1. Introduction
Surgical site infections (SSIs) have a great impact on postoperative morbidity and hospital stay and constitute a major economic burden to the health care system [
1]. Abdominal surgery carries the highest risk compared to other types of surgery, with an incidence between 4.1% and 26.7% [
2]. Among the main factors to determine this variability are underlying disease, type of intervention, comorbidity and immunosuppressive treatment. Factors such as surveillance and methods by which the data is collected also play an important role.
Several regimens for antibiotic treatment in surgery for secondary peritonitis and for the prophylaxis in elective abdominal surgery has been recommended. At our institution the combination of metronidazole and cefuroxime has been standard. The treatment goal should be to achieve concentrations above the minimum inhibitory concentration (MIC) of various bacterial strains in the peritoneal cavity during the treatment period. Studies suggest that a concentration of antibiotics that is above a given MIC value for at least more 40%–50% of the time (T>MIC) within the dosing interval result in an adequate clinical and microbiological efficacy [
3]. MIC values for metronidazole against anaerobe species varies considerably [
4] and with a breakpoint on 4 μg/mL according to The European Committee on Antimicrobial Susceptibility Testing (EUCAST) [
5]. For cefuroxime the MIC value for Gram- negative bacteria for most species are 4 μg/mL and higher for more resistant
Escherichia coli strains with a confidence interval of 8-16 μg/mL [
5].
The diffusion of antibiotics into the peritoneal fluid is an important issue in both the prophylaxis of surgical site infections in elective abdominal surgery and in surgery for secondary peritonitis. An inflamed peritoneal surface may influence the capability of antibiotics to pass across the peritoneal lining into the peritoneal cavity. This might have an important clinical implication in relation to dosing regimens to achieve optimal treatment results without a risk of overdosing. To our best knowledge this has not been investigated previously in patients undergoing acute surgery for secondary peritonitis or in elective surgery. Microdialysis is a known and well-established principle to measure the concentration of antibiotics in different tissue compartments (6).
The primary objective of this study was to investigate whether the concentrations of cefuroxime and metronidazole reached the MIC values for Gram-negative bacteria, Eschericia Coli and anaerobic bacteria, respectively, in the peritoneal cavity in patients undergoing either emergency surgery for secondary diffuse peritonitis or elective surgery for inflammatory bowel disease (IBD). The secondary objective was to investigate whether the sampling period in relation to antibiotic administration would have an influence on the results.
2. Results
The distribution of age, gender and body mass index (BMI) in each group is shown in
Table 1. The rate of missing samples in the IBD group was 5.6% for both metronidazole and cefuroxime. The values in the peritonitis group were 18% and 24% respectively. Only 4 out of 7 participants in the peritonitis group and 5 out of 11 participants in the IBD group had a full data set for the collection of dialysates. The reasons for missing data were mainly due to errors during sample collection. One sample was lost during analysis. Two participants did not complete the study period, one due to skin irritation around the microdialysis catheter that was removed prematurely and one due to the catheter falling out. No participants experienced surgical site infectious or other complications related to the microdialysis catheter, and all patients had an uncomplicated postoperative course.
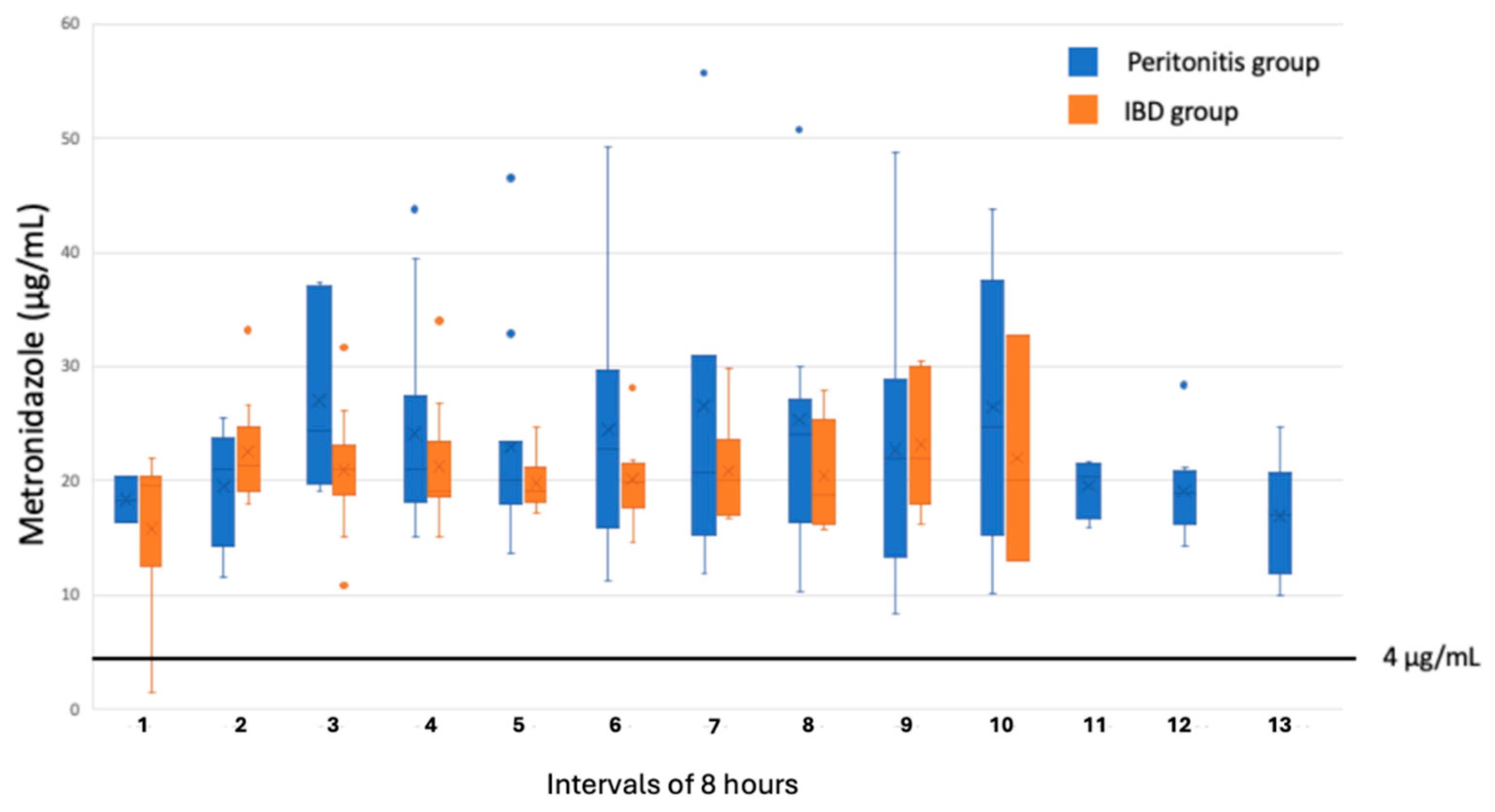
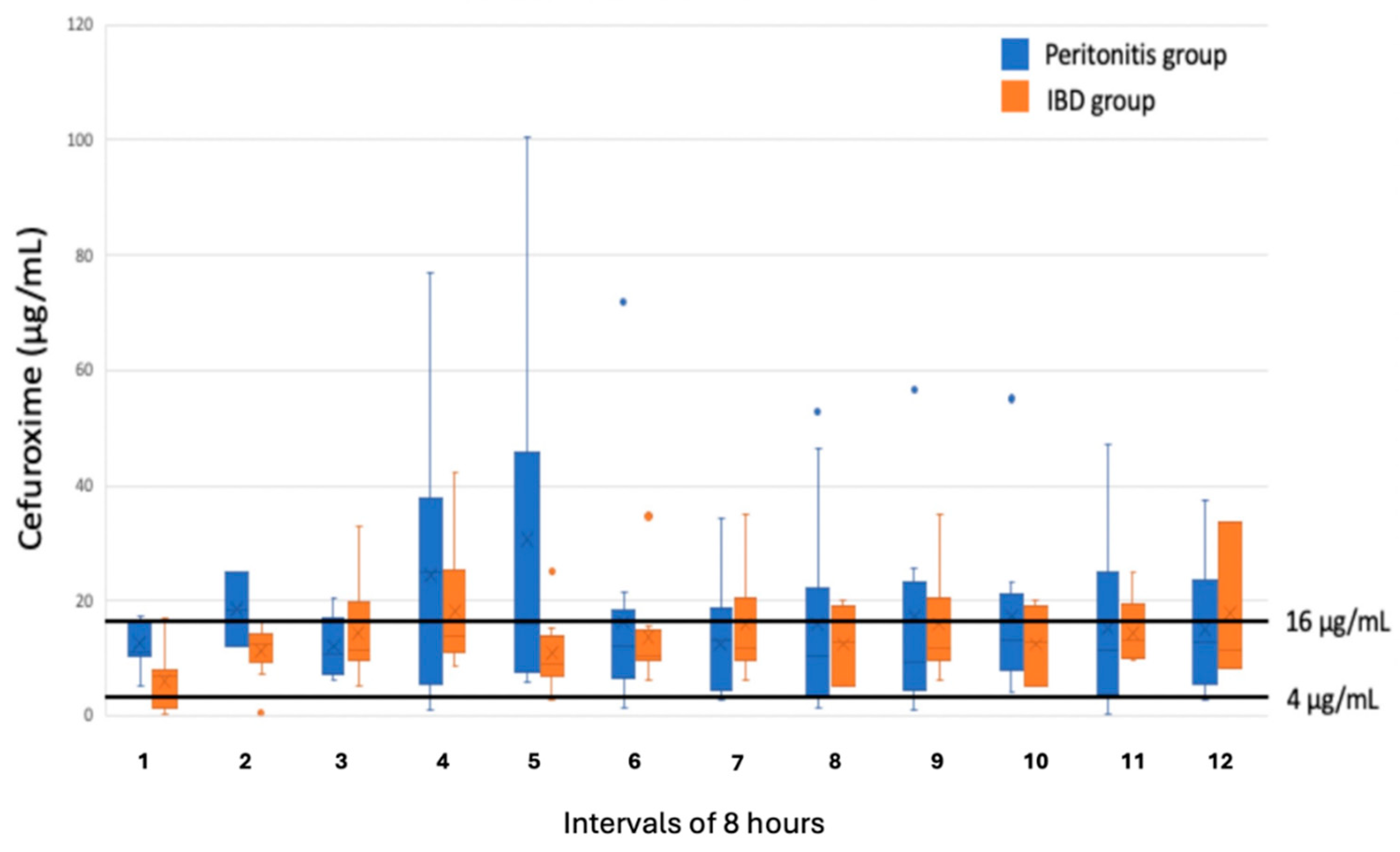
There were no significant differences in the concentrations of the measured concentration in the peritoneal fluid neither for cefuroxime nor for metronidazole between the two groups (
Figure 1 and
Figure 2). A concentration of metronidazole of ≥4 μg/mL was measured in all the samples collected from the peritonitis group and in 99% of the samples collected in the IBD group. For cefuroxime 88% of the samples in the peritonitis group and 93% of the samples in the IBD group a concentration ≥4 μg/mL was measured. A concentration ≥16 μg/mL this was only reached in 40% of the samples from the peritonitis group and in 23% from the IBD group.
In the peritonitis group the interindividual as well as the intraindividual variation in the concentrations measured was greater for both cefuroxime and metronidazole compared to the IBD group (
Table 2).
In the IBD group the mean difference in concentration between participant 1-4 (sampling started 4 hours after antibiotic administration) and participant 5-11 (sampling started immediately after antibiotic administration) was 1.5 for MTZ [ -2.1 to 2.9] and 2.1 for CEF [ -5 to 9.3].
3. Discussion
With the antibiotic regimen of cefuroxime and metronidazole used in the present study, a concentration of metronidazole of ≥4 μg/mL corresponding to the MIC value for most anaerobic strains was measured in almost all the collected microdialysates from both the IBD and the peritonitis group. Despite the relative long serum half-life time for metronidazole (6-10 hours) no tendency of accumulation in the peritoneal fluid was found in any of the patients from the two groups.
For cefuroxime a concentration on ≥4 μg/mL corresponding to the MIC value for most gram-negative strains was reached in >87% of the samples in both groups. A concentration of ≥16 μg/ml corresponding to the MIC value for mores more resistant bacterial strains was measured in only 40% of the samples in the peritonitis group and 23% in the IBD group. With the relatively few patients included in the present study we could not investigate the clinical relevance of these findings but all patients in our study didn’t develop any clinical signs of surgical site infectious or other infectious complications during the postoperative period.
Another finding in our study was that the sampling in relation to administration of the antibiotics in the IBD-group didn’t have any influence on the results. This is not surprising as the length of sampling interval on 8 hours corresponded to the interval of antibiotic administration.
From our results it is not possible to say anything about the fluctuations in the concentration over time within each sampling period of either antibiotic administered but only the cumulative concentration during a sampling period, which was equivalent to the time interval between administration of the drugs. Sampling periods with shorter intervals might have given more information on the fluctuation in the concentration of the antibiotics in relation to drug administration such as the length of the time period were the value of the concentration was above values for MIC (T>MIC), the area under the curve in relation to MIC (AUC/MIC) and of maximum concentration obtained in relation to MIC (C
max/MIC). These parameters are traditionally used to evaluate the pharmacokinetics in serum but more difficult to obtain in the target organs including the peritoneal fluid. Metronidazole is a concentration-dependent antibiotic, for which AUC/MIC is generally considered the most relevant pharmacodynamic target. In contrast, cefuroxime is a time-dependent antibiotic, where T>MIC is the most appropriate efficacy target. Our results focused exclusively on whether the measured concentrations exceed predefined MIC thresholds, which may oversimplify the pharmacodynamic interpretation. In addition, the clinical relevance of a given MIC value may be interpreted with caution [
7].
To achieve the traditional pharmacokinetic parameters mentioned above a sampling of microdialysates with much shorter intervals would have been necessary. This would have required a considerably higher perfusion rate to achieve sufficient material for the concentrations analysis. However, a higher perfusion rate compromises the relative recovery in microdialysis [
9]. An important limitation of our study is that no analysis of the recovery rate of the antibiotics administered were performed. An in vivo estimation of the relative recovery in our study would require a retrodialysis before or after treatment completion, which was considered as unethical to perform in our two patents groups. Previous in vitro studies have shown a high recovery rate of more than 75% for both cefuroxime and metronidazole using a similar microdialysis principle as used in the present study [
9,
10]. Other studies have shown no interference in recovery rate with a combined antibiotic administration [
11]. To our knowledge no other studies on recovery rate in peritoneal fluid has been published. The true concentrations in our study therefore might have been approximately 25% higher than measured values.
The advantage of the microdialysis principle is its ability to measure concentration of various administered compounds or drugs and their diffusion into the interstitial fluid of various tissue compartments where the drug is intended to elicit its effect. This has not been studied for the peritoneal fluid in humans, and one important finding was that the diffusion of both antibiotics investigated was similar in peritonitis compared to non-peritonitis, although with a higher intra- and intervariability in the peritonitis group.
The IBD patients were chosen as the control material because they receive standard postoperative antibiotic treatment for 3 days postoperatively due to an increased risk of surgical site infections, and that this therapeutic regimen is similar to that used for the treatment of secondary peritonitis at our institution. IBD patients had a lover coefficient of variation both interindividual and between individuals for both metronidazole and cefuroxime compared to the group of patients with peritonitis. This might indicate that an ongoing peritonitis may influence the penetration of antibiotics to the peritoneal cavity, A grading of the peritonitis was not performed which is another limitation of the study but a reliable statistical analysis on a possible correlation between the degree or extent of peritonitis would not have been possible with this low number of patients.
4. Material and Methods
4.1. Setting
This open, prospective observational single-centre study was conducted at the surgical department of Odense University Hospital. The study was conducted in accordance with the Declaration of Helsinki following approvals from the Regional Scientific Ethical Committees for Southern Denmark (ID: S- 20130018 and ID: S- S-20190095), and the Danish Data Protection Agency (19/36273).
4.2. Participants
The inclusion criteria were patients undergoing emergency surgery for secondary peritonitis due to a perforation on the small intestine, colon or rectum and elective surgery for IBD. The patients had to be older than 18 years. Exclusion criteria were an eGFR < 30 ml/min/1.73 m2 and known allergies to metronidazole or cefuroxime, disseminated cancer disease, postoperative vacuum assisted closure or peritonitis confined to less than two quadrants. Informed written consent was obtained from all participants prior to entry into the study. A total of 11 patients operated for inflammatory bowel disease and 7 patients operated for secondary peritonitis were included.
4.3. Peritoneal Microdialysis
At the end of surgery and before closure of the abdomen, an CMA 62 microdialysis catheter (M Dialysis AB, Stockholm, Sweden) was placed into the peritoneal cavity through a transcutaneous approach outside the incision with the tip of the catheter floating freely between intestinal loops. In patients with secondary peritonitis, the catheter was placed in the most severe affected quadrant. The catheter was perfused continuously with Perfusion Fluid (T1®, M Dialysis AB, Stockholm) via a CMA 106 or 107 microdialysis syringe pump (M Dialysis AB, Stockholm). The flow rate was set to 0.3 μL/min.
All patients received intravenous antibiotic with 3 g of cefuroxime (Zinacef®, Actavis, Gentofte, Denmark) and 1.5 g metronidazole (Baxter A/S, Allerød, Denmark) during induction of anaesthesia and were continued with 1.5 g cefuroxime and 500 mg metronidazole postoperatively with 8 hours interval for at least 3 days.
In the peritonitis group dialysates were collected every 8 hours starting immediately after the first antibiotic administration at 06.00 AM on postoperative day one. For the IBD patients sampling was started 4 hours (10.00 AM) after the first administration and continued for 8 hours intervals in the first 4 patients and in the last 7 patients immediately after first administration (06.00 AM). All samples were immediately stored at -80° C for later analysis.
4.4. Measuring the Concentrations of Cefuroxime and Metronidazole
4.4.1. Preparation of Stock Solutions, Calibration Samples, and Quality Control Samples
For cefuroxime and metronidazole (USP 1098209 and USP 1442009, from Sigma-Aldrich); a 500 μg/mL stock solution in methanol was prepared. A working solution of 5.0 μg/mL was prepared by diluting the stock solution with Milli-Q® water. By appropriate dilution with Milli-Q® water, a seven-point calibration curve of 0.02, 0.1, 0.5, 1.0, 5.0, 10.0, and 20.0 μg/mL was constructed. QC-samples were prepared from a 500 μg/mL methanolic stock solution and a 5.0 μg/mL aqueous working solution. QC samples were prepared in Milli-Q® water low, mid, and high level (0.05, 3.75, and 11.0 μg/mL) by appropriate dilution of the QC-working solution.
Internal standard stock solution (1000 μg/mL in methanol) was prepared by adding appropriate amounts of Cefuroxime-d3 and Metronidazole-d4 (Toronto Research Chemicals, Canada). An internal standard working solution (10 μg/mL) was prepared by dilution of stock solution with methanol.
4.4.2. Sample Preparation
To 25 μL blank, calibrator QC and patient sample, 12.5 μL internal standard working internal standard solution (10 μg/mL) was added. 75 μL acetonitrile was added to precipitate proteins. 625 μL 10 mM aqueous ammonium acetate with 5% methanol (v/v %) was added and mixed completely. Samples were centrifuged for 20 min. at 3750 g. After centrifugation the clear supernatant was transferred to HPLC vials.
4.4.3. Liquid Chromatography Mass Spectrometry Analysis
Cefuroxime and metronidazole were separated on a Phenomenex Gemini C18, 50 x 3.0 mm, 3 μm column. using gradient elution, mobile phase A (10 mM aqueous ammonium acetate with 5% methanol (v/v %)) and mobile phase B (10 mM ammonium acetate in methanol). Initial conditions were 97% A, and was maintained for 1.0 min, then B was increased to 95% over 5 min. The 95% B was kept for 1.0 min., then initial conditions was re-established within 0.3 min. Complete runtime was 10.0 min. Retention times for cefuroxime and mitronidazole was 6.35 min and 5.65 min. respectively.
Heated electrospray at 250 °C was used for ionization. Cefuroxime was detected by monitoring the transitions: m/z 423.6 to m/z 207.1 and m/z 423,6 to m/z 318.1. Metronidazole was detected by monitoring the transitions: m/z 172.0 to m/z 82.1 and m/z 172.0 to m/z 128.0.
Cefuroxime-d3 were detected by monitoring transitions the: m/z 426.6 to m/z 210.1 and m/z 426,6 to m/z 321.1. Metronidazole-d4 were detected by monitoring transitions: m/z 176.0 to m/z 82.1 and m/z 128.0 respectively.
4.5. Statistics
No power calculation was performed as this was an explorative observational study.
We registered the number of samples where the MIC in the collected dialysates reached 4 μg/mL for metronidazole and 4 and 16 μg/mL for cefuroxime, respectively.
We compared mean concentration between the two patient groups and between the two different collection methods in relation to antibiotic administration in the IBD group by linear mixed models. The variations of concentrations within each participant and variations between group members is reported as coefficient of variation (CV).
5. Conclusion
In conclusion the peritoneal microdialysis is a feasible method to study the diffusion of antibiotics into the peritoneal cavity. A method with measuring the accumulative concentration of the antibiotics in the peritoneal fluid corresponding to interval of drug administration may have the potential to add important information to the traditional pharmacodynamic parameters and relevant to reach optimal therapeutic effect.
Author Contributions
Conceptualization, K.J., M.B.E., J.E.S. and N.Q.; Methodology, K.J., M.B.E., P.D., P.B.F., J.E.S., A.R.A. and N.Q.; Validation, K.J., M.B.E., P.D., P.B.F., S.M., J.E.S. and N.Q.; Formal analysis, K.J., P.D., P.B.F., S.M., J.E.S., A.R.A. and N.Q.; Investigation, K.J., P.D., E.M.G., J.E.S., A.R.A. and N.Q.; Data curation, K.J., P.D., P.B.F., S.M., J.E.S. and N.Q.; Writing—original draft, K.J.; Writing—review & editing, M.B.E., P.D., Palle B. F., S.M., E.M.G., J.E.S., A.R.A. and N.Q.; Supervision, N.Q.; Project administration, K.J. and N.Q. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Danish Colitis-Crohn Association.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and by approvals from the Regional Scientific Ethical Committees for Southern Denmark (ID: S- 20130018 and ID: S- S-20190095), and the Danish Data Protection Agency (19/36273).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data is contained within the article.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Eckmann, C.; Kramer, A.; Assadian, O.; Flessa, S.; Huebner, C.; Michnacs, K.; Muehlendyck, C.; Podolski, K.M.; Wilke, M.; Heinlein, W.; Leaper, D.J. Clinical and Economic Burden of Surgical Site Infections in Inpatient Care in Germany. A Retrospective, Cross-sectional Analysis from 79 Hospitals. PLoS ONE 2022, 17, e0275970. [Google Scholar] [CrossRef] [PubMed]
- Marzoug, O.A.; Anees, A.; Malik, E. M. Assessment of Risk Factors Associated with Surgical Site Infection Following Abdominal Surgery. A Systematic Review. BMJ Surg. Interv. Health Technol. 2023, 5, e000182. [Google Scholar] [CrossRef] [PubMed]
- Craig, W.A. Does the Dose Matter? Clin. Infect. Dis. 2001, 33, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Reissier, S.; Penven, M.; Guérin, F.; Cattoir, V. Recent Trends in Antimicrobial Resistance Among Anaerobic Clinical Icolates. Microorganisms 2023, 11, 1474. [Google Scholar] [CrossRef] [PubMed]
- European Committee on Antimicrobial Susceptibility Testing. Available online: https://mic.eucast.org/search/?search%5Bmethod%5D=mic&search%5Bantibiotic%5D=58&search%5Bspecies%5D=-1&search%5Bdisk_content%5D=-1&search%5Blimit%5D=50 (accessed on 18 february 2026).
- Marchand, S.; Chauzy, A.; Dahyot-Fizelier, C.; Couet, W. Microdialysis as a Way to Measure Antibiotics Concentration in Tissues. Pharmacol. Res. 2016, 111, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Kowalaska-Krochmal, B.; Dudek-Vicher, R. The Minimum Inhibitory Concentration of Antibiotics. Methods, Interpretation, Clinical Relevance. Pathogens 2021, 10, 165. [Google Scholar]
- Dhanani, J.A.; Parker, S.L; Lipman, J.; Wallis, S.C.; Cohen, J.; Fraser, J.; Barnett, A.; Chew, M.; J.; Roberts, J.A. Recovery Rates of Combination Antibiotic Therapy using In Vitro Microdialysis Simulation In Vivo Conditions. J. Pharm. Anal. 2018, 6, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Skhirtladeze-Dworschak, K.; Hutschala, D.; Reining, G.; Dittrich, P.; Bartunek, A.; Dworschak, M.; Tschernko, E.M. Cefuroxime Plasma and Tissue Concentrations in Patients Undergoing Elective Cardiac Surgery. A Pilot Study. Br. J. of Clin. Pharmacol. 2019, 85, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Karjagin, J.; Pähkla, R.; Karki, T.; Starkopf, J. Distribution of Metronidazole in Muscle Tissue of Patients with Septic Shock and its Efficacy against Bactroides Fragiles In Vitro. J. Antimicrob. Chemother. 2005, 55, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Burau, D.; Petroff, D.; Simon, P.; Ehmann, L.; Weiser, C.; Dorn, C.; Kratzer, A.; Wrigge, H.; Kloft, C. Drug Combinations and Impact of Experimental Conditions on Relative Recovery in In Vitro Microdialysis Investigations. Eur. J. Pharm. Sci. 2019, 127, 252–260. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).