Submitted:
17 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
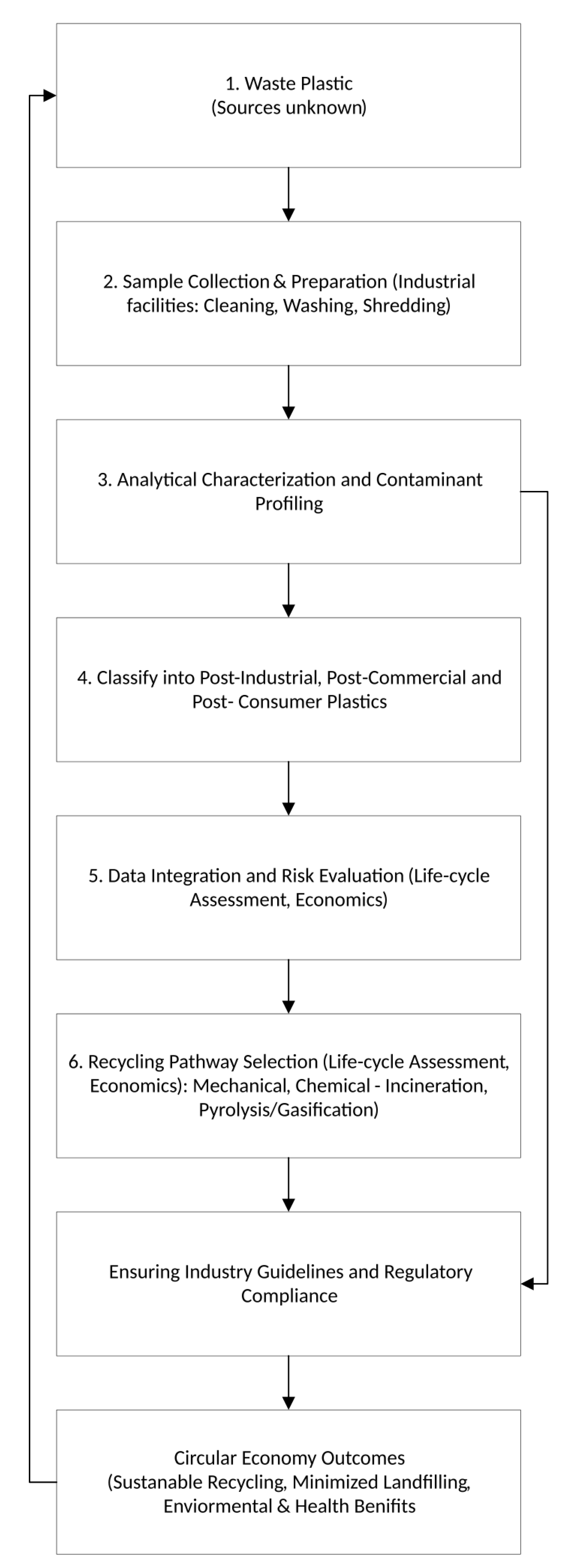
1.1. Post-consumer, post-industrial, and post-commercial plastic waste types
1.2. Literature Survey
1.3. Advanced Analytical Approaches for Mixed Plastics
1.4. Integrating Characterization with Recycling Strategies: Gaps and Outlook
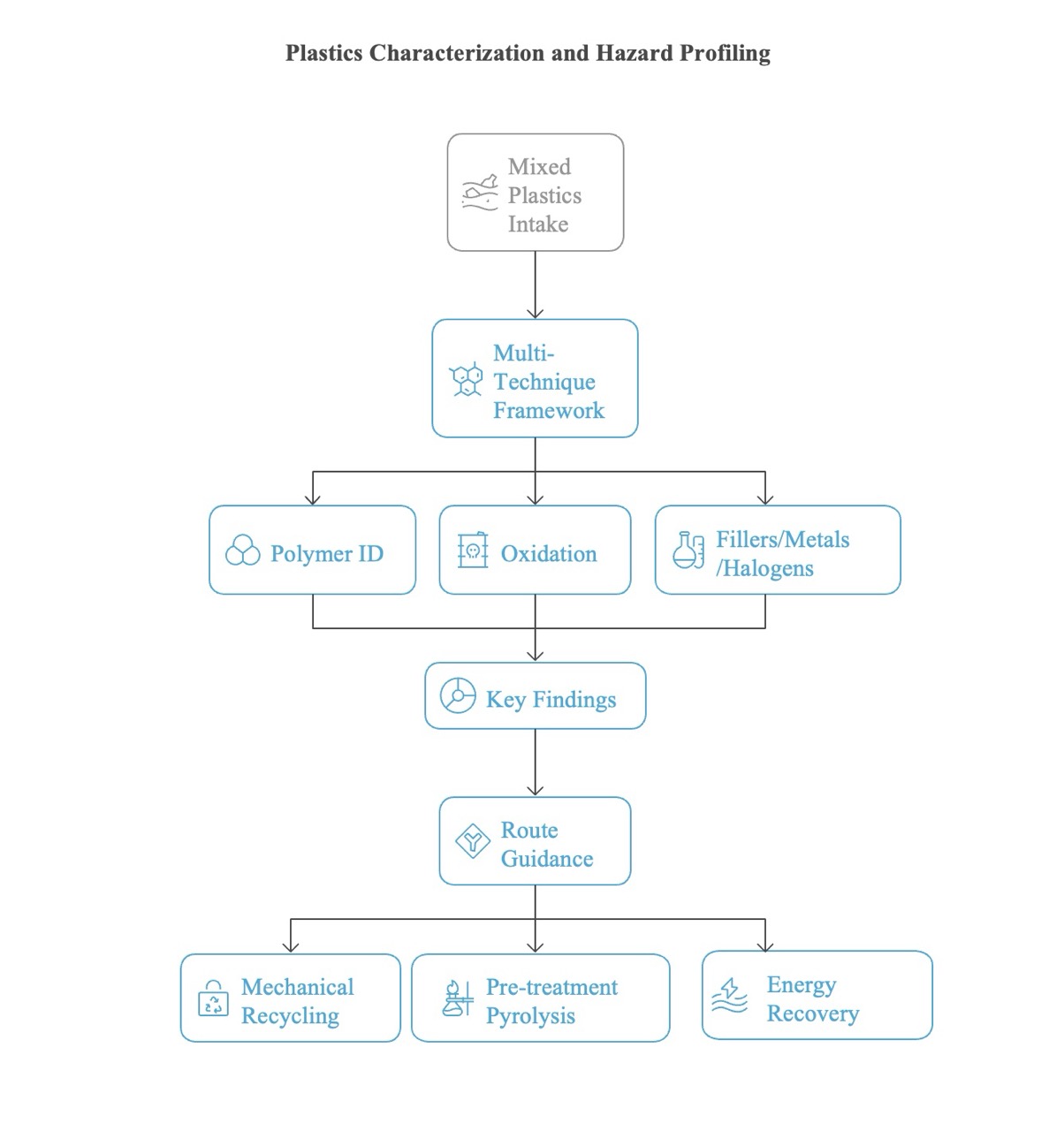
1.5. Integrated Analytical and Recycling Framework for Mixed Plastic Waste
1.6. Goals and Objectives of the Present Study
2. Materials and Methods
2.1. Plastic Waste Samples (P1–P9)
2.2. Analytical Methodology for Plastic Waste Characterization: Instrumentation and Experimental Procedures
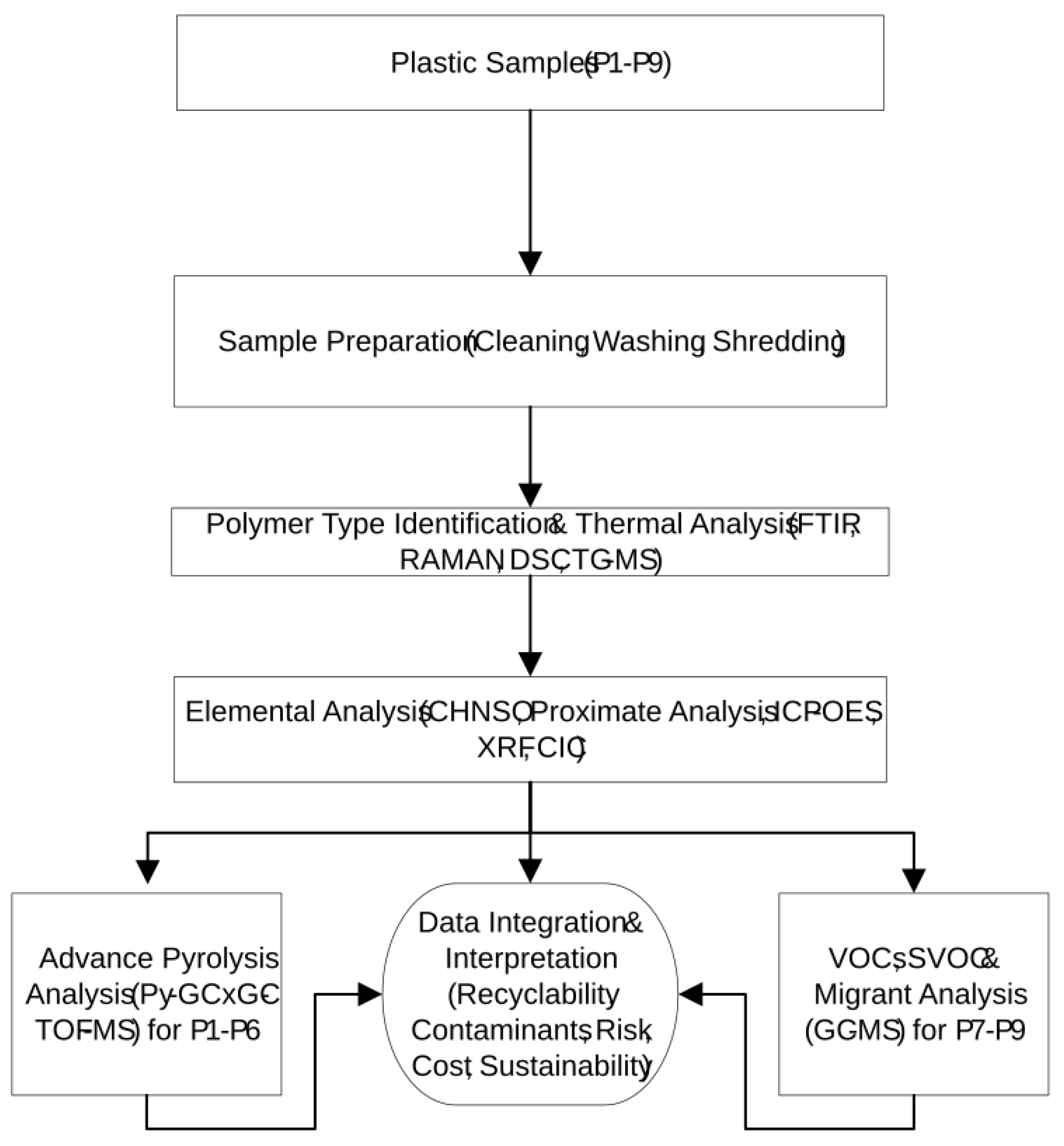
3. Results and Discussions
3.1. Identification of waste plastic samples (FTIR, Raman, Thermal Analysis)
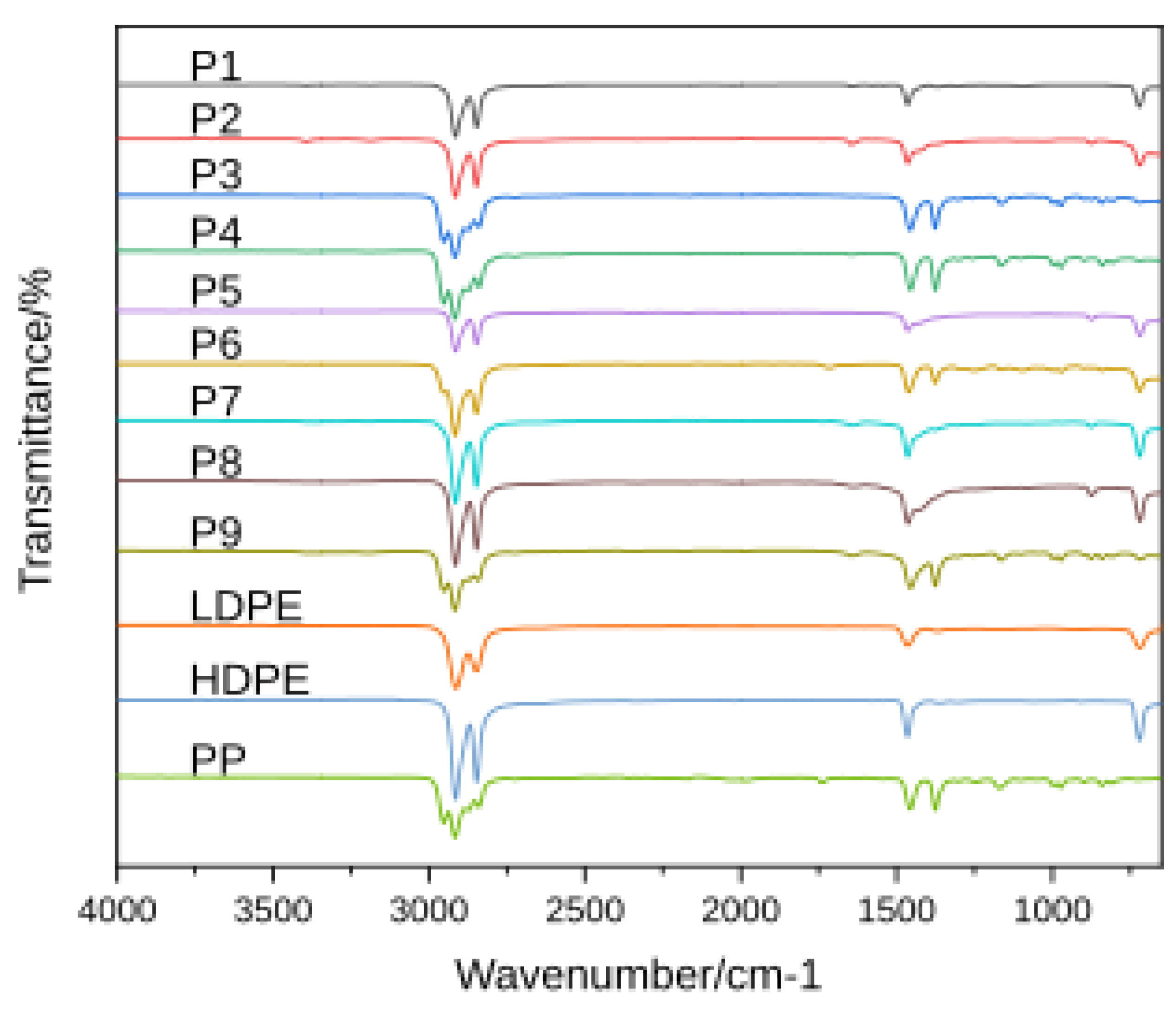
FTIR Spectroscopy
RAMAN spectroscopy
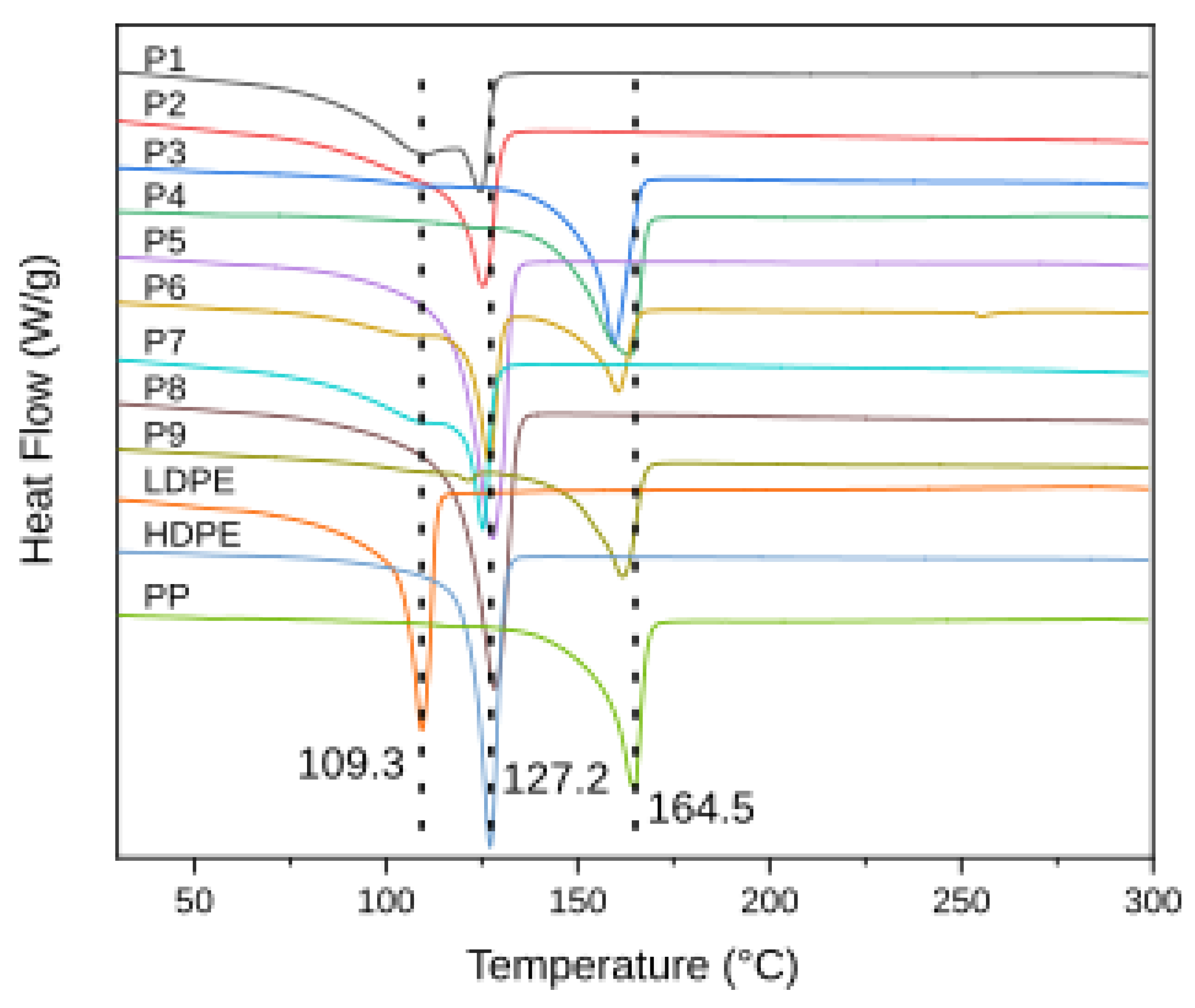
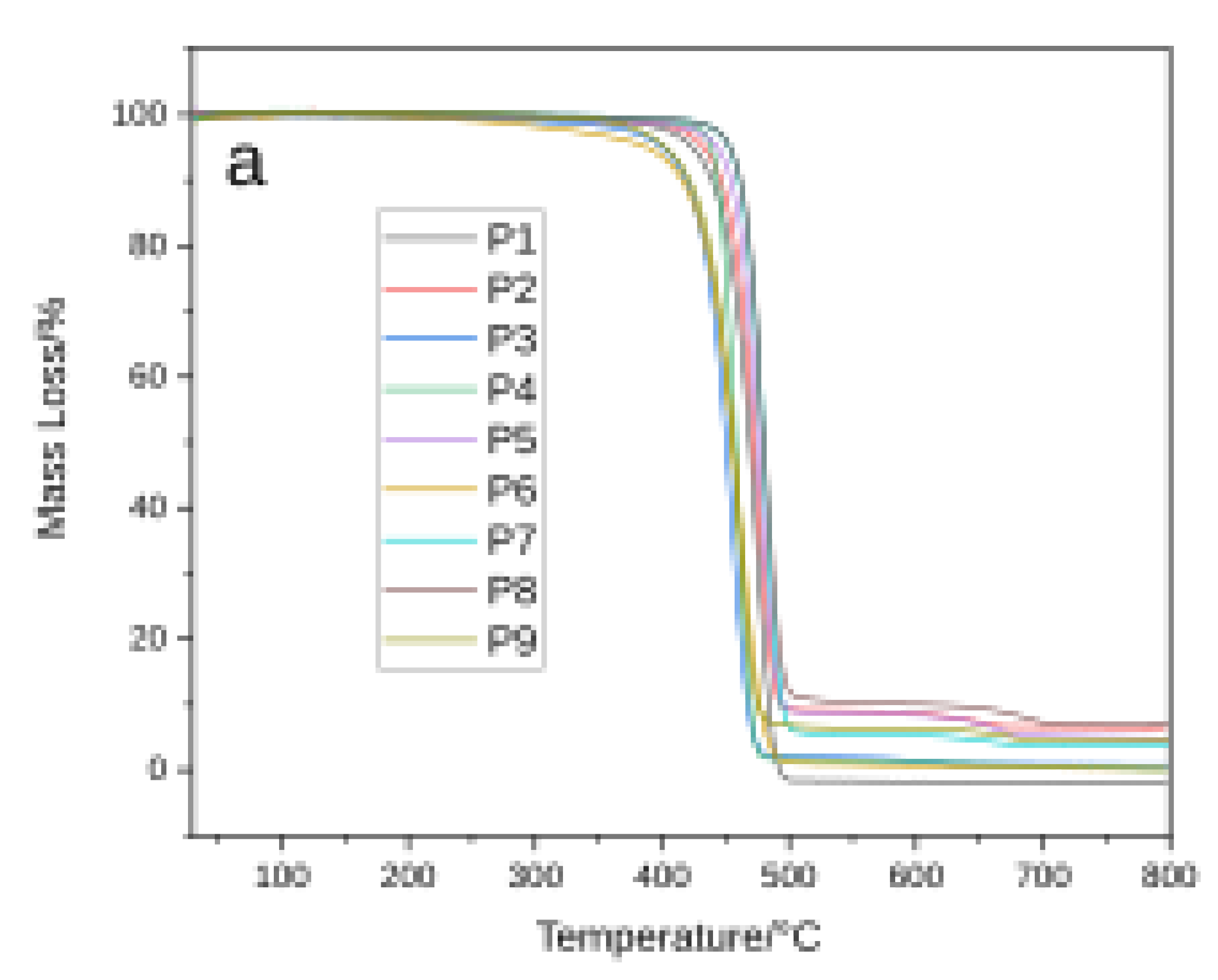
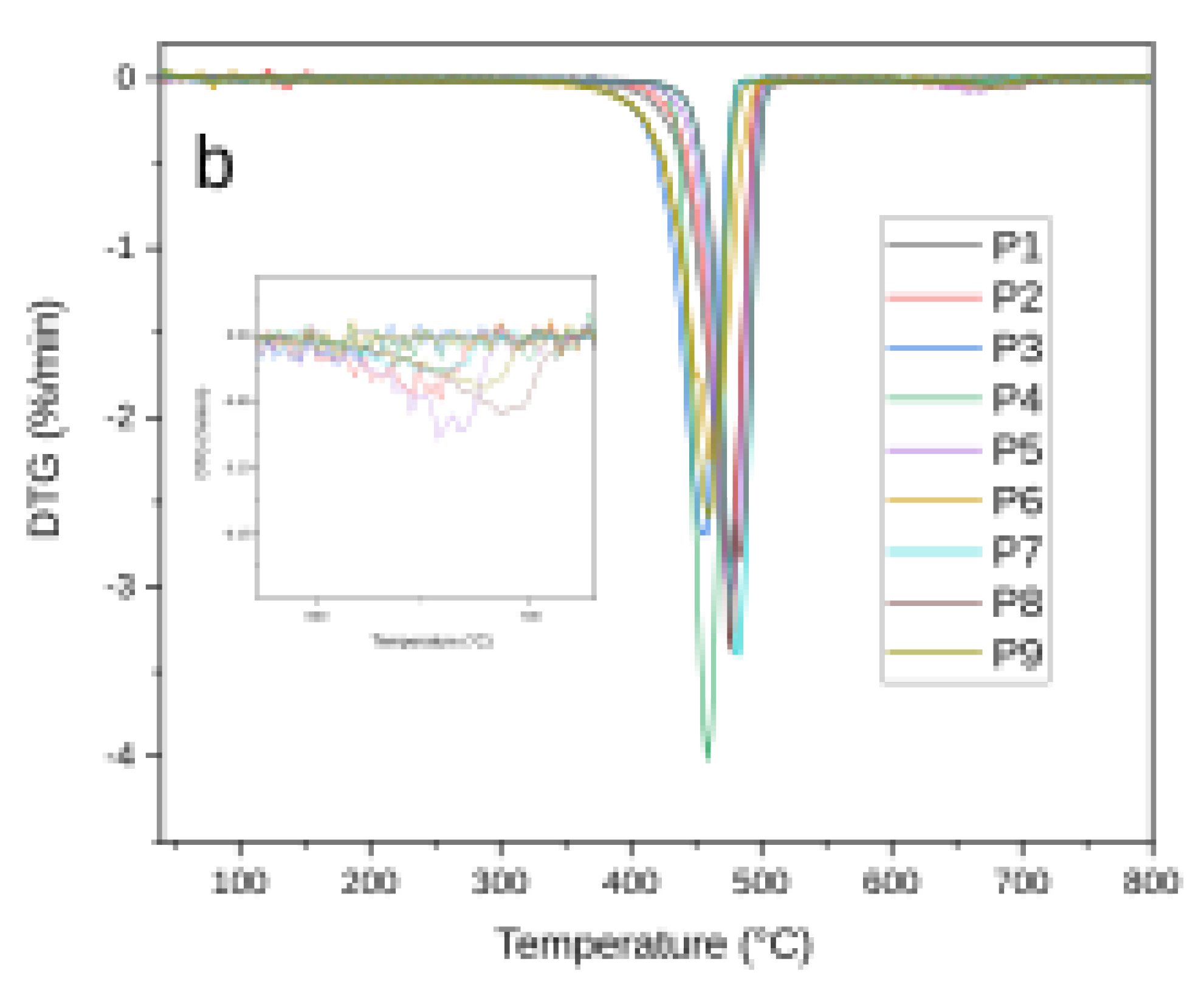
3.2. Thermal Analysis
| Sample | Dominant m/z Fragments | Key Evolved Species (assignment) | Interpretation (Polymer Type & Additives) |
|---|---|---|---|
| P1 | 28, 29, 41, 43 | C₂H₄ (ethylene), C₂H₆ (ethane), C₃H₆ (propylene), C₃H₈ (propane) | Pure PE (HDPE/LDPE); typical C₂–C₃ hydrocarbon gases; no filler (no CO₂ detected). |
| P2 | 18, 28, 29, 41, 43, 44 | H₂O (water), C₂H₄, C₂H₆, C₃H₆, C₃H₈, CO₂ (carbon dioxide) | HDPE; usual PE pyrolysis volatiles plus CO₂ release from CaCO₃ filler and elevated H₂O from additives/aging (NIAS). |
| P3 | 28, 41, 43 | C₂H₄, C₃H₆, C₃H₈ | Polypropylene; dominated by C₃ hydrocarbons (propylene/propane); no filler or moisture evident. |
| P4 | 28, 41, 43 | C₂H₄, C₃H₆, C₃H₈ | Polypropylene (similar to P3); mainly C₃ pyrolysis products; no filler present. |
| P5 | 28, 29, 41, 43, 44 | C₂H₄, C₂H₆, C₃H₆, C₃H₈, CO₂ | LDPE (with minor HDPE); typical PE volatiles plus CO₂ from significant CaCO₃ filler. |
| P6 | 28, 29, 41, 43 | C₂H₄, C₂H₆, C₃H₆, C₃H₈ | Mixed PE/PP; emits both C₂ and C₃ fragments consistent with a PE–PP blend; no notable CO₂ (minimal filler). |
3.3. Comprehensive elemental, trace metal, and halogen profiling
| Plastics | Proximate analysis (wt%) Proximate analysis (wt%) Proximate analysis (wt%) Proximate analysis (wt%) |
|||
|---|---|---|---|---|
| Plastics | Moisture | Volatile matter | Fixed Carbon | Ash |
| P1 | 0.07 | 99.41 | 0.02 | 0.50 |
| P2 | 0.12 | 93.29 | 0.01 | 6.58 |
| P3 | 0.05 | 96.80 | 0.93 | 2.22 |
| P4 | 0.07 | 98.92 | 0.90 | 0.11 |
| P5 | 0.07 | 89.38 | 0.34 | 10.21 |
| P6 | 0.13 | 97.00 | 0.99 | 1.88 |
| P7 | 0.06 | 96.10 | 1.01 | 2.83 |
| P8 | 0.02 | 92.62 | 2.70 | 4.66 |
| P9 | 0.20 | 95.58 | 0.95 | 3.37 |
Proximate Analysis
Ultimate analysis
| Plastics | Ultimate analysis (wt%) Ultimate analysis (wt%) Ultimate analysis (wt%) Ultimate analysis (wt%) Ultimate analysis (wt%) |
||||
|---|---|---|---|---|---|
| Plastics | C | H | N | O | Total |
| P1 | 85.90 | 14.03 | NDa | 1.23 | 101.16 |
| P2 | 79.90 | 12.68 | ND | 1.46 | 94.05 |
| P3 | 84.20 | 13.43 | ND | 0.92 | 98.55 |
| P4 | 86.04 | 13.66 | ND | 0.61 | 100.31 |
| P5 | 84.03 | 13.15 | ND | 1.37 | 98.55 |
| P6 | 84.71 | 13.36 | ND | 0.82 | 98.88 |
| P7 | 82.92 | 14.03 | 0.14 | 1.52 | 98.60 |
| P8 | 79.80 | 13.23 | 0.14 | 2.85 | 96.02 |
| P9 | 82.57 | 13.94 | 0.11 | 2.00 | 98.63 |
ICP-OES
Halogen Analysis (CIC)
| Sample | F (ppm) | Cl (ppm) | Br (ppm) | I (ppm) | |||||
|---|---|---|---|---|---|---|---|---|---|
| P1 | ND. | 488.14 | ND. | ND. | |||||
| P2# | ND. | ND. | ND. | ND. | |||||
| P3 | ND. | 120.94 | ND. | ND. | |||||
| P4 | ND. | 189.52 | ND. | ND. | |||||
| P5 | ND. | 67.57 | ND. | ND. | |||||
| P6 | ND. | 47.30 | ND. | ND. | |||||
| P7 | ND. | 179.36 | ND. | ND. | |||||
| P8 | ND. | 116.94 | ND. | ND. | |||||
| P9 | ND. | 62.41 | ND. | ND. | |||||
|
#P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. #P2 sample was not enough for this analysis. Hence no result. | |||||||||
| Detect limitation: 1ppm Detect limitation: 1ppm Detect limitation: 1ppm |
|||||||||
| ND less than 1ppm ND less than 1ppm ND less than 1ppm |
|||||||||
Elemental Signatures and Potential Sources in Plastic Waste Samples
| Element | Detected in Samples | Source Description |
|---|---|---|
| Al (Aluminum) | P1–P6 | Commonly present from aluminum foil layers in plastic packaging (metalized films or laminate foils used as food packaging)[66]. Aluminum-based additives are also sometimes used in polymers.[67] |
| Ca (Calcium) | P1–P5 | Frequently found as filler or stabilizer in plastics. For example, calcium carbonate (CaCO₃) is a common filler to improve mechanical properties and reduce cost, and calcium compounds (like calcium stearate) serve as PVC heat stabilizers[23,68,69]. |
| Cl (Chlorine) | P3, P4, P6 | Largely originates from PVC plastic and chlorinated additives. Polyvinyl chloride contains about 56–67% chlorine by weight, making chlorine a major component of PVC polymers[65]. Chlorinated flame retardants or plasticizers can also introduce chlorine into plastic waste. |
| Cu (Copper) | P1, P4, P6 | Often detected due to copper-based pigments and electrical components. For instance, copper phthalocyanine is a widely used blue/green pigment in plastics[70], and shredded e-waste plastics may contain embedded copper wiring or circuitry[71]. |
| Fe (Iron) | P1–P6 | Common from iron oxide pigments and steel wear debris. Iron oxides (e.g. Fe₂O₃) are used as color pigments (red, brown, black) in plastic products[72]. Additionally, metal particles from machinery (e.g. shredder or extruder wear) can contribute iron contamination in recycled plastics[73]. |
| K (Potassium) | P2, P3 | Potassium in plastic waste is often a residue from fertilizers or organic matter like MSW and sludge[74]. Plastic packaging used for potash or other K-containing fertilizers can carry fertilizer dust, and agricultural plastics contaminated with soil/manure may contain potassium from those sources[75]. |
| Mg (Magnesium) | P1, P3–P6 | Magnesium compounds are added to plastics as stabilizers and flame retardants. For example, magnesium hydroxide (Mg(OH)₂) is a common halogen-free flame retardant in polyolefin cables and plastics[76], and magnesium stearate is used as a lubricant/stabilizer in polymer processing[77] and as food additive[78]. |
| Na (Sodium) | P1–P6 | Sodium in plastic samples often comes from salt or other sodium-containing residues. Sea plastics can be encrusted with sodium chloride, and food packaging (e.g. for salty foods or brine) may leave Na deposits[79]. Sodium compounds are also used in some additives (e.g. sodium-based stabilizers or blowing agents). |
| Nb (Niobium) | P2 | Niobium is a less common element in plastics, but it can appear from electronic components. Niobium is used in some electronic capacitors (as a substitute for tantalum) and specialty alloys, so e-waste plastics or wiring can introduce niobium contaminants[80]. |
| P (Phosphorus) | P1–P6 | Widely present due to organophosphate additives. Phosphorus-based flame retardants and plasticizers are common – for example, tris(phenyl) phosphate (TPP) is used as a flame-retardant plasticizer in PVC and engineering plastics. Other phosphorous flame retardants (e.g. aryl phosphates, red phosphorus) are added to plastics to improve fire resistance[81]. |
| Ru (Ruthenium) | P3, P5 | Rarely present except via catalyst residues. Ruthenium is used as an industrial catalyst in certain chemical and polymer processes. For instance, Ru-based catalysts are employed in advanced plastic upcycling and hydrogenation (e.g. Ru/TiO₂ catalyzes polypropylene breakdown to hydrocarbons)[82]. Trace Ru in plastic waste may indicate residual catalyst contamination from such processes. |
| S (Sulfur) | P1, P3–P6 | Sulfur is found in plastics from both pigments and additives[83]. Certain inorganic pigments (like cadmium sulfide yellow or zinc sulfide in lithopone white) contain sulfur, and many rubber/plastic products are vulcanized with sulfur. Sulfur-based flame retardants and antioxidant additives (e.g. thioethers) also leave sulfur residues[53]. |
| Si (Silicon) | P1–P6 | Indicates silica-based fillers or silicone residues[84]. Silicon additives appear as fillers (e.g. talc is a Mg–silicate, and fumed silica is used to reinforce plastics) and in sealants/adhesives (silicone rubbers) present in plastic assemblies. X-ray analysis often finds silicon-based fillers added to improve plastic strength. |
| Sn (Tin) | P1 | Primarily from organotin stabilizers in PVC. Organotin compounds (like tributyltin and dibutyltin dilaurate) have been used as heat stabilizers in vinyl plastics[85]. The detection of Sn in plastic usually signals the presence of these PVC stabilizers. (Tin may also come from solder or metal foil contamination, but in polymers it’s chiefly from additives.) |
| Sr (Strontium) | P7, P8, P9 | Strontium (Sr) found in plastic waste likely originates from industrial contamination, recycled fillers, or pigments. Common sources include coal combustion residues, ceramics, and glass manufacturing, where Sr is used as a functional additive or colorant. Typically, found in MSW. |
| Ti (Titanium) | P2, P4–P6 | Titanium in plastics is mainly from titanium dioxide (TiO₂) pigment and UV stabilizers[86]. TiO₂ is the dominant white pigment in plastics, added for color and UV protection. It is routinely identified as a filler in plastic waste analyses. Other Ti additives (like organotitanate coupling agents) are also used for UV stabilization[87]. |
| V (Vanadium) | P3, P5 | Vanadium tends to enter plastics via hydrocarbon residues. Heavy fuel oils and lubricants contain vanadium (a trace element in crude oil), so plastic debris tainted with bunker fuel or engine oil can show V contamination[88,89]. Historically, some Ziegler-Natta polymerization catalysts used vanadium, but the most common pathway in waste is fuel/oil staining[90]. |
| Zn (Zinc) | P1–P6 | Ubiquitous in plastics as a stabilizer and pigment component. Zinc compounds (like zinc stearate and zinc oxide) are used as heat stabilizers in PVC and as UV absorbers[91]. Zinc oxide and sulfide appear in white pigments and fillers (e.g. lithopone = ZnS·BaSO₄)[92]. As a result, Zn is commonly detected in plastic waste elemental analyses. |
| Zr (Zirconium) | P2 | Typically comes from ceramic or coating contaminants. Zirconium-based compounds are used in ceramic materials and also as paint/ink additives. For example, zirconium chemicals in printing inks promote adhesion and heat resistance – printed plastic packaging or ceramic dust can thus introduce Zr[93]. It is not a deliberate plastic ingredient but enters via these contamination pathways. |
4. Discussion and Recycling Recommendations
5. Conclusion
| List of Acronyms | ||
| Acronym | Full Form | |
| ABS | Acrylonitrile Butadiene Styrene Acrylonitrile Butadiene Styrene |
|
| ATR | Attenuated Total Reflectance Attenuated Total Reflectance |
|
| BFR | Brominated Flame Retardants Brominated Flame Retardants |
|
| CERP | Clean Energy Research Platform Clean Energy Research Platform |
|
| CHNSO | Carbon, Hydrogen, Nitrogen, Sulfur, and Oxygen (Ultimate Elemental Analysis) Carbon, Hydrogen, Nitrogen, Sulfur, and Oxygen (Ultimate Elemental Analysis) |
|
| CIC | Combustion Ion Chromatography Combustion Ion Chromatography |
|
| DSC | Differential Scanning Calorimetry Differential Scanning Calorimetry |
|
| DTG | Derivative Thermogravimetry Derivative Thermogravimetry |
|
| EU | European Union European Union |
|
| FTIR | Fourier Transform Infrared Spectroscopy Fourier Transform Infrared Spectroscopy |
|
| GC-MS | Gas Chromatography–Mass Spectrometry Gas Chromatography–Mass Spectrometry |
|
| GC×GC–TOF–MS | Comprehensive Two-Dimensional Gas Chromatography with Time-of-Flight Mass Spectrometry Comprehensive Two-Dimensional Gas Chromatography with Time-of-Flight Mass Spectrometry |
|
| GPC | Gel Permeation Chromatography Gel Permeation Chromatography |
|
| HDPE | High-Density Polyethylene High-Density Polyethylene |
|
| ICP-OES | Inductively Coupled Plasma–Optical Emission Spectroscopy Inductively Coupled Plasma–Optical Emission Spectroscopy |
|
| LDPE | Low-Density Polyethylene Low-Density Polyethylene |
|
| MSW | Municipal Solid Waste Municipal Solid Waste |
|
| NIAS | Non-Intentionally Added Substances Non-Intentionally Added Substances |
|
| PSE | Physical Science and Engineering Division (KAUST) Physical Science and Engineering Division (KAUST) |
|
| PET | Polyethylene Terephthalate Polyethylene Terephthalate |
|
| PMMA | Polymethyl Methacrylate Polymethyl Methacrylate |
|
| POPs | Persistent Organic Pollutants Persistent Organic Pollutants |
|
| PP | Polypropylene Polypropylene |
|
| PS | Polystyrene | |
| PVC | Polyvinyl Chloride Polyvinyl Chloride |
|
| Py-GC/MS | Pyrolysis Gas Chromatography–Mass Spectrometry Pyrolysis Gas Chromatography–Mass Spectrometry |
|
| RoHS | Restriction of Hazardous Substances Restriction of Hazardous Substances |
|
| SCW | Supercritical Water Supercritical Water |
|
| SDG | Sustainable Development Goals Sustainable Development Goals |
|
| SVOCs | Semi-Volatile Organic Compounds Semi-Volatile Organic Compounds |
|
| TGA | Thermogravimetric Analysis Thermogravimetric Analysis |
|
| TG–MS | Thermogravimetry–Mass Spectrometry Thermogravimetry–Mass Spectrometry |
|
| UNEP | United Nations Environment Programme United Nations Environment Programme |
|
| VOCs | Volatile Organic Compounds Volatile Organic Compounds |
|
| WD-XRF | Wavelength Dispersive X-ray Fluorescence Wavelength Dispersive X-ray Fluorescence |
|
| WEEE | Waste Electrical and Electronic Equipment Waste Electrical and Electronic Equipment |
|
| XRF | X-ray Fluorescence X-ray Fluorescence |
|
Acknowledgments
Data availability
Declaration of Competing Interest:
References
- Stegmann, P., et al., Plastic futures and their CO2 emissions. Nature, 2022. 612(7939): p. 272-276.
- Jensen, A.C.a.M.S. Plastic pollution is a public health crisis. How do we reduce plastic waste? 2022.
- Chaine, C., et al., Recycling plastics from WEEE: a review of the environmental and human health challenges associated with brominated flame retardants. International journal of environmental research and public health, 2022. 19(2): p. 766.
- Dzierżyński, E., et al., Microplastics in the human body: Exposure, detection, and risk of carcinogenesis: A state-of-the-art review. Cancers, 2024. 16(21): p. 3703.
- Tsakona, M., Drowning in plastics: marine litter and plastic waste vital graphics. 2021.
- Turner, A., Black plastics: Linear and circular economies, hazardous additives and marine pollution. Environment international, 2018. 117: p. 308-318.
- Wang, S., International law-making process of combating plastic pollution: Status Quo, debates and prospects. Marine Policy, 2023. 147: p. 105376.
- OECD, Global Plastics Outlook: Economic Drivers, Environmental Impacts and Policy Options. 2022, Organisation for Economic Co-operation and Development (OECD) Publishing: Paris.
- Aanesen, M., et al., Insights from international environmental legislation and protocols for the global plastic treaty. Scientific Reports, 2024. 14(1): p. 2750. 1.
- Vince, J., et al., The Zero Draft Plastics Treaty: Gaps and challenges. Cambridge Prisms: Plastics, 2024. 2: p. e24.
- Sinkevičius, V. EU calls for agreement on global rules to end plastic pollution, in EU Environment newsletter. 2023, European Commission, Energy, Climate change, Environment.: Directorate-General for Environment, Brussel, Belgium.
- Wikipedia, Plastic bag ban. 2025, Wikipedia.
- Herberz, T., C.Y. Barlow, and M. Finkbeiner, Sustainability assessment of a single-use plastics ban. Sustainability, 2020. 12(9): p. 3746.
- Saxena, S., Pyrolysis and Beyond: Sustainable Valorization of Plastic Waste. Applications in Energy and Combustion Science, 2024: p. 100311.
- Hansen, E., et al., Hazardous substances in plastic materials. COWI in cooperation with Danish Technological Institute, 2013: p. 7-8.
- Ragaert, K., L. Delva, and K. Van Geem, Mechanical and chemical recycling of solid plastic waste. Waste management, 2017. 69: p. 24-58.
- Villanueva, A. and P. Eder, End-of-waste criteria for waste plastic for conversion. Institute for prospective Technological studies, 2014.
- Barrick, A., et al., Plastic additives: Challenges in ecotox hazard assessment. PeerJ, 2021. 9: p. e11300.
- Hahladakis, J.N., et al., An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. Journal of hazardous materials, 2018. 344: p. 179-199.
- Cook, E., C.A. Velis, and M. Derks, Plastic waste reprocessing for circular economy: A systematic review of risks to occupational and public health from legacy substances and extrusion. 2020.
- Santos, M., et al., The Hidden Risks of Recycled Plastic Toys: A Literature Review on Legacy Additives and Child Safety. Sustainability & Circularity NOW, 2025. 2(continuous publication).
- Sharkey, M., et al., Phasing-out of legacy brominated flame retardants: The UNEP Stockholm Convention and other legislative action worldwide. Environment International, 2020. 144: p. 106041.
- Xanthos, M., Functional fillers for plastics. 2010: John Wiley & Sons.
- Radhakrishnan, H., et al., Influence of Functional Additives, Fillers, and Pigments on Thermal and Catalytic Pyrolysis of Polyethylene for Waste Plastics Upcycling. Green Chemistry, 2025.
- Aguado, J., D. Serrano, and G. San Miguel, Analysis of products generated from the thermal and catalytic degradation of pure and waste polyolefins using Py-GC/MS. Journal of Polymers and the Environment, 2007. 15: p. 107-118.
- Sørensen, L., et al., UV degradation of natural and synthetic microfibers causes fragmentation and release of polymer degradation products and chemical additives. Science of the Total Environment, 2021. 755: p. 143170.
- Horodytska, O., A. Cabanes, and A. Fullana, Non-intentionally added substances (NIAS) in recycled plastics. Chemosphere, 2020. 251: p. 126373.
- Kato, L.S. and C.A. Conte-Junior, Safety of plastic food packaging: the challenges about non-intentionally added substances (NIAS) discovery, identification and risk assessment. Polymers, 2021. 13(13): p. 2077.
- Wiesinger, H., Z. Wang, and S. Hellweg, Deep dive into plastic monomers, additives, and processing aids. Environmental science & technology, 2021. 55(13): p. 9339-9351.
- Almroth, B.C., Scientists found hundreds of toxic chemicals in recycled plastics., in ScienceDaily. 2023, ScienceDaily: 2023.
- Almroth, B.C., et al., Addressing the toxic chemicals problem in plastics recycling. Cambridge Prisms: Plastics, 2025. 3: p. e3.
- Carmona, E., et al., A dataset of organic pollutants identified and quantified in recycled polyethylene pellets. Data in brief, 2023. 51: p. 109740.
- Geueke, B., et al., Systematic evidence on migrating and extractable food contact chemicals: most chemicals detected in food contact materials are not listed for use. CritiCal reviews in Food sCienCe and nutrition, 2023. 63(28): p. 9425-9435.
- Geueke, B., et al., Hazardous chemicals in recycled and reusable plastic food packaging. Cambridge Prisms: Plastics, 2023. 1: p. e7.
- Groh, K.J., et al., Overview of known plastic packaging-associated chemicals and their hazards. Science of the total environment, 2019. 651: p. 3253-3268.
- Groh, K.J., et al., Overview of intentionally used food contact chemicals and their hazards. Environment International, 2021. 150: p. 106225.
- Coniglio, M.A., et al., Non-intentionally added substances. 2020: Springer.
- Ong, H.-T., H. Samsudin, and H. Soto-Valdez, Migration of endocrine-disrupting chemicals into food from plastic packaging materials: an overview of chemical risk assessment, techniques to monitor migration, and international regulations. Critical reviews in food science and nutrition, 2022. 62(4): p. 957-979.
- Turner, A. and M. Filella, Hazardous metal additives in plastics and their environmental impacts. Environment International, 2021. 156: p. 106622.
- Nevondo, V., et al., Analytical insights into short-chain chlorinated paraffins in consumer products, leachates, and sediments in Gauteng, South Africa. International Journal of Environmental Science and Technology, 2025: p. 1-18.
- Bifulco, A., et al., Recycling of flame retardant polymers: Current technologies and future perspectives. Journal of Materials Science & Technology, 2024.
- Carney Almroth, B., et al., Chemical simplification and tracking in plastics. Science, 2023. 382(6670): p. 525-525.
- Wrona, M., et al., Identification of Intentionally and Non-intentionally Added Substances in Recycled Plastic Packaging Materials, in Food Packaging Materials: Current Protocols. 2024, Springer. p. 75-98.
- Núñez, S.S., et al., Heavy metals, PAHs and POPs in recycled polyethylene samples of agricultural, post-commercial, post-industrial and post-consumer origin. Waste Management, 2022. 144: p. 113-121.
- Long, F., et al., Online characterization of mixed plastic waste using machine learning and mid-infrared spectroscopy. ACS Sustainable Chemistry & Engineering, 2022. 10(48): p. 16064-16069. 48.
- Farrelly, T., et al., Independent science key to breaking stalemates in global plastics treaty negotiations. Cambridge Prisms: Plastics, 2025. 3: p. e6.
- Farrelly, T., et al., Global plastics treaty needs trusted science. Science, 2024. 384(6693): p. 281-281.
- Beccagutti, B., et al., Characterization of some real mixed plastics from WEEE: a focus on chlorine and bromine determination by different analytical methods. Sustainability, 2016. 8(11): p. 1107.
- Goedecke, C., et al., Evaluation of thermoanalytical methods equipped with evolved gas analysis for the detection of microplastic in environmental samples. Journal of Analytical and Applied Pyrolysis, 2020. 152: p. 104961.
- Hall, W.J. and P.T. Williams, Analysis of products from the pyrolysis of plastics recovered from the commercial scale recycling of waste electrical and electronic equipment. Journal of Analytical and Applied Pyrolysis, 2007. 79(1-2): p. 375-386.
- Hochegger, A. One-dimensional and comprehensive two-dimensional gas chromatographic approaches for the characterization of post-consumer recycled plastic materials . Analytical and Bioanalytical Chemistry 2023, 415(13), 2447–2457. [Google Scholar] [CrossRef]
- Kusch, P. Application of pyrolysis-gas chromatography/mass spectrometry (Py-GC/MS), in Comprehensive analytical chemistry; Elsevier, 2017; pp. 169–207. [Google Scholar]
- Cuthbertson, A.A. Characterization of polymer properties and identification of additives in commercially available research plastics . Green Chemistry 2024, 26(12), 7067–7090. [Google Scholar] [CrossRef]
- Gnoffo, C.; Frache, A. Identification of plastics in mixtures and blends through pyrolysis-gas chromatography/mass spectrometry . Polymers 2023, 16(1), 71. [Google Scholar] [CrossRef]
- Strien, J.R. Pyrolysis of Polyolefin-Enriched Mixed Plastic Waste Streams: Effects of Pretreatments and Presence of Hydrogen during Pyrolysis . Energy & Fuels 2024, 39(1), 686–698. [Google Scholar] [CrossRef]
- Lee, H.-J. Variation and uncertainty of microplastics in commercial table salts: critical review and validation . Journal of hazardous materials 2021, 402, 123743. [Google Scholar] [CrossRef]
- Roosen, M. Detailed analysis of the composition of selected plastic packaging waste products and its implications for mechanical and thermochemical recycling . Environmental science & technology 2020, 54(20), 13282–13293. [Google Scholar]
- Dahlbo, H. Recycling potential of post-consumer plastic packaging waste in Finland . Waste management 2018, 71, 52–61. [Google Scholar] [CrossRef]
- Budiyantoro, C.; et al. The effect of CaCO3 filler component on mechanical properties of polypropylene . IOP Conference Series: Materials Science and Engineering, 2018; IOP Publishing. [Google Scholar]
- Choi, S. The Effect of Coupling Agents and Graphene on the Mechanical Properties of Film-Based Post-Consumer Recycled Plastic . Polymers 2024, 16(3), 380. [Google Scholar] [CrossRef]
- Hernandez Fernandez, J., J. Carrascal Sanchez, and J. Lopez Martinez, Sustainable Catalysts from Industrial FeO Waste for Pyrolysis and Oxidation of Hospital Polypropylene in Cartagena . 2024.
- Uçar, S. The influence of the waste ethylene vinyl acetate copolymer on the thermal degradation of the waste polypropylene . Fuel processing technology 2008, 89(11), 1201–1206. [Google Scholar] [CrossRef]
- Kanan, S.; Samara, F. Dioxins and furans: A review from chemical and environmental perspectives . Trends in Environmental Analytical Chemistry 2018, 17, 1–13. [Google Scholar] [CrossRef]
- Gao, P. Pyrolysis of municipal plastic waste: Chlorine distribution and formation of organic chlorinated compounds . Science of The Total Environment 2024, 912, 169572. [Google Scholar] [CrossRef]
- Oberoi, S.; Malik, M. Polyvinyl chloride (PVC), chlorinated polyethylene (CPE), chlorinated polyvinyl chloride (CPVC), chlorosulfonated polyethylene (CSPE), polychloroprene rubber (CR)—Chemistry, applications and ecological impacts—I . In Ecological and Health Effects of Building Materials; 2022; pp. 33–52. [Google Scholar]
- Deshwal, G.K.; Panjagari, N.R. Review on metal packaging: Materials, forms, food applications, safety and recyclability . Journal of food science and technology 2020, 57(7), 2377–2392. [Google Scholar] [CrossRef]
- Saiyed, S.M.; Yokel, R.A. Aluminium content of some foods and food products in the USA, with aluminium food additives . Food additives and contaminants 2005, 22(3), 234–244. [Google Scholar] [CrossRef]
- DeArmitt, C., Functional fillers for plastics, in Applied plastics engineering handbook. 2011, Elsevier. p. 455-468.
- Ramakrishna, C., T. Thriveni, and A.J. Whan, The Role of Mineral Fillers for Enhancing the Mechanical Properties of Recycled Plastics . International Journal in IT & Engineering 2015, 3(3), 314–335.
- Cârdan, I. Blue micro-/nanoplastics abundance in the environment: a double threat as a Trojan horse for a plastic-Cu-phthalocyanine pigment and an opportunity for nanoplastic detection via micro-Raman spectroscopy . Environmental Science: Nano 2025, 12(4), 2357–2370. [Google Scholar] [CrossRef]
- Murali, A. Determination of metallic and polymeric contents in electronic waste materials and evaluation of their hydrometallurgical recovery potential . International Journal of Environmental Science and Technology 2022, 1–14. [Google Scholar] [CrossRef]
- Pfaff, G. Iron oxide pigments . Physical Sciences Reviews 2021, 6(10), 535–548. [Google Scholar] [CrossRef]
- Sadi, S.H.F. Recycling Waste Polymers from Automotive Shredder Residue (ASR); Application in Iron Making . 2013. [Google Scholar]
- Zhao, J. Slagging characteristics caused by alkali and alkaline earth metals during municipal solid waste and sewage sludge co-incineration . Energy 2020, 202, 117773. [Google Scholar] [CrossRef]
- Bley, H. Nutrient release, plant nutrition, and potassium leaching from polymer-coated fertilizer . Revista Brasileira de Ciência do Solo 2017, 41. [Google Scholar] [CrossRef]
- Li, Y. Recent advances in halogen-free flame retardants for polyolefin cable sheath materials . Polymers 2022, 14(14), 2876. [Google Scholar] [CrossRef]
- Grossman, R.F. Lubricants, in Polymer Modifiers and Additives; CRC Press, 2000; pp. 385–414. [Google Scholar]
- Hobbs, C.A. Magnesium stearate, a widely-used food additive, exhibits a lack of in vitro and in vivo genotoxic potential . Toxicology reports 2017, 4, 554–559. [Google Scholar] [CrossRef]
- Orriss, R. Effects of Contaminants in Ethylene Plants: Sodium and Iron . in AIChE Conference, Spring Meeting, 1996. [Google Scholar]
- Niu, B.; Xu, Z. Innovating e-waste recycling: From waste multi-layer ceramic capacitors to NbPb codoped and ag-Pd-Sn-Ni loaded BaTiO3 nano-photocatalyst through one-step ball milling process . Sustainable materials and technologies 2019, 21, p. e00101. [Google Scholar] [CrossRef]
- Mihajlović, I. Recent development of phosphorus flame retardants in thermoplastic blends and nanocomposites, in Flame retardants: Polymer blends, composites and nanocomposites; Springer, 2015; pp. 79–114. [Google Scholar]
- Kots, P.A. Polypropylene plastic waste conversion to lubricants over Ru/TiO2 catalysts . Acs Catalysis 2021, 11(13), 8104–8115. [Google Scholar] [CrossRef]
- Ranta-Korpi, M. Ash forming elements in plastics and rubbers.; VTT Technical Research Centre of Finland: Espoo, Finland, 2014; Volume 131. [Google Scholar]
- Chruściel, J.J. Silicon-Based Polymers and Materials; Walter de Gruyter GmbH & Co KG, 2022. [Google Scholar]
- Li, D.; Liu, P. Trends and prospects for thermal stabilizers in polyvinyl chloride . Journal of Vinyl and Additive Technology 2022, 28(4), 669–688. [Google Scholar] [CrossRef]
- Gázquez, M.J., S.M.P. Moreno, and J.P. Bolívar, TiO2 as white pigment and valorization of the waste coming from its production, in Titanium dioxide (Tio₂) and its applications; Elsevier, 2021; pp. 311–335.
- Alicja, K.; Grzegorz, C. Strontium leaching from municipal waste subjected to incineration . Environmental Geochemistry and Health 2024, 46(7), 220. [Google Scholar] [CrossRef]
- Vogel, C. Speciation of antimony and vanadium in municipal solid waste incineration ashes analyzed by XANES spectroscopy . Journal of Material Cycles and Waste Management 2024, 26(4), 2152–2158. [Google Scholar] [CrossRef]
- White, D.J. and L.S. Levy,Vanadium: environmental hazard or environmental opportunity? A perspective on some key research needs . Environmental Science: Processes & Impacts 2021, 23(4), 527–534. [CrossRef] [PubMed]
- Ishikura, H. Developments in vanadium-catalysed polymerisation reactions: A review . Inorganica Chimica Acta 2021, 515, 120047. [Google Scholar] [CrossRef]
- Morsy, A. Utilizing a blend of expandable graphite and calcium/zinc stearate as a heat stabilizer environmentally friendly for polyvinyl chloride . SPE Polymers 2024. 5, 1, 45–57. [Google Scholar] [CrossRef]
- Liu, W. Fabrication of anti-bacterial, hydrophobic and UV resistant galactomannan-zinc oxide nanocomposite films . Polymer 2021, 215, 123412. [Google Scholar] [CrossRef]
- Moles, P.J.M. Chemicals, The use of zirconium in Surface Coatings . Data sheet, 2002; 117776–1780. [Google Scholar]
| Sample ID | T0 (°C) | Tf (°C) | Tmax (°C) | ΔTG (%) |
| P1 | 365 | 502.2 | 475 | 99.99 |
| P2 | 376 | 503.3 | 476 | 90.77 |
| P2 | 611 | 673.3 | 658 | 2.32 |
| P2 | Total ΔTG Total ΔTG Total ΔTG |
93.09 | ||
| P3 | 353 | 485.5 | 458 | 98.23 |
| P4 | 416 | 486.3 | 459 | 99.23 |
| P5 | 392 | 519.9 | 477 | 91.23 |
| P5 | 620 | 679.9 | 667 | 3.69 |
| P5 | Total ΔTG Total ΔTG Total ΔTG |
94.92 | ||
| P6 | 355 | 504.7 | 462 | 99.29 |
| P7 | 417.9 | 520.4 | 482.9 | 94.89 |
| P7 | 660.4 | 1.3 | ||
| P7 | Total ΔTG Total ΔTG Total ΔTG |
96.19 | ||
| P8 | 410.3 | 535.3 | 475.3 | 93.25 |
| P8 | 687.8 | 3.12 | ||
| P8 | Total ΔTG Total ΔTG Total ΔTG |
96.37 | ||
| P9 | 376 | 503.3 | 460.7 | 95.6 |
| P9 | 680.7 | 1.86 | ||
| P9 | Total ΔTG Total ΔTG Total ΔTG |
97.46 | ||
| Sample | Mp | Mn | Mw | Mz |
| P1 | 147,040 | 44,959 | 238,891 | 683,064 |
| P2 | 137,869 | 44,937 | 228,606 | 596,549 |
| P3 | 156,596 | 76,526 | 197,794 | 365,870 |
| P4 | 188,593 | 56,685 | 250,303 | 575,682 |
| P5 | 99,747 | 43,309 | 235,918 | 899,948 |
| P6 | 122,743 | 48,623 | 170,115 | 375,282 |
| Sample | Identified Polymer(s) | Spectral Signatures (FTIR ) | Thermal Behavior (DSC & TGA/TG–MS) | Additives/Contamination Evidence | Suggested Origin |
|---|---|---|---|---|---|
| P1 | Poly(ethylene) – mixture of LDPE & HDPE | FTIR: CH₂ rocking and bending, C–H stretches indicative of PE; Raman confirms PE (matches standard). | DSC: two melting peaks at ~110 °C and 130 °C (LDPE & HDPE). TGA: single-stage decomposition ~475 °C with ~99% mass loss; TG–MS shows only hydrocarbon volatiles (C1–C3 alkanes/alkenes), typical of pure polyolefins. | No significant additives – no FTIR carbonyl (oxidation) band, negligible residue (<0.5% ash) indicating minimal inorganic content. | Post-Industrial |
| P2 | Poly(ethylene) – high-density (HDPE) | FTIR: CH₂ bending and rocking modes of PE, plus weak C=C and O–H bands suggesting slight oxidation; aromatic hints (trace impurities). Raman confirms PE polymer matrix. | DSC: single melting peak ~130 °C (HDPE). TGA: two-stage mass loss – main PE degradation ~476 °C, and a second minor step ~700 °C. TG–MS detects C1–C3 hydrocarbons and a notable CO₂ release in the >650 °C range, confirming CaCO₃ filler decomposition. Residual ash ~6.6%. | CaCO₃ filler evidenced by 6–7% ash and CO₂ evolution; mild oxidative degradation (C=C, O–H in FTIR). These indicate inorganic additives and weathering contaminants. | Post-Consumer |
| P3 | Poly(propylene) (PP) | FTIR: CH₂ and CH₃ stretching and bending vibrations characteristic of isotactic PP. Raman confirms PP identity (matches PP reference spectrum). | DSC: melting peak ~160 °C (PP). TGA: single-stage degradation around 460 °C (DTG peak) with ~99% mass loss. TG–MS shows predominant C3 hydrocarbon fragments (propylene/propane m/z 41, 43) and other alkane/alkene volatiles, as expected for pure PP pyrolysis. | No additives detected – FTIR shows no carbonyl or other extraneous bands; minimal residue (~1% ash) indicating virtually no filler. | Post-Industrial |
| P4 | Poly(propylene) (PP) | FTIR: CH₃ bending and CH₂ stretching bands consistent with PP. Raman signal was weak (fluorescence) but no contradictions to PP assignment. | DSC: melting peak ~160 °C (PP). TGA: single-stage decomposition ~458–460 °C, nearly complete mass loss (~99%). TG–MS profile shows only PP-derived volatiles (C3 hydrocarbons), with no secondary decomposition stage. | No significant additives – no signs of oxidation in FTIR; negligible inorganic residue (~0.9% ash). | Post-Industrial |
| P5 | Poly(ethylene) – high-density (HDPE) | FTIR: CH₂ bands of PE with broad C=O absorption (~1710 cm⁻¹) indicating oxidized polyethylene. Raman confirms PE polymer backbone (matches PE standard). | DSC: melting point ~125–130 °C (HDPE). TGA: multi-stage degradation – primary polymer breakdown ~477 °C (HDPE) and a second weight-loss event onset ~650–700 °C. TG–MS detects C1-C3 hydrocarbon volatiles and a strong CO₂ evolution peaking near 700 °C, corresponding to filler (CaCO₃) decomposition. ~10% residue remains. | Pronounced CaCO₃ filler presence (10.2% ash) confirmed by CO₂ release; clear oxidation signs (carbonyl in FTIR) from weathering. Possibly other additives (e.g. stabilizers) indicated by multi-step TGA profile. | Post-Consumer |
| P6 | Polyolefin blend – LDPE, HDPE & PP | FTIR: overlapping CH₂ (PE) and CH₃ (PP) bands; weak C=O stretch suggesting slight oxidation. Raman spectroscopy inconclusive (fluorescence interference), but overall spectra imply mixed PE/PP. | DSC: multiple melting points ~110 °C (LDPE), 130 °C (HDPE), 165 °C (PP) – confirming a PE/PP blend. TGA: broad multi-step degradation with an initial peak ~460 °C (PP) and shoulder ~470–480 °C (PE); nearly complete mass loss (~98%). TG–MS shows a composite volatile profile (mixed C2–C3 fragments from both PE and PP), no distinct second high-temperature gas release (low ash ~2%). | Minor oxidation (trace carbonyl in FTIR) but no substantial inorganic filler (residual <2%). The combined thermal profile reflects the PP/PE mixture without significant additives. | Post-Consumer |
| P7 | Poly(ethylene) – low & high density (LDPE & HDPE) | FTIR: CH₂ rocking/stretching of PE with an extra C=C stretching band, implying unsaturation (oxidative degradation or additive). (Raman not measured for this sample.) | DSC: dual melting peaks ~110 °C (LDPE) and 130 °C (HDPE). TGA: two-step decomposition – main polymer volatilization ~475 °C, plus a minor second loss up to ~750 °C. TG–MS (by analogy to similar samples) would show PE pyrolysis hydrocarbons and a small CO₂ release at high T (consistent with slight CaCO₃ content). Residue ~3%. | Inorganic filler (CaCO₃) is indicated by ~2.8% ash and a minor 700 °C decomposition event; FTIR C=C and slight O–H signals denote polymer oxidation. Overall additives are low-level (e.g. film filler, antioxidants byproducts). | Post-Consumer / Post-Commercial |
| P8 | Poly(ethylene) – high-density (HDPE) | FTIR: CH₂ vibrations of PE and faint C=C band (possible branching or oxidative unsaturation). (Raman not measured.) | DSC: melting peak ~125 °C (HDPE). TGA: nearly single-step degradation ~470 °C with a very small secondary weight loss around 700 °C; ~4–5% residue. TG–MS (inferred) predominantly shows C2–C3 hydrocarbon gases with a minor CO₂ signal from filler. | Moderate CaCO₃ filler present (~4.7% ash) evidenced by the slight high-T mass loss and expected CO₂ release; mild oxidation (unsaturation in FTIR) suggesting some aging or additive breakdown. | Post-Consumer / Post-Commercial |
| P9 | Polyolefin mix – HDPE & PP | FTIR: combined CH₂ (PE) and CH₃ (PP) bands confirm a PE/PP mixture; C=C stretching indicates unsaturation or additive. (Raman not measured.) | DSC: two melting transitions (~130 °C and 165 °C) corresponding to HDPE and PP. TGA: multi-stage decomposition – overlapping PE/PP degradation around 460–480 °C, plus a subtle secondary drop at higher temperature. ~3% residue remains. TG–MS was not run, but the sample is expected to release typical PE/PP pyrolysis gases (no large CO₂ peak, consistent with low filler). | Minor inorganic content (~3.4% ash, possibly pigment or trace filler); slight polymer oxidation (C=C in FTIR). No significant additives apart from the small contamination by the secondary polymer (mixed plastic stream). | Post-Commercial |
| Element (ppm) | Al | As | Ba | Ca | Cd | Fe | K | Mg | Na | P |
|---|---|---|---|---|---|---|---|---|---|---|
| P1 | 54.05 | ND | ND | 27.43 | ND | ND | ND | ND | 9.35 | 20.99 |
| P2 | 1155.80 | ND | ND | 26900.00 | ND | 28.28 | 4993.74 | 2718.60 | 9698.56 | 121.36 |
| P3 | 76.17 | ND | ND | 5362.95 | ND | 14.67 | ND | 25.57 | 44.19 | 38.34 |
| P4 | 37.81 | ND | ND | 881.50 | ND | 25.15 | ND | 31.06 | 85.88 | 40.51 |
| P5 | 213.93 | ND | ND | 15362.08 | ND | 14.52 | ND | 57.62 | 19.93 | 90.88 |
| P6 | 185.45 | ND | ND | 152.80 | ND | ND | ND | 20.44 | 16.51 | 26.76 |
| P7 | 216.80 | ND | 22.51 | 15401.03 | ND | 74.28 | 21.15 | 404.86 | 106.25 | 34.69 |
| P8 | 68.92 | ND | ND | 35608.29 | ND | 18.53 | ND | 75.49 | 24.40 | 70.97 |
| P9 | 216.86 | ND | 50.58 | 20896.63 | ND | 335.78 | 15.58 | 445.74 | 86.72 | 45.71 |
| Element (ppm) | Pb | S | Sb | Si | Sn | Sr | Te | Ti | Zn | Sum. /% |
| P1 | ND | ND | ND | 222.47 | ND | ND | ND | ND | ND | 0.03 |
| P2 | ND | 332.83 | ND | 827.45 | ND | ND | ND | 451.88 | 53.68 | 4.73 |
| P3 | ND | 91.17 | ND | 43.24 | ND | ND | ND | 208.8 | 35.72 | 0.59 |
| P4 | ND | 16.73 | ND | 74.01 | ND | ND | ND | 33.15 | 6.8 | 0.12 |
| P5 | ND | 279.02 | ND | 117.46 | ND | ND | ND | 277.05 | 30.65 | 1.65 |
| P6 | ND | 12.5 | ND | 94.02 | ND | ND | ND | 232.33 | 24.7 | 0.08 |
| P7 | 20.56 | 268.74 | ND | 733.47 | ND | 99.78 | ND | 1664.25 | 153.23 | 1.92 |
| P8 | ND | 451.26 | ND | 127.88 | ND | 12.18 | ND | 1124.93 | 52.51 | 3.76 |
| P9 | 9.98 | 291.14 | ND | 367.09 | ND | 10.88 | ND | 1828.17 | 28.91 | 2.46 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




