Submitted:
23 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethical Statment
2.2. Study Design and study sites
2.3. Participants recruitment and collection of information
2.4. Laboratory tests for HIV
2.5. In-house ELISA TLA Test
2.6. Data analysis
3. Results
3.1. Demographic and Clinical Characteristics of the Study Population
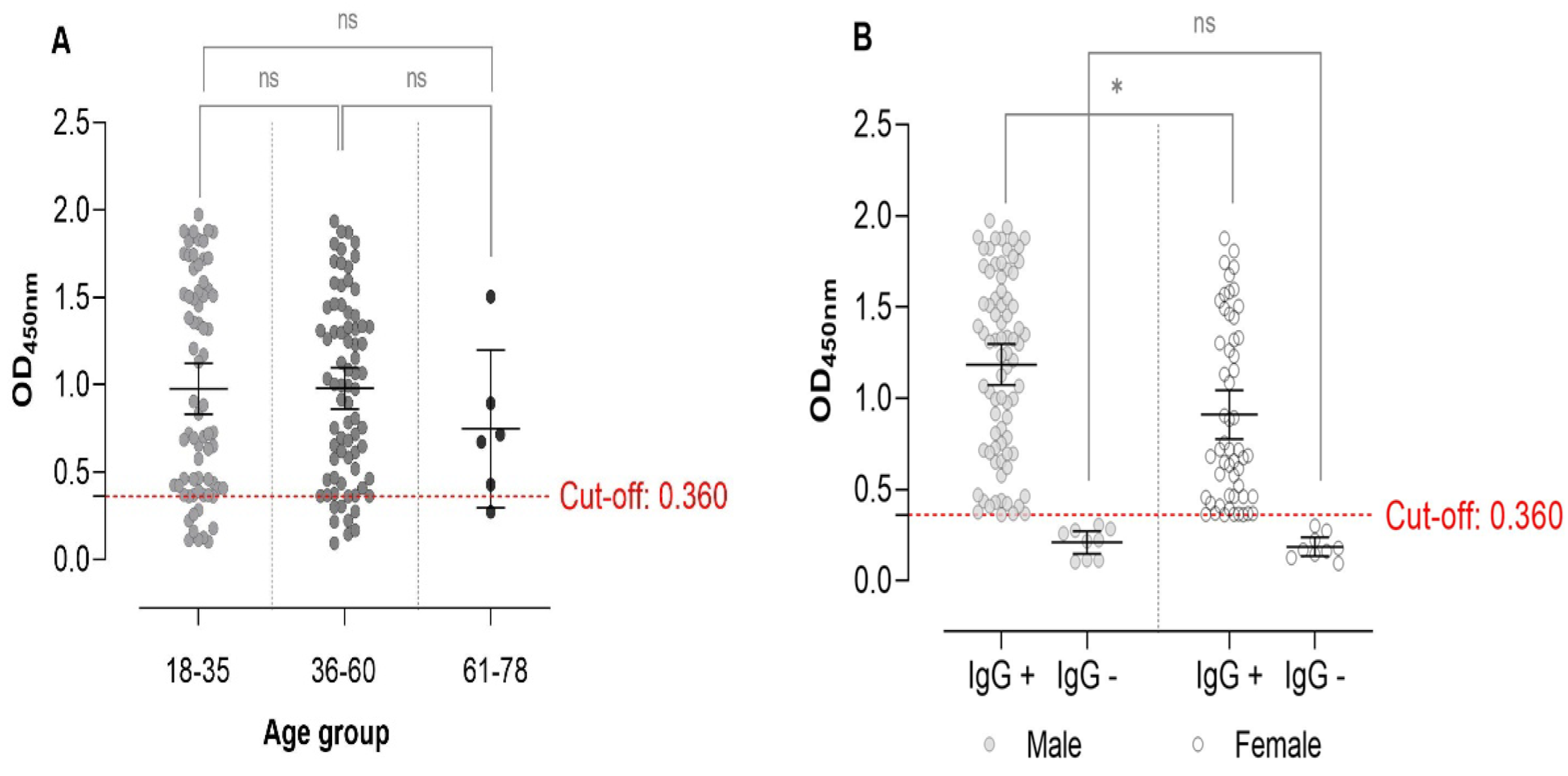
3.2. Seroprevalence of Toxoplasmosis in the Study Population
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HIV | Human Immunodeficiency Virus |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| IgG | Immunoglobulin G |
| TLA | Total Lysate Antigen |
| CI | Confidence Interval |
| OD450 | Optical Density at 450 nm |
| CD4 | Cluster of Differentiation 4 |
References
- Tenter, AM; Heckeroth, AR; Weiss, LM. Toxoplasma gondii: from animals to humans. Int J Parasitol. 2000, 30(12-13), 1217–58. [Google Scholar] [CrossRef] [PubMed]
- Hill, DE; Chirukandoth, S; Dubey, JP. Biology and epidemiology of Toxoplasma gondii in man and animals. Anim Health Res Rev. 2005, 6(1), 41–61. [Google Scholar] [CrossRef] [PubMed]
- Robert-Gangneux, F; Dardé, ML. Epidemiology of and diagnostic strategies for toxoplasmosis. Clin Microbiol Rev. 2012, 25(2), 264–96. [Google Scholar] [CrossRef] [PubMed]
- Dubey, JP. History of the discovery of the life cycle of Toxoplasma gondii. Int J Parasitol. 2009, 39(8), 877–82. [Google Scholar] [CrossRef]
- Dardé, ML; Peyron, F. Toxoplasma y toxoplasmosis. EMC - Pediatría 2013, 48(1), 1–12. [Google Scholar] [CrossRef]
- Basavaraju, A. Toxoplasmosis in HIV infection: an overview. Trop Parasitol 2016, 6(2), 129–35. [Google Scholar] [CrossRef]
- Botero-Rodríguez, F; Zárate-Velasco, AM; Cote Martínez, D; Gómez-Restrepo, C; Duarte Osorio, A. Infección por Toxoplasma gondii como factor de riesgo para desarrollar esquizofrenia: revisión de la literatura. Univ Med. 2023, 64(2). [Google Scholar] [CrossRef]
- Dian, S; Ganiem, AR; Ekawardhani, S. Cerebral toxoplasmosis in HIV-infected patients: a review. Pathog Glob Health 2023, 117(1), 14–23. [Google Scholar] [CrossRef]
- Molan, A; Nosaka, K; Hunter, M; Wang, W. Global status of Toxoplasma gondii infection: systematic review and prevalence snapshots. Trop Biomed. 2019, 36(4), 898–925. [Google Scholar]
- Zavala-Hoppe, A; Piguave-Cacao, R; Ponce-Macias, N. Epidemiología y factores de riesgo de la toxoplasmosis en los países de Latinoamérica. MQRInvestigar 2025, 9(1), e234. [Google Scholar] [CrossRef]
- Instituto Nacional de Salud (INS). Anuario estadístico 2023 [Internet]. INS: Lima, 2023; Available online: https://www.gob.pe/institucion/ins/informes-publicaciones/3833116-anuario-estadistico.
- Flores, BCA; Ferradas, C; Málaga-Machaca, ES; Diestra, A; Steinberg, HE; Calla, J; et al. Seroprevalence and factors associated with infection with Toxoplasma gondii in Iquitos and Lima, Peru. Am J Trop Med Hyg. 2025, 113(3), 600–6. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, HE; Bowman, NM; Diestra, A; Ferradas, C; Russo, P; Clark, DE; et al. Detection of toxoplasmic encephalitis in HIV positive patients in urine with hydrogel nanoparticles. PLoS Negl Trop Dis. 2021, 15(3), e0009199. [Google Scholar] [CrossRef]
- Steinberg, HE; Ramachandran, PS; Diestra, A; Pinchi, L; Ferradas, C; Kirwan, DE; et al. Clinical and metagenomic characterization of neurological infections of people with human immunodeficiency virus in the Peruvian Amazon. Open Forum Infect Dis. 2023, 10(11), ofad515. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Prieto, AJY; Quispe Roldan, JF; Sanchez Rosales, KIB; Casasola Lescano, X; Sanjinez Viera, YM; Giraldo Malca, UF. Climate variation in the Amazonian peneplain due to deforestation and urban expansion in Iquitos - Peru, between 1984 and 2023. In Proceedings of the 22nd LACCEI International Multi-Conference for Engineering, Education, and Technology: Sustainable Engineering for a Diverse, Equitable, and Inclusive Future at the Service of Education, Research, and Industry for a Society 5.0, San Jose, Costa Rica, 2024 Jul 17-19; LACCEI. [Google Scholar] [CrossRef]
- Sengupta, PP; Jacob, SS; Suresh, KP; Rajamani, S; Maharana, SM. Exploring global trends in human toxoplasmosis seroprevalence by meta-analysis. Exp Parasitol. 2025, 275, 108971. [Google Scholar] [CrossRef] [PubMed]
- Pari, J; Yauyo, R; Aquino, A; Adauto, M; Ochoa, S; Rojas, A; et al. Toxoplasma gondii infection in risk population groups from Andes Peruvian. Open J Med Microbiol. 2020, 10, 26–32. [Google Scholar] [CrossRef]
- Gontijo da Silva, M; Clare Vinaud, M; de Castro, AM. Prevalence of toxoplasmosis in pregnant women and vertical transmission of Toxoplasma gondii in patients from basic units of health from Gurupi, Tocantins, Brazil, from 2012 to 2014. PLoS One 2015, 10(11), e0141700. [Google Scholar] [CrossRef]
- Ministerio de Desarrollo e Inclusión Social (MIDIS). REDinforma report [Internet]. 8 Jan 2026. Available online: https://app.midis.gob.pe/RedInforma/Reporte/ReportePDF?vCodTema=1.
- Patz, JA; Graczyk, TK; Geller, N; Vittor, AY. Effects of environmental change on emerging parasitic diseases. Int J Parasitol. 2000, 30(12-13), 1395–405. [Google Scholar] [CrossRef]
- Yan, C; Liang, LJ; Zheng, KY; Zhu, XQ. Impact of environmental factors on the emergence, transmission and distribution of Toxoplasma gondii. Parasit Vectors 2016, 9, 137. [Google Scholar] [CrossRef]
- Liu, L; Liu, LN; Wang, P; Lv, TT; Fan, YG; Pan, HF. Elevated seroprevalence of Toxoplasma gondii in AIDS/HIV patients: a meta-analysis. Acta Trop. 2017, 176, 162–7. [Google Scholar] [CrossRef]
- Voyiatzaki, C; Zare Chormizi, AD; Tsoumani, ME; Efstathiou, A; Konstantinidis, K; Chaniotis, D; et al. Seroprevalence of Toxoplasma gondii among HIV positive patients under surveillance in Greek infectious disease units: a screening study with comparative evaluation of serological methods. Pathogens 2024, 13(5), 375. [Google Scholar] [CrossRef]
- Safarpour, H; Cevik, M; Zarean, M; Barac, A; Hatam-Nahavandi, K; Rahimi, MT; et al. Global status of Toxoplasma gondii infection and associated risk factors in people living with HIV. AIDS 2020, 34(3), 469–74. [Google Scholar] [CrossRef]
- Pan American Health Organization; World Health Organization. HIV/AIDS in the Americas: regional epidemiological update 2025 [Internet]. 2025. Available online: https://www.paho.org/en/topics/hivaids.
- Jimenez-Chunga, J; Gomez-Puerta, LA; Vargas-Calla, A; Castro-Hidalgo, J; Sánchez-Chicana, C; Calderón-Sánchez, M. Seroprevalence of Toxoplasma gondii, risk factors and knowledge about toxoplasmosis in undergraduate students from Lima, Peru. Acta Trop. 2024, 255, 107233. [Google Scholar] [CrossRef] [PubMed]
- Yu, CP; Chen, BC; Chou, YC; Hsieh, CJ; Lin, FH. The epidemiology of patients with toxoplasmosis and its associated risk factors in Taiwan during the 2007-2020 period. PLoS One 2023, 18(8), e0290769. [Google Scholar] [CrossRef] [PubMed]
- Shoukat, T; Awan, UA; Mahmood, T; Afzal, MS; Wasif, S; Ahmed, H; et al. Epidemiology of toxoplasmosis among the Pakistani population: a systematic review and meta-analysis. Pathogens 2022, 11(6), 675. [Google Scholar] [CrossRef]
- Gondim, LFP; Mineo, JR; Schares, G. Importance of serological cross-reactivity among Toxoplasma gondii, Hammondia spp., Neospora spp., Sarcocystis spp. and Besnoitia besnoiti. Parasitology 2017, 144(7), 851–68. [Google Scholar] [CrossRef]
- Ferra, BT; Chyb, M; Sołowińska, K; Holec-Gąsior, L; Skwarecka, M; Baranowicz, K; et al. The development of Toxoplasma gondii recombinant trivalent chimeric proteins as an alternative to Toxoplasma lysate antigen (TLA) in enzyme-linked immunosorbent assay (ELISA) for the detection of immunoglobulin G (IgG) in small ruminants. Int J Mol Sci. 2024, 25(8), 4384. [Google Scholar] [CrossRef]
- Dong, H; Zhang, J; Wang, Q; Shen, Y; Zhou, B; et al. Development of an indirect ELISA for detecting Toxoplasma gondii IgG antibodies based on a recombinant TgIMP1 protein. PLoS Negl Trop Dis. 2024, 18(8), e0012421. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Total, n (%) |
| Age Group | |
| 18 - 35 years | 70 (46.36) |
| 36 - 60 years | 75 (49.67) |
| 61 - 78 years | 6 (3.97) |
| Sex | |
| Male | 88 (58.28) |
| Female | 63 (41.72) |
| HIV Infection Status | |
| Positive | 92 (60.93) |
| Negative | 59 (39.07) |
| Seroprevalence of T. gondii | |
| Positive | 133 (88.08) |
| Negative | 18 (11.92) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




