Submitted:
24 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mathematical Model
2.1. Governing Equations for Fluid Flow
2.2. Turbulence Modeling
2.3. Heat Transfer Equations
2.4. Species Transport Equations
2.5. Multiphysics Couplings
2.5.1. Nonisothermal Flow Coupling
2.5.2. Thermal Connection for Nonlayered Shells
2.5.3. Reacting Flow with Dilute Species
2.6. Boundary Conditions for Respiratory Cycle
3. Numerical Implementation
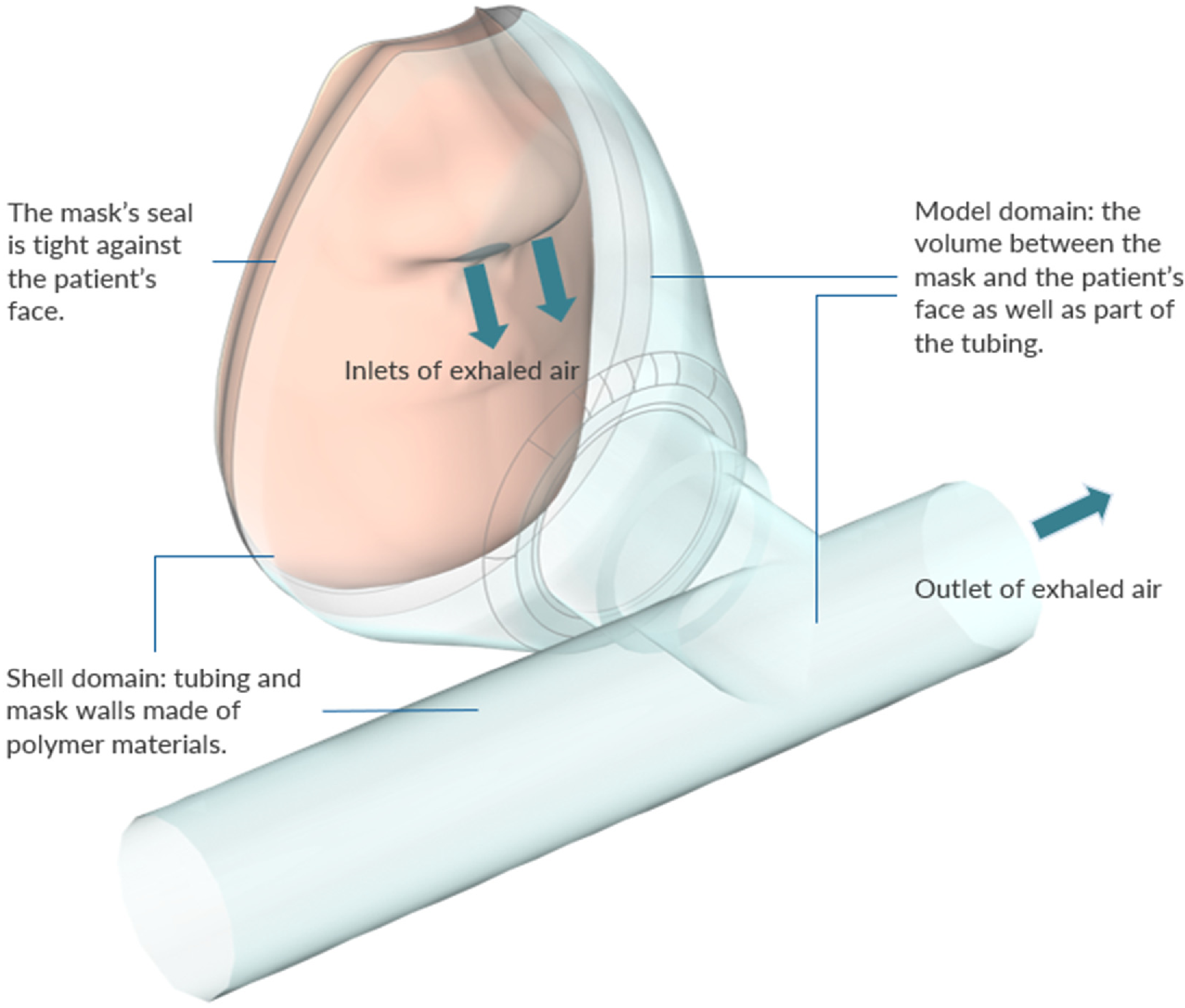
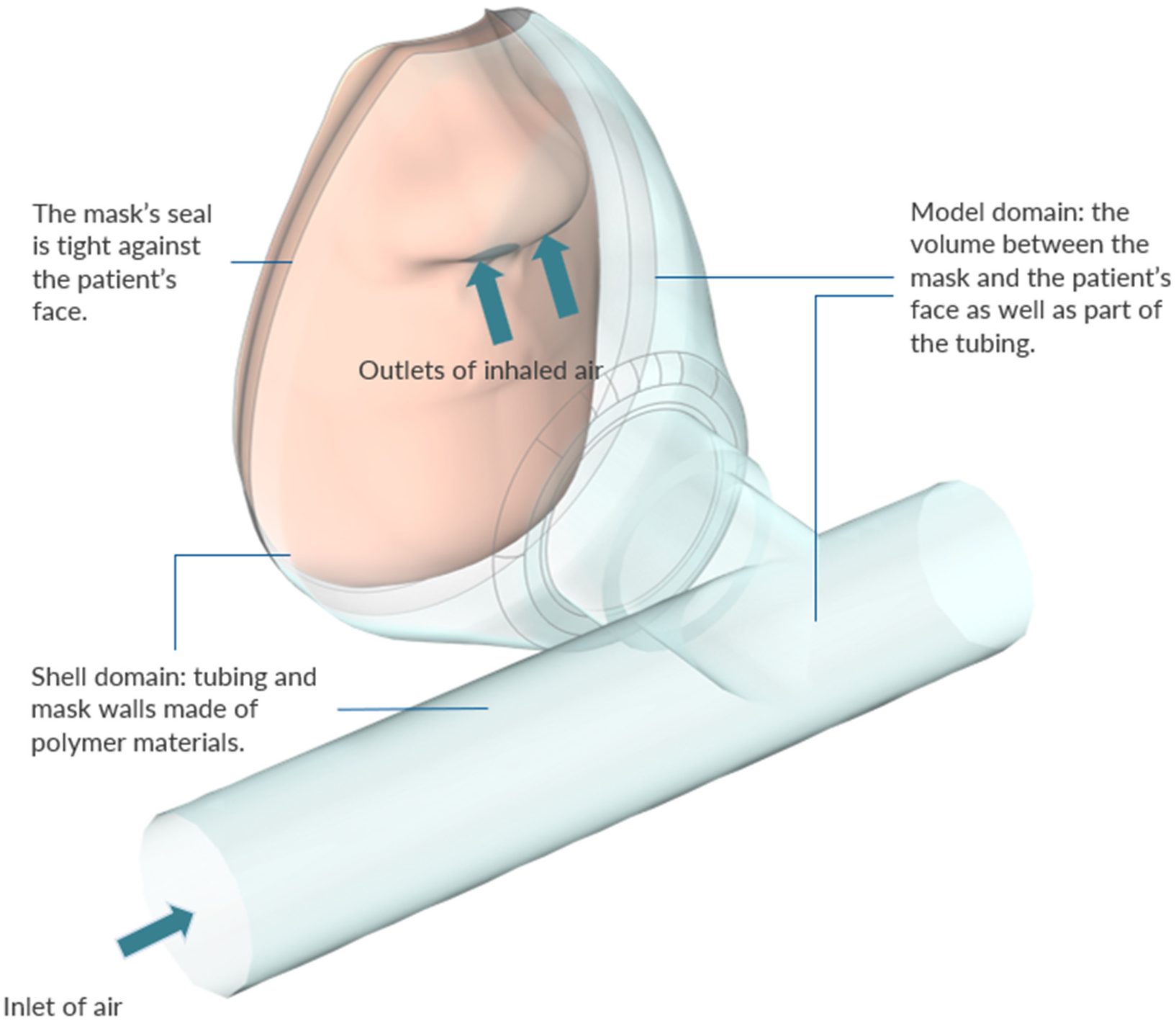
3.1. Computational Domain and Discretization
3.2. Study Setup
3.3. Solution Strategy
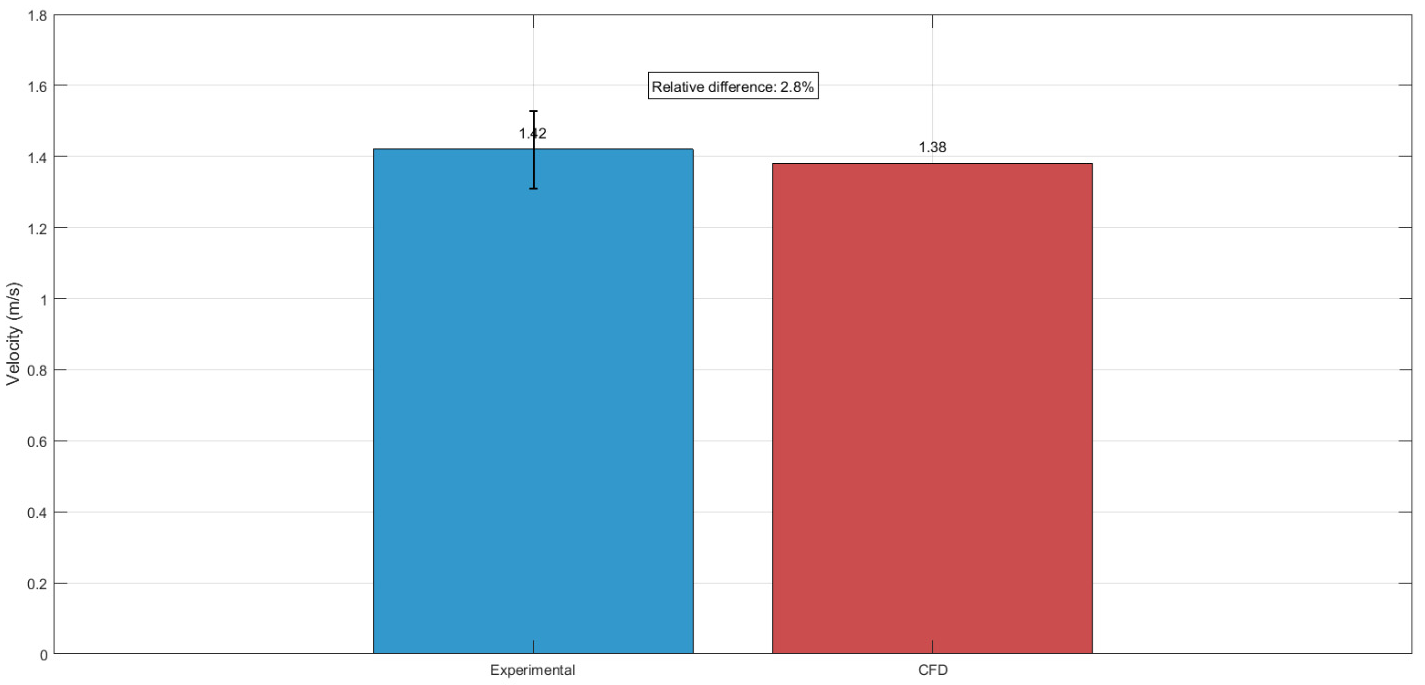
4. Model Validation
4.1. Experimental Validation Approach
4.2. Computational Setup for Validation
4.3. Validation Results
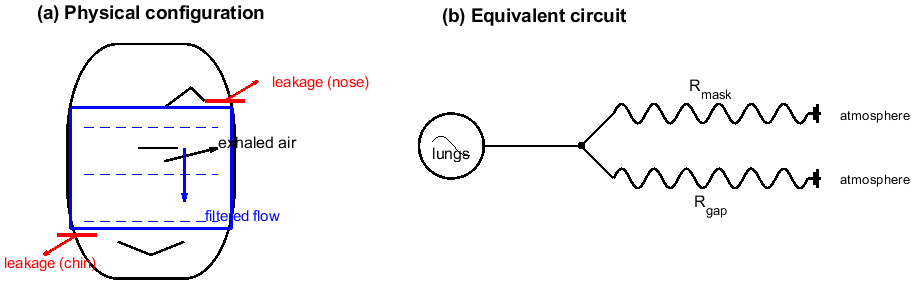
5. Reduced-Order Model For Mask Performance Analysis
5.1. Model Framework
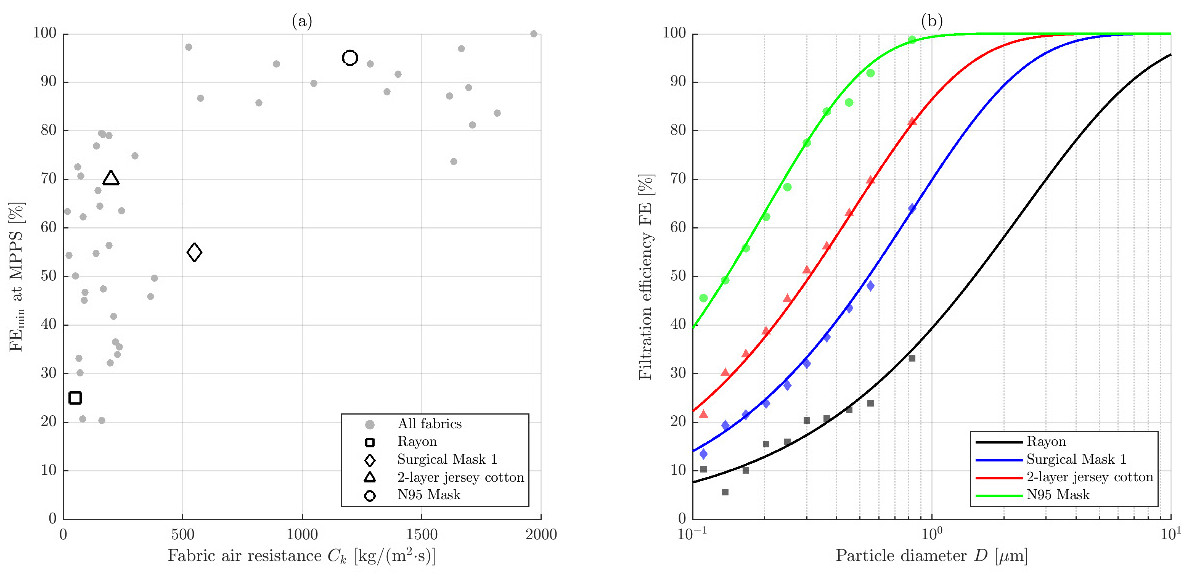
5.2. Mask Fabric Model
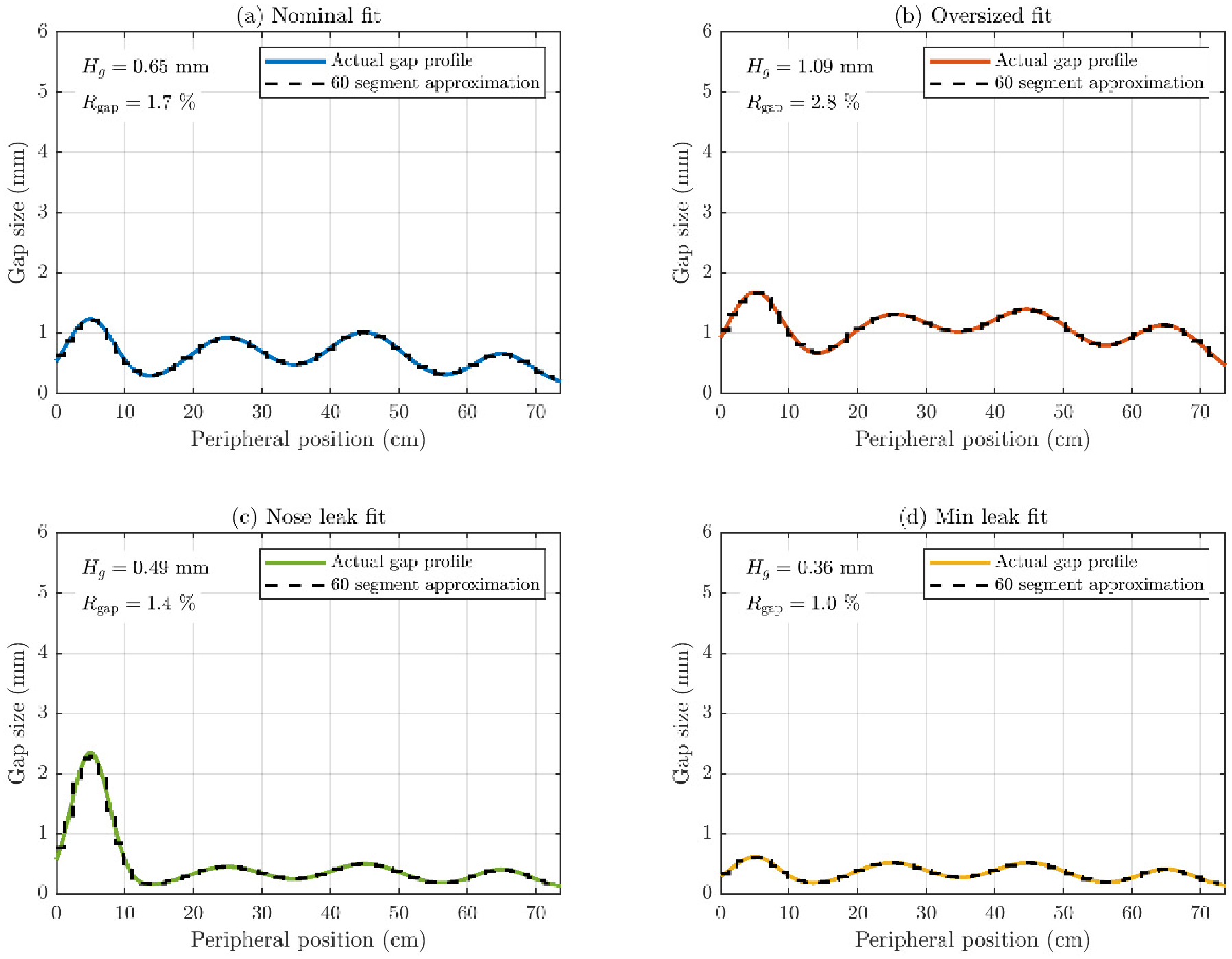
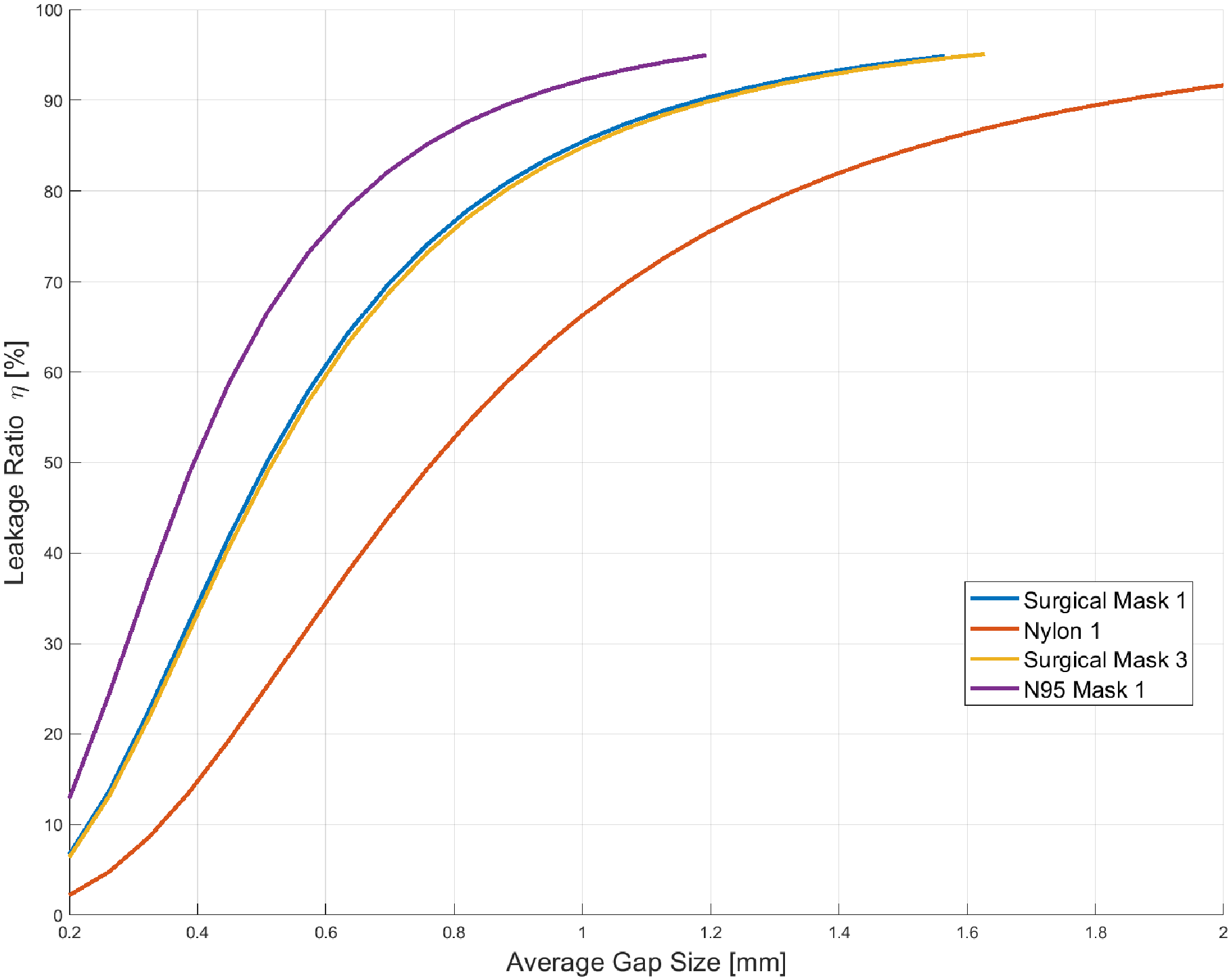
5.3. Peripheral Gap Model
5.4. Realistic Peripheral Gap Profiles
5.5. Lumped-Element System of Equations
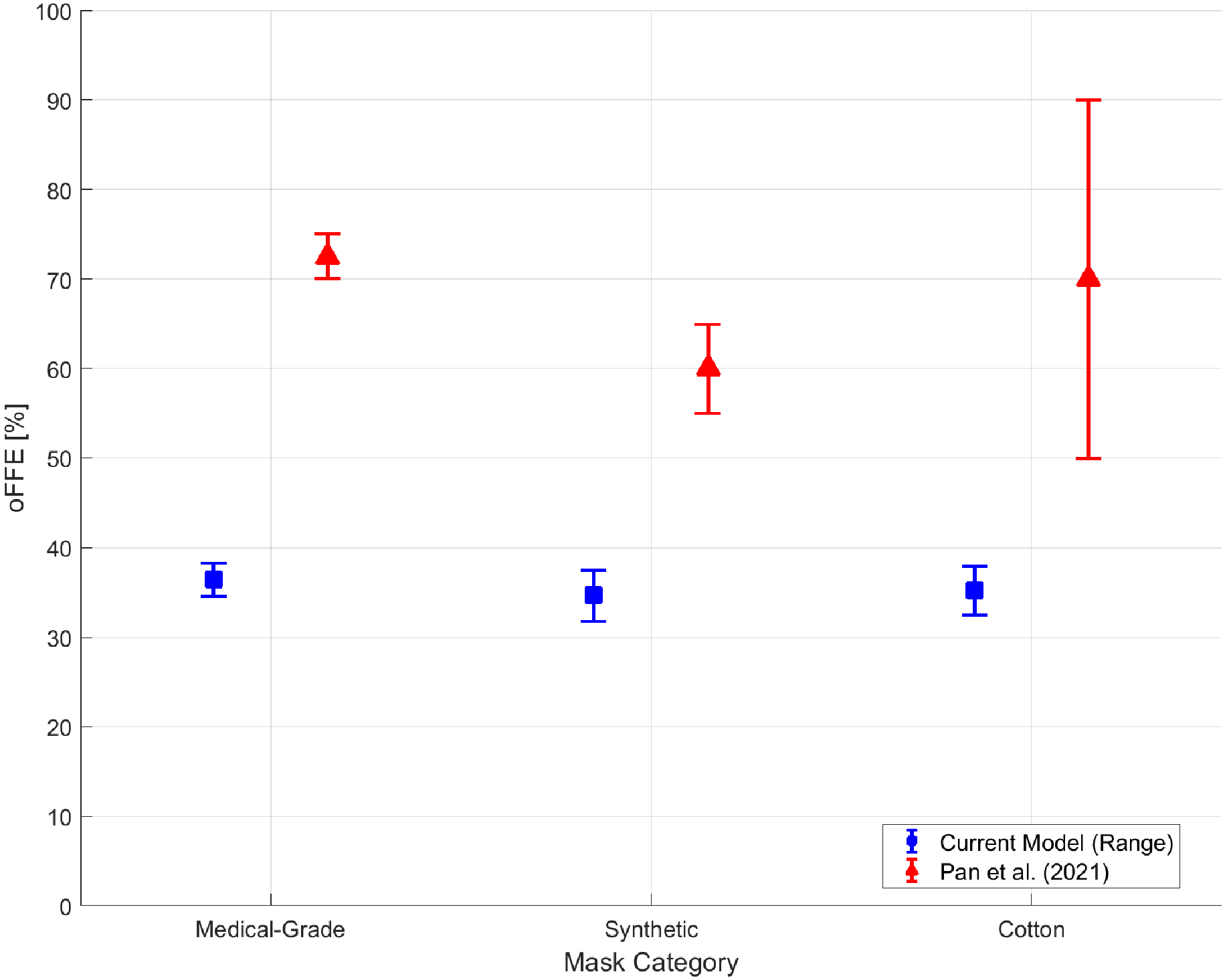
5.6. Outward Fitted Filtration Efficiency (oFFE)
5.7. Construction of Filtration Efficiency Functions
5.8. Validation of the Reduced-Order Model
5.9. Reduced-Order Model Results
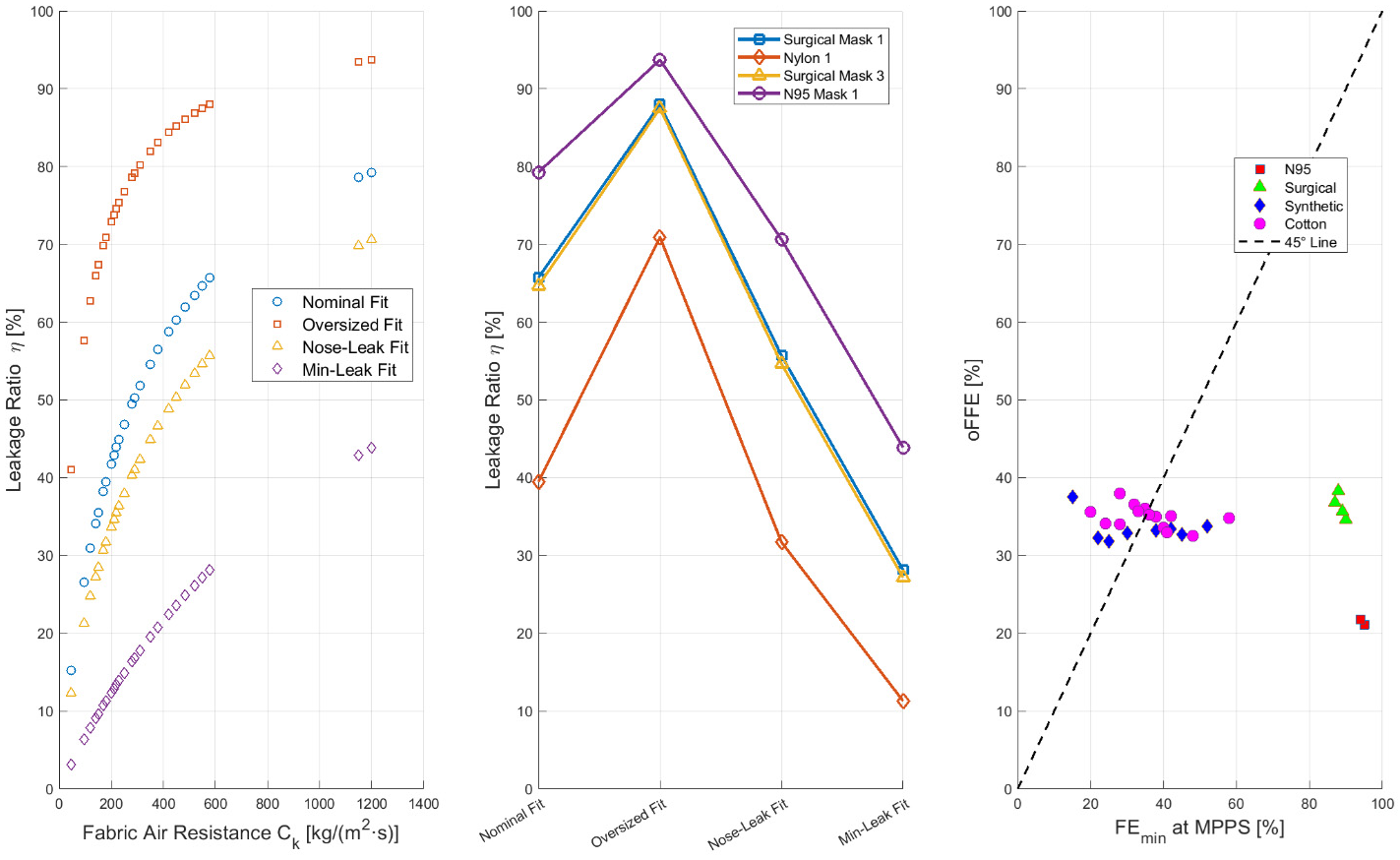
5.9.1. Peripheral Leakage Ratio
5.9.2. Local Leakage Velocity Distribution
5.9.3. Outward Fitted Filtration Efficiency
5.9.4. Implications for Transmission Reduction
6. Results and Discussion
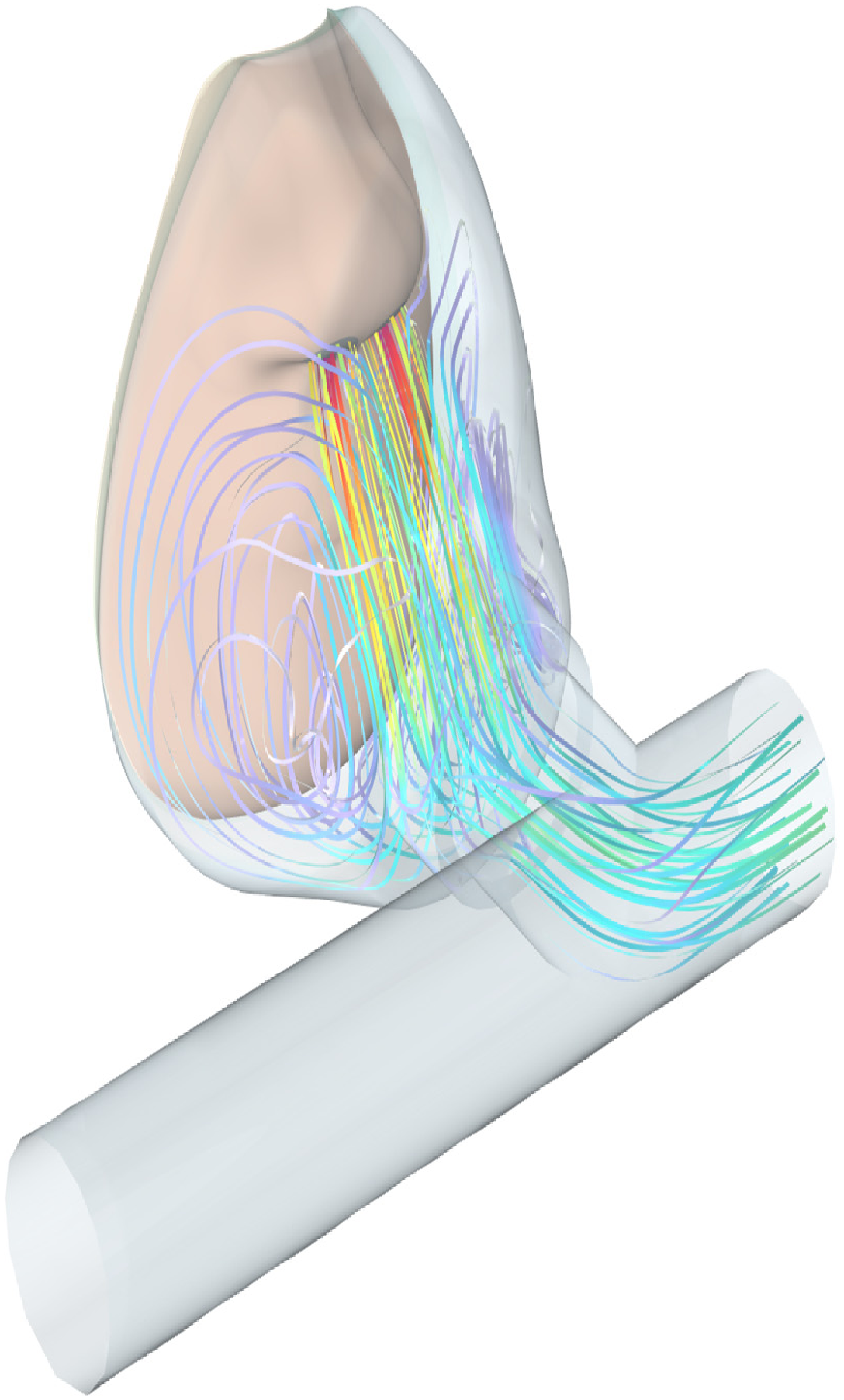
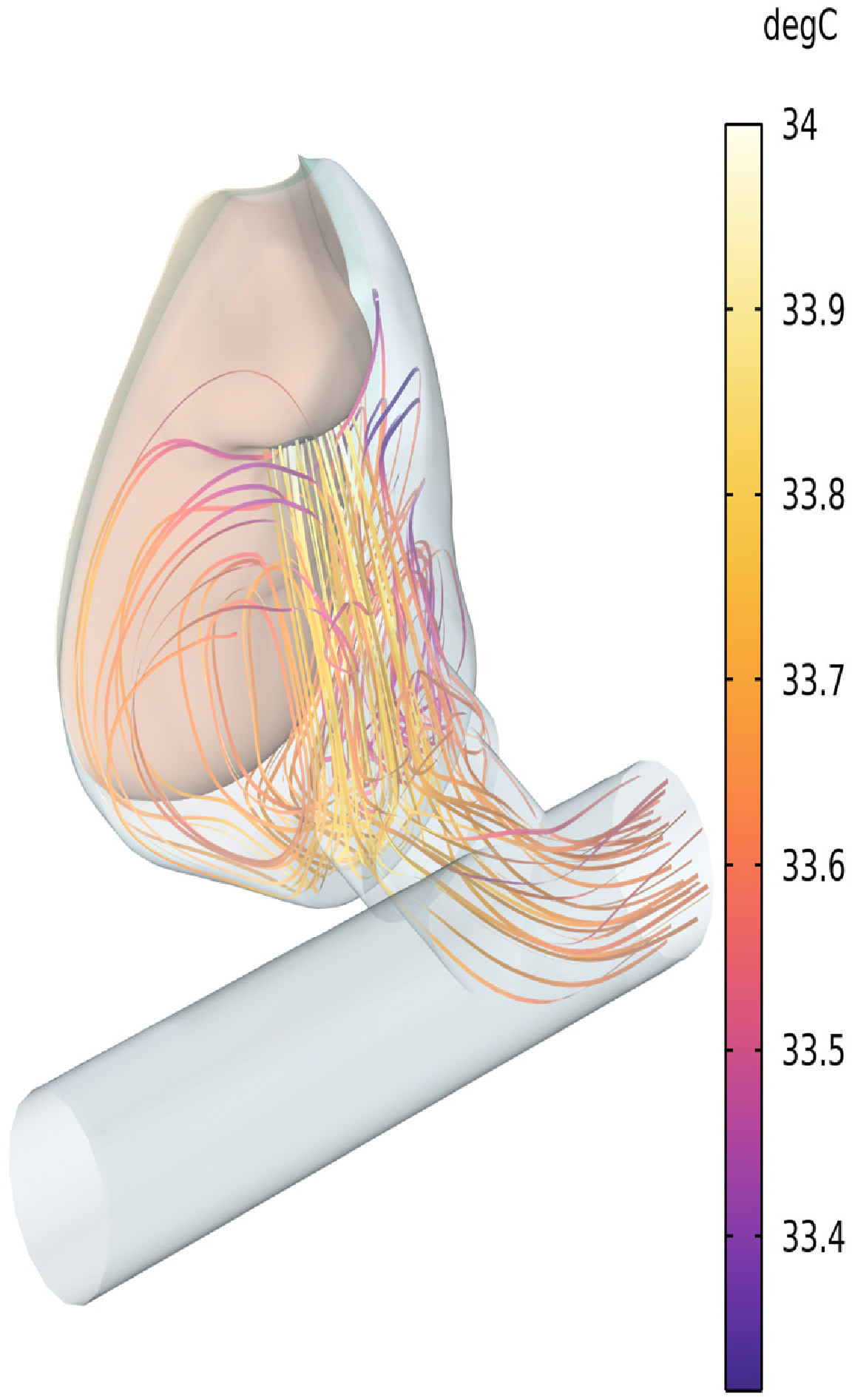
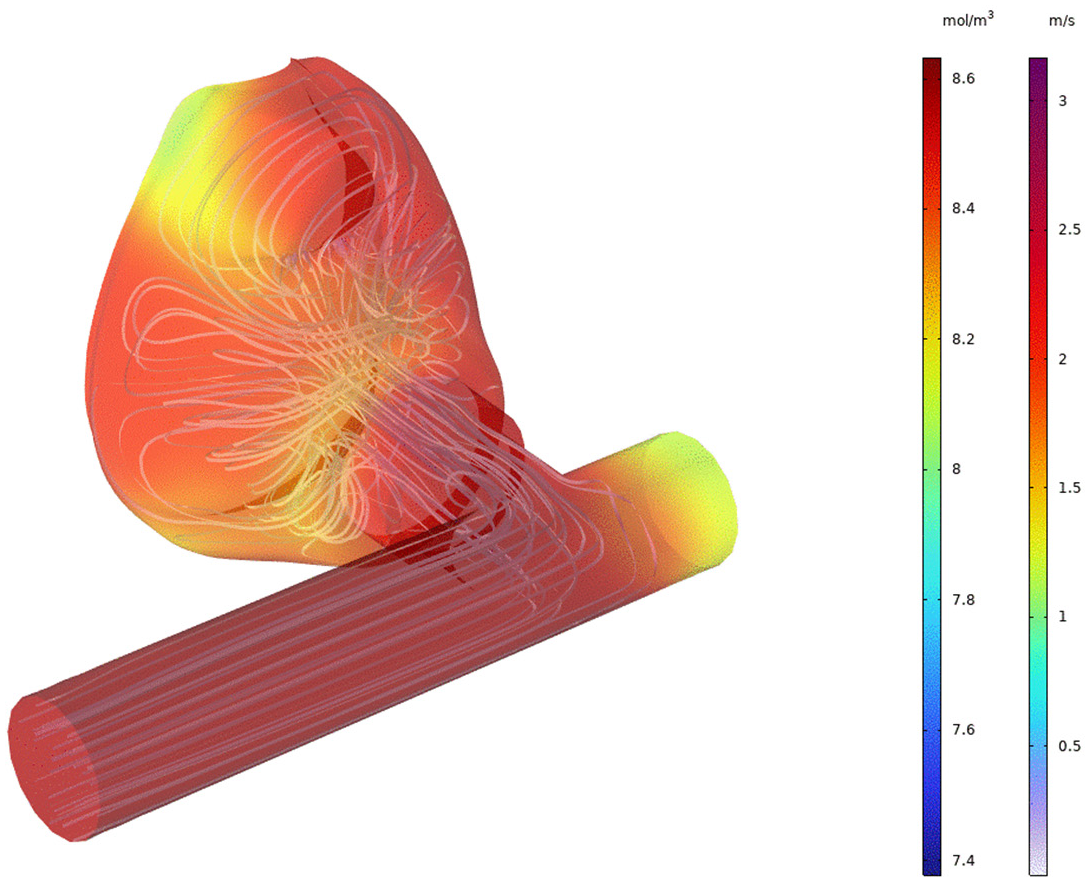
6.1. Flow Patterns from Full CFD
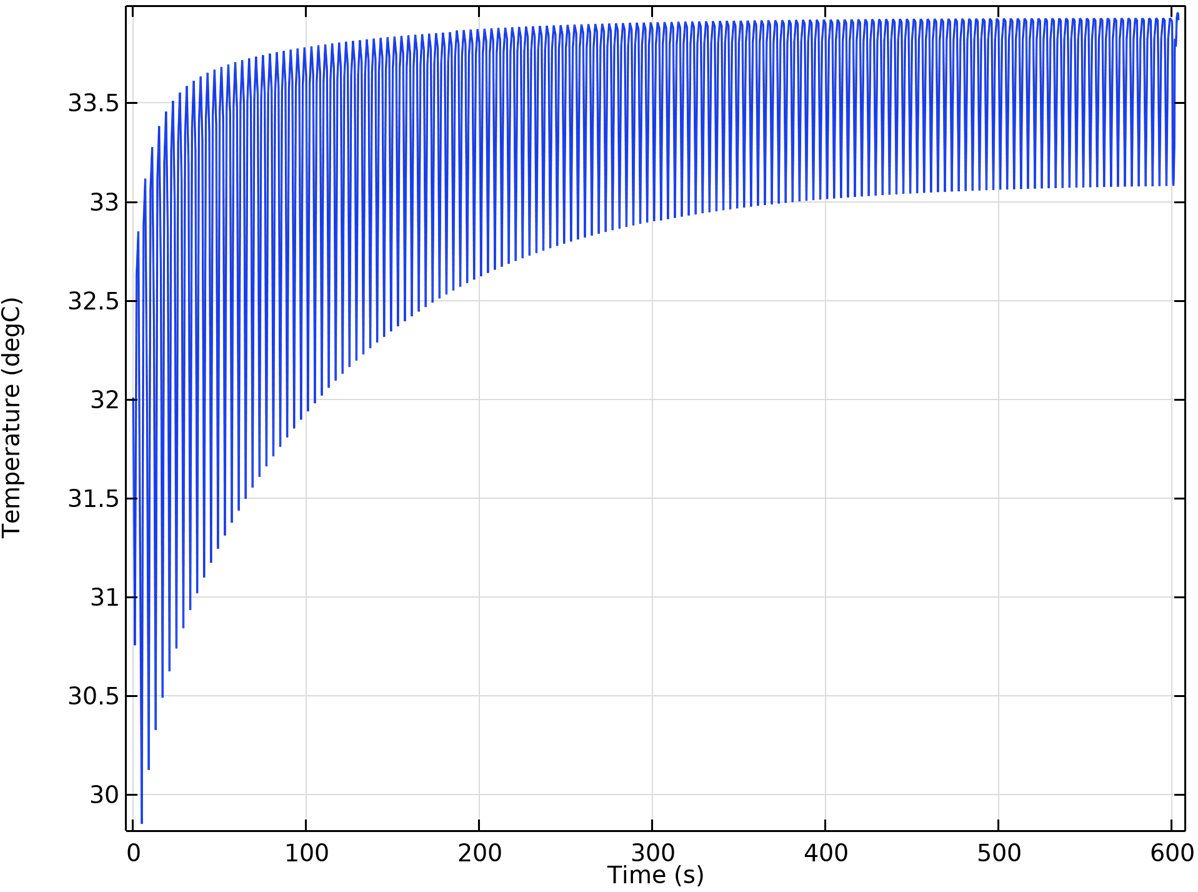
6.2. Temperature Field from Full CFD
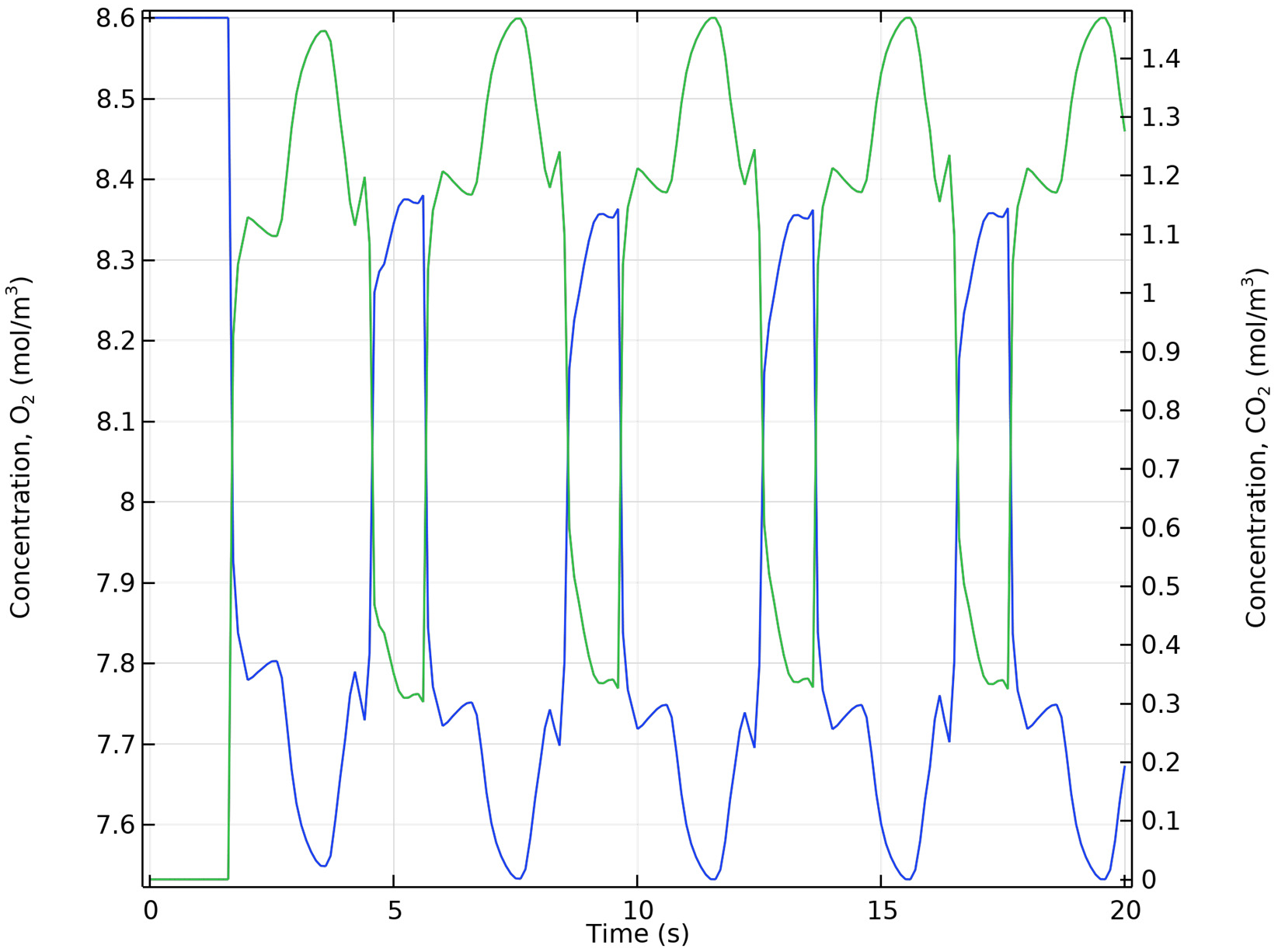
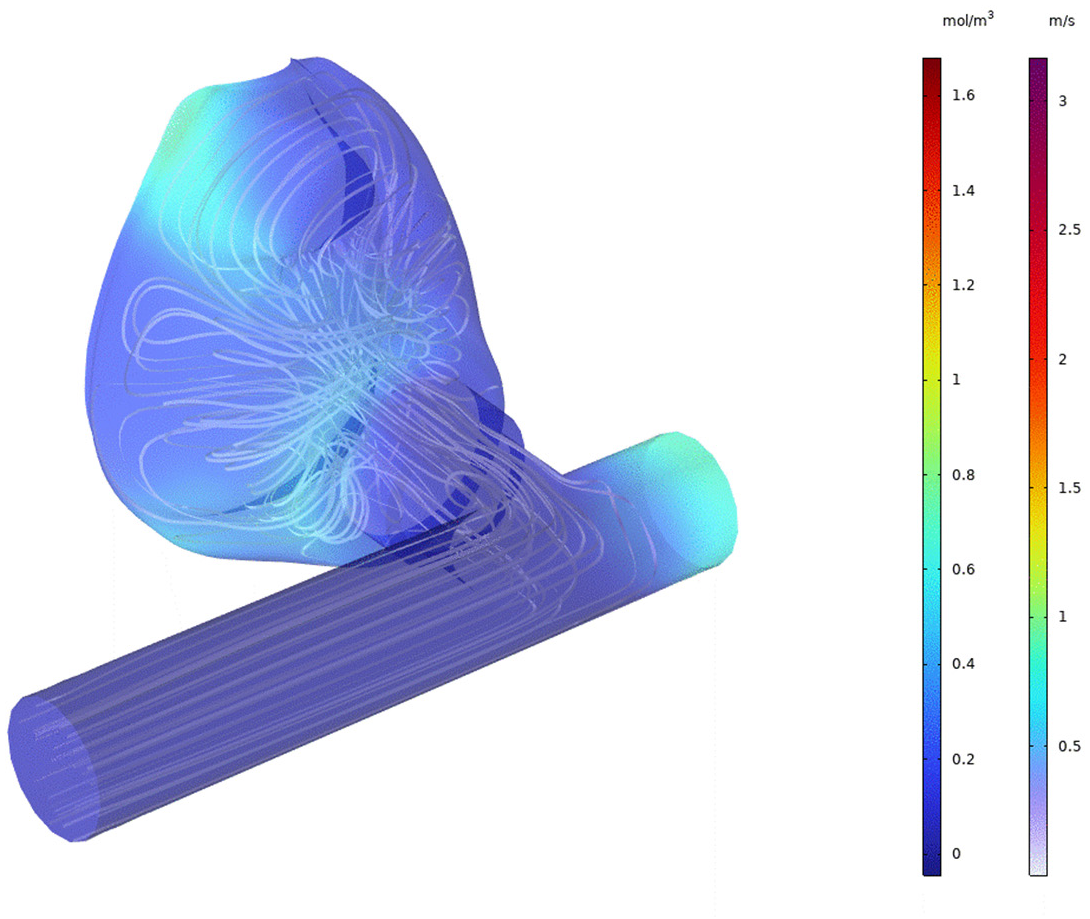
6.3. Species Transport from Full CFD
6.4. Synthesis of CFD and Reduced-Order Model Results
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| Symbol | Description | Units |
| ρ | Density | kg/m³ |
| u | Velocity vector | m/s |
| p | Pressure | Pa |
| τ | Stress tensor | Pa |
| F | Body force vector | N/m³ |
| k | Turbulent kinetic energy | m²/s² |
| ε | Turbulent dissipation rate | m²/s³ |
| μ | Dynamic viscosity | Pa·s |
| μT | Turbulent viscosity | Pa·s |
| T | Temperature | K |
| Cp | Specific heat capacity at constant pressure | J/(kg·K) |
| Cp,s | Specific heat capacity of solid | J/(kg·K) |
| kf | Thermal conductivity of fluid | W/(m·K) |
| ks | Thermal conductivity of solid | W/(m·K) |
| q | Heat flux vector | W/m² |
| Q | Heat source | W/m³ |
| Qs | Heat source in solid | W/m³ |
| ci | Concentration of species i | mol/m³ |
| cO2 | Oxygen concentration | mol/m³ |
| cCO2 | Carbon dioxide concentration | mol/m³ |
| Di | Diffusion coefficient of species i | m²/s |
| Di,ref | Reference diffusion coefficient of species i | m²/s |
| Ri | Reaction rate of species i | mol/(m³·s) |
| t | Time | s |
| d | Shell thickness | m |
| g | Gravitational acceleration vector | m/s² |
| β | Thermal expansion coefficient | 1/K |
| h | Heat transfer coefficient | W/(m²·K) |
| M | Molar mass | kg/mol |
| R | Universal gas constant | J/(mol·K) |
| Pk | Production of turbulent kinetic energy | kg/(m·s³) |
| Cμ, Cε1, Cε2 | Turbulence model constants | — |
| σk, σε | Turbulence model Prandtl numbers | — |
| I | Identity tensor | — |
| n | Unit normal vector | — |
| ω | Vorticity vector | 1/s |
| Γ | Circulation | m²/s |
| Nu | Nusselt number | — |
| Pei | Peclet number for species i | — |
| τthermal | Thermal time constant | s |
| Ck | Fabric air resistance | kg/(m²·s) |
| η | Leakage ratio | — |
| σ | Airflow adherence ratio | — |
| Stk | Stokes number | — |
| FE | Filtration efficiency | — |
| oFFE | Outward fitted filtration efficiency | — |
| Qe | Exhaled volume flow rate | m³/s |
| Qm | Mask penetration flow rate | m³/s |
| Qg | Gap leakage flow rate | m³/s |
| Hg | Gap height | m |
| Bg | Gap width | m |
| Lg | Gap length | m |
References
- Bird, R.B.; Stewart, W.E.; Lightfoot, E.N. Transport Phenomena, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2007; ISBN 978-0-470-11539-8.
- Wilcox, D.C. Turbulence Modeling for CFD, 3rd ed.; DCW Industries: La Cañada, CA, USA, 2006; ISBN 978-1-928729-08-2.
- Incropera, F.P.; DeWitt, D.P.; Bergman, T.L.; Lavine, A.S. Fundamentals of Heat and Mass Transfer, 6th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2007; ISBN 978-0-471-45728-2.
- Jamalabadi, M.A.; Hooshmand, P.; Bagheri, N.; KhakRah, H.; Dousti, M. Numerical Simulation of Williamson Combined Natural and Forced Convective Fluid Flow between Parallel Vertical Walls with Slip Effects and Radiative Heat Transfer in a Porous Medium. Entropy 2016, 18, 147. [CrossRef]
- Jamalabadi, M.Y.A. Effects of Micro- and Macro-Scale Viscous Dissipations with Heat Generation and Local Thermal Non-Equilibrium on Thermal Developing Forced Convection in Saturated Porous Media. J. Porous Media 2015, 18, 843–860. [CrossRef]
- Jamalabadi, M.Y.A. Thermohydraulic safety analysis of a research reactor by transport in porous media technique. Evolutions Mech. Eng. 2025, 6. [CrossRef]
- Finlay, W.H. The Mechanics of Inhaled Pharmaceutical Aerosols: An Introduction; Academic Press: London, UK, 2001; ISBN 978-0-12-256971-5.
- Kleinstreuer, C.; Zhang, Z. Airflow and Particle Transport in the Human Respiratory System. Annu. Rev. Fluid Mech. 2010, 42, 301–334. [CrossRef]
- Xi, J.; Barari, K.; Si, X.A.; Abdollahzadeh Jamalabadi, M.Y.; Park, J.H.; Rein, M. Inspiratory Leakage Flow Fraction for Surgical Masks with Varying Gaps and Filter Materials. Phys. Fluids 2022, 34, 041908. [CrossRef]
- Ni, C.; Solano, T.; Shoele, K.; Seo, J.H.; Mittal, R. Face Masks Provide High Outward Protection Despite Peripheral Leakage: Insights from a Reduced-Order Model of Face Mask Aerodynamics. Phys. Fluids 2023, 35, 061911. [CrossRef]
- Mittal, R.; Breuer, K.; Seo, J.H. The Flow Physics of Face Masks. Annu. Rev. Fluid Mech. 2023, 55, 193–211. [CrossRef]
- Zangmeister, C.D.; Radney, J.G.; Vicenzi, E.P.; Weaver, J.L. Filtration Efficiencies of Nanoscale Aerosol by Cloth Mask Materials Used to Slow the Spread of SARS-CoV-2. ACS Nano 2020, 14, 9188–9200. [CrossRef]
- Johnson, G.R.; Morawska, L.; Ristovski, Z.D.; Hargreaves, M.; Mengersen, K.; Chao, C.Y.H.; Wan, M.P.; Li, Y.; Xie, X.; Katoshevski, D.; et al. Modality of human expired aerosol size distributions. J. Aerosol Sci. 2011, 42, 839–851. [CrossRef]
- Pui, D.Y.H.; Romay-Novas, F.; Liu, B.Y.H. Experimental study of particle deposition in bends of circular cross section. Aerosol Sci. Technol. 1987, 7, 301–315. [CrossRef]
- Pan, J.; Harb, C.; Leng, W.; Marr, L.C. Inward and outward effectiveness of cloth masks, a surgical mask, and a face shield. Aerosol Sci. Technol. 2021, 55, 718–733. [CrossRef]
- Peric, R.; Peric, M. Analytical and Numerical Investigation of the Airflow in Face Masks used for Protection against COVID-19 Virus—Implications for Mask Design and Usage. J. Appl. Fluid Mech. 2020, 13, 1911–1923. [CrossRef]
- Solano, T.; Mittal, R.; Shoele, K. One size fits all: A simulation framework for face-mask fit on population-based faces. PLOS ONE 2021, 16, e0252143. [CrossRef]
- Clapp, P.W.; Sickbert-Bennett, E.E.; Samet, J.M.; Berntsen, J.; Zeman, K.L.; Anderson, D.J.; Weber, D.J.; Bennett, W.D. Evaluation of Cloth Masks and Modified Procedure Masks as Personal Protective Equipment for the Public During the COVID-19 Pandemic. JAMA Intern. Med. 2021, 181, 463–469. [CrossRef]
- Xi, J.; Si, X.A.; Nagarajan, R. Effects of mask-wearing on the inhalability and deposition of airborne SARS-CoV-2 aerosols in human upper airway. Phys. Fluids 2020, 32, 123312. [CrossRef]
- Cappa, C.D.; Asadi, S.; Barreda, S.; Wexler, A.S.; Ristenpart, W.D. Expiratory aerosol particle escape from surgical masks due to imperfect sealing. Sci. Rep. 2021, 11, 12110. [CrossRef]
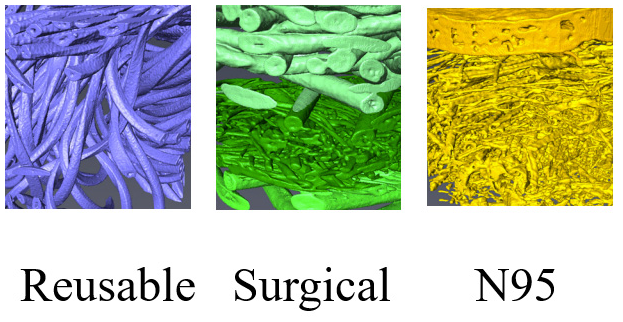
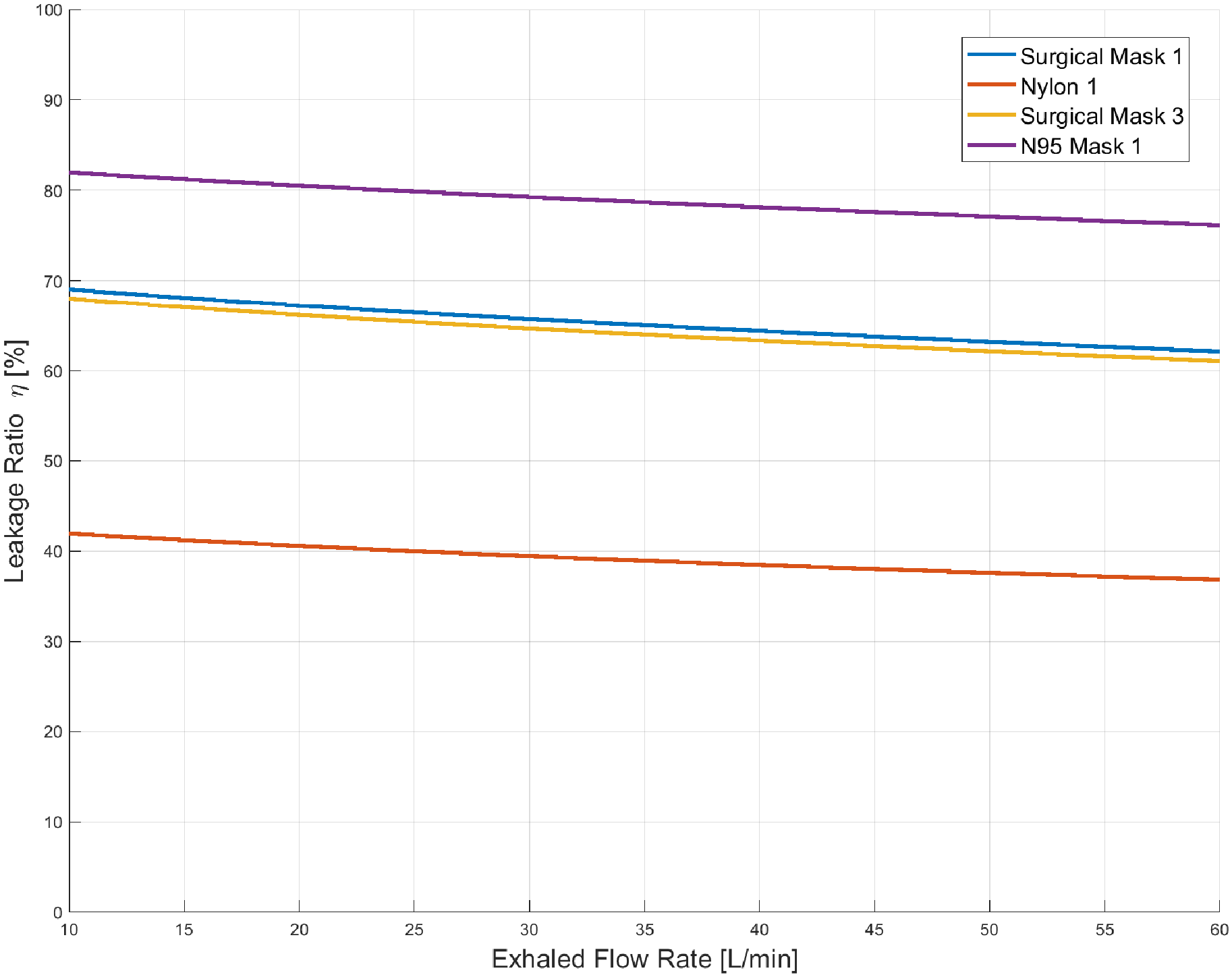
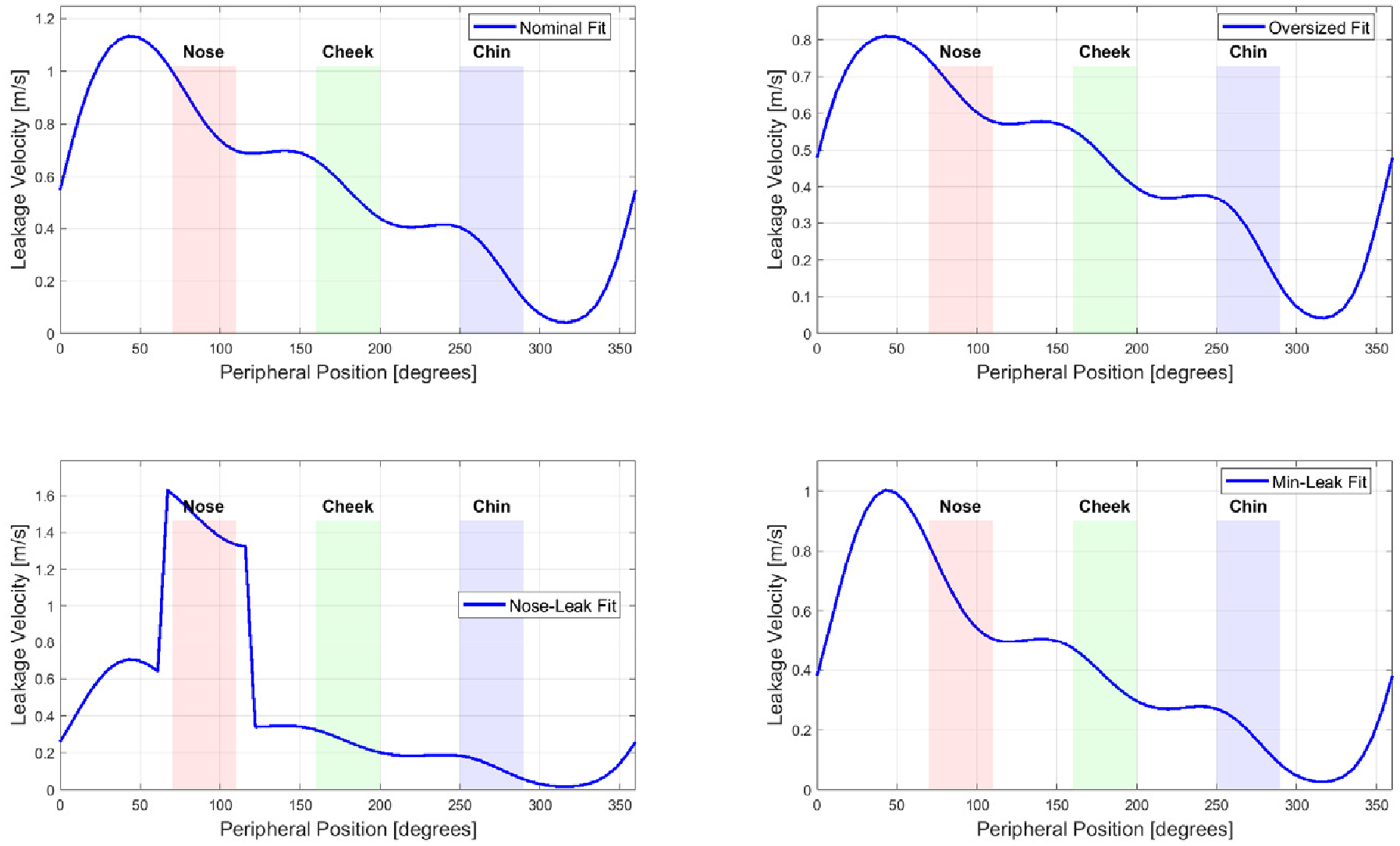
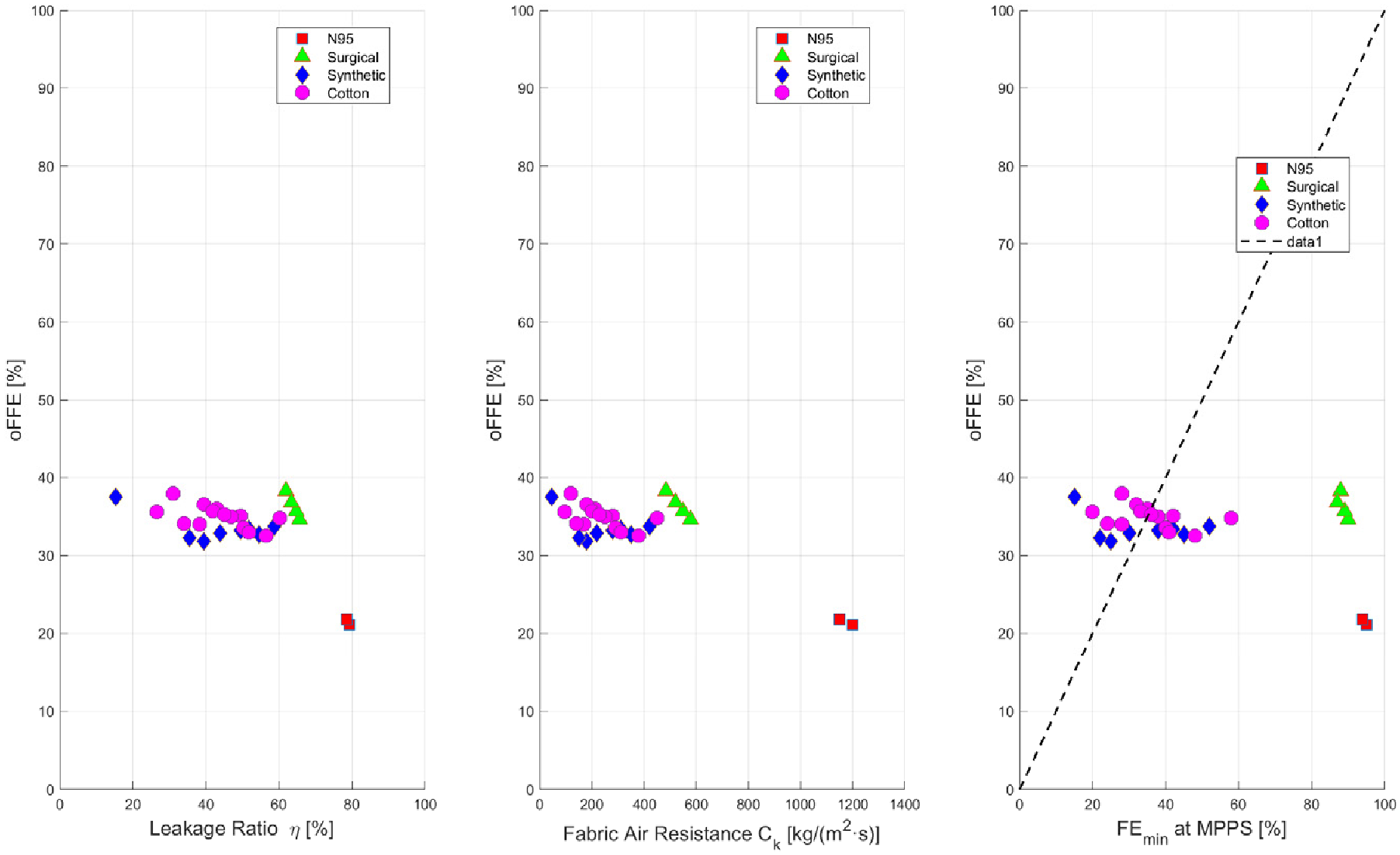
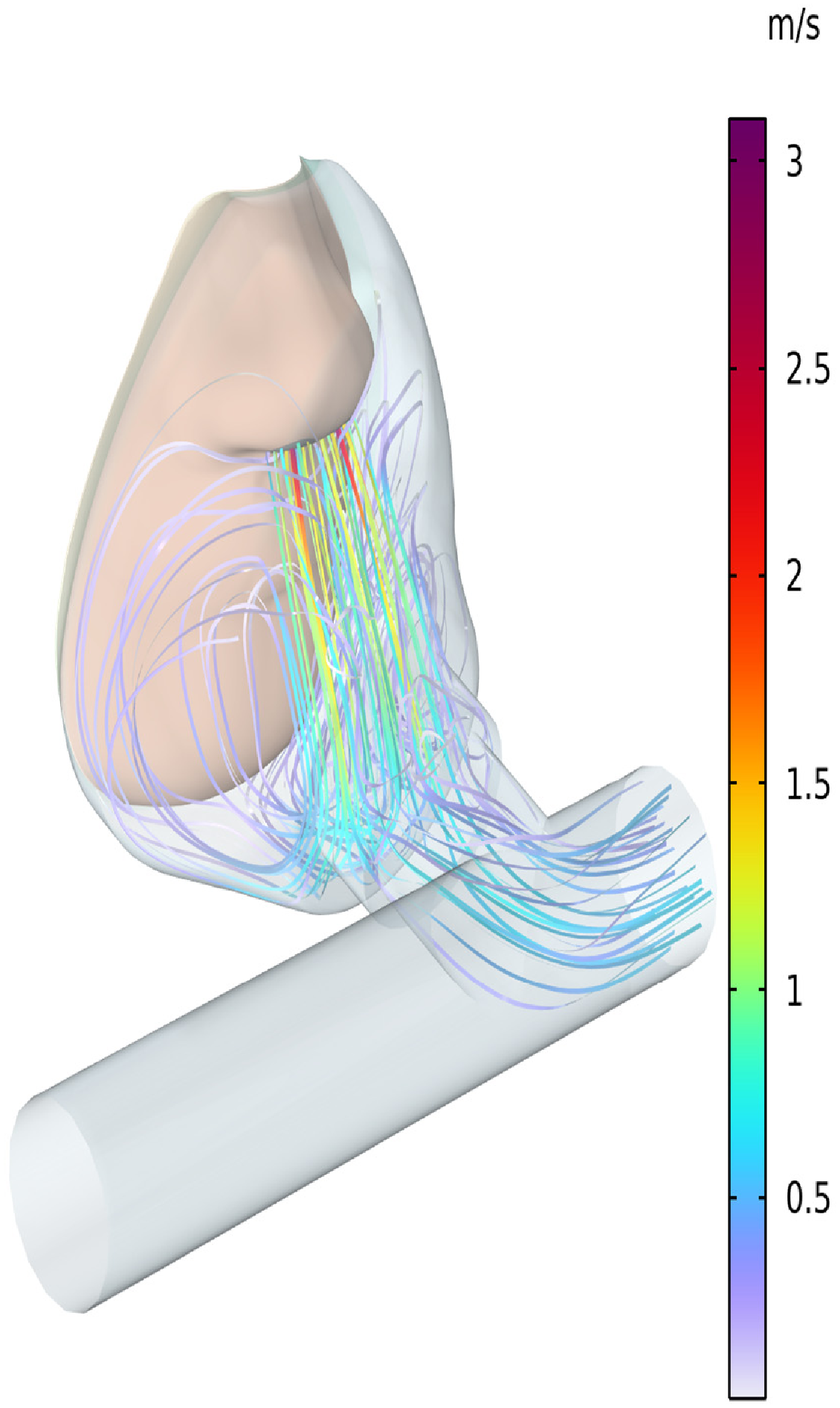
| Property | Reusable | Surgical | N95 |
| Volume fraction of fibres | 0.27 | 0.13 | 0.10 |
| Specific surface area (µm²/µm³) | 0.50 | 0.60 | 0.20 |
| Average length of fibres (µm) | 150 | 150 | 225 |
| Tortuosity of fibres | 6 | 6 | 12 |
| Average thickness of fibres (µm) | 30 | 30 | 45 |
| Average pore diameter (µm) | 0.30 | 0.20 | 0.38 |
| Permeability (µm²) | 30 | 20 | 30 |
| Facial temperature (°C) | 0.20 | 0.20 | 0.18 |
| Scenario | Surgical | Cloth | N95 | Rout (%) | Rout+in (%) |
| 1 | 25% | 75% | 0% | 52% | 66% |
| 2 | 50% | 50% | 0% | 58% | 72% |
| 3 | 75% | 25% | 0% | 65% | 77% |
| 4 | 33% | 33% | 33% | 70% | 87% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




