Submitted:
22 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Parasite Culture
2.2. Animals and Peritoneal Macrophage Isolation
2.3. Macrophage Infection
2.4. Light Microscopy and Giemsa Staining
2.4.1. Promastigotes
2.4.2. Intracellular Amastigotes
2.5. Scanning Electron Microscopy (SEM)
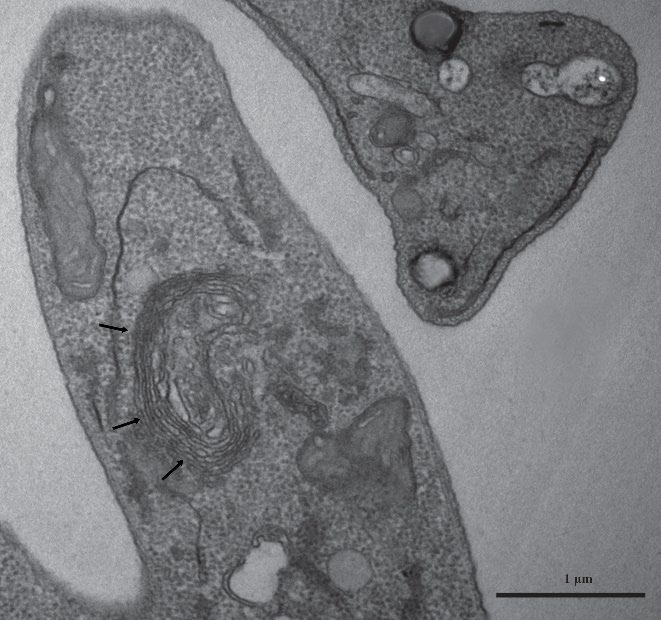
2.6. Transmission Electron Microscopy (TEM)
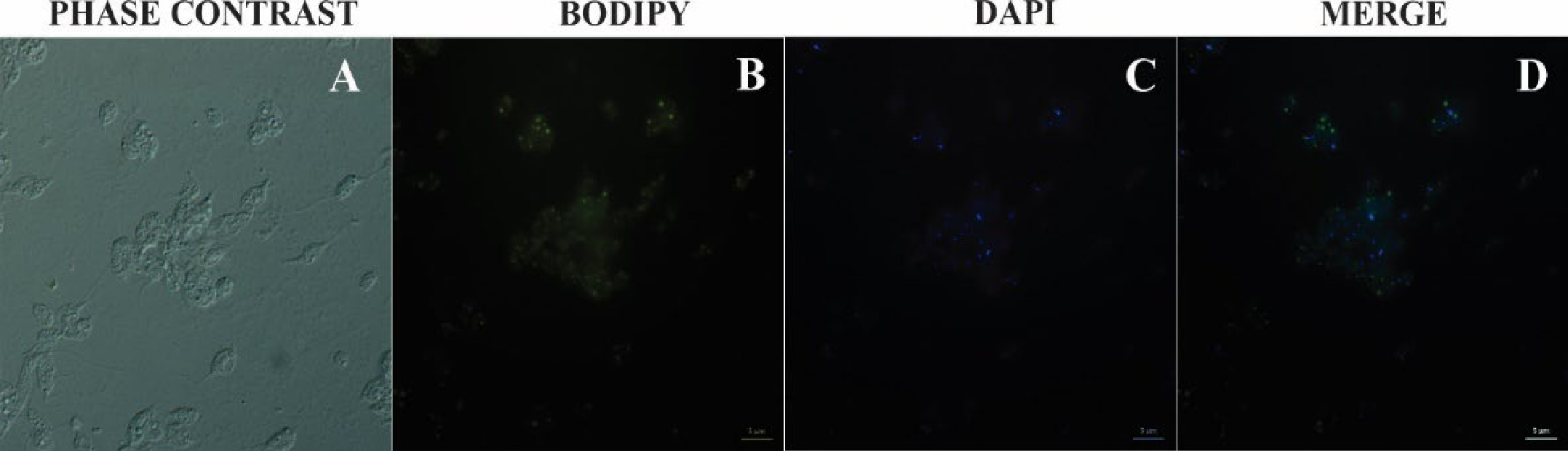
2.7. Bodipy Lipid Staining
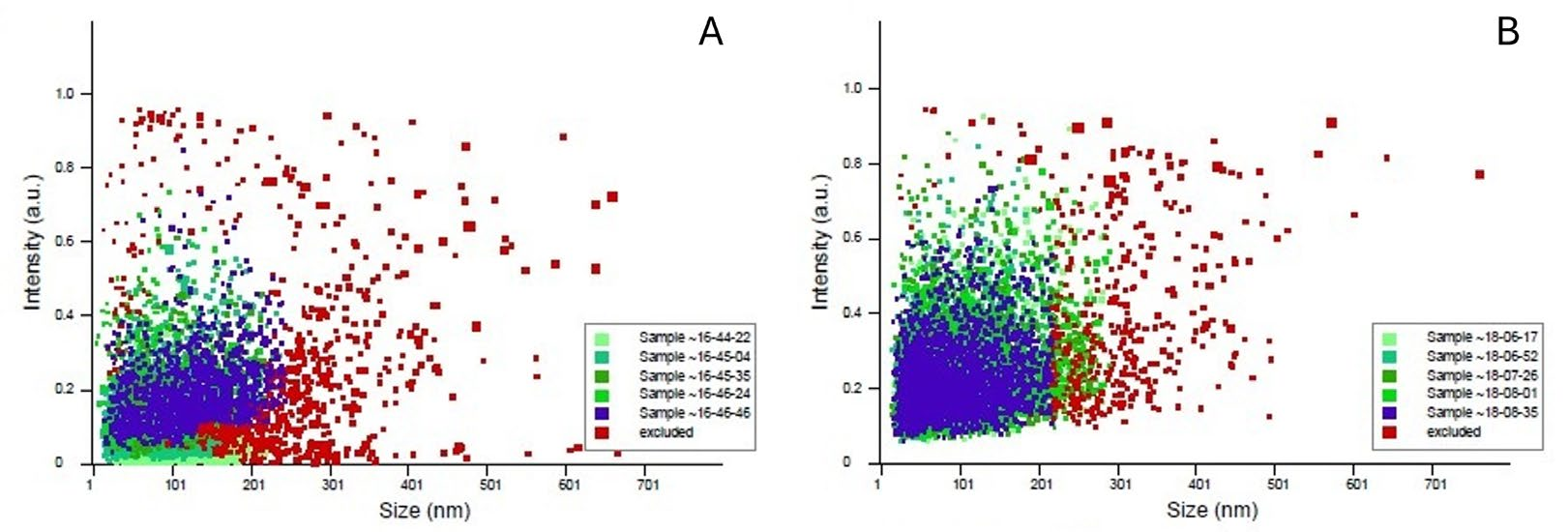
2.8. Nanoparticle Tracking Analysis (NTA)
3. Results
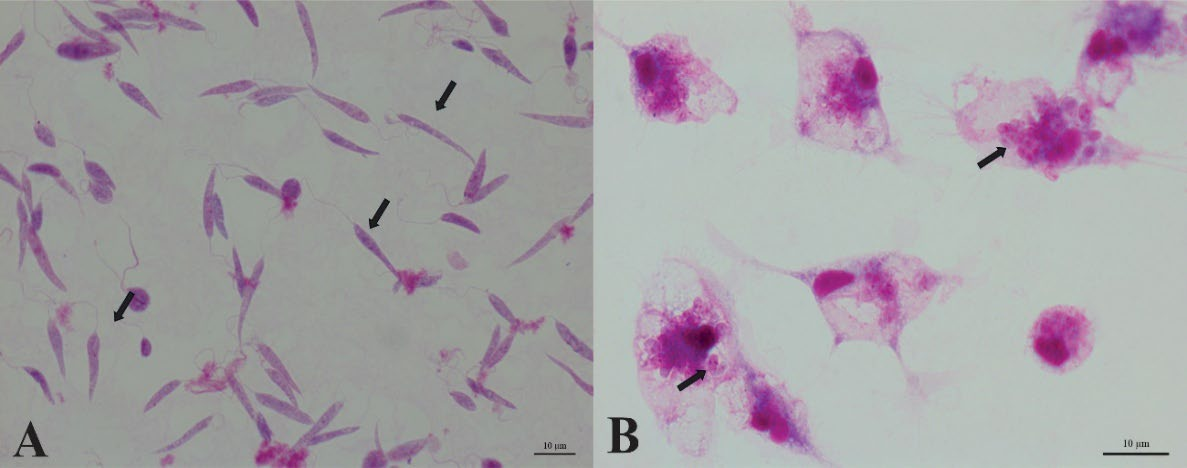
3.1. Promastigote Morphology
3.2. Intracellular Amastigotes
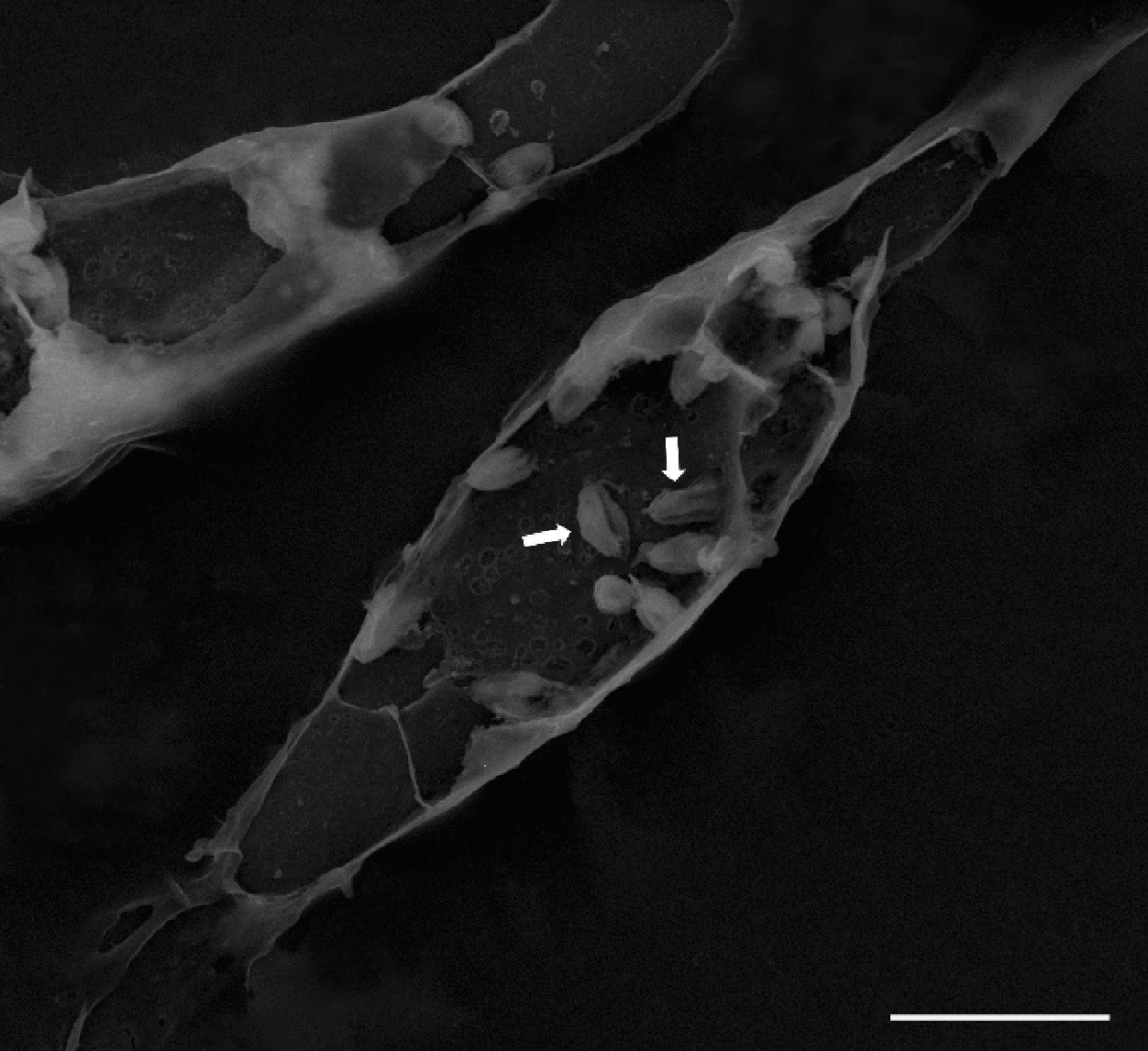
3.3. Lipid-Rich Structures
3.4. Bodipy Staining
3.5. Nanoparticle Tracking Analysis (NTA)
4. Discussion
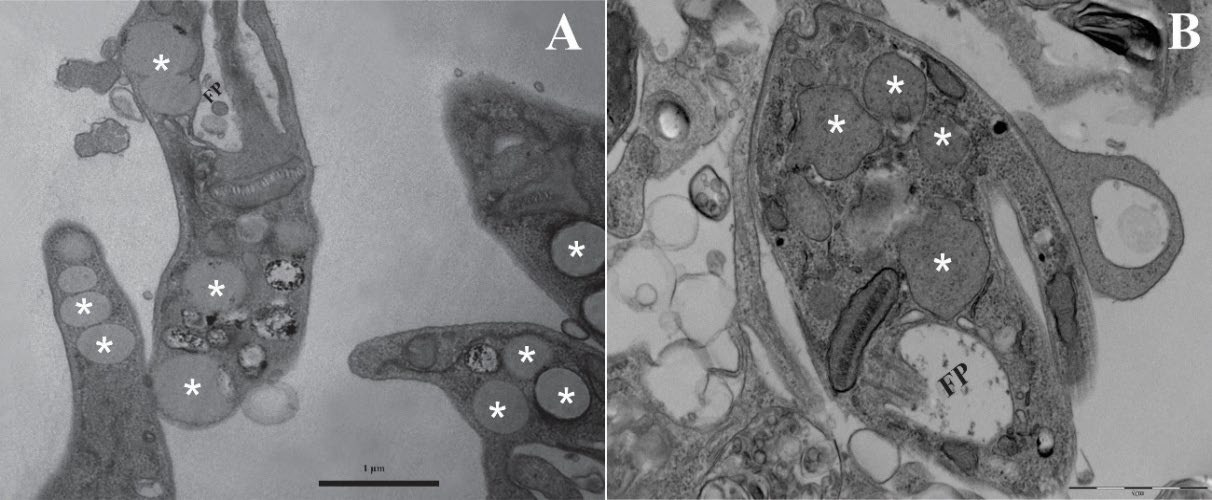
4.1. Lipid-Rich and Vesicle-like Structures in Promastigotes
4.2. Lipid-Rich Inclusions in Intracellular Amastigotes
4.3. Heterogeneity of Extracellular Particles Detected by NTA
4.4. Methodological Considerations and Limitations
- no isolation or purification of extracellular vesicles was performed;no biochemical or proteomic characterization was conducted;
- no functional assays were included;
- no organelle-specific markers were applied;
- no controls for particle purity or co-isolated contaminants were incorporated.
4.5. Implications for Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- World Health Organization (WHO). Leishmaniasis; WHO Fact Sheet, April 2017. Available online: https://www.who.int/mediacentre/factsheets/fs375/en/ (accessed on 24 July 2017).
- Atayde, V.D.; Hassani, K.; da Silva Lira Filho, A.; et al. Extracellular Vesicles in Leishmania Infection: Modulators of Host–Parasite Interaction. PLoS Pathog. 2023, 19, e1012636. [CrossRef]
- Silverman, J.M.; Clos, J.; de’Oliveira, C.C.; et al. Leishmania Exosomes Modulate Innate Immunity and Promote Infection. Proc. Natl. Acad. Sci. USA 2010, 107, 21635–21640. [CrossRef]
- Parreira de Aquino, G.; et al. Lipid Metabolism in Leishmania: Implications for Pathogenesis and Drug Resistance. Microb. Cell 2021, 8, 88–104. [CrossRef]
- Chowdhury, S.; et al. Host–Parasite Lipid Interactions in Leishmaniasis: Mechanisms and Therapeutic Perspectives. Int. J. Mol. Sci. 2022, 23, 2414. [CrossRef]
- Rodrigues, J.C.F.; et al. Metabolic Adaptations of Leishmania Parasites: Lipid Remodeling and Survival Strategies. Microorganisms 2023, 13, 531. [CrossRef]
- Carroll, S.B. Evo Devo and the Evolution of Animal Diversity. Cell 2005, 120, 201–207. [CrossRef]
- Hutchinson, G.E. Concluding Remarks. Cold Spring Harb. Symp. Quant. Biol. 1957, 22, 415–427. [CrossRef]
- Kumar, A.; et al. Lipid-Mediated Modulation of Host Immunity by Leishmania spp. Exp. Parasitol. 2021, 223, 107999. [CrossRef]
- Macedo, A.M.; et al. Lipidomic Signatures of Leishmania Extracellular Vesicles Reveal Species-Specific Patterns. Int. J. Mol. Sci. 2023, 24, 10637. [CrossRef]
- Pal, P.; Das, S.; Chatterjee, N.; Bose, D.; Saha, K.D. Studies on the Anti-Inflammatory Effect of Leishmanial Lipid In Vitro and In Vivo. Prajnan O Sadhona 2015, 2, 1–XX.
- Serrano, A.; et al. Lipid Droplets and Vesicular Trafficking in Leishmania: Ultrastructural Insights. Micron 2021, 142, 103089. [CrossRef]
- Sacks, D.; et al. Lipid Bodies in Leishmania: Structure, Function, and Role in Host Interaction. Mol. Biochem. Parasitol. 2009, 165, 1–9. [CrossRef]
- Parreira de Aquino, G.; et al. Lipid Remodeling in Leishmania: A Microbial Cell Perspective. Microb. Cell 2021, 8, 1–12. [CrossRef]
- Booth, L.-A.; Smith, T.K. Lipid Metabolism in Trypanosoma cruzi: A Review. Mol. Biochem. Parasitol. 2020, 240, 111324. [CrossRef]
- Bird, A. Perceptions of Epigenetics. Nature 2007, 447, 396–398. [CrossRef]
- Gonçalves, R.; et al. Extracellular Vesicles from Protozoan Parasites: Biogenesis, Composition, and Function. Front. Microbiol. 2016, 7, 427. [CrossRef]
- Costa, D.L.; et al. Lipid-Rich Extracellular Vesicles in Leishmania Infection: Implications for Pathogenesis. Int. J. Parasitol. 2024, 54, 1–18. [CrossRef]
- Gabriel, Á.M.; Galué Parra, A.; Pereira, W.L.A.; Pedersen, K.W.; da Silva, E.O. Leishmania 360°: Guidelines for Exosomal Research. Microorganisms 2021, 9, 2081. [CrossRef]
- Gabriel, Á.M.; Galvão, G.R.; Galué Parra, A.; Casseb, L.M.N.; Pereira, W.L.A.; Pedersen, K.W.; Aguiar, D.C.F.; Gonçalves, E.C.; da Silva, E.O. Pathogenesis of Canine Leishmaniasis: Diagnostic Accuracy and Experimental Models Targeting Leishmania Lipid-Bound Vesicles. Acad. Biol. 2025, 3(1), 1–XX. [CrossRef]
- Santos, L.C.; et al. Metabolomic Profiling of Leishmania Species Reveals Lipid Remodeling During Infection. Metabolites 2024, 14, 658. [CrossRef]
- Kim, S.E.; Ibarra-Meneses, A.V.; Fernandez-Prada, C.; Huan, T. Global Lipidomics Reveals the Lipid Composition Heterogeneity of Extracellular Vesicles from Drug-Resistant Leishmania. Metabolites 2024, 14, 658. [CrossRef]
- International Society for Extracellular Vesicles (ISEV). MISEV2023: Minimal Information for Studies of Extracellular Vesicles. J. Extracell. Vesicles 2023. Available online: https://isevjournals.onlinelibrary.wiley.com/doi/10.1002/jev2.12404 (accessed on 7 January 2026).
- Schmid-Hempel, P. Evolutionary Parasitology; Oxford University Press: Oxford, UK, 2011. ISBN 978-0199229482.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



