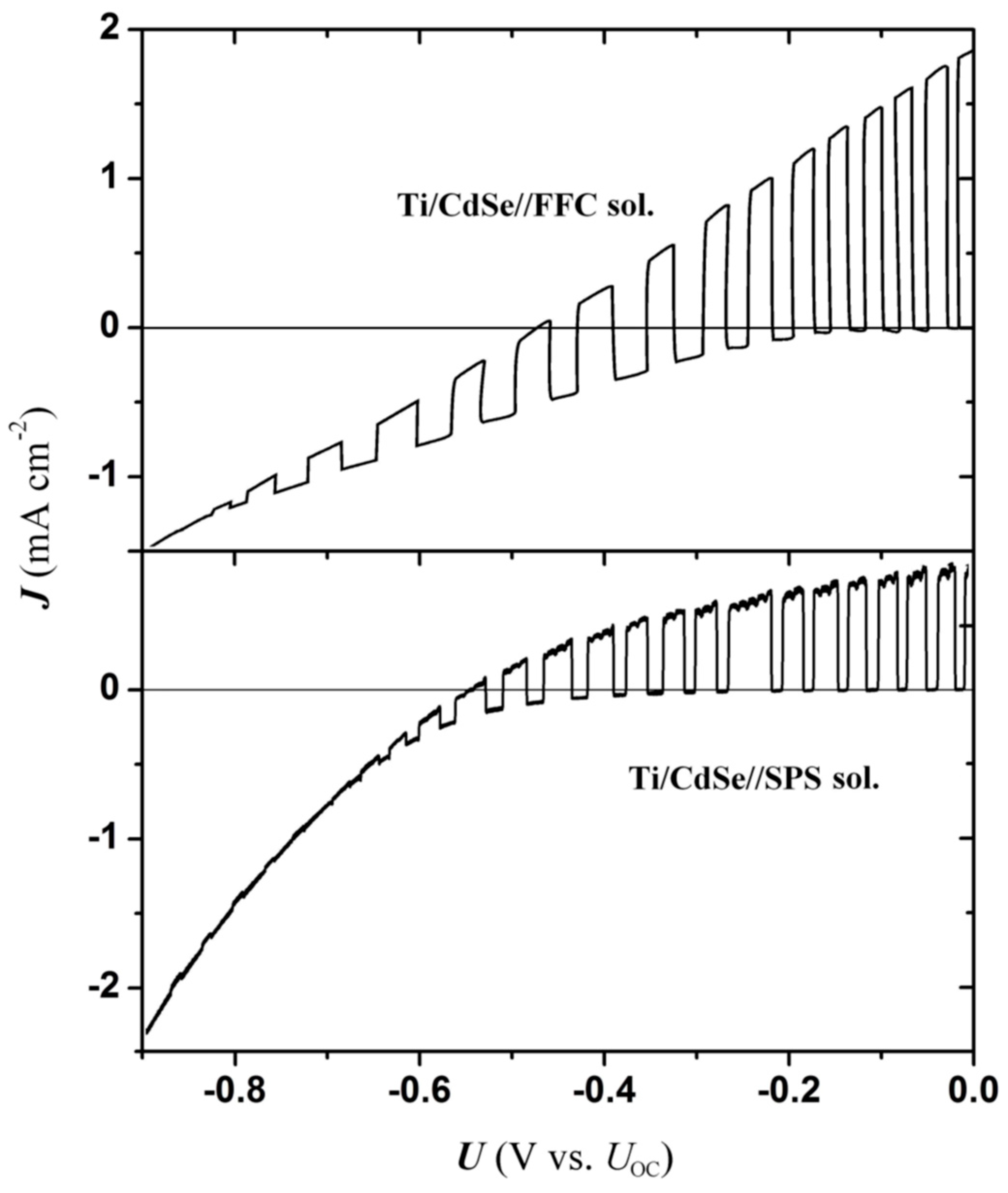
In order to extract information from EI measurements and evaluate their significance, some prior knowledge of interface energetics is needed. Characteristic energies of the electrodes, i.e., the band edge positions of CdSe films as-prepared by electrodeposition (polycrystalline; pc-films), were estimated through the flat-band potential (
UFB) values as-determined from the onset potential (
Uonset) in
photocurrent voltammetry measurements in common electrolytes (e.g.,
Figure 1). The open circuit potential
UOC in the dark was taken as the zero point for each measurement cell and considered equal to the redox potential for the employed electrolyte (
UR/O). The
UFB position was taken 0.10 V more negative than the
Uonset value, on account of the electron back-diffusion current [
7]. The conduction band edge (
UCB) of the SC was taken as lying at −0.12 V relative to
UFB [
8], and the valence band edge (
UVB) was located through the known value of the energetic band gap of CdSe (1.70 eV). The assessed values of
UFB,
UCB, and
UVB are given in
Table 1, along with selected data from the literature regarding
n-CdSe single crystal (sc) electrodes in the respective electrolytes.
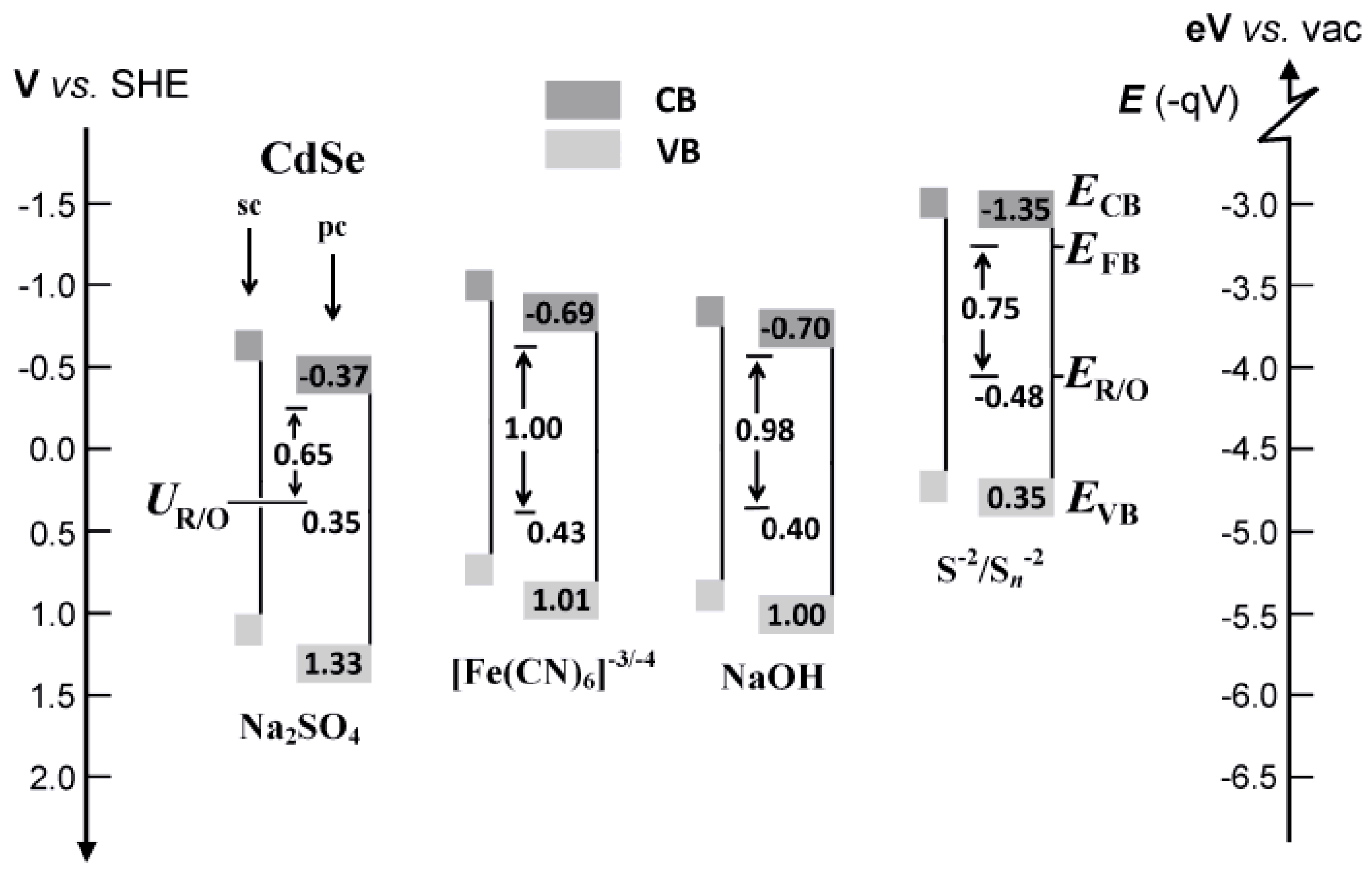
The overall resulting interface energetics is depicted in the scheme of
Figure 2. It is seen that our estimated band edges for the polycrystalline electrodes are systematically shifted to slightly more positive potentials compared to those reported for single crystal electrodes; otherwise the agreement is rather good. From these data it appears that use of the depletion layer model (Mott–Schottky) is justified at the first place, since the U
R/O values lie in the band gap range of the semiconductor electrode.
3.2. Different Electrolytes
Mott–Schottky plots in the dark and under illumination for CdSe/(TiO
2)Ti electrodes and different liquid contacts are shown in
Figure 3. The graphs were very similar for the CdSe/Ti electrodes. In these diagrams, the potential is scanned to the negative direction starting from the open circuit potential
UOC in the dark taken as the zero point (actually
UOC =
UR/O vs Ag/AgCl). Since the SC electrode is
n-type, the scan direction corresponds to a Fermi level displacement from the dark equilibrium position, towards the flat band potential
UFB of the semiconductor.
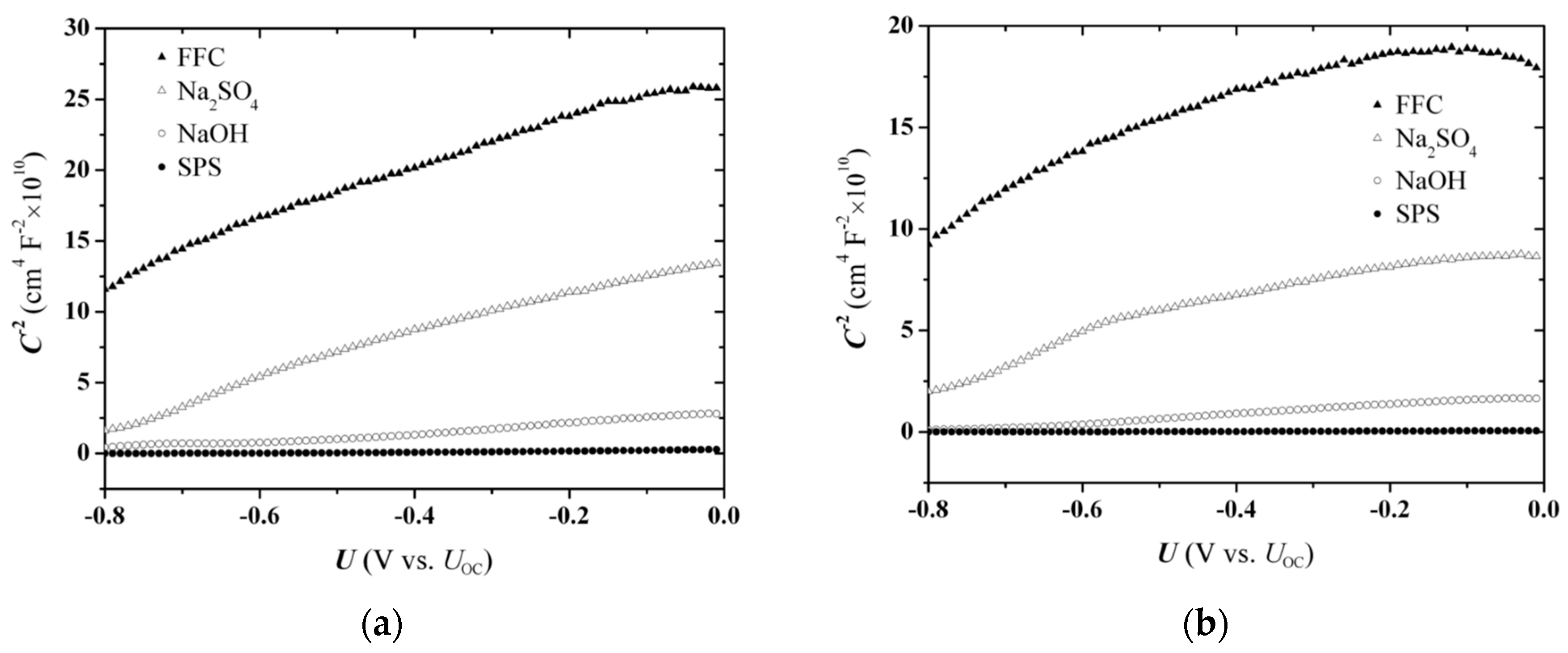
It is seen in
Figure 3 that the capacitance
C increases in the order: FFC < Na
2SO
4 < NaOH < SPS, both for the dark and illuminated cells. In principle, the smaller
C (higher
C−2) measured in the FFC system compared to the SPS should be expected, on account of the more positive redox potential of this electrolyte (
Table 1), causing larger band bending in the SC and thereby wider space charge region. Assuming that the recorded capacitance equals
CSC, the
U-intercepts of the plots in
Figure 3 should correspond to the flat band potentials
UFB vs.
UOC in the dark, i.e., to the maximum band bending (
UFB −
UR/O) for each CdSe//electrolyte junction as given in the scheme of
Figure 2. Drawing of intercept lines at the
proper capacitance scale shows that the
UFB vs.
UOC values for the redox solutions, found at
ca. −0.75 V (
cf. Figure 4a for CdSe/TiO
2 in SPS solution) and −1.00 V for FFC, approach those predicted in
Figure 2. However, the
U-intercepts for the FFC and Na
2SO
4 solutions are found at quite more negative values than expected from
Figure 2; i.e., at −1.60 rather than −1.00 V for FFC and −0.80 rather than −0.65 V for Na
2SO
4. It seems that abnormally high
C−2 values are involved in this case, which could be due to surface modification of the electrode during the measurement process.
Indeed, the CdSe surface in contact to FFC has been known [
14] to undergo abundant formation of stable corrosion products, such as ferro-ferricyanide compounds and amorphous Se from the hole-mediated decomposition of CdSe, which add a substantial dielectric capacitance component
in series to the SC space charge. It must be emphasized, however, that the absence of similar effects for SPS is only due to the fact that corrosion products build up on the electrode surface much more slowly than in the FFC case, while also any produced Se dissolves easily in the polysulfide electrolyte (
Section 3.5). To be sure, the photovoltammetric (onset potential) measurements were considered more reliable, because their time scale is much smaller compared to the EI experiment, so the results in
Table 1 correspond to the initially measured values, before time-dependent phenomena step in (also [
2]).
Illumination of the electrodes during measurement results in an overall increase in
C (decrease in
C−2) at a given applied potential (
Figure 3b). Considering that band-edge unpinning is very probable under conditions of intense anodic (photo)corrosion, this change of junction capacitance can be attributed to the displacement of band edges to the positive direction and consequent decrease of the band bending and space charge layer width, due to trapping of photoinduced holes at surface states. At any rate, the presence of a redox couple in the electrolyte should conduce to the stability of the system as enhancing the hole-scavenging ability for the
n-type SC. “Stability” tests involving successive voltage-stepping scans were used to check each CdSe//electrolyte system. In particular, three EI recording cycles were performed involving a first scan in the dark (“dark 1”) followed by a second scan under illumination and a third again in the dark (“dark 2”). The relative increment Δ
C/
C between “dark 1” and “dark 2” was estimated at several selected potentials and taken as a measure of the photocorrosion effect. From “dark 1” to “dark 2”, the resulting plots always shifted to lower
C−2 values, indicating that all systems are prone to photocorrosion. The SPS system was always more stable as giving a Δ
C/
C ratio around 1 compared to 1.5 for the FFC and the background electrolytes.
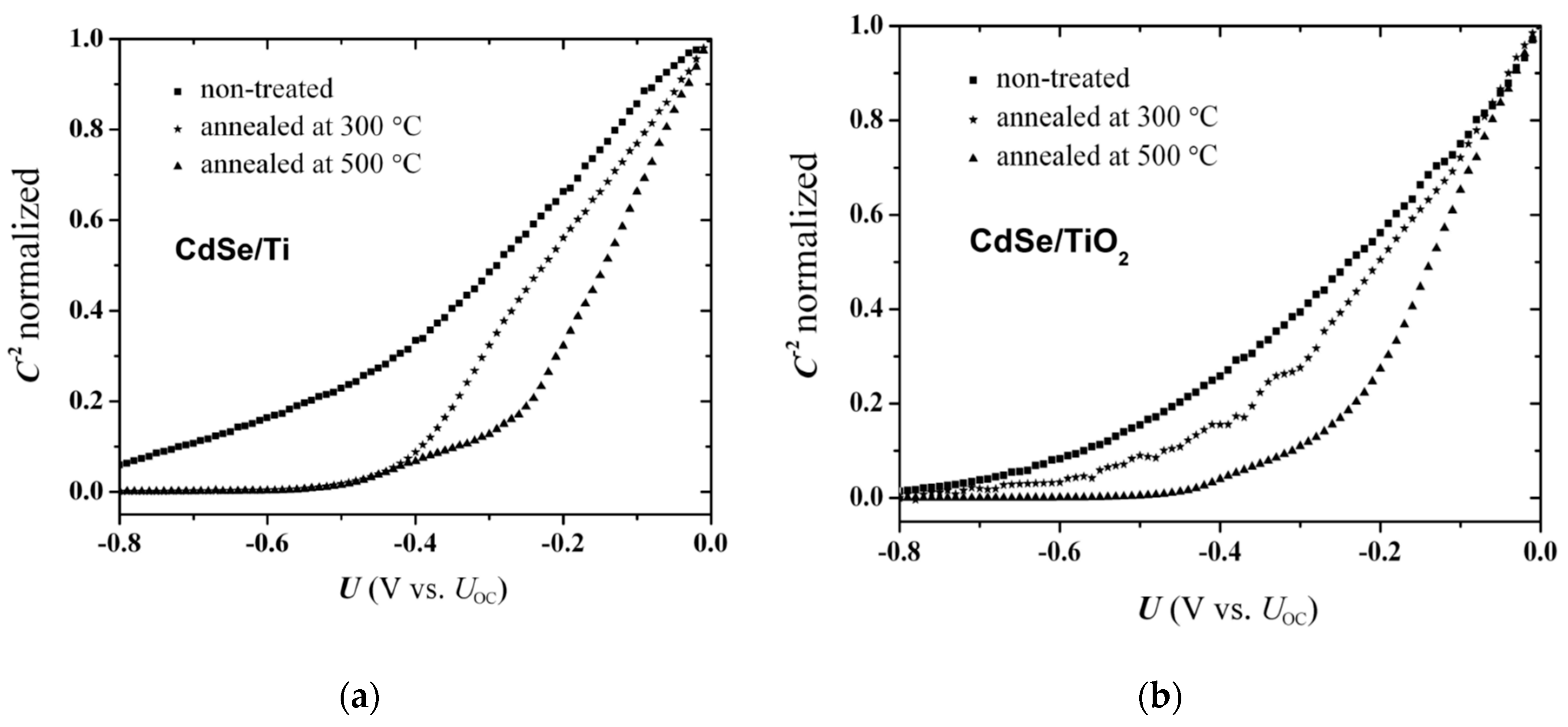
3.3. Effect of Annealing on the C−2 vs. U Plots
From the preceding remarks, it is clear that the foremost prerequisite to obtain reliable EI data is the selection of a redox electrolyte appropriate for the specific electrode type (i.e., CdSe in the present case). The
C−2 vs.
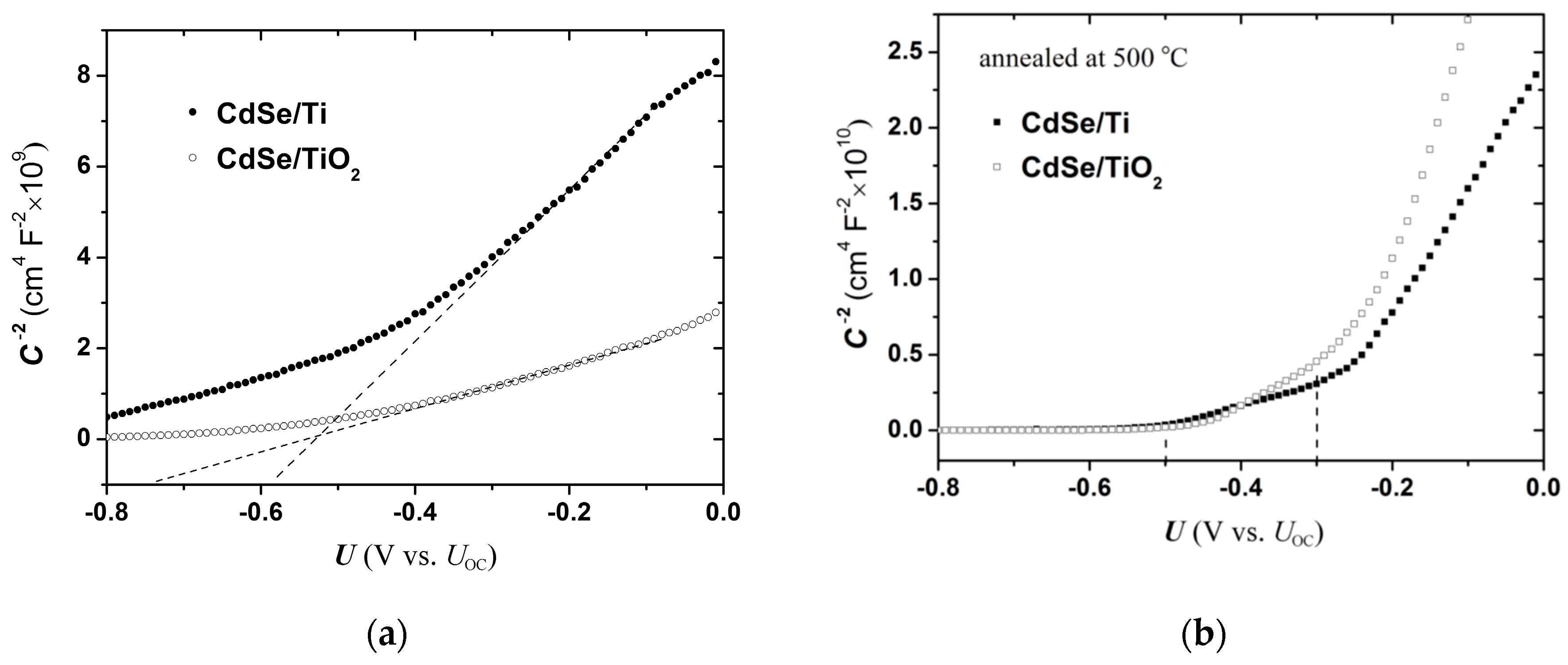
U plots for the SPS cell, which is the most photocorrosion-resistant system of those presently investigated, are depicted in their appropriate capacitance scale in
Figure 4, for CdSe/Ti and CdSe/TiO
2 electrodes, as-prepared (
Figure 4a) and annealed at 500 °C (
Figure 4b). Similar graphs are given in the diagrams of
Figure 5 with ordinate scale normalized to unity, for as-prepared and annealed (at 300 and 500 °C) electrodes. The shapes of the plots in these diagrams are not unusual in the literature, and encompass curved rather than straight lines for the non-treated electrodes, as well as annealing-induced features on the slope and the
U-axis intercept. Recall that the annealing treatment affects only the CdSe layer, while at temperatures above 400 °C, a wurtzite CdSe structure is obtained. Thermal oxidation of the Ti substrate to rutile TiO
2 during treatment of CdSe/Ti must be ruled out in view of the oxygen-free annealing atmosphere.
Specifying a single slope for the curved lines in
Figure 4a to estimate the active donor concentration
ND in the non-treated electrodes from Eq. 1, is rather tricky. On the basis of the ideal model, and disregarding any changes of the electrode during the measurement, the shape of the plots may imply that the active donor concentration
ND is not uniform across the CdSe layer and decreases with the distance from the SC/EL interface towards the SC bulk; the lower
ND values correspond to the higher slope (∝ 1/
ND) obtained at the more positive potentials, whereat the depletion region extends deeper in the SC electrode. After annealing the electrodes, the Mott–Schottky lines become steeper and present an upright segment at potentials positive to about −0.25 V (
Figure 4b). In addition, annealed electrodes present an appreciable shift of the projected
U-intercept to less negative values compared to the non-treated electrode (
Figure 5), the magnitude depending on the annealing temperature.
The steep change of Mott–Schottky slope as recorded in aqueous electrolyte for electroplated polycrystalline CdSe electrodes has been addressed in the literature (e.g., [
15]); however, no explanation is known to us. Within the depletion-layer approximation –
cf. assumption (vi) of the ideal model – the observed segmented line shape could be interpreted by assuming either (1) the existence of two donor types, D1 and D2, in the
same semiconductor phase, associated to two discrete energy levels within the bandgap, a shallow and a deep; or (2) response of both the
n-type CdSe layer and TiO
2 substrate, in sequence. In case (1), as the potential increases and the Fermi level moves down the energy gap, a Mott-Schottky slope of
would be obtained upon reaching the shallow level, and a slope of
upon reaching the deep level; this sequence would cause however a decrease in the slope, contrary to the observed. In case (2), the slope would change from
to
, and since the TiO
2 substrate has a much lower donor concentration than CdSe, this would be in accord with the observed direction in slope change. However, comparison between the CdSe/(TiO
2)Ti and CdSe/Ti electrodes (
Figure 4 and
Figure 5) suggests that the response regards solely the CdSe layer, since a similar slope change is observed regardless of the substrate.
Since, in general, the non-linearity of Mott–Schottky plots is largely connected to a disordered solid state structure, it can be assumed that in non-treated electrodes, the donor concentration drops gradually from the electrolyte interface towards the SC bulk due to the more defective (donor-rich) character of the upper CdSe layers, whereas in the annealed samples the
ND is high at the surface contacting the electrolyte and abruptly drops in the bulk. In other words, the thermal treatment, by restoring a uniform crystallized layer, eliminates lattice imperfections with a donor character, thus steeping the Mott–Schottky plot. Calculations based on the ideal model as a rough approximation (
Appendix A) may corroborate this claim, as indicating that the high donor concentration region has practically zero thickness; hence it should correspond to the inter-phase region of the CdSe//SPS contact.
The elimination of bulk donor states would be expected to move the Fermi level towards the intrinsic position in the middle of the bandgap, which explains the positive shift of the projected flat band potential (
Figure 5). This model suggests that the high-slope line is representative of the bulk SC properties and allows quantitative predictions to be made from the slope and intercept of this line. On the other hand, the low-slope regions are complicated by the occurrence of surface states, which introduce energetic states close to the conduction band of the SC, presumably with density that follows an exponential decay from the band edge to lower energies within the bandgap (this “band tailing” in the polycrystalline electrode has been evidenced clearly by photocurrent spectroscopy). At the more negative potentials, charging of the surface states will make
CSS the main component of the overall capacitance, and as the potential is scanned more positive and the Fermi level moves deeper in the bandgap, the exponential decrease of surface state density will be followed by an exponential increase of
C−2 with potential. At some point, though,
CSC dominates and a linear Mott–Schottky behavior is established [
1], pretty much as the curve shape indicates. The reconstructive effect of annealing partly restores, thus, a linear behavior of the electrode at the more positive potentials, where the measured capacitance corresponds to
CSC.
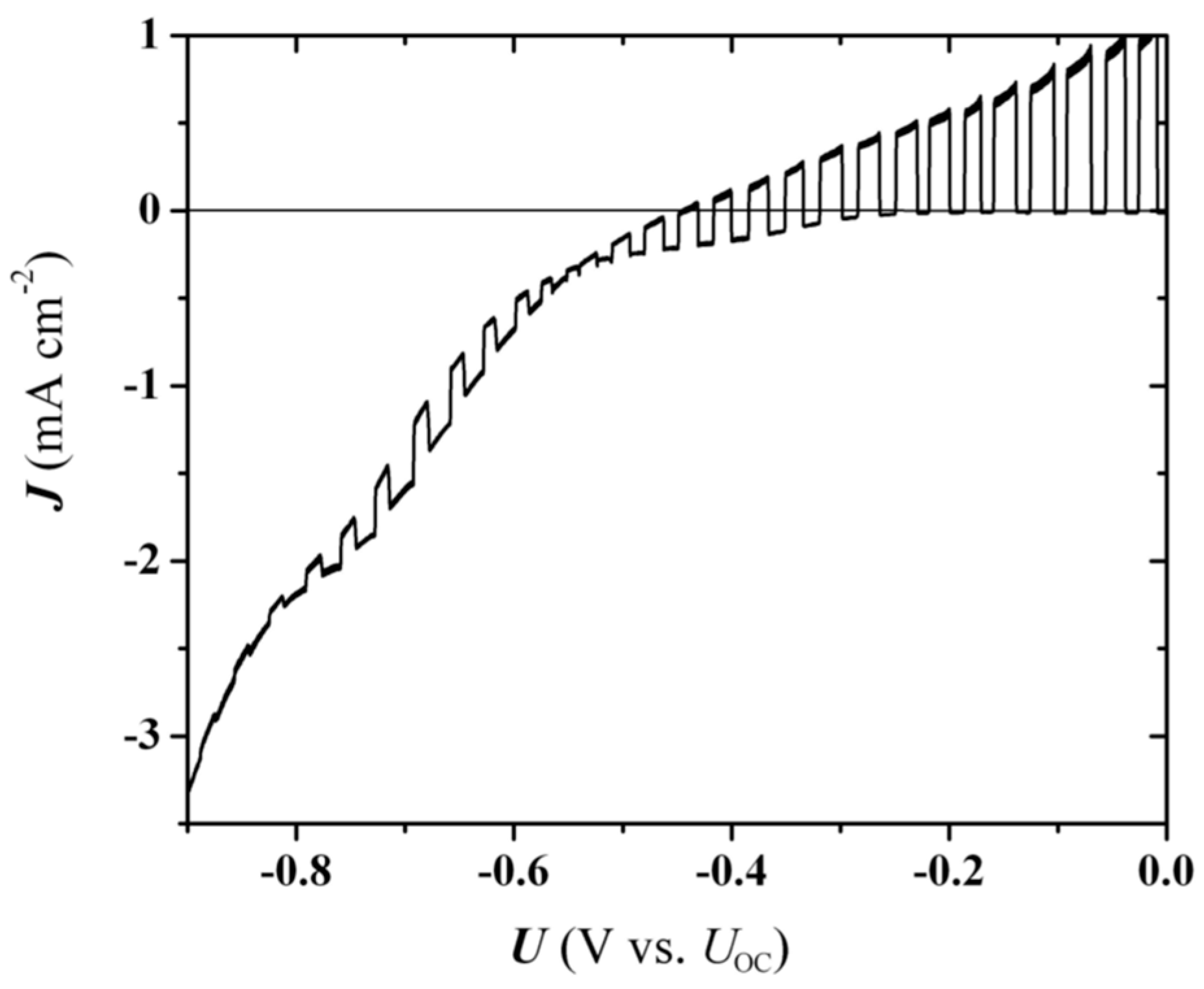
Further insight can be gained here with the assistance of photocurrent voltammetry (PV) data. For non-treated CdSe/Ti samples, the
U-axis intercept of
Figure 4a indicates a
UFB lower than −0.60 V, close to the PV result (
Figure 1), which gives a
Uonset at about the same potential (
UFB ≈
Uonset − 0.1 V). But, the PV curve for the annealed at 500 °C electrode (
Figure 6) presents an appreciable photocurrent inversion from anodic to cathodic at more positive potentials, which is not observed with the non-treated electrode. In principle, the photocurrent switch signifies a transition from depletion to accumulation zone as the potential crosses the Fermi level. It occurs at approximately −0.50 (
Figure 6), i.e., more negative than the claimed intercept of the upright line in
Figure 4b, making the
UFB estimation based on the higher slope of
Figure 4b more ambiguous than considered. But, even if the quantitative agreement is not good, this result corroborates a shift of the Fermi level deeper (more positive) into the CdSe bandgap in the annealed samples. The photocurrent inversion is no longer observed already after two PV cycles so it can be considered to be implicit in the pristine CdSe phase before degradation starts in the PEC. Actually, evolving photocorrosion modifies the electrode interface, building up a dielectric Se layer (
Section 3.5), which, although not restricting the anodic photocurrent as being a good hole-conductor, inhibits the cathodic photocurrent by imposing an energetic barrier to the CdSe conduction band electrons (
ECB of Se lies approximately 1.0 to 1.2 eV above the
ECB of CdSe [
10,
16]).
An alternative way to account for the uncertainty in the position of the Fermi level in terms of the photocurrent switching and the segmented appearance of the Mott–Schottky plot is to invoke the so-called Photoelectrochemical Photocurrent Switching (PEPS) effect, which describes the observed behavior as depending on the relative positions of the Fermi level and the surface state energy, rather than the position of the Fermi level itself with respect to the band edges. Hints on this effect and its significance are illustrated in the
Appendix B.
3.4. Frequency Dependence and Electron Density Calculation
Impedance measurements performed with the CdSe//SPS system show strong variation of the Mott–Schottky slope with frequency, implying that the assumption (v) of the ideal model is not valid due to dielectric relaxation; that is, frequency dependence of the dielectric constant
ε. When the determination of
ND is not possible from a single experiment at one frequency, a slope averaging procedure can be attempted as suggested by Gomes and Cardon [
1], by plotting
C−2 vs. U at different EI frequencies and specifying limiting slope values: the maximum slope is given by 2/[
eεoε(0)
ND] and the minimum slope by 2
ε(0)/[
eε0εi2ND], where
ε(0) and
εi are the relative permittivities for zero and limiting high frequency, respectively. Since the dielectric constant drops asymptotically from
ε(0) to
εi, within the applied range of frequencies, the higher frequencies correspond to higher Mott-Schottky slopes [
1,
17]. The donor density is calculated from the geometrical average of the two limiting terms (square root of their product), which is 2/[
eε0εiND].
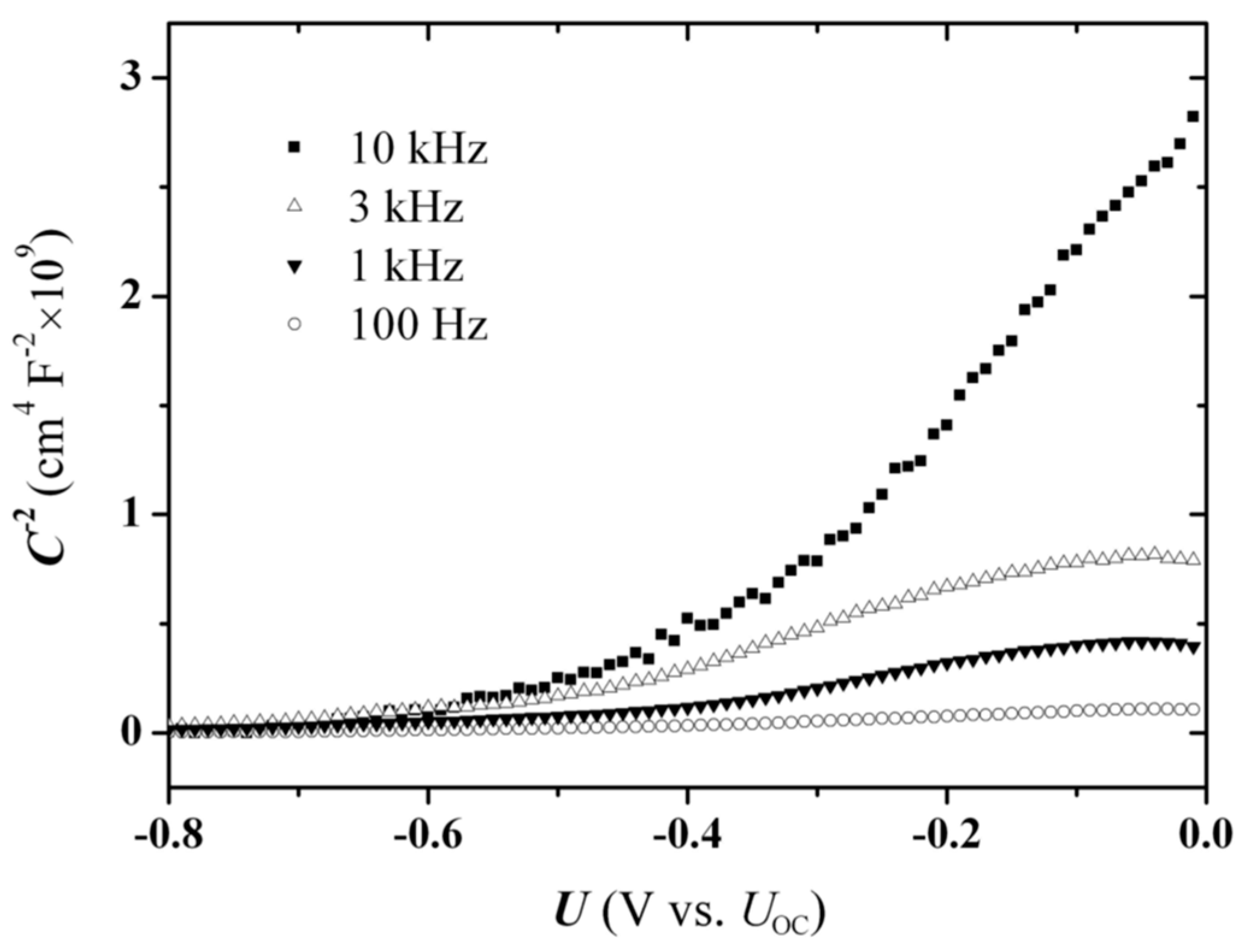
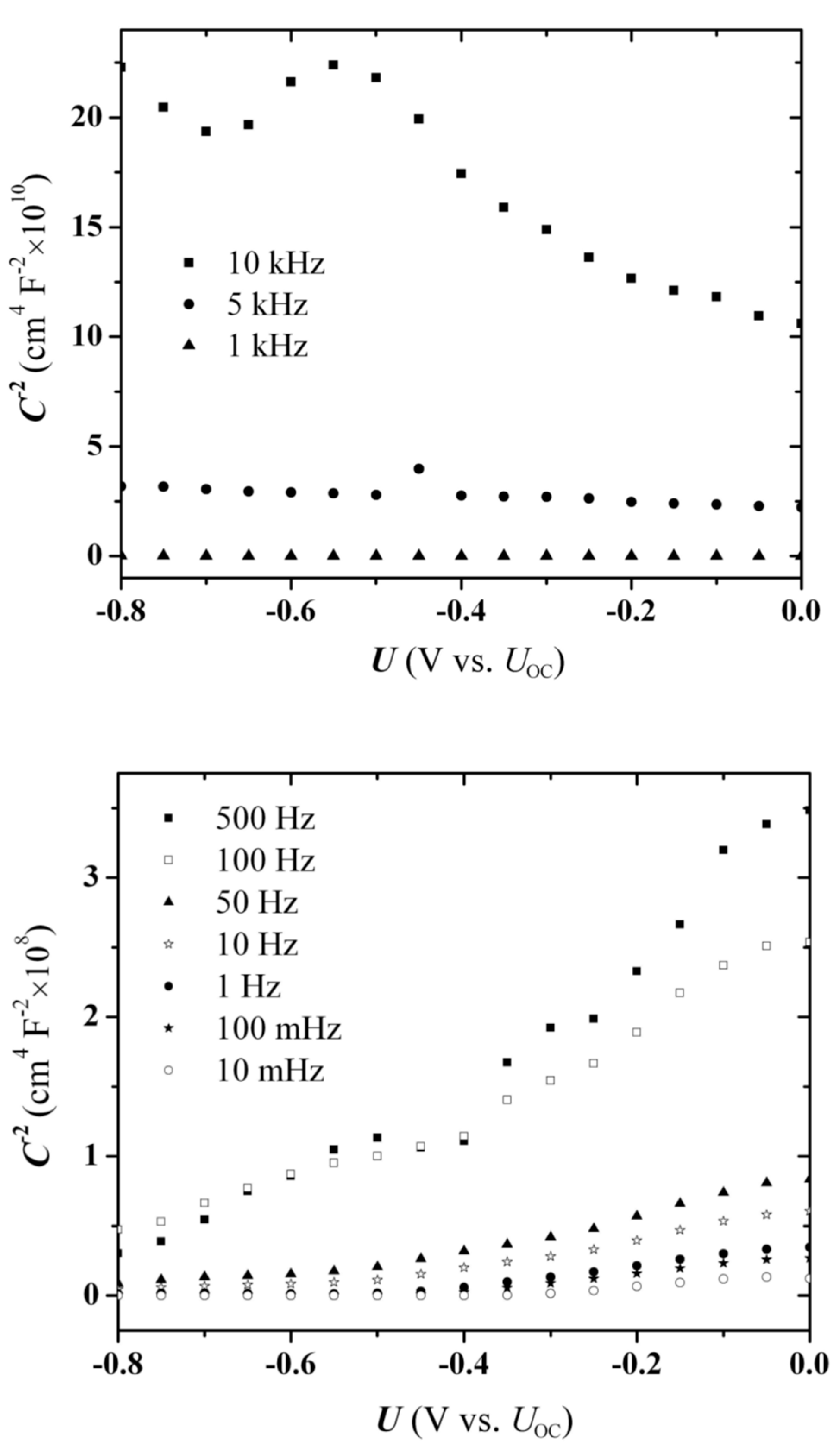
The EI experiments were performed with successive voltage scans of varying constant frequencies in the direction from high to low frequency. In order to examine also the time dependence of the process, two series of measurements were carried out, one with 10 mV-step scans (
Figure 7) and the other with 50 mV-step scans (
Figure 8). The step-duration was maintained at 20 s, therefore the 50 mV sequence, allowing a faster scanning of the fixed potential range, involved a smaller number of recording points (
Appendix C). At any rate, according to the Gomes–Cardon procedure, and using a high frequency relative permittivity value of
εi for CdSe equal to 6.20, the donor density in the semiconductor was estimated from the 10 mV
-stepping experiments as 5.39 × 10
18 cm
−3, and from the 50 mV
-stepping experiments, as 4.36 × 10
18 cm
−3. These values are reasonably close to each other and indicate an approximate electron density of 5 × 10
18 cm
−3.
The first three cycles of the fast-scan experiments were excluded from the electron-density calculation procedure, due to the presence of a moving summit, i.e., a local capacitance drop, which appears clearly at −0.60 V during the 10 kHz cycle (
Figure 8a), and moves towards −0.40 V during the subsequent two cycles (the scaling is not clear in the diagram) before disappearing (
Figure 8b). This peak was absent in the “slow” experiments with the SPS system, i.e., when the voltage scans were carried out in 10 mV steps (
Figure 7), and needs a special consideration.
3.5. Surface Corrosion Effects: Formation of Dielectric Layer on the Electrode
The former observation signifies an ongoing chemical modification of the CdSe electrode during measurement that becomes visible on the Mott–Schottky plots, insofar as the SPS solution is concerned, only when increasing the scan rate of the potential (by an effect analogous to redox peak amplification with higher scan rate in cyclic voltammetry). In the less “stabilizing” environment of other electrolytes, this feature is prominent even with the “slow” 10 mV
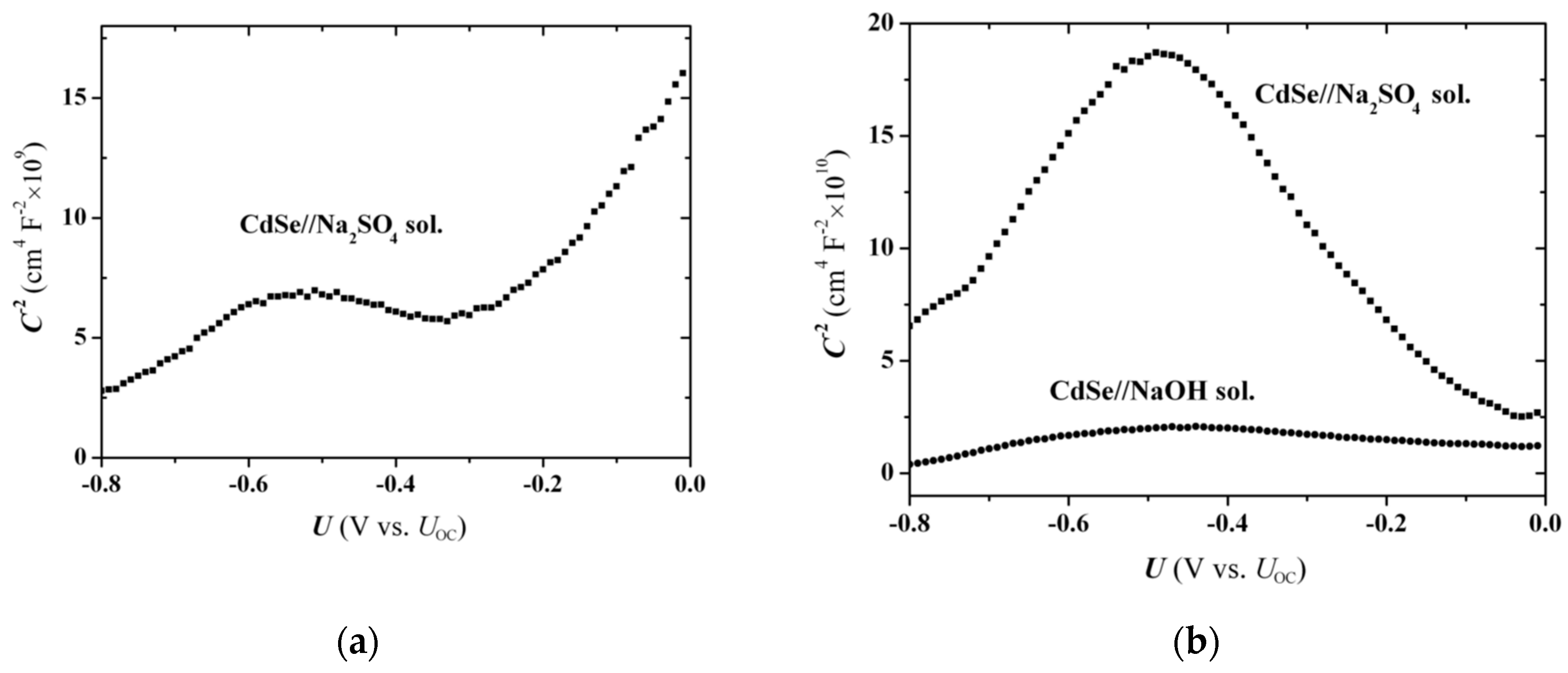
-stepping scans. Thus, the slight bending observed at voltages around −0.55 V in
Figure 3a for the Na
2SO
4 solution (TiO
2 substrate), which becomes more intense with illumination (
Figure 3b), may be considered to have a common origin with the peak in
Figure 8a. The peak is further enhanced for annealed electrodes processed in Na
2SO
4 solution (
Figure 9a at
ca. −0.55 V) and even more intense when a Ti substrate is used instead of TiO
2 (
Figure 9b); in this case a broad hump can also be seen on the curve regarding the NaOH solution (at
ca. −0.50 V). Overall, for fixed potential-scan rate, the
peak location remains at about the same potential, independent of substrate, electrolyte, or application of thermal annealing. On the other hand, the
peak height is affected by all of the previous factors inasmuch as they change the electrode conductivity and surface-hole concentration. Thus, the use of metallic Ti substrate increases the electrode conductivity and also the peak height, while a similar effect is observed when the hole-scavenging ability of the solution is low, and also upon illumination; that is, factors that increase the hole concentration at the SC surface.
The local capacitance drop observed in
Figure 3,
Figure 8 and
Figure 9 between −0.60 and −0.40 V can be attributed to the formation of a surface dielectric layer. In their
C−2 vs.
U plots, recorded with potentiodynamic EI spectroscopy on anodic TiO
2 layers in contact with acidic electrolyte, Bondarenko et al. [
18] observed a slight bowing, similar to the one appearing at about −0.60 V in
Figure 3a and b for TiO
2/CdSe//Na
2SO
4, which they attributed to the presence of a thin layer of different composition generated on the electrode surface by H
+ intercalation in the TiO
2 lattice. In our case, the dielectric layer is probably amorphous selenium produced by (photo)corrosion during the measurement process; CdSe is known to undergo a hole-mediated decomposition, which is catalyzed by light [
10,
13,
19]:
The dynamic features of the EI measurement process with our SC electrodes, involving a decrease in peak height and displacement of its location to more positive potentials during successive voltage scans (
Figure 8a), may be illustrated by considering transitions from high to low capacitance lines as the corrosion product progressively passivates the electrode and its growth rate decreases. The elements of this analysis have been previously addressed; in their general approach, Fabregat-Santiago et al. [
20] suggested that complete coverage of a SC electrode by a thin dielectric surface layer results in a parallel shift of the Mott-Schottky plot to higher
C−2 values, due to capacitance drop, whereas an incomplete coverage of the electrode manifests itself as a slope increase, i.e., again a capacitance drop.
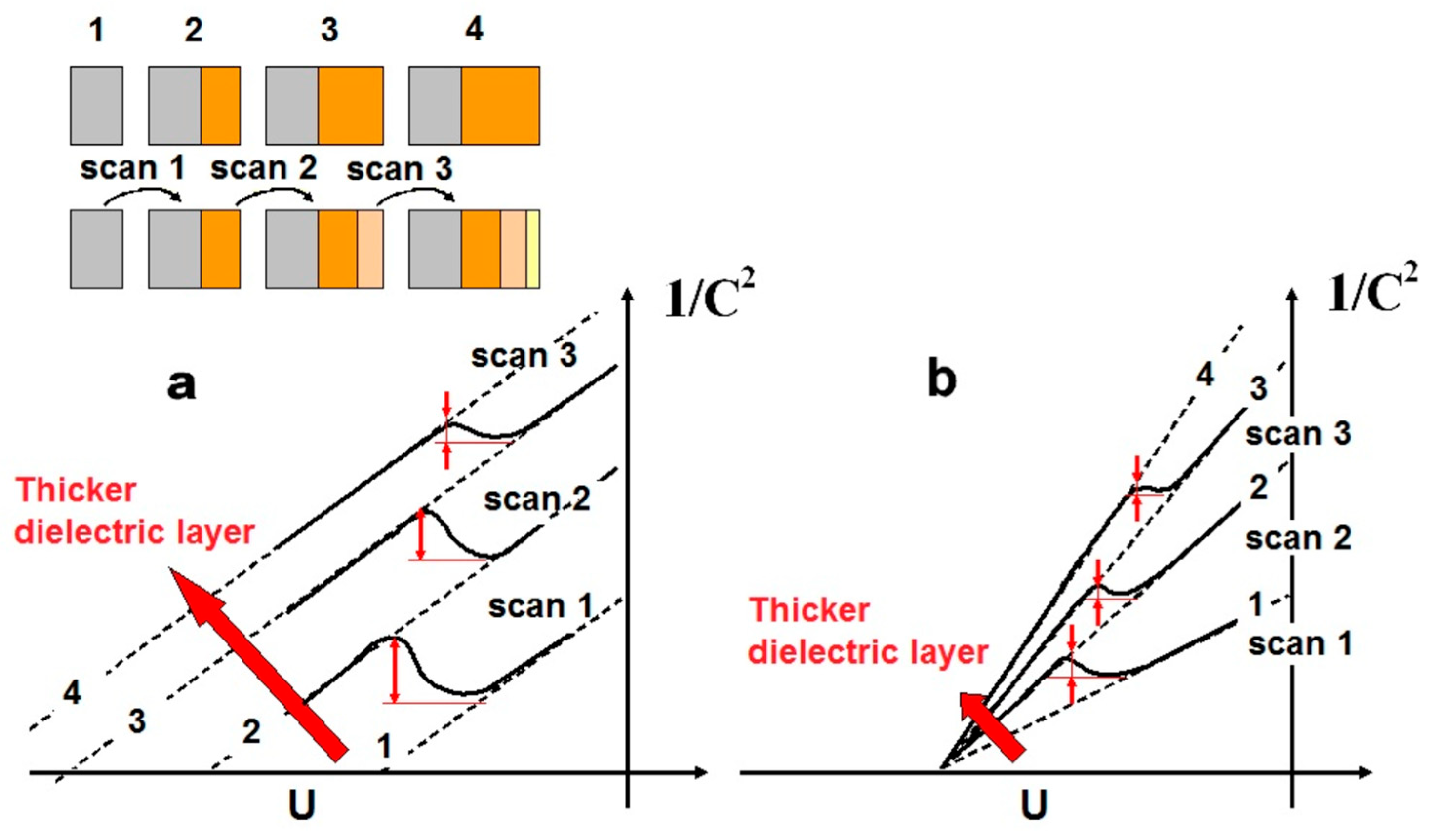
The evolution of the Mott–Schottky line is modeled in
Figure 10; the 1/
C2 plot follows initially the Mott–Schottky line of the bare CdSe electrode (dashed line 1) and gradually, as the scan proceeds towards negative potentials, goes over the dashed line 2 of a completely (
Figure 10a) or partly covered electrode (
Figure 10b). The transition is accompanied by a curve bending as shown and formation of a peak; in effect, the larger the increase of the Se layer thickness during a single scan, the more pronounced is the peak and more negative the potential it occurs. Thus, the peak height progressively decreases as the dielectric layer added at each consecutive scan is thinner, while the peak is displaced to more positive potentials. Growth of the Se layer ceases when the electrolyte dissolves the chalcogen as fast as it is formed, i.e., when the rate of Se production by the photocorrosion reaction becomes equal to its dissolution rate. In the presence of a redox couple in solution (as SPS), the equilibrium point is reached soon and the thickness of the Se layer remains small since the oxidation of the reduced form of the redox couple competes with the anodic corrosion of CdSe. By contrast, in the background solutions (and also FFC), the corrosion rate is high and the equilibrium point corresponds to a significantly larger thickness of the dielectric layer. As a result, whereas in SPS the observed peak quickly disappears and can be observed only in fast potential-scan experiments, in the other solutions it can be observed even by slow potential scanning.