Submitted:
18 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
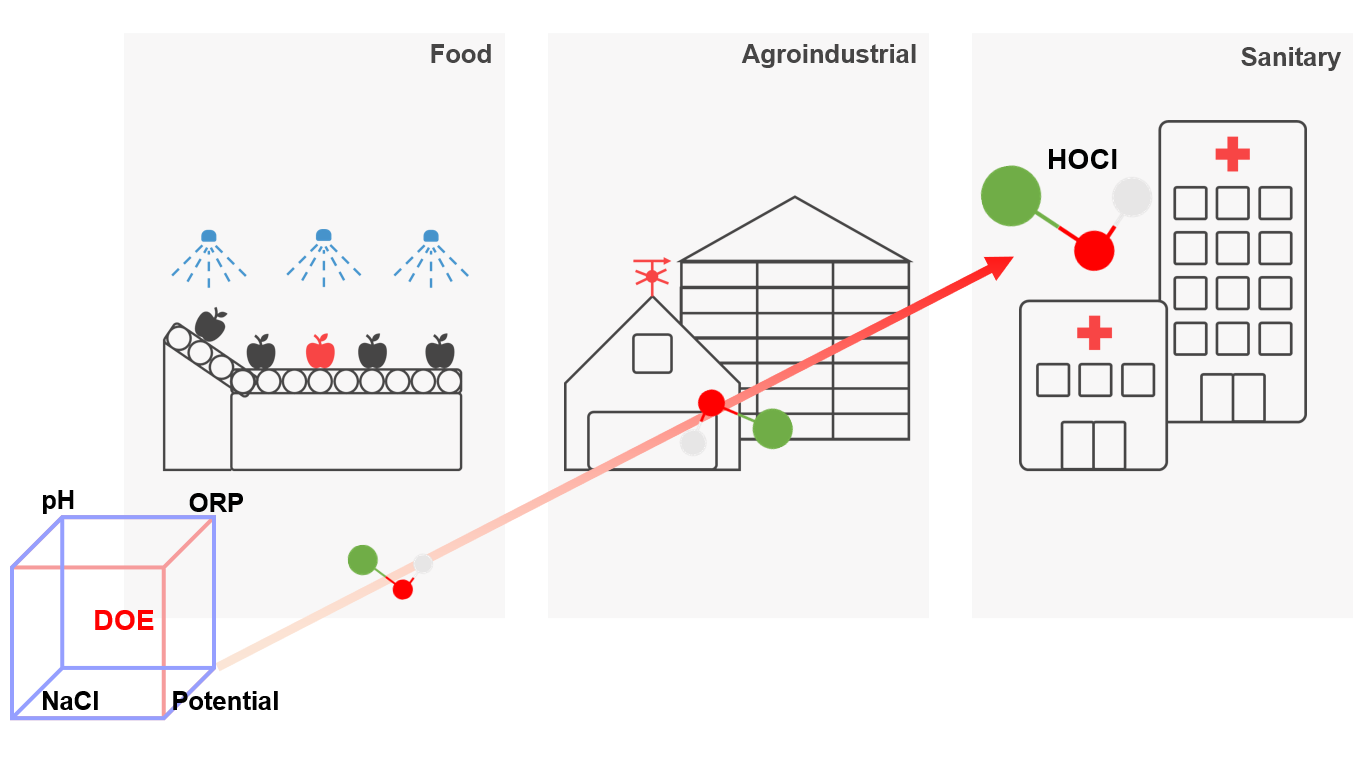
1. Introduction
2. Materials and Methods
2.1. SOW Production
2.2. DOE Study
2.3. Stability Monitoring
2.4. Microbial Culture
2.5. Agar Diffusion Assay
3. Results
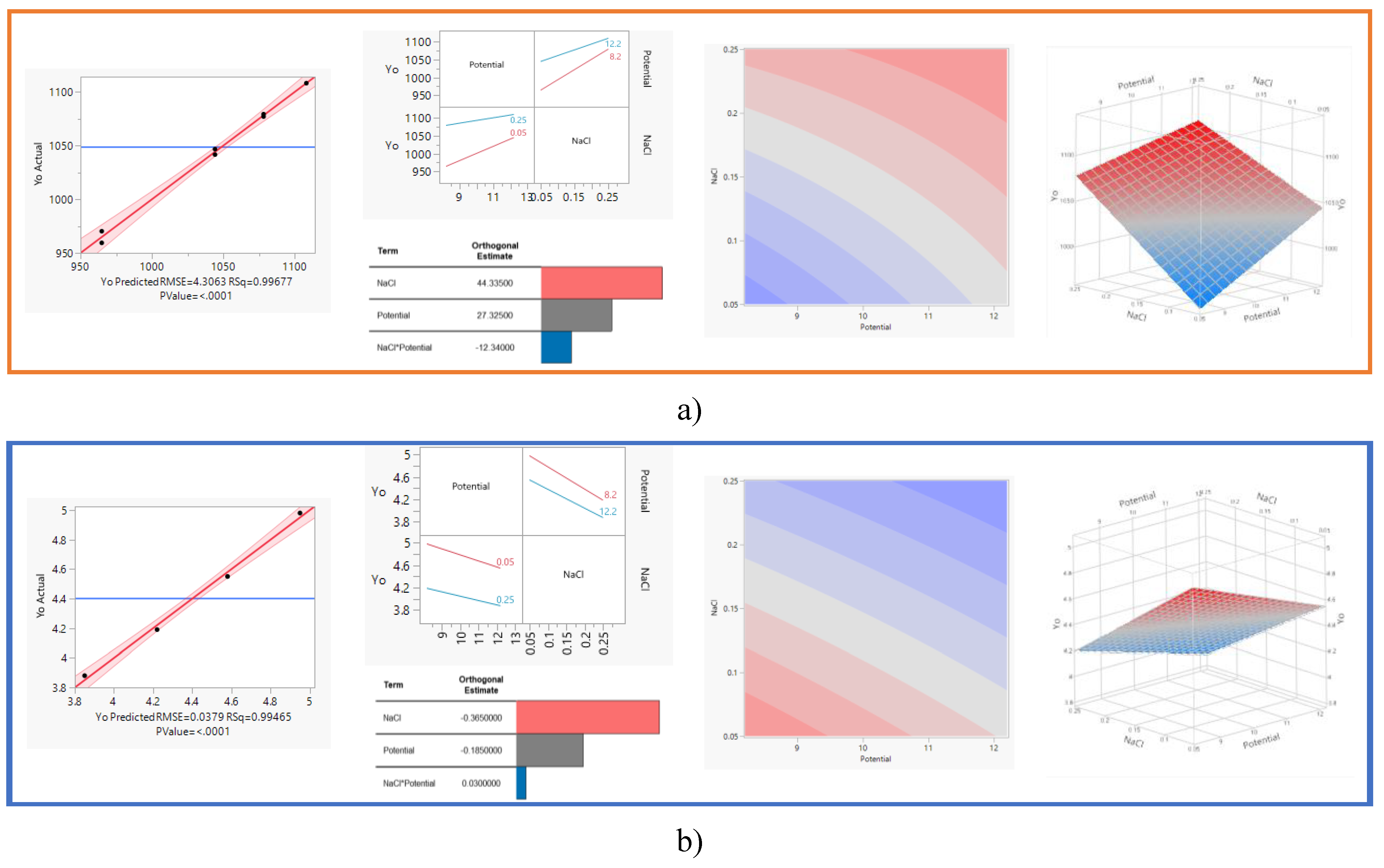
3.1. DOE Study
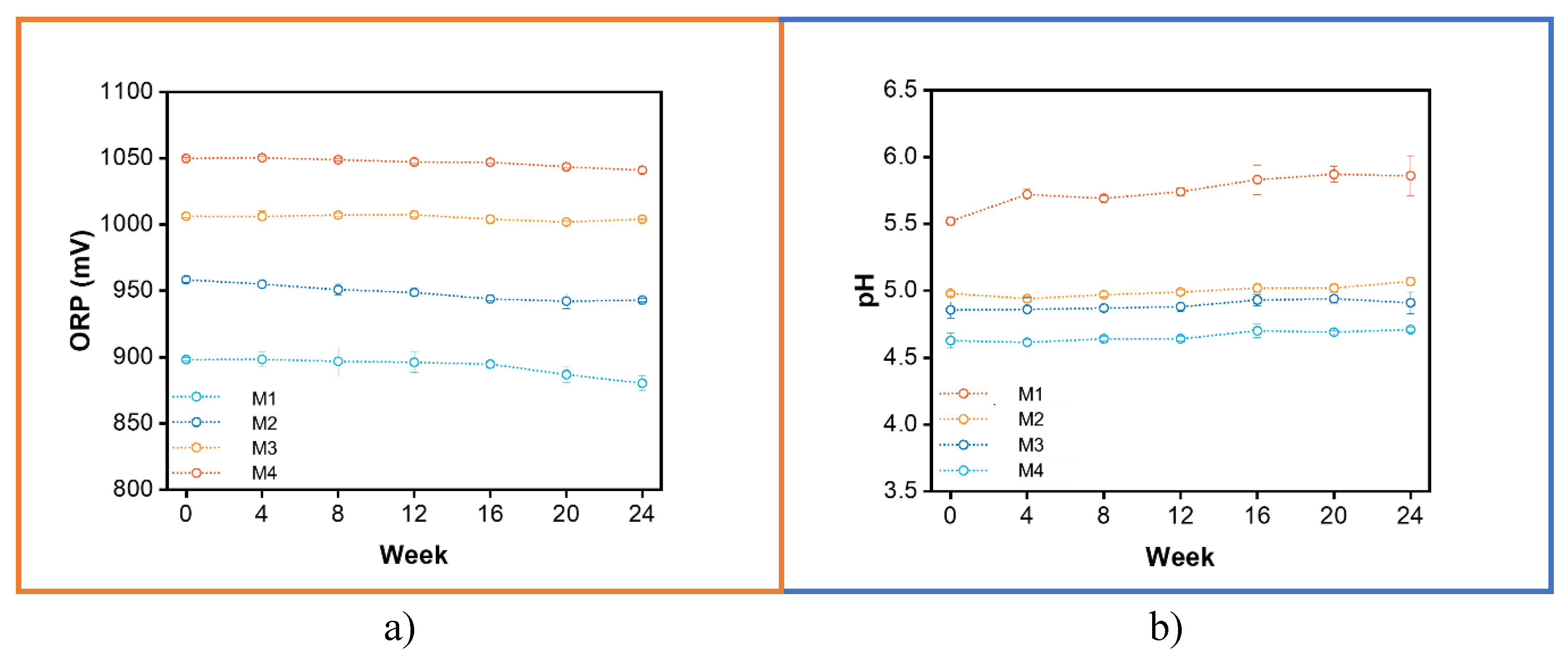
3.2. SOW Stability
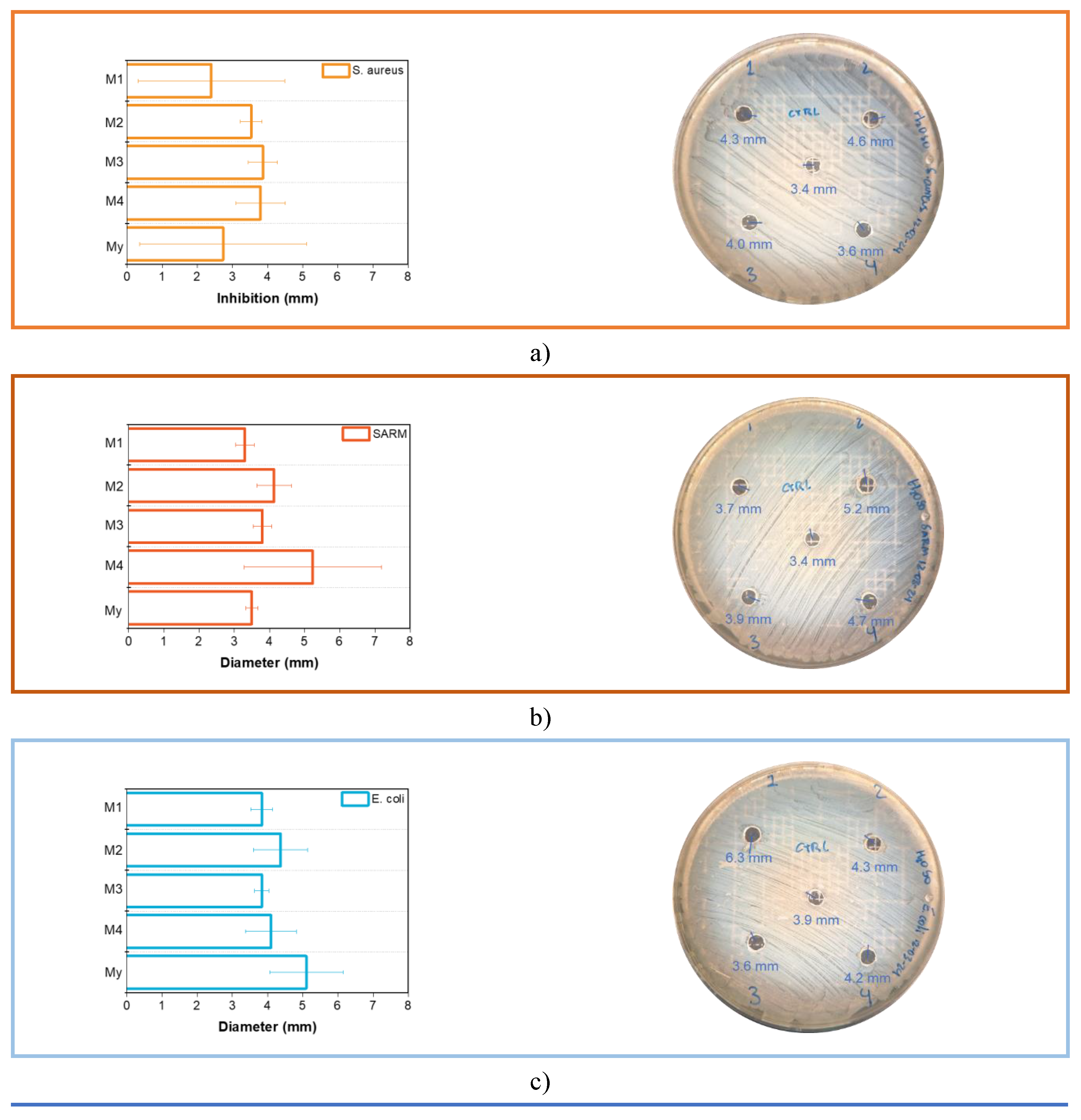
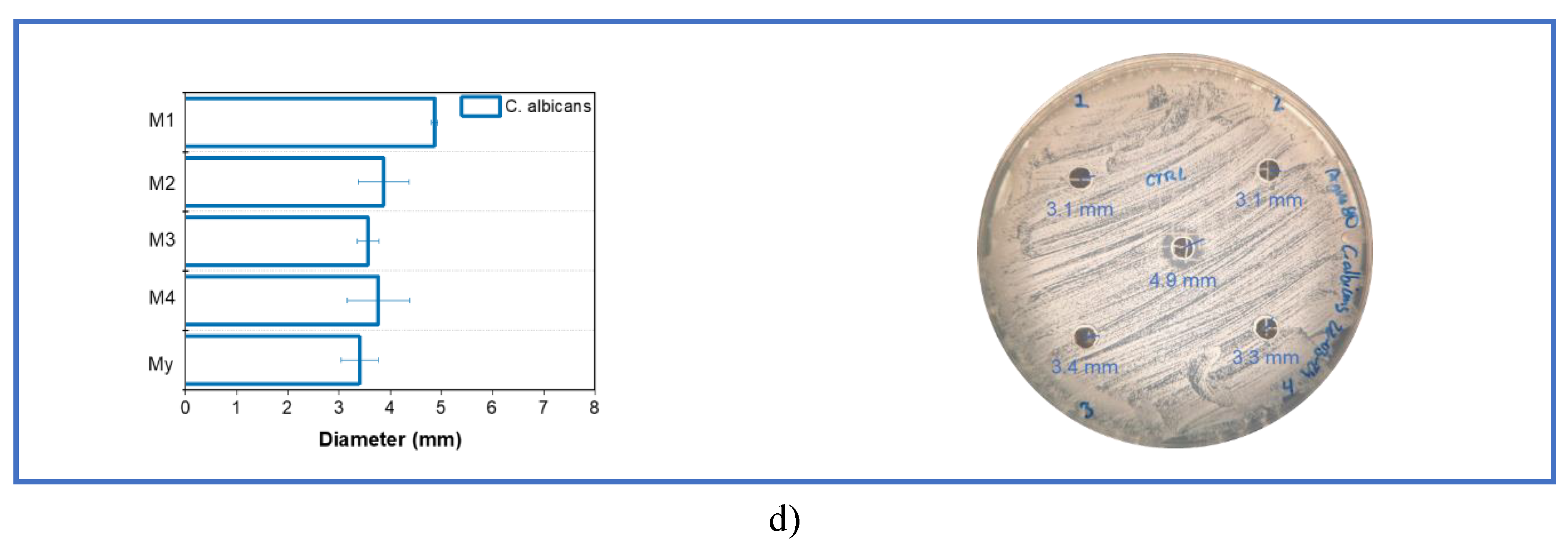
3.3. Microbial Evaluation
4. Discussion
5. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rebezov, M.; Saeed, K.; Khaliq, A.; Rahman, S.J.U.; Sameed, N.; Semenova, A.; et al. Application of Electrolyzed Water in the Food Industry: A Review. Appl. Sci. 2022, 12, 6639. [Google Scholar] [CrossRef]
- Yan, P.; Daliri, E.B.M.; Oh, D.H. New Clinical Applications of Electrolyzed Water: A Review. Microorganisms 2021, 9, 136. [Google Scholar] [CrossRef]
- Saxena, J.; Williams, T. Electrolysed water (hypochlorous acid) generation and efficacy against food-borne pathogens. Sustain. Food Technol. 2023, 1, 603–609. [Google Scholar] [CrossRef]
- Saikumar, A.; Singh, A.; Kaur, K.; Kumar, N.; Sharma, S.; Dobhal, A.; et al. Numerical optimization of hypochlorous acid (HOCl) treatment parameters and its effect on postharvest quality characteristics of tomatoes. J. Agric. Food Res. 2023, 14, 100762. [Google Scholar] [CrossRef]
- Haralović, V.; Mokos, M.; Špoljar, S.; Dolački, L.; Šitum, M.; Lugović-Mihić, L. Hypochlorous Acid: Clinical Insights and Experience in Dermatology, Surgery, Dentistry, Ophthalmology, Rhinology, and Other Specialties. Biomedicines 2025, 13, 2921. [Google Scholar] [CrossRef]
- Palau, M.; Muñoz, E.; Lujan, E.; Larrosa, N.; Gomis, X.; Márquez, E.; et al. In Vitro and In Vivo Antimicrobial Activity of Hypochlorous Acid against Drug-Resistant and Biofilm-Producing Strains. Microbiol. Spectr. 2022, 10, e02365-22. [Google Scholar] [CrossRef]
- López-Labrador, F.X.; Blasco, A.; Villamayor, L.M.; Gaume, G.; Ros-Lis, J.V. Use of Electrolysed Water as Disinfection Technology in Front of Various Microorganisms and SARS-COV-2. Food Environ. Virol. 2025, 17, 49. [Google Scholar] [CrossRef]
- Cárdenas, A.M.; Campos-Bijit, V.; Di Francesco, F.; Schwarz, F.; Cafferata, E.A.; Vernal, R. Electrolyzed water for the microbiologic control in the pandemic dental setting: a systematic review. BMC Oral Health 2022, 22, 579. [Google Scholar] [CrossRef] [PubMed]
- Valdez-Salas, B.; Salvador-Carlos, J.; Beltrán-Partida, E.; Curiel-Álvarez, M.A.; Valdez-Salas, E.; Cheng, N. Pitting Corrosion of Austenitic Stainless Steels in Electroactivated Water. Int. J. Corros. 2024, 2024, 5591969. [Google Scholar] [CrossRef]
- Ampiaw, R.E.; Yaqub, M.; Lee, W. Electrolyzed water as a disinfectant: A systematic review of factors affecting the production and efficiency of hypochlorous acid. J. Water Process Eng. 2021, 43, 102228. [Google Scholar] [CrossRef]
- Tong, W.; Forster, M.; Dionigi, F.; Dresp, S.; Sadeghi Erami, R.; Strasser, P.; et al. Electrolysis of low-grade and saline surface water. Nat. Energy 2020, 5, 367–377. [Google Scholar] [CrossRef]
- Iram, A.; Wang, X.; Demirci, A. Electrolyzed Oxidizing Water and Its Applications as Sanitation and Cleaning Agent. Food Eng. Rev. 2021, 13, 411–427. [Google Scholar] [CrossRef] [PubMed]
- Issa-Zacharia, A. Application of Slightly Acidic Electrolyzed Water as a Potential Sanitizer in the Food Industry. J. Food Qual. 2024, 2024, 1–18. [Google Scholar] [CrossRef]
- Ramírez Orejel, J.C.; Cano-Buendía, J.A. Applications of Electrolyzed Water as a Sanitizer in the Food and Animal-By Products Industry. Processes 2020, 8, 534. [Google Scholar] [CrossRef]
- Li, Z.; Li, X.; Yang, D.; Li, S.; Yu, K.; Yan, W.; et al. Revolutionizing Strongly Acidic Electrolyzed Water Production: A Breakthrough in Membrane-Free Pump-Suction Electrochemical System. Sep. Purif. Technol. 2025, 361, 131449. [Google Scholar] [CrossRef]
- Sanzani, S.; Catalano, P.; Tanucci, G.; Giametta, F.; Ippolito, A.; Bianchi, B. Design and Construction and Energy Consumption Study of a New Electrolyzed Water Cell Generator Prototype for Food Disinfection. Sci. 2024, 6, 43. [Google Scholar] [CrossRef]
- Umimoto, K.; Nagata, S.; Yanagida, J. Development of device producing electrolyzed water for home care. J. Phys. Conf. Ser. 2013, 450, 012035. [Google Scholar] [CrossRef]
- Salvador Carlos, J.; Valdez Salas, B.; Beltran Partida, E.A.; Curiel Alvarez, M.; Cheng, N.; Valdez Salas, E. Rapid optimization of disinfectants by photographic image-based analysis and design of experiments. DYNA 2024, 99, 154–158. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, C.; Yoon, J. The effect of electrode material on the generation of oxidants and microbial inactivation in the electrochemical disinfection processes. Water Res. 2009, 43, 895–901. [Google Scholar] [CrossRef]
- Gombas, D.; Luo, Y.; Brennan, J.; Shergill, G.; Petran, R.; Walsh, R.; et al. Guidelines To Validate Control of Cross-Contamination during Washing of Fresh-Cut Leafy Vegetables. J. Food Prot. 2017, 80, 312–330. [Google Scholar] [CrossRef] [PubMed]
- Damalerio, R.G.; Orbecido, A.H.; Uba, M.O.; Cantiller, P.E.L.; Beltran, A.B. Storage Stability and Disinfection Performance on Escherichia coli of Electrolyzed Seawater. Water 2019, 11, 980. [Google Scholar] [CrossRef]
- Castro, D.; Ferreri, I.; Carvalho, I.; Henriques, M. Long-lasting multi-surface disinfectant: Evaluation of efficiency and durability. Results Eng. 2022, 16, 100649. [Google Scholar] [CrossRef]
- Rossi-Fedele, G.; Dogramaci, E.J.; Steier, L.; De Figueiredo, J.A.P. Some factors influencing the stability of Sterilox®, a super-oxidised water. Br. Dent. J. 2011, 210, E23. [Google Scholar] [CrossRef]
- Len, S.V.; Hung, Y.C.; Chung, D.; Anderson, J.L.; Erickson, M.C.; Morita, K. Effects of Storage Conditions and pH on Chlorine Loss in Electrolyzed Oxidizing (EO) Water. J. Agric. Food Chem. 2002, 50, 209–212. [Google Scholar] [CrossRef]
- Block, Z.; Eyles, A.; Corkrey, R.; Stanley, R.; Ross, T.; Kocharunchitt, C. Effect of Storage Conditions on Shelf Stability of Undiluted Neutral Electrolyzed Water. J. Food Prot. 2020, 83, 1838–1843. [Google Scholar] [CrossRef]
- Sen, T.S.; Huat, L.L.; Andrianto, V.K. Effects of storage conditions on superoxidised water. MOJ Food Process. Technol. 2023, 11, 51–54. [Google Scholar] [CrossRef]
- Ashby, L.V.; Springer, R.; Hampton, M.B.; Kettle, A.J.; Winterbourn, C.C. Evaluating the bactericidal action of hypochlorous acid in culture media. Free Radic. Biol. Med. 2020, 159, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, C.S.; Milne, T.J.; Tompkins, G.R.; Cannon, R.D.; Lamping, E. Molecular Mechanism of Action of HOCl from Neutral-pH Electrolysed Oxidising Water Against Candida albicans. J. Fungi 2025, 11, 761. [Google Scholar] [CrossRef]
- Kim, C.; Hung, Y.C.; Brackett, R.E. Roles of Oxidation–Reduction Potential in Electrolyzed Oxidizing and Chemically Modified Water for the Inactivation of Food-Related Pathogens. J. Food Prot. 2000, 63, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Valdez-Salas, B.; Beltran-Partida, E.; Cheng, N.; Salvador-Carlos, J.; Valdez-Salas, E.A.; Curiel-Alvarez, M.; et al. Promotion of Surgical Masks Antimicrobial Activity by Disinfection and Impregnation with Disinfectant Silver Nanoparticles. Int. J. Nanomed. 2021, 16, 2689–2702. [Google Scholar] [CrossRef]
- Landa-Solis, C.; González-Espinosa, D.; Guzmán-Soriano, B.; Snyder, M.; Reyes-Terán, G.; Torres, K.; et al. Microcyn™: a novel super-oxidized water with neutral pH and disinfectant activity. J. Hosp. Infect. 2005, 61, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Velazquez-Meza, M.E.; Hernández-Salgado, M.; Sánchez-Alemán, M.A. Evaluation of the Antimicrobial Activity of a Super Oxidized Solution in Clinical Isolates. Microb. Drug Resist. 2015, 21, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Chekabab, S.M.; Paquin-Veillette, J.; Dozois, C.M.; Harel, J. The ecological habitat and transmission of Escherichia coli O157:H7. FEMS Microbiol. Lett. 2013, 341, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Elomri, A.; Kerbache, L.; El Omri, A. Impacts of COVID-19 on Global Supply Chains: Facts and Perspectives. IEEE Eng. Manag. Rev. 2020, 48, 153–166. [Google Scholar] [CrossRef]
- Moosavi, J.; Fathollahi-Fard, A.M.; Dulebenets, M.A. Supply chain disruption during the COVID-19 pandemic: Recognizing potential disruption management strategies. Int. J. Disaster Risk Reduct. 2022, 75, 102983. [Google Scholar] [CrossRef]
|
Potential (V) |
NaCl (%wt.) |
ORP (mV) |
| 8.2 | 0.25 | 1079.3 ± 0.62 |
| 8.2 | 0.25 | 1077.1 ± 1.30 |
| 12.2 | 0.25 | 1108.17 ± 0.11 |
| 8.2 | 0.05 | 959.43 ± 3.12 |
| 8.2 | 0.05 | 950.27 ± 4.07 |
| 12.2 | 0.05 | 1046.73 ± 2.82 |
| 12.2 | 0.05 | 1041.63 ± 1.73 |
| 12.2 | 0.25 | 1120.7 ± 1.21 |
|
Potential (V) |
NaCl (%wt.) |
pH |
| 8.2 | 0.05 | 4.98 ± 0.16 |
| 12.2 | 0.25 | 3.88 ± 0.05 |
| 12.2 | 0.05 | 4.55 ± 0.13 |
| 8.2 | 0.05 | 4.94 ± 0.07 |
| 8.2 | 0.25 | 4.13 ± 0.04 |
| 12.2 | 0.05 | 4.51 ± 0.07 |
| 8.2 | 0.25 | 4.19 ± 0.05 |
| 12.2 | 0.25 | 3.84 ± 0.05 |
| Sample name | Target ORP (mV) |
Potential (V) |
NaCl (g) |
Resulted ORP (mV) |
Resulted pH |
| M1 | 950 | 8.2 | 0.05 | 964 ± 7.6 | 5.0 ± 0.03 |
| M2 | 1000 | 8.2 | 0.1158 | 1012 ± 8.4 | 4.8 ± 0.07 |
| M3 | 1050 | 10 | 0.1607 | 1060 ± 10.7 | 4.5 ± 0.06 |
| M4 | 1100 | 12.2 | 0.1927 | 1108 ± 1.5 | 4.1 ± 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




