Submitted:
23 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Biological Material and Study Design
2.2. Lyophilization and Sample Preparation
2.3. Biphasic Extraction of Polar and Apolar Metabolites
2.4. NMR Sample Preparation
2.5. 1H NMR Data Acquisition
2.6. Spectral Processing and Data Handling
2.7. Metabolite Annotation Strategy
2.8. Statistical Analysis and Chemometric Modelling
2.9. Pathway Analysis (MetaboAnalyst)
2.10. Apolar Extract Quantification and Lipid Index Calculation
3. Results
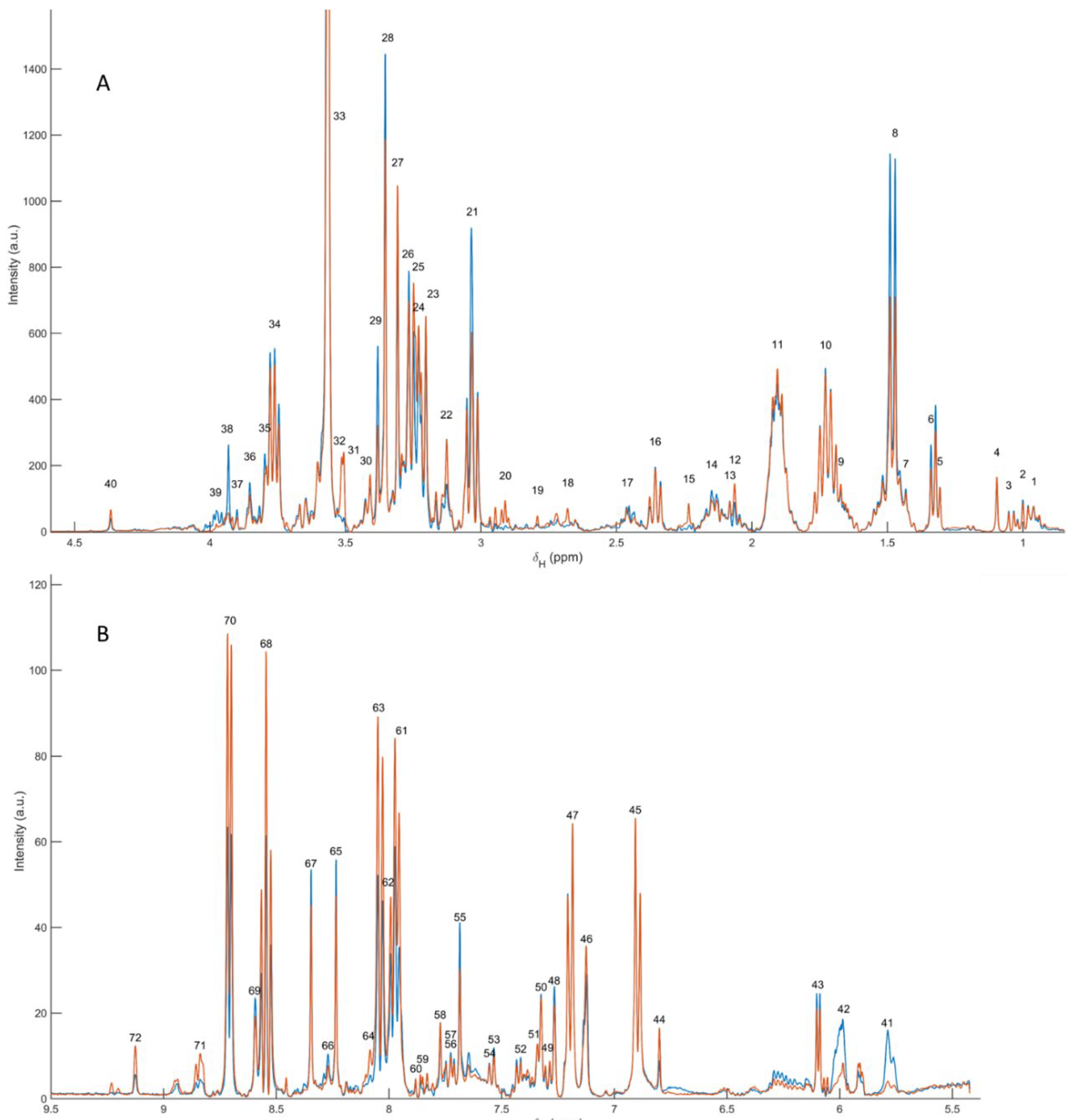
3.1. 1H NMR Spectral Overview and Metabolite Annotation (Polar Extracts)
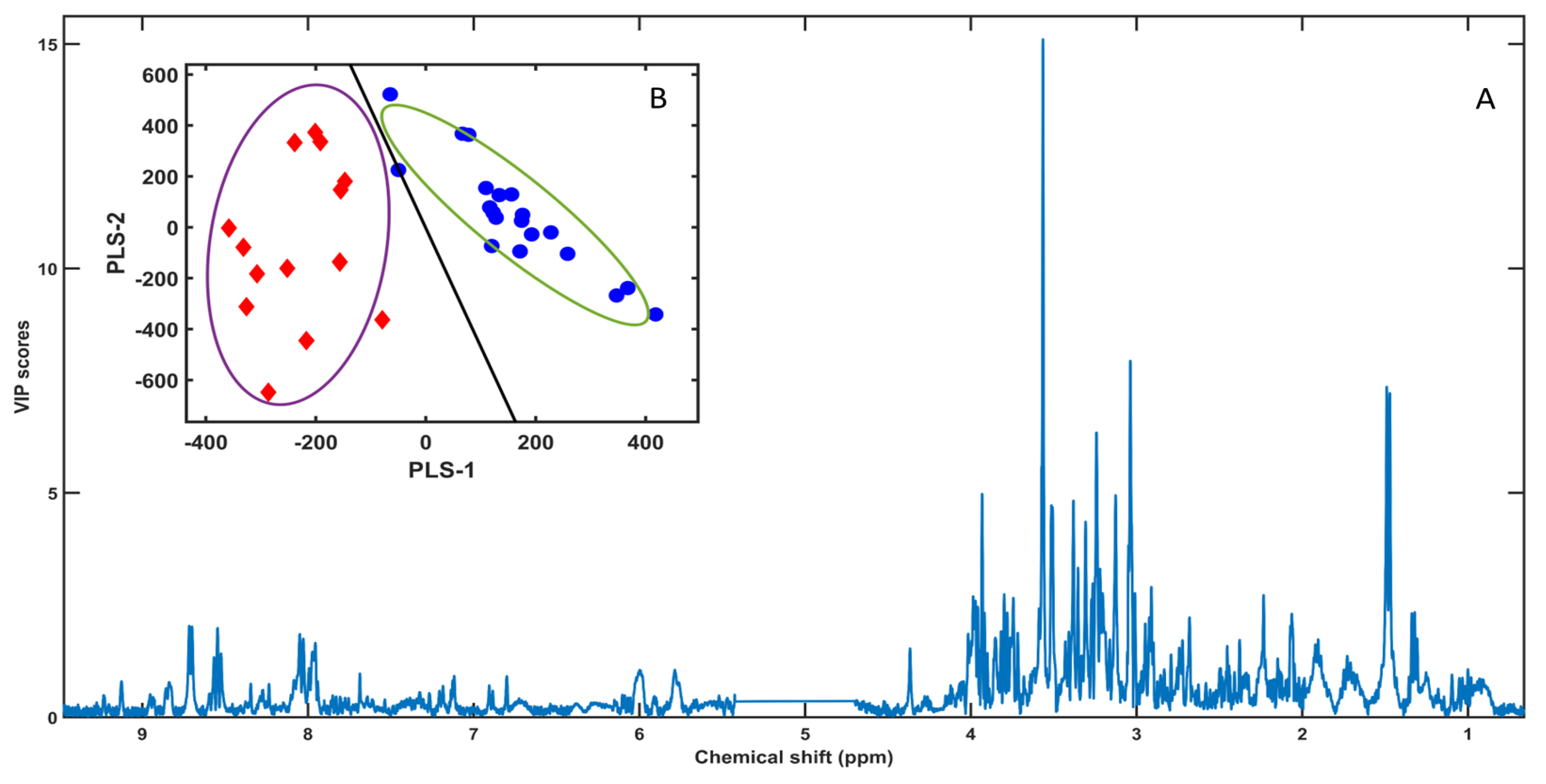
3.2. PLS-LDA Model
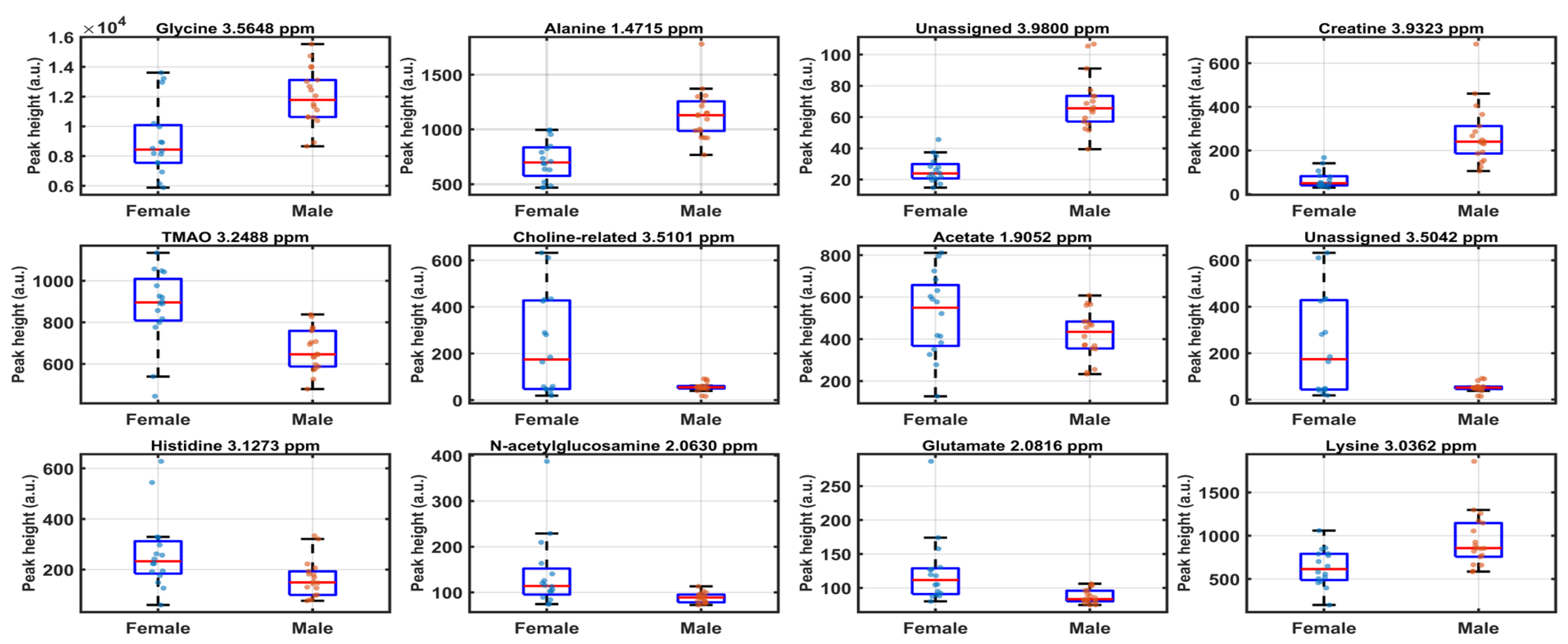
3.3. SPA-Based Variable Selection and COSS Ranking
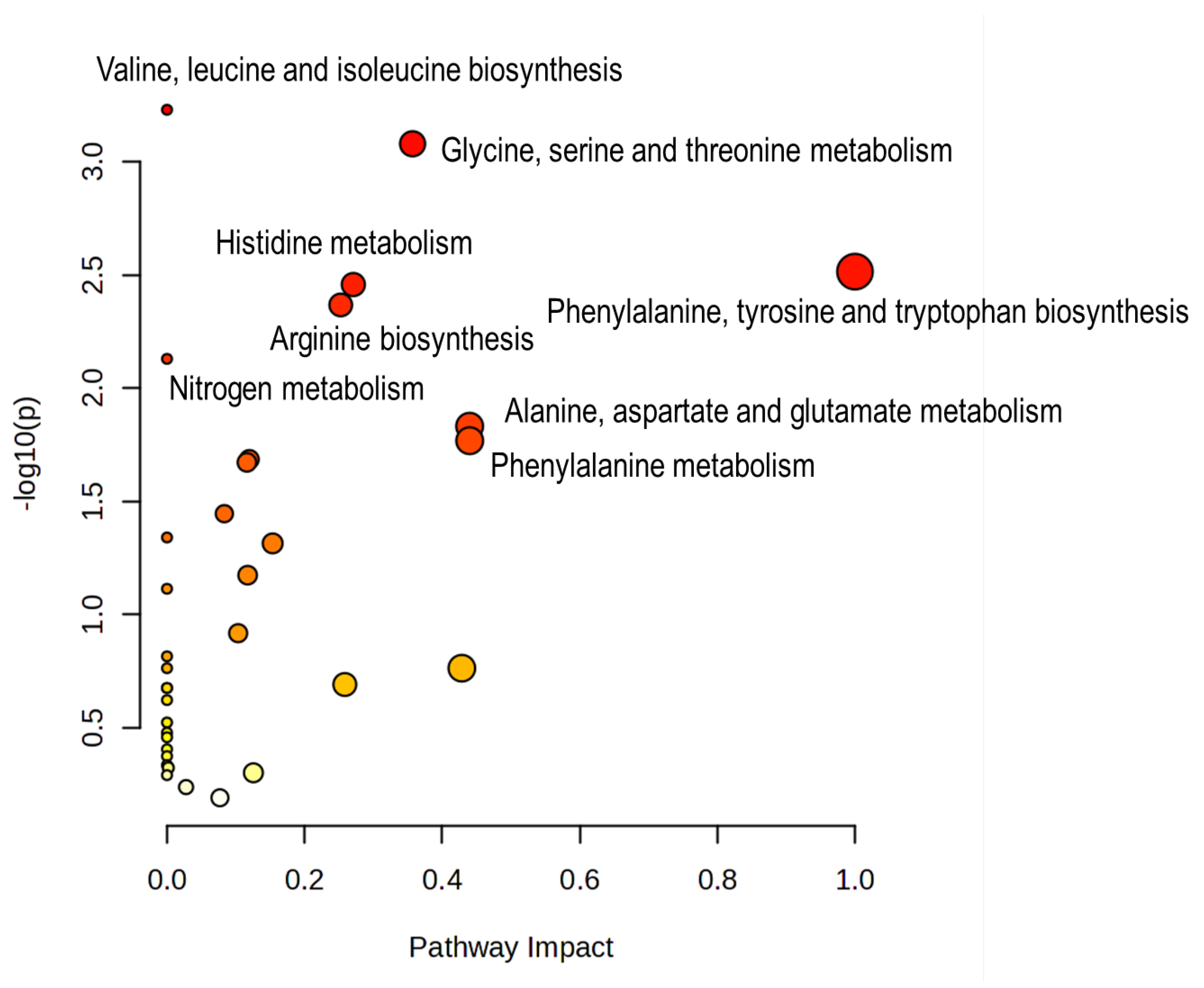
3.4. Pathway Analysis of Sex-Discriminatory Polar Metabolites
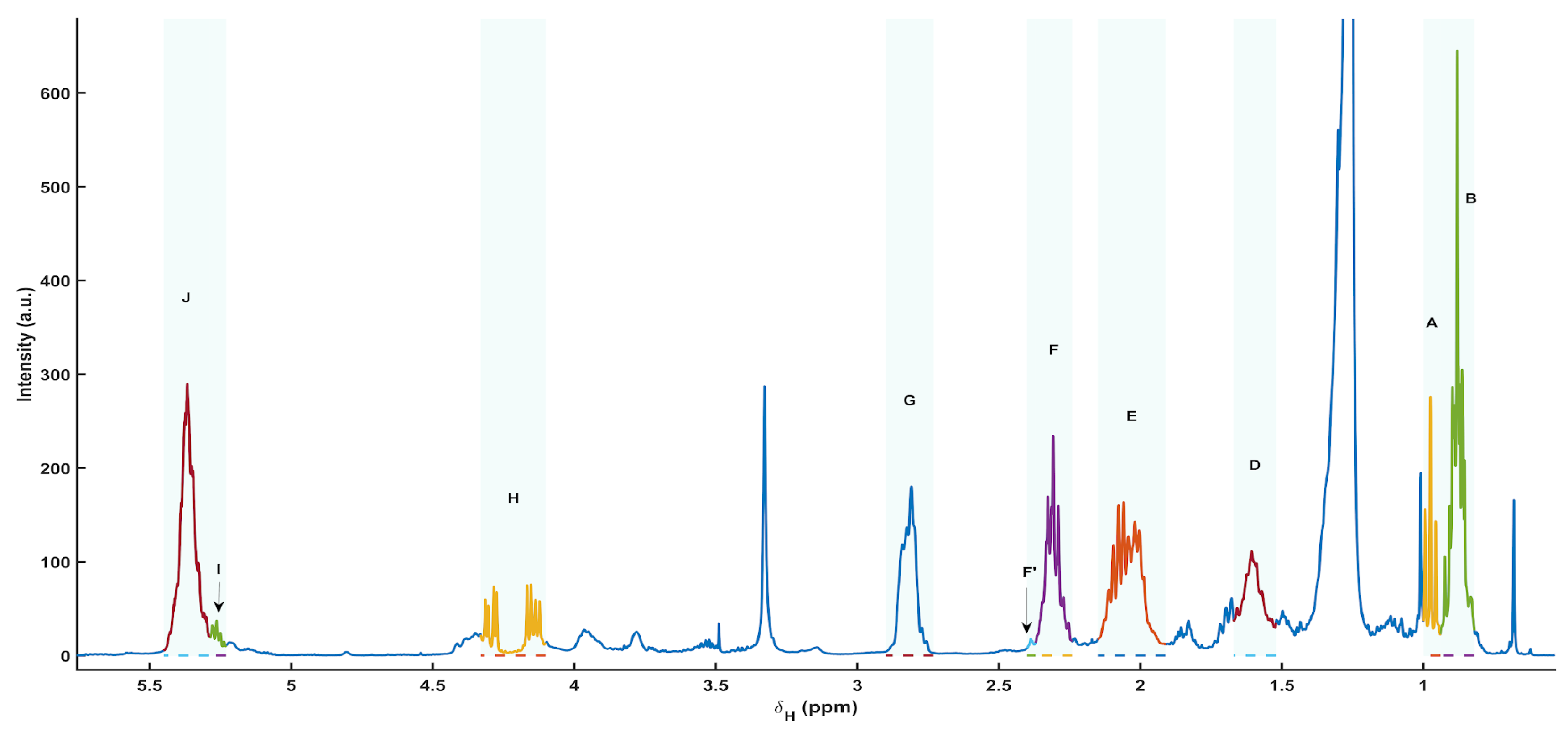
3.5. 1H NMR Spectral Overview and Metabolite Annotation (Apolar Extracts)
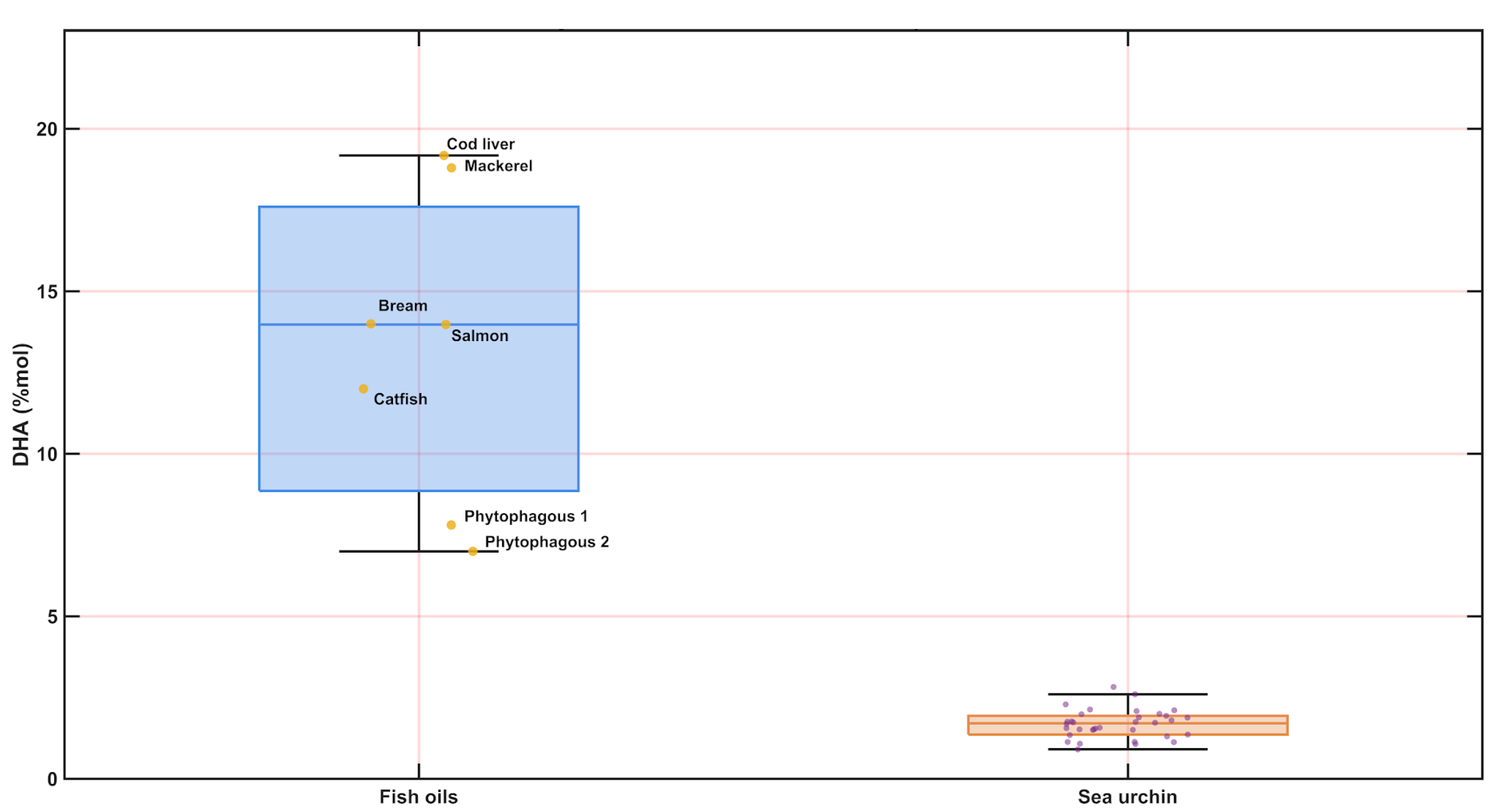
3.6. Lipid Class Estimation Using Bratu Equations and Comparison with Fish Oils
3.7. Sex-Related Differences and Multivariate Separability (Apolar Metrics)
4. Discussion
4.1. Multivariate Evidence of Robust Sex Separation and Model Validation
4.2. Discriminant Variables Converge on Osmolyte Balance, Amino-Acid Pools, and Energy Buffering
4.3. Pathway Analysis: Strong Amino-Acid and One-Carbon Signatures, with an Important Caveat on Directionality
4.4. Integrating Sex Biology of P. lividus Gonads with the Observed Metabolite Shifts
4.5. Interpretive Scope, Limitations, and Recommended Confirmatory Steps
4.6. Interpreting Apolar 1H NMR Lipid Signatures in Paracentrotus lividus Gonads: Ecological Drivers, Limitations, and Next Steps
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baião, L.F.; Moura, A.P.; Rocha, C.; Valente, L.M.P.; Cunha, L.M. Dimensions for the valorisation of sea urchin (paracentrotus lividus) gonads production through the eyes of experienced chefs. International Journal of Gastronomy and Food Science 2021, 26, 100438. [Google Scholar] [CrossRef]
- Camacho, C.; Correia, T.; Teixeira, B.; Mendes, R.; Valente, L.M.P.; Pessoa, M.F.; Nunes, M.L.; Gonçalves, A. Nucleotides and free amino acids in sea urchin paracentrotus lividus gonads: Contributions for freshness and overall taste. Food Chem 2023, 404, 134505. [Google Scholar] [CrossRef]
- Rocha, F.; Peres, H.; Diogo, P.; Ozório, R.; Valente, L.M.P. The effect of sex, season and gametogenic cycle on gonad yield, biochemical composition and quality traits of paracentrotus lividus along the north atlantic coast of portugal. PeerJ 2019, 7. [Google Scholar] [CrossRef]
- Powers, R.; Andersson, E.R.; Bayless, A.L.; Brua, R.B.; Chang, M.C.; Cheng, L.L.; Clendinen, C.S.; Cochran, D.; Copié, V.; Cort, J.R.; et al. Best practices in nmr metabolomics: Current state. Trends Analyt Chem 2024, 171. [Google Scholar] [CrossRef]
- Sumner, L.W.; Amberg, A.; Barrett, D.; Beale, M.H.; Beger, R.; Daykin, C.A.; Fan, T.W.M.; Fiehn, O.; Goodacre, R.; Griffin, J.L.; et al. Proposed minimum reporting standards for chemical analysis. Metabolomics 2007, 3, 211–221. [Google Scholar] [CrossRef]
- Nagana Gowda, G.A.; Raftery, D. Quantitative nmr methods in metabolomics. Handb Exp Pharmacol 2023, 277, 143–164. [Google Scholar] [CrossRef] [PubMed]
- Carbonell-Garzón, E.; Ibanco-Cañete, R.; Sanchez-Jerez, P.; Egea, F.C.M. Osmolytes vs. Anabolic reserves: Contrasting gonadal metabolomes in two sympatric mediterranean sea urchins. metabo 2025, 15, 787. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, W.; Ding, B.; Zhang, Y.; Huang, X.; Liu, X.; Zuo, R.; Chang, Y.; Ding, J. Comparative lipidomics profiling of the sea urchin, strongylocentrotus intermedius. Comp Biochem Physiol Part D Genomics Proteomics 2021, 40, 100900. [Google Scholar] [CrossRef]
- Amai, S.; Yuki, K.; Gowda, S.G.B.; Gowda, D.; Hui, S.-P. Lipidomic profiling of edible japanese sea urchins by lc–ms. foods 2025, 14, 2268. [Google Scholar] [CrossRef]
- Haug, K.; Cochrane, K.; Nainala, V.C.; Williams, M.; Chang, J.; Jayaseelan, K.V.; O’Donovan, C. Metabolights: A resource evolving in response to the needs of its scientific community. Nucleic Acids Research 2020, 48, D440–D444. [Google Scholar] [CrossRef]
- Bingol, K.; Li, D.W.; Zhang, B.; Brüschweiler, R. Comprehensive metabolite identification strategy using multiple two-dimensional nmr spectra of a complex mixture implemented in the colmarm web server. Anal Chem 2016, 88, 12411–12418. [Google Scholar] [CrossRef] [PubMed]
- Badaoui, W.; Marhuenda-Egea, F.C.; Valero-Rodriguez, J.M.; Sanchez-Jerez, P.; Arechavala-Lopez, P.; Toledo-Guedes, K. Metabolomic and lipidomic tools for tracing fish escapes from aquaculture facilities. ACS Food Science & Technology 2024, 4, 871–879. [Google Scholar] [CrossRef]
- Aursand, M.; Standal, I.B.; Gribbestad, I.S.; Martinez, I. Omega-3 fatty acid content of intact muscle of farmed atlantic salmon (salmo salar) examined by 1h mas nmr spectroscopy; 2018; pp. 1917–1925. [Google Scholar] [CrossRef]
- Igarashi, T.; Aursand, M.; Sacchi, R.; Paolillo, L.; Nonaka, M.; Wada, S. Determination of docosahexaenoic acid and n-3 fatty acids in refined fish oils by h-1-nmr spectroscopy: Iupac interlaboratory study. Journal of Aoac International 2002, 85, 1341–1354. [Google Scholar] [CrossRef]
- Cravero, B.H.; Prez, G.; Lombardo, V.A.; Guastaferri, F.V.; Delprato, C.B.; Altabe, S.; de Mendoza, D.; Binolfi, A. A high-resolution 13c nmr approach for profiling fatty acid unsaturation in lipid extracts and in live caenorhabditis elegans. Journal of Lipid Research 2024, 65, 100618. [Google Scholar] [CrossRef]
- Nieva-Echevarría, B.; Goicoechea, E.; Manzanos, M.J.; Guillén, M.D. Usefulness of 1h nmr in assessing the extent of lipid digestion. Food Chemistry 2015, 179, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Ibanco-Cañete, R.; Carbonell-Garzón, E.; Sanchez-Jerez, P.; Egea, F.C.M. Identification of sex-dependent aroma compounds in gonads of commercially valuable sea urchins: Implications for gastronomical use of Paracentrotus lividus. Jmse 2025, 13, 2160. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Li, H.D.; Xu, Q.S.; Liang, Y.Z. Libpls: An integrated library for partial least squares regression and linear discriminant analysis. Chemometrics and Intelligent Laboratory Systems 2018, 176, 34–43. [Google Scholar] [CrossRef]
- Li, H.-D.; Zeng, M.-M.; Tan, B.-B.; Liang, Y.-Z.; Xu, Q.-S.; Cao, D.-S. Recipe for revealing informative metabolites based on model population analysis. Metabolomics 2010, 6, 353–361. [Google Scholar] [CrossRef]
- Wang, Q.; Li, H.-D.; Xu, Q.-S.; Liang, Y.-Z. Noise incorporated subwindow permutation analysis for informative gene selection using support vector machines. Analyst 2011, 136, 1456–1463. [Google Scholar] [CrossRef] [PubMed]
- Pang, Z.; Lu, Y.; Zhou, G.; Hui, F.; Xu, L.; Viau, C.; Spigelman, A.F.; MacDonald, P.E.; Wishart, D.S.; Li, S.; et al. Metaboanalyst 6.0: Towards a unified platform for metabolomics data processing, analysis and interpretation. Nucleic Acids Research 2024, 52, W398–W406. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. Kegg: Kyoto encyclopedia of genes and genomes. Nucleic Acids Research 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Bratu, A.; Mihalache, M.; Hanganu, A.; Chira, N.; Todasca, C.; Rosca, S. Quantitative determination of fatty acids from fish oils using gc-ms method and 1h-nmr spectroscopy. UPB Scientific Bulletin, Series B: Chemistry and Materials Science 2013, 75, 23–32. [Google Scholar]
- Szymańska, E.; Saccenti, E.; Smilde, A.K.; Westerhuis, J.A. Double-check: Validation of diagnostic statistics for pls-da models in metabolomics studies. Metabolomics 2012, 8, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Broadhurst, D.I.; Kell, D.B. Statistical strategies for avoiding false discoveries in metabolomics and related experiments. Metabolomics 2006, 2, 171–196. [Google Scholar] [CrossRef]
- Westerhuis, J.A.; Hoefsloot, H.C.J.; Smit, S.; Vis, D.J.; Smilde, A.K.; van Velzen, E.J.J.; van Duijnhoven, J.P.M.; van Dorsten, F.A. Assessment of plsda cross validation. Metabolomics 2008, 4, 81–89. [Google Scholar] [CrossRef]
- Podbielski, I.; Schmittmann, L.; Sanders, T.; Melzner, F. “Acclimation of marine invertebrate osmolyte systems to low salinity: A systematic review & meta-analysis.” Frontiers in Marine Science Volume 9 - 2022 (2022): https://www.frontiersin.org/journals/marine-science/articles/10.3389/fmars.2022.934378.
- Yancey, P.H. Organic osmolytes as compatible, metabolic and counteracting cytoprotectants in high osmolarity and other stresses. Journal of Experimental Biology 2005, 208, 2819–2830. [Google Scholar] [CrossRef]
- Ellington, W.R. Arginine kinase and creatine kinase appear to be present in the same cells of an echinoderm muscle. Journal of Experimental Biology 1991, 158, 591–597. [Google Scholar] [CrossRef]
- Martínez-Pita, I.; García, F.J.; Pita, M.-L. The effect of seasonality on gonad fatty acids of the sea urchins paracentrotus lividus and arbacia lixula (echinodermata: Echinoidea). Journal of Shellfish Research 2010, 29, 517–525. [Google Scholar] [CrossRef]
- Anedda, R.; Siliani, S.; Melis, R.; Loi, B.; Baroli, M. Lipid metabolism of sea urchin paracentrotus lividus in two contrasting natural habitats. Scientific Reports 2021, 11, 14174. [Google Scholar] [CrossRef] [PubMed]
- Siliani, S.; Melis, R.; Loi, B.; Guala, I.; Baroli, M.; Sanna, R.; Uzzau, S.; Roggio, T.; Addis, M.F.; Anedda, R. Influence of seasonal and environmental patterns on the lipid content and fatty acid profiles in gonads of the edible sea urchin paracentrotus lividus from sardinia. Marine Environmental Research 2016, 113, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Sanna, R.; Siliani, S.; Melis, R.; Loi, B.; Baroli, M.; Roggio, T.; Uzzau, S.; Anedda, R. The role of fatty acids and triglycerides in the gonads of paracentrotus lividus from sardinia: Growth, reproduction and cold acclimatization. Marine Environmental Research 2017, 130, 113–121. [Google Scholar] [CrossRef]
- Rodrı́guez-Bernaldo de Quiros, A.; López-Hernández, J.; Simal-Lozano, J. Separation of phospholipid classes in sea urchin, paracentrotus lividus by high-performance liquid chromatography. Journal of Chromatography B 2002, 770, 71–75. [Google Scholar] [CrossRef]
- Montero-Torreiro, M.F.; Garcia-Martinez, P. Seasonal changes in the biochemical composition of body components of the sea urchin, paracentrotus lividus, in lorbé (galicia, north-western spain). Journal of the Marine Biological Association of the United Kingdom 2003, 83, 575–581. [Google Scholar] [CrossRef]
- Raposo, A.I. G.; Ferreira, S.M.F.; Ramos, R.; Santos, P.M.; Anjos, C.; Baptista, T.; Tecelão, C.; Costa, J.L.; Pombo, A. Effect of three diets on the gametogenic development and fatty acid profile of paracentrotus lividus (lamarck, 1816) gonads. Aquaculture Research 2019, 50, 2023–2038. [Google Scholar] [CrossRef]
- Baião, L.F.; Rocha, F.; Costa, M.; Sá, T.; Oliveira, A.; Maia, M.R.G.; Fonseca, A.J.M.; Pintado, M.; Valente, L.M.P. Effect of protein and lipid levels in diets for adult sea urchin paracentrotus lividus (lamarck, 1816). Aquaculture 2019, 506, 127–138. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



