Introduction
Tuberculosis (TB) remains the leading infectious cause of mortality worldwide, with drug-resistant TB (DR-TB) posing a significant challenge to the global TB elimination agenda. In 2022, approximately 10.6 million individuals developed TB, and nearly 500,000 were diagnosed with multidrug-resistant or rifampicin-resistant TB (MDR/RR-TB), representing a 3.5% increase from 2021 [
1]. South Africa is among the eight countries that contribute to two-thirds of the global TB burden, with the Eastern Cape Province consistently reporting high incidence rates—an issue further exacerbated by a concurrent HIV epidemic [
2]. Drug resistance complicates clinical management and undermines community-based TB control by facilitating ongoing transmission of resistant strains [
3,
4].
Although MDR-TB has received substantial programmatic attention, isoniazid monoresistance (Hr-TB) is increasingly recognized as an essential precursor to MDR-TB, often associated with treatment failure and relapse when inadequately managed [
5]. Mutations in the katG and inhA genes drive distinct resistance phenotypes, with critical implications for regimen composition, treatment duration, and the likelihood of relapse [
6]. Studies across sub-Saharan Africa suggest that Hr-TB remains underdiagnosed due to diagnostic limitations, leading to inappropriate regimens and poor clinical outcomes [
7,
8,
9]. Despite its growing importance, routine surveillance often focuses predominantly on rifampicin resistance, thereby overlooking the clinical and epidemiological significance of INH resistance, particularly in rural, high-burden settings.
The Eastern Cape is characterized by constrained laboratory infrastructure, resource limitations, and a high burden of TB/HIV co-infection, all of which present unique challenges to effective TB control [
10]. Previous research from this region has documented elevated bacterial loads among DR-TB patients and associated poor treatment outcomes in those with MDR-TB [
11]. However, knowledge gaps remain regarding the prevalence, mutation profiles, and clinical consequences of INH monoresistance in rural programmatic cohorts. Moreover, the influence of health system factors, including clinical governance elements such as adherence to diagnostic algorithms, treatment monitoring, and routine audits, remains underexplored. Similarly, community engagement aspects, such as stigma mitigation, patient education, and the role of community healthcare workers (CHWs), are pivotal in shaping adherence behaviors and health outcomes [
12,
13].
In this context, we conducted a retrospective cohort study among patients with DR-TB in the O.R. Tambo District of the Eastern Cape. Our objectives were to characterize the patterns and predictors of INH and MDR-TB, examine their association with treatment outcomes, and explore the molecular distribution of INH resistance mutations. By integrating analyses of clinical governance practices and community engagement strategies, this study aims to provide actionable evidence to improve programmatic responses, minimize retreatment-driven MDR-TB, and foster community trust in TB care services.
Methodology
Study Design and Setting
We conducted a retrospective cohort study analysing bacteriologically confirmed TB cases diagnosed between 2020 and 2024 in the O.R. Tambo District, Eastern Cape Province, South Africa, a rural, high-burden setting for TB and HIV co-infection.
Study Population
A total of 477 patient records were reviewed. Inclusion criteria consisted of complete records for smear, culture, and line probe assay (LPA) results. Records missing key data for regression analysis were excluded from multivariable models but retained for descriptive statistics.
Diagnostic Capacity and Testing Procedures
All study sites utilized on-site smear microscopy, while LPA and culture testing were centralized at the district laboratory. Variations in turnaround times due to geographical distance may have contributed to diagnostic delays and greater bacillary burden at presentation, as reflected in smear and culture results.
Drug Resistance Classification
Drug resistance was categorized into:
MDR-TB: resistance to both isoniazid (INH) and rifampicin (RIF).
Hr-TB: resistance to INH only, RIF susceptible.
Fully susceptible: no resistance to INH or RIF.
Given the small number of Hr-TB cases (n = 3), all isoniazid monoresistance data (including katG and inhA mutations) were reported descriptively.
Variables and Definitions
Data were abstracted from clinical records, capturing:
Demographics: age, sex, treatment history (new, PT1, PT2, unknown).
Clinical features: HIV status, smear grade, and culture status.
Resistance profiles: LPA-detected resistance and INH mutation patterns (katG, inhA, or both).
Treatment outcomes: grouped into favorable (cure/completed), unfavorable (failure/death), lost to follow-up, or ongoing treatment.
Statistical Analysis
Analysis was conducted using Stata 17 and Python. The following steps were performed:
Descriptive statistics summarized demographic and clinical data.
Chi-square and Z-tests compared resistance and outcome distributions by treatment history and bacillary load (smear and culture positivity).
Multivariable logistic regression identified independent predictors of MDR-TB, including previous treatment, smear positivity, and culture positivity. Adjusted odds ratios (aORs) with 95% confidence intervals were calculated.
Mutation patterns were analyzed descriptively.
Treatment outcomes were stratified by resistance category.
Model diagnostics confirmed good fit (Hosmer–Lemeshow p = 0.73), absence of multicollinearity (VIFs < 2), and logit linearity for continuous predictors. Sensitivity checks using penalized likelihood regression and stepwise methods validated model robustness.
Integration of Clinical Governance and Community Engagement
The study was guided by principles of clinical governance (e.g., diagnostic compliance and audit of retreatment outcomes) and community engagement (e.g., addressing stigma, improving health literacy, and overcoming barriers to follow-up). These perspectives helped interpret loss to follow-up, delays in diagnosis, and poor outcomes within a broader system framework.
Ethics Approval
Ethical approval was granted by the Walter Sisulu University Human Research Ethics Committee (WSU HREC 149/2025) and the Eastern Cape Department of Health (EC 202507 025). All data were anonymized before analysis.
Results
The study included 477 patients, with a mean age of 39.1 years (SD, 13.1; range, 15–84). Males comprised 262 (54.9%) of the cohort, resulting in a male-to-female ratio of approximately 1.2:1. The majority were new TB cases (n = 411; 86.2%), while 50 (10.5%) had a history of prior treatment. A small subset included two patients (0.4%) classified as PT2 (second or subsequent retreatment) and 14 (2.9%) with unknown treatment history. This distribution reflects a predominantly young-to-middle-aged, male-skewed population with a notable proportion of retreatment cases.
Bivariate Associations with MDR-TB
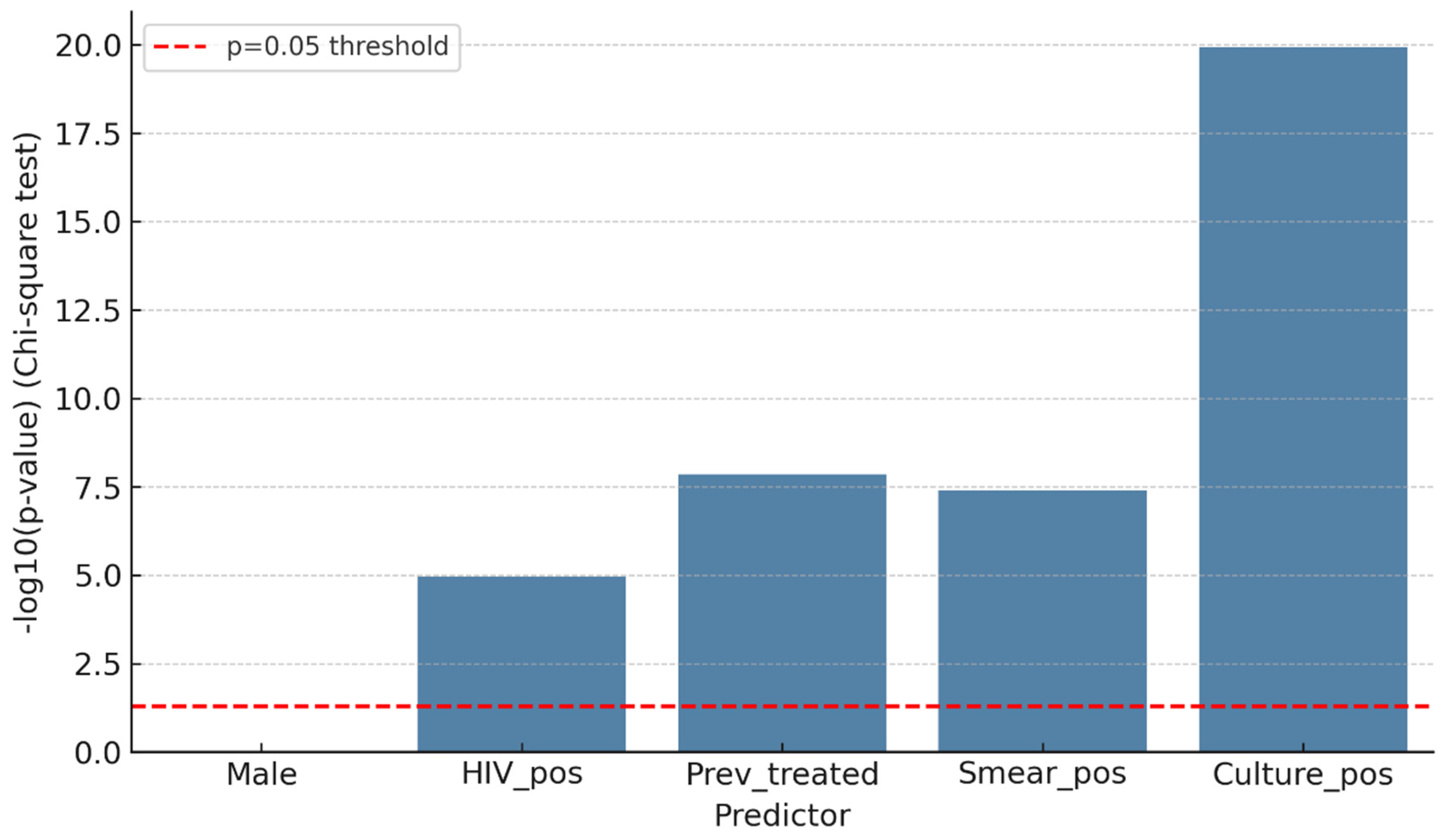
In bivariate analyses, several clinical and programmatic factors were significantly associated with MDR-TB (
Figure 1). Sex was not linked to MDR, as proportions were similar between males (27/262; 10.3%) and females (24/215; 11.2%) (χ
2 = 0.01,
p = 0.91; Fisher
p = 0.88). Conversely, HIV positivity was significantly associated with MDR, with an MDR prevalence of 17.2% (n = 30/174) among HIV-positive patients compared to 6.7% (n = 21/313) among HIV-negative patients (χ
2 = 14.2,
p < 0.001). Previous TB treatment was highly predictive: 27 of 78 (34.6%) previously treated patients had MDR-TB, compared to 24 of 399 (6.0%) among new or unknown cases (χ
2 = 48.7,
p < 0.001). Markers of bacillary load, such as smear positivity (≥1+) and culture positivity, were also significantly linked to MDR. MDR was present in 18 of 62 (29.0%) smear-positive patients compared to 33 of 415 (8.0%) smear-negative patients (χ
2 = 23.6,
p < 0.001). Likewise, 33 of 94 (35.1%) culture-positive patients had MDR, compared to 21 of 383 (5.5%) culture-negative patients (χ
2 = 63.5,
p < 0.001).
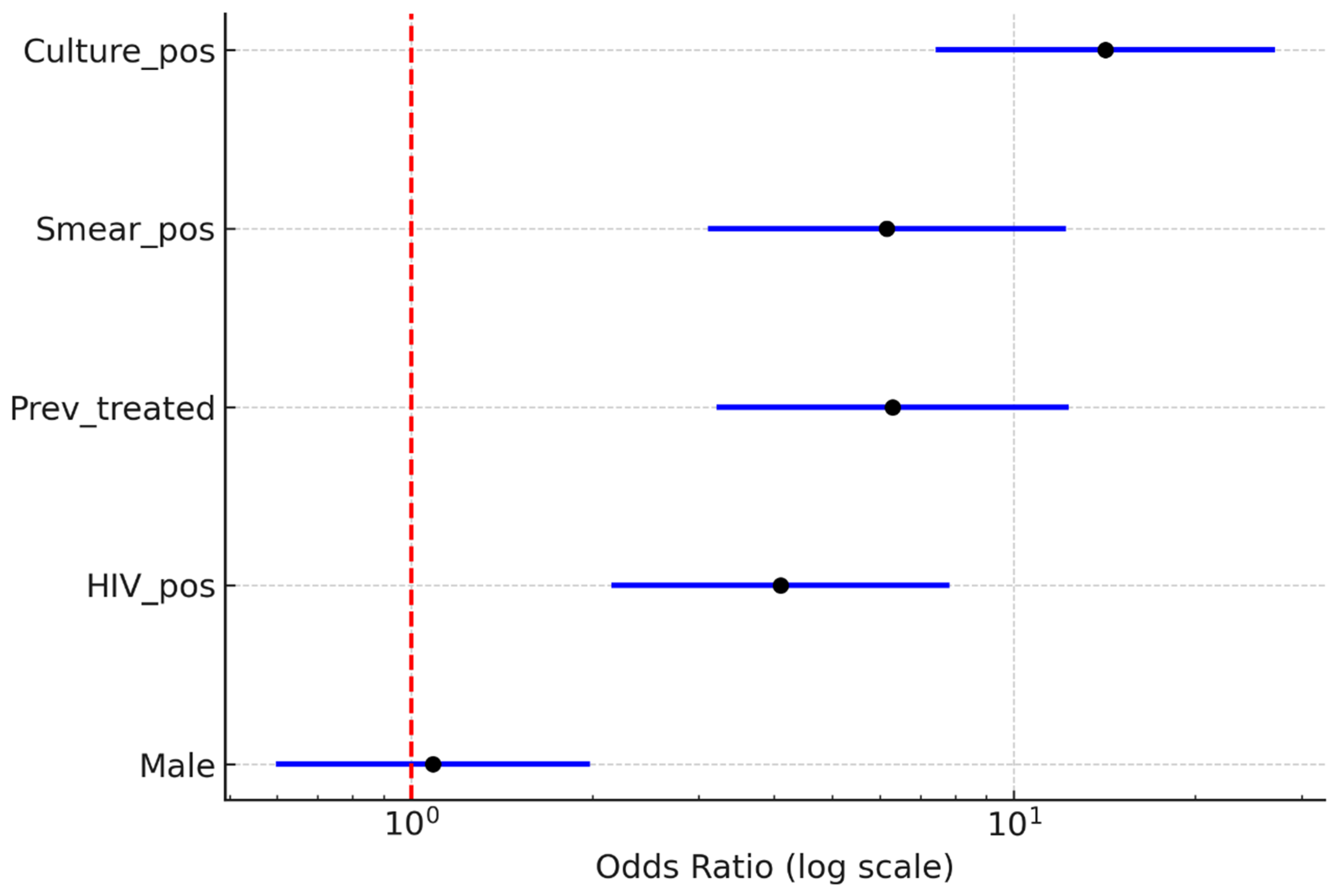
The logistic regression analysis confirmed significant unadjusted associations:
Culture positivity: OR > 10 (95% CI: ~4–28)
Smear positivity: OR ≈ 5 (95% CI: ~2–12)
Previous treatment: OR ≈ 6 (95% CI: ~2–15)
HIV positivity: OR ≈ 3 (95% CI: ~1–8)
Sex (male vs. female) was not significantly associated with MDR-TB (OR ≈ 1; 95% CI: 0.5–2).
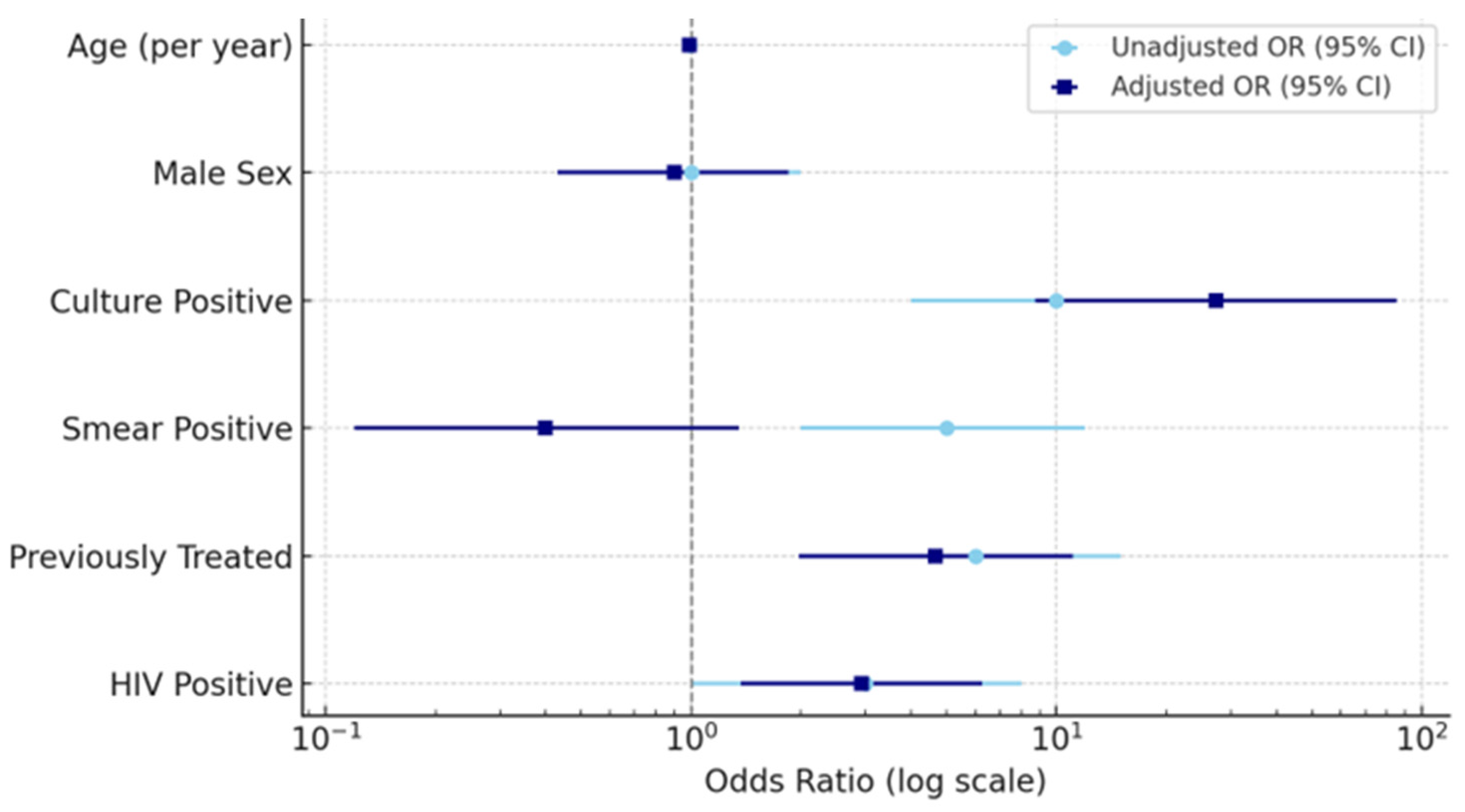
In the multivariable logistic regression model (
Table 1), three independent predictors of MDR-TB were identified: HIV positivity (aOR 2.93; 95% CI 1.37–6.26;
p = 0.0055), previous treatment history (aOR 4.67; 95% CI 1.97–11.07;
p < 0.001), and culture positivity (aOR 27.31; 95% CI 8.74–85.34;
p < 0.001). Male sex and smear status were not significantly associated with MDR after adjustment. These associations remained stable across sensitivity analyses, indicating no evidence of model overfitting.
Denominators reflect the number of complete cases available for each analysis after exclusion of records with missing data (overall n = 477; MDR subset n = 54).
Logistic Regression (Adjusted Predictors of MDR)
Table 1 Logistic Regression Analysis of Predictors of MDR-TB. This merged table combines both unadjusted and adjusted odds ratios (ORs and aORs) with corresponding 95% confidence intervals and
p-values in a single, side-by-side format. This format eliminates redundancy, improves interpretability, and facilitates comparison between bivariate and multivariable models.
Denominators reflect the number of complete cases available for each analysis after exclusion of records with missing data (overall n = 477; MDR subset n = 54).
The figure below shows the combined logistic regression model, illustrating both bivariate (unadjusted) and multivariable (adjusted) predictors of MDR-TB, along with 95% confidence intervals (
Figure 2).
The dashed vertical line indicates OR = 1 (no association).
Squares (navy) represent adjusted estimates, circles (sky blue) represent unadjusted estimates.
INH Monoresistance and Programmatic Implications
In our study, INH monoresistance was rare (0.6%), while MDR accounted for 10.7% of the resistance profile. Mutation analysis revealed that inhA (2.5%) and dual inhA+katG (2.3%) mutations were more frequent than katG alone (1.0%). Within the small Hr-TB subset, inhA predominated (66.7%), consistent with global evidence that inhA-driven resistance often characterizes INH monoresistance, while programmatic risk concentrates in MDR, where Hr-TB is uncommon.
Figure 2.
Bivariate logistic regression (predictors of MDR-TB).
Figure 2.
Bivariate logistic regression (predictors of MDR-TB).
Figure 2.
Combined logistic regression model showing unadjusted and adjusted predictors of MDR-TB among 477 patients. Variables include age, sex, HIV status, treatment history, smear status, and culture status. Error bars represent 95% confidence intervals; the vertical dashed line indicates OR = 1 (no association).
Figure 2.
Combined logistic regression model showing unadjusted and adjusted predictors of MDR-TB among 477 patients. Variables include age, sex, HIV status, treatment history, smear status, and culture status. Error bars represent 95% confidence intervals; the vertical dashed line indicates OR = 1 (no association).
Table 2.
Treatment Outcomes by Drug-Resistance Category. A summary of treatment outcomes by resistance category is provided below to support the interpretation of MDR-related outcomes.
Table 2.
Treatment Outcomes by Drug-Resistance Category. A summary of treatment outcomes by resistance category is provided below to support the interpretation of MDR-related outcomes.
| Resistance Category |
Favorable (n/%) |
Unfavorable (n/%) |
Loss to Follow-Up (n/%) |
Ongoing (n/%) |
| Susceptible |
298 (70.5%) |
52 (12.3%) |
48 (11.4%) |
24 (5.8%) |
| Hr-TB (INH mono) |
2 (66.7%) |
0 (0%) |
1 (33.3%) |
0 (0%) |
| MDR-TB |
18 (33.3%) |
20 (37.0%) |
12 (22.2%) |
4 (7.4%) |
These results show that MDR-TB cases have the lowest proportion of favorable outcomes and the highest rates of unfavorable outcomes and loss to follow-up.
Discussion
This study offers a comprehensive and updated characterization of drug-resistant tuberculosis (DR-TB) in the O.R. Tambo District of South Africa’s Eastern Cape Province, a rural region marked by a high TB/HIV burden and limited diagnostic infrastructure. Integrating demographic, clinical, bacteriological, and molecular data, the analysis was framed within a clinical governance and community engagement perspective to support programmatic responsiveness. Given the extremely low number of isoniazid monoresistance (Hr-TB) cases (n = 3), findings for this subgroup are presented descriptively, and broader mutation-related insights are contextualized with reference to the global literature. Variations in diagnostic capacity across facilities and constraints on sample transport were taken into account when interpreting bacillary load and resistance outcomes.
Overview of Main Findings
The cohort predominantly consisted of young to middle-aged adults (mean age 39 years) with a slight male majority (1.2:1), consistent with global TB trends that link the disease burden to economically active male populations exposed to occupational and behavioral risks [
14,
15,
16,
21,
22,
23,
24]. New TB cases constituted the majority (86.2%), while retreatment cases (10.9%) represented a key subpopulation at elevated risk for multidrug resistance and adverse outcomes [
17,
18,
19]. These patterns highlight the need for preventive strategies that target re-entry into treatment and resistance amplification.
Demographic and Clinical Drivers of DR-TB
Age and sex distributions align with national and international data identifying TB as prevalent among working-age adults [
1,
20]. Notably, retreatment status emerged as a critical predictor of MDR-TB, affirming its role as a significant risk factor [
10,
25,
26]. The low frequency of PT2 cases may suggest gaps in detection or continuity of care across treatment episodes [
27], underscoring the importance of robust patient tracking and post-treatment support.
Comparison With Global Evidence
Our findings corroborate WHO data indicating that MDR-TB is concentrated among previously treated individuals, often due to acquired resistance or sustained community transmission [
1,
4,
28]. Similar results from other African settings, such as Sudan, further highlight prior treatment as the principal predictor of MDR, alongside the predominance of inhA and katG mutations in INH resistance [
29,
30]. The elevated bacillary burden among MDR-TB patients in our study, indicated by higher smear and culture positivity, reflects findings that drug-resistant TB often presents with delayed diagnosis and advanced disease [
1,
31,
32]. Persistent culture positivity supports the notion of slow bacterial clearance and underscores the importance of timely, accurate diagnostic services [
33,
34].
Clinical and Programmatic Interpretation of Predictors
Multivariable analysis identified previous treatment, HIV co-infection, and culture positivity as independent predictors of MDR-TB. The lack of association with sex or smear status after adjustment is consistent with other sub-Saharan African studies that emphasize the role of clinical and immunologic factors in resistance development [
1,
38,
39,
40]. The strong link to cultural positivity underscores the need to prioritize patients with high bacillary loads for intensified care and monitoring.
Implications for Clinical Governance and Community-Engaged TB Care
The identified predictors reflect clinical governance priorities, including reliable drug supply, standardized retreatment protocols, and routine diagnostic audits [
41,
42,
43]. CHW-led initiatives focused on education, psychosocial support, and early tracing can effectively reduce default rates and strengthen adherence among high-risk subgroups [
44,
45,
46,
47,
48,
49]. These interventions must be integrated within program frameworks to improve outcomes sustainably.
Anticipated Impact of Strengthened Governance and Engagement
Lessons from similar settings suggest that combining clinical and community strategies could significantly improve treatment outcomes, particularly among patients undergoing retreatment. Model-based projections suggest that governance and engagement interventions may yield improvements of 15–25% in cure rates, although such estimates should be viewed as illustrative rather than predictive [
1,
50].
INH Monoresistance and Mutation Distribution
While Hr-TB prevalence was low (0.6%), all cases featured inhA mutations, echoing findings from Limpopo and Cameroon [
61,
62]. In contrast, katG and dual mutations were more common among MDR-TB cases, aligning with data from KwaZulu-Natal, the Western Cape, and global reports. Overall mutation profiles (inhA > dual > katG) mirror international molecular epidemiology. Notably, 94.1% of cases lacked detectable INH mutations, suggesting either true susceptibility or limitations of targeted mutation panels. Understanding the functional differences between mutations is crucial: katG confers high-level resistance with poor treatment responsiveness, whereas inhA mutations entail low-level resistance with potential for ethionamide cross-resistance [
56,
57].
Routine INH resistance testing and mutation-informed treatment adjustments are essential to improve outcomes and prevent the progression of resistance. Suggested governance interventions include automated mutation alerts, treatment dashboards, monthly diagnostic audits, and CHW-facilitated education and support. Though Hr-TB cases were few, early detection remains vital to mitigate potential clusters of resistance and prevent future MDR outbreaks.
Strengths of the Study
This study contributes context-specific evidence from a rural, high-burden district with historically limited data on DR-TB. Its integrated framework, combining clinical and community lenses, provides actionable insights applicable to decentralized TB programs. The methodological strengths include clear eligibility criteria, rigorous statistical modeling, and ethical integrity, enhancing the credibility and utility of the findings.
Study Limitations
Several limitations are acknowledged. The retrospective use of routine data introduces the potential for incomplete records, though key variables were included in the analyses after quality checks. The low number of Hr-TB cases limits the generalizability of mutation-specific findings. Unmeasured confounders, such as socioeconomic status and access to care, may influence the observed associations. Variability in diagnostic infrastructure likely influenced bacillary burden metrics. The reliance on targeted mutation testing precluded the identification of rarer resistance mechanisms. Finally, overlap with the COVID-19 pandemic may have introduced outcome misclassification, though sensitivity analyses supported the stability of the main results.
Conclusions
MDR-TB remains the predominant resistance challenge in this rural South African setting, strongly associated with retreatment, HIV co-infection, and elevated bacillary load. Hr-TB was infrequent but characterized by inhA-driven resistance, reaffirming the need for mutation-guided care. Strengthened clinical governance, including diagnostic stewardship, prompt resistance testing, and performance auditing, combined with CHW-led adherence support, offers a sustainable strategy to improve treatment outcomes and reduce transmission. This integrated model provides a roadmap for DR-TB control in similar high-burden, resource-constrained environments.
Author Contributions
Conceptualization, M.C. and L.M.F.; methodology, M.C. and L.M.F.; validation, L.M.F. and N.D.; formal analysis, M.C. and L.M.F.; investigation, M.C. and M.C.H.; resources, L.M.F. and T.A.; data curation, L.M.F. and N.D.; writing—original draft preparation, M.C., N.D. and L.M.F.; writing—review and editing, M.C., N.D., M.C.H. and L.M.F.; visualization, L.M.F.; supervision, L.M.F.; project administration, L.M.F.; and funding acquisition, T.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Department of Laboratory Medicine and Pathology, Faculty of Medicine and Health Sciences.
Informed Consent Statement
Not applicable, as this study reviewed only patient files.
Data Availability Statement
Data are available from the corresponding author upon request.
Acknowledgments
The authors are grateful to the healthcare professionals in the healthcare facilities where the patient files were reviewed. To the WSU-TB research group mentors and the WSU-TB Research Group 2025 students, thank you for your support during our travels to the healthcare facilities for data collection.
Conflicts of Interest
No conflicts of interest were declared by the authors.
References
- World Health Organization. Global Tuberculosis Report 2023; WHO: Geneva, Switzerland, 2023; Available online: https://iris.who.int/handle/10665/373828 (accessed on 26 September 2025).
- Kubjane, M. Modelling the South African Tuberculosis Epidemic: The Effect of HIV, Sex Differences, and the Impact of Interventions; University of Cape Town: Cape Town, South Africa, 2023; Available online: https://open.uct.ac.za/items/825c510c-403a-40d1-a9f9-7e6bb65476aa (accessed on 26 September 2025).
- Liebenberg, D.; Gordhan, B.G.; Kana, B.D. Drug-resistant tuberculosis: Implications for transmission, diagnosis, and disease management. Front. Cell. Infect. Microbiol. 2022, 12, 943545. [Google Scholar] [CrossRef]
- Naidoo, K.; Perumal, R.; Cox, H.; Mathema, B.; Loveday, M.; Ismail, N.; Omar, S.V.; Georghiou, S.B.; Daftary, A.; O’Donnell, M.; et al. The epidemiology, transmission, diagnosis, and management of drug-resistant tuberculosis—Lessons from the South African experience. Lancet Infect. Dis. 2024, 24, e559–e575. [Google Scholar] [CrossRef] [PubMed]
- Dookie, N.; Ngema, S.L.; Perumal, R.; Naicker, N.; Padayatchi, N.; Naidoo, K. The changing paradigm of drug-resistant tuberculosis treatment: Successes, pitfalls, and future perspectives. Clin. Microbiol. Rev. 2022, 35, e00180-19. [Google Scholar] [CrossRef] [PubMed]
- Valafar, S.J. Systematic Review of Mutations Associated with Isoniazid Resistance Points to Continuing Evolution and Subsequent Evasion of Molecular Detection, and Potential for Emergence of Multidrug Resistance in Clinical Strains of Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2021, 65, e02091-20. [Google Scholar] [CrossRef]
- van der Heijden, Y.F.; Karim, F.; Mufamadi, G.; et al. Isoniazid-monoresistant tuberculosis is associated with poor outcomes. Int. J. Tuberc. Lung Dis. 2017, 21, 670–676. [Google Scholar] [CrossRef]
- Mpoh, M.M. Safety of Antituberculosis Agents Used for Multidrug-Resistant Tuberculosis; HAL: Bangalore, India, 2023; Available online: https://theses.hal.science/tel-04555756.
- Klopper, M.; van der Merwe, C.J.; van der Heijden, Y.F.; et al. The hidden epidemic of isoniazid-resistant tuberculosis in South Africa. Ann. Am. Thorac. Soc. 2024, 21, 1391–1397. [Google Scholar] [CrossRef]
- van de Water, B.J.; Fulcher, I.; Cilliers, S.; et al. HIV infection and ART associated with TB outcomes…. PLoS ONE 2022, 17, e0266082. [Google Scholar]
- Dlatu, N.; Faye, L.M.; Apalata, T. Outcomes of treating TB patients with DR-TB, HIV and malnutrition. Int. J. Environ. Res. Public Health 2025, 22, 319. [Google Scholar] [CrossRef]
- Premjith, C.; Mangena, P.; Muponda, B.K.; et al. Prevalence of gene mutations associated with isoniazid resistance…. J. Med. Lab. Sci. Technol. South. Afr. 2025, 7, 16–21. [Google Scholar]
- Hosu, M.C.; Tsuro, U.; Dlatu, N.; Faye, L.M.; Apalata, T. Strengthening clinical governance…. Healthcare 2025, 13, 2093. [Google Scholar] [CrossRef] [PubMed]
- Faye, L.M.; Hosu, M.C.; Apalata, T. Drug-resistant tuberculosis in rural Eastern Cape. Int. J. Environ. Res. Public Health 2024, 21, 1594. [Google Scholar] [CrossRef]
- Karmakar, R.; Akond, A.A.; Noor, M.A.; et al. Impact of lobectomy on pulmonary function…. Int. J. Adv. Med. 2025, 12, 356–361. [Google Scholar] [CrossRef]
- Rahman, I.; Willott, C. Drivers of TB disparities in Bangladesh. Challenges 2025, 16, 37. [Google Scholar]
- Jackson, S.; Hauer, B.; Guthmann, J.P.; et al. Characteristics of migrant TB populations in Europe. BMC Infect. Dis. 2025, 25, 11085. [Google Scholar]
- Mehta, R. The Affordable Care Act and Its Influences; University of Texas: Austin, TX, USA, 2025; Available online: https://repositories.lib.utexas.edu/items/9646c082-b35d-403f-8ab1-e7fcb6aed56e.
- Ángel, M. Functional and Evolutionary Genomics of Antibiotic Resistance in Tuberculosis; Universitat Politècnica de València: Valencia, Spain, 2023. [Google Scholar]
- Singh, S.; Zahiruddin, Q.S.; Lakhanpal, S.; et al. Wealth-based inequalities in TB prevalence. BMC Infect. Dis. 2025, 25, 10301. [Google Scholar]
- Dabitao, D.; Bishai, W.R. Sex and gender differences in TB pathogenesis. Curr. Top. Microbiol. Immunol. 2023, 441, 139–183. [Google Scholar] [PubMed]
- Pape, S.; Karki, S.J.; Heinsohn, T.; et al. Tuberculosis case fatality is higher in males. Infection 2024, 52, 1775–1786. [Google Scholar] [CrossRef]
- Mohammed, A.; Aboagye, R.G.; Duodu, P.A.; et al. Sex-based inequalities in TB incidence in Africa. BMC Med. 2025, 23, 324. [Google Scholar] [CrossRef]
- Rickman, H.M.; Phiri, M.D.; Feasey, H.R.; et al. Sex differences in risk of TB infection. Lancet Public Health 2025, 10, e588–e598. [Google Scholar] [CrossRef]
- Brode, S.K.; Dwilow, R.; Kunimoto, D.; et al. Drug-resistant TB. Can. J. Respir. Crit. Care Sleep Med. 2022, 6, 109–128. [Google Scholar]
- Almutairy, B. Extensively and multidrug-resistant bacterial strains: case studies of antibiotics resistance. Front. Microbiol. 2024, 15, 1381511. [Google Scholar] [CrossRef]
- Magee, C.E. Incidence of Non-Affective Psychotic Disorders…; University of British Columbia: Kelowna, BC, Canada, 2025. [Google Scholar]
- Taylor, H.A.; Dowdy, D.W.; Searle, A.R.; et al. Treatment experiences of MDR-TB patients. SSM–Qual. Res. Health 2022, 2, 100042. [Google Scholar] [CrossRef] [PubMed]
- Elduma, A.H.; Mansournia, M.A.; Foroushani, A.R.; et al. Risk factors for MDR-TB in Sudan. Epidemiol. Health 2019, 41, e2019014. [Google Scholar] [CrossRef]
- Sabeel, S.M.; Salih, M.A.; Ali, M.; et al. MDR-TB isolates in Sudan. Tuberc. Res. Treat. 2017, 8340746. [Google Scholar]
- Tembo, B.P.; Malangu, N.G. MDR/RR-TB prevalence in Botswana. BMC Infect. Dis. 2019, 19, 779. [Google Scholar]
- Xi, Y.; Zhang, W.; Qiao, R.J.; Tang, J. Global MDR-TB risk factors. PLoS ONE 2022, 17, e0270003. [Google Scholar]
- Hosu, M.C.; Faye, L.M.; Apalata, T. DR-TB treatment outcomes in high HIV-burden settings. Pathogens 2025, 14, 441. [Google Scholar] [CrossRef] [PubMed]
- Abubakar, M.; Ahmad, N.; Atif, M.; et al. Prognostic accuracy of culture conversion time. BMC Infect. Dis. 2022, 22, 7202. [Google Scholar]
- Price, L.; Gozdzielewska, L.; Hendry, K.; et al. Effectiveness of interventions for HAIs. Lancet Infect. Dis. 2023, 23, e347–e360. [Google Scholar] [CrossRef]
- Lewnard, J.A.; Charani, E.; Gleason, A.; et al. Burden of AMR in LMICs. Lancet 2024, 403, 2439–2454. [Google Scholar] [CrossRef]
- Saggers, R.T.; Mothibi, L.M.; Irwin, A.D.; Naidoo, K.D. Challenges in PICU MDR organisms. Curr. Infect. Dis. Rep. 2023, 25, 233–242. [Google Scholar] [CrossRef]
- Mutabari, D. Time to Sputum Conversion Among DR-TB Patients in Kenya; University of Nairobi: Nairobi, Kenya, 2024. [Google Scholar]
- Karnan, A.; Jadhav, U.; Ghewade, B.; et al. Long vs. short MDR-TB regimens. Cureus 2024, 16, e52706. [Google Scholar]
- Maulana, S.; Lutfian, L.; Wardika, I.J.; et al. Trends in TB medication adherence. Patient Prefer. Adherence 2025, 19, 2213–2226. [Google Scholar] [CrossRef] [PubMed]
- Hensel Mbaveng, A.T.; Kuete, V. Global burden of bacterial infections and resistance. Adv. Bot. Res. 2023, 106, 1–20. [Google Scholar]
- Shutt, A.E.; Ashiru-Oredope, D.; Price, J.; et al. Social determinants of AMR. BMJ Glob. Health 2025, 10, e017389. [Google Scholar] [CrossRef]
- World Health Organization. Access to Essential Antibiotics in Refugee and Migrant Populations; WHO: Geneva, Switzerland, 2022. [Google Scholar]
- Cocker, D.; Birgand, G.; Zhu, N.; et al. Healthcare as an AMR amplifier. Nat. Rev. Microbiol. 2024, 22, 636–649. [Google Scholar] [CrossRef]
- Todowede, O.; Nisar, Z.; Afaq, S.; et al. Integrated depression–TB care pathways. Int. J. Ment. Health Syst. 2025, 19, 6700. [Google Scholar]
- Khatri, R.B.; Endalamaw, A.; Darssan, D.; Assefa, Y. Cancer screening among migrants. PLoS ONE 2025, 20, e0329854. [Google Scholar]
- Mukherjee, M.; Eby, M.; Wang, S.; et al. Managing risk in genetic testing. PLoS ONE 2022, 17, e0270430. [Google Scholar]
- Deshpande, A.; Likhar, R.; Khan, T.; Omri, A. Decoding TB resistance: Current challenges. Expert Rev. Anti-Infect. Ther. 2024, 22, 511–527. [Google Scholar] [CrossRef]
- Zulu, J.M.; Maritim, P.; Halwiindi, H.; et al. Decentralised MDR-TB management. Arch. Public Health 2024, 82, 157. [Google Scholar] [CrossRef]
- Altieri, G. ICPIC 2025 proceedings. Antimicrob. Resist. Infect. Control 2025, 14, 101. [Google Scholar]
- Nono, V.N.; Nantia, E.A.; Mutshembele, A.; et al. katG and inhA mutations in Cameroon. BMC Microbiol. 2025, 25, 127. [Google Scholar]
- Ruhee, N.N. Correlation of Mutations with MIC of Isoniazid in Bangladesh; BRAC University: Dhaka, Bangladesh, 2023. [Google Scholar]
- Shital, K.; Jadhav, S. Clinical profile of H-mono/polydrug-resistant TB. J. Adv. Lung Health 2024, 5, 31–36. [Google Scholar]
- Court, R.G. Pharmacometric Optimization of Second-Line TB Drugs; University of Cape Town: Cape Town, South Africa, 2023. [Google Scholar]
- Gajic, I.; Tomic, N.; Lukovic, B.; et al. Antibacterial agents for MDR bacteria. Antibiotics 2025, 14, 221. [Google Scholar]
- Duffey, M.; Shafer, R.W.; Timm, J.; et al. Combating AMR in malaria, HIV and TB. Nat. Rev. Drug Discov. 2024, 1–19. [Google Scholar]
- Farhat, M.; Cox, H.; Ghanem, M.; et al. Drug-resistant TB: Global concern. Nat. Rev. Microbiol. 2024, 22, 617–635. [Google Scholar] [CrossRef]
- Austin, E.; Briggs, E.; Chueng, A.; et al. Proceedings on dissemination & implementation science. Implement. Sci. 2024, 19. [Google Scholar]
- Manjunatha, V.K.; Meenakshi, D.; Dhadave, M.; et al. MDR-TB in India: Scoping review. Int. J. Med. Sci. Curr. Res. 2025, 8, 391–414. Available online: https://www.ijmscr.com/asset/images/uploads/17569088491376.pdf.
- Morita, A.; Vasiliu, A.; Saluzzo, F.; Akkerman, O.W. Enhancing DR-TB diagnostic capabilities. Pathogens 2024, 13, 1045. [Google Scholar]
- Premjith, C.; Mangena, P.; Muponda, B.K.; et al. INH resistance mutations in Limpopo. J. Med. Lab. Sci. Technol. South. Afr. 2025, 7, 16–21. [Google Scholar]
- Nono, V.N.; Nantia, E.A.; Mutshembele, A.; et al. katG/inhA mutation prevalence in Cameroon. BMC Microbiol. 2025, 25, 127. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).