Submitted:
24 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials Selection
2.2. Characterizations
2.3. Time -Temperature Fire Test
3. Results and Discussion
3.1. Physicochemical Characteristics
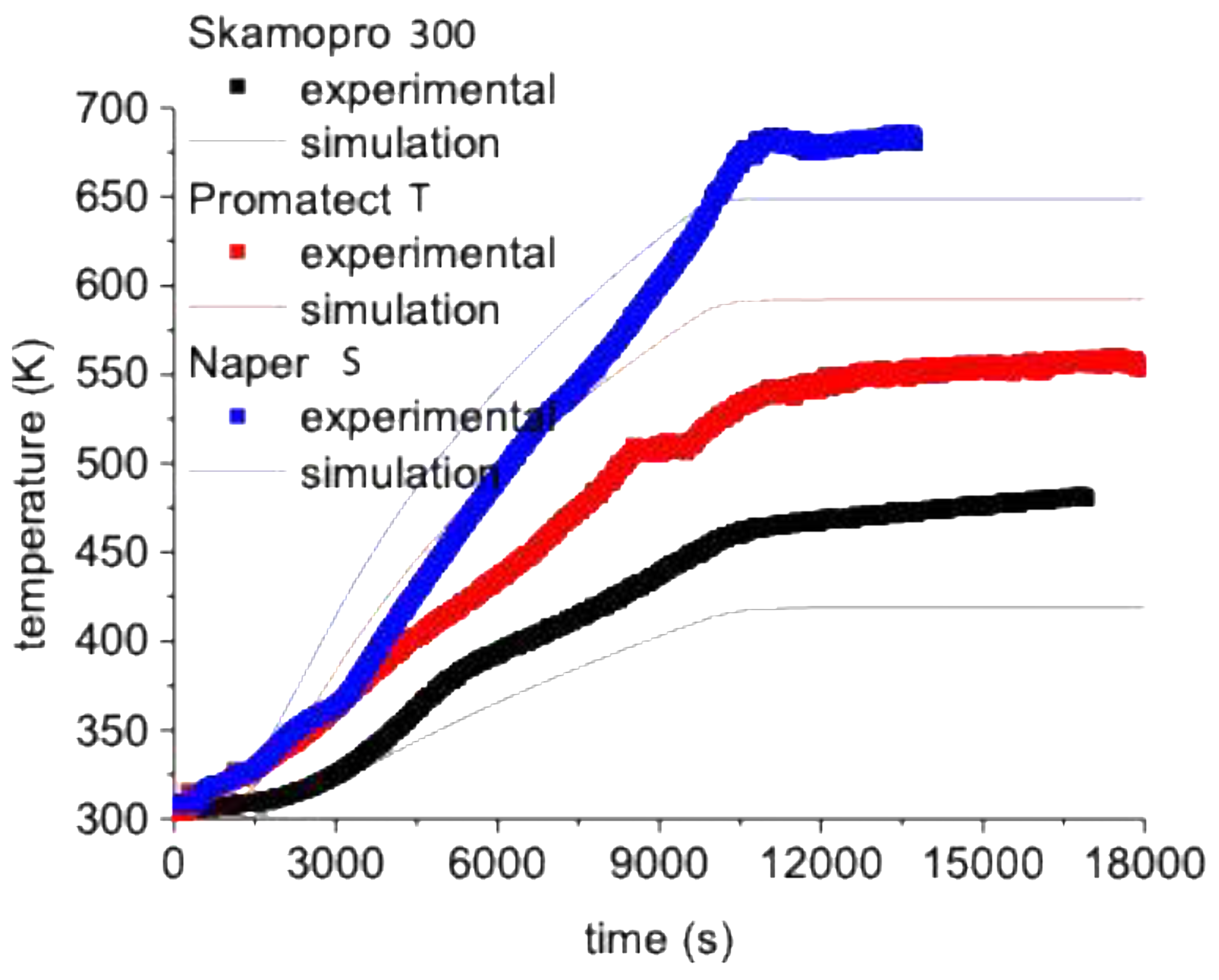
3.2. Time -Temperature Fire Test
3.3. Finite Element Modelling (FEM) for Simulating Heat Exchange During Fire
| Heating rate (K/s) (3000- 6000 s) | Maximum temperature (K) | |||||
| experimental | Simulation | Error (%) | experimental | Simulation | Errror (%) | |
| Skampro-300 | 0.019 | 0.014 | 26 | 480 | 420 | 12 |
| Promatect T | 0.025 | 0.029 | 16 | 550 | 590 | 7 |
| Naper-S | 0.039 | 0.035 | 10 | 680 | 650 | 4 |
4. Conclusions
References
- V. Kodur, Properties of Concrete at Elevated Temperatures, ISRN Civil Engineering 2014 (2014) e468510. [CrossRef]
- K. Sakr, E. EL-Hakim, Effect of high temperature or fire on heavy weight concrete properties, Cement and Concrete Research 35 (2005) 590–596. [CrossRef]
- M.C. Yew, M.K. Yew, R.K.K. Yuen, Experimental Analysis of Lightweight Fire-Rated Board on Fire Resistance, Mechanical, and Acoustic Properties, Fire 6 (2023). [CrossRef]
- K. Mróz, I. Hager, K. Korniejenko, Material Solutions for Passive Fire Protection of Buildings and Structures and Their Performances Testing, Procedia Engineering 151 (2016) 284–291. [CrossRef]
- A. Di Padova, A. Tugnoli, V. Cozzani, T. Barbaresi, F. Tallone, Identification of fireproofing zones in Oil&Gas facilities by a risk-based procedure, Journal of Hazardous Materials 191 (2011) 83–93. [CrossRef]
- R. Gellert, 8 - Inorganic mineral materials for insulation in buildings **This chapter is dedicated to Dr Walter F. Cammerer on the occasion of his 90th birthday., in: M.R. Hall (Ed.), Materials for Energy Efficiency and Thermal Comfort in Buildings, Woodhead Publishing, 2010: pp. 193–228. [CrossRef]
- I.H. Aziz, M.M. Al Bakri Abdullah, H.C. Yong, L.Y. Ming, K. Hussin, A. Surleva, E.A. Azimi, Manufacturing parameters influencing fire resistance of geopolymers: A review, Proceedings of the Institution of Mechanical Engineers, Part L: Journal of Materials: Design and Applications 233 (2019) 721–733. [CrossRef]
- D.I. Kolaitis, E.K. Asimakopoulou, M.A. Founti, Fire behaviour of gypsum plasterboard wall assemblies: CFD simulation of a full-scale residential building, Case Studies in Fire Safety 7 (2017) 23–35. [CrossRef]
- Y. Wang, Y.-J. Chuang, C.-Y. Lin, The Performance of Calcium Silicate Board Partition Fireproof Drywall Assembly with Junction Box under Fire, Advances in Materials Science and Engineering 2015 (2015) 642061. [CrossRef]
- L. Kristanto, H. Sugiharto, S.W.D. Agus, S.A. Pratama, Calcium Silicate Board as Wall-facade, Procedia Engineering 171 (2017) 679–688. [CrossRef]
- M. Gravit, E. Golub, The fire resistant ceiling construction in a hydrocarbon fire, MATEC Web Conf. 245 (2018) 03004. [CrossRef]
- R. Prieler, H. Gerhardter, M. Landfahrer, C. Gaber, C. Schluckner, M. Eichhorn-Gruber, G. Schwabegger, C. Hochenauer, Development of a numerically efficient approach based on coupled CFD/FEM analysis for virtual fire resistance tests—Part B: Deformation process of a steel structure, Fire and Materials 44 (2020) 704–723. [CrossRef]
- 13. Cell Dimensions and Composition of Nanocrystalline Calcium Silicate Hydrate Solid Solutions. Part 2: X-Ray and Thermogravimetry Study - Garbev - 2008 - Journal of the American Ceramic Society - Wiley Online Library, (n.d.). (accessed December 13, 2018). [CrossRef]
- R. Maddalena, C. Hall, A. Hamilton, Effect of silica particle size on the formation of calcium silicate hydrate using thermal analysis, Thermochimica Acta (2018). [CrossRef]
- G. Taglieri, C. Mondelli, V. Daniele, E. Pusceddu, A. Trapananti, Synthesis and X-Ray Diffraction Analyses of Calcium Hydroxide Nanoparticles in Aqueous Suspension, Advances in Materials Physics and Chemistry 03 (2013) 108. [CrossRef]
- E. Tajuelo Rodriguez, K. Garbev, D. Merz, L. Black, I.G. Richardson, Thermal stability of C-S-H phases and applicability of Richardson and Groves’ and Richardson C-(A)-S-H(I) models to synthetic C-S-H, Cement and Concrete Research 93 (2017) 45–56. [CrossRef]
- S. Grangeon, F. Claret, C. Lerouge, F. Warmont, T. Sato, S. Anraku, C. Numako, Y. Linard, B. Lanson, On the nature of structural disorder in calcium silicate hydrates with a calcium/silicon ratio similar to tobermorite, Cement and Concrete Research 52 (2013) 31–37. [CrossRef]
- M.A. Trezza, Hydration study of ordinary portland cement in the presence of zinc ions, Materials Research 10 (2007) 331–334. [CrossRef]
- V. Jokanović, B. Čolović, M. Mitrić, D. Marković, B. Ćetenović, Synthesis and Properties of a New Dental Material Based on Nano-Structured Highly Active Calcium Silicates and Calcium Carbonates, International Journal of Applied Ceramic Technology 11 (2014) 57–64. [CrossRef]
- R. Puntharod, C. Sankram, N. Chantaramee, P. Pookmanee, K.J. Haller, Ceramic Processing Research, (n.d.) 4.
- P. Yu, R.J. Kirkpatrick, B. Poe, P.F. McMillan, X. Cong, Structure of Calcium Silicate Hydrate (C-S-H): Near-, Mid-, and Far-Infrared Spectroscopy, Journal of the American Ceramic Society 82 (1999) 742–748. [CrossRef]
- Wollastonite R040131 - RRUFF Database: Raman, X-ray, Infrared, and Chemistry, (n.d.). http://rruff.info/wollastonite/display=default/R040131 (accessed December 19, 2018).
- C. Zhang, A. Usmani, Heat transfer principles in thermal calculation of structures in fire, Fire Safety Journal 78 (2015) 85–95. [CrossRef]
- S.W. Churchill, R.U. Churchill, A comprehensive correlating equation for heat and component transfer by free convection, AIChE Journal 21 (1975) 604–606. [CrossRef]
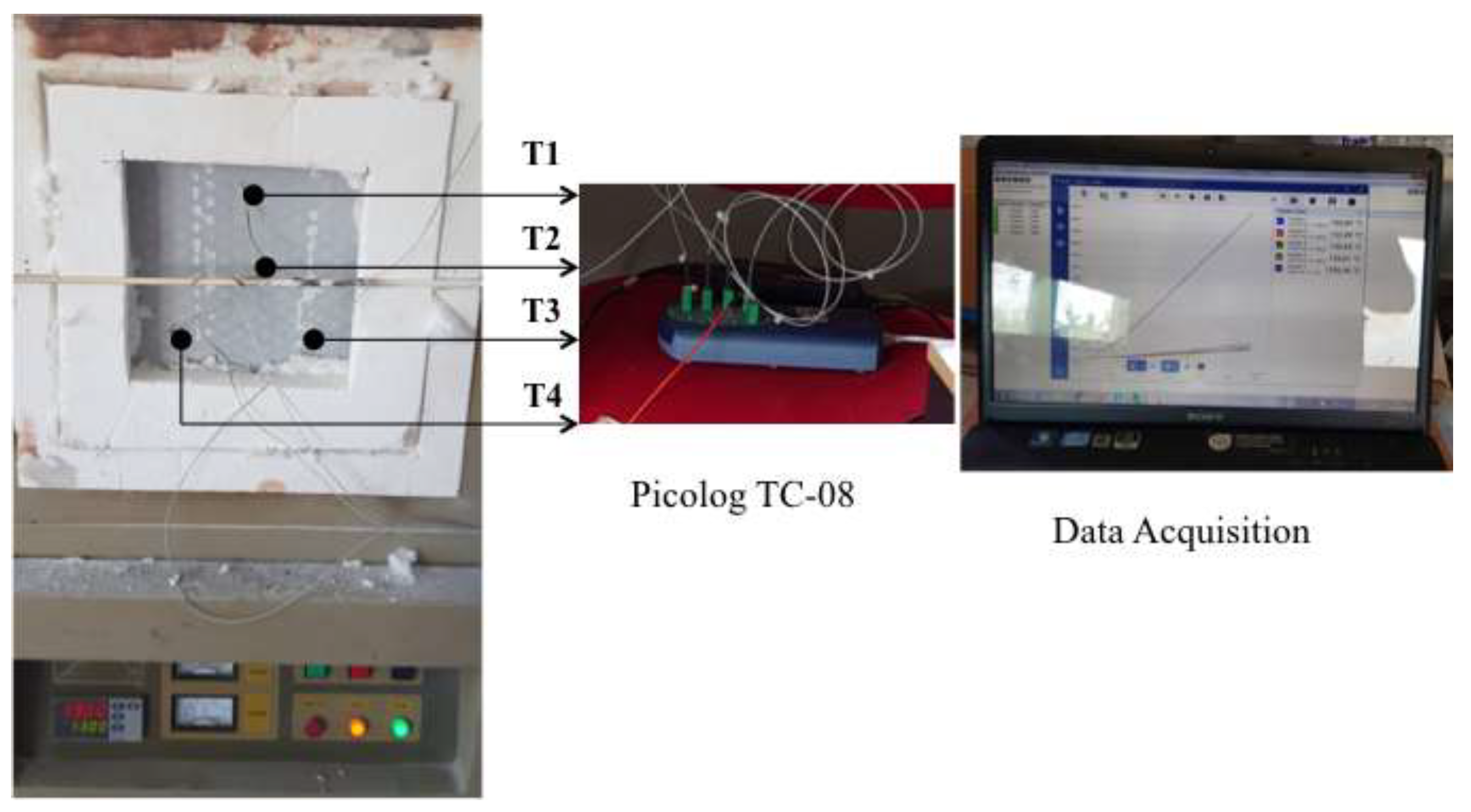
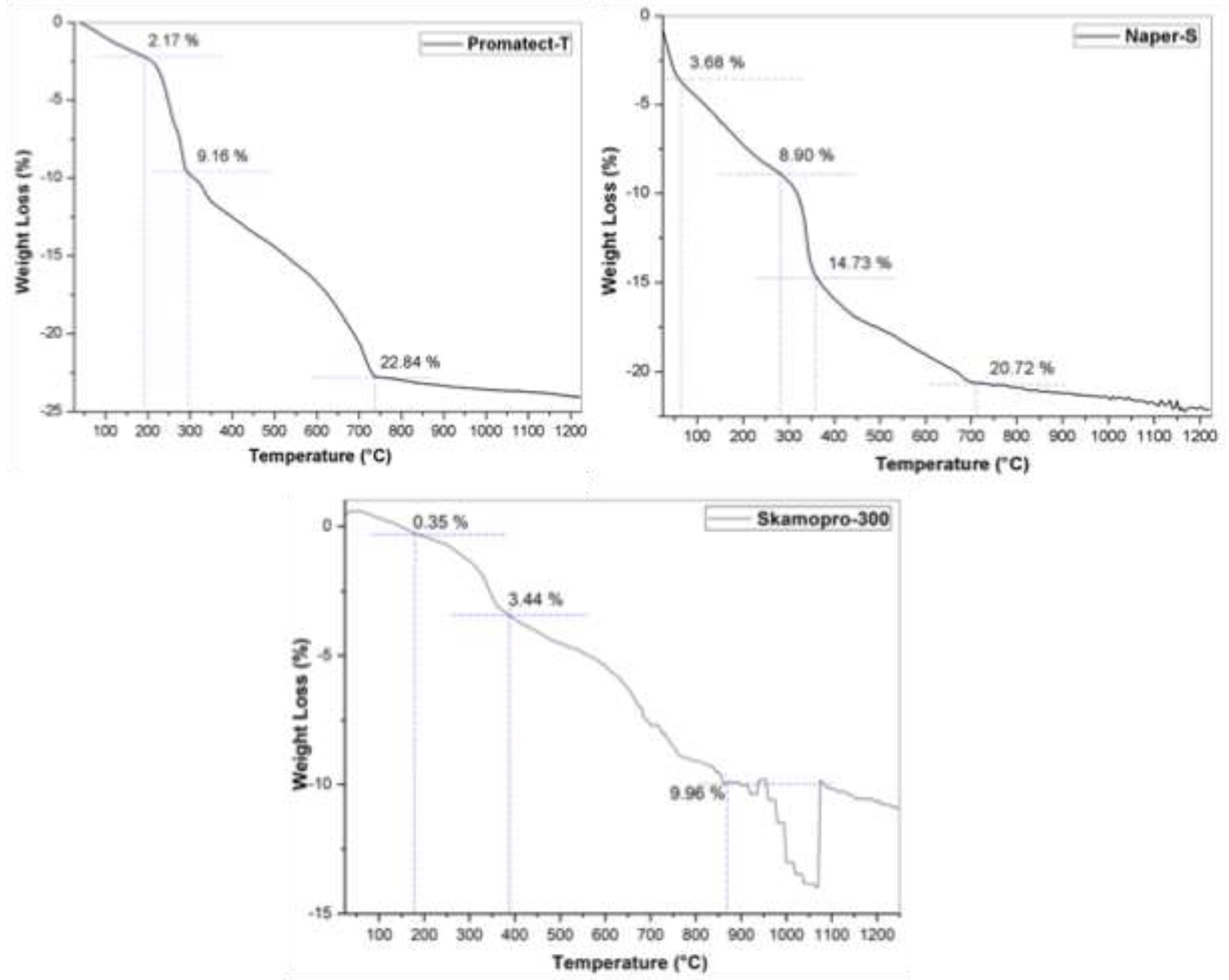
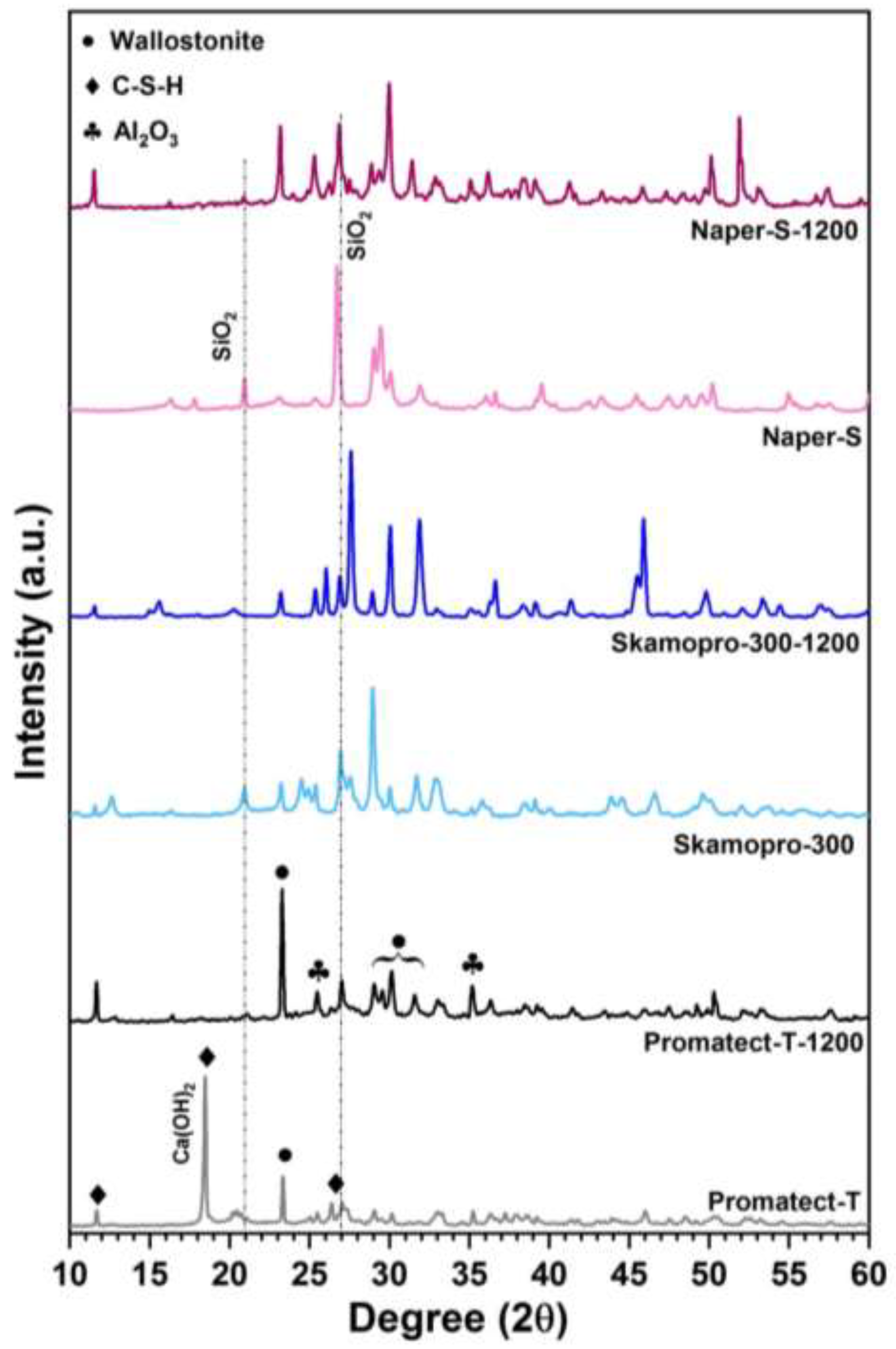
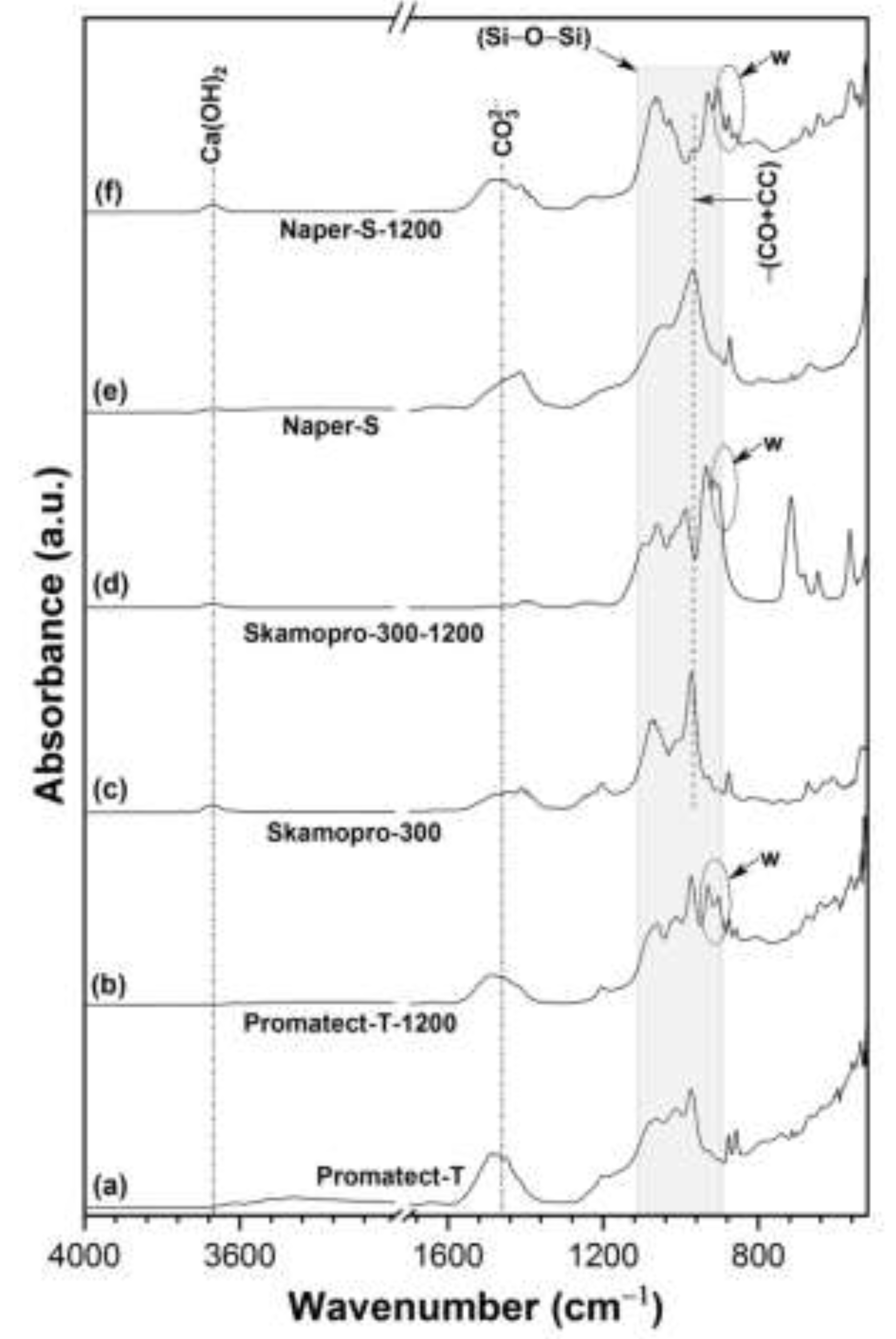

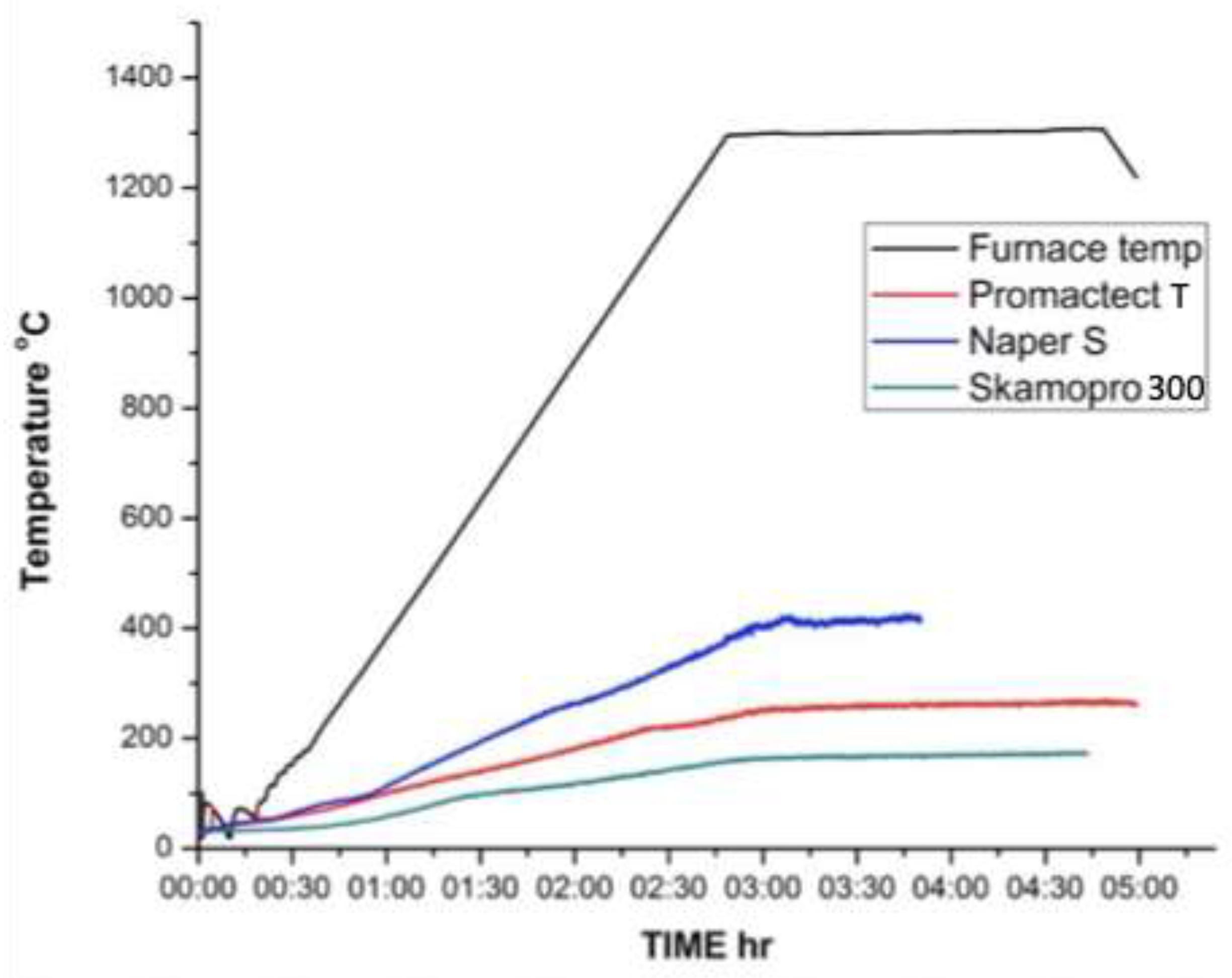

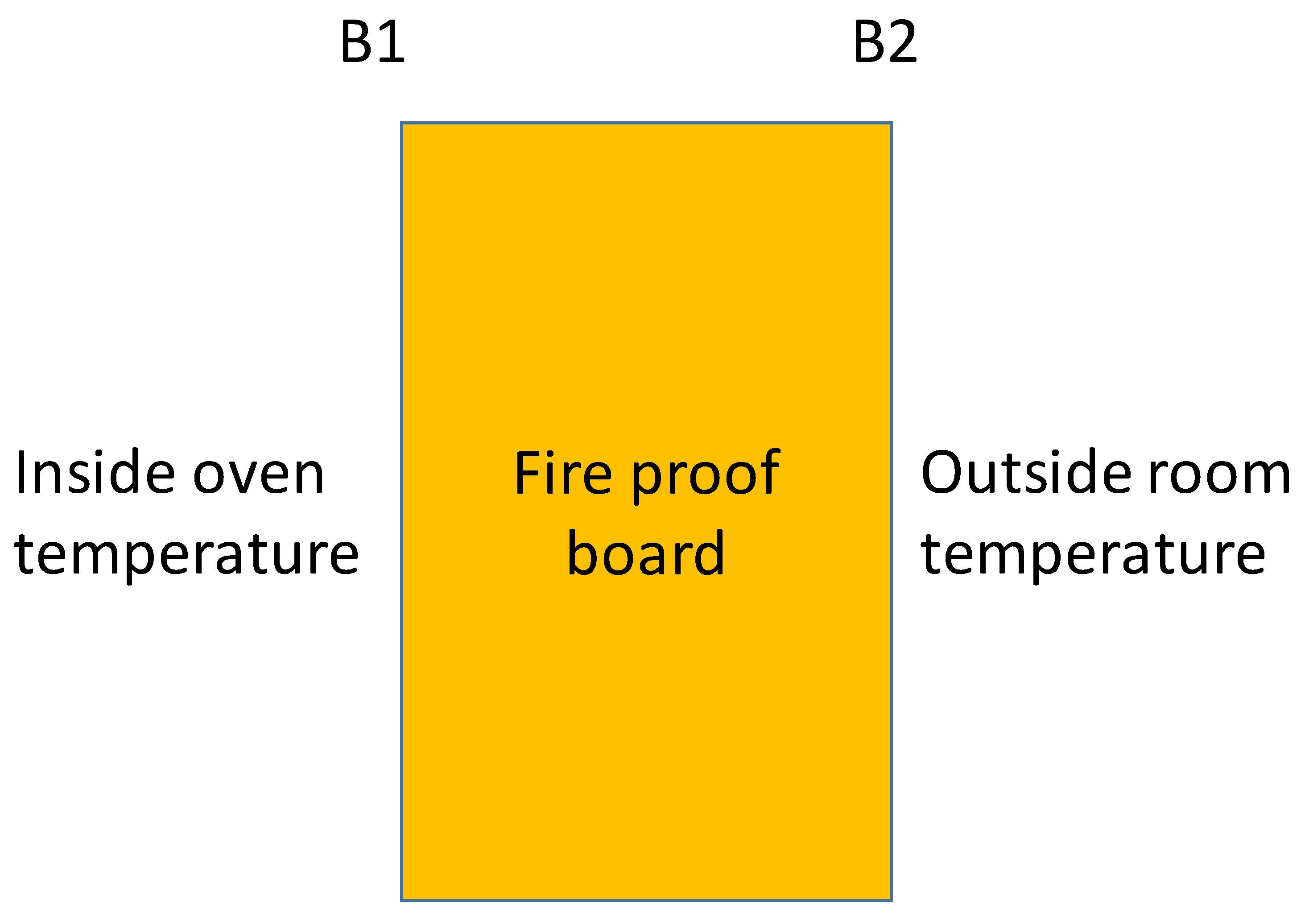
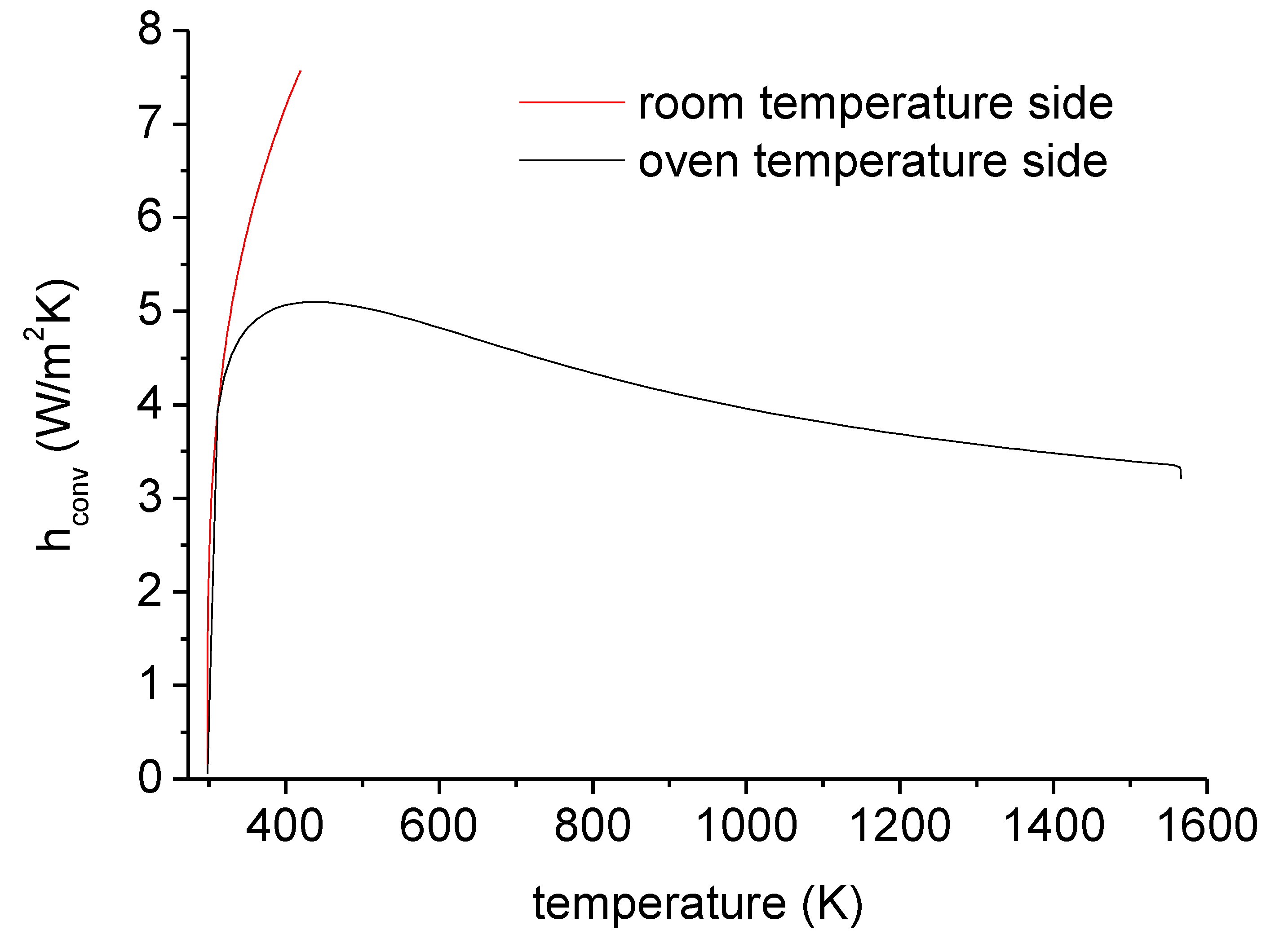
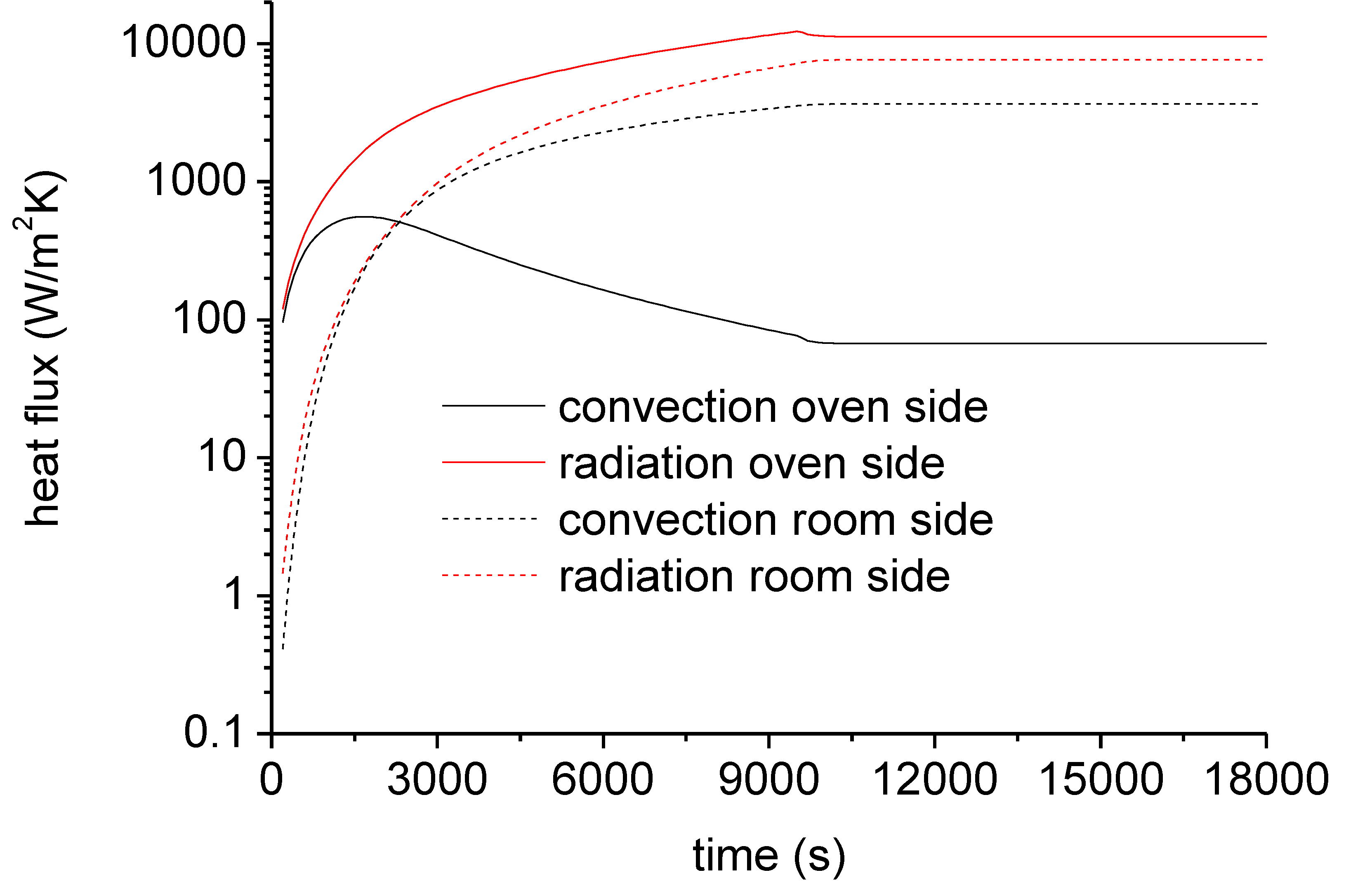
| Products Name | Composition |
Board Thickness (mm) |
Thermal Conductibvity (W/mK) |
Density (Kg/m3) |
Thermal Conductibvity (W/mK) |
Fire Performance Classification |
|
Promatect-T Promat International, Brussels, Belgium |
Calcium Silicate- aluminate | 25 | 0.21 at 20 °C | 900 | 0.21 at 20 °C | Class 1 (BS 476: part 7, EN 13051-1:A1) |
|
Naper S Global Building, Treviso, Italy |
Cement matrix based calcium silicate board | 20 | 0.25 at 20 °C | 1200 | 0.25 at 20 °C | A1 (EN 12467) |
|
Skamopro-300 Skamol- Østergade, Denmark |
Calcium silicate board | 50 | 0.072 at 10 °C | 300 | 0.072 at 10 °C | A1 (EN 13501-1:2007 + A1:2009) |
Sample
|
Promatect-T | Naper-S | Skamopro-300 | |||
wt%
|
As received | 1200 °C | As received | 1200 °C | As received | 1200 °C |
| SiO2 | 27.04 | 28.91 | 49.21 | 52.78 | 45.40 | 46.74 |
| CaO | 39.86 | 35.04 | 41.96 | 38.51 | 52.27 | 51.00 |
| Al2O3 | 30.63 | 34.01 | 4.39 | 4.43 | 1.03 | 1.21 |
| Fe2O3 | 0.94 | 0.72 | 2.35 | 2.41 | 0.28 | 0.22 |
| Cr2O3 | 0.01 | 0.02 | 0.05 | 0.06 | 0.06 | 0.07 |
| K2O | 0.32 | 0.15 | 0.78 | 0.92 | 0.42 | 0.16 |
| TiO2 | 0.09 | 0.07 | 0.16 | 0.17 | 0.01 | 0.01 |
| MgO | 0.65 | 0.72 | 0.63 | 0.72 | 0.52 | 0.58 |
| SO3 | 0.25 | 0.12 | 0.30 | -- | -- | -- |
| P2O5 | 0.10 | 0.17 | 0.14 | -- | -- | -- |
| Promatect-T | Naper S | Skamopro-300 | |
| keff (W/mK) | 0.21 | 0.25 | 0.072 |
| ρ(Kg/m3) | 900 | 1200 | 300 |
| cp (J/Kgk) | 1050 | 850 | 1050 |
| Thickness (m) | 25*10-3 | 20*10-3 | 50*10-3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




