Submitted:
23 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
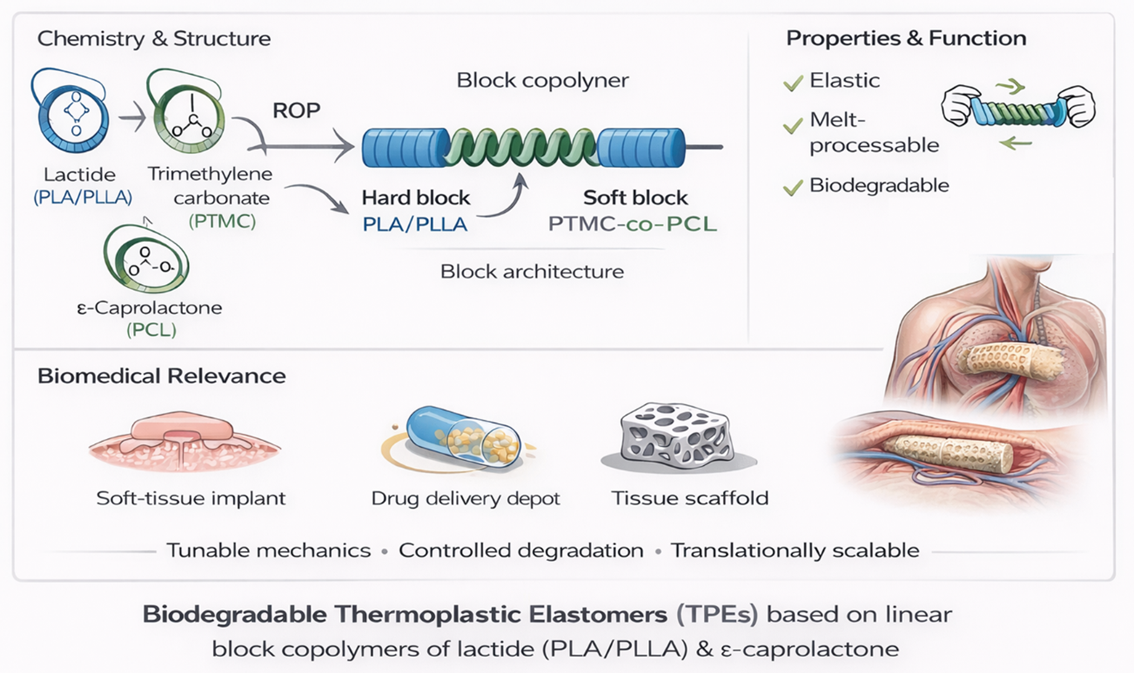
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. 3D Printing
2.3. Differential Scanning Calorimetry
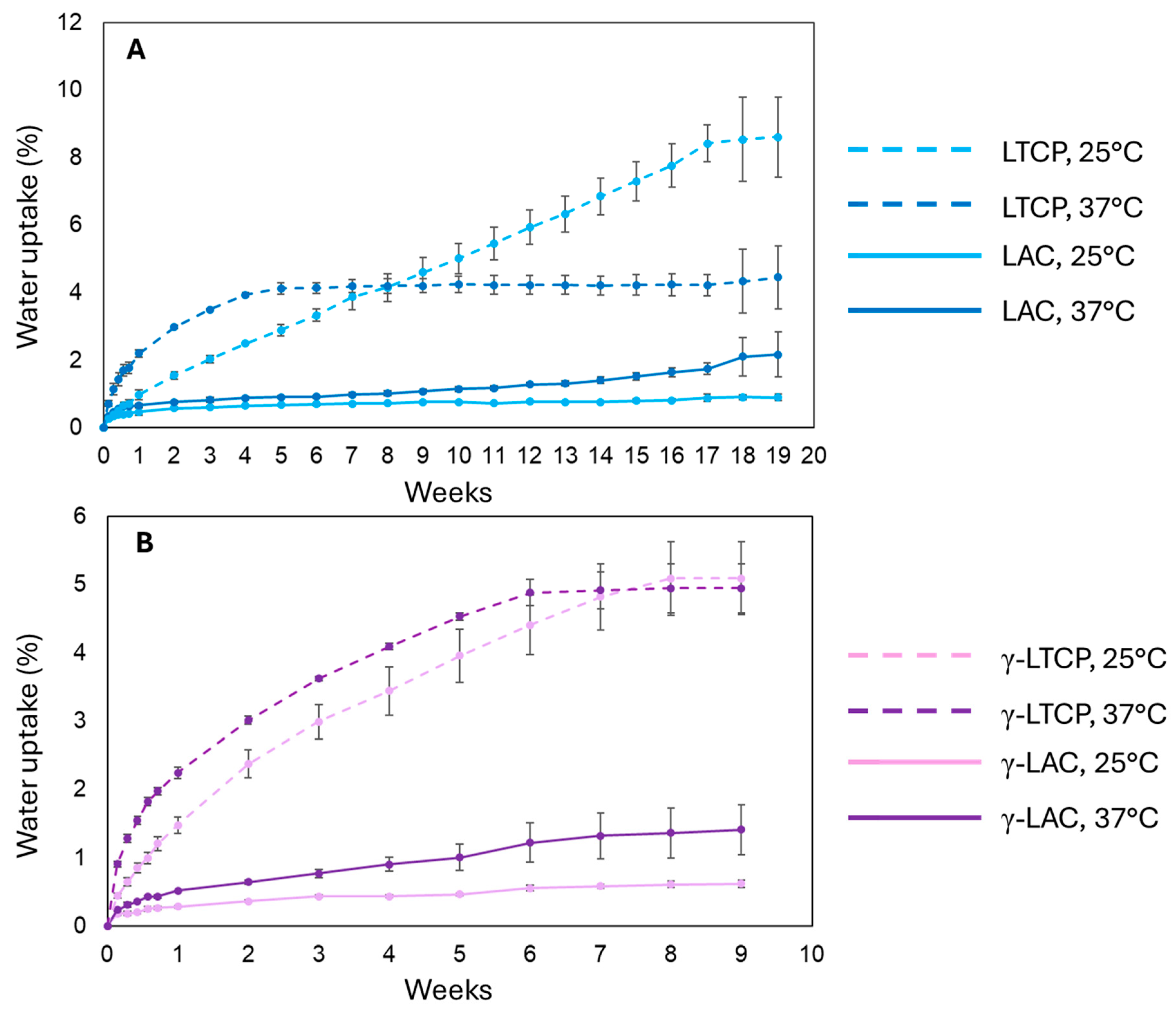
2.4. Water Uptake
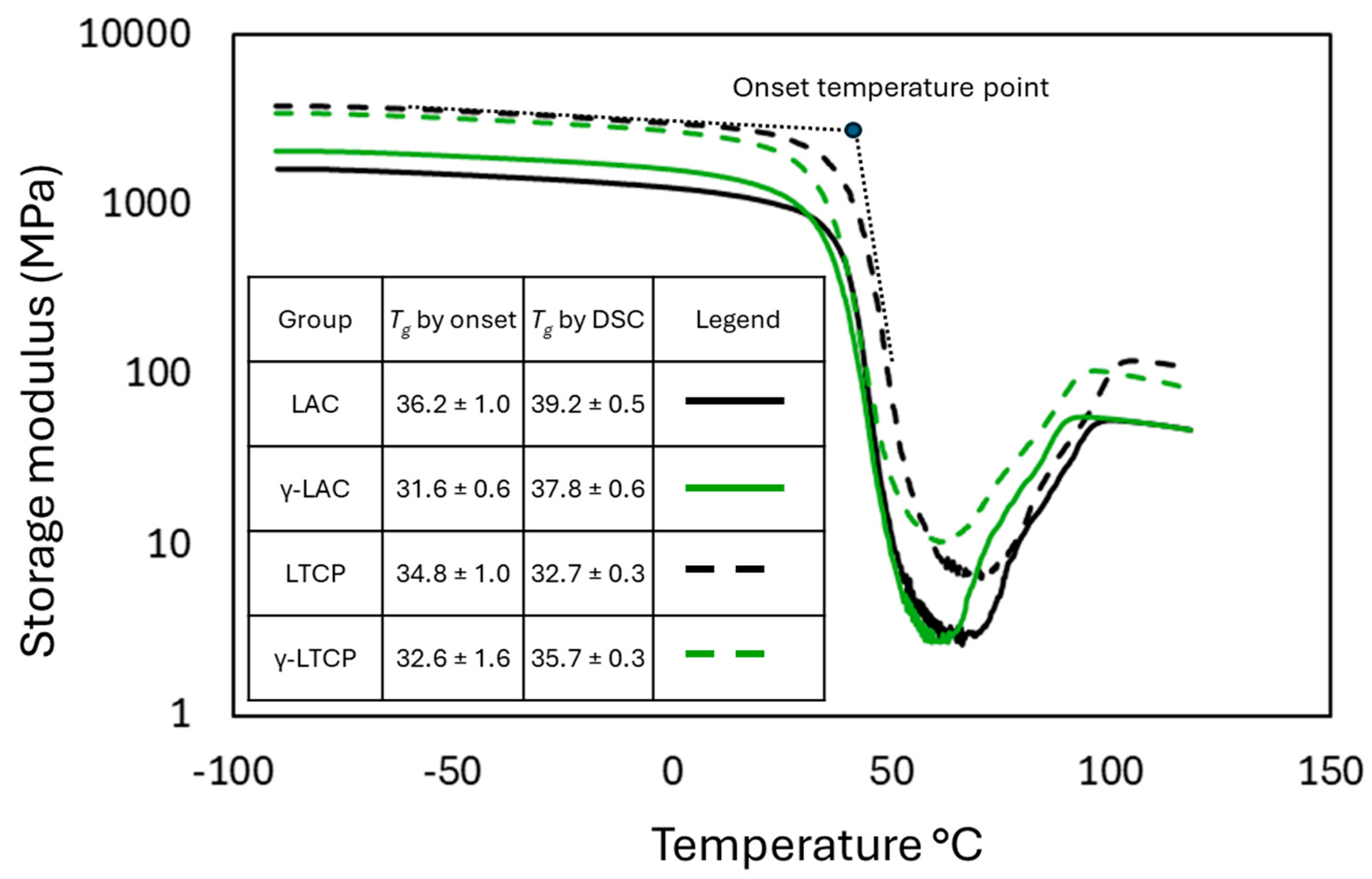
2.5. Dynamic Mechanical Analysis
2.6. Gel Permeation Chromatography
2.7. Mechanical Testing and Recovery Rate
3. Results and Discussion
3.1. Structural Characterization
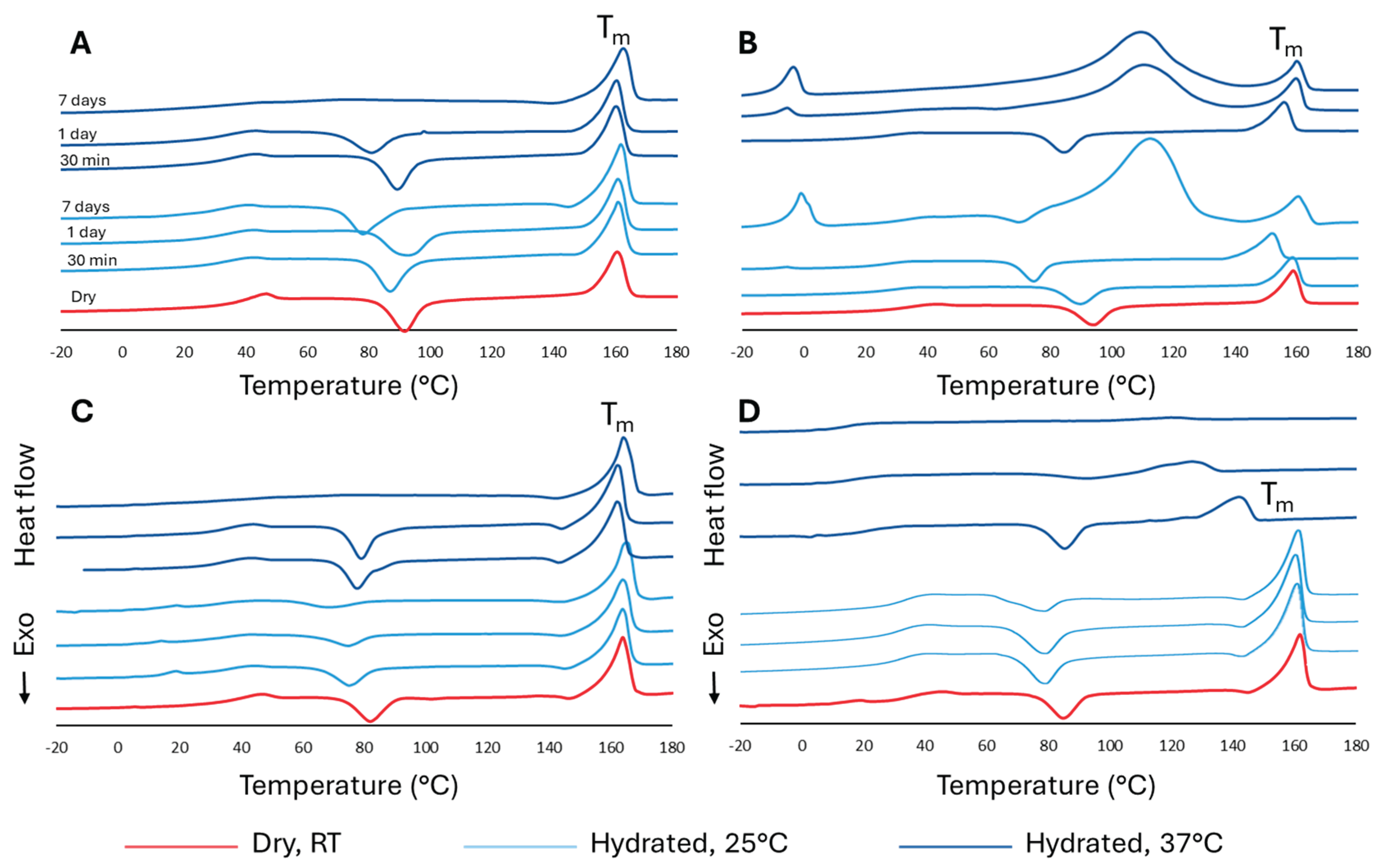
3.2. Thermal Characterization
3.3. Water Uptake
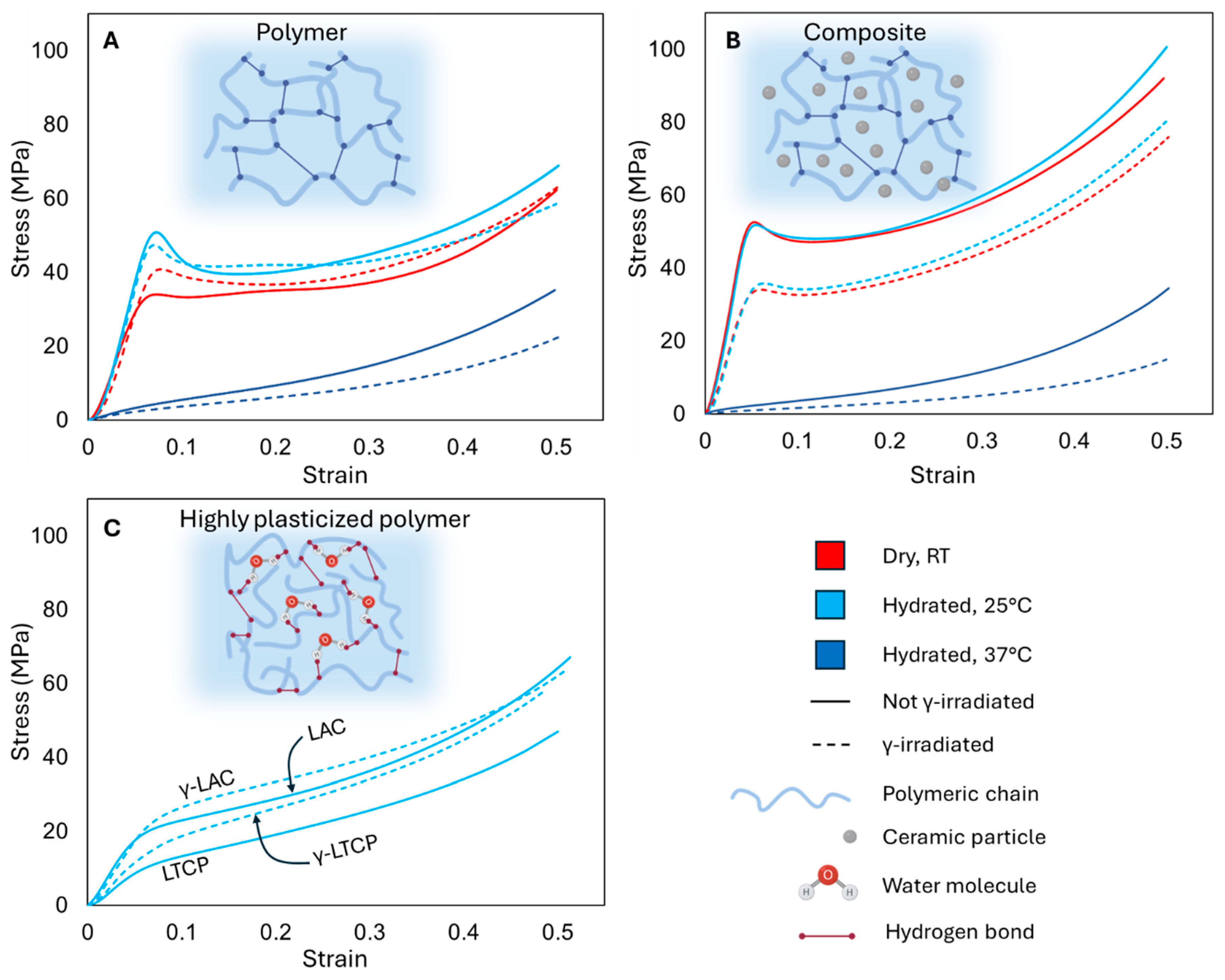
3.4. Mechanical Properties
3.4.1. Effect of γ-Irradiation
3.4.2. Effect of Hydration
3.4.3. Effects of Water Saturation
3.4.4. Effect of Temperature
3.4.5. Effect of Ceramic Particles
3.4.6. Combined Effects of Irradiation and Ceramic Particles
3.4.7. Combined Effects of Irradiation, Ceramic Particles, and Physiological Conditions
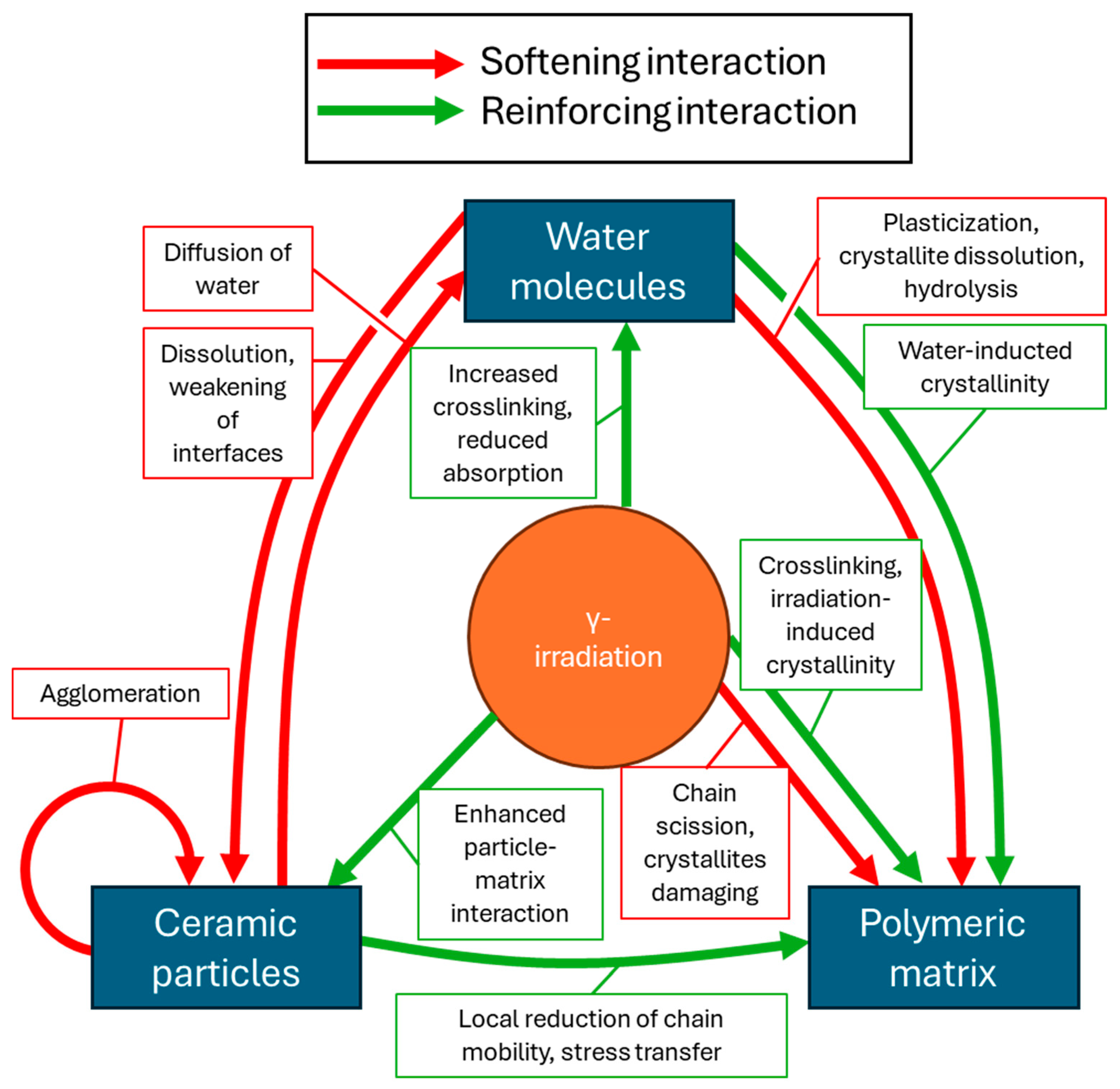
3.4.8. Molecular-Scale Interactions in Irradiated Composites in Physiological Conditions
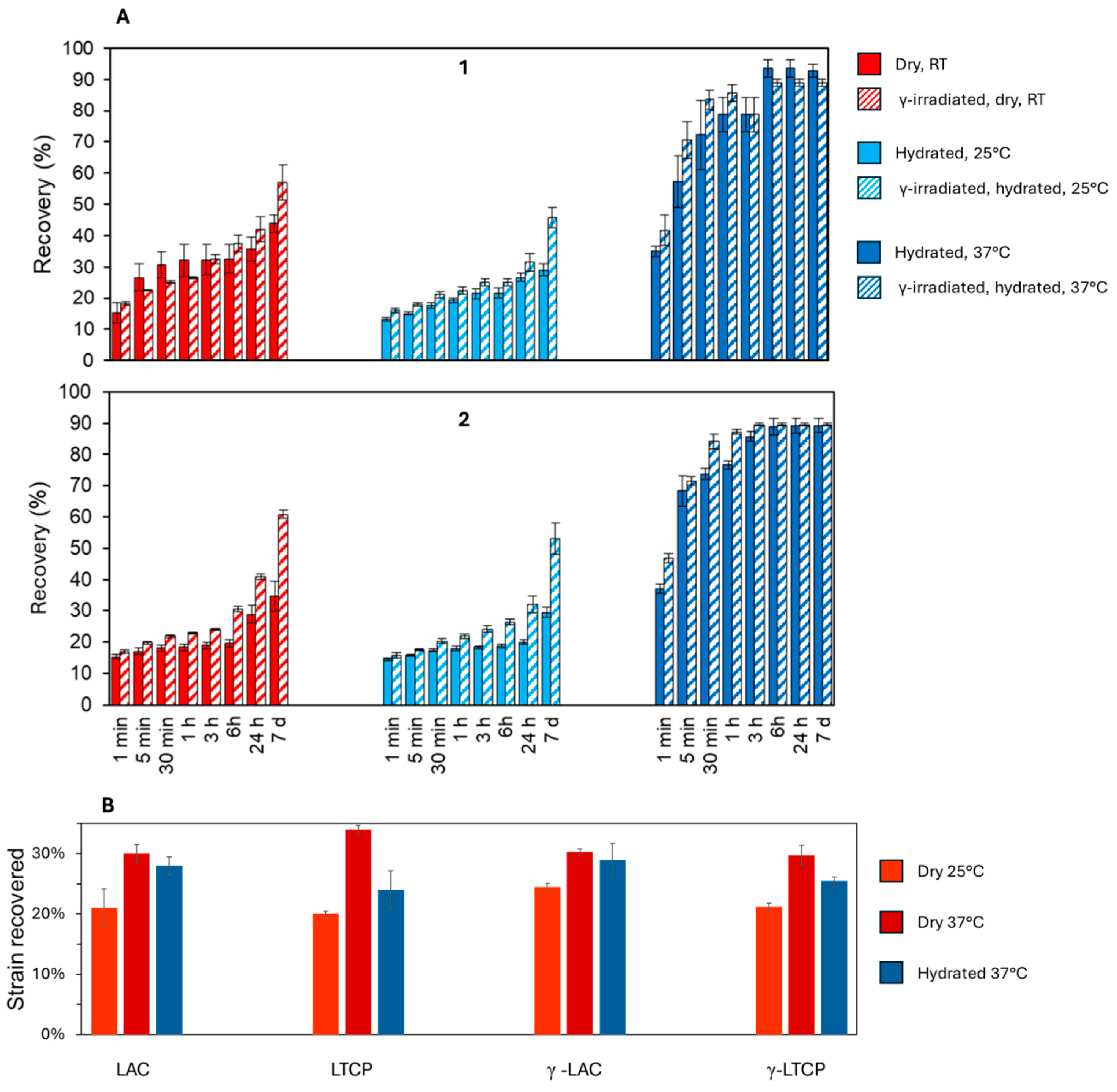
3.4. Recovery
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| β-TCP | β-tricalcium phosphate |
| ASTM | American society for testing and materials |
| CE | Conformité Européenne |
| DCM | Dichloromethane |
| DMA | Dynamic mechanical analysis |
| DSC | Differential scanning calorimetry |
| FDA | Food and drug administration |
| GPC | Gel permeation chromatography |
| ND | Not detectable |
| PBS | Phosphate-buffered saline |
| PCL | Polycaprolactone |
| PDI | Polydispersity index |
| PDLLA | Poly-d,l-lactic acid |
| PLA | Poly-lactic acid |
| PLGA | Poly(lactic-co-glycolic acid) |
| PLLA | Poly-L-Lactic Acid |
| PTMC | Poly-trimethylene carbonate |
| RT | Room temperature |
| SGBR | Scaffold guided bone regeneration |
| STL | Stereolithography |
| TPEs | Thermoplastic elastomers |
References
- Widjaja, L.K.; Kong, J.F.; Chattopadhyay, S.; Lipik, V.T.; Liow, S.S.; Abadie, M.J.M.; Venkatraman, S.S. Triblock copolymers of ε-caprolactone, L-lactide, and trimethylene carbonate: Biodegradability and elastomeric behavior. Journal of Biomedical Materials Research Part A 2011, 99A(1), 38–46. [Google Scholar] [CrossRef] [PubMed]
- Widjaja, L.K.; Kong, J.F.; Chattopadhyay, S.; Lipik, V.T.; Liow, S.S.; Abadie, M.J.M.; Venkatraman, S.S. Triblock copolymers of ε-caprolactone, trimethylene carbonate, and L-lactide: Effects of using random copolymer as hard-block. Journal of the Mechanical Behavior of Biomedical Materials 2012, 6, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Grijpma, D.W.; Feijen, J. Triblock copolymers based on 1,3-trimethylene carbonate and lactide as biodegradable thermoplastic elastomers. Macromolecular Chemistry and Physics 2004, 205(7), 867–875. [Google Scholar] [CrossRef]
- Zhang, Z.; Grijpma, D.W.; Feijen, J. Thermoplastic elastomers based on poly(lactide)–poly(trimethylene carbonate-co-caprolactone)–poly(lactide) triblock copolymers and their stereocomplexes. Journal of Controlled Release 2006, 116(2), e29–e31. [Google Scholar] [CrossRef]
- Seifi, E.; Mohseni, M.; Samson, K.D.; Cavelier, S.; Ayyachi, T.; Hutmacher, D.W. Comprehensive physicochemical evaluation of 3D-printed medical-grade poly (lactic acid)-based composite: Mechanical, thermal, and morphological properties. Journal of the American Ceramic Society 2025, 108(11), e70069. [Google Scholar] [CrossRef]
- Hill, D. Design engineering of biomaterials for medical devices. 1998. [Google Scholar]
- Sharma; Anup, N.; Tekade, R.K. Chapter 21 - Achieving sterility in biomedical and pharmaceutical products (part-II): radiation sterilization. In The Future of Pharmaceutical Product Development and Research; Tekade, R.K., Ed.; Academic Press, 2020; pp. 789–848. [Google Scholar]
- Bento, S. A.; Gaspar, M.C.; Coimbra, P.; de Sousa, H.C.; Braga, M. E. M. A review of conventional and emerging technologies for hydrogels sterilization. International Journal of Pharmaceutics 2023, 634, 122671. [Google Scholar] [CrossRef]
- Noah, E.M.; Chen, J.; Jiao, X.; Heschel, I.; Pallua, N. Impact of sterilization on the porous design and cell behavior in collagen sponges prepared for tissue engineering. Biomaterials 2002, 23(14), 2855–2861. [Google Scholar] [CrossRef]
- Dai, Z.; Ronholm, J.; Tian, Y.; Sethi, B.; Cao, X. Sterilization techniques for biodegradable scaffolds in tissue engineering applications. Journal of Tissue Engineering 2016, 7, 2041731416648810. [Google Scholar] [CrossRef]
- Bansal, N.; Arora, S. Exploring the impact of gamma rays and electron beam irradiation on physico-mechanical properties of polymers & polymer composites: A comprehensive review. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms 2024, 549, 165297. [Google Scholar] [CrossRef]
- Babanalbandi; Hill, D.; O'donnell, J.; Pomery, P.; Whittaker, A. An electron spin resonance study on γ-irradiated poly (l-lactic acid) and poly (d, l-lactic acid). Polymer Degradation and Stability 1995, 50(3), 297–304. [Google Scholar] [CrossRef]
- Hooper, K.A.; Cox, J.D.; Kohn, J. Comparison of the effect of ethylene oxide and γ-irradiation on selected tyrosine-derived polycarbonates and poly (L-lactic acid). Journal of Applied Polymer Science 1997, 63(11), 1499–1510. [Google Scholar] [CrossRef]
- Gupta, M.C.; Deshmukh, V.G. Radiation effects on poly (lactic acid). Polymer 1983, 24(7), 827–830. [Google Scholar] [CrossRef]
- Rouif, S. Radiation cross-linked plastics: a versatile material solution for packaging, automotive. Electrotechnic and Electronics, Radiation Physics and Chemistry 2004, 71(1-2), 527–530. [Google Scholar]
- Rouif, S. Radiation cross-linked polymers: Recent developments and new applications. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms 2005, 236(1-4), 68–72. [Google Scholar] [CrossRef]
- Seguchi, T.; Yagi, T.; Ishikawa, S.; Sano, Y. New material synthesis by radiation processing at high temperature—polymer modification with improved irradiation technology. Radiation Physics and Chemistry 2002, 63(1), 35–40. [Google Scholar] [CrossRef]
- Plikk, P.; Odelius, K.; Hakkarainen, M.; Albertsson, A.C. Finalizing the properties of porous scaffolds of aliphatic polyesters through radiation sterilization. Biomaterials 2006, 27(31), 5335–5347. [Google Scholar] [CrossRef]
- Cottam, E.; Hukins, D.W.; Lee, K.; Hewitt, C.; Jenkins, M.J. Effect of sterilisation by gamma irradiation on the ability of polycaprolactone (PCL) to act as a scaffold material. Medical Engineering & Physics 2009, 31(2), 221–226. [Google Scholar]
- Yunoki, S.; Ikoma, T.; Monkawa, A.; Ohta, K.; Tanaka, J.; Sotome, S.; Shinomiya, K. Influence of γ irradiation on the mechanical strength and in vitro biodegradation of porous hydroxyapatite/collagen composite. Journal of the American Ceramic Society 2006, 89(9), 2977–2979. [Google Scholar] [CrossRef]
- Di Foggia, M.; Corda, U.; Plescia, E.; Taddei, P.; Torreggiani, A. Effects of sterilisation by high-energy radiation on biomedical poly-(ε-caprolactone)/hydroxyapatite composites. Journal of Materials Science: Materials in Medicine 2010, 21(6), 1789–1797. [Google Scholar] [CrossRef]
- Selim, M.; Bullock, A.J.; Blackwood, K.A.; Chapple, C.R.; MacNeil, S. Developing biodegradable scaffolds for tissue engineering of the urethra. BJU International 2011, 107(2), 296–302. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, E.; Stawarz, A.; Rimnac, C. Gamma radiation sterilization reduces the fatigue crack propagation resistance of human cortical bone. In Transactions of the Annual Meeting - Orthopaedic Research Society; 2002; pp. 551–551. [Google Scholar]
- Mitchell, E.J.; Stawarz, A.M.; Kayacan, R.; Rimnac, C.M. The effect of gamma radiation sterilization on the fatigue crack propagation resistance of human cortical bone. Journal of Bone and Joint Surgery 2004, 86(12), 2648–2657. [Google Scholar] [CrossRef] [PubMed]
- Loty, B.; Courpied, J.; Tomeno, B.; Postel, M.; Forest, M.; Abelanet, R. Bone allografts sterilised by irradiation: Biological properties, procurement and results of 150 massive allografts. International Orthopaedics 1990, 14(3), 237–242. [Google Scholar] [CrossRef] [PubMed]
- Narkis, M.; Sibony-Chaouat, S.; Siegmann, A.; Shkolnik, S.; Bell, J. Irradiation effects on polycaprolactone. Polymer 1985, 26(1), 50–54. [Google Scholar] [CrossRef]
- Mohammadian-Kohol, M.; Nasrabadi, M.N.; Navarchian, A.H.; Jabbari, I.; Seyedhabashi, M.R. A study of the effects of gamma irradiation on the structural and mechanical properties of polyvinyl butyral film. Radiation Physics and Chemistry 2018, 152, 1–5. [Google Scholar] [CrossRef]
- Kantoǧlu, Ö.; Güven, O. Radiation induced crystallinity damage in poly (L-lactic acid). Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms 2002, 197(3-4), 259–264. [Google Scholar] [CrossRef]
- Dadbin, S.; Kheirkhah, Y. Gamma irradiation of melt processed biomedical PDLLA/HAP nanocomposites. Radiation Physics and Chemistry 2014, 97, 270–274. [Google Scholar] [CrossRef]
- T.a., A.S.f. Materials, Standard test method for compressive properties of rigid plastics. 2023. [Google Scholar]
- I.O.f. Standardization, 11137-1:2025 Sterilization of health care products—Radiation—Part 1: requirements for the development, validation and routine control of a sterilization process for medical devices. 2025.
- Fischer, E.; Sterzel, H.J.; Wegner, G. Investigation of the structure of solution grown crystals of lactide copolymers by means of chemical reactions. Kolloid-Zeitschrift und Zeitschrift für Polymere 1973, 251(11), 980–990. [Google Scholar] [CrossRef]
- Sato, K.; Kobayashi, S.; Kusakari, M.; Watahiki, S.; Oikawa, M.; Hoshiba, T.; Tanaka, M. The relationship between water Structure and blood compatibility in poly (2-methoxyethyl acrylate)(PMEA) analogues. Macromolecular Bioscience 2015, 15(9), 1296–1303. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, L.; Bruzaud, S.; Kaci, M.; Bourmaud, A.; Gautier, N.; Grohens, Y. The effects of gamma irradiation on the morphology and properties of polylactide/Cloisite 30B nanocomposites. Polymer Degradation and Stability 2013, 98(1), 348–355. [Google Scholar] [CrossRef]
- Nugroho, P.; Mitomo, H.; Yoshii, F.; Kume, T. Degradation of poly (L-lactic acid) by γ-irradiation. Polymer Degradation and Stability 2001, 72(2), 337–343. [Google Scholar] [CrossRef]
- Aouat, T.; Kaci, M.; Lopez-Cuesta, J.-M.; Devaux, E.; Mahlous, M. The effect of gamma-irradiation on morphology and properties of melt-spun poly (lactic acid)/cellulose fibers. Polymer Degradation and Stability 2019, 160, 14–23. [Google Scholar] [CrossRef]
- Milicevic, D.; Trifunovic, S.; Galovic, S.; Suljovrujic, E. Thermal and crystallization behaviour of gamma irradiated PLLA. Radiation Physics and Chemistry 2007, 76(8-9), 1376–1380. [Google Scholar] [CrossRef]
- Khattari, Z.; Mahdy, E.A. Probabilistic approach to assessing the radiation shielding capabilities of carbonated hydroxyapatite modified by carbonate-to-phosphate ratio and temperature. Preprint. 2024. [Google Scholar]
- Fu, G.; Zeng, L.; Jiang, J.; Xia, Z.; Jing, B.; Zhang, X. Preparation and characterization of nanocomposites based on poly (ε-caprolactone) and the surface grafted nanohydroxyapatite with the comb-shaped poly (ε-caprolactone) brushes. Polymers and Polymer Composites 2012, 20(5), 463–470. [Google Scholar] [CrossRef]
- Wilberforce, S.I.; Finlayson, C.E.; Best, S.M.; Cameron, R.E. The influence of hydroxyapatite (HA) microparticles (m) and nanoparticles (n) on the thermal and dynamic mechanical properties of poly-l-lactide. Polymer 2011, 52(13), 2883–2890. [Google Scholar] [CrossRef]
- Hodge, R.; Edward, G.H.; Simon, G.P. Water absorption and states of water in semicrystalline poly (vinyl alcohol) films. Polymer 1996, 37(8), 1371–1376. [Google Scholar] [CrossRef]
- González-Campos, J.B.; Prokhorov, E.; Luna-Bárcenas, G.; Fonseca-García, A.; Sanchez, I.C. Dielectric relaxations of chitosan: The effect of water on the α-relaxation and the glass transition temperature. Journal of Polymer Science Part B: Polymer Physics 2009, 47(22), 2259–2271. [Google Scholar] [CrossRef]
- Beltrán, F.; De La Orden, M.; Lorenzo, V.; Pérez, E.; Cerrada, M.L.; Urreaga, J.M. Water-induced structural changes in poly (lactic acid) and PLLA-clay nanocomposites. Polymer 2016, 107, 211–222. [Google Scholar] [CrossRef]
- Pan, P.; Zhu, B.; Kai, W.; Dong, T.; Inoue, Y. Effect of crystallization temperature on crystal modifications and crystallization kinetics of poly (L-lactide). Journal of Applied Polymer Science 2008, 107(1), 54–62. [Google Scholar] [CrossRef]
- Badia, J.; Santonja-Blasco, L.; Martínez-Felipe, A.; Ribes-Greus, A. Hygrothermal ageing of reprocessed polylactide. Polymer Degradation and Stability 2012, 97(10), 1881–1890. [Google Scholar] [CrossRef]
- Vyavahare, O.; Ng, D.; Hsu, S.L. Analysis of structural rearrangements of poly (lactic acid) in the presence of water. The Journal of Physical Chemistry B 2014, 118(15), 4185–4193. [Google Scholar] [CrossRef] [PubMed]
- Qiao, C.; Ma, X.; Zhang, J.; Yao, J. Effect of hydration on water state, glass transition dynamics and crystalline structure in chitosan films. Carbohydrate Polymers 2019, 206, 602–608. [Google Scholar] [CrossRef]
- Qiao, D.; Zhang, B.; Huang, J.; Xie, F.; Wang, D.K.; Jiang, F.; Zhao, S.; Zhu, J. Hydration-induced crystalline transformation of starch polymer under ambient conditions. International Journal of Biological Macromolecules 2017, 103, 152–157. [Google Scholar] [CrossRef]
- Xiang; Lv, C.; Zhou, H. Changes in crystallization behaviors of poly (vinyl alcohol) induced by water content. Journal of Vinyl and Additive Technology 2020, 26(4), 613–622. [Google Scholar] [CrossRef]
- Alariqi, S.A.S.; Kumar, A.P.; Rao, B.S.M.; Singh, R.P. Effect of γ-dose rate on crystallinity and morphological changes of γ-sterilized biomedical polypropylene. Polymer Degradation and Stability 2009, 94(2), 272–277. [Google Scholar] [CrossRef]
- Zhudi, Z.; Wenxue, Y.; Xinfang, C. Study on increase in crystallinity in γ-irradiated poly(vinylidene fluoride). Radiation Physics and Chemistry 2002, 65(2), 173–176. [Google Scholar] [CrossRef]
- Kaouach, H. γ-irradiation induced effects on structural, morphological and photoluminescence properties of PEO-PVA blended polymer films. Optik 2023, 274, 170563. [Google Scholar] [CrossRef]
- Dadbin, S.; Naimian, F. Gamma radiation induced property modification of poly (lactic acid)/hydroxyapatite bio-nanocomposites. Polymer International 2014, 63(6), 1063–1069. [Google Scholar] [CrossRef]
- Harris, A.M.; Lee, E.C. Heat and humidity performance of injection molded PLA for durable applications. Journal of Applied Polymer Science 2010, 115(3), 1380–1389. [Google Scholar] [CrossRef]
- Comeau, S.J.; McCoy, J.D.; Kropka, J.M. The effect of water sorption on the calorimetric signature of aging in DGEBA epoxies. Polymer Degradation and Stability 2023, 216, 110473. [Google Scholar] [CrossRef]
- López; Ayyachi, T.; Brouwers, T.; Åberg, J.; Wistrand, A.F.; Engqvist, H. 1-Year pullout strength and degradation of ultrasound welded vs tapped craniomaxillofacial fixation screws. Polymer Testing 2022, 109, 107519. [Google Scholar] [CrossRef]
- McBrierty, V.; Martin, S.; Karasz, F. Understanding hydrated polymers: The perspective of NMR. Journal of Molecular Liquids 1999, 80(2-3), 179–205. [Google Scholar] [CrossRef]
- Zhou, J.; Lucas, J.P. Effects of water on a graphite/epoxy composite. Journal of Thermoplastic Composite Materials 1996, 9(4), 316–328. [Google Scholar] [CrossRef]
- Alizadeh-Osgouei, M.; Li, Y.; Wen, C. A comprehensive review of biodegradable synthetic polymer-ceramic composites and their manufacture for biomedical applications. Bioactive Materials 2019, 4, 22–36. [Google Scholar] [CrossRef]
- Cavelier, S.; Dargaville, B.L.; Hutmacher, D.W. Water interactions in hydrated aliphatic polyester composite scaffolds. Advanced Materials 2025, n/a(n/a), e11614. [Google Scholar] [CrossRef]
- Blasi, P.; D'Souza, S.S.; Selmin, F.; DeLuca, P.P. Plasticizing effect of water on poly (lactide-co-glycolide). Journal of Controlled Release 2005, 108(1), 1–9. [Google Scholar] [CrossRef]
- D’Amico, S. Influence of humidity on glass transition of PLA films. Research and Reviews in Materials Science and Chemistry 2014, 3(2), 115–122. [Google Scholar]
- Mastalygina, E.E.; Aleksanyan, K.V. Recent approaches to the plasticization of poly(lactic acid) (PLA) (a review). Polymers 2024, 16(1), 87. [Google Scholar] [CrossRef]
- Wang, W.; Lu, H.; Liu, Y.; Leng, J. Sodium dodecyl sulfate/epoxy composite: water-induced shape memory effect and its mechanism. Journal of Materials Chemistry A 2014, 2(15), 5441–5449. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Chen, H.; Yang, G.; Zheng, X.; Zhou, S. Water-induced shape-memory poly (d, l-lactide)/microcrystalline cellulose composites. Carbohydrate Polymers 2014, 104, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Benavides, R.; Urbano, R.; Morales-Acosta, D.; Martínez-Pardo, M.; Carrasco, H.; Paula, M.; Da Silva, L. Effect of gamma radiation on crosslinking, water uptake and ion exchange on polystyrene-co-acrylic acid copolymers useful for fuel cells. International Journal of Hydrogen Energy 2019, 44(24), 12525–12528. [Google Scholar] [CrossRef]
- Kumar, K.S.; Singh, I. Effect of gamma-ray irradiation on the mechanical, thermal, and morphological behavior of sisal fiber/bio-PBS composites. Construction and Building Materials 2024, 450, 138703. [Google Scholar] [CrossRef]
- Alothman, O.Y.; Almajhdi, F.N.; Fouad, H. Effect of gamma radiation and accelerated aging on the mechanical and thermal behavior of HDPE/HA nano-composites for bone tissue regeneration. Biomedical Engineering Online 2013, 12 95. [Google Scholar] [CrossRef]
- Carraher, C.E., Jr. Seymour/Carraher's polymer chemistry; CRC press, 2003. [Google Scholar]
- Courtney, T.H. Mechanical behavior of materials; Waveland Press, 2005. [Google Scholar]
- Kumar, V.; Ali, Y.; Sonkawade, R.; Dhaliwal, A. Effect of gamma irradiation on the properties of plastic bottle sheet. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms 2012, 287, 10–14. [Google Scholar] [CrossRef]
- Zaki, M.F.; Elshaer, Y.H.; Taha, D.H. The alterations in high density polyethylene properties with gamma irradiation. Radiation Physics and Chemistry 2017, 139, 90–96. [Google Scholar] [CrossRef]
- Banjo, A.D.; Agrawal, V.; Auad, M.L.; Celestine, A.-D.N. Moisture-induced changes in the mechanical behavior of 3D printed polymers. Composites Part C: Open Access 2022, 7, 100243. [Google Scholar] [CrossRef]
- (!!! INVALID CITATION !!! [70, 71]).
- (!!! INVALID CITATION !!! [69, 72]). Seymour/Carraher's polymer chemistry, CRC press, 2003.
- Treloar, L.G. The physics of rubber elasticity; 1975. [Google Scholar]
- James, H.M.; Guth, E. Theory of the elastic properties of rubber. The Journal of Chemical Physics 1943, 11(10), 455–481. [Google Scholar] [CrossRef]
- Zhang, Y.; Tanner, K.E. Impact behavior of hydroxyapatite reinforced polyethylene composites. Journal of Materials Science: Materials in Medicine 2003, 14(1), 63–8. [Google Scholar] [CrossRef]
- Fu, S.-Y.; Feng, X.-Q.; Lauke, B.; Mai, Y.-W. Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate–polymer composites. Composites Part B: Engineering 2008, 39(6), 933–961. [Google Scholar] [CrossRef]
- Jiao, Z.; Luo, B.; Xiang, S.; Ma, H.; Yu, Y.; Yang, W. 3D printing of HA/PCL composite tissue engineering scaffolds. Advanced Industrial and Engineering Polymer Research 2019, 2(4), 196–202. [Google Scholar] [CrossRef]
- Ferri, J.; Gisbert, I.; García-Sanoguera, D.; Reig, M.; Balart, R. The effect of beta-tricalcium phosphate on mechanical and thermal performances of poly (lactic acid). Journal of Composite Materials 2016, 50(30), 4189–4198. [Google Scholar] [CrossRef]
- Kontou, E. Micromechanics model for particulate composites. Mechanics of Materials 2007, 39(7), 702–709. [Google Scholar] [CrossRef]
- Cui, Y.; Liu, Y.; Cui, Y.; Jing, X.; Zhang, P.; Chen, X. The nanocomposite scaffold of poly (lactide-co-glycolide) and hydroxyapatite surface-grafted with L-lactic acid oligomer for bone repair. Acta Biomaterialia 2009, 5(7), 2680–2692. [Google Scholar] [CrossRef]
- Choi, D.; Marra, K.G.; Kumta, P.N. Chemical synthesis of hydroxyapatite/poly (ε-caprolactone) composites. Materials Research Bulletin 2004, 39(3), 417–432. [Google Scholar] [CrossRef]
- Kobayashi, S.; Yamadi, S. Strain rate dependency of mechanical properties of TCP/PLLA composites after immersion in simulated body environments. Composites Science and Technology 2010, 70(13), 1820–1825. [Google Scholar] [CrossRef]
- Hao, J.; Yuan, M.; Deng, X. Biodegradable and biocompatible nanocomposites of poly (ϵ-caprolactone) with hydroxyapatite nanocrystals: Thermal and mechanical properties. Journal of Applied Polymer Science 2002, 86(3), 676–683. [Google Scholar] [CrossRef]
- Dziadek, M.; Menaszek, E.; Zagrajczuk, B.; Pawlik, J.; Cholewa-Kowalska, K. New generation poly (ε-caprolactone)/gel-derived bioactive glass composites for bone tissue engineering: Part I. Material properties, Materials Science and Engineering: C 2015, 56, 9–21. [Google Scholar] [CrossRef]
- Hong, Z.; Zhang, P.; He, C.; Qiu, X.; Liu, A.; Chen, L.; Chen, X.; Jing, X. Nano-composite of poly (L-lactide) and surface grafted hydroxyapatite: mechanical properties and biocompatibility. Biomaterials 2005, 26(32), 6296–6304. [Google Scholar] [CrossRef]
- Liu, Z.; Kwok, K.W.; Li, R.; Choy, C. Effects of coupling agent and morphology on the impact strength of high density polyethylene/CaCO3 composites. Polymer 2002, 43(8), 2501–2506. [Google Scholar] [CrossRef]
- Lewis, J.A. Colloidal processing of ceramics. Journal of the American Ceramic Society 2000, 83(10), 2341–2359. [Google Scholar] [CrossRef]
- Jakus, A.E.; Rutz, A.L.; Jordan, S.W.; Kannan, A.; Mitchell, S.M.; Yun, C.; Koube, K.D.; Yoo, S.C.; Whiteley, H.E.; Richter, C.-P. Hyperelastic “bone”: A highly versatile, growth factor–free, osteoregenerative, scalable, and surgically friendly biomaterial. Science Translational Medicine 2016, 8(358), 358ra127–358ra127. [Google Scholar] [CrossRef] [PubMed]
- Jakus, A.E.; Shah, R.N. Multi and mixed 3 D-printing of graphene-hydroxyapatite hybrid materials for complex tissue engineering. Journal of Biomedical Materials Research Part A 2017, 105(1), 274–283. [Google Scholar] [CrossRef]
- Iorshase, M.; Aliyu, A.; Utume, L.; Adamu, E.; Sani, M.; Salami, W. Impacts of high-dose gamma irradiation on the mechanical, structural and thermal properties of doum fiber reinforced High-Density Polyethylene (HDPe). Recent Advances in Natural Sciences 2025, 200. [Google Scholar] [CrossRef]
- Li, R.; Gu, Y.; Wang, Y.; Yang, Z.; Li, M.; Zhang, Z. Effect of particle size on gamma radiation shielding property of gadolinium oxide dispersed epoxy resin matrix composite. Materials Research Express 2017, 4(3), 035035. [Google Scholar] [CrossRef]
- Ali, M.R.; Chowdhury, M.A.; Ali, M.O. Gamma irradiation effects on mechanical and environmental performance of Al2O3-TiO2 reinforced jute-glass hybrid composites. Next Materials 2026, 10, 101416. [Google Scholar] [CrossRef]
- Suljovrujić, E.; Ignjatović, N.; Uskoković, D. Gamma irradiation processing of hydroxyapatite/poly-L-lactide composite biomaterial. Radiation Physics and Chemistry 2003, 67(3-4), 375–379. [Google Scholar] [CrossRef]
- Suljovrujić, E.; Ignjatović, N.; Uskoković, D.; Mitrić, M.; Mitrović, M.; Tomić, S. Radiation-induced degradation of hydroxyapatite/poly L-lactide composite biomaterial. Radiation Physics and Chemistry 2007, 76(4), 722–728. [Google Scholar] [CrossRef]
- Drozdov, A.D.; Dorfmann, A. The stress–strain response and ultimate strength of filled elastomers. Computational Materials Science 2001, 21(3), 395–417. [Google Scholar] [CrossRef]
- Zhou, S.; Zheng, X.; Yu, X.; Wang, J.; Weng, J.; Li, X.; Feng, B.; Yin, M. Hydrogen bonding interaction of poly (D, L-lactide)/hydroxyapatite nanocomposites. Chemistry of materials 2007, 19(2), 247–253. [Google Scholar] [CrossRef]
- Wang, M.; Bonfield, W. Chemically coupled hydroxyapatite–polyethylene composites: structure and properties. Biomaterials 2001, 22(11), 1311–1320. [Google Scholar] [CrossRef]
- Dhakal, H.N.; Zhang, Z. Polymer Matrix Composites: Moisture Effects and Dimensional Stability. In Wiley Encyclopedia of Composites; pp. 1–7.
- Senatov, F.; Zadorozhnyy, M.Y.; Niaza, K.; Medvedev, V.; Kaloshkin, S.; Anisimova, N.Y.; Kiselevskiy, M.; Yang, K.-C. Shape memory effect in 3D-printed scaffolds for self-fitting implants. European Polymer Journal 2017, 93, 222–231. [Google Scholar] [CrossRef]
- Rychter, P.; Pamula, E.; Orchel, A.; Posadowska, U.; Krok-Borkowicz, M.; Kaps, A.; Smigiel-Gac, N.; Smola, A.; Kasperczyk, J.; Prochwicz, W. Scaffolds with shape memory behavior for the treatment of large bone defects. Journal of Biomedical Materials Research Part A 2015, 103(11), 3503–3515. [Google Scholar] [CrossRef] [PubMed]
- Bao, M.; Lou, X.; Zhou, Q.; Dong, W.; Yuan, H.; Zhang, Y. Electrospun biomimetic fibrous scaffold from shape memory polymer of PDLLA-co-TMC for bone tissue engineering. ACS Applied Materials & Interfaces 2014, 6(4), 2611–2621. [Google Scholar]
- Huang, N.; Deng, H.; Liu, B.; Wang, D.; Zhao, Z. Features and futures of X-ray free-electron lasers. The Innovation 2021, 2(2). [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Omi, T.; Akai, H.; Kubota, Y.; Takahashi, Y.; Suzuki, Y.; Hirata, Y.; Yamamoto, K.; Yukawa, R.; Horiba, K. Element selectivity in second-harmonic generation of GaFeO 3 by a soft-X-ray free-electron laser. Physical Review Letters 2018, 120(22), 223902. [Google Scholar] [CrossRef]
- Fogarty, A.C.; Laage, D. Water dynamics in protein hydration shells: The molecular origins of the dynamical perturbation. The Journal of Physical Chemistry B 2014, 118(28), 7715–7729. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, Y.-Y.; Jiang, S.; Yan, W.; Li, S.; Zheng, H.; Lu, J.-B.; Hu, H.-S.; Yang, J.; Zhang, W. Experimental determination of structural motifs of interference-free water undecamer cluster (H2O) 11. Nature Communications 2025. [Google Scholar] [CrossRef]
- Zong; Nebgen, B.R.; Lin, S.-C.; Spies, J.A.; Zuerch, M. Emerging ultrafast techniques for studying quantum materials. Nature Reviews Materials 2023, 8(4), 224–240. [Google Scholar] [CrossRef]
- Zhang, Y.-Y.; Zheng, H.; Wang, T.; Jiang, S.; Yan, W.; Wang, C.; Zhao, Y.; Lu, J.-B.; Hu, H.-S.; Yang, J. Spectroscopic and Theoretical Identifications of Two Structural Motifs of (H2O) 10 Cluster. The Journal of Physical Chemistry Letters 2024, 15(11), 3055–3060. [Google Scholar] [CrossRef]
| Group Name | Geometry and composition | Irradiation |
|---|---|---|
| LAC | Lactoprene®, bulk-like cylindrical 12.7 × 25.4 mm | No |
| γ-LAC | γ-irradiated | |
| LTCP | Lactoprene®/β-TCP (60:40), bulk-like cylindrical 12.7 × 25.4 mm | No |
| γ-LTCP | γ-irradiated |
| LAC | γ-LAC | LTCP | γ-LTCP | |
|---|---|---|---|---|
| Mn | 156,830 ± 2,082 | 73,705 ± 314 | 115,505 ± 4,041 | 77,018 ± 3,749 |
| Mw (g/mol) | 253,406 ± 491 | 142,462 ± 907 | 205,690 ± 1,465 | 146,140 ± 707 |
| PDI | 1.61 ± 0.02 | 1.93 ± 0.01 | 1.79 ± 0.05 | 1.90 ± 0.08 |
| Condition | Group | |||
|---|---|---|---|---|
| LAC | LTCP | γ-LAC | γ-LTCP | |
| Dry | 1.1 ± 0.5 | 4.3 ± 0.2 | 23.9 ± 2.5 | 16.8 ± 0.5 |
| 25°C – 30 min | 2.8 ± 0.1 | 7.1 ± 0.8 | 27.5 ± 1.4 | 14.5 ± 3 |
| 25°C – 1 d | 3.1 ± 1.2 | 7.7 ± 0.3 | 36.5 ± 2.4 | 15.8 ± 1.6 |
| 25°C – 7 d | 11.8 ± 1.5 | ND | 37.0 ± 2.0 | 30.1 ± 2.7 |
| 37°C – 30 min | 2.2 ± 0.5 | 6.4 ± 0.5 | 25.3 ± 0.9 | 1.9 ± 0.3 |
| 37°C – 1 d | 18.1 ± 0.1 | ND | 23.5 ± 1.9 | 3.95 ± 1.5 |
| 37°C – 7 d | ND | ND | ND | ND |
| Condition | Group | |||
|---|---|---|---|---|
| LAC | LTCP | γ-LAC | γ-LTCP | |
| Dry | 39.2 ± 0.5 | 32.7 ± 0.3 | 37.8 ± 0.6 | 35.7 ± 0.3 |
| 25°C – 30 min | 29.2 ± 0.6 | 24.3 ± 1.8 | 34.4 ± 1.2 | 28.4 ± 2.2 |
| 25°C – 1 d | 33.8 ± 0.9 | 26.2 ± 0.4 | 34.3 ± 0.7 | 30.1 ± 1 |
| 25°C – 7 d | 29.0 ± 0.8 | ND | 33.6 ± 0.6 | 31.4 ± 0.2 |
| 37°C – 30 min | 36.4 ± 0.6 | 26.3 ± 0.6 | 33.5 ± 1.0 | 23.7 ± 0.9 |
| 37°C – 1 d | 31.1 ± 0.6 | ND | 33.4 ± 0.7 | 16.4 ± 0.3 |
| 25°C – 7 d | 27.4 ± 0.7 | ND | 34.4 ± 2.3 | 14.6 ± 0.7 |
| Testing conditions | Group | Compressive modulus (MPa) | Max stress (MPa) | Yield Stress (MPa) | Energy dissipated (MJ/m3) |
|---|---|---|---|---|---|
| Dry, RT | LAC | 630.7 ± 40.6 | 60.5 ± 3.1 | 26.7 ± 3.5 | 18.4 ± 0.7 |
| γ-LAC | 861.0 ± 36.1 | 61.3 ± 2.3 | 41.3 ± 0.6 | 19.2 ± 0.9 | |
| LTCP | 1142.7 ± 93.9 | 89.2 ± 2.9 | 41.6 ± 2.0 | 27.1 ± 1.0 | |
| γ-LTCP | 795.2 ± 43.4 | 74.8 ± 1.4 | 32.6 ± 1.3 | 21.3 ± 0.3 | |
| Wet, 25°C | LAC | 806.3 ± 45.0 | 71.4 ± 4.7 | 43.2 ± 1.0 | 22.8 ± 1.7 |
| γ-LAC | 867.9 ± 75.1 | 58.0 ± 6.7 | 43.4 ± 3.0 | 20.9 ± 0.4 | |
| LTCP | 1177.2 ± 69.5 | 95.1 ± 5.9 | 43.7 ± 2.0 | 29.1 ± 1.1 | |
| γ-LTCP | 965.6 ± 65.4 | 83.7 ± 2.7 | 38.5 ± 2.2 | 23.9 ± 1.1 | |
| Water saturated, 25°C | LAC | 432.2 ± 22.6 | 66.1 ± 1.6 | ND | 18.2 ± 0.4 |
| γ-LAC | 391.5 ± 43.0 | 61.9 ± 2.8 | ND | 18.5 ± 0.8 | |
| LTCP | 219.8 ± 10.3 | 46.9 ± 0.1 | ND | 10.7 ± 0.6 | |
| γ-LTCP | 278.3 ± 2.2 | 55.8 ± 1.5 | ND | 14.2 ± 0.3 | |
| Wet, 37°C | LAC | 55.0 ± 11.2 | 24.1 ± 3.9 | ND | 5.4 ± 0.9 |
| γ-LAC | 43.7 ± 8.8 | 22.0 ± 5.8 | ND | 4.6 ± 1.1 | |
| LTCP | 77.3 ± 9.3 | 33.2 ± 1.8 | ND | 5.7 ± 0.4 | |
| γ-LTCP | 26.0 ± 5.7 | 17.2 ± 3.2 | ND | 2.9 ± 0.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





