Submitted:
19 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
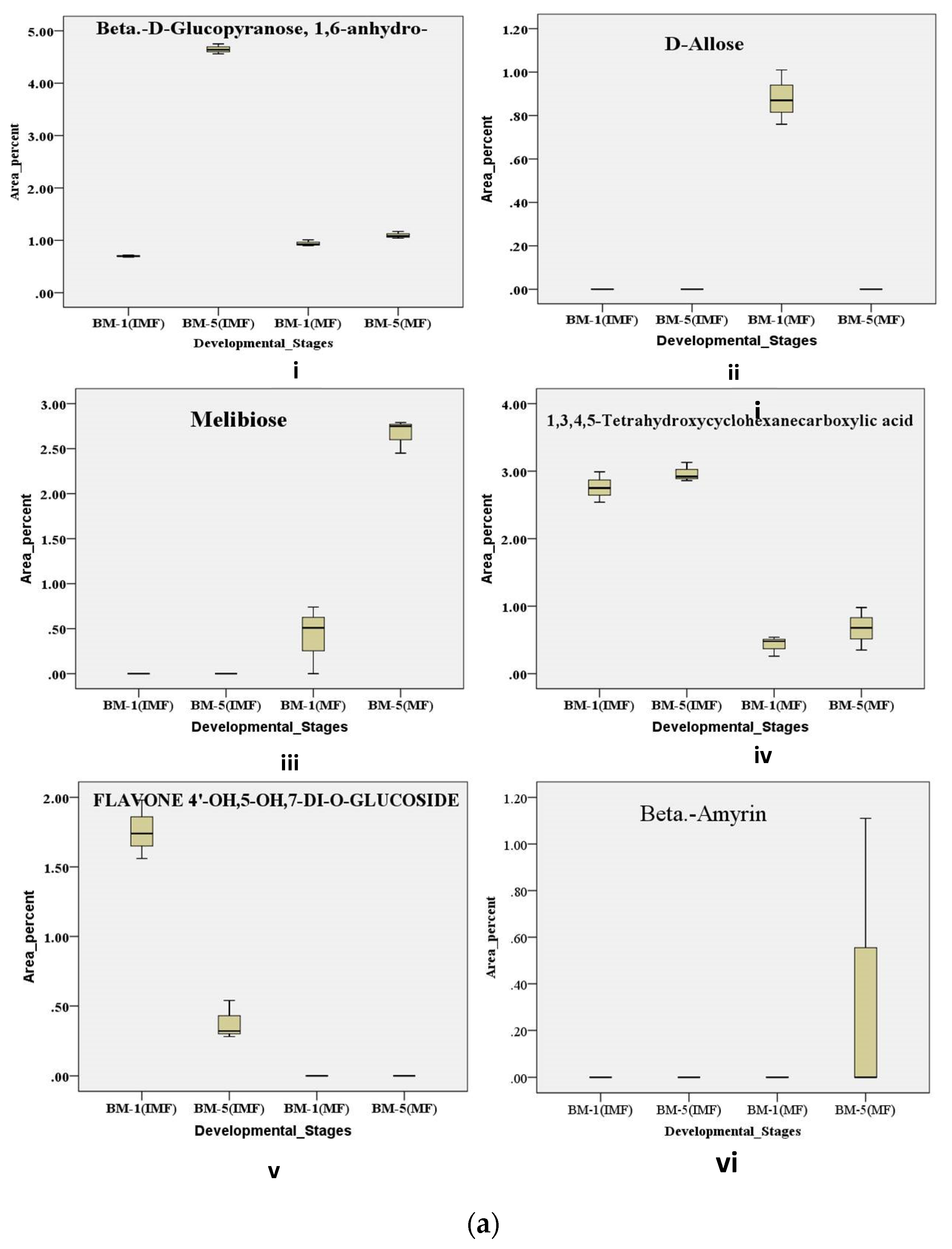
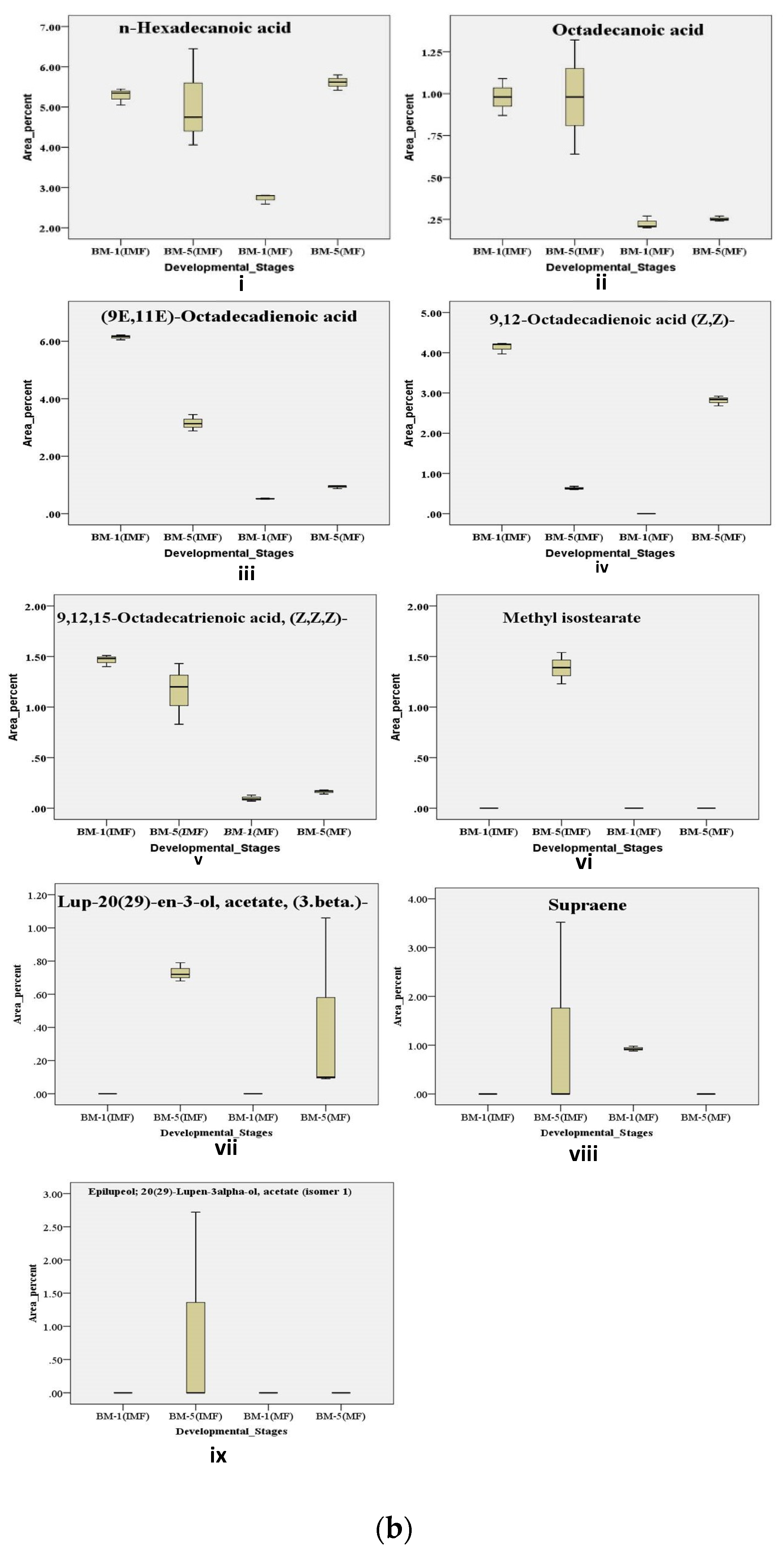
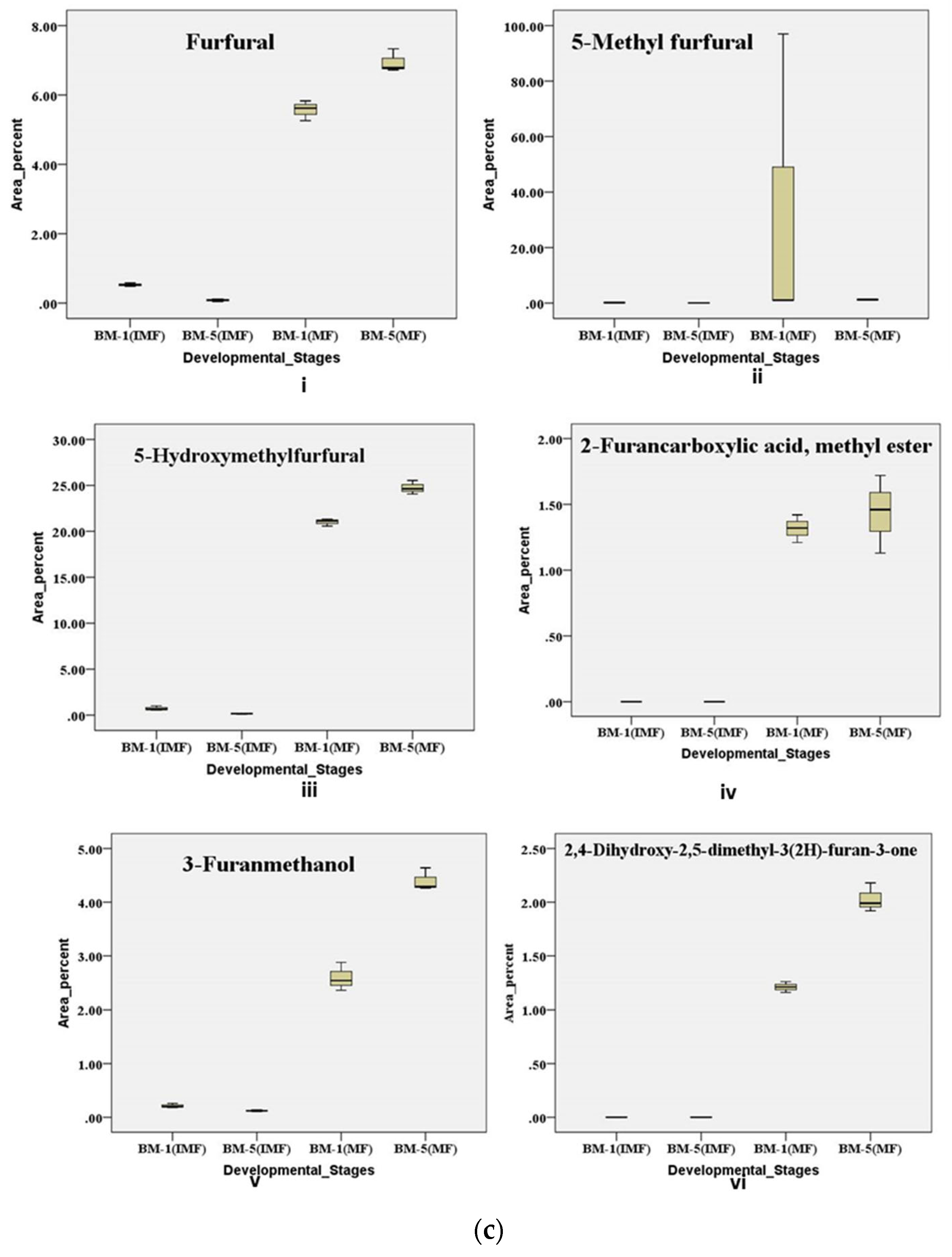
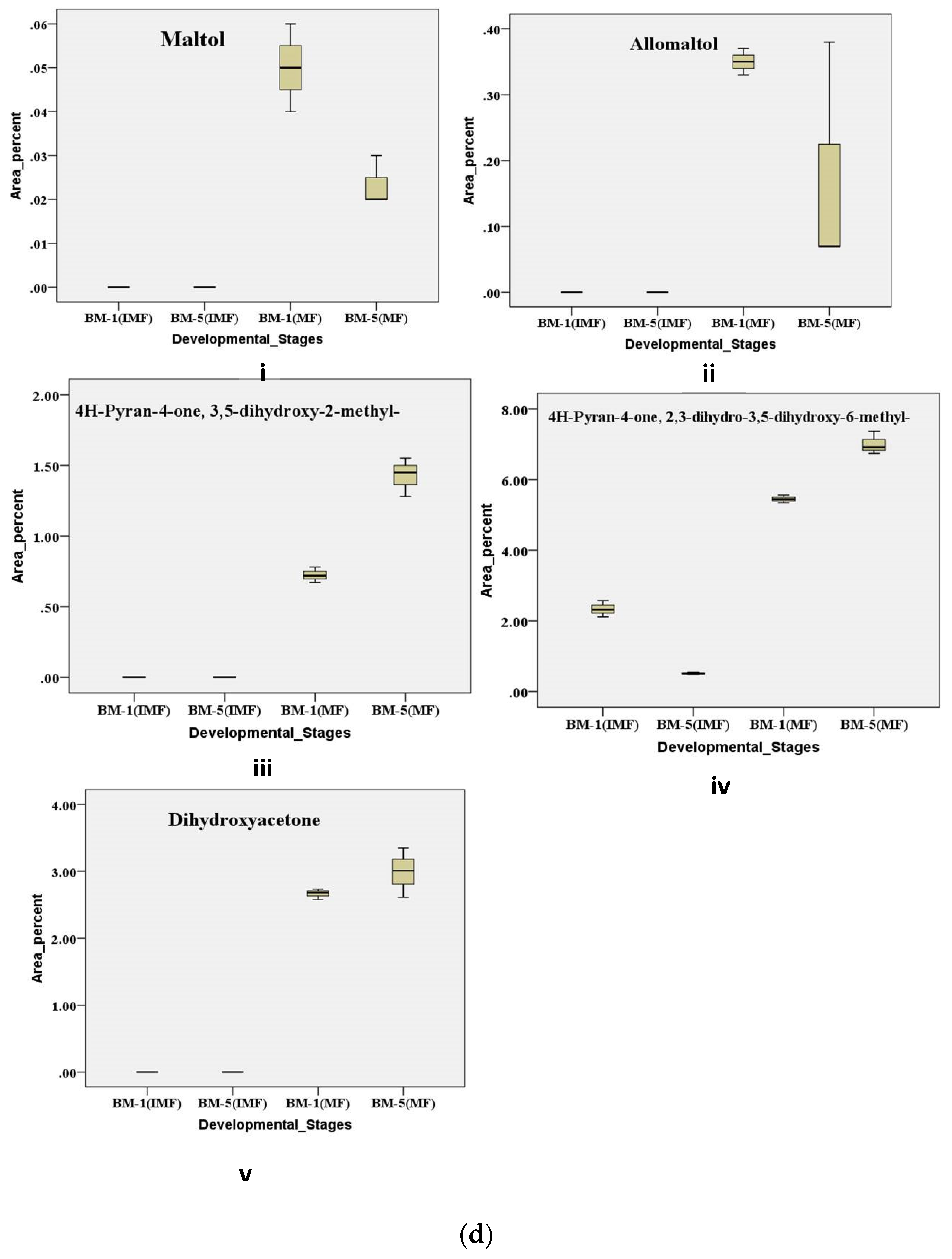
Variations in sweetness and bitterness among Madhuca longifolia flowers strongly influence their processing value and market acceptance, yet the chemo-diversity underlying these traits remains poorly characterized. This study aimed to unravel accession- and stage-specific differences by integrating physico-biochemical, elemental, and metabolic profiling across thirteen accessions (BM-1 to BM-13) from BUAT, Banda. Sensory and textural evaluations revealed wide diversity, with BM-5 displaying superior sweetness and aroma, whereas BM-6, BM-7, and BM-10 were differentiated by firmness, elasticity, and gumminess. Biochemical analyses across flower development showed BM-5 consistently maintained higher sugars and β-carotene, while BM-1 exhibited marked reductions in sugars and total phenolics content; antioxidant activity increased with maturity, with BM-5 remaining the most stable. ICP-MS elemental analysis confirmed BM-5 as mineral-rich compared with lower-performing accessions. GC–MS metabolomic profiling of contrasting accessions (BM-1 and BM-5) across stages identified 376 volatile and semi-volatile metabolites, and multivariate analyses (PCA, VIP, volcano plots, pathway enrichment) revealed distinct stage- and accession-dependent patterns. Mature BM-5 was enriched in fermentation- and aroma-related metabolites such as melibiose, furfural, 5-HMF, and furaneol, whereas BM-1 accumulated defense-linked compounds including catechol, benzyl nitrile, and maltol. Overall, the integrated chemo-diversity landscape identifies BM-5 as a superior accession with high processing potential and value-addition prospects.
Keywords:
1. Introduction
2. Results
2.1. Texture and Sensory Analysis of Flowers from Different Accessions of M. longifolia
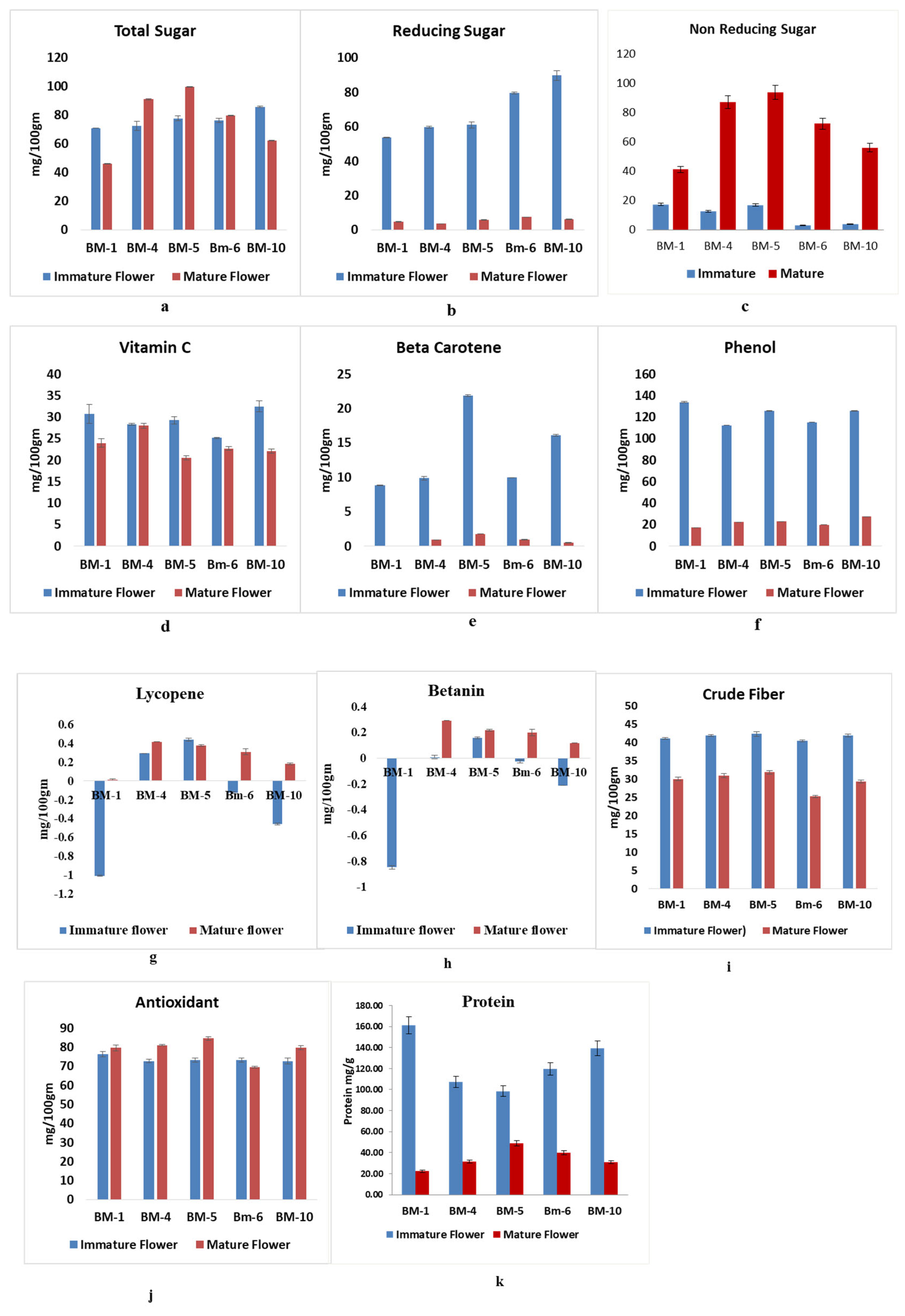
2.2. The Biochemical Analysis of Primary Metabolites During Developmental Stages of Flowering in Selected Accession of M. longifolia
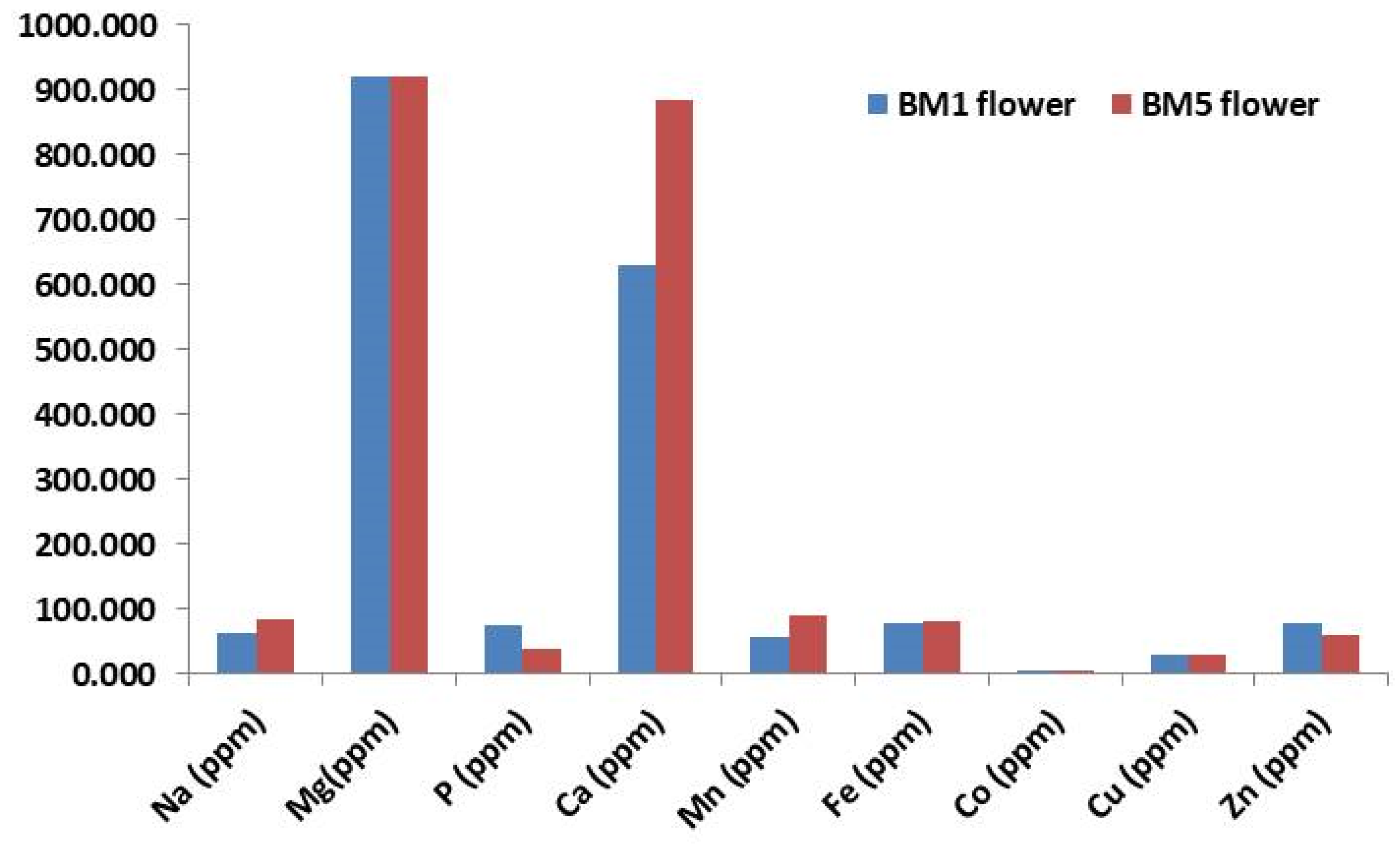
2.3. Elemental Composition of M. longifolia Flower in Contrasting Accessions by ICP–MS
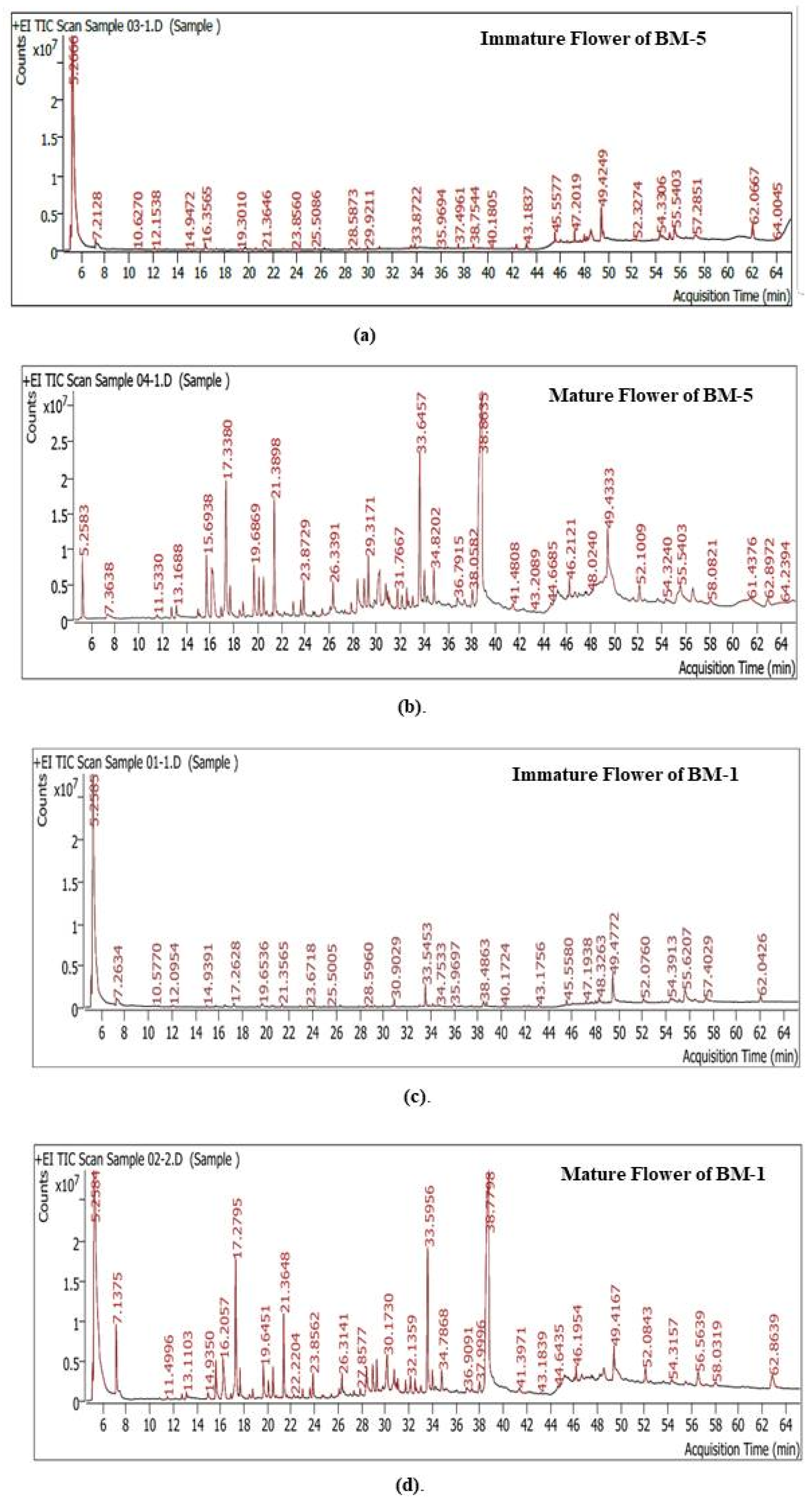
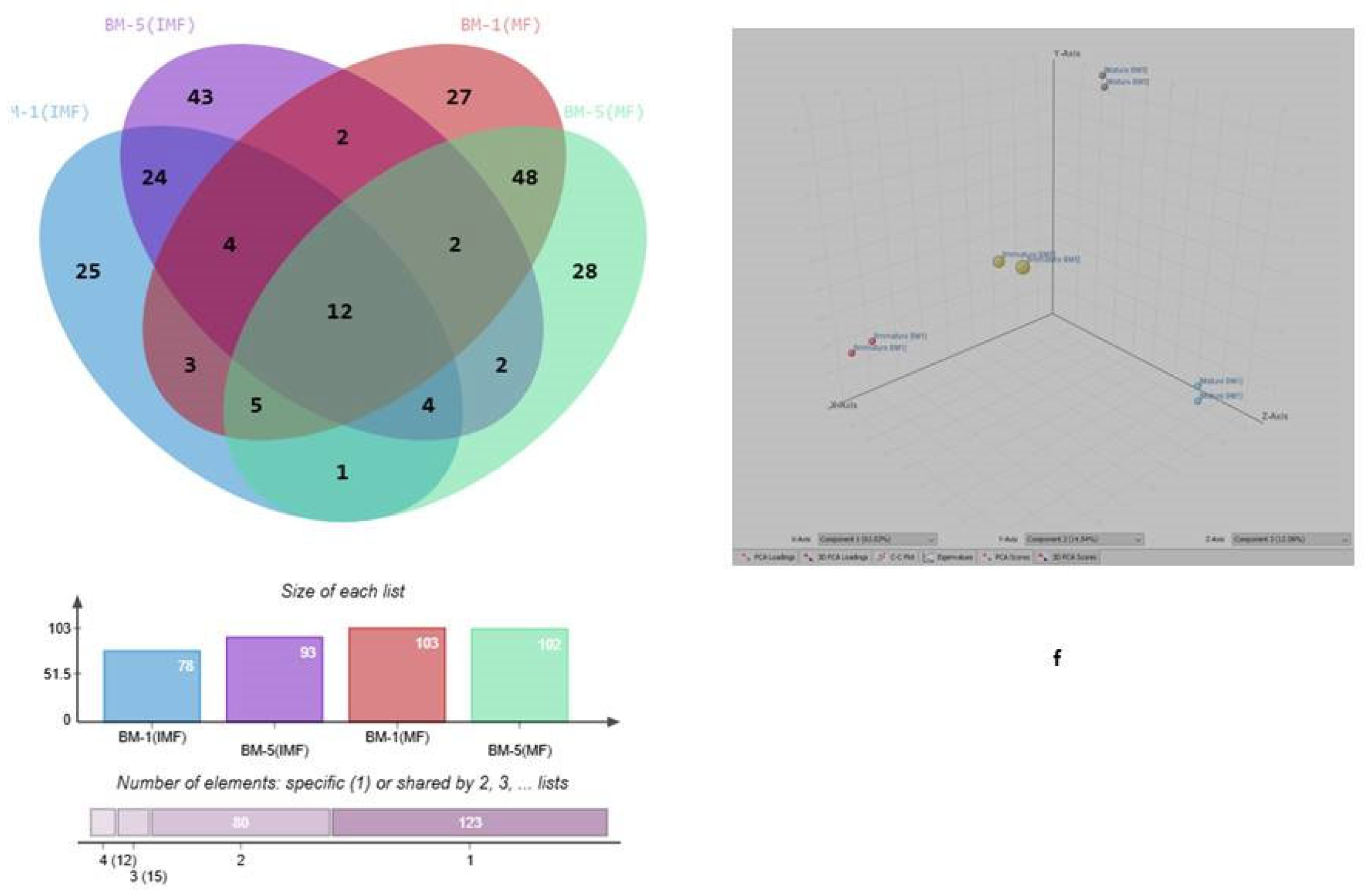
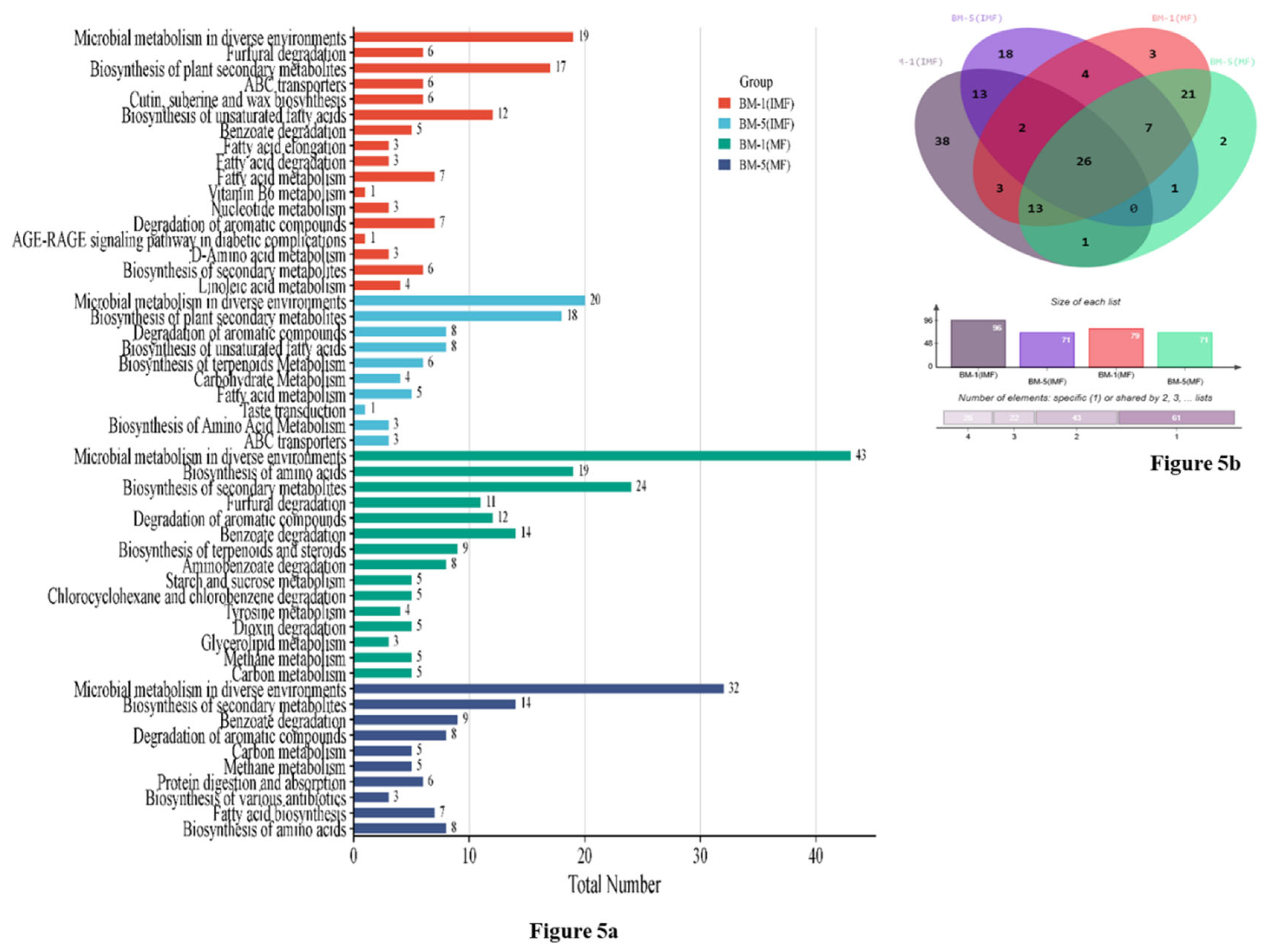
2.4. GC–MS-Based Metabolite Profiling and Pathway Enrichment Analysis During Developmental Stages of Flowering in Contrasting M. longifoliaaccessions
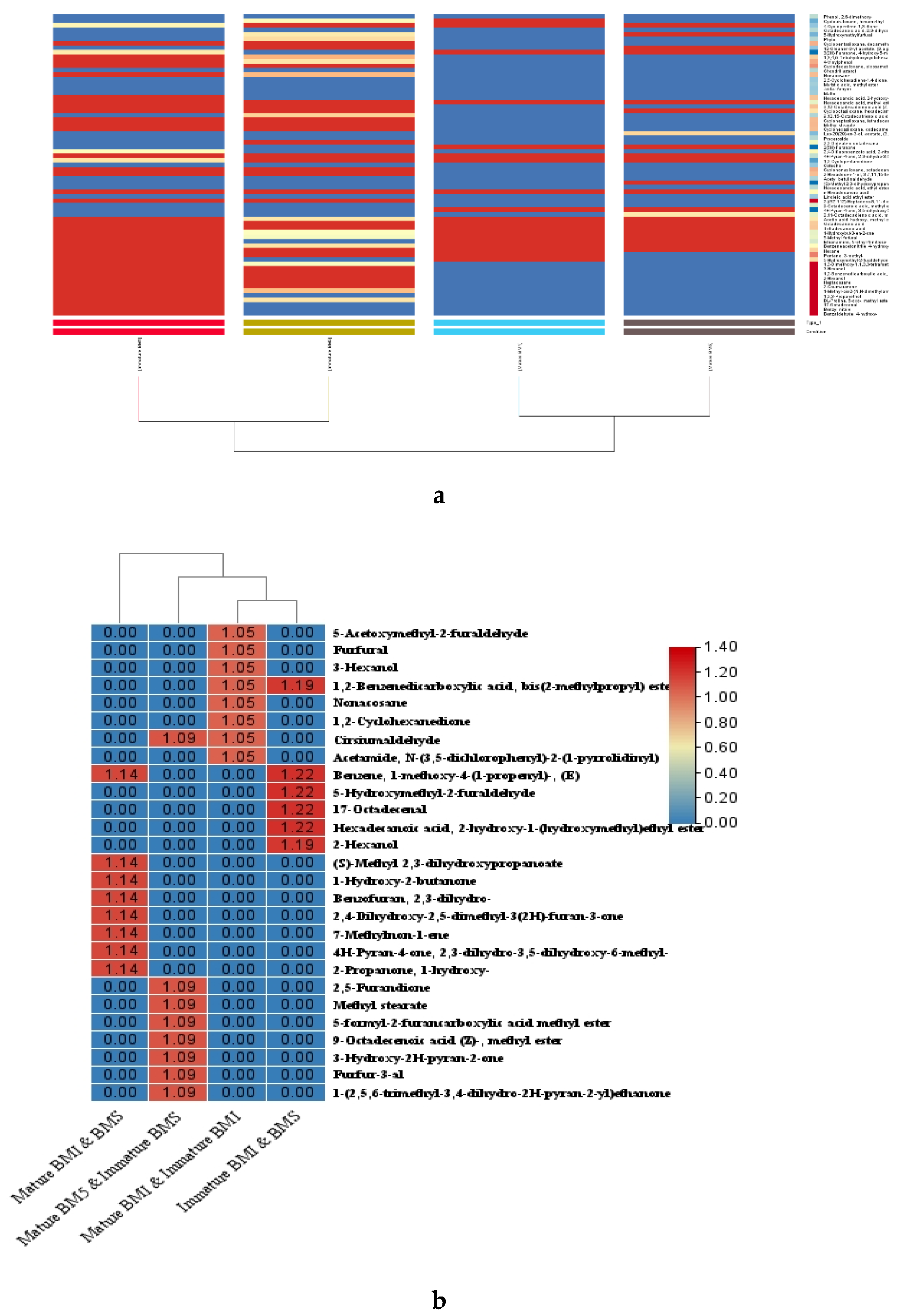
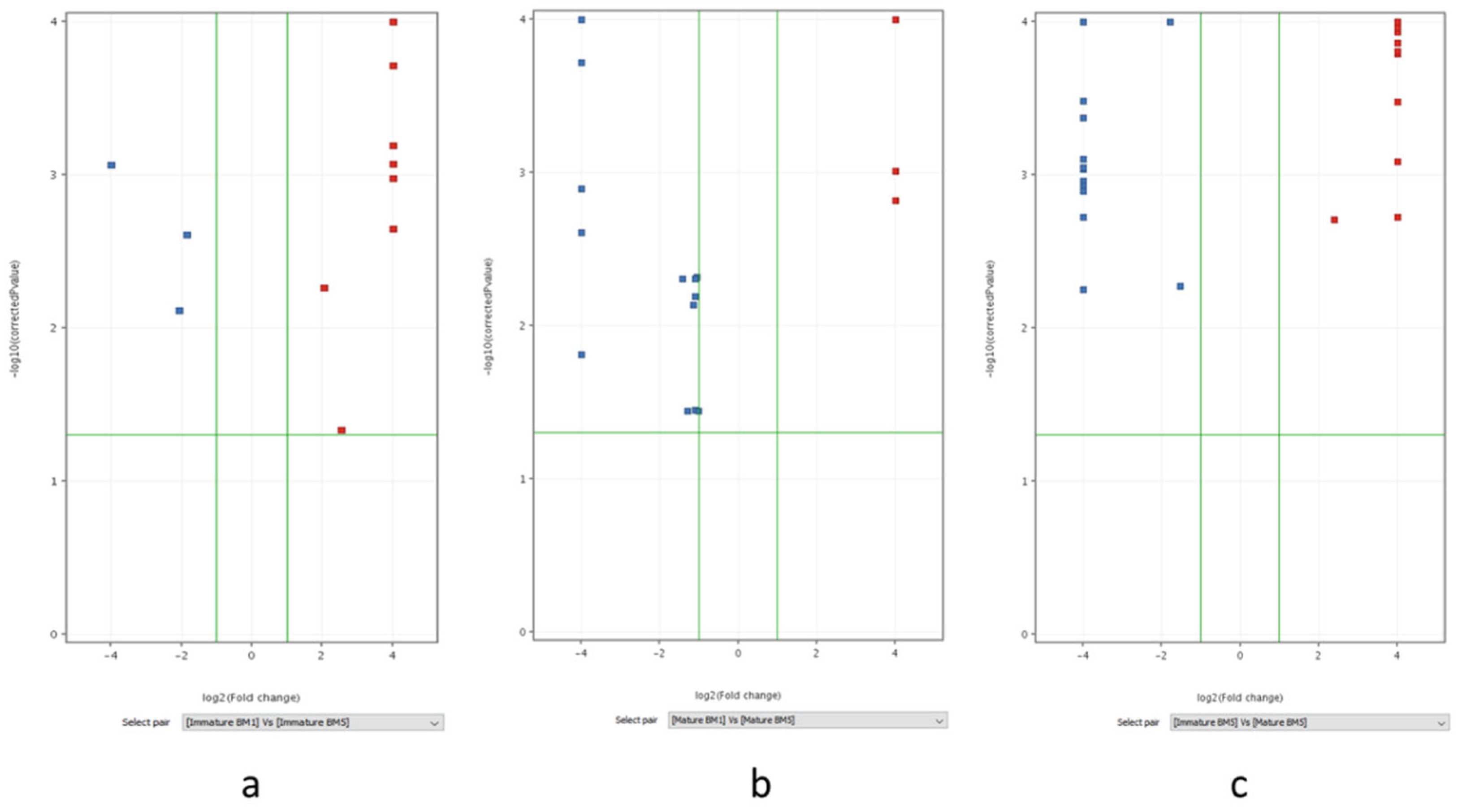
2.5. Hierarchical Clustering, and Variable Importance in Projection (VIP) Score, Analysis in and Volcano Plot-Based Differential Metabolite Profiling Developmental Stages of Flowering in Contrasting Accession of M. longifolia
2.6. Regulation of Key Metabolite During Developmental Stages of Flowering in Contrasting Accession of M. longifolia
2.6.1. Metabolic Regulation of Sugars and Its Derivatives During Developmental Stages of Flowering in Contrasting Accession of M. longifolia
2.6.2. Metabolic Regulation of Fatty Acids Derivative Contributes During Developmental Stages of Flowering in Contrasting Accession of M. longifolia
2.6.3. Metabolic regulation of Furan and Furan Derivative Contributes During Developmental Stages of Flowering in Contrasting Accession of M. longifolia
2.6.4. Metabolic Regulation of Pyranone and Its Derivative Contributes During Developmental Stages of Flowering in Contrasting Accession of M. longifolia
3. Discussion
3.1. Texture and Sensory Analysis of Flowers Collected from Different Accessions of M. longifolia
3.2. The Biochemical Analysis of Primary Metabolites in Developmental Stages of Flowering in Contracting Accession of M. longifolia
3.3. Elemental Composition of M. longifolia Flower in Contrasting Accessions by ICP–MS
3.4. GC–MS-Based Secondary Metabolite Profiling and KEGG Pathway Mapping in Developing Flowers of Contrasting M. longifolia Accessions (BM1&BM5)
3.5. Integrated Hierarchical Clustering, VIP Score, and Volcano Plot Analyses, Reveals Developmental Regulation of Key Metabolites in Contrasting M. longifolia Accessions
4. Materials and Methods
4.1. Collection of Immature and Mature Flower of M. longifolia
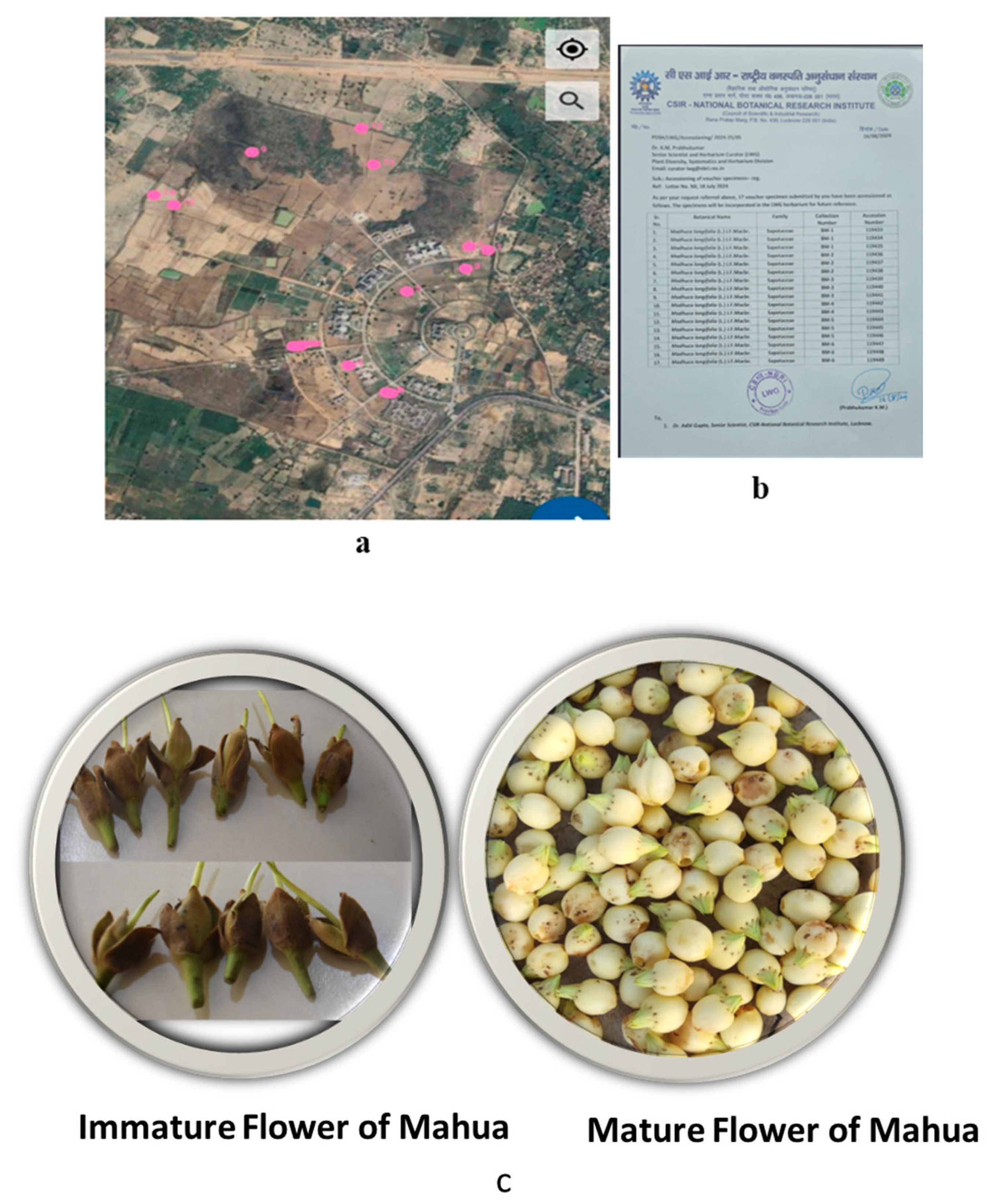
4.2. Sensory and Instrumental Texture Analysis
4.3. Biochemical Analysis of Primary Metabolites
4.3.1. Carbohydrates
4.3.2. Protein
4.3.3. Ascorbic Acid
4.3.4. Antioxidant Activity
4.3.5. β-Carotene
4.3.6. Total Phenolic Content
4.4. Mineral Analysis by ICP-MS
4.5. GC-MS Analysis
4.5.1. Extraction of Volatile Compounds
4.5.2. GC-MS Analysis and Data Interpretation
4.6. Statistical, KEGG, Variable Importance in Projection (VIP) and Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviation
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| GC–MS | Gas Chromatography–Mass Spectrometry |
| PCA | Principal Component Analysis |
| VIP | Variable Importance in Projection |
References
- Gupta, A.; Sanwal, N.; Sharma, N.; Sahu, J.K.; P, H.; Kheto, A. Dynamics of functional and physicochemical properties of Madhuca longifolia flower juice under Saccharomyces cerevisiae fermentation. Food and Humanity 2024, 2, 100244. [Google Scholar] [CrossRef]
- Mishra, A.; Poonia, A. mahua (Madhuca longifolia) flowers: Review on processing and biological properties. Nutr. Food Sci. 2019, 49, 1153–1163. [Google Scholar] [CrossRef]
- Gupta, A.; Chaudhary, R.; Sharma, S. Potential applications of mahua (Madhuca indica) biomass. Waste Biomass Valorization 2012, 3, 175–189. [Google Scholar] [CrossRef]
- Singh, D.; Yadav, E.; Kumar, V.; Verma, A. Madhuca longifolia-embedded silver nanoparticles attenuate diethylnitrosamine (DEN)-induced renal cancer via regulating oxidative stress. Curr. Drug Deliv. 2021, 18, 634–644. [Google Scholar] [CrossRef]
- Baschali, A.; Tsakalidou, E.; Kyriacou, A.; Karavasiloglou, N.; Matalas, A.L. Traditional low-alcoholic and non-alcoholic fermented beverages consumed in European countries: A neglected food group. Nutr. Res. Rev. 2017, 30, 1–24. [Google Scholar] [CrossRef]
- Jayabalan, R.; Waisundara, V.Y. Kombucha as a functional beverage. In Functional and Medicinal Beverages; Academic Press: London, UK, 2019; pp. 413–446. [Google Scholar]
- Singh, V.; Kumar, S.; Rai, A.K. Sensory analysis of bar samples prepared from mahua (Madhuca longifolia) flower syrup using fuzzy logic. Nutrafoods 2018, 17, 137–144. [Google Scholar]
- Pooja; Purohit, A.; Kaur, S.; Yadav, S.K. Identification of a yeast Meyerozymacaribbica M72 from mahua flower for efficient transformation of rice straw into ethanol. Biomass Convers. Biorefin. 2023, 13, 12591–12603. [Google Scholar] [CrossRef]
- Cejudo-Bastante, M.J.; Rodríguez-Pulido, F.J.; Heredia, F.J.; González-Miret, M.L. Assessment of sensory and texture profiles of grape seeds at real maturity stages using image analysis. Foods 2021, 10, 1098. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Liu, M.; Bai, L.; Shang, W.; Ren, R.; Zhao, Z.; Sun, Y. Establishing a berry sensory evaluation model based on machine learning. Foods 2023, 12, 3502. [Google Scholar] [CrossRef]
- Quispe-Sanchez, L.; Mena-Chacon, L.M.; Hernandez-Diaz, E.; Siche, R.; Yoplac, I.; Chuquilín-Goicochea, R.; Vigo, C.N.; Juarez-Contreras, L.; Oliva-Cruz, M. Physicochemical, functional, and sensory properties of berries at different maturity stages. Appl. Food Res. 2025, 5, 101265. [Google Scholar] [CrossRef]
- Singh, V.; Guizani, N.; Al-Zakwani, I.; Al-Shamsi, Q.; Al-Alawi, A.; Rahman, M.S. Sensory texture of date fruits as a function of physicochemical properties and its use in date classification. Acta Aliment. 2015, 44, 119–125. [Google Scholar] [CrossRef]
- Gatti, E.; Di Virgilio, N.; Magli, M.; Predieri, S. Integrating sensory analysis and hedonic evaluation for apple quality assessment. J. Food Qual. 2011, 34, 126–132. [Google Scholar] [CrossRef]
- Swain, M.R.; Kar, S.; Sahoo, A.K.; Ray, R.C. Ethanol fermentation of mahula (Madhuca latifolia L.) flowers using free and immobilized yeast Saccharomyces cerevisiae. Microbiol. Res. 2007, 162, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Rakesh, S. Madhuca longifolia var. latifolia (Roxb.) A. Chev: A plant with medicinal boon. Int. J. Ayurvedic Med. 2023, 14, 593–605. [Google Scholar]
- Li, M.; Li, P.; Ma, F.; Dandekar, A.M.; Cheng, L. Sugar metabolism and accumulation in the fruit of transgenic apple trees with decreased sorbitol synthesis. Hortic. Res. 2018, 5, 60. [Google Scholar] [CrossRef]
- Lungade, P.; Karadbhajne, S.V. Mahua flower (Madhuca indica): Approach of functional, nutritional characteristics and an accompaniment to food products. J. Pharm. Negat. Results 2022, 13, 9093–9104. [Google Scholar]
- Suryawanshi, Y.C.; Mokat, D.N. Chemical composition of essential oil of Madhuca longifolia var. latifolia (Roxb.) A. Chev. flowers. J. Essent. Oil Bear. Plants 2019, 22, 1034–1039. [Google Scholar] [CrossRef]
- Ferrara, G.; Marcotuli, V.; Didonna, A.; Stellacci, A.M.; Palasciano, M.; Mazzeo, A. Ripeness prediction in table grape cultivars by using a portable NIR device. Horticulturae 2022, 8, 613. [Google Scholar] [CrossRef]
- Herrera, J.; Guesalaga, A.; Agosin, E. Shortwave–near infrared spectroscopy for non-destructive determination of maturity of wine grapes. Meas. Sci. Technol. 2003, 14, 689–697. [Google Scholar] [CrossRef]
- Kureel, R.S.; Kishor, R.; Dutt, D.; Pandey, A. Mahua: A Potential Tree Borne Oilseed; National Oilseeds and Vegetable Oils Development Board, Ministry of Agriculture, Government of India: Gurugram, India, 2009; pp. 1–21. [Google Scholar]
- Sinha, J.; Singh, V.; Singh, J.; Rai, A.K. Phytochemistry, ethnomedical uses and future prospects of Mahua (Madhuca longifolia) as a food: A review. J. Nutr. Food Sci. 2017, 7, 573. [Google Scholar] [CrossRef]
- Kassaye, M.; Hagos, M.; Chandravanshi, B.S. Determination of β-carotene in five commonly used Ethiopian vegetables using UV–Vis spectrophotometric method. Chem. Int. 2023, 9, 111–119. [Google Scholar]
- Sultana, R.; Polash, M.A.S.; Sakil, M.A.; Shorna, S.I.; Rahman, M.S.; Rahman, M.A.; Hossain, M.A. Health-promoting pigments and bioactive compounds of six vegetables grown in Bangladesh. Asian J. Med. Biol. Res. 2019, 5, 280–285. [Google Scholar] [CrossRef]
- Suwanaruang, T. Analyzing lycopene content in fruits. Agric. Agric. Sci. Procedia 2016, 11, 46–48. [Google Scholar] [CrossRef]
- Sandate-Flores, L.; Rodríguez-Hernández, D.V.; Rostro-Alanis, M.; Melchor-Martínez, E.M.; Brambila-Paz, C.; Sosa-Hernández, J.E.; Iqbal, H.M.N. Evaluation of three methods for betanin quantification in fruits from cacti. MethodsX 2022, 9, 101746. [Google Scholar] [CrossRef]
- Choo, K.Y.; Ong, Y.Y.; Lim, R.L.H.; Tan, C.P.; Ho, C.W. Study on bioaccessibility of betacyanins from red dragon fruit (Hylocereuspolyrhizus). Food Sci. Biotechnol. 2019, 28, 1163–1169. [Google Scholar] [CrossRef]
- Qiu, Y.; Wang, R.; Zhang, E.; Shang, Y.; Feng, G.; Wang, W.; Ma, Y.; Bai, W.; Zhang, W.; Xu, Z.; Shi, W.; Niu, X. Carotenoid biosynthesis profiling unveils the variance of flower coloration in Tageteserecta and enhances fruit pigmentation in tomato. Plant Sci. 2024, 347, 112207. [Google Scholar] [CrossRef]
- Jayasree, B.; Harishankar, N.; Rukmini, C. Chemical composition and biological evaluation of Mahua flowers. Indian J. Nutr. Diet. 1998, 35, 1–6. [Google Scholar]
- Muzolf-Panek, M.; Waśkiewicz, A. Relationship between phenolic compounds, antioxidant activity and color parameters of red table grape skins using linear ordering analysis. Appl. Sci. 2022, 12, 6146. [Google Scholar] [CrossRef]
- Nile, S.H.; Kim, S.H.; Ko, E.Y.; Park, S.W. Polyphenolic contents and antioxidant properties of different grape (Vitisvinifera, V. labrusca, and V. hybrid) cultivars. Biomed Res. Int. 2013, 2013, 718065. [Google Scholar] [CrossRef]
- Dwivedi, A.; Priyadarshini, A.; Induar, S. Mahua (Madhuca longifolia) flower and its application in food industry: A review. Int. J. Chem. Stud. 2022, 10, 80–84. [Google Scholar]
- Zheng, J.; Yu, X.; Maninder, M.; Xu, B. Total phenolics and antioxidants profiles of commonly consumed edible flowers in China. Int. J. Food Prop. 2018, 21, 1524–1540. [Google Scholar] [CrossRef]
- Zhang, C.; Guo, X.; Guo, R.; Zhu, L.; Qiu, X.; Yu, X.; Chai, J.; Gu, C.; Feng, Z. Insights into the effects of extractable phenolic compounds and Maillard reaction products on the antioxidant activity of roasted wheat flours with different maturities. Food Chemistry 2023, 17, 100548. [Google Scholar] [CrossRef]
- Bolchini, S.; Morozova, K.; Ferrentino, G.; Scampicchio, M. Assessing antioxidant properties of Maillard reaction products: Methods and potential applications as food preservatives. Eur. Food Res. Technol. 2025, 251, 2039–2059. [Google Scholar] [CrossRef]
- Sood, S.; Methven, L.; Cheng, Q. Role of taste receptors in salty taste perception of minerals and amino acids and developments in salt reduction strategies: A review. Crit. Rev. Food Sci. Nutr. 2025, 65, 3444–3458. [Google Scholar] [CrossRef]
- Jakubczyk, K.; Janda, K.; Watychowicz, K.; Łukasiak, J.; Wolska, J. Garden nasturtium (Tropaeolummajus L.)—A source of mineral elements and bioactive compounds. Rocz. Panstw. Zakl. Hig. 2018, 69, 119–126. [Google Scholar] [PubMed]
- Sood, S.; Methven, L.; Cheng, Q. Role of taste receptors in salty taste perception of minerals and amino acids and developments in salt reduction strategies: A review. Crit. Rev. Food Sci. Nutr. 2025, 65, 3444–3458. [Google Scholar] [CrossRef]
- Ahern, G.P.; Brooks, I.M.; Miyares, R.L.; Wang, X.-B. Extracellular cations sensitize and gate capsaicin receptor TRPV1 modulating pain signaling. J. Neurosci. 2005, 25, 5109–5116. [Google Scholar] [CrossRef] [PubMed]
- Riera, C.E.; Vogel, H.; Simon, S.A.; Damak, S.; le Coutre, J. Sensory attributes of complex tasting divalent salts are mediated by TRPM5 and TRPV1 channels. J. Neurosci. 2009, 29, 2654–2662. [Google Scholar] [CrossRef]
- Medler, K.F. Calcium signaling in taste cells. Biochim. Biophys. Acta Mol. Cell Res. 2015, 1853, 2025–2032. [Google Scholar] [CrossRef] [PubMed]
- Keast, R.S. The effect of zinc on human taste perception. J. Food Sci. 2003, 68, 1871–1877. [Google Scholar] [CrossRef]
- Orth, H.N.; Pirkwieser, P.; Benthin, J.; Koehler, M.; Sterneder, S.; Parlar, E.; Schaudy, E.; Lietard, J.; Michel, T.; Boger, V.; Dunkel, A.; Somoza, M.M.; Somoza, V. Bitter taste receptor TAS2R43 co-regulates mechanisms of gastric acid secretion and zinc homeostasis. Int. J. Mol. Sci. 2024, 26, 6017. [Google Scholar] [CrossRef] [PubMed]
- Huseynli, L.; Walser, C.; Blumenthaler, L.; Vene, K.; Dawid, C. Toward a comprehensive understanding of flavor of sunflower products: A review of confirmed and prospective aroma- and taste-active compounds. Foods 2025, 14, 1940. [Google Scholar] [CrossRef]
- Quan, W.; Jin, J.; Qian, C.; Li, C.; Zhou, H. Characterization of volatiles in flowers from four Rosa chinensis cultivars by HS-SPME-GC×GC-QTOFMS. Front. Plant Sci. 2023, 14, 1060747. [Google Scholar] [CrossRef]
- Li, S.; Dong, M.; Yang, J.; Cheng, X.; Shen, X.; Liu, S.; Han, B. Selective hydrogenation of 5-(hydroxymethyl)furfural to 5-methylfurfural over single atomic metals anchored on Nb2O5. Nat. Commun. 2021, 12, 584. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.; Benfodda, Z.; Bénimélis, D.; Fontaine, J.X.; Molinié, R.; Meffre, P. Extraction and identification of volatile organic compounds in scentless flowers of 14 Tillandsia species using HS-SPME/GC-MS. Metabolites 2022, 12, 628. [Google Scholar] [CrossRef] [PubMed]
- Lo, M.M.; Benfodda, Z.; Bénimélis, D.; Fontaine, J.X.; Molinié, R.; Meffre, P. Extraction and identification of volatile organic compounds emitted by fragrant flowers of three Tillandsia species by HS-SPME/GC-MS. Metabolites 2021, 11, 594. [Google Scholar] [CrossRef]
- Shi, T.; Yue, Y.; Shi, M.; Chen, M.; Yang, X.; Wang, L. Exploration of floral volatile organic compounds in six typical Lycoris taxa by GC–MS. Plants 2019, 8, 422. [Google Scholar] [CrossRef]
- Perchuk, I.; Shelenga, T.; Gurkina, M.; Miroshnichenko, E.; Burlyaeva, M. Composition of primary and secondary metabolite compounds in seeds and pods of asparagus bean (Vignaunguiculata (L.) Walp.) from China. Molecules 2020, 25, 3778. [Google Scholar] [CrossRef]
- Darwish, A.G.; Das, P.R.; Olaoye, E.; Gajjar, P.; Ismail, A.; Mohamed, A.G.; Tsolova, V.; Hassan, N.A.; El Kayal, W.; Walters, K.J. Untargeted flower volatilome profiling highlights differential pollinator attraction strategies in muscadine. Front. Plant Sci. 2025, 16, 1548564. [Google Scholar] [CrossRef]
- Chautá, A.; Kumar, A.; Mejia, J.; Stashenko, E.E.; Kessler, A. Defensive functions and potential ecological conflicts of floral stickiness. Sci. Rep. 2022, 12, 19848. [Google Scholar] [CrossRef]
- Sasidharan, R.; Junker, R.R.; Eilers, E.J.; Müller, C. Floral volatiles evoke partially similar responses in both florivores and pollinators and are correlated with non-volatile reward chemicals. Ann. Bot. 2023, 132, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Basallo, O.; Perez, L.; Lucido, A.; Sorribas, A.; Vilaprinyo, E.; Albacete, A.; Fraser, P.D.; Christou, P.; Capell, T.; Alves, R. Changing biosynthesis of terpenoid precursors in rice through synthetic biology. Front. Plant Sci. 2023, 14, 1133299. [Google Scholar] [CrossRef]
- Dudareva, N.; Negre, F.; Nagegowda, D.A.; Orlova, I. Plant volatiles: Recent advances and future perspectives. Crit. Rev. Plant Sci. 2006, 25, 417–440. [Google Scholar] [CrossRef]
- Wang, Q.; Quan, S.; Xiao, H. Towards efficient terpenoid biosynthesis: Manipulating IPP and DMAPP supply. Bioresour. Bioprocess. 2019, 6, 1–13. [Google Scholar] [CrossRef]
- Liu, S.; Sun, H.; Ma, G.; Zhang, T.; Wang, L.; Pei, H.; Li, X.; Gao, L. Insights into flavor and key influencing factors of Maillard reaction products: A recent update. Front. Nutr. 2022, 9, 973677. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.T.; Zheng, X.; Li, S. Tea aroma formation. Food Sci. Hum. Wellness 2015, 4, 9–27. [Google Scholar] [CrossRef]
- Yang, W.; Zhang, C.; Li, C.; Huang, Z.Y.; Miao, X. Pathway of 5-hydroxymethyl-2-furaldehyde formation in honey. J. Food Sci. Technol. 2019, 56, 2417–2425. [Google Scholar] [CrossRef]
- Yang, J.; Jiang, D.; Shui, X.; Lei, T.; Zhang, H.; Zhang, Z.; Zhang, Q. Effect of 5-HMF and furfural additives on bio-hydrogen production by photo-fermentation from giant reed. Bioresour. Technol. 2022, 347, 126743. [Google Scholar] [CrossRef]
- Chen, C.; Lv, M.; Hu, H.; Huai, L.; Zhu, B.; Fan, S.; Zhang, J. 5-Hydroxymethylfurfural and its downstream chemicals: A review of catalytic routes. Adv. Mater. 2024, 36, 2311464. [Google Scholar] [CrossRef] [PubMed]
- Pagare, P.P.; McGinn, M.; Ghatge, M.S.; Shekhar, V.; Alhashimi, R.T.; Pierce, B.D.; Abdulmalik, O.; Zhang, Y.; Safo, M.K. The antisickling agent, 5-hydroxymethyl-2-furfural: Other potential pharmacological applications. Med. Res. Rev. 2024, 44, 2707–2729. [Google Scholar] [CrossRef]
- Fagbemi, K.O.; Aina, D.A.; Olutunmbi, M.; Naidoo, K.K.; Coopoosamy, R.M.; Olajuyigbe, O.O. Bioactive compounds, antibacterial and antioxidant activities of methanol extract of Tamarindusindica Linn. Sci. Rep. 2022, 12, 9432. [Google Scholar] [CrossRef]
- Shapla, U.M.; Solayman, M.; Alam, N.; Khalil, M.I.; Gan, S.H. 5-Hydroxymethylfurfural (HMF) levels in honey and other food products: Effects on bees and human health. Chem. Cent. J. 2018, 12, 35. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Lee, B.H.; Wei, K. 5-Hydroxymethylfurfural mitigates lipopolysaccharide-stimulated inflammation via suppression of MAPK, NF-κB and mTOR activation in RAW 264.7 cells. Molecules 2019, 24, 275. [Google Scholar] [CrossRef]
- Sancheti, S.V.; Yadav, G.D. Synthesis of environment-friendly, sustainable, and nontoxic bio-lubricants: A critical review of advances and a path forward. Biofuels Bioprod. Biorefin. 2022, 16, 1172–1195. [Google Scholar] [CrossRef]
- Sharma, B.K.; Karmakar, G.; Shah, R.; Ghosh, P.; Sarker, M.I.; Erhan, S.Z. Sustainable lubricant formulations from natural oils: A short review. Renew. Sustain. Energy Rev. 2023, 173, 113095. [Google Scholar]
- Câmara, J.S.; Perestrelo, R.; Ferreira, R.; Berenguer, C.V.; Pereira, J.A.M.; Castilho, P.C. Plant-derived terpenoids: A plethora of bioactive compounds with several health functions and industrial applications—A comprehensive overview. Molecules 2024, 29, 3861. [Google Scholar] [CrossRef]
- Dalimunthe, A.; Gunawan, M.C.; Utari, Z.D.; Dinata, M.R.; Halim, P.; Pakpahan, N.E.S.; Sitohang, A.I.; Sukarno, M.A.; Harahap, Y.; Setyowati, E.P.; Park, M.N.; Yusoff, S.D.; Zainalabidin, S.; Prananda, A.T.; Mahadi, M.K.; Kim, B.; Harahap, U.; Syahputra, R.A. In-depth analysis of lupeol: Delving into the diverse pharmacological profile. Front. Pharmacol. 2024, 15, 1461478. [Google Scholar] [CrossRef]
- Sowa, R.M. The Pharmacological Potential of Lupeol and Derivatives: A Corroborative Analysis. Doctoral Dissertation, BRAC University, Dhaka, Bangladesh, 2022. [Google Scholar]
- Liu, K.; Zhang, X.; Xie, L.; Deng, M.; Chen, H.; Song, J.; Luo, J. Lupeol and its derivatives as anticancer and anti-inflammatory agents: Molecular mechanisms and therapeutic efficacy. Pharmacol. Res. 2021, 164, 105373. [Google Scholar] [CrossRef]
- Romero-Estrada, A.; Maldonado-Magaña, A.; González-Christen, J.; Bahena, S.M.; Garduño-Ramírez, M.L.; Rodríguez-López, V.; Alvarez, L. Anti-inflammatory and antioxidative effects of six pentacyclictriterpenes isolated from the Mexican copal resin of Burseracopallifera. BMC Complement. Altern. Med. 2016, 16, 422. [Google Scholar] [CrossRef]
- Liao, Y.; Zeng, L.; Tan, H.; Cheng, S.; Dong, F.; Yang, Z. Biochemical pathway of benzyl nitrile derived from L-phenylalanine in tea (Camellia sinensis) and its formation in response to post-harvest stresses. J. Agric. Food Chem. 2020, 68, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Balabanlı, Z.Y. Investigating the Aqueous Behavior of D-Glucose, D-Fructose and D-Allulose by Molecular Dynamics Simulations and Nuclear Magnetic Resonance Relaxometry. Master’s Thesis, Middle East Technical University, Ankara, Turkey, 2022. [Google Scholar]
- Ma, X.L.; Wang, X.C.; Zhang, J.N.; Liu, J.N.; Ma, M.H.; Ma, F.L.; She, Y. A study of flavor variations during the flaxseed roasting procedure by developed real-time SPME GC–MS coupled with chemometrics. Food Chem. 2023, 410, 135453. [Google Scholar] [CrossRef]
- Van Laar, A.; Grootaert, C.; Rajkovic, A.; Desmet, T.; Beerens, K.; Van Camp, J. Rare sugar metabolism and impact on insulin sensitivity along the gut–liver–muscle axis in vitro. Nutrients 2023, 15, 1593. [Google Scholar] [CrossRef]
- Afzal, S.; Chaudhary, N.; Singh, N.K. Role of soluble sugars in metabolism and sensing under abiotic stress. In Plant Growth Regulators: Signalling under Stress Conditions; Springer: Cham, Switzerland, 2021; pp. 305–334. [Google Scholar]
- Tan, M.J.; Ye, J.M.; Turner, N.; Hohnen-Behrens, C.; Ke, C.Q.; Tang, C.P.; Chen, T.; Weiss, H.C.; Gesing, E.R.; Ye, Y. Antidiabetic activities of triterpenoids isolated from bitter melon associated with activation of the AMPK pathway. Chem. Biol. 2008, 15, 263–273. [Google Scholar] [CrossRef]
- Clifford, M.N.; Kerimi, A.; Williamson, G. Bioavailability and metabolism of chlorogenic acids (acyl-quinic acids) in humans. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1299–1352. [Google Scholar] [CrossRef]
- Agati, G.; Brunetti, C.; Di Ferdinando, M.; Ferrini, F.; Pollastri, S.; Tattini, M. Functional roles of flavonoids in photoprotection: New evidence, lessons from the past. Plant Physiol. Biochem. 2013, 72, 35–45. [Google Scholar] [CrossRef]
- Gomathi, D.; Kalaiselvi, M.; Ravikumar, G.; Devaki, K.; Uma, C. GC–MS analysis of bioactive compounds from the whole plant ethanolic extract of Evolvulusalsinoides (L.) L. J. Food Sci. Technol. 2015, 52, 1212–1217. [Google Scholar]
- Yadav, S.; Suneja, P.; Hussain, Z.; Abraham, Z.; Mishra, S.K. Prospects and potential of Madhuca longifolia (Koenig) J.F. Macbride for nutritional and industrial purpose. Biomass Bioenergy 2011, 35, 1539–1544. [Google Scholar] [CrossRef]
- Saeed, N.M.; El-Demerdash, E.; Abdel-Rahman, H.M.; Algandaby, M.M.; Al-Abbasi, F.A.; Abdel-Naim, A.B. Anti-inflammatory activity of methyl palmitate and ethyl palmitate in different experimental rat models. Toxicol. Appl. Pharmacol. 2012, 264, 84–93. [Google Scholar] [CrossRef]
- Schwab, W. Natural 4-hydroxy-2,5-dimethyl-3(2H)-furanone (Furaneol®). Molecules 2013, 18, 6936–6951. [Google Scholar] [CrossRef] [PubMed]
- Yin, T.; Song, C.; Li, H.; Wang, S.; Wei, W.; Meng, J.; Liu, Q. Unveiling stage-specific flavonoid dynamics underlying drought tolerance in sweet potato (Ipomoea batatas L.) via integrative transcriptomic and metabolomic analyses. Plants 2025, 14, 2383. [Google Scholar] [CrossRef]
- Chan, P.N.A. Chemical properties and applications of food additives: Flavor, sweeteners, food colors, and texturizers. In Handbook of Food Chemistry; Springer: Berlin, Heidelberg, Germany, 2015; pp. 101–129. [Google Scholar]
- Ahn, H.; Lee, G.; Han, B.C.; Lee, S.H.; Lee, G.S. Maltol, a natural flavor enhancer, inhibits NLRP3 and non-canonical inflammasome activation. Antioxidants 2022, 11, 1923. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Lee, K.G. Formation and reduction of furan in Maillard reaction model systems consisting of various sugars, amino acids, and furan precursors. J. Agric. Food Chem. 2014, 62, 5978–5982. [Google Scholar] [CrossRef] [PubMed]
- Gatti, E.; Di Virgilio, N.; Magli, M.; Predieri, S. Integrating sensory analysis and hedonic evaluation for apple quality assessment. J. Food Qual. 2011, 34, 126–132. [Google Scholar] [CrossRef]
- Manickavasagan, A.; Ganeshmoorthy, K.; Claereboudt, M.R.; Al-Yahyai, R.; Khriji, L. Non-destructive measurement of total soluble solid content of dates using near infrared imaging. Emir. J. Food Agric. 2014, 26, 970–978. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Nelson, N. A photometric adaptation of the Somogyi method for the determination of glucose. J. Biol. Chem. 1952, 153, 375–380. [Google Scholar] [CrossRef]
- Somogyi, M. Notes on sugar determination. J. Biol. Chem. 1952, 195, 19–23. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Harris, L.J.; Ray, S.N. Determination of ascorbic acid in plant materials. Biochem. J. 1935, 29, 2013–2019. [Google Scholar]
- Gupta, E.; Vajpayee, G.; Purwar, S.; Shakyawar, S.; Alok, S.; Sundaram, S. Phytochemical screening and in vitro studies of antioxidant and antimicrobial activity of extracts of dried Stevia rebaudiana leaves. Int. J. Pharm. Sci. Res. 2017, 8, 3354–3360. [Google Scholar] [CrossRef]
- Hagos, M.; Redi-Abshiro, M.; Chandravanshi, B.S.; Yaya, E.E. Development of analytical methods for determination of β-carotene in pumpkin (Cucurbita maxima) flesh, peel, and seed powder samples. Int. J. Anal. Chem. 2022, 2022, 9363692. [Google Scholar] [CrossRef]
- Kamtekar, S.; Keer, V.; Patil, V. Estimation of phenolic content, flavonoid content, antioxidant and α-amylase inhibitory activity of marketed polyherbal formulation. J. Appl. Pharm. Sci. 2014, 4, 061–065. [Google Scholar]
- Ahmad, I.; Rawoof, A.; Dubey, M.; Ramchiary, N. ICP-MS based analysis of mineral elements composition during fruit development in Capsicum germplasm. J. Food Compos. Anal. 2021, 101, 103977. [Google Scholar] [CrossRef]
- Ma, C.; Li, J.; Chen, W.; Wang, W.; Qi, D.; Pang, S.; Miao, A. Study of aroma formation and transformation during the manufacturing process of oolong tea by SPME–GC–MS combined with chemometrics. Food Res. Int. 2018, 108, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.K.; Zhang, D.D.; Liu, Y.L. Survey of polycyclic aromatic hydrocarbons of vegetable oils and oilseeds by GC–MS in China. Food Addit. Contam. Part A 2016, 33, 603–611. [Google Scholar] [CrossRef]
- Wold, S.; Sjöström, M.; Eriksson, L. PLS-regression: A basic tool of chemometrics. Chemom. Intell. Lab. Syst. 2001, 58, 109–130. [Google Scholar] [CrossRef]
- Jaiswal, R.P.; Chugh, V.; Nagar, S.; Purwar, S.; Azam, A.; Verma, A. Screening of metabolites and metabolic pathways in five different Ocimum species from the same origin using GC–MS. Biochem. Res. Int. 2025, 2025, 7121687. [Google Scholar] [CrossRef]
- Lucero, M.; Estell, R.; Tellez, M.; Fredrickson, E. A retention index calculator simplifies identification of plant volatile organic compounds. Phytochem. Anal. 2009, 20, 378–384. [Google Scholar] [CrossRef]
- Kanehisa, M.; Araki, M.; Goto, S.; Hattori, M.; Hirakawa, M.; Itoh, M.; Katayama, T.; Kawashima, S.; Okuda, S.; Tokimatsu, T.; Yamanishi, Y. KEGG for linking genomes to life and the environment. Nucleic Acids Res. 2007, 36, D480–D484. [Google Scholar] [CrossRef]
- Xie, C.; Mao, X.; Huang, J.; Ding, Y.; Wu, J.; Dong, S.; Kong, L.; Gao, G.; Li, C.Y.; Wei, L. KOBAS 2.0: A web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 2011, 39, W316–W322. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



