Submitted:
17 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Plastic Samples and Pyrolysis Procedure
2.2. GC×GC–TOF–MS Analysis of Pyrolysis Oils
2.3. VOC and SVOC Analysis (Headspace and Leachate GC–MS)
2.4. Data Interpretation and Literature Comparison
3. Results and Discussion
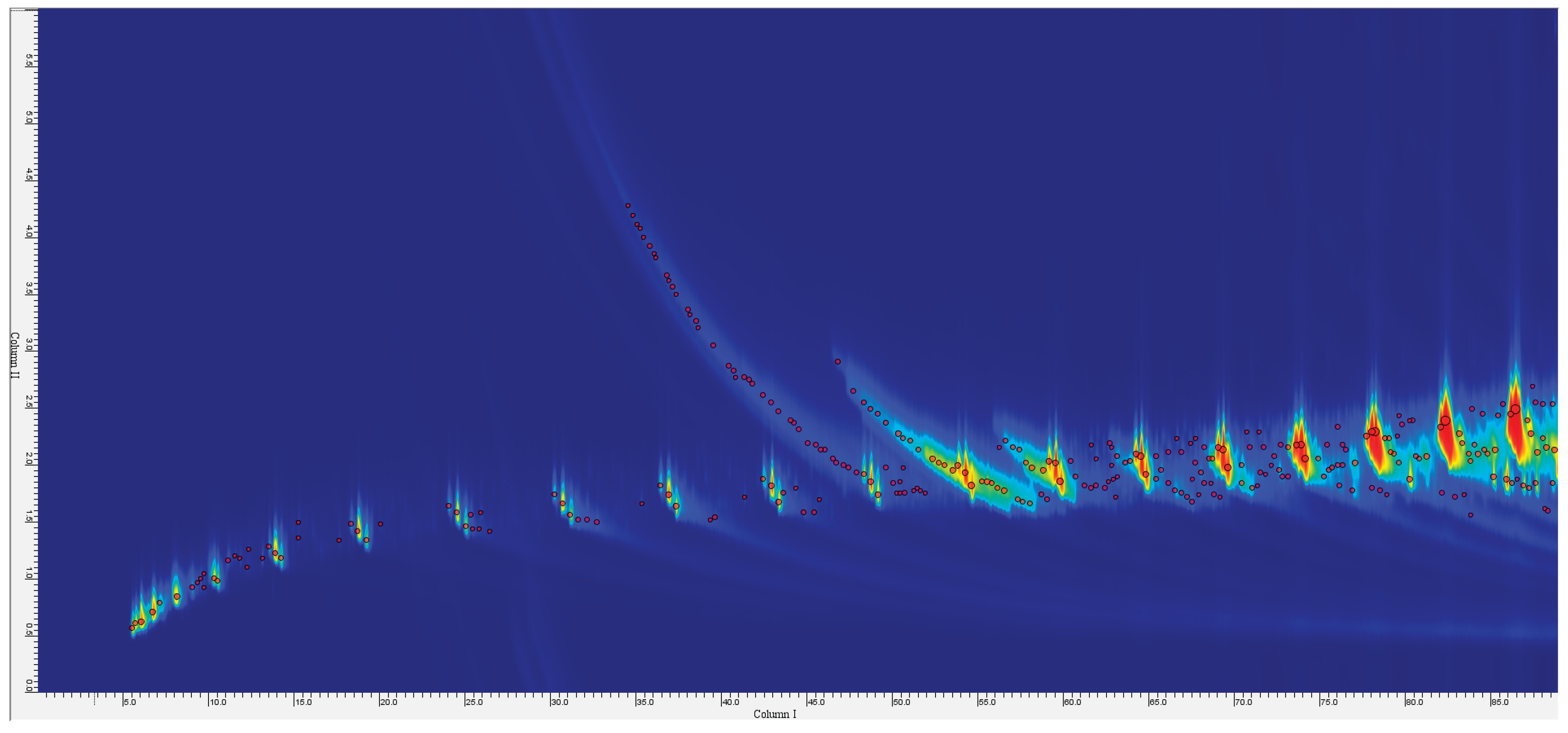
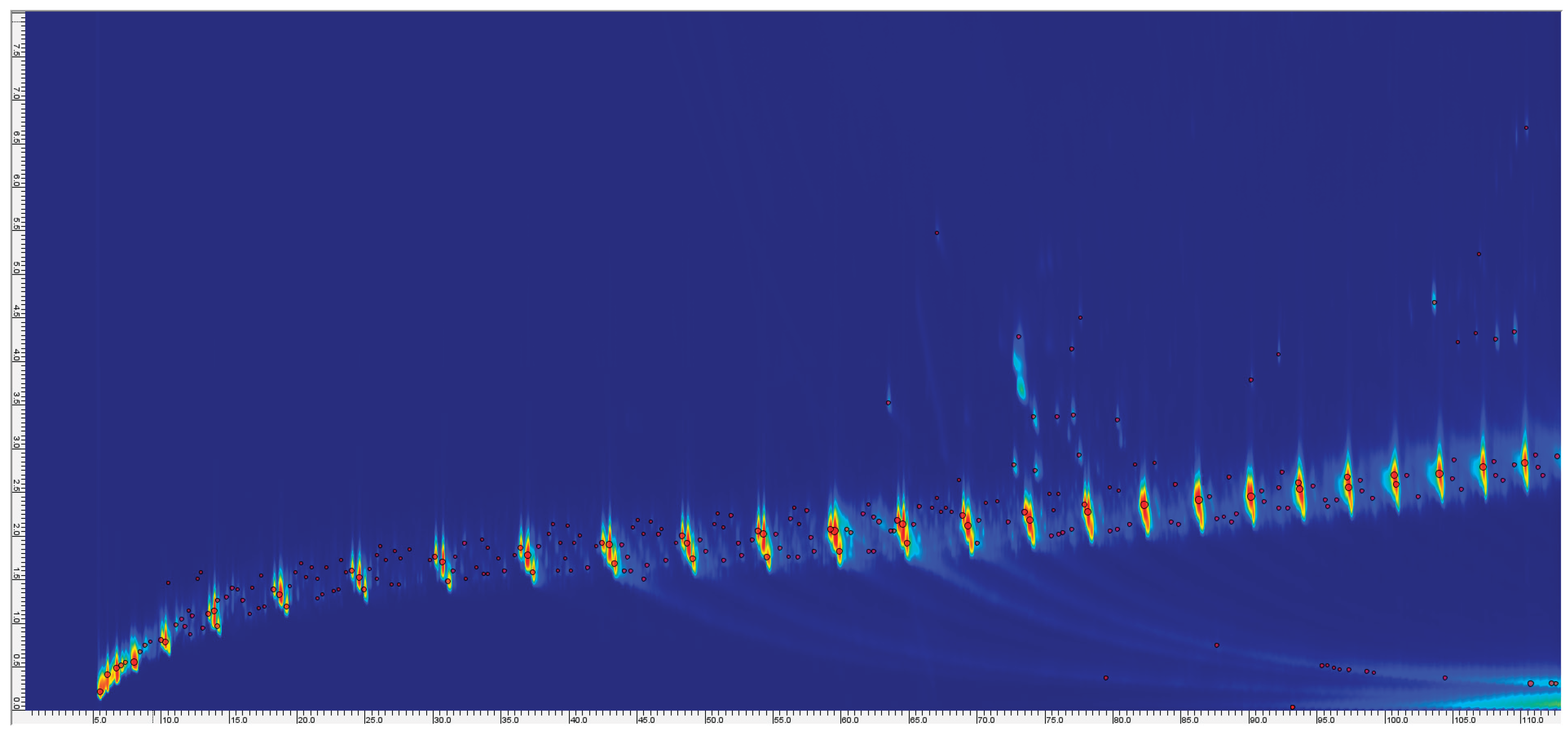
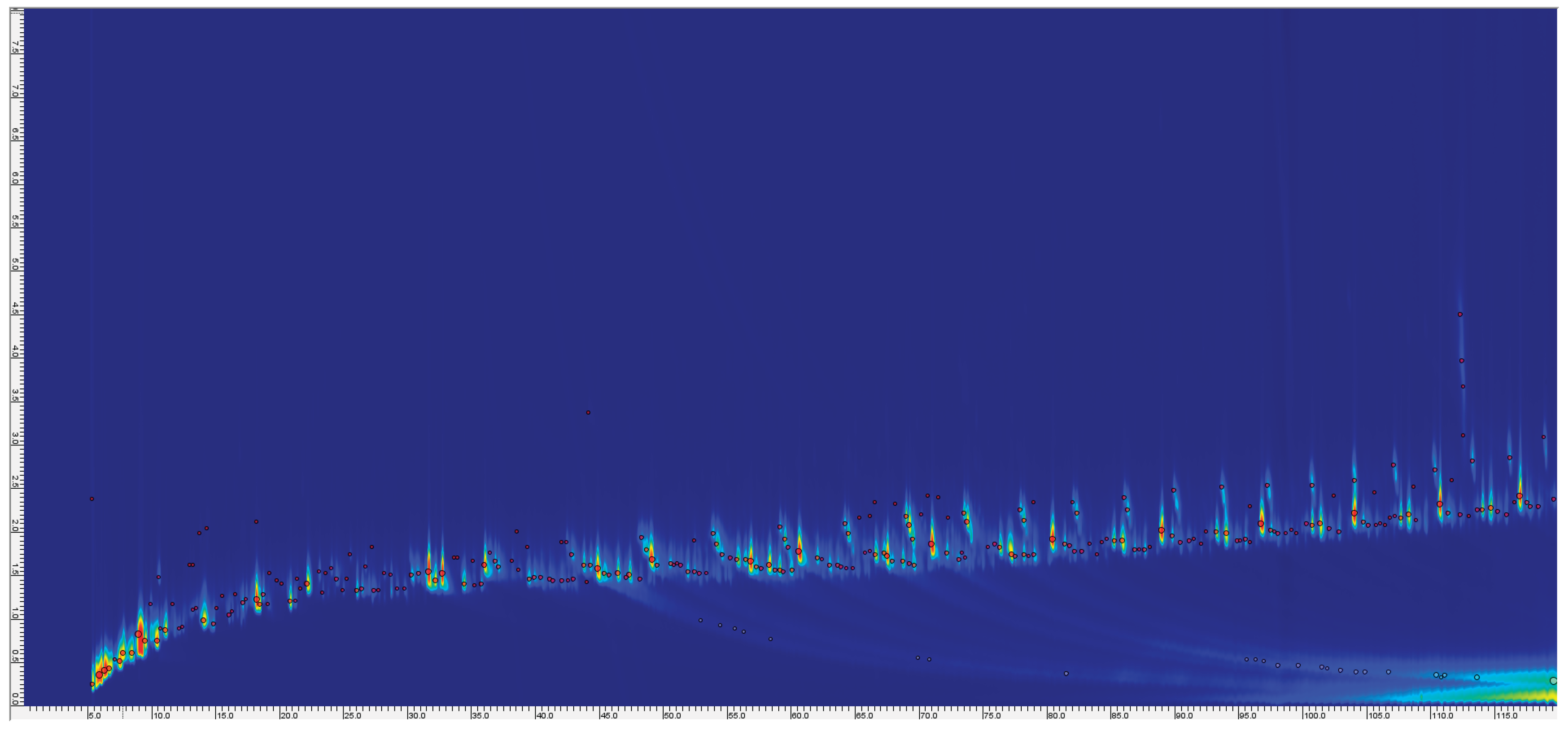
3.1. GC×GC–TOF–MS Analysis of Pyrolysis Oils (Samples P1–P6)
3.1.1. Effect of Pyrolysis Temperature on Pyrolysis Oil Composition (450 °C vs 650 °C)
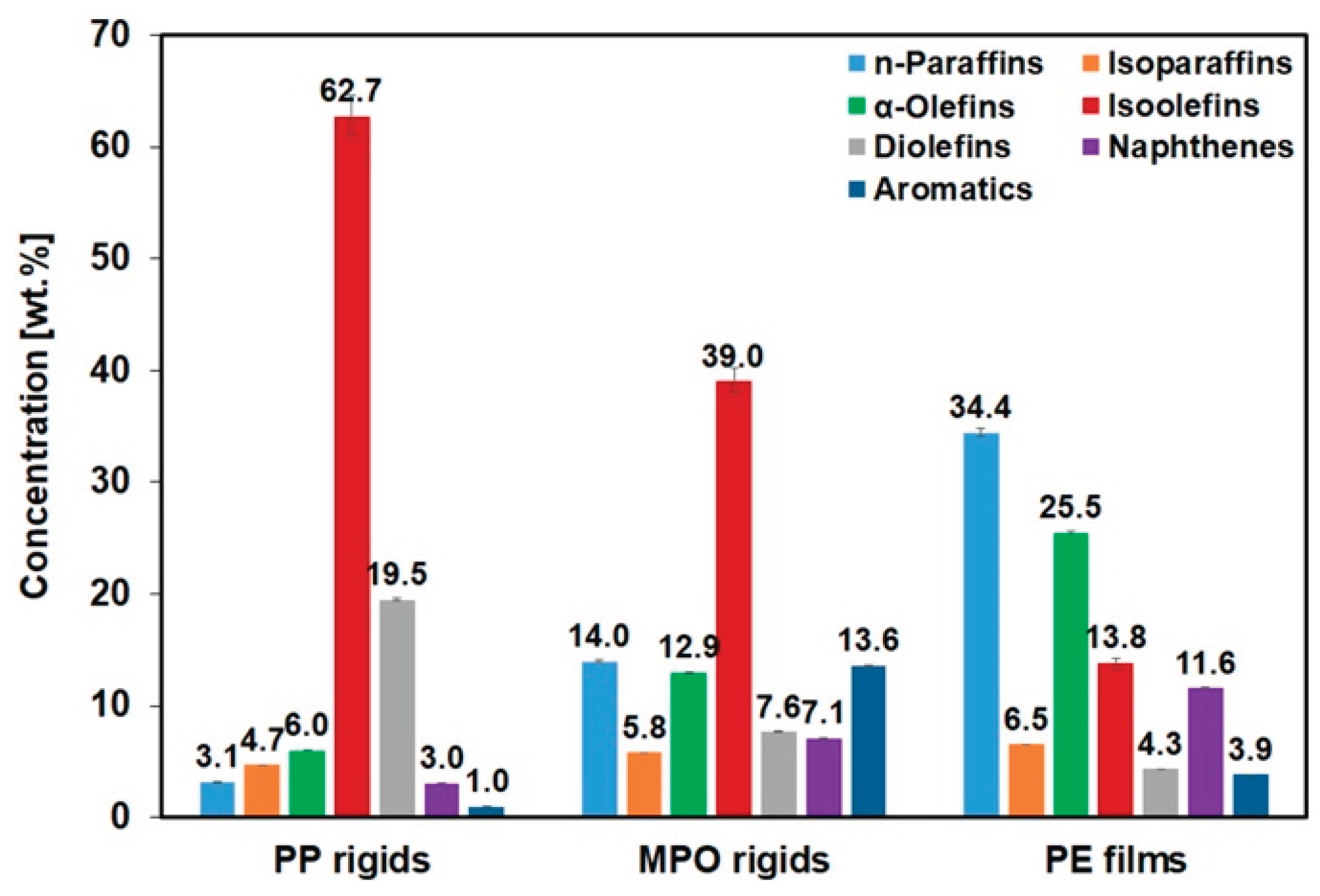
3.1.2. Overview of Pyrolysis Oil Composition at 650 °C
3.1.3. Repeat and Comparative Analysis
3.1.4. Broader Differences Between Samples (Effect of Feedstock):
3.1.5. Additive-Derived Compounds and NIAS in Oils
3.2. VOC and Volatile, Semi-Volatile, and Migrating Organic Compounds
3.2.1. Volatile and Semi-Volatile Organic Compounds (VOCs and SVOCs)
3.2.2. Migrants in Aqueous Food Simulant (Water Migrants)
4. Conclusions
Supplementary Materials
Acknowledgments
Data Availability
Declaration of Competing Interest
Nomenclature
| FT-IR | Fourier Transform Infrared Spectroscopy |
| DSC | Differential Scanning Calorimetry |
| TGA | Thermal Gravimetric Analysis |
| CCD | Charge-Coupled Device |
| NIAS | Non-Intentionally Added Substances |
| LDPE | Low-Density Polyethylene |
| HDPE | High-Density Polyethylene |
| PP | Polypropylene |
| PVC | Polyvinyl Chloride |
| UV | Ultraviolet |
| PBDEs | Polybrominated Diphenyl Ethers |
| PFAS | Perfluoroalkyl Substances |
| POPs | Persistent Organic Pollutants |
| SCCPs | Short-Chain Chlorinated Paraffins |
| PCBs | Polychlorinated Biphenyls |
| REACH | Registration, Evaluation, Authorization, and Restriction of Chemicals |
| UNEP | United Nations Environment Programme |
| PBDE | Polybrominated Diphenyl Ethers |
| POP | Persistent Organic Pollutants |
| SCCP | Short-Chain Chlorinated Paraffins |
| PCB | Polychlorinated Biphenyls |
| Py-GCxGC TOF MS | Pyrolysis coupled with Comprehensive Two-Dimensional Gas Chromatography Time-of-Flight Mass Spectrometry |
| GC | Gas Chromatography |
| TOF-MS | Time-of-Flight Mass Spectrometry |
| GC×GC | Comprehensive Two-Dimensional Gas Chromatography |
| CDS | Chemical Delivery Systems (specific to the Pyroprobe 6150 model) |
| HP-5MS UI | High-Performance 5% Phenyl Methylpolysiloxane Ultra Inert |
| BPX-50 | Biphenyl Polysilphenylenesiloxane (mid-polar column) |
| EI+ | Electron Ionization (Positive Mode) |
| NIST | National Institute of Standards and Technology |
| EPA | Environmental Protection Agency |
| NIH | National Institutes of Health |
| m/z | Mass-to-Charge Ratio |
| Hz | Hertz |
| GCImageTM | Gas Chromatography Image (software) |
| eV | Electron Volt |
References
- Llorenç Milà i Canals, A.C., Peggy Lefort, Allan Meso, Andrew David Raine, Aphrodite Smagadi, Steven Stone, Elisa Tonda, Yoni Shiran, José de la Fuente, Julia Koskella, Anne Titia Bove, Eline Boon, Steve Fletcher, Antaya March, Keiron Roberts, Andrea Bassi, Sheila Aggarwal-Khan, Andrea Hinwood, Vincent Aloysius, Sandra Averous, Stephanie Gerretsen, Claudia Giacovelli, Toma Iida, Pushpam Kumar, Stéphanie Laruelle, Silvana Loayza León, Ekaterina Poleshchuk, Heidi Savelli, Himanshu Sharma, Ran Xie, Xiaozhen Xu, Natalie Harms, Kei Ohno Woodall, Kabir Arora, Nao Takeuchi, Anjali Acharya, Milagros Aimé, Turning off the Tap. How the world can end plastic pollution and create a circular economy. 2023, United Nations Environment Programme (2023): Nairobi.
- Saxena, S., Pyrolysis and Beyond: Sustainable Valorization of Plastic Waste. Applications in Energy and Combustion Science, 2024: p. 100311. [CrossRef]
- Almroth, B.C., Scientists found hundreds of toxic chemicals in recycled plastics., in ScienceDaily. 2023, ScienceDaily: 2023.
- Almroth, B.C., et al., Addressing the toxic chemicals problem in plastics recycling. Cambridge Prisms: Plastics, 2025. 3: p. e3.
- Geueke, B., K. Groh, and J. Muncke, Food packaging in the circular economy: Overview of chemical safety aspects for commonly used materials. Journal of cleaner production, 2018. 193: p. 491-505. [CrossRef]
- Geueke, B., et al., Hazardous chemicals in recycled and reusable plastic food packaging. Cambridge Prisms: Plastics, 2023. 1: p. e7.
- Groh, K.J., et al., Overview of known plastic packaging-associated chemicals and their hazards. Science of the total environment, 2019. 651: p. 3253-3268. [CrossRef]
- Groh, K.J., et al., Overview of intentionally used food contact chemicals and their hazards. Environment International, 2021. 150: p. 106225. [CrossRef]
- Hahladakis, J.N., et al., An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. Journal of hazardous materials, 2018. 344: p. 179-199. [CrossRef]
- Jones, K.C., Persistent organic pollutants (POPs) and related chemicals in the global environment: some personal reflections. Environmental Science & Technology, 2021. 55(14): p. 9400-9412. [CrossRef]
- Roland Weber, N.M.A., Nicolò Aurisano, Zhanyun Wang, Magali Outters, Kimberley De Miguel, Martin Schlummer, Markus Blepp, Helene Wiesinger, Helena Andrade, Martin Scheringer, Peter Fantke, Chemicals in Plastics - A Technical Report. 2023, United Nations Environment Programme and Secretariat of the Basel, Rotterdam and Stockholm Conventions (2023): Geneva.
- Spring, M., et al., Effective progress and implementation of the INC-5 plastics treaty through scientific guidance. Nature Sustainability, 2025: p. 1-3. [CrossRef]
- Aanesen, M., et al., Insights from international environmental legislation and protocols for the global plastic treaty. Scientific Reports, 2024. 14(1): p. 2750. [CrossRef]
- Farrelly, T., et al., Independent science key to breaking stalemates in global plastics treaty negotiations. Cambridge Prisms: Plastics, 2025. 3: p. e6. [CrossRef]
- Farrelly, T., et al., Global plastics treaty needs trusted science. Science, 2024. 384(6693): p. 281-281. [CrossRef]
- Vince, J., et al., The Zero Draft Plastics Treaty: Gaps and challenges. Cambridge Prisms: Plastics, 2024. 2: p. e24.
- Aiping Chen, S.S., Vasilios G. Samaras, and Bassam Dally, Advanced Analytical Framework for Feedstock Characterization and Hazardous Contaminant Profiling in Mixed Plastic Waste: Implications for Recycling Strategy K.A.U.o.S.a. Technology, Editor. 2025.
- Akoueson, F., et al., Identification and quantification of plastic additives using pyrolysis-GC/MS: A review. Science of the Total Environment, 2021. 773: p. 145073. [CrossRef]
- Barrick, A., et al., Plastic additives: Challenges in ecotox hazard assessment. PeerJ, 2021. 9: p. e11300. [CrossRef]
- Bill, A., et al., Characterizing plastics from large household appliances: Brominated flame retardants, other additives and density profiles. Resources, Conservation and Recycling, 2022. 177: p. 105956. [CrossRef]
- Cuthbertson, A.A., et al., Characterization of polymer properties and identification of additives in commercially available research plastics. Green Chemistry, 2024. 26(12): p. 7067-7090. [CrossRef]
- Radhakrishnan, H., et al., Influence of Functional Additives, Fillers, and Pigments on Thermal and Catalytic Pyrolysis of Polyethylene for Waste Plastics Upcycling. Green Chemistry, 2025. [CrossRef]
- Staub, C. Study: E-plastics additives present in home goods. Plastics Recycling update, 2004.
- Turner, A. and M. Filella, Hazardous metal additives in plastics and their environmental impacts. Environment International, 2021. 156: p. 106622. [CrossRef]
- Wiesinger, H., Z. Wang, and S. Hellweg, Deep dive into plastic monomers, additives, and processing aids. Environmental science & technology, 2021. 55(13): p. 9339-9351.
- Horodytska, O., A. Cabanes, and A. Fullana, Non-intentionally added substances (NIAS) in recycled plastics. Chemosphere, 2020. 251: p. 126373. [CrossRef]
- Kato, L.S. and C.A. Conte-Junior, Safety of plastic food packaging: the challenges about non-intentionally added substances (NIAS) discovery, identification and risk assessment. Polymers, 2021. 13(13): p. 2077. [CrossRef]
- Geueke, B., et al., Systematic evidence on migrating and extractable food contact chemicals: most chemicals detected in food contact materials are not listed for use. CritiCal reviews in Food sCienCe and nutrition, 2023. 63(28): p. 9425-9435. [CrossRef]
- He, Y.-J., et al., Migration of (non-) intentionally added substances and microplastics from microwavable plastic food containers. Journal of hazardous materials, 2021. 417: p. 126074. [CrossRef]
- Kim, H.S., et al., Comparison of migration and cumulative risk assessment of antioxidants, antioxidant degradation products, and other non-intentionally added substances from plastic food contact materials. Food Packaging and Shelf Life, 2023. 35: p. 101037. [CrossRef]
- Ong, H.-T., H. Samsudin, and H. Soto-Valdez, Migration of endocrine-disrupting chemicals into food from plastic packaging materials: an overview of chemical risk assessment, techniques to monitor migration, and international regulations. Critical reviews in food science and nutrition, 2022. 62(4): p. 957-979. [CrossRef]
- Phelps, D.W., et al., Per-and polyfluoroalkyl substances in food packaging: migration, toxicity, and management strategies. Environmental Science & Technology, 2024. 58(13): p. 5670-5684.
- Ureel, Y., et al. Detailed Characterization of Plastic Pyrolysis Oils and Their Contaminants by FT-ICR MS and GCxGC. in 2024 AIChE Spring Meeting & 20th Global Congress on Process Safety (AIChE Spring). 2024.
- Beccaria, M., et al., Analysis of mixed plastic pyrolysis oil by comprehensive two-dimensional gas chromatography coupled with low-and high-resolution time-of-flight mass spectrometry with the support of soft ionization. Talanta, 2023. 252: p. 123799. [CrossRef]
- Núñez, S.S., et al., Heavy metals, PAHs and POPs in recycled polyethylene samples of agricultural, post-commercial, post-industrial and post-consumer origin. Waste Management, 2022. 144: p. 113-121. [CrossRef]
- Gao, P., et al., Pyrolysis of municipal plastic waste: Chlorine distribution and formation of organic chlorinated compounds. Science of The Total Environment, 2024. 912: p. 169572. [CrossRef]
- Nevondo, V., et al., Analytical insights into short-chain chlorinated paraffins in consumer products, leachates, and sediments in Gauteng, South Africa. International Journal of Environmental Science and Technology, 2025: p. 1-18. [CrossRef]
- Oberoi, S. and M. Malik, Polyvinyl chloride (PVC), chlorinated polyethylene (CPE), chlorinated polyvinyl chloride (CPVC), chlorosulfonated polyethylene (CSPE), polychloroprene rubber (CR)—Chemistry, applications and ecological impacts—I. Ecological and Health Effects of Building Materials, 2022: p. 33-52.
- Hochegger, A., et al., One-dimensional and comprehensive two-dimensional gas chromatographic approaches for the characterization of post-consumer recycled plastic materials. Analytical and Bioanalytical Chemistry, 2023. 415(13): p. 2447-2457. [CrossRef]
- Dao Thi, H., M.R. Djokic, and K.M. Van Geem, Detailed group-type characterization of plastic-waste pyrolysis oils: by comprehensive two-dimensional gas chromatography including linear, branched, and di-olefins. Separations, 2021. 8(7): p. 103. [CrossRef]
- Kusenberg, M., et al., Assessing the feasibility of chemical recycling via steam cracking of untreated plastic waste pyrolysis oils: Feedstock impurities, product yields and coke formation. Waste management, 2022. 141: p. 104-114. [CrossRef]
- Kusenberg, M., et al., A comprehensive experimental investigation of plastic waste pyrolysis oil quality and its dependence on the plastic waste composition. Fuel Processing Technology, 2022. 227: p. 107090. [CrossRef]
- Rung, C., et al., Identification and Evaluation of (Non-) Intentionally Added Substances in Post-Consumer Recyclates and Their Toxicological Classification. Recycling, 2023. 8(1): p. 24. [CrossRef]
- NIST, NIST 23 Tandem Mass Spectral Libraries, N.I.o.S.a.T. (NIST), Editor.
- Chen, Z.-F., et al., Identification of recycled polyethylene and virgin polyethylene based on untargeted migrants. Food Packaging and Shelf Life, 2021. 30: p. 100762. [CrossRef]
- Germinario, G., I.D. van der Werf, and L. Sabbatini, Pyrolysis gas chromatography mass spectrometry of two green phthalocyanine pigments and their identification in paint systems. Journal of Analytical and Applied Pyrolysis, 2015. 115: p. 175-183. [CrossRef]
- Strien, J.R., et al., Pyrolysis of Polyolefin-Enriched Mixed Plastic Waste Streams: Effects of Pretreatments and Presence of Hydrogen during Pyrolysis. Energy & Fuels, 2024. 39(1): p. 686-698. [CrossRef]
- Toraman, H.E., et al., Detailed compositional characterization of plastic waste pyrolysis oil by comprehensive two-dimensional gas-chromatography coupled to multiple detectors. Journal of Chromatography A, 2014. 1359: p. 237-246. [CrossRef]
- Jung, S.-H., et al., Pyrolysis of a fraction of waste polypropylene and polyethylene for the recovery of BTX aromatics using a fluidized bed reactor. Fuel processing technology, 2010. 91(3): p. 277-284. [CrossRef]
- Saha, B., et al., Review on production of liquid fuel from plastic wastes through thermal and catalytic degradation. Journal of the Energy Institute, 2024: p. 101661. [CrossRef]
- Hoang, N.M. and K. Park, Applications of Tert-Butyl-Phenolic Antioxidants in Consumer Products and Their Potential Toxicities in Humans. Toxics, 2024. 12(12): p. 869. [CrossRef]
- Khan, R., B.A. Perez, and H.E. Toraman, Comparative analysis of additive decomposition using one-dimensional and two–dimensional gas chromatography: Part II-Irgafos 168 and zinc stearate. Journal of Chromatography A, 2024. 1732: p. 465244. [CrossRef]
- Van Waeyenberg, J., et al., High molecular weight product formation in polyolefin chemical recycling: a comprehensive review on primary and secondary products. ACS Sustainable Chemistry & Engineering, 2024. 12(30): p. 11074-11092. [CrossRef]
- Xayachak, T., et al., Pyrolysis for plastic waste management: An engineering perspective. Journal of Environmental Chemical Engineering, 2022. 10(6): p. 108865. [CrossRef]
- Dulal, N., Study on the Migration of Slip Agents on the Surface of High-Density Polyethylene Beverage Closures. 2018, RMIT University.
- Chen, Y., et al., Recent advances in non-targeted screening of compounds in plastic-based/paper-based food contact materials. Foods, 2023. 12(22): p. 4135. [CrossRef]
- Maruyama, F., et al., Screening of phthalates in polymer materials by pyrolysis GC/MS. Analytical Sciences, 2015. 31(1): p. 3-5. [CrossRef]
- Zou, L., et al., Chemical recycling of polyolefins: a closed-loop cycle of waste to olefins. National Science Review, 2023. 10(9): p. nwad207. [CrossRef]
- Aiping Chen, S.S., Vasileios Samaras, Bassam Dally, Comprehensive Characterization of Post-Consumer, Post-Commercial, and Post-Industrial Plastics: Towards Tailored Recycling Solutions, T.K. King Abdullah University of Science & Technology, Editor. 2025.
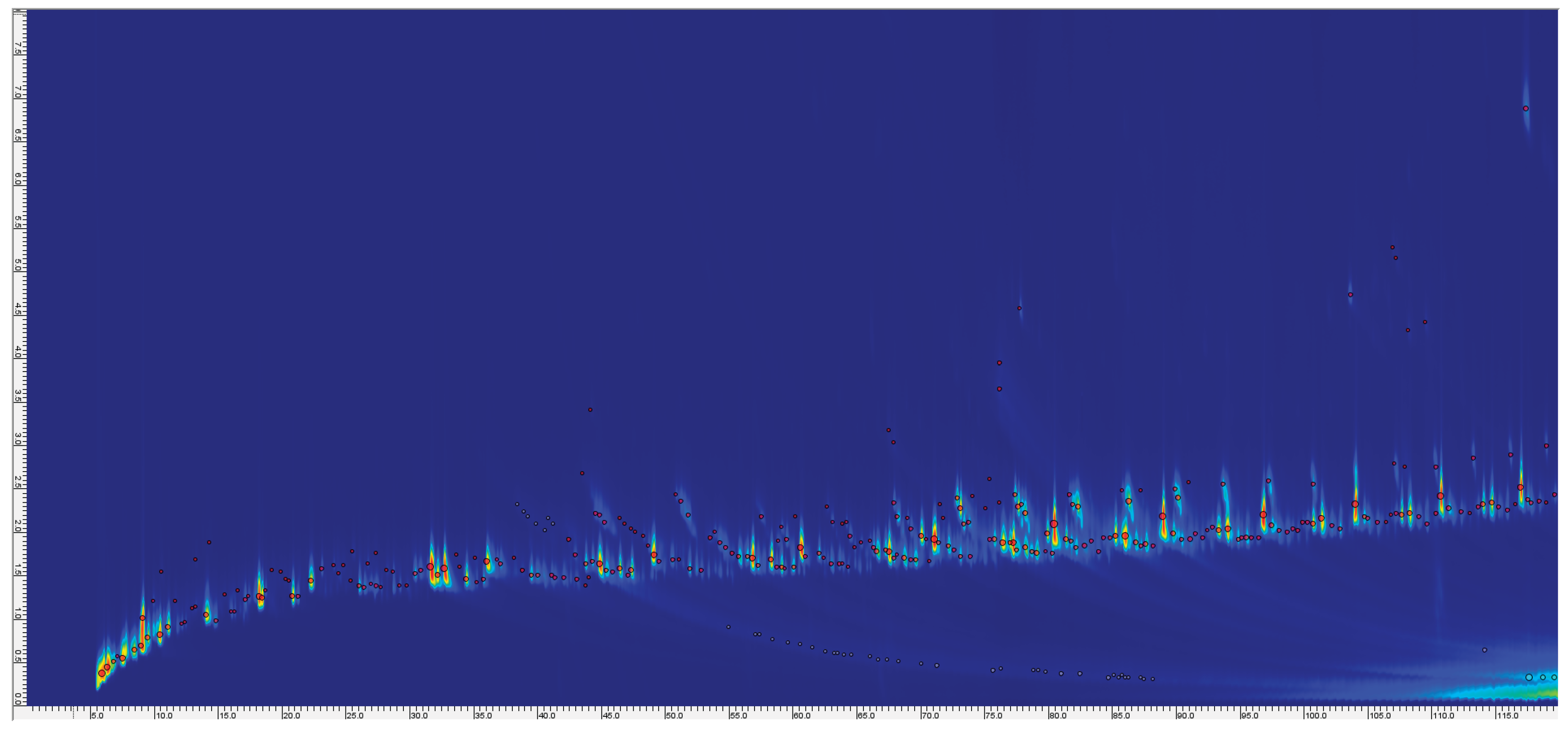
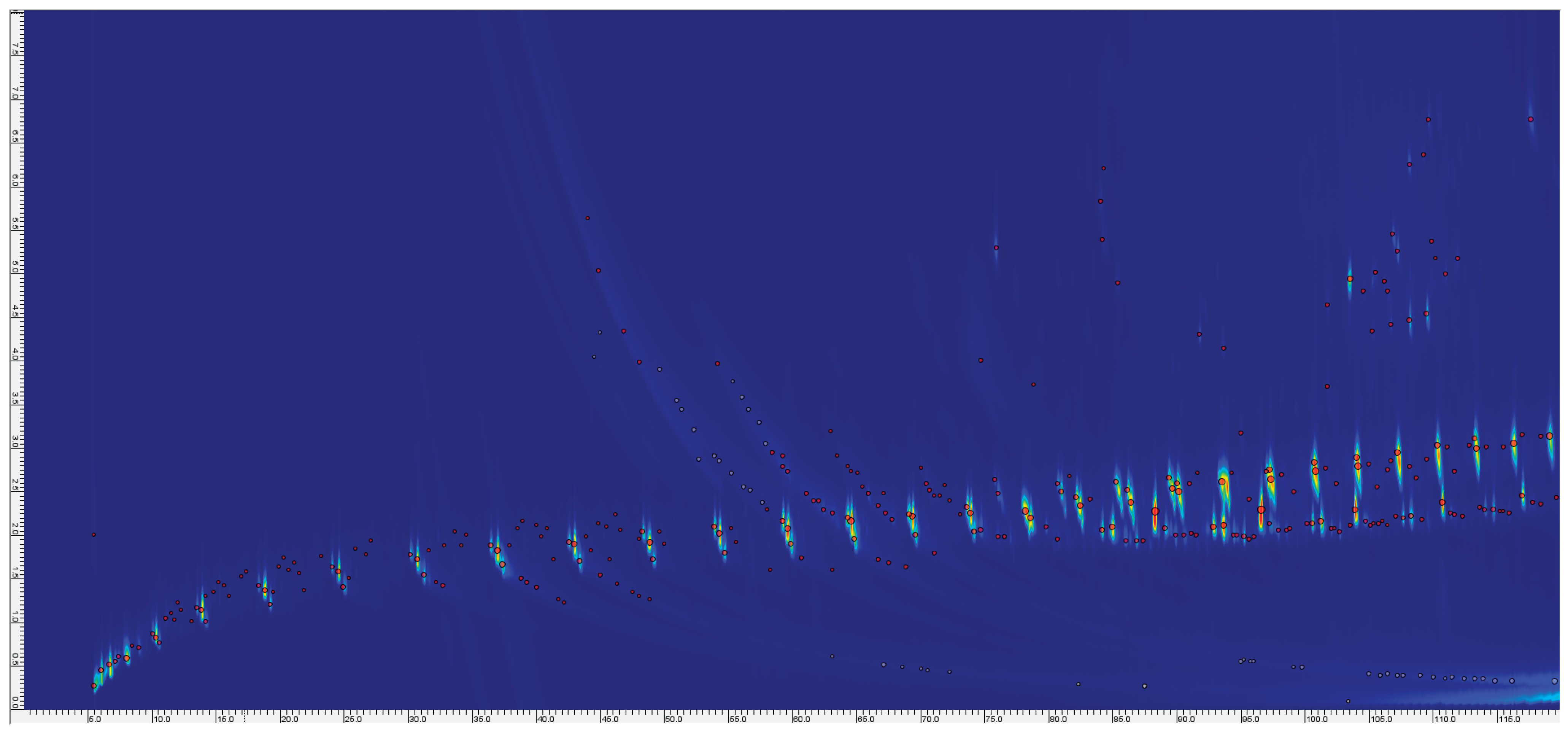
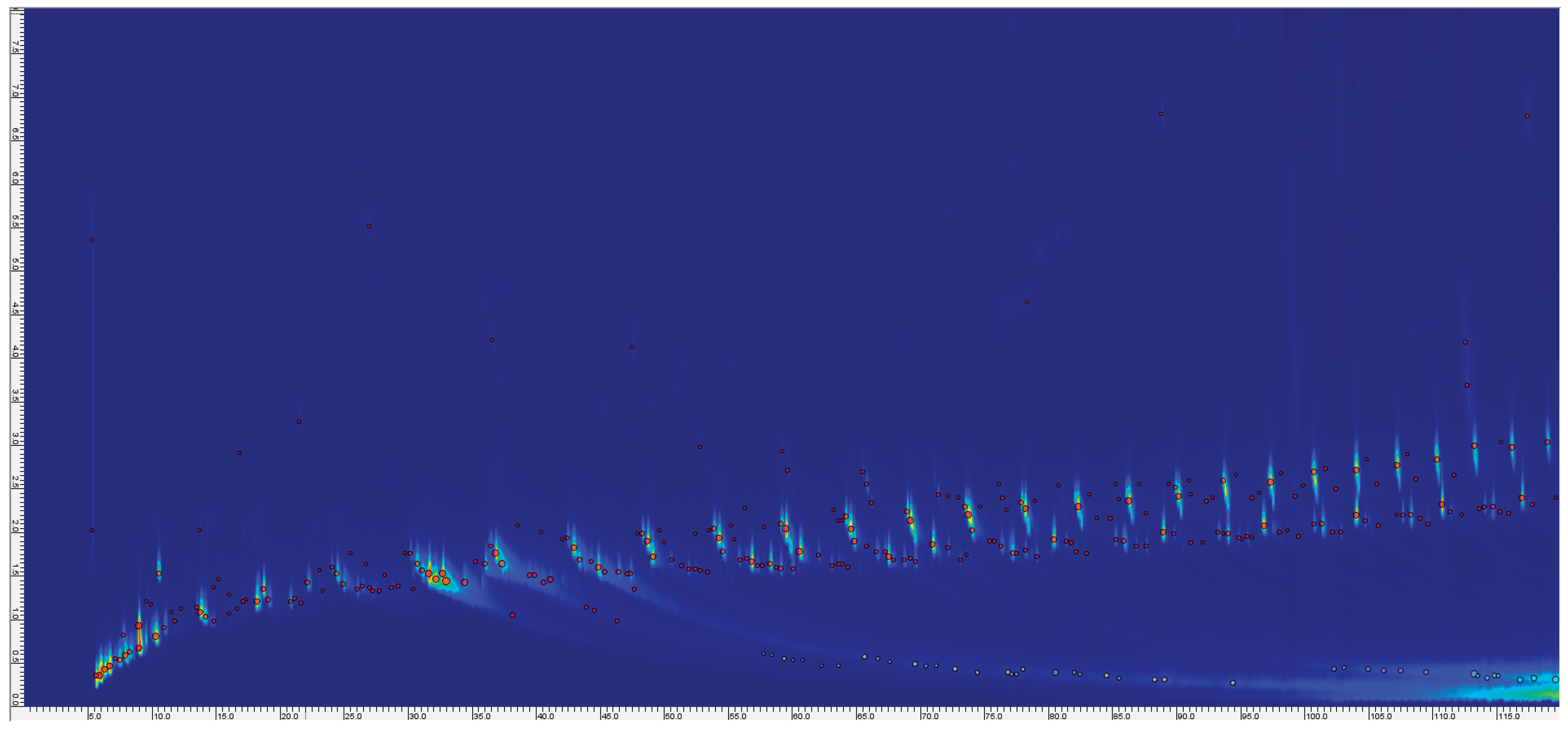
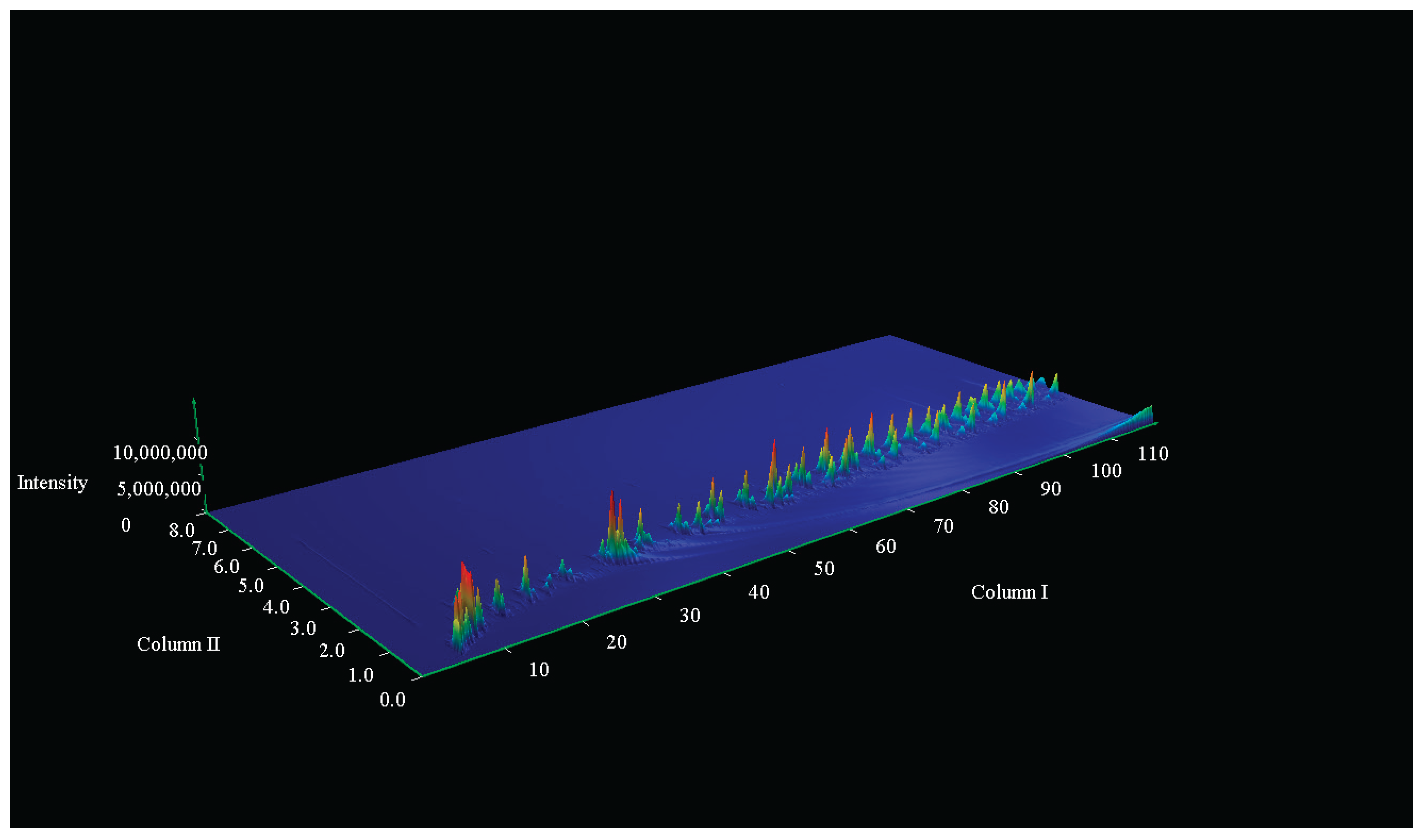
| Name | Mass (mg) Before pyrolysis | Mass (mg) After pyrolysis | Temperature (°C) |
|---|---|---|---|
| P1_450 | 5.517 | 5.544 | 450 |
| P2_450 | 4.582 | 4.611 | 450 |
| P3_450 | 4.27 | 4.36 | 450 |
| P4_450 | 4.029 | 4.027 | 450 |
| P5_450 | 3.992 | 4.032 | 450 |
| P6_450 | 4.464 | 4.495 | 450 |
| P1_650 | 2.611 | 1.452 | 650 |
| P2_650 | 2.085 | 0.274 | 650 |
| P3_650 | 2.577 | 0.477 | 650 |
| P4_650 | 2.409 | 0.395 | 650 |
| P5_650 | 2.083 | 0.596 | 650 |
| P6_650 | 2.121 | 0.389 | 650 |
| Compound | P1 | P2 | P3 | P4 | P5 | P6 | |
|---|---|---|---|---|---|---|---|
| Alcohols | |||||||
| 1-Decanol, 2-hexyl- | 35.63 | 13.22 | 2.5 | 4.56 | 17.14 | ||
| 1-Eicosanol | 5.61 | 2.1 | 13.42 | 7.92 | |||
| n-Nonadecanol-1 | 3.16 | 3.37 | |||||
| 2-Hexyl-1-octanol | 2.75 | 3.16 | 5.12 | ||||
| 2-Ethyl-1-dodecanol | 2.24 | ||||||
| 11-Dodecen-1-ol, 2,4,6-trimethyl-, | 11-Dodecen-1-ol, 2,4,6-trimethyl-, | 2.36 | 2.85 | ||||
| 11-Hexadecen-1-ol, (Z)- | 2.36 | ||||||
| 1-Octanol, 2-butyl- | 2.16 | 2.37 | |||||
| 2-Isopropyl-5-methyl-1-heptanol | 2-Isopropyl-5-methyl-1-heptanol | 6.08 | 7.9 | 3.76 | |||
| n-Heptadecanol-1 | 2.54 | ||||||
| Tricosan-2-ol | 12.12 | 13.97 | |||||
| Olefins | |||||||
| 1-Docosene | 9.04 | 5.45 | 4.76 | ||||
| 1,19-Eicosadiene | 4.27 | 9.31 | 9.44 | 2.99 | |||
| 1-Tetracosene | 2.95 | 3.13 | 2.19 | 11 | 2.23 | ||
| 1-Nonadecene | 2.85 | 3.77 | 8.77 | 2.47 | |||
| 1-Heptadecene | 4.21 | ||||||
| 1-Hexacosene | 8.77 | 3.25 | |||||
| 2,4-Dimethyl-1-heptene | 2.36 | ||||||
| Cetene | 2.89 | ||||||
| Nonacos-1-ene | 16.65 | 18.67 | 10.47 | ||||
| Pentacos-1-ene | 3.3 | ||||||
| Esters | |||||||
| Oxalic acid, allyl octadecyl ester | 2.4 | 11.92 | 3.11 | 9 | |||
| Carbonic acid, eicosyl vinyl ester | Carbonic acid, eicosyl vinyl ester | 9.15 | 9.03 | 8.1 | |||
| Oxalic acid, allyl hexadecyl ester | Oxalic acid, allyl hexadecyl ester | 4.95 | |||||
| Oxalic acid, allyl tridecyl ester | Oxalic acid, allyl tridecyl ester | 6.07 | |||||
| Alkenal | |||||||
| cis-4-Decenal | 2.89 | ||||||
| E-15-Heptadecenal | 3.34 | ||||||
| Cycloparaffins | |||||||
| Cyclotetradecane, 1,7,11-trimethyl-4-(1-methylethyl)- | Cyclotetradecane, 1,7,11-trimethyl-4-(1-methylethyl)- | Cyclotetradecane, 1,7,11-trimethyl-4-(1-methylethyl)- | 15.78 | 10.8 | |||
| Paraffins | |||||||
| Nonadecane | 3.05 | 2.78 | |||||
| Alkyne | |||||||
| 1-Octadecyne | 4.19 | 4.72 |
| Category | Concentration (area %) | Concentration (area %) | Concentration (area %) | Concentration (area %) | Concentration (area %) | Concentration (area %) |
|---|---|---|---|---|---|---|
| Category | P1 | P2 | P3 | P4 | P5 | P6 |
| Alcohol | 53.52 | 23.92 | 20.59 | 22.12 | 17.75 | 32.09 |
| α-olefines | 25.5 | 46.34 | 26.49 | 25.8 | 35.82 | 34.08 |
| Diolefins | 4.57 | 8 | 1.11 | 1.31 | 4.53 | 2.54 |
| Iso-olefins | 1.51 | 1.83 | 5.75 | 3.73 | 3.03 | 6.07 |
| Naphthenes | 0.27 | 0.94 | 21.56 | 27.29 | 20.34 | 8.8 |
| n-paraffins | 1.8 | 8.67 | 2.64 | |||
| Iso-paraffins | 0.19 | 0.21 | ||||
| Ester | 5.59 | 4.79 | 14.47 | 10.7 | 4.28 | 10.27 |
| Alkyne | 4.31 | 1.34 | 2.9 | 1.81 | 0.72 | 0.64 |
| Oxirane | 2.73 | 1.08 | 0.68 | 1.8 | 5.61 | 0.99 |
| Aromatics | 0.06 | 0.11 | 0.09 | 0.45 | 0.85 | |
| Si | 2.45 | 0.11 | ||||
| Cyclic alcohols | 5.7 | 3.03 | 0.47 | 1 | ||
| Ether | 0.34 | 0.99 | 2.25 | 2.36 |
| Compound name | Peak intensity | Peak intensity | Peak intensity | Classification | Classification | Classification | Classification |
|---|---|---|---|---|---|---|---|
| Compound name | P7 | P8 | P9 | Type | Type | Possible origin | Odorants |
| Hydrocarbons | |||||||
| 10-Heneicosene (c,t) | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| 1-Dodecene | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| 1-Nonadecene | 2 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| 1-Tetradecene | 1 | 3 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| 2,4-Dimethyl-1-heptene | 1 | 2 | 1 | C | Polymer degradation | Polymer degradation | Strong pungent plastic odor |
| 3-Dpdecene | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| 3-Eicosene, (E)- | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Cetene | 1 | 3 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Decane | 1 | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Decane, 4-methyl- | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Dodecane | 1 | 3 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Dodecane, 2,7,10-trimethyl- | Dodecane, 2,7,10-trimethyl- | 3 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Eicosane, 2-methyl- | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Heptacosane | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Heptadecane, 2,6,10,15-tetramethyl- | Heptadecane, 2,6,10,15-tetramethyl- | 1 | AD | Lubricant | Lubricant | ||
| Heptadecane, 4-methyl- | Heptadecane, 4-methyl- | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Heptane, 2,3-dimethyl- | Heptane, 2,3-dimethyl- | 1 | C | Processing aid | Processing aid | ||
| Heptane, 2,4-dimethyl- | Heptane, 2,4-dimethyl- | 3 | C | Processing aid | Processing aid | ||
| Heptane, 4-methyl- | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Hexadecane | 1 | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Hexadecane, 2,6,11,15-tetramethyl- | Hexadecane, 2,6,11,15-tetramethyl- | 1 | AD | Lubricant | Lubricant | ||
| Hexane, 2,3,5-trimethyl- | Hexane, 2,3,5-trimethyl- | 1 | C | Processing aid | Processing aid | ||
| Nonadecane | 2 | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Octadecane, 2-methyl- | 1 | 1 | 1 | PD | Processing aid | Processing aid | |
| Octane | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Octane, 4-methyl- | 2 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Tetradecane | 2 | 1 | 2 | PD | Polymer degradation | Polymer degradation | Polymer degradation |
| Tridecane | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| Undecane | 1 | 3 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Undecane, 4-methyl- | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | ||
| α-Methylstyrene | 3 | 2 | 2 | PD | Polymer degradation | Polymer degradation | Polymer degradation |
| Tetradecane, 4-methyl- | Tetradecane, 4-methyl- | 1 | PD | Polymer degradation | Polymer degradation | Polymer degradation | |
| Alcohols | |||||||
| 1-Decanol, 2-hexyl- | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
||
| 1-Butanol | 1 | C | Cosmetics | Cosmetics | |||
| 1-Octanol, 2,7-dimethyl- | 1-Octanol, 2,7-dimethyl- | 2 | C | Cosmetics / Industrial Solvent | Cosmetics / Industrial Solvent | Camphor-like odor | |
| 1-Octanol, 2-butyl- | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
||
| 2,2-dimethylphenylmethanol | 1 | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
|
| 2-Hexyl-1-octanol | 1 | C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
||
| 2-Undecanethiol, 2-methyl- | 2-Undecanethiol, 2-methyl- | 1 | C | Daily chemical products related | Daily chemical products related | Pungent and garlic-like odor | |
| Cyclobutanol | 1 | 1 | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
| Aldehydes and Ketones | Aldehydes and Ketones | ||||||
| 2-Pentenal, 2-methyl- | 1 | C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
||
| Decanal | 1 | PD or C | Cosmetics | Cosmetics | |||
| Heptanal | 1 | PD or C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
||
| Hexanal | 1 | PD or C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
||
| Nonanal | 1 | PD or C | Cosmetics | Cosmetics | |||
| Octanal | 1 | PD or C | Cosmetics | Cosmetics | |||
| Pentanal | 1 | 1 | PD or C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
|
| 2-Heptanone, 4,6-dimethyl- | 2-Heptanone, 4,6-dimethyl- | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
|
| 2-Heptanone, 4-methyl- | 2-Heptanone, 4-methyl- | 1 | C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
|
| Acetophenone | 1 | 1 | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
| Cyclopentanone, 2-(1-methylpropyl)- | Cyclopentanone, 2-(1-methylpropyl)- | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
|
| Others | |||||||
| 1-Propanol, 2-amino-, (±)- | 1 | C | Industrial solvent/Adhesive | Industrial solvent/Adhesive | Industrial solvent/Adhesive | ||
| 2,3-Epoxybutane | 1 | 1 | C | Pharmaceutical related | Pharmaceutical related | Pharmaceutical related | |
| 2,6-Di-tert-butylbenzoquinone | 2,6-Di-tert-butylbenzoquinone | 1 | AD | Oxidation phosphite additives |
Oxidation phosphite additives |
Oxidation phosphite additives |
|
| 2,6-di-tert-butyl-4-ethylphenol | 1 | AD | Anotioxidate | Anotioxidate | |||
| 2-Aminononadecane | 1 | 1 | 1 | C | Lubricant additive | Lubricant additive | Mild amine odor. |
| 2-Bromo dodecane | 1 | AD | Plasticizer intermediate | Plasticizer intermediate | Plasticizer intermediate | ||
| 2-Butanamine, 3-methyl- | 2-Butanamine, 3-methyl- | 1 | C | Pharmaceutical related | Pharmaceutical related | Strong fishy odor | |
| 2-Ethylacrolein | 1 | C | Processing aid | Processing aid | Pungent and acrid odor | ||
| 2-Octanamine | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
||
| 2-Pentanamine, 4-methyl- | 1 | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
|
| 3-(Prop-2-enoyloxy)dodecane | 1 | C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
||
| 3,3-Dimethyl-4-methylamino-butan-2-one | 1 | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
|
| 3,5-di-tert-Butyl-4-hydroxybenzaldehyde | 1 | AD | Irganox 1076 oligomer | Irganox 1076 oligomer | Irganox 1076 oligomer | ||
| Benzene, (1-methoxy-1-methylethyl)- | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
||
| Benzene, 1,3-bis(1,1-dimethylethyl)- | 1 | 1 | 1 | C | Processing aid | Processing aid | |
| Benzene, 1,3-bis(1-methylethenyl)- | 1 | C | Processing aid | Processing aid | |||
| Benzene, 1-ethynyl-4-methyl- | Benzene, 1-ethynyl-4-methyl- | 1 | C | Processing aid | Processing aid | ||
| Butanal, 3-hydroxy- | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
||
| Caprolactam | 2 | PD | Processing aid | Processing aid | |||
| Cis-bicyclo[4.2.0]octane | Cis-bicyclo[4.2.0]octane | 1 | 1 | C | Processing aid | Processing aid | |
| Cyclotrisiloxane, hexamethyl- | 1 | 1 | C | Cosmetics/Lubricant | Cosmetics/Lubricant | Cosmetics/Lubricant | |
| Ethanone, 1-(2,3-dihydro-1H-inden-5-yl)- | 1 | C | Daily chemical products related | Daily chemical products related | Pungent odor | ||
| Ethyne, fluoro- | 1 | C | Processing aid | Processing aid | |||
| Furan, 2,5-dihydro-2,5-dimethyl- | Furan, 2,5-dihydro-2,5-dimethyl- | 1 | C | Processing aid | Processing aid | ||
| Hexacosyl acetate | 1 | C | Flavor and fragrance agent |
Flavor and fragrance agent |
Flavor and fragrance agent |
||
| Hydroxylamine, O-decyl | Hydroxylamine, O-decyl | 1 | 1 | C | Processing aid | Processing aid | Ammonia-like odor |
| N-dl-Alanylglycine | 1 | C | Pharmaceutical/Food related |
Pharmaceutical/Food related |
Pharmaceutical/Food related |
||
| n-Hexylmethylamine | 1 | 1 | C | Processing aid | Processing aid | ||
| Pentanamide | C | Pharmaceutical related | Pharmaceutical related | Pharmaceutical related | |||
| Phenol, 2-(3-hydroxy-3-methyl-1-butenyl)-, (Z)- | 1 | C | Daily chemical products related |
Daily chemical products related |
Daily chemical products related |
||
| Phenol, 2,4-bis(1,1-dimethylethyl)- | 1 | 1 | AD | Antioxidant | Antioxidant | ||
| Propanamide | C | Pharmaceutical related | Pharmaceutical related | Pharmaceutical related | |||
| R-(-)-Cyclohexylethylamine | 1 | 1 | 1 | C | Processing aid | Processing aid | Strong ammonia-like odor |
| sec-Butylamine | 1 | C | Processing aid | Processing aid |
| Compound name | Peak intensity | Peak intensity | Peak intensity | Classification | Classification |
|---|---|---|---|---|---|
| Compound name | P7 | P8 | P9 | Type | Possible origin |
| Hydrocarbons | |||||
| Cetene | 1 | 2 | PD | ||
| Decane, 4-methyl- | 1 | PD | |||
| Dodecane | 2 | PD | |||
| Dodecane, 2,6,10-trimethyl- | 1 | C | Cosmetic related | ||
| Dodecane, 2,6,11-trimethyl- | 2 | C | Cosmetic related | ||
| Eicosane | 2 | PD | |||
| Heptacosane | 3 | 2 | PD | ||
| Heptadecane, 2,6,10,15-tertamethyl- | 1 | C | Processing aid | ||
| Hexadecane | 1 | 1 | PD | ||
| Hexadecane, 2,6,10,14-tetramethyl- | 1 | C | Processing aid | ||
| Octadecane | 1 | 1 | PD | ||
| Octadecane, 3-ethyl-5-(2-ethylbutyl)- | 1 | C | Processing aid | ||
| Octadecane, 6-methyl- | 1 | 1 | PD | ||
| Octane, 4-methyl- | 1 | PD | |||
| Pentadecane | 2 | PD | |||
| Pentadecane, 3-methyl- | 1 | PD | |||
| Tetradecane | 1 | 1 | PD | ||
| Tetradecane, 2,6,10-trimethyl- | 1 | 2 | C | Cosmetic or pharmaceutical related | |
| Undecane | 2 | PD | |||
| Undecane, 4-methyl- | 2 | PD | |||
| 1-Nonadecene | 2 | 3 | PD | ||
| 1-Octadecene | 3 | PD | |||
| 1-Pentadecene,2-methyl- | 1 | PD | |||
| 17-Pentatriacontene | 1 | 2 | 1 | AD | Lubricant |
| Esters | |||||
| 1-Propene-1,2,3-tricarboxylic acid, tributyl ester | 3 | 2 | 1 | AD | Plasticizer |
| 9-Octadecenoic acid (Z)-, tetradecyl ester | 1 | C | - | ||
| Decanedioic acid, bis(2,2,6,6- tetramethyl-4-piperidinyl) ester |
Decanedioic acid, bis(2,2,6,6- tetramethyl-4-piperidinyl) ester |
Decanedioic acid, bis(2,2,6,6- tetramethyl-4-piperidinyl) ester |
2 | C | Pharmaceutical related |
| Hexadecanoic acid, 1-(hydroxymethyl) -1,2-ethanediyl ester |
Hexadecanoic acid, 1-(hydroxymethyl) -1,2-ethanediyl ester |
1 | C | Cosmetic related or lubricant | |
| Hexadecanoic acid, butyl ester | 1 | 1 | C | Daily chemical products related | |
| Isopropyl myristate | 1 | C | Cosmetic related | ||
| Octadecanoic acid, 2-hydroxy-1,3-propanediyl ester | 3 | 1 | C | Cosmetic related | |
| Octadecanoic acid, 4-hydroxy-, methyl ester | 1 | C | Daily chemical products related | ||
| Octadecanoic acid, butyl ester | 2 | 2 | C | Daily chemical products related | |
| Oleic acid, 3-(octadecyloxy)propyl ester | 2 | 1 | C | Cosmetic related | |
| Oleic acid, eicosyl ester | 1 | 1 | C | Cosmetic related | |
| Oxalic acid, ethyl 2-isopropylphenyl ester | 2 | AD | Plasticizer | ||
| Trichloroacetic acid, pentadecyl ester | 1 | A | Plasticizer | ||
| Alcohols | |||||
| 1,4:3,6-Dianhydro-α-d-glucopyranose | 1 | C | Food related | ||
| 1-Dodecanol, 3,7,11-trimethyl- | 1 | C | Daily chemical products related | ||
| 1-Eicosanol | 2 | C | Daily chemical products related | ||
| 1-Hexadecanol, 2-methyl- | 1 | 2 | 1 | C | Daily chemical products related |
| 1-Octanol, 2,2-dimethyl- | 1 | AD | Plasticizer | ||
| 2,2,6,6-Tetramethyl-4-piperidinol | 1 | C | Pharmaceutical related | ||
| 2-Hexadecanol | 1 | AD | Lubricant | ||
| 2-Methyl-E,E-3,13-Octadecadien-1-ol | 1 | C | Pharmaceutical related | ||
| 3-Hexadecanol | 1 | C | Cosmetic related or surfactant | ||
| 3-Isopropyl-6,7-dimethyltricyclo[4.4.0.0(2,8)] decane-9,10-diol |
3-Isopropyl-6,7-dimethyltricyclo[4.4.0.0(2,8)] decane-9,10-diol |
1 | AD | Plasticizer or flame retardant. | |
| Behenic alcohol | 2 | 1 | C | Cosmetic related or lubricant | |
| Ethanol, 2-(octadecyloxy)- | 1 | 2 | C | Cosmetic related or lubricant | |
| Ethyl iso-allocholate | 1 | 1 | 1 | C | Pharmaceutical related |
| n-Tetracosanol-1 | 2 | 2 | C | Pharmaceutical related | |
| Octacosanol | 1 | 2 | C | Pharmaceutical related | |
| tert-Hexadecanethiol | 1 | 1 | C | Processing aid | |
| α-N-Normethadol | 1 | C | Pharmaceutical related | ||
| Benzene derivatives | |||||
| 1,3-Benzenedicarboxylic acid, bis(2-ethylhexyl) ester | 1 | A | Plasticizer | ||
| 1,3-Diacetylbenzene | 1 | C | Pharmaceutical related | ||
| 1,4-Benzenediol, 2,6-bis(1,1-dimethylethyl)- | 1 | A | Antioxidant | ||
| 3,5-di-tert-Butyl-4-hydroxyacetophenone | 1 | 1 | A | Antioxidant and stabilizer | |
| 4[h]-Pyridone, 1-benzyl-3,5-dichloro-2,6-dimethyl- | 1 | C | Pesticide | ||
| Benzene, 1,1'-(1,1,2,2-tetramethyl-1,2-ethanediyl)bis- | 2 | C | Processing aid | ||
| Benzene, 1,3-bis(1,1-dimethylethyl)- | 1 | AD | Antioxidant | ||
| Benzeneethanol, α-methyl-3-(1-methylethyl)- | 1 | C | Daily chemical products related | ||
| Benzenepropanoic acid, 3,5-bis(1,1-dimethyl ethyl)-4-hydroxy-, octadecyl ester |
Benzenepropanoic acid, 3,5-bis(1,1-dimethyl ethyl)-4-hydroxy-, octadecyl ester |
3 | C | Pharmaceutical related | |
| Benzenmethanol, a,a-dimethyl- | 1 | C | Processing aid | ||
| Benzestrol | 1 | C | Pharmaceutical related | ||
| Diisooctyl phthalate | 1 | 2 | A | Plasticizer | |
| Phenol, 2,4-bis(1,1-dimethylethyl)- | 1 | 2 | 1 | AD | Antioxidant |
| Phenol, 2,6-bis(1,1-dimethylethyl)-4-ethyl- | 1 | AD | Antioxidant | ||
| Phenol, 2,6-di-tert-butyl-4-ethyl- | 1 | AD | Antioxidant | ||
| Phenol, 2-methyl-4-(1,1,3,3-tetramethylbutyl)- | 2 | AD | Antioxidant | ||
| Phenol, 4-(1,1,3,3-tetramethylbutyl)- | 1 | AD | Antioxidant or stabilizer | ||
| Phenol, 4-(1,1-dimethylpropyl)- | 1 | C | Pharmaceutical related | ||
| Phthalic acid, butyl tetradecyl ester | 1 | A | Plasticizer | ||
| p-Octylacetophenone | 1 | C | Cosmetic related | ||
| γ-Chlorobutyrophenone | 1 | C | Surfactant | ||
| Acids, Aldehydes, Amides, Ketones | |||||
| Octadecanoic acid | 2 | C | Daily chemical products related | ||
| cis-13-Eicosenoic acid | 1 | C | Food related | ||
| n-Hexadecanoic acid | 1 | 1 | C | Daily chemical products related | |
| Pterin-6-carboxylic acid | 1 | C | Pharmaceutical related | ||
| 10-Octadecenal | 1 | C | Daily chemical products related | ||
| 5-Octadecenal | 1 | 1 | C | Daily chemical products related | |
| 8-Octadecenal | 1 | C | Daily chemical products related | ||
| E-15-Heptadecenal | 3 | C | Food related | ||
| 13-Docosenamide, (Z)- | 1 | 3 | C | Daily chemical products related | |
| Cis-11-Eicosenamide | 1 | C | Lubricant | ||
| 2,2,7,7Tetramethyltricyclo[6.2.1.0(1,6)] undec-4-en-3-one |
2,2,7,7Tetramethyltricyclo[6.2.1.0(1,6)] undec-4-en-3-one |
1 | C | Pharmaceutical related | |
| 2-Pentanone, 4-hydroxy-4-methyl- | 2 | C | - | ||
| 7,9-Di-tert-butyl-1-oxaspiro(4,5)deca-6,9-diene-2,8-dione | 1 | 2 | 1 | C | Processing aid |
| Others | |||||
| 2-Bromo dodecane | 1 | AD | Plasticizer | ||
| 2-Trifluoroacetoxytridecane | 1 | A | Plasticizer | ||
| Caprolactam | 3 | 1 | PD | Processing aid | |
| Octadecanal, 2-bromo- | 1 | 1 | 1 | AD | Plasticizer or lubricant |
| Octasiloxane, 1,1,3,3,5,5,7,7,9,9,11, 11,13,13,15,15-hexadecamethyl- |
Octasiloxane, 1,1,3,3,5,5,7,7,9,9,11, 11,13,13,15,15-hexadecamethyl- |
1 | A | Lubricant | |
| Octatriacontyl pentafluoropropionate | 1 | 1 | C | Surfactant or lubricant, toxic | |
| Silane, diethylheptyloxyoctadecyloxy- | 1 | 3 | 2 | Processing aid | |
| Triallyl isocyanurate | 1 | AD | Flame retardant or plasticizer | ||
| Tributyl acetylcitrate | 1 | 3 | A | Plasticizer |
| Compound Name | Peak Intensity | Peak Intensity | Peak Intensity | Possible origin |
|---|---|---|---|---|
| Compound Name | P7 | P8 | P9 | Possible origin |
| Benzene derivatives | ||||
| 1,2-Dimethoxy-4-(1-methoxy-1-propenyl)benzene | 1,2-Dimethoxy-4-(1-methoxy-1-propenyl)benzene | 1,2-Dimethoxy-4-(1-methoxy-1-propenyl)benzene | 1 | Pharmaceutical related |
| 2,4-Dimethyl-5,6-dimethoxy-8-aminoquinoline | 1 | Pharmaceutical related | ||
| 3,5-di-tert-Butyl-4-hydroxycinnamic acid | 3,5-di-tert-Butyl-4-hydroxycinnamic acid | 2 | Antioxidant and stabilizer | |
| Benzenesulfonamide, N-ethyl-2-methyl- | Benzenesulfonamide, N-ethyl-2-methyl- | 1 | Sweetener in food industry | |
| Benzyl alcohol, α,α-dimethyl-p-isopropyl- | 2 | Cosmetic or Pharmaceutical related | ||
| Ethanone, 1,1'-(1,4-phenylene)bis- | 1 | Plasticizer or UV absorber | ||
| Phenol, 2,6-bis(1,1-dimethylethyl)- | Phenol, 2,6-bis(1,1-dimethylethyl)- | 1 | 1 | Antioxidant |
| p-Octylacetophenone | 2 | 2 | Food industry | |
| Acids, Alcohols, Esters, Ketones | Acids, Alcohols, Esters, Ketones | |||
| Dimethylmuconic acid | 1 | Pharmaceutical related | ||
| 1-(3-Methoxymethyl-2,4,6-trimethylphenyl)ethanol | 1 | |||
| 1,4:3,6-Dianhydro-α-d-glucopyranose | 1,4:3,6-Dianhydro-α-d-glucopyranose | 2 | Food related | |
| Dibutyl itaconate | 1 | Plasticizer | ||
| Furan, 2-[(2-ethoxy-3,4-dimethyl-2-cyclohexen-1-ylidene)methyl]- | 1 | Cosmetic or food related | ||
| 2,5-Cyclohexadiene-1,4-dione, 2,6-bis (1,1-dimethylethyl)- |
2,5-Cyclohexadiene-1,4-dione, 2,6-bis (1,1-dimethylethyl)- |
1 | 1 | Pharmaceutical related |
| 2,5-di-tert-Butyl-1,4-benzoquinone | 2,5-di-tert-Butyl-1,4-benzoquinone | 1 | 1 | Stabilizer |
| 7,9-Di-tert-butyl-1-oxaspiro(4,5)deca-6,9- diene-2,8-dione |
7,9-Di-tert-butyl-1-oxaspiro(4,5)deca-6,9- diene-2,8-dione |
3 | 3 | Processing aid hazardous |
| Others | ||||
| 4-Piperidinol, 2,2,6,6-tetramethyl- | 1 | 3 | Processing aid | |
| Caprolactam | 3 | 2 | 2 | Processing aid |
| Cyclobarbital | 1 | Pharmaceutical related | ||
| Hexasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11-dodecamethyl- | 1 | 1 | 1 | Plasticizer |
| l-Guanidinosuccinimide | 1 | Pharmaceutical related | ||
| Morpholinomethyl urea i | 1 | Corrosion inhibitor or flame retardant. | ||
| Octasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11,13,13,15,15-hexadecamethyl- | 1 | 1 | 1 | Plasticizer or lubricant |
| Pregn-5-ene-3,11,20-trione, cyclic 3,20-bis (1,2-ethanediyl acetal) |
Pregn-5-ene-3,11,20-trione, cyclic 3,20-bis (1,2-ethanediyl acetal) |
Pregn-5-ene-3,11,20-trione, cyclic 3,20-bis (1,2-ethanediyl acetal) |
1 | Pharmaceutical related |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




