1. Introduction
The global population continues to expand at an unprecedented rate, and contemporary food production systems are increasingly unable to meet this rising demand. According to the United Nations, the global population is projected to reach approximately 8.5 billion by 2030 and 9.7 billion by 2050, before peaking at around 10.4 billion in the 2080s [
1]. This sustained demographic growth inevitably increases the demand for food, disrupting the equilibrium between existing production capacity and global requirements. Furthermore, environmental factors, including global warming and natural disasters, have diminished agricultural yields, exacerbating the instability of the global food supply [
2].
Over the past several decades, agricultural production has intensified to meet the rapidly growing food demand, resulting in a substantial increase in the environmental burden. Current food systems face critical sustainability challenges, with greenhouse gas emissions and water pollution substantially affecting the global environment [
3,
4]. There is a detrimental interplay between population growth and environmental issues. Specifically, efforts to intensify agriculture in order to boost yields often result in environmental pollution and ecosystem degradation, creating a vicious cycle that undermines the long-term sustainability of food production. To address these challenges, there is an urgent need to develop innovative food production technologies that go beyond traditional agricultural frameworks.
Recently, “cellular agriculture” has attracted considerable attention as a novel production paradigm capable of tackling these issues. This field involves the indoor production of cellular biomass or specific extracts for food and industrial applications by culturing cells derived from living organisms [
5]. Cultured meat produced from animal cells has seen substantial progress in research and development, gaining considerable social recognition. Animal cellular agriculture originated in 2002 when the feasibility of a culture system for goldfish muscle protein was evaluated as a potential food source for astronauts [
6]. A major milestone followed in 2013, when Dr. Mark Post unveiled the world’s first cultured humburger produced from bovine myogenic stem cells in the Netherlands [
7].
In plant cell research, it has been demonstrated that culturing cells of
Coffea arabica and roasting the harvested biomass can produce a beverage base. Although it exhibits fewer nutty and earthy notes characteristic of the conventional coffee beans, it possesses distinct aromatic profiles, such as caramelized sugar and caramel, resulting from the high sugar content of the raw material [
8,
9]. Furthermore, cell cultures of
Rubus species (e.g., cloudberries and lingonberries) have been reported to contain higher levels of protein, dietary fiber, and essential amino acids than their whole-fruit counterparts, offering both high nutritional value and favorable palatability. Although nutrient-dense foods such as macroalgae are typically classified as “superfoods,” these findings suggest that cellular agriculture could serve as a novel and sustainable source of such superfoods [
10].
Ensuring product safety is a critical challenge in the social implementation of these technologies as viable food production systems. Currently, definitive international standards and regulatory frameworks for the safety assessment of cellular agricultural products, including cultured meat, have yet to be finalized; instead, regulatory authorities in various countries are in the process of developing their own independent systems. For example, the U.S. Food and Drug Administration (FDA) proposed a joint regulatory framework with the USDA Food Safety and Inspection Service (USDA-FSIS). The FDA evaluates the entire process, from cell collection to proliferation and differentiation, requiring the submission of rigorous data concerning cell line stability, all culture medium components, impurities generated during manufacturing, and comprehensive hazard analyses [
11]. Similarly, the Centre for Food Safety (CFS) in Hong Kong has established more specific management criteria for media components. The CFS requires explicit identification and purity standards for antimicrobials, growth factors, and modifying agents added to the media. Furthermore, it mandates toxicological evidence to demonstrate that the residual levels of any non-food-grade components pose no harm to human health [
12].
These regulatory trends highlight the challenges of directly repurposing reagent-grade medium components, traditionally used in tissue engineering and biopharmaceutical development, for food production. To address these issues, we developed and validated a novel, cost-effective, and safe culture medium composed entirely of food-grade ingredients. This study aimed to reconcile cost reduction with safety assurance in plant cellular agriculture, thereby establishing a feasible pathway for commercial scale-up and social implementation.
2. Materials and Methods
In the present study, a food-grade (FG) basal medium was formulated by substituting the essential components of conventional plant cell culture media with food-grade raw materials. Subsequently, plant cell cultures were performed using the developed medium, and their performance was evaluated by comparing callus fresh weight and gene expression profiles with those of conventional media.
The N6 medium, developed by Chu et al. in 1975, is a widely recognized standard basal medium for plant cell culture [
13]. It is noteworthy that the “N6CI medium” used as the positive control in this study is not the unmodified N6 basal medium ; rather, it is an in-house modification of the N6 medium, supplemented with casamino acids and proline to optimize rice callus induction. Hereafter, this variant is referred to as “N6CI medium.” The addition of casamino acids and proline is known to significantly enhance the proliferation and regeneration efficiency of cultured rice cells [
14] and has been established as the standard optimized protocol in our laboratory. Therefore, to rigorously validate the practical utility of the FG medium as a viable alternative, its performance was compared with that of the optimized N6CI medium, rather than the standard N6 formulation.
Basal media provide a comprehensive set of nutrients essential for plant cell proliferation, categorized as macronutrients, micronutrients, carbon sources, vitamins, and amino acids (
Table 1). For practical culture applications, these basal formulations were supplemented with appropriate types and concentrations of phytohormones tailored to the specific plant species.
2.1. Design of FG-N6CI (Food-Grade Basal Medium)
In this experiment, the FG-N6CI basal medium was designed based on the composition of the conventional N6CI basal medium. The components of the FG-N6CI basal medium were selected by substituting traditional reagents with either food-grade additives or food ingredients or by omitting them entirely, depending on their regulatory status.
The suitability of each N6CI component as a food additive was verified according to “Japan’s Specifications and Standards for Food Additives, 10th Ed. (2024)”[
15] and is listed by the Japan Food Chemical Research Foundation [
16,
17]. Based on this regulatory review, the components were categorized into three groups: “Authorized,” “Conditionally Authorized,” and “Unauthorized” (
Table 2).
The FG-N6CI basal medium was formulated using food additives for “Authorized” and “Conditionally Authorized” components, whereas “Unauthorized” components were either substituted with general food ingredients or excluded. However, the phytohormones added to the basal medium were of reagent grade.
Four components—MnSO4, H3BO3, KI, and Casamino acids—were identified as unauthorized food additives. Suitable food-grade alternatives were selected based on their physiological roles in the culture medium as follows:
MnSO
4 serves as the primary source of manganese in plant tissues. It was replaced with manganese-enriched edible yeast (Oriental Yeast Co., Ltd., Tokyo, Japan) as a food-grade manganese source. The composition of manganese yeast is provided in
Table A1.
H
3BO
3 functions as a boron source. For its substitution, a boron supplement, “Boron 3 mg Veg Capsules” (NOW Foods, Bloomingdale, IL, USA), containing calcium borogluconate (CaBG) as the primary ingredient, was utilized. Each capsule (370 mg) contained 3 mg boron (NOW Foods). Nutritional profiles of the supplements are presented in
Table A2.
KI provides both potassium and iodine. Commercially available kombu (kelp) powder was selected as a natural source of these elements. Typical kombu contains approximately 225 mg of iodine per 100 g [
18], and the required amount of kombu powder was calculated based on reported concentrations [
18].
Casamino acids, a mixture of amino acids and small peptides obtained from the acid hydrolysis of casein, provide essential nitrogen and amino acids in plant cell cultures. A previous study reported that substituting casamino acids with yeast extract during the vegetative growth of kuritake mushrooms resulted in improved yields and growth rates [
19]. Based on this precedent, the yeast extract “Hi-Max GL” (Fuji Foods Corporation, Yokohama, Japan) was selected as a food-grade alternative to supply both amino acids and nitrogen simultaneously. Its composition is provided in
Table A3.
2.2. Medium Composition
FG-N6CI (experimental group)
It comprised food additive-grade macro-elements (KNO
3, (NH
4)
2SO
4, MgSO
4·7H
2O, KH
2PO
4, CaCl
2·2H
2O), trace elements (FeSO
4·7H
2O, ZnSO
4·7H
2O, EDTA·2Na), and vitamins/amino acids (myo-inositol, niacin, vitamin B6, vitamin B1, glycine, and proline). Food ingredients were used as substitutes for the unauthorized components, as described above. Sucrose (sugar) and agar “Kanten Cook” (Ina Food Industry, Ina, Japan) were of food grade. The pH was adjusted to 5.8 with food-grade KOH (
Table 3) (detailed sourcing is presented in
Table A4).
It comprised reagent-grade chemicals. Basal salts were supplied as Chu’s N6 Basal Salt Mixture (PhytoTech LABS, Lenexa, KS, USA). The vitamins, amino acids (casamino acids, proline, and glycine), Sucrose and agar were of reagent grade. The pH was adjusted to 5.8 with reagent-grade KOH (
Table 4) (see
Table A5 for detailed sourcing).
The negative control consisted of water, plant hormones (2,4-D), and food-grade agar without added nutrients. No pH adjustments were performed.
All media contained 2.0 mg/L 2,4-D and were autoclaved at 121 °C, 220 kPa for 20 min.
2.3. Plant Materials and Culture
Rice seeds (Oryza sativa L. ‘Taichung 65’) were dehusked, sterilized with sodium hypochlorite solution/Tween 20 for 30 min, and then washed with sterile water. Seeds were plated on the N6CI medium to induce callus formation (28 °C, continuous light). Induced calli were separated from the seeds and subcultured on test media (FG-N6CI, N6CI, and NC) with a standardized initial fresh weight of 0.1 g per plate (n = 13).
2.4. Measurements
Callus fresh weight (FW) was measured every 7 days for 35 days. Statistical analysis was performed using a one-way ANOVA, followed by Tukey’s honest significant difference test (p < 0.05). For gene expression analysis, total RNA was extracted using the RNeasy Plant Mini Kit (Qiagen, Hilden, Germany), treated with DNase, and reverse-transcribed using SuperScript II (Thermo Fisher, Waltham, MA, USA). RT-qPCR was performed using TB Green Premix Ex Taq II (Takara Bio, Kusatsu, Japan), and the expression of
OsHDA710 and
OsTIR1 was normalized to
Actin7 expression. The primers used are listed in
Table A6.
2.5. Cost Calculation
The costs (JPY/L) were calculated based on the catalog prices of the components. For N6CI, calculations were made for both “mixture-based” (using commercial salt mix) and “reagent-based” (individual chemicals) preparations.
3. Results
3.1. Callus Proliferation
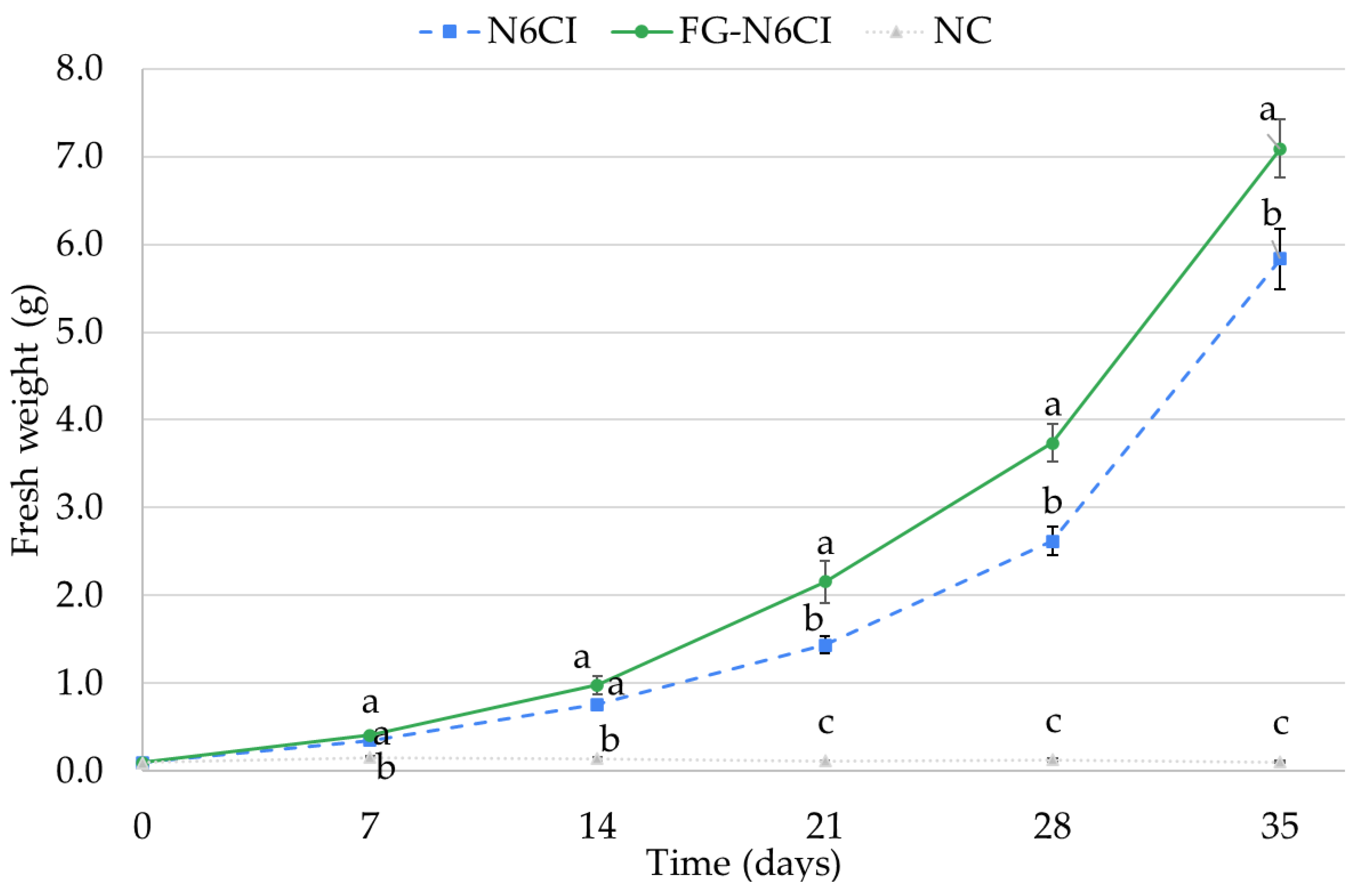
Figure 1 illustrates the time course of the changes in callus fresh weight under each condition. In the NC medium, almost no increase in callus weight was observed throughout the 35-day cultivation period. In contrast, both the FG-N6CI and N6CI media exhibited remarkable increase in fresh weight as the cultivation period progressed. The growth curves for these two media showed a clear divergence around day 21, with FG-N6CI demonstrating a superior growth rate than N6CI. At the final point (day 35), the average fresh weight of FG-N6CI reached 7.1 g, which was significantly higher than the 5.8 g observed in the N6CI medium. This result suggests that FG-N6CI promotes callus proliferation more effectively than the N6CI medium.
3.2. Callus Morphology
Morphological observations of calli after 35 days of culture are shown in
Figure 2. Distinct differences were evident among the treatment groups. In the NC medium, only a few microscopic callus fragments were observed across the Petri dish, indicating that proliferation was almost completely suppressed (
Figure 2-C). In the N6CI medium, yellow granular callus masses were observed growing at a moderate density (
Figure 2-B). In contrast, FG-N6CI exhibited more vigorous growth than the N6CI medium (
Figure 2-A). The calli in FG-N6CI appeared pale yellow and friable and densely covered the surface of the Petri dish. These morphological findings are consistent with the fresh weight data described above.
3.3. Gene Expression Analysis
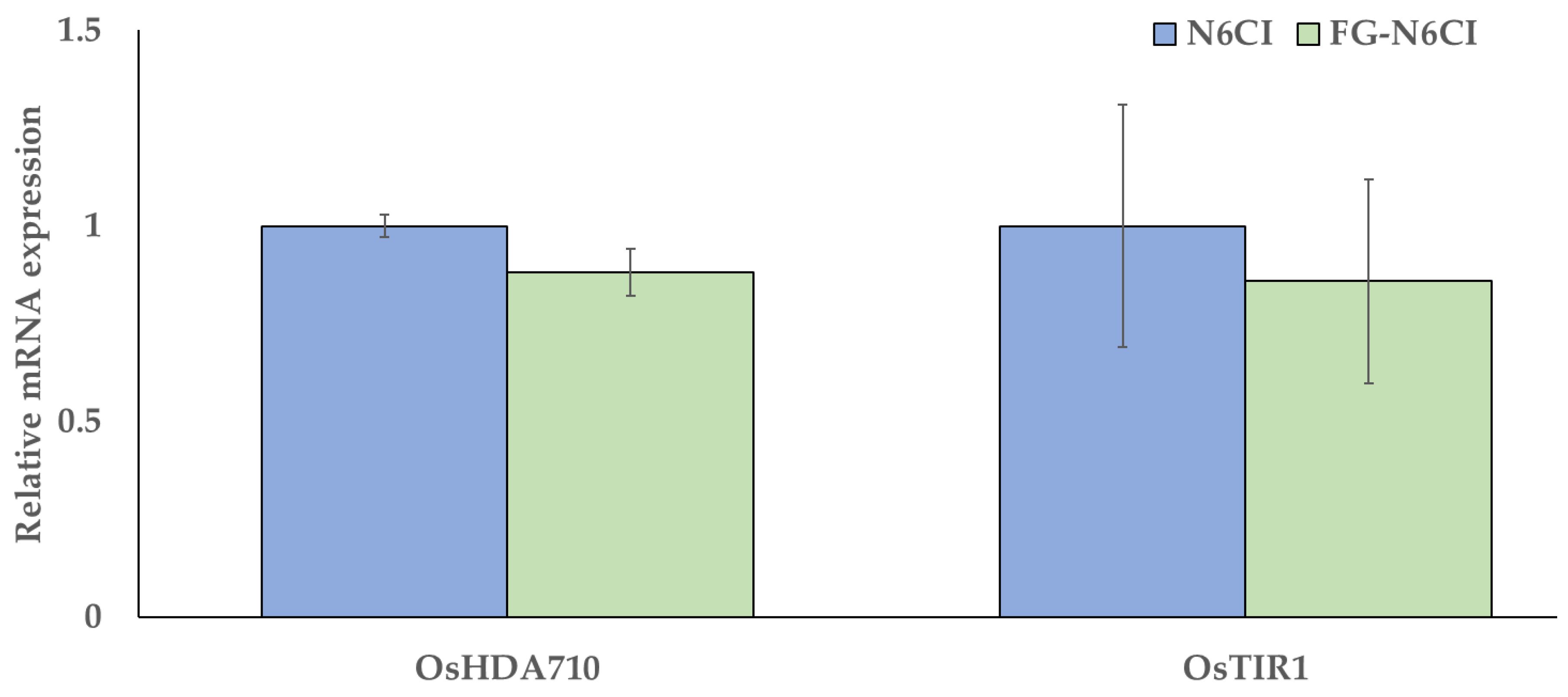
Total RNA was extracted using the RNeasy Plant Mini Kit (Qiagen). Sufficient amounts of RNA were obtained from calli cultured in N6CI and FG-N6CI; however, calli from the NC medium yielded only trace amounts of RNA, which were insufficient for cDNA synthesis. Therefore, RT-qPCR analysis was performed to compare the gene expression levels between the N6CI and FG-N6CI groups. The analysis targeted two genes involved in callus formation: OsHDA710 (histone deacetylase 710 [Os02g0215200]) and OsTIR1 (transport inhibitor response 1 [Os05g0150500]). Gene expression levels are presented as relative values, with those of calli cultured in N6CI medium set at 1. Data are shown as mean ± standard error (SE).
The expression of OsHDA710 in calli cultured in FG-N6CI showed a lower mean value than that in calli cultured in the N6CI medium, but no significant difference was observed. Similarly, although the expression of OsTIR1 in the FG-N6CI group was slightly lower than that in the N6CI group, the difference was not statistically significant (
Figure 3). These results suggested that calli cultured in FG-N6CI maintained a physiological and genetic state conducive to healthy growth, comparable to that of calli grown in the standard reagent-grade medium.
3.4. Comparison of the Costs of Culture Media
Costs were calculated based on catalog prices. For the N6CI basal medium used as a reference, costs were estimated for two scenarios: preparation using individual reagent components and preparation using the commercially available premixed salt powder utilized in this study. The raw material costs per liter (JPY/L) are listed in
Table 5. All values were rounded to two decimal places, and components with calculated costs of less than 0.01 JPY are indicated as “< 0.01”.
The cost of the N6CI basal medium prepared using the commercial premixed powder was approximately 795 JPY/L, whereas that of FG-N6CI was approximately 224 JPY/L, representing a 72% cost reduction. Even when compared with the version prepared with individual reagents (approximately 571 JPY/L), FG-N6CI achieved a 61% reduction in cost.
4. Discussion
4.1. High Nitrogen-Utilizing Capacity of Rice
Rice is adapted to anaerobic paddy fields and excels in utilizing ammonium nitrogen (NH
4+) [
20]. Although high ammonium levels can be toxic to many plants, causing rhizosphere acidification and cation competition [
21], rice efficiently takes up ammonium via OsAMT transporters and assimilates it into amino acids [
22,
23]. N6CI uses reagent-grade ammonium sulfate. Successful growth on FG-N6CI confirms that food-additive-grade ammonium sulfate provided an equally effective ammonium supply without toxicity.
4.2. Impact of Specific Food Ingredients (Molecular Perspectives)
The superior growth observed in FG-N6CI likely resulted from the active molecular components present in the food-grade substitutions.
4.2.1. Yeast Extract and Energy Savings via Salvage Pathway
Casamino acids (in N6CI) provide a balanced nitrogen source. The substitute Yeast Extract (HIMAX GL) contains glutamate, nucleotides (IMP/GMP), and NaCl [
24]. Glutamate acts as a signaling molecule in root apical meristems and promotes cell division [
25]. The presence of nucleotides provides metabolic advantages. The de novo synthesis of purine nucleotides in plants requires a complex 12-step reaction that consumes substantial amounts of ATP. In contrast, nucleotides provided by yeast extract can be directly recycled via the salvage pathway mediated by enzymes such as hypoxanthine-guanine phosphoribosyl transferase (HGPRT) [
26]. This bypass allows cells to save substantial energy during the rapid division phase, potentially channeling resources into biomass accumulation, which aligns with the higher fresh weight observed in FG-N6CI.
4.2.2. Kelp (Kombu) and Phytohormone Synergy
Kelp replaced KI with iodine/potassium but likely provided secondary benefits. Seaweed extracts are biostimulants that contain natural cytokinins (e.g., zeatin riboside) and auxins (IAA). Specifically, extracts from
Ascophyllum nodosum contain substantial amounts of zeatin riboside (66 µg/g) and IAA (50 mg/g) [
27]. However, a recent quantitative analysis using LC-MS/MS revealed that the actual hormone levels in commercial extracts are much lower. Specifically, an analysis of extracts from the Norwegian Ascophyllum nodosum detected IAA at a concentration of 615 ng/g DW [
28]. While both media contained 2,4-D (synthetic auxin), N6CI essentially operated as an “auxin-only” system. The addition of kelp to FG-N6CI introduced trace cytokinins, which likely shifted the hormonal balance from a pure auxin state to a synergistic auxin-cytokinin state. This balance is known to be more effective for inducing rapid cell division [
29,
30].
4.2.3. Manganese Yeast and Transporter Availability
MnSO
4 releases free Mn
2+, which can be problematic in complex media. Free Mn
2+ competes with Fe
2+ and Zn
2+ for uptake via natural resistance-associated macrophage proteins (NRAMP) or zinc-regulated/iron-regulated transporter-like protein (ZIP) transporters [
31,
32,
33]. Additionally, free Mn
2+ is susceptible to oxidation to unavailable forms, such as MnO
2 [
33]. In contrast, in manganese-enriched yeast, manganese is chelated intracellularly by proteins or polyphosphates, which protects it from oxidation [
34]. Plants possess yellow stripe-like (YSL) transporters specifically designed to take up metal-organic complexes [
31]. We speculated that the chelated manganese in FG-N6CI bypassed competitive ion channels (NRAMP/ZIP) and was efficiently absorbed via the YSL pathway, ensuring stable micronutrient availability despite the complex composition of the medium.
4.2.4. Boron Supplement and Molecular Mimicry
Calcium borogluconate (CaBG) was used as the source of boron. In biological systems, boron is typically transported not as free boric acid but as a complex with sugar alcohols (e.g., boron sorbitol) [
35]. CaBG is a complex of boron and gluconic acid (a sugar acid). This structure may exhibit “molecular mimicry” of the natural boron-sugar transport complexes, allowing it to be recognized and transported more efficiently by plant boron transporters and facilitating phloem mobility [
35] compared to inorganic boric acid. Such efficient delivery supported cell wall synthesis and division.
4.3. Mechanism Underlying Callus Morphology (Friable vs. Compact)
FG-N6CI induced Friable calli (ideal for liquid suspension), whereas N6CI induced compact calli.
Gelling Agent Hardness: The physical properties of the gelling agent are likely the decisive factor. Food-grade agar (FG-N6CI) produced a physically harder gel than reagent-grade agar (N6CI). Hard media restrict passive water uptake and increase resistance to cell expansion. This mechanical stress forces the proliferating cells to separate rather than adhere, resulting in a friable texture [
36,
37]. Conversely, softer media (N6CI) allow cells to expand and adhere tightly, forming compact clumps [
36].
Implications for Scaling: Friable calli are technically superior for industrial-scale applications. They disperse easily in liquid suspension bioreactors, ensuring uniform nutrient access and preventing the formation of large necrotic centers, which are common in compact clumps [
38].
4.4. Economic and Regulatory Advantages
This 72% cost reduction demonstrates that food-grade substitution is economically effective. Beyond material costs, FG-N6CI reduces significant “regulatory costs.” Using unapproved reagents (e.g., unverified MnSO4 or KI) for food production would require extensive safety testing, such as GLP toxicity studies, potentially costing millions of dollars and years [
39]. FG-N6CI, composed entirely of food ingredients, inherently bypasses these hurdles. In jurisdictions such as the US or Singapore, this “Safe by Design” approach significantly diminishes the barrier to regulatory approval [
39], accelerating the path to social implementation.
5. Conclusions
We developed “FG-N6CI,” a food-grade medium for rice cellular agriculture, which achieved 72% cost reduction and superior growth (7.1 g vs 5.8 g) compared to standard N6CI. The successful application of FG-N6CI is not solely attributable to elemental substitution but likely arises from the specific molecular benefits of food-grade ingredients, including energy savings via the nucleotide salvage pathway (yeast extract), hormonal synergy (kelp), chelated nutrient uptake (manganese yeast), and molecular mimicry of natural transport forms (boron supplement). This study demonstrates that food-grade media can outperform reagent-grade standards, offering a scientifically robust and economically viable platform for sustainable food production.
6. Patents
A patent application concerning the preparation and use of FG-N6CI described in this work has been filed.
Author Contributions
M.M.: Investigation, Analysis, Writing—Original Draft. K.I.: Conceptualization, Supervision, Editing.
Funding
This research received no external funding.
Data Availability Statement
Data is contained within the article.
Acknowledgments
The authors used Gemini 3 Pro (Google) to assist with English translation and language refinement of an existing Japanese draft. The authors reviewed and edited all outputs and take full responsibility for the final manuscript. We would like to thank Editage (
www.editage.jp) for English language editing.
Conflicts of Interest
A Japanese patent application (Application No. [JP2025-137089]) has been filed by Tohoku University, with Author M.M and Author K.I listed as inventors.
Appendix A
Table A1.
Nutritional and elemental composition of the manganese-enriched yeast.
Table A1.
Nutritional and elemental composition of the manganese-enriched yeast.
| Alternative source |
Component |
Content (%) |
| Manganese yeast (5% Mn) |
Protein |
45.2 |
| |
Lipid |
2.9 |
| |
Carbohydrates |
33.6 |
| |
Moisture |
3.3 |
| |
Ash |
15.0 |
| |
Manganese |
5.4 |
| |
Sodium |
0.095 |
| |
Salt (as NaCl) |
0.2 |
Table A2.
Composition and ingredients of the boron supplement.
Table A2.
Composition and ingredients of the boron supplement.
| Alternative source |
Component |
Content (mg/capsule) |
| Boron supplement |
Boron (as calcium borogluconate) |
3 |
| |
Rice flour |
N.D.1
|
| |
Stearic acid (vegetable source) |
N.D.1
|
Table A3.
Chemical composition of the yeast extract (Hi-Max GL).
Table A3.
Chemical composition of the yeast extract (Hi-Max GL).
| Alternative source |
Component |
Content (%) |
| Yeast extract (Hi-Max GL) |
Monosodium glutamate |
21.2 |
| |
Nucleic acids (IMP-2Na + GMP-2Na)1
|
2.0 |
| |
Sodium chloride (NaCl) |
1.9 |
| |
Moisture |
4.5 |
Table A4.
Suppliers and Product Details of FG-N6CI Components.
Table A4.
Suppliers and Product Details of FG-N6CI Components.
| Component |
Supplier |
Product Number / Product Name |
| KNO3
|
Hayashi Pure Chemical Ind., Ltd., Osaka, Japan |
47007015 |
| (NH4)2SO4
|
Junsei Chemical Co., Ltd., Tokyo, Japan |
83111-2201 |
| KH2PO4
|
Hayashi Pure Chemical Ind., Ltd. |
47005285 |
| MgSO4・7H2O |
Hayashi Pure Chemical Ind., Ltd. |
470040050 |
| CaCl2・2H2O |
Junsei Chemical Co., Ltd. |
18229-2201 |
| EDTA・2Na |
Chelest Corporation, Osaka, Japan |
Chelest F-NA |
| FeSO4・7H2O |
Hayashi Pure Chemical Ind., Ltd. |
47002765 |
| Manganese-enriched yeast |
Medience Corporation, Tokyo, Japan |
Manganese yeast 5% |
| Boron supplement (CaBG) |
NOW Foods |
1410CA / Boron 3 mg |
| ZnSO4・7H2O |
Kanto Chemical Co., Inc., Tokyo, Japan |
58008-17 |
| Kelp |
Yamamoto Food Co., Ltd., Hiroshima, Japan |
497966060007 |
| Niacin (Nicotinic acid) |
Amosea Co., Ltd., Tokyo, Japan |
Niacin Premium |
| Vitamin B6 supplement (Pyridoxine-HCl) |
Nutricost, Vineyard, UT, USA |
Vitamin B6 (Pyridoxine HCl) |
| Glycine |
Nippon Garlic Co., Ltd., Takasaki, Japan |
2023120102 |
| Vitamin B1 supplement (Thiamine-HCl) |
Kyowa Yakuhin Co., Ltd., Osaka, Japan |
Simply B1 |
|
myo-inositol |
NOW Foods |
0527 |
| Yeast extract |
Fuji Foods Corporation |
Hi-Max GL |
| L-Proline |
Marugo Corporation, Osaka, Japan |
L-Proline |
| Table sugar |
Wellneo Sugar Co., Ltd., Tokyo, Japan |
(Cup brand) White Sugar |
| 2,4-D |
Merck, Darmstadt, Germany |
D7299 |
| Agar |
Ina Food Industry Co., Ltd. |
Kanten Cook |
| KOH |
Junsei Chemical Co., Ltd. |
1310-58-3 |
Table A5.
Suppliers and Product Details of Conventional N6CI Medium Components.
Table A5.
Suppliers and Product Details of Conventional N6CI Medium Components.
| Component |
Supplier |
Product Number / Product Name |
| Chu (N6) Basal Salt Mixture (Powder) |
PhytoTech Labs |
C416 |
| Nicotinic acid |
Nacalai Tesque, Inc., Kyoto, Japan |
243-26 |
| Pyridoxine HCl |
FUJIFILM Wako Pure Chemical Corp., Osaka, Japan |
163-05402 |
| Glycine |
FUJIFILM Wako Pure Chemical Corp. |
077-00735 |
| Thiamine HCl |
FUJIFILM Wako Pure Chemical Corp. |
203-00851 |
|
myo-inositol |
FUJIFILM Wako Pure Chemical Corp. |
094-00281 |
| Casamino acid |
Nihon Pharmaceutical Co., Ltd., Tokyo, Japan |
393-02145 |
| L-Proline |
FUJIFILM Wako Pure Chemical Corp. |
165-04605 |
| Sucrose |
FUJIFILM Wako Pure Chemical Corp. |
192-00017 |
| 2,4-D |
Merck |
D7299 |
| Agar |
FUJIFILM Wako Pure Chemical Corp. |
016-11875 |
| KOH |
Nacalai Tesque, Inc. |
28616-45 |
Table A6.
Primers.
| Name |
Sequence (5’ to 3’) |
| Rac-7_F |
ACACCGTGCCAATCTATGAAGG |
| Rac-7-R |
ACAATTTCCCGTTCAGCAGTGG |
| OsHDA710-F |
ATCTCTCAGGCAAAGGTCATGC |
| OsHDA710-R |
ACCACCACCAAGAAGAAGCAAC |
| OsTIR1-F |
TTGGATGTCGTCGTGCTTGTTG |
| OsTIR1-R |
AACAGGTGTTTCATCCGGAAGC |
References
- United Nations Department of Economic and Social Affairs; United Nations Department of Economic and Social Affairs; Population Division. Population Division World Population Prospects 2024: Summary of Results; New York, 2024. [Google Scholar]
- FAO The State of Food and Agriculture. Addressing Land Degradation across Landholding Scales; The State of Food and Agriculture (SOFA); FAO: Rome, Italy, 2025; ISBN 978-92-5-140142-2. [Google Scholar]
- Tubiello, F.N.; Rosenzweig, C.; Conchedda, G.; Karl, K.; Gütschow, J.; Xueyao, P.; Obli-Laryea, G.; Wanner, N.; Qiu, S.Y.; Barros, J.D.; et al. Greenhouse Gas Emissions from Food Systems: Building the Evidence Base. Environ. Res. Lett. 2021, 16, 065007. [Google Scholar] [CrossRef]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Controlling Eutrophication: Nitrogen and Phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef]
- McNulty, M.J.; Stout, A.J.; Kaplan, D.L. Meating the Moment: Challenges and Opportunities for Cellular Agriculture to Produce the Foods of the Future. EMBO Rep 2025, 26, 3229–3235. [Google Scholar] [CrossRef]
- Benjaminson, M.A.; Gilchriest, J.A.; Lorenz, M. In Vitro Edible Muscle Protein Production System (Mpps): Stage 1, Fish. Acta Astronautica 2002, 51, 879–889. [Google Scholar] [CrossRef]
- Post, M.J. An Alternative Animal Protein Source: Cultured Beef. Annals of the New York Academy of Sciences 2014, 1328, 29–33. [Google Scholar] [CrossRef]
- Aisala, H.; Kärkkäinen, E.; Jokinen, I.; Seppänen-Laakso, T.; Rischer, H. Proof of Concept for Cell Culture-Based Coffee. J. Agric. Food Chem. 2023, 71, 18478–18488. [Google Scholar] [CrossRef] [PubMed]
- Khushvakov, J.; Opitz, S.E.W.; Plüss, N.; Sun, J.; Manthey, L.J.; Rischer, H.; Yeretzian, C. Analytical Platform to Determine Similarities and Dissimilarities between Cell-Cultured Coffee and Farm-Grown Coffee. ACS Food Sci. Technol. 2024, 4, 1890–1903. [Google Scholar] [CrossRef]
- Nordlund, E.; Lille, M.; Silventoinen, P.; Nygren, H.; Seppänen-Laakso, T.; Mikkelson, A.; Aura, A.-M.; Heiniö, R.-L.; Nohynek, L.; Puupponen-Pimiä, R.; et al. Plant Cells as Food – A Concept Taking Shape. Food Research International 2018, 107, 297–305. [Google Scholar] [CrossRef]
- US Food and Drug Administration; US Food and Drug Administration; US Department of Agriculture. US Department of Agriculture Formal Agreement between FDA and USDA Regarding Oversight of Human Food Produced Using Animal Cell Technology Derived from Cell Lines of USDA-Amenable Species; 2019. [Google Scholar]
-
Centre for Food Safety (CFS) Technical Guidance Notes on the Safety Assessment of Cultured Meat; Centre for Food Safety (CFS), Food and Environmental Hygiene Department, HKSAR, 2025.
- Chu, C.C.; Wang, C.C.; Sun, C.S.; Hsu, C.; Yin, K.C.; Chu, C.Y.; Bi, F.Y. Establishment of an Efficient Medium for Anther Culture of Rice through Comparative Experiments on the Nitrogen Sources. Scientia Sinica 1975, 18, 659–668. [Google Scholar] [CrossRef]
- Khaleda, L.; Al-Forkan, M. Stimulatory Effects of Casein Hydrolysate and Proline in in Vitro Callus Induction and Plant Regeneration from Five Deepwater Rice (Oryza Sativa L.). Biotechnology 2006, 5, 379–384. [Google Scholar] [CrossRef]
- Ministry of Health, Labour and Welfare Japan’s Specifications and Standards for Food Additives 2024. 2024.
- Japan Food Chemical Research Foundation List of Designated Additives (Annexed Table 1 of the Regulations). 2023.
- Japan Food Chemical Research Foundation Usage Standards and Storage Standards for Each Additive. 2023.
- Kikuchi, Y.; Takebayashi, T.; Sasaki, S. Iodine Concentration in Current Japanese Foods and Beverages. Jpn. J. Hyg. 2008, 63, 724–734. [Google Scholar] [CrossRef]
- Yoshida, H.; Fujimoto, S.; Hayashi, J. Studies on the Physiological and Chemical Characteristics of Kuritake Mushroom (Naematoloma Sublateritium) (Fr.) Karst.). Part 1. Effects of Carbon and Nitrogen Sources on the Vegetative Growth of Kuritake Mushroom (Naematoloma Sublateritium). JOURNAL OF FOOD SCIENCE AND TECHNOLOGY 1990, 37, 695–701. [Google Scholar] [CrossRef]
- Zhu, Y.; Qi, B.; Hao, Y.; Liu, H.; Sun, G.; Chen, R.; Song, S. Appropriate NH4+/NO3– Ratio Triggers Plant Growth and Nutrient Uptake of Flowering Chinese Cabbage by Optimizing the pH Value of Nutrient Solution. Front. Plant Sci. 2021, 12, 656144. [Google Scholar] [CrossRef] [PubMed]
- Britto, D.T.; Kronzucker, H.J. NH4+ Toxicity in Higher Plants: A Critical Review. Journal of Plant Physiology 2002, 159, 567–584. [Google Scholar] [CrossRef]
- Duan, Y.H.; Zhang, Y.L.; Ye, L.T.; Fan, X.R.; Xu, G.H.; Shen, Q.R. Responses of Rice Cultivars with Different Nitrogen Use Efficiency to Partial Nitrate Nutrition. Annals of Botany 2007, 99, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Hou, M.; Yu, M.; Li, Z.; Ai, Z.; Chen, J. Molecular Regulatory Networks for Improving Nitrogen Use Efficiency in Rice. IJMS 2021, 22, 9040. [Google Scholar] [CrossRef]
- Tao, Z.; Yuan, H.; Liu, M.; Liu, Q.; Zhang, S.; Liu, H.; Jiang, Y.; Huang, D.; Wang, T. Yeast Extract: Characteristics, Production, Applications and Future Perspectives. J. Microbiol. Biotechnol. 2023, 33, 151–166. [Google Scholar] [CrossRef]
- Forde, B.G.; Lea, P.J. Glutamate in Plants: Metabolism, Regulation, and Signalling. Journal of Experimental Botany 2007, 58, 2339–2358. [Google Scholar] [CrossRef]
- Witte, C.-P.; Herde, M. Nucleotide Metabolism in Plants. Plant Physiol. 2020, 182, 63–78. [Google Scholar] [CrossRef]
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed Extracts as Biostimulants of Plant Growth and Development. J Plant Growth Regul 2009, 28, 386–399. [Google Scholar] [CrossRef]
- Wally, O.S.D.; Critchley, A.T.; Hiltz, D.; Craigie, J.S.; Han, X.; Zaharia, L.I.; Abrams, S.R.; Prithiviraj, B. Regulation of Phytohormone Biosynthesis and Accumulation in Arabidopsis Following Treatment with Commercial Extract from the Marine Macroalga Ascophyllum Nodosum. J Plant Growth Regul 2013, 32, 324–339. [Google Scholar] [CrossRef]
- Taratima, W.; Plaikhunto, K.; Wichachai, C.; Maneeratta, P. Optimization of Rice (Oryza Sativa L.) ‘Tubtim Chumphae’, for Callus Induction, Proliferation and Plantlet Regeneration. Asian J. of Plant Sciences 2022, 21, 727–734. [Google Scholar] [CrossRef]
- Ikeuchi, M.; Sugimoto, K.; Iwase, A. Plant Callus: Mechanisms of Induction and Repression. Plant Cell 2013, 25, 3159–3173. [Google Scholar] [CrossRef]
- Socha, A.L.; Guerinot, M.L. Mn-Euvering Manganese: The Role of Transporter Gene Family Members in Manganese Uptake and Mobilization in Plants. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Qin, L.; Han, P.; Chen, L.; Walk, T.C.; Li, Y.; Hu, X.; Xie, L.; Liao, H.; Liao, X. Genome-Wide Identification and Expression Analysis of NRAMP Family Genes in Soybean (Glycine Max L.). Front. Plant Sci. 2017, 8, 1436. [Google Scholar] [CrossRef]
- Askarian, M.; Mousavi, F.; Dufault-Bedard, V.; Houlachi, G.; Alamdari, H. Kinetic Study of Manganese Oxidative Precipitation Reaction by Using SO2/Air Gas Mixture. Metals 2024, 14, 412. [Google Scholar] [CrossRef]
- Andreeva, N.; Kulakovskaya, E.; Zvonarev, A.; Penin, A.; Eliseeva, I.; Teterina, A.; Lando, A.; Kulakovskiy, I.V.; Kulakovskaya, T. Transcriptome Profile of Yeast Reveals the Essential Role of PMA2 and Uncharacterized Gene YBR056W-A (MNC1) in Adaptation to Toxic Manganese Concentration. Metallomics 2017, 9, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Pan, Z.-Y.; Hussain, S.B.; Han, Z.-X.; Peng, S.-A.; Liu, Y.-Z. Foliar Supplied Boron Can Be Transported to Roots as a Boron-Sucrose Complex via Phloem in Citrus Trees. Front. Plant Sci. 2020, 11, 250. [Google Scholar] [CrossRef]
- Muzika, N.S.; Kamai, T.; Williams, L.E.; Kleiman, M. Characterization of Gelling Agents in Callus Inducing Media: Physical Properties and Their Effect on Callus Growth. Physiologia Plantarum 2024, 176, e14312. [Google Scholar] [CrossRef]
- Jadhav, P.V.; Dudhare, M.S.; Saluja, T.; Sarawgi, A.K.; Saxena, R.; Chandel, G. Assessment of Critical Factors Influencing Callus Induction, in Vitro Regeneration and Selection of Bombarded Indica Rice Genotypes. Journal of Agricultural Biotechnology and Sustainable Development 2011, 3, 44–59. [Google Scholar]
- Sathish, S.; Venkatesh, R.; Safia, N.; Sathishkumar, R. Studies on Growth Dynamics of Embryogenic Cell Suspension Cultures of Commercially Important Indica Rice Cultivars ASD16 and Pusa Basmati. 3 Biotech 2018, 8, 194. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.; Lee, G.; Jeong, D.-W. A Study on Safety Evaluation System of Cultured Foods among Alternative Proteins. Food Sci Biotechnol 2025, 34, 365–371. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).