1. Introduction
Nutrition plays a fundamental role in the development and maintenance of brain function, with profound implications for cognitive well-being across the lifespan [
1,
2]. An adequate intake of nutrients is essential for optimal brain performance, as it influences neurotransmitter synthesis, neuronal activity, and the structural integrity of cell membranes [
2]. Within this context, minerals stand out for their critical role in neurological health. Essential minerals such as iron (Fe), zinc (Zn), magnesium (Mg), copper (Cu), and selenium (Se) serve as cofactors in numerous biochemical processes vital to brain function. For instance, iron is indispensable for normal neurocognitive development and myelin production [
3]; zinc is associated with attention, learning, and memory [
4]; and magnesium is crucial for nervous system function and processes such as memory and attention, partly because magnesium deficiency has been linked to chronic low-grade brain inflammation [
5].
Several studies have documented associations between specific mineral levels and cognitive performance at different stages of the life course. For example, lower magnesium intake has been associated with an increased risk of cognitive decline in older adults, due to its role in oxidative stress and neuroinflammation [
5,
6]. In this regard, data from a large cohort of 2,508 adults in the NHANES study revealed that individuals with high daily magnesium intake exhibited better global cognitive test scores, particularly among women and individuals with adequate vitamin D levels [
7]. Similarly, among older adults, adequate copper intake (around 1.3 mg/day) has been associated with reduced cognitive decline over time, suggesting a potential neuroprotective role for this mineral [
8]. These findings highlight the brain's sensitivity to variations in mineral availability and support the notion that even subtle imbalances can have significant consequences for cognitive function.
Historically, much of the research in nutritional science has focused on the detrimental effects of mineral deficiencies, consistently documenting their negative impact on brain development, neuronal plasticity, and cognitive performance [
9,
10]. This perspective has informed the design of nutritional interventions and public health policies aimed at preventing deficiency states, especially during critical periods such as childhood and old age. However, the consequences of nutritional excess, particularly the accumulation of certain minerals, have received considerably less attention [
10]. Emerging evidence indicates that elevated concentrations of minerals such as iron, copper, or manganese may foster a pro-oxidative environment, triggering oxidative stress and neuroinflammation, as well as disruptions in cellular signaling pathways [
11,
12]. These processes can alter neuronal homeostasis and potentially compromise the structure and function of the nervous system, with deleterious effects on cognition.
This gap in the literature is particularly relevant in light of the globalization of dietary patterns, the widespread use of fortified foods, and increasing reliance on dietary supplements, all of which contribute to more frequent exposure to supraphysiological levels of certain minerals. Therefore, the primary objective of this article is to explore the influence of mineral intake on cognitive function.
2. Materials and Methods
This cross-sectional study was conducted with a sample of 239 Spanish adults, within the framework of the research project Personalized Obesity Treatment by Learning and Analysing a Multimodal Space of the Disease with an Integral Approach (Tech4Diet-Person). The overarching aim of Tech4Diet-Person is to improve obesity treatment through personalized interventions based on deep multimodal learning and continuous patient monitoring, by developing digital tools that support tailored nutrition and motivation for long-term lifestyle changes. This project represents the continuation of a research line initiated in two previous studies: 4D Modelling and Visualization of the Human Body for the Improvement of Adherence to Dietary-Nutritional Treatment of Obesity through Low-Cost Technologies (TIN2017-89069-R, Tech4Diet, 2017) and Predictive Models of the Morphological Evolution of the Human Body to Improve Adherence (PID2020-119144RB-100, Tech4Diet-Predict, 2020).
The study protocol was reviewed and approved by the Ethical Committee of the University of Alicante (File Nos. UA-2016-06-30 and UA-2021-11-18). Participants were recruited following strict ethical standards and current data protection regulations, ensuring respect for privacy, confidentiality, and the voluntary nature of participation. All individuals were fully informed about the study’s objectives, procedures, and conditions, and were reminded of their right to withdraw at any time without consequence. Written informed consent was obtained from all participants before enrolment. The signed consent forms were securely stored by the research team in accordance with institutional and ethical guidelines. No minors were included in the study. All recruitment procedures and assessments were carried out at the Faculty of Health Sciences of the University of Alicante between January 10, 2022, and February 20, 2024. Data collection included a semi-structured interview for sociodemographic information, dietary-nutritional questionnaires, and neuropsychological tests to assess current nutritional status and cognitive function. The sample size was estimated a priori using G*Power 3.1. A power analysis for a non-parametric comparison between two independent groups, assuming a medium effect size (d = 0.25), an alpha level of 0.05, and a power of 0.90, indicated that a minimum of 130 individuals.
Inclusion criteria were being an adult between 18 and 65 years of age and having Spanish as a native language. Exclusion criteria included: (i) the presence of a professionally monitored endocrine-metabolic disorder; (ii) a history of neurological disease or acquired brain injury (e.g., stroke, traumatic brain injury, or neurodegenerative conditions such as Parkinson’s disease); (iii) a history of severe psychopathology according to DSM-5 criteria; (iv) current psychiatric treatment; and (v) pregnancy and/or lactation in women.
Sociodemographic and Lifestyle Variables
Sociodemographic variables were collected through a specific semi-structured interview, which included questions regarding the following aspects: sex (female, male), age (years), body mass index (BMI), marital status (single, married, divorced, widowed), educational level (primary, secondary, university), and employment status (employed, unemployed, retired, homemaker).
Cognitive Assessment
The General Cognitive Assessment Battery (CAB) from CogniFit was used, a scientifically validated computerized neuropsychological tool that evaluates multiple cognitive domains through standardized tasks [
13]. The CogniFit battery has been widely applied in both clinical and research contexts, as its component tests have been validated against various standardized neuropsychological assessments [
14]. Moreover, numerous studies have employed CogniFit’s cognitive activities with healthy populations (including children, adults, and older adults) with the aim of assessing or enhancing cognitive functioning. These studies are characterized by high methodological rigor, granting CogniFit a robust level of empirical evidence in the field of cognitive interventions [
15,
16]
The CAB is administered individually via a digital device (computer, mobile phone, or tablet) and takes approximately 40 minutes to complete. The system generates a comprehensive report consisting of three sections: well-being indicators (physical, psychological, and social well-being), cognitive profile, and conclusions (including a description of different indices, specific recommendations, and a personalized action plan).
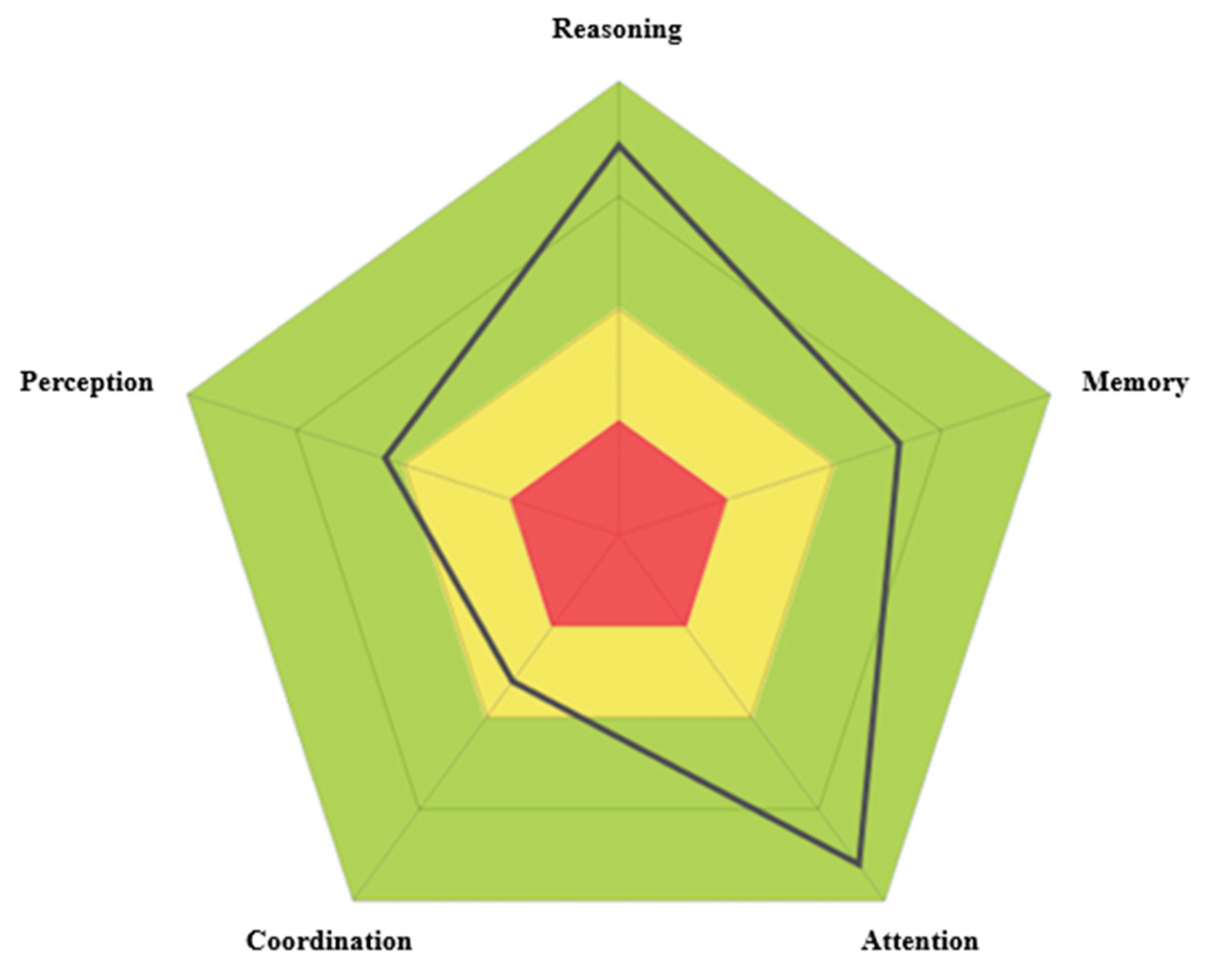
The cognitive profile is presented in a circular diagram for each evaluated domain (
Figure 1), providing normalized quantitative scores based on a large population database, adjusted for the participant's age and sex [
14]. Although CogniFit scores are calculated in percentiles, they are presented on a unified scale ranging from 0 to 800. In this scale, higher scores reflect better cognitive performance. Scores between 0 and 200 (red zone) indicate performance significantly below the expected average, interpreted as a cognitive weakness in the assessed domain. Scores between 200 and 400 (yellow zone) reflect performance below the average, suggesting optimizable cognitive abilities. Scores between 400 and 600 (first green zone) correspond to adequate cognitive performance, indicating that the assessed ability is within the normative range. Finally, scores above 600 (second green zone) are considered cognitive strengths, as they reflect above-average functioning.
The total cognitive profile score was calculated as the mean of five core cognitive domains: reasoning, memory, attention, coordination, and perception. Each of these domains is composed of the average performance across several basic cognitive functions. For the purposes of this study, only selected functions were analyzed, as defined in the report generated by the CogniFit platform:
- Reasoning: Defined as the ability to efficiently process acquired information. It evaluates executive function and is composed of the following sub-functions:
- Planning: The ability to mentally anticipate the correct way to execute a task.
- Processing speed: The amount of time it takes an individual to complete a mental task.
- Cognitive flexibility: The brain’s capacity to adapt behavior and thinking to novel, changing, or unexpected situations.
Working memory: This function refers to the ability to temporarily store and manipulate the necessary information for performing complex cognitive tasks. Although the CogniFit platform categorizes it under the domain of memory, the scientific literature widely recognizes it as an executive function due to its anatomical localization in the prefrontal cortex [
17] and its involvement in cognitive control and decision-making processes [
18]. For this reason, and to encompass a comprehensive analysis of executive functions, working memory was included in this study as part of the executive function category.
Dietary Intake Assessment
To evaluate dietary intake, a semi-quantitative Food Frequency Questionnaire (FFQ) comprising 93 items was used. This instrument was specifically developed for the Nutrition and Health Survey of the Valencian Community (ENCV) and has been validated and employed in several epidemiological studies involving Spanish adult populations [
19].
The questionnaire gathered detailed information on the frequency and usual quantity of consumption of various food groups, including bread, fats, meats, dairy products, and beverages. Consumption frequency was recorded based on the participants' intake over the year preceding the assessment. Response options ranged from "once a day" to "more than six times a day" for daily intake; "once a week", "2–4 times per week", or "5–6 times per week" for weekly intake; and "1–3 times per month" or "occasional/none" for monthly intake. For seasonal foods, such as fruits and vegetables, the questionnaire specified the frequency during their natural season.
Statistical Analysis
Frequencies and percentages were calculated for categorical variables, both overall and stratified by groups. Categorical variables were compared across groups using the Chi-square test (Χ²), and effect sizes were assessed using Cramér’s V (with > 0.1 considered small, > 0.3 moderate, and > 0.5 large) [
20]. Quantitative variables were described using means and standard deviations (SD), both globally and disaggregated by groups. Group differences were analysed using the non-parametric Mann–Whitney U test, and effect sizes were assessed using Rosenthal’s r (with > 0.1 considered small, > 0.3 moderate, and > 0.5 large) [
21].
To quantify dietary intake, the frequencies reported in the FFQ were converted into daily frequencies. This conversion involved assigning an average numerical value to each response category: for example, "never or rarely" was translated to 0.0; "1–3 times per month" to 0.07 (2/30); "once a week" to 0.14 (1/7); "2–4 times per week" to 0.42 (3/7); "5–6 times per week" to 0.78 (5.5/7); and "once a day" to 1.0 (1/1). These values, representing daily consumption frequency, were multiplied by the corresponding portion size (in grams) for each food item to calculate the estimated daily intake of each food.
Once the daily intake of each food was calculated, the energy and nutrient content per portion was estimated using the Spanish Food Composition Database (Red BEDCA) [
22], considering only the edible portion. Subsequently, total individual intake of energy and nutrients was obtained by summing the values for all food items.
To compare intake between groups, z-scores were calculated for each cognitive function, interpreted according to standard normal distribution tables. The segmentation criterion was defined as a z-score below -1.5, indicating low cognitive performance [
23]. Two groups were thus established for each cognitive function analysed: global cognitive function, reasoning, cognitive flexibility, processing speed, planning, and working memory. For the variable "global cognitive function," the low performance group included individuals with a z-score below -1.5 in at least one of the five core cognitive domains assessed by CogniFit: memory, attention, reasoning, coordination, and/or perception. Following group segmentation, the Mann–Whitney U test was applied to compare differences in mineral intake between groups. The significance level was set at α = 0.05, and effect size was calculated using the r statistic, where 0.10 indicates a small effect, 0.30 medium, and 0.50 large [
20].
To explore the relationship between sociodemographic variables and iron intake in relation to cognitive performance, a binary logistic regression analysis was conducted. Cognitive performance was used as the dependent variable (coded as 0 = normal, 1 = low). The independent variables included in the model were iron intake (Fe), marital status, employment status, educational level, and age. The enter method was used to simultaneously evaluate the combined effect of all predictors on the likelihood of low cognitive performance. The model reported B coefficients, standard errors, Wald statistics, degrees of freedom, p-values, and odds ratios (Exp(B)) for each predictor.
All statistical analyses were performed using IBM SPSS Statistics for Windows, version 21.0 (IBM Corp., Armonk, NY; 2012). Values of p < 0.05 were considered statistically significant.
3. Results
The sample consisted of 239 Spanish adults, with a mean age of 45.85 years (SD = 10.136), including 91 men (37.2%) and 152 women (62.8%). Most participants were married or in a stable partnership (64.0 %), had higher education degrees (51.5 %), and were employed (83.7%).
As shown in
Table 1, out of the total participants, 200 exhibited normal cognitive performance, while 39 showed low performance (defined as a z-score below -1.5 in at least one of the five cognitive domains assessed by CogniFit: memory, attention, reasoning, coordination, and/or perception). The low cognitive performance group had a significantly higher mean age than the normal performance group (M = 50.67, SD = 10.27 vs. M = 44.91, SD = 9.86; p < 0.001), with a moderate effect size (r = 0.279). Likewise, the low cognitive performance group had a significantly higher mean BMI than the normal performance group (M = 34.89, SD = 6.85 vs. M = 31.20, SD = 6.23; p = 0.002), with a moderate effect size (r = 0.28). No significant differences were found in sex distribution (p = 0.202).
Regarding marital status, significant differences were observed between groups (p = 0.039), with a lower proportion of single individuals and a higher proportion of divorced individuals in the low performance group; the effect size was small (V = 0.188). Significant differences were also found in educational level (p < 0.001), with the low performance group having a higher percentage of participants with primary education and fewer with higher education; the effect size was moderate (V = 0.337). Additionally, significant differences in employment status were identified (p = 0.005), with a higher proportion of retired and unemployed individuals in the low performance group; the effect size was moderate (V = 0.208).
Table 2A and
Table 2B present the differences in mineral intake according to cognitive performance across various domains. Specifically, iron intake was significantly higher in the low performance group compared to the normal group for global cognitive function (23.07 ± 7.41 mg vs. 20.38 ± 5.84 mg; p = 0.037; r = 0.135), reasoning (23.38 ± 5.79 mg vs. 20.65 ± 6.19 mg; p = 0.037; r = 0.135), and cognitive flexibility (22.98 ± 6.28 mg vs. 20.58 ± 6.15 mg; p = 0.037; r = 0.135). This pattern was partially replicated in the domain of working memory, where the difference did not reach statistical significance (p = 0.072) but followed a similar trend (24.14 ± 8.68 mg vs. 20.52 ± 5.85 mg).
Additionally, a significant difference was observed in zinc intake related to working memory, with the low performance group reporting significantly higher intake compared to the normal group (14.75 ± 5.00 mg vs. 12.52 ± 3.39 mg; p = 0.026; r = 0.144). Despite achieving statistical significance, all effect sizes were small (r < 0.20).
|
Table 2B. Daily mineral intake (mg/day) by cognitive performance in processing speed, planning, and working memory |
| Mineral |
Processing speed |
|
Planning |
|
Working memory |
|
| |
Normal (n=226) |
Low (n=13) |
p / r |
Normal (n=215) |
Low (n=24) |
p / r |
Normal (n=219) |
Low (n=20) |
p / r |
| Calcium |
1130.47 (405.56) |
1239.00 (782.53) |
0.779 |
1145.93 (441.99) |
1058.09 (341.04) |
0.333 |
1128.94 (408.29) |
1217.78 (649.56) |
0.978 |
| Iron |
20.72 (6.03) |
22.55 (8.63) |
0.353 |
20.75 (6.08) |
21.40 (7.10) |
0.652 |
20.52 (5.85) |
24.14 (8.68) |
0.072 |
| Potassium |
3951.01 (1079.04) |
4137.25 (1468.80) |
0.763 |
3964.97 (1111.28) |
3929.72 (1028.70) |
0.885 |
3933.81 (1079.47) |
4260.34 (1304.03) |
0.395 |
| Magnesium |
427.35 (112.01) |
476.06 (161.15) |
0.190 |
429.83 (115.90) |
431.36 (112.46) |
0.768 |
427.32 (112.57) |
459.31 (141.89) |
0.314 |
| Sodium |
9245.30 (4267.92) |
10209.25 (3442.14) |
0.400 |
9222.91 (4246.72) |
9910.70 (4087.52) |
0.417 |
9305.44 (4254.43) |
9213.36 (4013.20) |
0.903 |
| Phosphorus |
1917.80 (531.29) |
2005.22 (806.02) |
0.980 |
1927.81 (536.72) |
1879.52 (641.08) |
0.390 |
1902.67 (515.20) |
2140.30 (811.10) |
0.259 |
| Iodine |
265.54 (85.60) |
274.06 (74.12) |
0.674 |
264.54 (86.45) |
277.99 (71.19) |
0.235 |
266.32 (85.54) |
262.49 (79.48) |
0.808 |
| Selenium |
127.45 (46.75) |
127.38 (52.17) |
0.987 |
128.02 (44.61) |
122.73 (63.80) |
0.185 |
125.97 (43.26) |
143.60 (76.30) |
0.627 |
| Zinc |
12.64 (3.47) |
13.96 (5.32) |
0.400 |
12.72 (3.58) |
12.60 (3.80) |
0.851 |
12.52 (3.39) |
14.75 (5.00) |
0.026 / 0.144 |
Table 3 displays the impact of sociodemographic variables and iron intake on predicting low cognitive performance through binary logistic regression analysis. Although some sociodemographic variables, such as education level, showed significant effects (χ² = 10.147; df = 2; p = 0.006), iron intake remained significantly associated with low cognitive performance even after adjusting for the other variables. Specifically, higher iron intake was associated with an increased likelihood of low cognitive performance (B = 0.128; p = 0.011; odds ratio = 1.13), even when simultaneously considering BMI, daily calorie intake (kcal), marital status, employment status, and educational level. Other variables such as BMI (p=0.074), kcal (p=0.222), age (p = 0.151), marital status (p = 0.764), and employment status (p = 0.419) were not significant predictors in the model.
4. Discussion
The aim of this study was to explore the influence of mineral intake on cognitive function. The findings revealed a significant association between higher intake of iron and zinc and low cognitive performance in a sample of Spanish adults, even after adjusting for sociodemographic variables such as age, education level, marital status, and employment status. This is particularly noteworthy as it challenges the common notion that only mineral deficiencies are detrimental to cognitive function, suggesting instead a potential nonlinear and dose-dependent relationship.
Evidence from the present study indicates that individuals with low cognitive performance had on average, a higher intake of iron—both in global cognition and in specific domains such as reasoning and cognitive flexibility. This pattern may play a role in cognitive performance and aligns with previous research documenting how iron overload can contribute to cognitive decline [
24]. Several neurophysiological mechanisms may explain this relationship, with oxidative stress being one of the most prominent. Excess iron acts as a catalyst in the Fenton reaction, promoting the formation of highly reactive hydroxyl radicals. These radicals cause oxidative damage to essential cellular components such as lipids, proteins, and DNA, particularly affecting vulnerable brain regions like the hippocampus and prefrontal cortex, which are key for higher-order functions such as memory and reasoning [
25]. Other implicated mechanisms include neuroinflammation—since iron activates microglia and fosters a pro-inflammatory environment via the overexpression of cytokines such as IL-6 and TNF-α [
26]; disruption of synaptogenesis and plasticity—given that iron excess disturbs calcium homeostasis and neurotransmitter release (e.g., glutamate and dopamine), thereby impairing synaptic plasticity crucial for learning and memory [
27] and structural brain degeneration—as iron accumulation has been associated with grey matter atrophy and altered functional connectivity [
28].
Moreover, excessive brain iron accumulation has been widely reported in several neurodegenerative diseases. In conditions such as Alzheimer's and Parkinson’s disease, elevated iron levels have been detected in specific brain regions, supporting its potential neurotoxic role [
29]. In Alzheimer's disease, iron contributes to oxidative damage to the tau protein, promoting its hyperphosphorylation via the activation of related kinases. In this way, iron not only directly affects tau but also enhances its pathological modification through enzymatic phosphorylation pathways [
30]. In Parkinson's disease, iron binds directly to the presynaptic protein alpha-synuclein, which is involved in neurotransmitter release regulation. This interaction promotes the aggregation of alpha-synuclein into insoluble fibrillar forms, increasing its expression and reducing its degradation. Consequently, numerous studies have reported marked iron accumulation in the substantia nigra of patients with Parkinson’s disease—a region critical for motor control and highly susceptible to oxidative stress [
31].
A significant association was also found between zinc intake and working memory performance. Although the effect size was small, this finding aligns with existing literature recognizing zinc as a neuroactive nutrient with dual effects. Zinc plays a role in glutamatergic neurotransmission, modulating NMDA and AMPA receptors and supporting synaptic plasticity in key regions such as the hippocampus and prefrontal cortex [
32]. However, zinc can also be neurotoxic when accumulated excessively. High extracellular concentrations disrupt calcium homeostasis, inhibit NMDA channels, and lead to mitochondrial dysfunction, resulting in oxidative stress and neuronal apoptosis [
33]. This neurotoxic imbalance can impair synaptic dynamics essential for maintaining complex cognitive functions such as attention and working memory [
34].
Despite the relevance of these findings, several methodological and analytical limitations should be considered when interpreting the results. First, the cross-sectional design precludes causal inferences regarding the relationship between mineral intake and cognitive performance. Second, dietary intake was assessed via a self-reported food frequency questionnaire (FFQ), which may be subject to recall bias or social desirability bias. Lastly, although the sample size was adequate for regression and group comparison analyses, the subsamples with low cognitive performance were considerably smaller, potentially affecting the stability of estimates and limiting the generalizability of the results. Future studies should address these limitations by employing longitudinal designs, using biomedical assessments of micronutrient levels, and comparing more homogeneous cognitive performance groups.