Submitted:
23 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
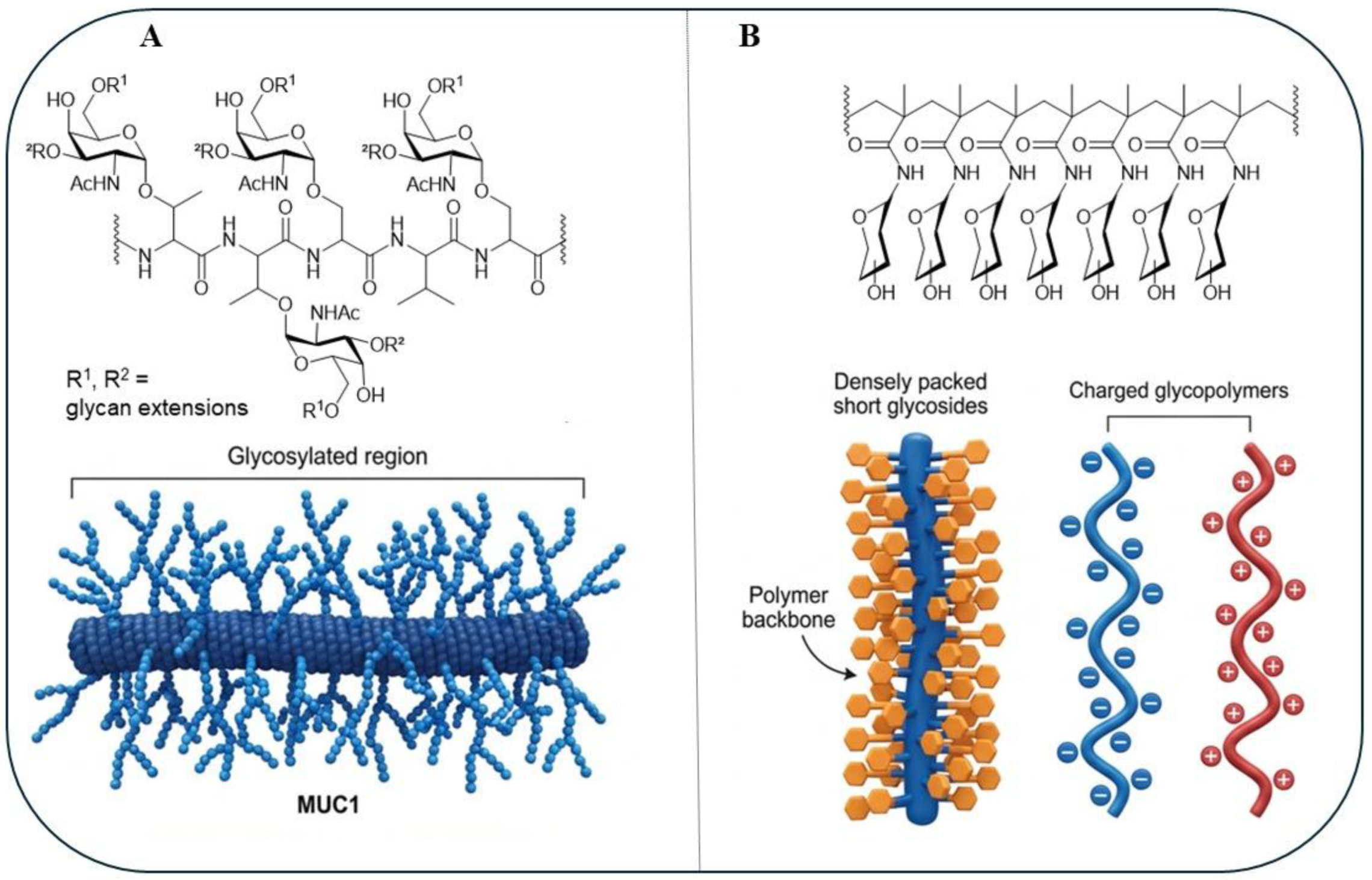
1. Introduction
2. Controlled Synthesis of Glycopolymers
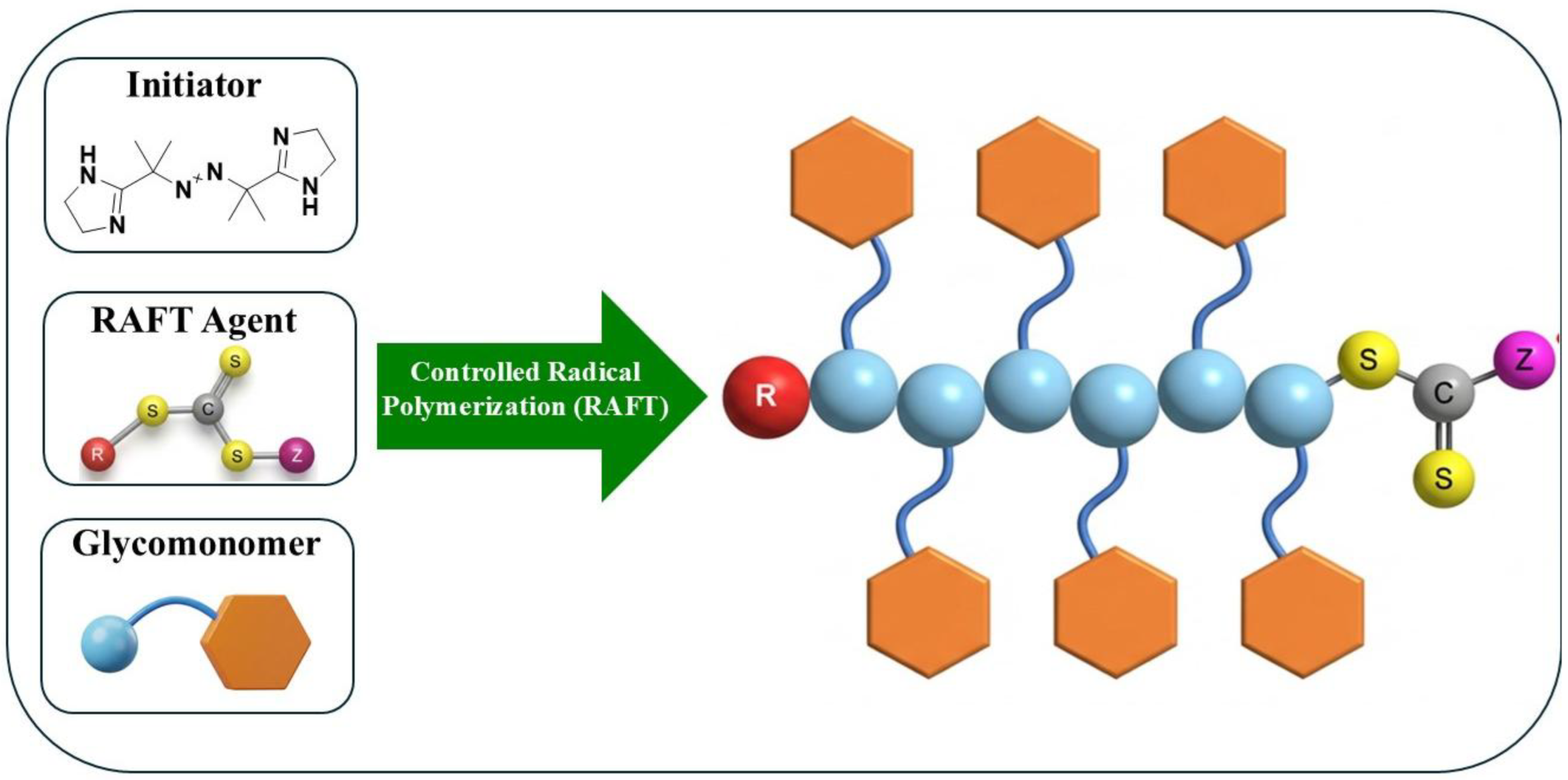
2.1. The RAFT Polymerization Route
2.2. Surface Modification Strategies: Anchoring Glycopolymers to Biomaterials
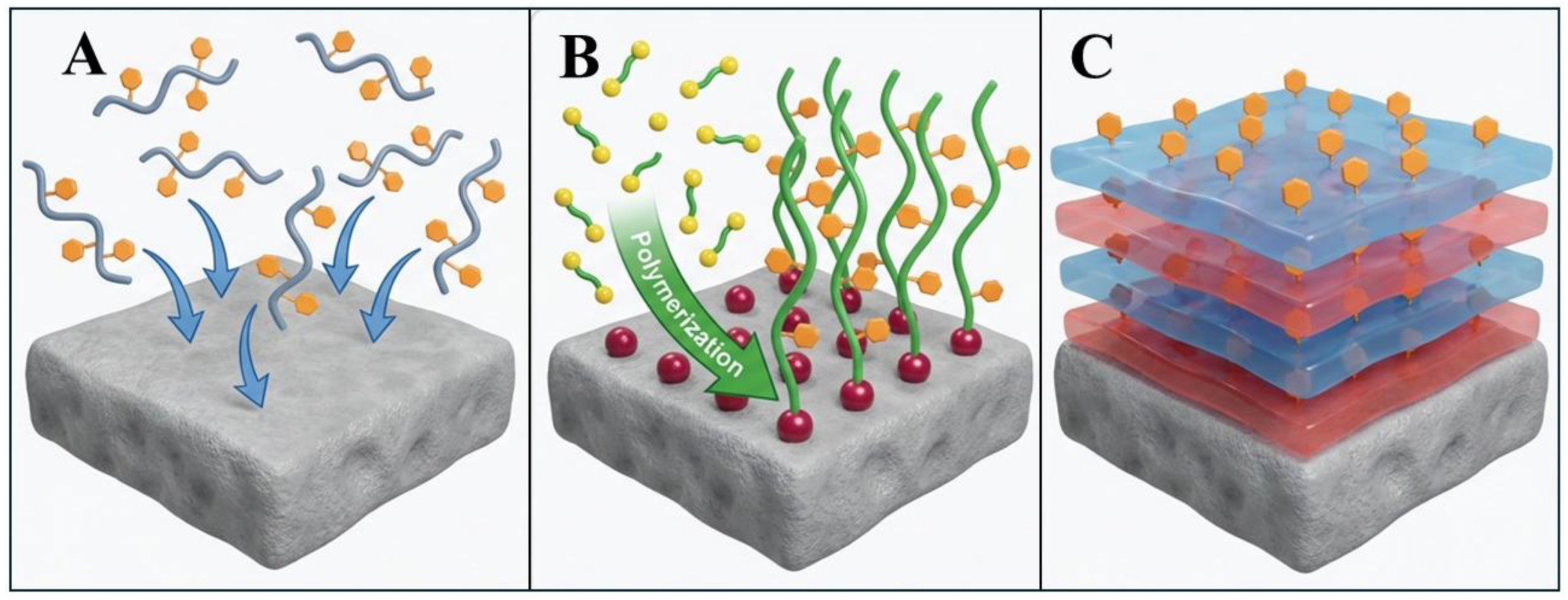
2.2.1. Grafting-to and Grafting-from Approaches
2.2.2. Layer-by-Layer (LbL) Assembly
3. Application to Common Biomaterials
3.1. Tailoring the Biointerface: From Bio-Inert to Bio-Specific
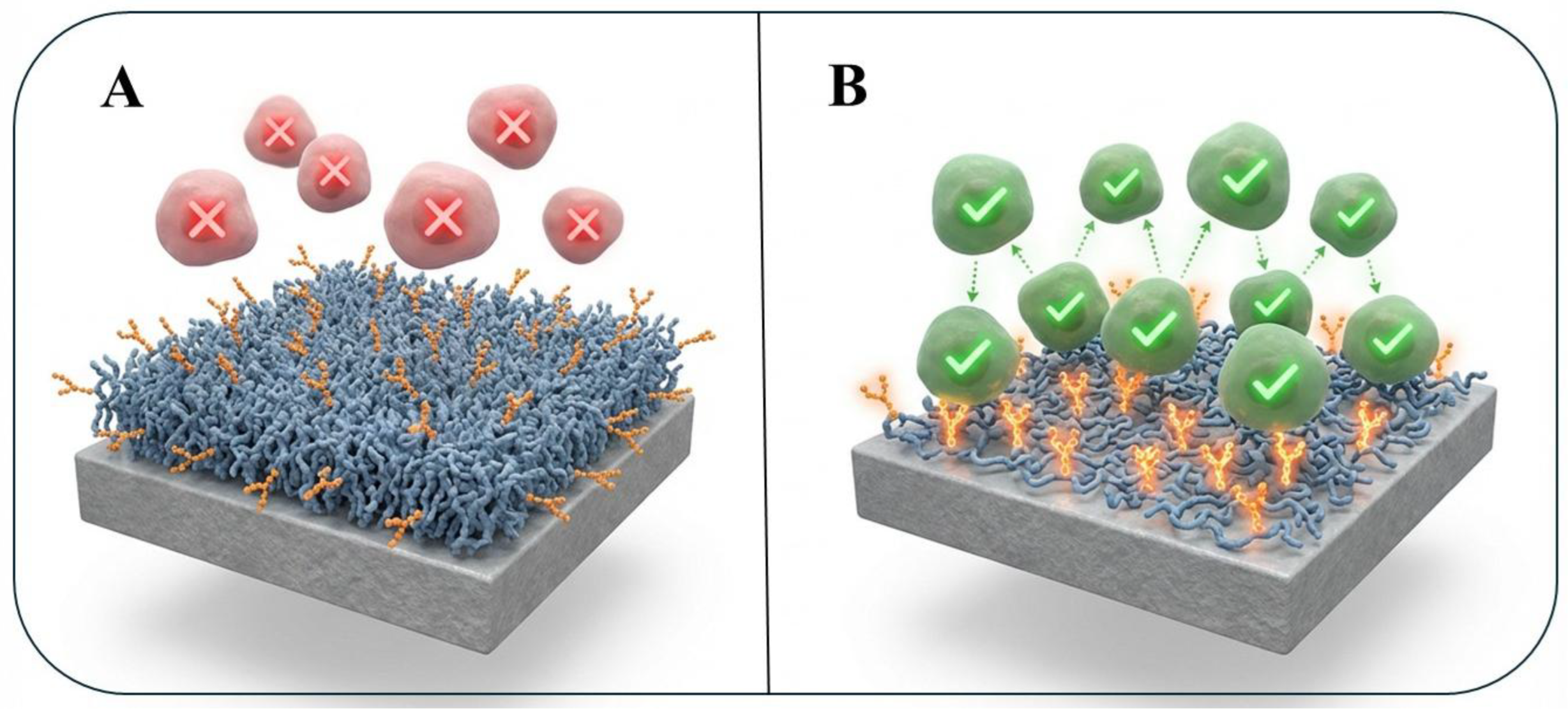
3.2. Creating Bio-Inert, Cell-Repellent Surfaces
3.3. Designing Bio-Specific, Cell-Adhesive Surfaces
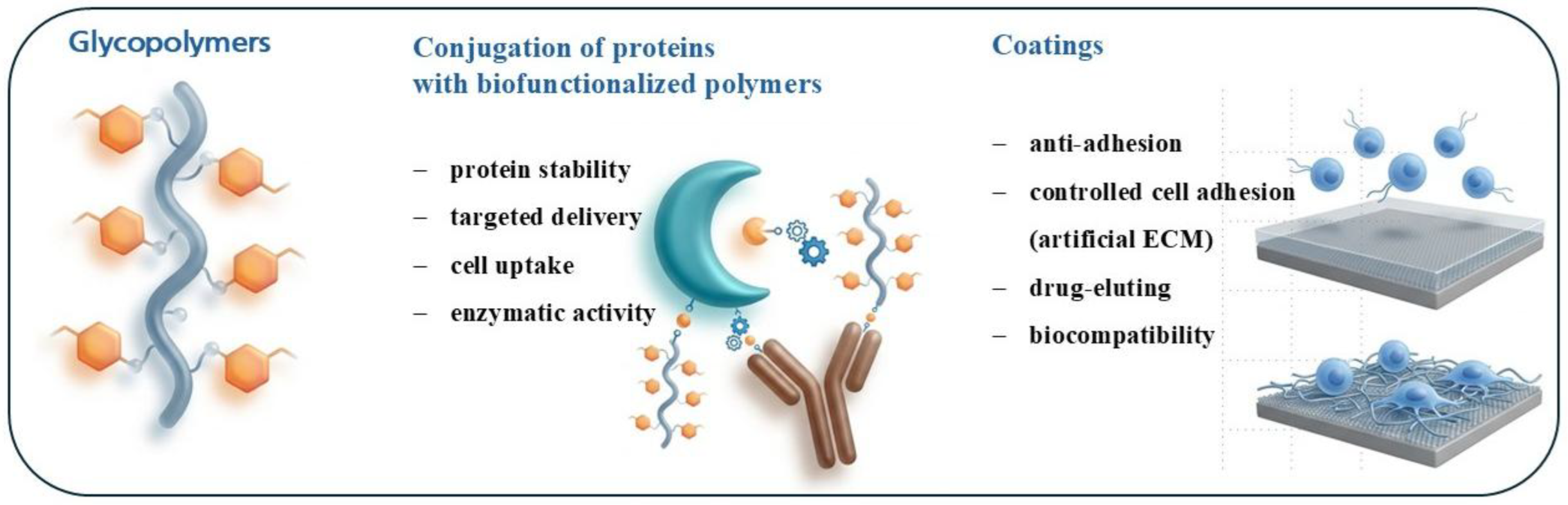
4. Glycopolymers as Platform Technology
4.1. Applications of Glycopolymer-Modified Biomaterials
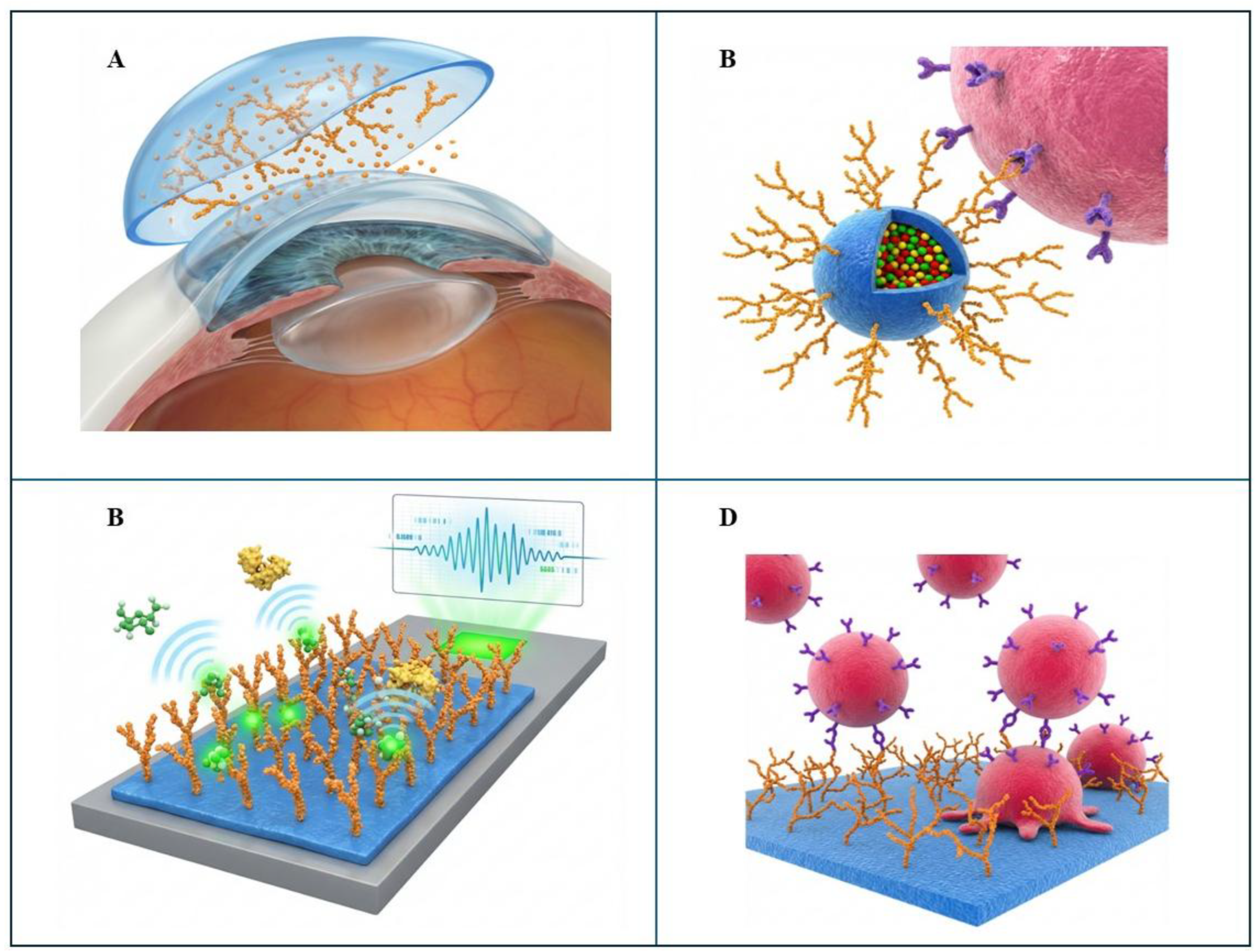
4.1.1. Advanced Contact Lenses
4.1.2. Targeted Drug and Gene Delivery
4.1.3. Biosensing and Diagnostics
4.1.4. Tissue Engineering and Regenerative Medicine
4.2. Soluble Glycopolymers as Therapeutics
5. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ishihara, K.; Shi, X.; Fukazawa, K.; Yamaoka, T.; Yao, G.; Wu, J. Y., Biomimetic-Engineered Silicone Hydrogel Contact Lens Materials. ACS Applied Bio Materials 2023, 6 (9), 3600-3616. [CrossRef]
- Anderson, J. M., Biological Responses to Materials. Annual Review of Materials Research 2001, 31 (Volume 31, 2001), 81-110.
- Hu, X.; Wang, T.; Li, F.; Mao, X., Surface modifications of biomaterials in different applied fields. RSC Advances 2023, 13 (30), 20495-20511.
- Liping, T.; Paul, T.; Wenjing, H., Surface Chemistry Influences Implant Biocompatibility. Current Topics in Medicinal Chemistry 2008, 8 (4), 270-280.
- Su, L.; Feng, Y.; Wei, K.; Xu, X.; Liu, R.; Chen, G., Carbohydrate-Based Macromolecular Biomaterials. Chemical Reviews 2021, 121 (18), 10950-11029. [CrossRef]
- Miura, Y.; Hoshino, Y.; Seto, H., Glycopolymer Nanobiotechnology. Chemical Reviews 2016, 116 (4), 1673-1692.
- Purcell, S. C.; Godula, K., Synthetic glycoscapes: addressing the structural and functional complexity of the glycocalyx. Interface Focus 2019, 9 (2), 20180080.
- Moniaux, N.; Escande, F.; Porchet, N.; Aubert, J.-P.; Batra, S. K., Structural organization and classification of the human mucin genes. Front. Biosci. (Landmark Ed) 2001, 6 (3), 1192-1206.
- Hattrup, C. L.; Gendler, S. J., Structure and Function of the Cell Surface (Tethered) Mucins. Annual Review of Physiology 2008, 70 (Volume 70, 2008), 431-457. [CrossRef]
- Rosencrantz, R. R. S., Joachim, Novel bioinspired polymeric material concepts and applications for functionalized contact lenses and keratoprostheses. Global Contact 2025, 3-25, 18-22.
- Spain, S. G.; Cameron, N. R., A spoonful of sugar: the application of glycopolymers in therapeutics. Polymer Chemistry 2011, 2 (1), 60-68. [CrossRef]
- Thalji, M. R.; Ibrahim, A. A.; Chong, K. F.; Soldatov, A.; Ali, G. A. M., Glycopolymer-Based Materials: Synthesis, Properties, and Biosensing Applications. Topics Curr Chem 2022, 380 (5).
- Vázquez-Dorbatt, V.; Lee, J.; Lin, E.-W.; Maynard, H. D., Synthesis of Glycopolymers by Controlled Radical Polymerization Techniques and Their Applications. ChemBioChem 2012, 13 (17), 2478-2487.
- Pramudya, I.; Chung, H., Recent progress of glycopolymer synthesis for biomedical applications. Biomaterials Science 2019, 7 (12), 4848-4872. [CrossRef]
- Hoffmann, M.; Bonda, L.; Fels, I.; Anhlan, D.; Hrincius, E.; Hermsen, D.; Ludwig, S.; Schelhaas, M.; Snyder, N. L.; Hartmann, L., Controlling the Sulfation Density of Glycosaminoglycan Glycopolymer Mimetics Enables High Antiviral Activity against SARS-CoV-2 and Reduces Anticoagulant Activity. Biomacromolecules 2025, 26 (8), 5169-5181.
- Polizzotti, B. D.; Kiick, K. L., Effects of Polymer Structure on the Inhibition of Cholera Toxin by Linear Polypeptide-Based Glycopolymers. Biomacromolecules 2006, 7 (2), 483-490.
- Jones, M. W.; Otten, L.; Richards, S. J.; Lowery, R.; Phillips, D. J.; Haddleton, D. M.; Gibson, M. I., Glycopolymers with secondary binding motifs mimic glycan branching and display bacterial lectin selectivity in addition to affinity. Chemical Science 2014, 5 (4), 1611-1616.
- Amani, H.; Arzaghi, H.; Bayandori, M.; Dezfuli, A. S.; Pazoki-Toroudi, H.; Shafiee, A.; Moradi, L., Controlling Cell Behavior through the Design of Biomaterial Surfaces: A Focus on Surface Modification Techniques. Advanced Materials Interfaces 2019, 6 (13), 1900572. [CrossRef]
- Ha, C.-S.; Gardella, J. A., Surface Chemistry of Biodegradable Polymers for Drug Delivery Systems. Chemical Reviews 2005, 105 (11), 4205-4232.
- Yang, Q.; Xu, Z.-K.; Dai, Z.-W.; Wang, J.-L.; Ulbricht, M., Surface Modification of Polypropylene Microporous Membranes with a Novel Glycopolymer. Chemistry of Materials 2005, 17 (11), 3050-3058.
- Sharon, N., Lectins: Carbohydrate-specific Reagents and Biological Recognition Molecules. Journal of Biological Chemistry 2007, 282 (5), 2753-2764. [CrossRef]
- Laederach, A.; Reilly, P. J., Modeling protein recognition of carbohydrates. Proteins: Structure, Function, and Bioinformatics 2005, 60 (4), 591-597. [CrossRef]
- Yu, K.; Lai, B. F. L.; Kizhakkedathu, J. N., Carbohydrate Structure Dependent Hemocompatibility of Biomimetic Functional Polymer Brushes on Surfaces. Advanced Healthcare Materials 2012, 1 (2), 199-213.
- Okada, M., Molecular design and syntheses of glycopolymers. Progress in Polymer Science 2001, 26 (1), 67-104.
- Miura, Y., Design and synthesis of well-defined glycopolymers for the control of biological functionalities. Polymer Journal 2012, 44 (7), 679-689.
- Moad, G.; Rizzardo, E.; Thang, S. H., Living Radical Polymerization by the RAFT Process. ChemInform 2005, 36 (40).
- Boyer, C.; Bulmus, V.; Davis, T. P.; Ladmiral, V.; Liu, J.; Perrier, S., Bioapplications of RAFT Polymerization. Chemical Reviews 2009, 109 (11), 5402-5436.
- Chiefari, J.; Chong, Y. K.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T. P. T.; Mayadunne, R. T. A.; Meijs, G. F.; Moad, C. L.; Moad, G.; Rizzardo, E.; Thang, S. H., Living Free-Radical Polymerization by Reversible Addition−Fragmentation Chain Transfer: The RAFT Process. Macromolecules 1998, 31 (16), 5559-5562.
- Moad, G.; Rizzardo, E.; Thang, S. H., Toward Living Radical Polymerization. Accounts of Chemical Research 2008, 41 (9), 1133-1142. [CrossRef]
- Bernard, J.; Hao, X.; Davis, T. P.; Barner-Kowollik, C.; Stenzel, M. H., Synthesis of Various Glycopolymer Architectures via RAFT Polymerization: From Block Copolymers to Stars. Biomacromolecules 2006, 7 (1), 232-238.
- Rosencrantz, S.; Tang, J. S. J.; Schulte-Osseili, C.; Böker, A.; Rosencrantz, R. R., Glycopolymers by RAFT Polymerization as Functional Surfaces for Galectin-3. Macromolecular Chemistry and Physics 2019, 220 (20), 1900293.
- Park, H.; Rosencrantz, R. R.; Elling, L.; Böker, A., Glycopolymer Brushes for Specific Lectin Binding by Controlled Multivalent Presentation of N-Acetyllactosamine Glycan Oligomers. Macromolecular Rapid Communications 2015, 36 (1), 45-54. [CrossRef]
- Min, E. H.; Ting, S. R. S.; Billon, L.; Stenzel, M. H., Thermo-responsive glycopolymer chains grafted onto honeycomb structured porous films via RAFT polymerization as a thermo-dependent switcher for lectin Concanavalin a conjugation. Journal of Polymer Science Part A: Polymer Chemistry 2010, 48 (15), 3440-3455.
- Korzhikov-Vlakh, V.; Sinitsyna, E.; Arkhipov, K.; Levit, M.; Korzhikova-Vlakh, E.; Tennikova, T. Improvement in Biological Performance of Poly(Lactic Acid)-Based Materials via Single-Point Surface Modification with Glycopolymer Surfaces [Online], 2024, p. 1008-1028. [CrossRef]
- Wang, Y.; Narain, R.; Liu, Y., Study of Bacterial Adhesion on Different Glycopolymer Surfaces by Quartz Crystal Microbalance with Dissipation. Langmuir 2014, 30 (25), 7377-7387.
- Tu, C.-X.; Gao, C.-Y., Recent Advances on Surface-modified Biomaterials Promoting Selective Adhesion and Directional Migration of Cells. Chinese Journal of Polymer Science 2021, 39 (7), 815-823.
- Barbey, R.; Klok, H.-A., Room Temperature, Aqueous Post-Polymerization Modification of Glycidyl Methacrylate-Containing Polymer Brushes Prepared via Surface-Initiated Atom Transfer Radical Polymerization. Langmuir 2010, 26 (23), 18219-18230. [CrossRef]
- Meng, X.-L.; Fang, Y.; Wan, L.-S.; Huang, X.-J.; Xu, Z.-K., Glycopolymer Brushes for the Affinity Adsorption of RCA120: Effects of Thickness, Grafting Density, and Epitope Density. Langmuir 2012, 28 (38), 13616-13623.
- Yang, Q.; Kaul, C.; Ulbricht, M., Anti-nonspecific Protein Adsorption Properties of Biomimetic Glycocalyx-like Glycopolymer Layers: Effects of Glycopolymer Chain Density and Protein Size. Langmuir 2010, 26 (8), 5746-5752.
- Yoshihara, E.; Nabil, A.; Iijima, M.; Ebara, M., A Comparative Study of “Grafting to” and “Grafting from” Conjugation Methods for the Preparation of Antibody-Temperature-Responsive Polymer Conjugates. ACS Omega 2024, 9 (20), 22043-22050.
- Le-Masurier, S. P.; Duong, H. T. T.; Boyer, C.; Granville, A. M., Surface modification of polydopamine coated particles via glycopolymer brush synthesis for protein binding and FLIM testing11Electronic supplementary information (ESI) available: NMR spectra for BIAzTC synthesis; GPC, ATR-FTIR, and NMR of PPFS using BIAzTC; TGA curves for polymer brush particles. Polymer Chemistry 2015, 6 (13), 2504-2511. [CrossRef]
- Rubio, N.; Au, H.; Leese, H. S.; Hu, S.; Clancy, A. J.; Shaffer, M. S. P., Grafting from versus Grafting to Approaches for the Functionalization of Graphene Nanoplatelets with Poly(methyl methacrylate). Macromolecules 2017, 50 (18), 7070-7079.
- L. M. Gonçalves, J.; J. Castanheira, E.; P. C. Alves, S.; Baleizão, C.; Farinha, J. P., Grafting with RAFT—gRAFT Strategies to Prepare Hybrid Nanocarriers with Core-shell Architecture. Polymers 2020, 12 (10), 2175.
- Edmondson, S.; Osborne, V. L.; Huck, W. T. S., Polymer brushes via surface-initiated polymerizations. Chemical Society Reviews 2004, 33 (1), 14-22.
- Lau, K. H. A.; Ren, C.; Park, S. H.; Szleifer, I.; Messersmith, P. B., An Experimental–Theoretical Analysis of Protein Adsorption on Peptidomimetic Polymer Brushes. Langmuir 2012, 28 (4), 2288-2298.
- Li, C.; Han, J.; Ryu, C. Y.; Benicewicz, B. C., A Versatile Method To Prepare RAFT Agent Anchored Substrates and the Preparation of PMMA Grafted Nanoparticles. Macromolecules 2006, 39 (9), 3175-3183.
- Ohno, K.; Ma, Y.; Huang, Y.; Mori, C.; Yahata, Y.; Tsujii, Y.; Maschmeyer, T.; Moraes, J.; Perrier, S., Surface-Initiated Reversible Addition–Fragmentation Chain Transfer (RAFT) Polymerization from Fine Particles Functionalized with Trithiocarbonates. Macromolecules 2011, 44 (22), 8944-8953.
- Tang, J. S. J.; Smaczniak, A. D.; Tepper, L.; Rosencrantz, S.; Aleksanyan, M.; Dähne, L.; Rosencrantz, R. R., Glycopolymer Based LbL Multilayer Thin Films with Embedded Liposomes. Macromolecular Bioscience 2022, 22 (4), 2270013.
- Boyer, C.; Bousquet, A.; Rondolo, J.; Whittaker, M. R.; Stenzel, M. H.; Davis, T. P., Glycopolymer Decoration of Gold Nanoparticles Using a LbL Approach. Macromolecules 2010, 43 (8), 3775-3784.
- Kempe, K.; Xiang, S. D.; Wilson, P.; Rahim, M. A.; Ju, Y.; Whittaker, M. R.; Haddleton, D. M.; Plebanski, M.; Caruso, F.; Davis, T. P., Engineered Hydrogen-Bonded Glycopolymer Capsules and Their Interactions with Antigen Presenting Cells. ACS Applied Materials & Interfaces 2017, 9 (7), 6444-6452.
- Zelikin, A. N., Drug Releasing Polymer Thin Films: New Era of Surface-Mediated Drug Delivery. ACS Nano 2010, 4 (5), 2494-2509. [CrossRef]
- Korogiannaki, M.; Samsom, M.; Schmidt, T. A.; Sheardown, H., Surface-Functionalized Model Contact Lenses with a Bioinspired Proteoglycan 4 (PRG4)-Grafted Layer. ACS Applied Materials & Interfaces 2018, 10 (36), 30125-30136.
- Lynge, M. E.; Baekgaard Laursen, M.; Hosta-Rigau, L.; Jensen, B. E. B.; Ogaki, R.; Smith, A. A. A.; Zelikin, A. N.; Städler, B., Liposomes as Drug Deposits in Multilayered Polymer Films. ACS Applied Materials & Interfaces 2013, 5 (8), 2967-2975. [CrossRef]
- Luo, J.; Li, X.; Dong, S.; Zhu, P.; Liu, W.; Zhang, S.; Du, J., Layer-by-layer coated hybrid nanoparticles with pH-sensitivity for drug delivery to treat acute lung infection. Drug Delivery 2021, 28 (1), 2460-2468.
- Jones, L.; Senchyna, M.; Glasier, M.-A.; Schickler, J.; Forbes, I.; Louie, D.; May, C., Lysozyme and Lipid Deposition on Silicone Hydrogel Contact Lens Materials. Eye & Contact Lens 2003, 29 (1), S75-S79. [CrossRef]
- Tighe, B. J., A Decade of Silicone Hydrogel Development: Surface Properties, Mechanical Properties, and Ocular Compatibility. Eye & Contact Lens 2013, 39 (1), 4-12.
- Zare, M.; Ghomi, E. R.; Venkatraman, P. D.; Ramakrishna, S., Silicone-based biomaterials for biomedical applications: Antimicrobial strategies and 3D printing technologies. Journal of Applied Polymer Science 2021, 138 (38), 50969.
- Ishihara, K.; Fukazawa, K.; Sharma, V.; Liang, S.; Shows, A.; Dunbar, D. C.; Zheng, Y.; Ge, J.; Zhang, S.; Hong, Y.; Shi, X.; Wu, J. Y., Antifouling Silicone Hydrogel Contact Lenses with a Bioinspired 2-Methacryloyloxyethyl Phosphorylcholine Polymer Surface. ACS Omega 2021, 6 (10), 7058-7067.
- Spadafora, A.; Korogiannaki, M.; Sheardown, H., Antifouling silicone hydrogel contact lenses via densely grafted phosphorylcholine polymers. Biointerphases 2020, 15 (4).
- Rickert, C. A.; Wittmann, B.; Fromme, R.; Lieleg, O., Highly Transparent Covalent Mucin Coatings Improve the Wettability and Tribology of Hydrophobic Contact Lenses. ACS Applied Materials & Interfaces 2020, 12 (25), 28024-28033. [CrossRef]
- Chen, S.; Jiang, S., An New Avenue to Nonfouling Materials. Advanced Materials 2008, 20 (2), 335-338.
- Kim, S.-H.; Kim, J.-H.; Akaike, T., Regulation of cell adhesion signaling by synthetic glycopolymer matrix in primary cultured hepatocyte. FEBS Letters 2003, 553 (3), 433-439.
- Yu, K.; Kizhakkedathu, J. N., Synthesis of Functional Polymer Brushes Containing Carbohydrate Residues in the Pyranose Form and Their Specific and Nonspecific Interactions with Proteins. Biomacromolecules 2010, 11 (11), 3073-3085.
- Zhu, J.; Marchant, R. E., Dendritic Saccharide Surfactant Polymers as Antifouling Interface Materials to Reduce Platelet Adhesion. Biomacromolecules 2006, 7 (4), 1036-1041.
- Yang, Q.; Xu, Z.-K.; Hu, M.-X.; Li, J.-J.; Wu, J., Novel Sequence for Generating Glycopolymer Tethered on a Membrane Surface. Langmuir 2005, 21 (23), 10717-10723.
- Imbia, A. S.; Ounkaew, A.; Zeng, H.; Liu, Y.; Narain, R., Stable Antifouling and Antibacterial Coating Based on Assembly of Copper-Phenolic Networks. ACS Applied Bio Materials 2025, 8 (1), 527-534.
- Trosan, P.; Tang, J. S. J.; Rosencrantz, R. R.; Daehne, L.; Smaczniak, A. D.; Staehlke, S.; Chea, S.; Fuchsluger, T. A., The Biocompatibility Analysis of Artificial Mucin-Like Glycopolymers. International Journal of Molecular Sciences 2023, 24 (18), 14150. [CrossRef]
- Wang, B.; Lin, Q.; Jin, T.; Shen, C.; Tang, J.; Han, Y.; Chen, H., Surface modification of intraocular lenses with hyaluronic acid and lysozyme for the prevention of endophthalmitis and posterior capsule opacification. RSC Advances 2015, 5 (5), 3597-3604.
- Schmidbauer, J. M.; Vargas, L. G.; Peng, Q.; Escobar-Gomez, M.; Werner, L.; Arthur, S. N.; Apple, D. J., Posterior Capsule Opacification. International Ophthalmology Clinics 2001, 41 (3), 109-131.
- Mammen, M.; Choi, S.-K.; Whitesides, G. M., Polyvalent Interactions in Biological Systems: Implications for Design and Use of Multivalent Ligands and Inhibitors. Angewandte Chemie International Edition 1998, 37 (20), 2754-2794.
- Ting, S. R. S.; Chen, G.; Stenzel, M. H., Synthesis of glycopolymers and their multivalent recognitions with lectins. Polymer Chemistry 2010, 1 (9), 1392-1412.
- Becer, C. R., The Glycopolymer Code: Synthesis of Glycopolymers and Multivalent Carbohydrate–Lectin Interactions. Macromolecular Rapid Communications 2012, 33 (9), 742-752.
- Cairo, C. W.; Gestwicki, J. E.; Kanai, M.; Kiessling, L. L., Control of Multivalent Interactions by Binding Epitope Density. Journal of the American Chemical Society 2002, 124 (8), 1615-1619.
- Thapa, B.; Kumar, P.; Zeng, H.; Narain, R., Asialoglycoprotein Receptor-Mediated Gene Delivery to Hepatocytes Using Galactosylated Polymers. Biomacromolecules 2015, 16 (9), 3008-3020.
- Huang, X.; Leroux, J.-C.; Castagner, B., Well-Defined Multivalent Ligands for Hepatocytes Targeting via Asialoglycoprotein Receptor. Bioconjugate Chemistry 2017, 28 (2), 283-295.
- Dhande, Y. K.; Wagh, B. S.; Hall, B. C.; Sprouse, D.; Hackett, P. B.; Reineke, T. M., N-Acetylgalactosamine Block-co-Polycations Form Stable Polyplexes with Plasmids and Promote Liver-Targeted Delivery. Biomacromolecules 2016, 17 (3), 830-840. [CrossRef]
- Santo, D.; Cordeiro, R. A.; Mendonça, P. V.; Serra, A. C.; Coelho, J. F. J.; Faneca, H., Glycopolymers Mediate Suicide Gene Therapy in ASGPR-Expressing Hepatocellular Carcinoma Cells in Tandem with Docetaxel. Biomacromolecules 2023, 24 (3), 1274-1286.
- Yang, W.; Mou, T.; Shao, G.; Wang, F.; Zhang, X.; Liu, B., Copolymer-Based Hepatocyte Asialoglycoprotein Receptor Targeting Agent for SPECT. Journal of Nuclear Medicine 2011, 52 (6), 978-985. [CrossRef]
- D’Souza, A. A.; Devarajan, P. V., Asialoglycoprotein receptor mediated hepatocyte targeting — Strategies and applications. Journal of Controlled Release 2015, 203, 126-139.
- Wu, J.; Nantz, M. H.; Zern, M. A., Targeting hepatocytes for drug and gene delivery: emerging novel approaches and applications. Front. Biosci. (Landmark Ed) 2002, 7 (4), 717-725.
- Gu, Y.; Liu, B.; Liu, Q.; Hang, Y.; Wang, L.; Brash, J. L.; Chen, G.; Chen, H., Modular Polymers as a Platform for Cell Surface Engineering: Promoting Neural Differentiation and Enhancing the Immune Response. ACS Applied Materials & Interfaces 2019, 11 (51), 47720-47729.
- Trinadh, M.; Kannan, G.; Rajasekhar, T.; Sesha Sainath, A. V.; Dhayal, M., Synthesis of glycopolymers at various pendant spacer lengths of glucose moiety and their effects on adhesion, viability and proliferation of osteoblast cells. RSC Advances 2014, 4 (70), 37400-37410. [CrossRef]
- Kaplan, A. V.; Baim, D. S.; Smith, J. J.; Feigal, D. A.; Simons, M.; Jefferys, D.; Fogarty, T. J.; Kuntz, R. E.; Leon, M. B., Medical Device Development. Circulation 2004, 109 (25), 3068-3072.
- Windecker, S.; Fraser, A. G.; Szymanski, P.; Gilard, M.; Lüscher, T. F.; Abid, L.; Brennan, J.; Byrne, R.; Crotti, L.; Drossart, I.; Franke, J.; Cossellu, M. G.; Kirtane, A. J.; Kurucova, J.; Krucoff, M.; McGauran, G.; Myers, P. O.; O’Connor, D. B.; Parma, R.; Piscoi, P.; Rao, A.; Rappagliosi, A.; Stefanini, G.; Samset, E.; Vincent, A.; von Bardeleben, R. S.; Weidinger, F., Priorities for medical device regulatory approval: a report from the European Society of Cardiology Cardiovascular Round Table. European Heart Journal 2025, 46 (16), 1469-1479.
- Heneghan, C.; Thompson, M., Rethinking medical device regulation. Journal of the Royal Society of Medicine 2012, 105 (5), 186-188.
- Guillon, M., Are Silicone Hydrogel Contact Lenses More Comfortable Than Hydrogel Contact Lenses? Eye & Contact Lens 2013, 39 (1), 86-92.
- Cheng, Q.; Peng, Y.-Y.; Asha, A. B.; Zhang, L.; Li, J.; Shi, Z.; Cui, Z.; Narain, R., Construction of antibacterial adhesion surfaces based on bioinspired borneol-containing glycopolymers. Biomaterials Science 2022, 10 (7), 1787-1794.
- Cheng, W.; Yang, C.; Ding, X.; Engler, A. C.; Hedrick, J. L.; Yang, Y. Y., Broad-Spectrum Antimicrobial/Antifouling Soft Material Coatings Using Poly(ethylenimine) as a Tailorable Scaffold. Biomacromolecules 2015, 16 (7), 1967-1977. [CrossRef]
- Ariga, K.; McShane, M.; Lvov, Y. M.; Ji, Q.; Hill, J. P., Layer-by-layer assembly for drug delivery and related applications. Expert Opinion on Drug Delivery 2011, 8 (5), 633-644.
- Qin, Q.; Lang, S.; Huang, X., Synthetic linear glycopolymers and their biological applications. Journal of Carbohydrate Chemistry 2021, 40 (1-3), 1-44. [CrossRef]
- Van Bruggen, C.; Hexum, J. K.; Tan, Z.; Dalal, R. J.; Reineke, T. M., Nonviral Gene Delivery with Cationic Glycopolymers. Accounts of Chemical Research 2019, 52 (5), 1347-1358.
- Nakayama, Y., Hyperbranched Polymeric “Star Vectors” for Effective DNA or siRNA Delivery. Accounts of Chemical Research 2012, 45 (7), 994-1004. [CrossRef]
- Bellato, F.; Bellio, G.; Asnicar, D.; Catania, R.; Pecchielan, L.; Marcenta, L.; Zanon, M.; Cielo, A.; Zainotto, M.; Pirazzini, M.; Ferrarini, A.; Mantovani, G.; Mastrotto, F., Mannose Targeting and Hydrophobic Tuning of Polycationic Vectors for Efficient Immunostimulatory CpG Delivery. ACS Applied Nano Materials 2025, 8 (47), 22637-22656.
- Ahmed, M.; Narain, R., The effect of polymer architecture, composition, and molecular weight on the properties of glycopolymer-based non-viral gene delivery systems. Biomaterials 2011, 32 (22), 5279-5290.
- Singhsa, P.; Diaz-Dussan, D.; Manuspiya, H.; Narain, R., Well-Defined Cationic N-[3-(Dimethylamino)propyl]methacrylamide Hydrochloride-Based (Co)polymers for siRNA Delivery. Biomacromolecules 2018, 19 (1), 209-221.
- Wang, W.; Guo, H.; Geng, J.; Zheng, X.; Wei, H.; Sun, R.; Tian, Z., Tumor-released Galectin-3, a Soluble Inhibitory Ligand of Human NKp30, Plays an Important Role in Tumor Escape from NK Cell Attack. Journal of Biological Chemistry 2014, 289 (48), 33311-33319.
- dos Santos, S. N.; Sheldon, H.; Pereira, J. X.; Paluch, C.; Bridges, E. M.; El-Cheikh, M. C.; Harris, A. L.; Bernardes, E. S., Galectin-3 acts as an angiogenic switch to induce tumor angiogenesis via Jagged-1/Notch activation. Oncotarget 2017, 8 (30), 49484-49501.
- Bumba, L.; Laaf, D.; Spiwok, V.; Elling, L.; Křen, V.; Bojarová, P., Poly-N-Acetyllactosamine Neo-Glycoproteins as Nanomolar Ligands of Human Galectin-3: Binding Kinetics and Modeling. International Journal of Molecular Sciences 2018, 19 (2), 372. [CrossRef]
- Miura, Y.; Nagao, M.; Matsumoto, H., De novo designed glycopolymer by precise polymer synthesis. Chemistry Letters 2023, 53 (2).
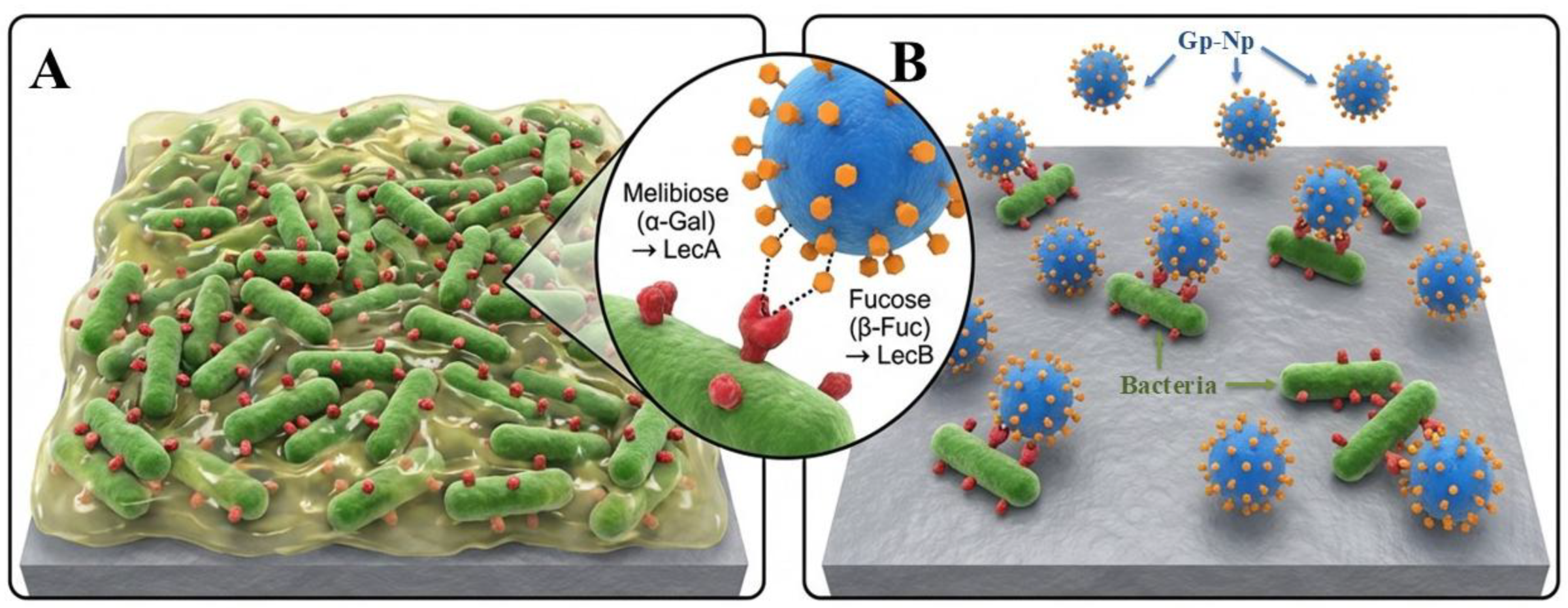
- Gerling-Driessen, U. I. M.; Hoffmann, M.; Schmidt, S.; Snyder, N. L.; Hartmann, L., Glycopolymers against pathogen infection. Chemical Society Reviews 2023, 52 (8), 2617-2642.
- Terada, Y.; Futamata, M.; Tsutsui, K.; Aoki, H., Evaluation of Preferential Cytokine Adsorption onto Biosensing Surface Modified with Glycopolymer. Biosensors 2025, 15 (3), 178.
- Xia, X.; Hu, Y.; Jiang, N.; Yetisen, A. K., Optical Contact Lenses Biosensors. ACS Sensors 2025, 10 (10), 7231-7251.
- Sugiaman, V. K.; Jeffrey; Naliani, S.; Pranata, N.; Djuanda, R.; Saputri, R. I., Polymeric Scaffolds Used in Dental Pulp Regeneration by Tissue Engineering Approach. Polymers 2023, 15 (5), 1082.
- Gerberich, B. G.; Bhatia, S. K., Tissue scaffold surface patterning for clinical applications. Biotechnology Journal 2013, 8 (1), 73-84.
- Putri, N. R. E.; Chen, H.; Kawazoe, N.; Rose, F. R. A. J.; Wildman, R. D.; Chen, G., Tailoring cell behaviour by surface micropatterning and interconnected porous structure of gelatin/nano-silica/PLGA 3D composite scaffold for bone tissue engineering. RSC Advances 2025, 15 (35), 28581-28591.
- Stöbener, D. D.; Uckert, M.; Cuellar-Camacho, J. L.; Hoppensack, A.; Weinhart, M., Ultrathin Poly(glycidyl ether) Coatings on Polystyrene for Temperature-Triggered Human Dermal Fibroblast Sheet Fabrication. ACS Biomaterials Science & Engineering 2017, 3 (9), 2155-2165. [CrossRef]
- Ali, A. A.; Al Bostami, R. D.; Al-Othman, A., Nanogel-based composites for bacterial antibiofilm activity: advances, challenges, and prospects. RSC Advances 2024, 14 (15), 10546-10559. [CrossRef]
- Rosencrantz, S.; Tang, J. S. J.; Koenig, K.; Chea, S.; Rosencrantz, R. R., Glyco-Nanogels for Modulating Pseudomonas aeruginosa Biofilm. Macromolecular Rapid Communications n/a (n/a), e00807.
- Ma, R.; Lai, Y.-x.; Li, L.; Tan, H.-l.; Wang, J.-l.; Li, Y.; Tang, T.-t.; Qin, L., Bacterial inhibition potential of 3D rapid-prototyped magnesium-based porous composite scaffolds–an in vitro efficacy study. Scientific Reports 2015, 5 (1), 13775. [CrossRef]
- Jiang, Y.; Nie, C.; Zheng, B.; Khatri, V.; Puccio, D.; Long, Y.; Dimde, M.; Haag, R.; Bhatia, S., Heteromultivalent Nanogels as Highly Potent Inhibitors of Pseudomonas Aeruginosa. Angewandte Chemie International Edition 2025, 64 (52), e13121. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



