1. Introduction
Infantile hemangiomas (IH) are the most common lesions of in infants [
1]. They occur mostly in premature babies with the body weight of under 1800gramms [
2], more often in Caucasian girls.
These benign vascular tumors undergo a phase of rapid proliferation, which may continue up to 8-12 months, then a slow spontaneous involution may begin, lasting 4-7 years, and therefore most patients need no any treatment [
1,
3]. However, the diffuse hemangiomatosis of internal organs can probably lead to developing severe life-threatening conditions (LTC). Multiple skin hemangiomas in diffuse neonatal hemangiomatosis can combine with lesions in the internal organs, primarily the liver [
4]. Diffuse hemangiomas can lead to the abdominal compartment syndrome (ACS) that develops along with the increased liver; as well as the heart failure resulting from the increasing venous return and increased preload on the right heart; and the respiratory failure [
5]. Severe respiratory failure and respiratory distress syndrome may occur in case of IH localization in the upper respiratory tract due to the compression of the organs [
6,
7].
Severe acquired thyroid dysfunction is also possible as a result of the overexpression of type 3 deiodinase (an enzyme that destroys thyroxine), which leads to increased deiodination of thyroxine and triiodothyronine to biologically inactive T3 and diiodothyronine T2. Hypothyroidism is always observed in diffuse forms of hemangioma, while it is rarely noted in multifocal variants of hemangioma [
2,
8]. Timely diagnosis of IH and initiation of the appropriate therapy are ultimately important for saving a child's life. The first-line therapy for the diffuse lesions includes the non-selective β-adrenoblocker Propranolol that has shown its effectiveness in the IH treatment [
7,
9].
We present a clinical case of observation and treatment of a patient with a complicated course of IH, which required differential diagnosis with malignant tumors and immediate initiation of the specific treatment and the concomitant therapy to stop the LTC. The consent of legal representatives for the publication was obtained.
2. Case Presentation
In June 2024 a girl M. aged 1 month with a bulky formation in the abdominal cavity was admitted to the L.A. Durnov Research Institute of Pediatric Oncology and Hematology of the “N.N.Blokhin National Research Medical Center of oncology” the Ministry of Health of Russia (hereinafter referred to as the RI PO&H). The girl was born in the natural childbirth after an uncomplicated pregnancy. On the whole, intrauterine development had no complications and ultrasound examinations revealed no structural or vascular abnormalities.
The child's parents noted an increase in the size of the belly from the 15th day of life. Ultrasound (US) examination of the abdominal organs performed in the local clinic revealed total liver damage by multiple hypoechoic round-shaped formations up to 1.8 cm in diameter and confirmed by the computed tomography (CT).
After admission to the RI PO&H the child was hospitalized in the intensive care unit (ICU) taking into account the severe condition probably caused by ACS (the belly was significantly enlarged as a result of the severe hepatomegalia; abdominal circumference at admission was 47.5 cm, the liver protruded 5 cm from the edge of the costal arch). Physical examination revealed the hemangiomas of the facial skin at the brow arch on the left, acrocyanosis, tachypnea (respiratory rate of over 60/min), pronounced venous network on the anterior abdominal wall, and significantly enlarged abdomen, which was dense and tense at palpation.
Laboratory tests showed a high level of α-fetoprotein (AFP) (10564.1 IU/ml), increased levels of neuron-specific enolase (NSE) (49.93 ng/ml) and β-chorionic gonadotropin (β-HCG) (0.1 mIU/ml), and thrombocytopenia (48×109/ L). According to the coagulogram, hypofibrinogenemia was detected up to 0.4 g / L and hypocoagulation in all chains. US examination of the abdominal organs and retroperitoneal space revealed hepatomegalia (oblique vertical dimension of 143 mm), diffusely heterogeneous parenchyma with multiple areas of reduced echogenicity without clear contours with the diameters of 10-24 mm. The US picture corresponds to hemangiomatosis of the liver.
The assessment of the hormonal profile showed an increased level of the thyroid-stimulating hormone (TSH) of 26.41 mIU/ L, the level of thyroxine (svT4) of 26.1 pmol/ L, and triiodothyronine (svT3) of 4.8 pmol/L. US examination of the thyroid showed no any abnormal signs. Decreased function of the thyroid gland was most likely associated with the main disease. Replacement hormone therapy with levothyroxine at a dose of 2 µg / kg / day was initiated on the base of these findings.
CT data of the abdominal organs and retroperitoneal space with intravenous contrast showed that liver parenchyma was totally altered as a result of multiple foci up to 4 cm in diameter with indistinct uneven contours; contrasting determined the centripetal nature of accumulation, which was typical for hemangiomatosis.
EchoCT data detected a congenital heart defect: a secondary defect of the atrial septal of 7-8 mm with a left-right discharge, a slight dilation of the left atrium (z-score +2.34), and pulmonary hypertension (systolic pressure in the pulmonary artery of 40-43 mmHg). Left ventricular (LV) contractility was satisfactory: LV ejection fraction according to Simpson of 72%. The ECG results showed a sinus rhythm and the normal position of the electrical cardiac axis. The analysis of the acid-base status at the admission showed mixed acidosis (pH=7.119), hyperlactatemia (3.5 mmol/L), and electrolyte disturbances.
The electro-cardio-monitor recording in 12 hours after admission to the ICU revealed a condition with negative dynamics such as depressed consciousness, increasing respiratory failure, the sinus rhythm with a tendency to bradycardia up to 70 beats/min. The treatment included administration at a slow rate of atropine at a dose of 0.01 mg/kg, furosemide ─ 1 mg / kg, and dexamethasone ─ 0.2 mg /kg. Given the increase in respiratory failure, the clinical picture of pulmonary edema, the depressed consciousness, and the development of life-threatening bradycardia, the decision was taken to start artificial ventilation of the patient, which lead to the stabilization of the child's condition and normalization of the hemodynamic parameters. Analgesic therapy was added including midazolam 1 µg/ kg/min and morphine 0.02 mg/kg/h. Taking into account the patient's type, the stratification of the infection risk caused by polyresistant pathogens and the risk of invasive candidiasis, antibacterial prophylaxis was initiated with a protected beta-lactam antibiotic (cefepime/sulbactam 80 mg/kg/day for sulbactam) and antifungal therapy ─ with fluconazole (12 mg/kg loading dose, then 8 mg/kg).
A clinical and radiological diagnosis was established according to the clinical picture and the comprehensive examination data: diffuse neonatal hemangiomatosis of the liver with skin lesions; congenital heart disease: a secondary defect of the atrial septum; pulmonary hypertension; circulatory insufficiency I; functional class II according to ROSS Classification; respiratory failure grade 1. Surgical intervention for the diagnostic morphological verification was impossible considering the localization, size, and the severity of the process, as well as the high risks of intra- and postoperative complications including fatal outcome. Taking into account the benign nature of the lesion, the child was given the therapy with a non-selective β-adrenoblocker Propranolol in the dosage form of a solution for an oral intake Hemangiol® to treat total liver damage with multiple hemangiomas.
Generally, the recommended starting dose is 1 mg/ kg/ day, divided into 2 doses of 0.5 mg/kg, with a gradual increase by 1 mg/ kg per week up to the therapeutic dose of 3 mg/kg [
10]. In our case, due to the serious patient’s condition, the medical consultation made a prescription for the starting drug dose of 0.5 mg / kg / day orally (in 3 doses), with a gradual daily increase by 0.5 mg / kg / day every 3 days until the maximum therapeutic dose of 3 mg / kg / day was reached. The daily ECG monitoring registered no alterations in the rhythm and conduction.
Propranolol therapy was performed along with the hormone therapy (L-thyroxine), infusion therapy, prophylactic measures against hemorrhagic complications, hepatoprotective therapy, diuretic therapy, and replacement transfusions of erythrocyte suspension, freshly frozen plasma, cryoprecipitate, 20% albumin solution and single i.v. human immunoglobulin. The replacement transfusions were needed within the first 7 days, and then the laboratory parameters returned to normal. From day 3 after normalization of the lactate levels, enteral loading was performed through a nasogastric tube with an adapted milk mixture starting from 5 ml/ hour with an extended infusion and then expanding to the physiological norm for 7 days. From day 7, the child received auxiliary ventilation; the dose of analgesic therapy was gradually decreased.
On day 10 of the therapy, a marked positive trend was registered according to the cessation of ACS (abdominal circumference: 42 cm, liver: +3 cm from the edge of the costal arch) and effective independent breathing (the child was extubated). On day 14, the child was transferred to a specialized department in the stable condition to continue Propranolol therapy and accompanying therapy.
Upon transfer to the specialized department, the child had clear consciousness and no fever. Her breathing was spontaneous and effective, without dyspnea. Blood gas results were normal and oxygenation while breathing ambient air was stable. The child’s hemodynamics was also stable. She was ingesting enteral loading through an AMS Nutrilon 1 nasogastric tube at a rate of 30 ml/hour with a 4-hour interval at night. The child was urinating while receiving spironolactone at a dose of 2 mg/kg/day. Her stools were unaided, mushy, and regular, with no abnormalities. Her weight upon transfer was 4.3 kg; and on admission, 4.5 kg.
The table below presents the patient's laboratory test results while staying in the ICU. The patient's severe thrombocytopenia resolved with the treatment, and a significant decrease in ALT liver enzyme levels was registered (
Table 1).
A marked positive trend was observed when the maximum therapeutic dose of Propranolol 3 mg/kg/day was reached; the results demonstrated a reduction in the size of facial skin hemangiomas from 1.5 cm in diameter to 0.8 cm, cessation of ACS (abdominal circumference was 38 cm), normalization of platelet levels in accordance with the age norm, and a decrease in TSH, svT3, and svT4 levels. The patient showed satisfactory tolerance of the therapy. No adverse events were recorded during the entire follow-up period.
According to the instrumental examination the dynamics on the 30th day of therapy was as following:
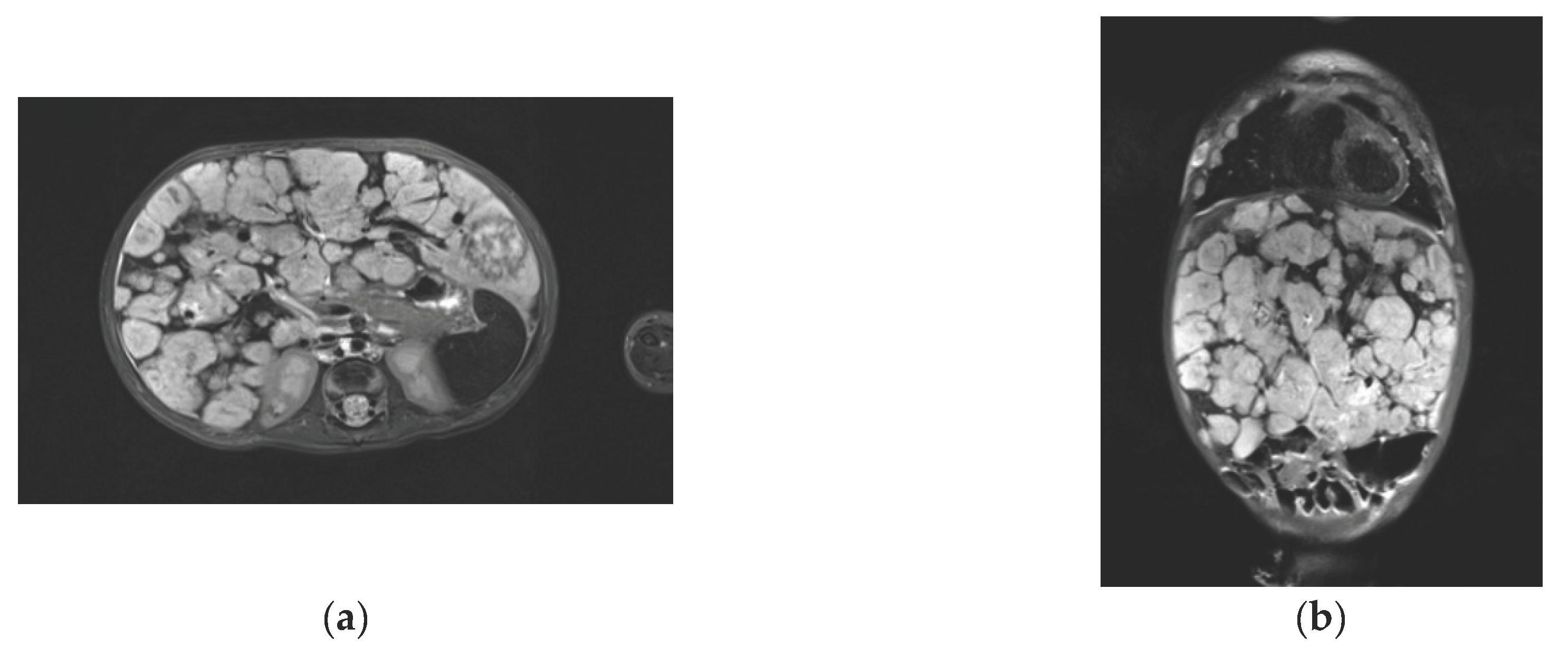
US examination of the liver — OTS (subcostal approach, oblique-transverse scanning along the edge of the costal arch at the level of the midclavicular line) reduction to 105 mm; reduction of the size of multiple focal formations to 8×12 mm; EchoCG — reduction of the size of the atrial septal defect to 5-6 mm; reduction of the heart cavities to normal size; no signs of pulmonary hypertension; MRI of the abdominal organs demonstrated a picture of liver hemangiomatosis with positive dynamics in comparison with the CT picture upon admission. The picture of the liver parenchyma had multiple irregularly shaped bulky formations up to 3.7 cm subtotally replacing the parenchyma. The centripetal type of the accumulation of the contrast by focal formations was revealed with intravenous multiphase contrasting (
Figure 1a,b).
3. Discussion
Hemangiomatosis of internal organs is rare, but in some cases it is a severe diagnosis that causes many life-threatening conditions, and therefore rapid diagnosis and timely start of the therapy are important. In certain cases, an individual approach to titrating dosages and prescribing accompanying therapy is required.
It is important to bear in mind that skin IH can be associated with visceral lesions. The most common visceral organ involved in the pathological process is the liver [
3,
11]. At present classification includes focal, multifocal, and diffuse forms of liver hemangiomas [
12].
Diffuse IH of the liver is always accompanied by a severe clinical course that occurs due to ACS, posing a threat to the patient's life, which requires a multimodal approach to treatment [
4,
13].
In some cases, hemangiomas may be part of PHACE syndrome (posterior fossa abnormalities, hemangioma, and arterial, cardiac, eye, and sternal abnormalities), including posterior cranial fossa malformation, hemangiomas, heart malformations, and eye malformations. This syndrome is associated with preeclampsia or placental abnormalities [
13,
14]. Another syndrome manifested by hemangiomas is lumbar spine syndrome — LUMBAR — (lumbar spine syndrome — multiple hemangiomas of the lower body, developmental abnormalities of the genitourinary system, bone deformities, vascular malformations) [
13,
14,
15,
16]. A number of patients with diffuse hemangiomatosis with liver damage, have persistent thrombocytopenia and hypocoagulation, which is associated with Kazabach—Merritt syndrome [
13,
14,
15,
16]. Secondary hypothyroidism may also occur, requiring hormone replacement therapy, but this complication is resolved along with the tumor involution, and therefore the drug dose is reduced until it is completely discontinued [
17].
The diffuse hemangiomatous lesions are often accompanied by hypothyroidism, probably due to the high levels of type 3 iodothyronine deiodinase activity produced by hemangiomas [
18]. According to Kassarjian et al. [
19] hypothyroidism in patients with infantile hepatic hemangiomas (IHH) is a poor prognostic factor associated with an increased likelihood of complications including sepsis, multiple organ failure, pulmonary hypertension, and intracranial hemorrhage.
These patients are more likely to have a massive tumor volume, leading to compression of neighboring organs and vessels, rather than the more common pattern of high-release congestive heart failure. Obviously, in patients with extensive infantile liver hemangioma, thyroid function should be evaluated. In the present study, the patient with an advanced IHH with no morphological changes in the thyroid gland had signs of hypothyroidism that manifested in a decrease in the level of the thyroid hormones. The patient received thyroxine replacement therapy on the base of these data. Previously, corticosteroids in combination with digoxin and diuretics were used as the first line for IHH treatment [
20]. However, side effects such as stomach irritation, osteoporosis, psychological disorders, and abnormal sedimentation limited the duration of this strategy. In addition, a significant proportion of patients (23.1%) was resistant to steroids [
21]. In the past, interferon α-2 (INFα-2) was used in cases that cannot be treated with steroids, as it inhibited the proliferation and migration of endothelial cells, causing a decrease in the size of intracranial hypervascular formation [
22]. However, INFα could cause serious complications such as spastic diplegia and developmental disorders in children under 3 years of age. INFα is no longer recommended in current guidelines for the treatment of hypervascular formations [
23]. In 2008 Leaute-Labreze et al. [
24] reported the results of a study where they accidentally discovered that Propranolol, a non-selective antagonist of β-1 and β-2 adrenergic receptors, showed an anti-proliferative effect in infantile hemangioma. Later, French researchers designed a drug for the IH treatment in infants on the base of Propranolol in the form of an oral solution, Hemangiol®. Currently, the therapy with this drug is recommended as the therapy of choice for patients with IH who require systemic therapy [
24,
25,
26]. Hemangiol® (international non-patented name (INN) — Propranolol) is the only officially registered drug for the IH treatment in children in Europe and the USA, which is included in European, American and Russian clinical guidelines for the treatment of this disease [
26,
27]. The drug was registered in Russia in 2015 and is also successfully used in clinical practice in children from 5 weeks of life [
1,
2]. The exact mechanisms underlying Propranolol's potential to cause hemangioma involution are unclear. This phenomenon may be associated with vasoconstriction mediated by pericytes, a decrease in the level of endothelial and basic fibroblast growth factor, as well as inactivation of the renin-angiotensin system [
28]. In 2014, the FDA approved Propranolol for the systemic treatment of idiopathic hyperhidrosis. Since then, Propranolol has been considered an effective and safe drug for the treatment of idiopathic hyperhidrosis and has replaced steroids [
29]. The recommended starting dose of Propranolol is 0.6 mg/kg twice daily with a gradual increase over 2 weeks to a maintenance dose of 1.7 mg/kg twice daily. The European Expert Group recommended starting Propranolol therapy at a dose of 1.0 mg/kg daily, and the target dose should be from 2.0 to 3.0 mg/kg daily and divided into two or three drug intakes [
23]. However, the dosage should be adjusted to the patient's condition, especially in patients with posterior cranial fossa malformations, hemangioma, arterial abnormalities, aortic coarctation/heart defects, eye abnormalities, and adverse reactions such as sleep disorders [
8,
30]. Treatment should continue at least 6 months until the child reaches the age of 12 months. During the period of dose reduction or after discontinuation of the drug, 10-25% of patients may experience repeated growth, which may occur even 6 months after the end of the therapy. A large multicenter retrospective cohort study reported that patients who stopped therapy at <12 months of age, especially those under 9 months of age, had the highest risk of regrowth. The lowest risk of recurrence was observed in patients whose treatment was discontinued between the ages of 12 and 15 months [
31]. Despite the fact that a hemangioma is a benign neoplasm, the diffuse form of the disease with massive liver damage leads to mortality rates of about 70% [
3,
11,
25]. Rapidly growing lesions can compress the inferior vena cava, leading to hepatomegalia or bloating, secondary liver failure, difficulty in breathing, and multiple organ failure. These tumors with a high degree of vascularization can form arteriovenous shunts inside the lesions, causing aggressive high blood volume and life-threatening congestive heart failure [
32]. Multiple lesions can also lead to platelet accumulation in tumors, which causes anemia, thrombocytopenia, and coagulopathy [
33]. High mortality is associated with the development of multiple organ failure in patients with ACS, as well as with DIC syndrome (disseminated intravascular coagulation syndrome) in case of coagulopathy with liver damage.
In some cases, it may be difficult to diagnose a pathological lesion despite the fact that US and MRI can be used to accurately make a differential diagnosis; rare cases require a diagnostic biopsy of the lesion. The treatment of patients is primarily aimed at accelerating tumor involution in order to reduce the lesion, stop LTC, and prevent compression of vital organs.
4. Conclusions
Hemangiomas of internal organs in newborns are a rare and potentially life-threatening condition that requires a multidisciplinary approach to diagnosis and treatment. It is necessary to take into account the concomitant diseases and monitor the treatment in collaboration with a cardiologist and other specialists. At the first stages of treatment it may also be necessary to invite an anesthesiologist-resuscitator for examination and participation in the treatment of patients with LTC with impaired functions of organs and systems. Timely detection and adequate treatment of IH significantly improve the prognosis and quality of life of the patient.
Author Contributions
Conceptualization, T.T.V. and A.P.K.; Methodology, T.S.B.; Validation, R.R.T. and N.V. M.; Formal Analysis, R.V.M.; Investigation, L.R.T. and V.A. A.; Resources, E.V.C.; Data Curation, T.V.G. and G.M.M.; Writing – Original Draft Preparation, R.R.T. and M.V.K.; Writing – Review & Editing, V.V.R., I.Z.S. and M.V.K.; Visualization, D.A.K.; Supervision, K.I.K.; Project Administration, S.R.V.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Ethics Committee of N.N. Blokhin National Medical Research Center of Oncology.
Informed Consent Statement
Informed consent was obtained from the legal representatives of the patient involved in the study. Written informed consent has been obtained from the legal representatives of patient to publish this paper.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ACS |
abdominal compartment syndrome |
| AFP |
α-fetoprotein |
| ALT |
alanine aminotransferase |
| CT |
computed tomography |
| DIC |
disseminated intravascular coagulation syndrome |
| ECG |
electrocardiography |
| FDA |
Food and Drug Administration |
| i.v. |
intravenous |
| ICU |
intensive care unit |
| IH |
infantile hemangioma |
| IHH |
infantile hepatic hemangiomas |
| INFα-2 |
interferon α-2 |
| INN |
international non-patented name |
| LTC |
life-threatening conditions |
| LV |
left ventricular |
| MRI |
magnetic resonance imaging |
| NSE |
neuron-specific enolase |
| OTS |
oblique-transverse scanning |
| TSH |
thyroid-stimulating hormone |
| US |
ultrasound |
| β-HCG |
β-chorionic gonadotropin |
References
- Sandru, F.; Turenschi, A.; Constantin, A.T.; et al. Infantile Hemangioma: A Cross-Sectional Observational Study. Life (Basel) 2023, 13, 1868. [Google Scholar] [CrossRef]
- Lopriore, E.; Markhorst, D.G. Diffuse neonatal haemangiomatosis: new views on diagnostic criteria and prognosis. Acta Paediatr. 1999, 88, 93–97. [Google Scholar] [CrossRef]
- Kilcline, C.; Frieden, I.J. Infantile hemangiomas: how common are they? A systematic review of the medical literature. Pediatr Dermatol 2008, 25, 168–173. [Google Scholar] [CrossRef]
- Wen, R.; Zhou, Z.Z.; Chen, W.J. Clinical and Pathological Features of Congenital Hepatic Hemangioma in Children: A Retrospective Analysis. Fetal Pediatr Pathol. 2023, 42, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Dotan, M.; Lorber, A. Congestive heart failure with diffuse neonatal hemangiomatosis-case report and literature review. Acta Paediatr. 2013, 102, 232–238. [Google Scholar] [CrossRef]
- Christison-Lagay, E.R.; Burrows, P.E.; Alomari, A.; et al. Hepatic hemangiomas: subtype classification and development of a clinical practice algorithm and registry. J Pediatr Surg. 2007, 42, 62–67; discussion 67–68. [Google Scholar] [CrossRef]
- Rahbar, R.; Nicollas, R.; Roger, G.; et al. The biology and management of subglottic hemangioma: past, present, future. Laryngoscope 2004, 114, 1880–1891. [Google Scholar] [CrossRef] [PubMed]
- Kulungowski, A.M.; Alomari, A.I.; Chawla, A.; et al. Lessons from a liver hemangioma registry: subtype classification. J Pediatr Surg. 2012, 47, 165–170. [Google Scholar] [CrossRef]
- Kotlukova, N.P.; Konstantinova, N.K.; Trunina, I.I.; et al. Modern strategy and tactics of management of children with infantile hemangiomas. Pediatrician Practice 2020, 4, 4–12, (in Russ.). [Google Scholar]
- Tan, S.T.; Itinteang, T.; Leadbitter, P. Low-dose propranolol for infantile haemangioma. JPlastic, Reconstruct. & Aesthet. Surg. 2011, 64, 292–299. [Google Scholar]
- Munden, A.; Butschek, R.; Tom, W.L.; et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014, 170, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lin, X.; Jin, Y.; et al. Deep infantile hemangiomas and early venous malformations: differential diagnosis by 3D CT angiography. Ann Plast Surg. 2010, 64, 755–758. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.D.; Baselga, E.; McCuaig, C.; et al. Rebound Growth of Infantile Hemangiomas After Propranolol Therapy. Pediatrics 2016, 137, 20151754. [Google Scholar] [CrossRef]
- Ross, K.R. Vascular Birthmarks—Hemangiomas and Malformations. Arch Dis Child. 1989, 64, 1524. [Google Scholar] [CrossRef]
- Boscolo, E.; Bischoff, J. Vasculogenesis in infantile hemangioma. Angiogenesis 2009, 12, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Greenberger, S.; Bischoff, J. Pathogenesis of infantile haemangioma. Br J Dermatol. 2013, 169, 12–19. [Google Scholar] [CrossRef]
- Dubois, J.; Garel, L.; Grignon, A.; et al. Imaging of hemangiomas and vascular malformations in children. Acad Radiol. 1998, 5, 390–400. [Google Scholar] [CrossRef]
- Huang, S.A.; Tu, H.M.; Harney, J.W.; et al. Severe hypothyroidism caused by type 3 iodothyronine deiodinase in infantile hemangiomas. N Engl J Med. 2000, 343, 185–189. [Google Scholar] [CrossRef]
- Kassarjian, A.; Zurakowski, D.; Dubois, J; Paltiel, H.J.; Fishman, S.J.; Burrows, P.E. Infantile hepatic hemangiomas: clinical and imaging findings and their correlation with therapy. AJR Am J Roentgenol. 2004, 182, 785–795. [Google Scholar] [CrossRef]
- Kuroda, T.; Hoshino, K.; Nosaka, S.; Shiota, Y.; Nakazawa, A.; Takimoto, T. Critical hepatic hemangioma in infants: recent nationwide survey in Japan. Pediatr Int. 2014, 56, 304–308. [Google Scholar] [CrossRef]
- Ji, Y.; Chen, S.; Xianng, B.; Xu, Z.; Jiang, X.; Liu, X.; et al. Clinical features and management of multifocal hepatic hemangiomas in children: a retrospective study. Sci Rep. 2016, 6, 31744. [Google Scholar] [CrossRef]
- Kalpatthi, R.; Germak, J.; Mizelle, K.; Yeager, N. Thyroid abnormalities in infantile hepatic hemangioendotheliomas. Pediatr Blood Cancer 2007, 49, 1021–1024. [Google Scholar] [CrossRef]
- Krowchuk, D.P.; Frieden, I.J.; Mancini, A.J.; Darrow, D.H.; Blei, F.; Greene, A.K.; et al. Clinical practice guideline for the management of infantile hemangiomas. Pediatrics 2019, 143, 20183475. [Google Scholar] [CrossRef]
- Léauté-Labrèze, C.; Dumas; De la Roque, E.; Hubiche, T.; et al. Propranolol for severe hemangiomas of infancy. N Engl J Med. 2008, 358, 2649–2651. [Google Scholar] [CrossRef]
- Gonzalez Martinez, O.G.; Langer, P.D.; Milman, T. Propranolol-induced Involution of Infantile Hemangioma. Ophthalmology 2023, 130, 1359. [Google Scholar] [CrossRef]
- Hoeger, P.H.; Harper, J.I.; Baselga, E.; et al. Treatment of infantile haemangiomas: recommendations of a European expert group. Eur J Pediatr. 2015, 174, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Kotlukova, N.P.; Belysheva, T.S.; Shats, L.I.; et al. Possibilities of medical treatment of infantile hemangiomas in Russia. Russian Journal of Pediatric Hematology and Oncology 2022, 9, 22–28, (in Russ.). [Google Scholar] [CrossRef]
- Ji, Y.; Chen, S.; Xu, C.; Li, L.; Xiang, B. The use of propranolol in the treatment of infantile haemangiomas: an update on potential mechanisms of action. Br J Dermatol 2015, 172, 24–32. [Google Scholar] [CrossRef]
- Zavras, N.; Dimopoulou, A.; Machairas, N.; Paspala, A.; Vaos, G. Infantile hepatic hemangioma: current state of the art, controversies, and perspectives. Eur J Pediatr. 2020, 179, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Garzon, M.C.; Epstein, L.G.; Heyer, G.L.; Frommelt, P.C.; Orbach, D.B.; Baylis, A.L.; et al. PHACE syndrome: consensus-derived diagnosis and care recommendations. J Pediatr. 2016, 178, 24–33. [Google Scholar] [CrossRef]
- Shah, S.D.; Baselga, E.; McCuaig, C.; Pope, E.; Coulie, J.; Boon, L.M.; et al. Rebound growth of infantile hemangiomas after propranolol therapy. Pediatrics 2016, 137, 20151754. [Google Scholar] [CrossRef] [PubMed]
- Rialon, K.L.; Murillo, R.; Fevurly, R.D.; Kulungowski, A.M.; Christison-Lagay, E.R.; Zurakowski, D.; et al. Risk factors for mortality in patients with multifocal and diffuse hepatic hemangiomas. J Pediatr Surg. 2015, 50, 837–841. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, A.; Durkin, N.; Deganello, A.; Sellars, M.E.; Makin, E.; Davenport, M. Historical and contemporary management of infantile hepatic hemangioma: A 30-year single-center experience. Ann Surg. 2022, 275, 250–255. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |