1. Introduction
Upper gastrointestinal bleeding (UGIB) is a frequent reason for emergency admissions and remains a condition linked to notable rates of illness, death, and extensive use of medical resources [
1]. The incidence of UGIB is estimated at 80-150 per 100,000 people annually, with mortality rates ranging from 2% to 15% [
2,
3]. Despite advances in endoscopic techniques and supportive care, mortality rates have remained relatively stable over the past two decades, highlighting the need for improved risk stratification tools [
4]. UGIB is classified as variceal bleeding and non-variceal bleeding according to the etiology [
5]. Variceal bleeding results from the rupture of varices in the esophagus or stomach, commonly associated with portal hypertension due to cirrhosis. It accounts for approximately 70% of UGIB episodes in patients with cirrhosis and carries a higher mortality risk than non-variceal causes[
6].
Although peptic ulcer is the most common cause of non-variceal upper gastrointestinal bleeding, it arises from a disruption in the balance between gastric acid secretion, mucosal defense mechanisms, and pepsin activity [
7]. Early risk stratification in the management of UGIB is important to identify patients who will benefit from close follow-up and treatment, and those who may have a high risk of complications. Risk stratification helps clinicians to determine the need for intensive care unit admission, length of hospital stay, and duration of follow-up [
8,
9]. Identifying accessible and reliable predictors of adverse outcomes remains an important goal in UGIB management. Complete blood count (CBC) parameters are used worldwide, are inexpensive and are routinely investigated in UGIB patients. Multiple biomarkers derived from CBC have been studied as indicators of systemic inflammation in various diseases [
10]. Platelet-to-Lymphocyte Ratio (PLR), Neutrophil-to- Lymphocyte Ratio (NLR), and Monocyte-to-Lymphocyte Ratio (MLR) have shown prognostic value in inflammatory situations, including cancer, renal failure, and acute inflammatory conditions [
11,
12,
13]. These ratios illustrate the balance of adaptive immune responses and innate that can be disrupted during acute bleeding episodes [
10]. More complex inflammatory indices have been developed to demonstrate the prognostic significance of CBC-derived biomarkers. One of these, the Systemic Immune Inflammation Index (SII), is calculated as the product of neutrophil and platelet counts divided by the lymphocyte count. SII has demonstrated prognostic value in various malignancies and cardiovascular diseases [
14,
15]. Similarly, the Systemic Inflammation Response Index (SIRI), calculated as (neutrophil × monocyte)/lymphocyte, represents another composite marker incorporating monocytes, which play a crucial role in tissue repair and inflammation resolution.SIRI has been associated with outcomes in certain cancers and inflammatory conditions, including an elevated risk of pneumonia following subarachnoid hemorrhage [
16,
17]. Compared to NLR or PLR, the systemic immune-inflammation index (SII) and the systemic inflammation response index (SIRI), which are relatively newer biomarkers, have a greater potential to effectively assess systemic inflammation [
18]. Despite the potential relevance of systemic inflammation in UGIB outcomes, the prognostic value of these inflammatory biomarkers in UGIB has been insufficiently explored.A recent study by Bozan and colleagues evaluated SII and CRP/albumin ratio in patients with gastrointestinal bleeding but found limited predictive ability for the Forrest classification [
19]. However, in this study, they did not evaluate the relationship that may exist between the available indices and critical clinical outcomes such as mortality. The inflammatory cascade triggered by acute bleeding, tissue injury, and ischemiareperfusion plays a significant role in determining clinical outcomes in UGIB. Therefore, accessible biomarkers reflecting systemic inflammation might provide valuable information for risk assessment. Given that inflammatory biomarkers are derived from routinely collected CBC and reflect pathophysiological processes relevant to UGIB, they merit further investigation as potential prognostic tools.
This study aims to assess the importance of NLR, PLR, MLR, especially SII and SIRI in predicting ICU admission rates and mortality in UGIB patients. By identifying simple, accessible biomarkers that can help determine risk in UGIB, we hope to contribute to improving patient triage, resource allocation and ultimately clinical outcomes in UGIB. This study represents an important step towards understanding whether inflammatory indices can provide valuable prognostic information in this vulnerable patient population.
2. Materials and Methods
This retrospective cohort study was conducted at xxx University Hospital, a tertiary care institution in Turkey. The study population consisted of adult patients (18 years and older) diagnosed and treated for UGIB between May 2016 and December 2021. This study was approved by the Interventional Clinical Research Ethics Committee of xxx University (approval no: 76244175-050.04-393103, date: November 29, 2024). The study was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent could not be obtained because the study was planned retrospectively. Patients were identified through the hospital’s electronic medical record system using International Classification of Diseases (ICD) codes related to UGIB. Inclusion criteria were: (1) age ≥18 years, (2) presentation with symptoms suggestive of UGIB (hematemesis, melena, or hematochezia with hemodynamic instability), (3) endoscopic confirmation of upper gastrointestinal bleeding source, and (4) availability of complete blood count results at admission. Exclusion criteria were: (1) lower gastrointestinal bleeding, (2) patients with active hematological malignancies or receiving chemotherapy, (3) patients with known active infection at admission, (4) patients who received blood transfusions prior to initial blood sampling, and (5) incomplete clinical or laboratory data.Demographic data, comorbidities, laboratory parameters, endoscopic findings, treatments, and clinical outcomes were extracted from electronic medical records. For patients with liver cirrhosis, liver function parameters including serum bilirubin, albumin, and international normalized ratio (INR) were recorded.
2.1. Clinical Management Protocol
All patients were initially managed according to the institution’s protocol for UGIB. This included hemodynamic assessment and resuscitation, blood typing and cross-matching, and administration of intravenous proton pump inhibitors for suspected non-variceal bleeding or vasoactive drugs (terlipressin or octreotide) for suspected variceal bleeding. All patients underwent endoscopic evaluation within 24 hours of hospital admission. For patients with hemodynamic instability (systolic blood pressure <90 mmHg despite fluid resuscitation or heart rate >100 beats/minute) or massive hematemesis, urgent endoscopy was performed within 12 hours. All endoscopic procedures were performed by experienced gastroenterologists. Patients were admitted to the ICU based on the following criteria: (1) hemodynamic instability requiring vasopressor support, (2) respiratory failure requiring mechanical ventilation or (3) severe ongoing bleeding requiring close monitoring. The decision for ICU admission was made by the attending physician based on clinical judgment and institutional protocols.
2.2. Endoscopic Classification and Treatment
Non-variceal bleeding sources, particularly peptic ulcers, were classified according to the Forrest classification: Forrest Ia (spurting bleeding), Ib (oozing bleeding), IIa (non-bleeding visible vessel), IIb (adherent clot), IIc (flat pigmented spot), and III (clean ulcer base). Variceal bleeding was classified according to the location (esophageal, gastroesophageal, or isolated gastric varices [IGV]).Endoscopic treatment was provided according to the bleeding source. For variceal bleeding, band ligation was the primary treatment for esophageal varices, while sclerotherapy (using cyanoacrylate) was used for gastric varices. For non-variceal bleeding, various endoscopic techniques were employed, including injection therapy (epinephrine), thermal coagulation, hemoclips, or combination therapy based on the endoscopist’s judgment and the nature of the lesion. Treatment failure was defined as persistent bleeding despite initial endoscopic therapy or recurrent bleeding within 30 days. Management of treatment failure included repeat endoscopic therapy, angiographic embolization, or surgery depending on clinical circumstances and institutional resources.
2.3. Laboratory Measurements and Inflammatory Biomarkers
Peripheral venous blood samples were obtained from all patients at the time of their admission to the emergency department, prior to the initiation of any medical intervention or blood transfusion. CBC analyses were conducted using an automated hematology analyzer (Beckman Coulter LH 780, USA). Based on the CBC parameters, the following inflammation-based hematologic indices were calculated for each patient:
NLR: Absolute neutrophil count/absolute lymphocyte count
PLR: Absolute platelet count/absolute lymphocyte count
MLR: Absolute monocyte count/absolute lymphocyte count
SII: (Neutrophil count × Platelet count) / Lymphocyte count
SIRI: (Neutrophil count × Monocyte count) / Lymphocyte count
2.4. Outcome Measures
The primary outcome measure was 30-day all-cause mortality. Secondary outcomes included need for ICU admission, hospital length of stay, ICU length of stay, need for blood transfusion, and endoscopic treatment failure. Mortality data were obtained from hospital records and the national death registry.
2.5. Statistical Analysis
Categorical variables are presented as frequencies and percentages. The normality of distribution for continuous variables was assessed using the Shapiro-Wilk test. Normally distributed continuous variables are presented as mean ± standard deviation, while non-normally distributed variables are presented as median with interquartile range or minimum maximum range. Comparisons between survivors and non-survivors for categorical variables were performed using the Chi-square test or Fisher’s exact test as appropriate. For continuous variables, the Student’s t-test was used for normally distributed data and the Mann-Whitney U test for non-normally distributed data. The Kruskal-Wallis test was used for comparing inflammatory markers across Forrest classification groups. Receiver operating characteristic (ROC) curve analysis was performed to evaluate the discriminative ability of inflammatory biomarkers for predicting 30-day mortality. The area under the curve (AUC), optimal cutoff values (determined by Youden’s index), sensitivity, specificity, and 95% confidence intervals were calculated. Multivariable logistic regression analysis was conducted to assess the independent association of inflammatory biomarkers with 30-day mortality after adjusting for potential confounders, including age, comorbidities, and bleeding etiology (variceal vs. non-variceal). For patients with variceal bleeding, liver function parameters were included in the model. Results are presented as adjusted odds ratios (OR) with 95% confidence intervals. Subgroup analyses were performed for variceal and nonvariceal bleeding to evaluate the performance of inflammatory biomarkers in these distinct clinical entities. A p-value <0.05 was considered statistically significant for all analyses. Statistical analyses were performed using SPSS version 26 (IBM Inc., Chicago, IL, USA).
3. Results
3.1. Patient Characteristics and Clinical Outcomes
The study included 307 patients with endoscopically confirmed UGIB. The demographic, clinical, and laboratory characteristics of the study population, stratified by 30-day survival status (
Table 1). The mean age was 57.96 ± 18.7 years, with non-survivors being significantly older (65.20 ± 17.45 vs. 56.75 ± 18.66 years,
p=0.005). Males constituted 73% of the cohort, with a higher proportion among non-survivors (81.8% vs. 71.4%), though this difference was not statistically significant (
p=0.098). Admission hemoglobin and hematocrit levels were significantly lower in non-survivors compared to survivors (hemoglobin: 8.34 ± 2.07 vs. 9.20 ± 2.32 g/dL,
p=0.012; hematocrit: 25.50 ± 6.84% vs. 28.52 ± 7.15%,
p=0.006). Non-survivors required more blood transfusions (mean 5.57 vs. 2.62 units,
p=0.003) and had longer hospital stays (6.36 vs. 3.71 days,
p=0.009) and intensive care unit stays (6.16 vs. 2.29 days,
p<0.001). The overall 30-day mortality rate was 14.3% (44/307). A total of 234 patients (76.2%) received blood transfusions. The primary causes of death included hemorrhagic shock (n=17, 38.6%), multiple organ failure (n=15, 34.1%), and hepatic failure (n=12, 27.3%). The main criteria for ICU admission were hemodynamic instability requiring vasopressor support (n=73, 61.9%), respiratory failure requiring mechanical ventilation (n=28, 23.7%), and close monitoring for high-risk bleeding (n=17, 14.4%).
3.2. Endoscopic Findings and Treatments
Endoscopic findings and treatments are summarized in
Table 2. Esophageal variceal bleeding was identified in 116 patients (37.8%), with a significantly higher proportion among non-survivors compared to survivors (63.6% vs. 33.5%,
p<0.001). Among patients with non-variceal bleeding, the distribution across Forrest classifications differed significantly between survivors and non-survivors (
p=0.006), with higher proportions of Forrest 1a and 1b classifications among non-survivors. For patients with variceal bleeding, liver function parameters indicated more severe liver disease among non-survivors, with higher median bilirubin (3.2 vs. 1.8 mg/dL, p<0.001), lower albumin (2.6 vs. 3.1 g/dL,
p=0.002), and higher INR values (1.8 vs. 1.4, p<0.001) compared to survivors. Endoscopic band ligation was performed in 94 patients (30.6%), sclerotherapy in 116 (37.8%), thermal therapy in 32 (10.4%), and hemoclip application in 26 (8.5%). Combined endoscopic therapy was used in 38 patients (12.4%). No endoscopic intervention was required in 94 patients (30.6%). Endoscopic treatment failure, defined as persistent or recurrent bleeding despite initial endoscopic therapy, occurred in 36 patients (11.7%). Among these, 24 (66.7%) underwent repeat endoscopic therapy, 11 (30.6%) underwent angiographic embolization, and 1 (2.8%) required surgical intervention.
3.3. Inflammatory Biomarkers and Clinical Outcomes
The relationship between inflammatory biomarkers and survival status is shown in
Table 1. Nonsurvivors had significantly higher values of NLR (median 13.13 vs. 5.88,
p<0.001), MLR (median 0.75 vs. 0.46,
p=0.002), SII (median 2544 vs. 1268,
p=0.030), and SIRI (median 9.23 vs. 3.96,
p=0.003) compared to survivors. PLR showed a trend toward higher values in non-survivors, but this difference did not reach statistical significance (median 251.24 vs. 154.66,
p=0.078). When stratified by bleeding etiology, the association between inflammatory biomarkers and mortality remained significant in both variceal and non-variceal groups for NLR, MLR, and SIRI (all
p<0.05). However, SII showed a significant association with mortality only in the non-variceal group (
p=0.022) but not in the variceal group (
p=0.217). Analysis of inflammatory biomarkers across Forrest classification groups (
Table 3) showed that only SIRI values demonstrated a statistically significant difference (
p=0.037), with higher median values observed in Forrest 1 (3.96, 95% CI: 0.67-43.78) compared to Forrest 2 (2.48, 95% CI: 0.03-34.80) and Forrest 3 (2.22, 95% CI: 0-47.39). The other inflammatory markers, including SII (
p=0.490), NLR (
p=0.862), PLR (
p=0.520), and MLR (
p=0.218), did not show statistically significant differences across the Forrest classification groups.
3.4. Predictive Performance of Inflammatory Biomarkers
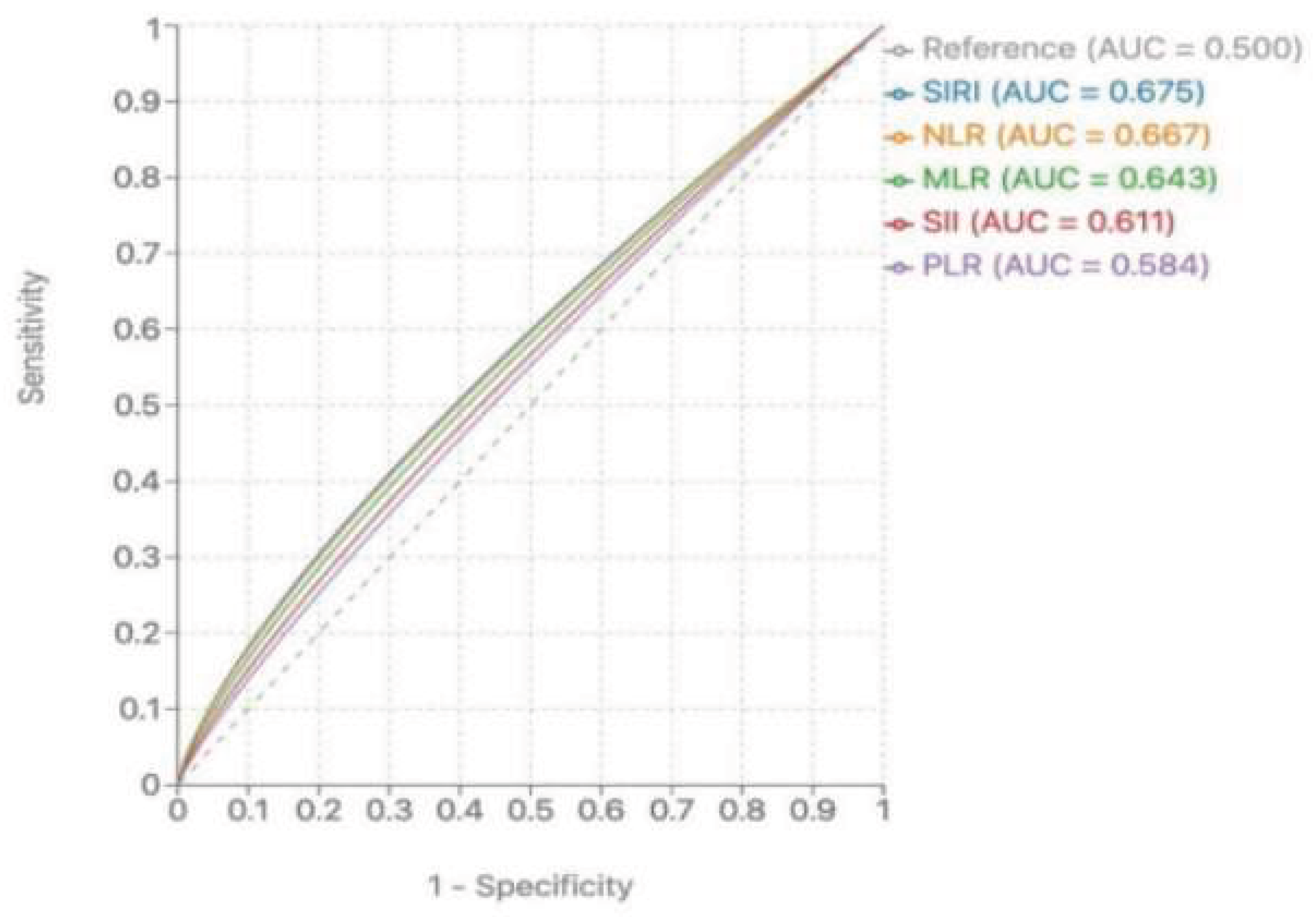
ROC curve analysis was performed to evaluate the predictive performance of inflammatory biomarkers for 30-day mortality (Table 4,
Figure 1). Among the inflammatory biomarkers, SIRI demonstrated the highest AUC (0.675, 95% CI: 0.584-0.766,
p<0.001), followed by NLR (0.667, 95% CI: 0.574-0.760,
p<0.001), MLR (0.643, 95% CI: 0.552-0.735,
p=0.002), SII (0.611, 95% CI: 0.519-0.704,
p=0.019), and PLR (0.584, 95% CI: 0.493-0.675,
p=0.078). The optimal cutoff values for SII and SIRI in predicting mortality were determined to be 1056.8 (sensitivity 61.4%, specificity 60.8%) and 3.19 (sensitivity 65.9%, specificity 63.1%), respectively. In the subgroup analysis by bleeding etiology, all inflammatory biomarkers showed slightly better discriminative performance in non-variceal bleeding compared to variceal bleeding. In non-variceal bleeding, the AUC for SIRI was 0.692 (95% CI: 0.572-0.813,
p=0.003) and for SII was 0.635 (95% CI: 0.511-0.759,
p=0.032), while in variceal bleeding, the AUC for SIRI was 0.641 (95% CI: 0.519-0.763,
p=0.021) and for SII was 0.583 (95% CI: 0.455-0.711,
p=0.157).
3.5. Multivariable Analysis
Multivariable logistic regression analysis was performed to assess the independent association of inflammatory biomarkers with 30-day mortality after adjusting for established risk factors. After adjusting for age, comorbidities, and bleeding etiology (variceal vs. non-variceal), both SIRI (adjusted OR 1.08 per unit increase, 95% CI: 1.02-1.14, p=0.009) and NLR (adjusted OR 1.06 per unit increase, 95% CI: 1.01-1.11, p=0.014) remained independently associated with mortality. SII, MLR, and PLR lost their significant association with mortality in the multivariable model. In the etiology-specific multivariable analysis, SIRI remained an independent predictor of mortality in non-variceal bleeding (adjusted OR 1.11, 95% CI: 1.03-1.20, p=0.005) but showed a weaker association in variceal bleeding (adjusted OR 1.05, 95% CI: 0.99-1.12, p=0.091) after adjusting for the same covariates plus liver function parameters.
4. Discussion
UGIB remains a significant clinical challenge with substantial morbidity and mortality worldwide [
19,
20]. Effective risk stratification is essential for optimizing resource allocation and improving patient outcomes. This study investigated the potential utility of inflammatory biomarkers derived from complete blood count as prognostic indicators in patients with UGIB. The mean age of the errors included in the study was 57.96 years, which is consistent with previous epidemiological studies on UGIB [
8,
21]. However, we observed a higher proportion of variceal bleeding (37.8%) compared to prior reports, which typically document prevalence rates of 10-30% [
22,
23]. This disparity is probably attributable to our hospital’s role as a tertiary referral center specializing in liver diseases, which leads to a greater proportion of patients presenting with portal hypertension. The relatively high 30-day mortality rate (14.3%) in our cohort, compared to 7.75% reported by Lau et al. [
23], may also be attributed to this higher proportion of variceal bleeding, which generally carries a poorer prognosis than nonvariceal causes.
When examining the relationship between inflammatory biomarkers and clinical outcomes, we found that both SII and SIRI demonstrated statistically significant associations with 30-day mortality. Nonsurvivors had notably higher SII and SIRI values compared to survivors. Additionally, SIRI values showed significant differences across Forrest classification groups (
p=0.037), suggesting a potential relationship with the severity of non-variceal bleeding. In contrast, SII, NLR, PLR, and MLR did not demonstrate significant differences across Forrest classification groups. However, the discriminative ability of these biomarkers for predicting mortality was limited, with AUC values of 61.1% for SII and 67.5% for SIRI. According to established interpretation guidelines for AUC values, scores below 0.7 indicate poor discrimination [
24]. Our findings partially differ from those of Bozan et al., who reported no significant correlation between SII and the Forrest classification in UGIB patients [
21]. While we also found no significant relationship between SII and Forrest classification (
p=0.49), we did observe that SIRI values differed significantly across Forrest groups (
p=0.037). This discrepancy might be related to our larger sample size or differences in patient characteristics. The limited predictive performance of inflammatory biomarkers in our study deserves careful consideration. Several factors may contribute to this finding. First, UGIB represents a heterogeneous condition with diverse etiologies and pathophysiological mechanisms. The inflammatory response may vary substantially between variceal and non-variceal bleeding, as well as across different non-variceal causes [
25,
26]. Our subgroup analysis revealed that the predictive value of inflammatory markers was somewhat better in non-variceal bleeding (SII AUC 63.5%, SIRI AUC 69.2%) compared to variceal bleeding (SII AUC 58.3%, SIRI AUC 64.1%), though still below the threshold for good discrimination. Second, systemic inflammation represents just one aspect of UGIB pathophysiology. Multiple other factors, including age, comorbidities, hemodynamic parameters, and endoscopic findings, contribute significantly to patient outcomes [
27,
28]. In our multivariable analysis, after adjusting for age, comorbidities, and bleeding source, SIRI remained an independent predictor of mortality (adjusted OR 1.08, 95% CI 1.02-1.14,
p=0.009), suggesting it may capture a distinct aspect of risk not fully accounted for by traditional factors. Third, the timing of inflammatory marker measurement may influence their predictive value. We analyzed blood samples collected at admission, but the inflammatory response evolves dynamically during the course of UGIB. Serial measurements might better capture this temporal dimension and potentially improve prognostic accuracy. Despite these limitations, inflammatory biomarkers offer certain advantages as prognostic tools. They are derived from routine blood tests, making them universally available and cost-effective. Their calculation is straightforward and can be performed rapidly, potentially facilitating early risk assessment before more comprehensive evaluation is complete. Additionally, the pathophysiological basis of these markers provides biological plausibility for their association with outcomes in UGIB. There are several important limitations to our study that must be acknowledged. First, the retrospective, single-center design may introduce selection bias and limit the generalizability of the improvement. Second, the heterogeneous nature of our study population, including both variceal and non-variceal bleeding, may obscure etiology-specific associations. Third, factors that could influence the course of the disease, including specific comorbidities, history of medication use (especially antithrombotic and NSAIDs) and liver function parameters, were not complete for all patients. Fourth, inflammatory markers were analyzed only at admission, precluding assessment of dynamic changes that might enhance prognostic value. Future research directions should include prospective, multicenter studies with larger sample sizes and more homogeneous patient populations. Investigating the dynamic changes in inflammatory biomarkers during the course of UGIB might reveal patterns with enhanced predictive value. Additionally, exploring the potential complementary role of these biomarkers within existing clinical assessment frameworks could leverage their distinct pathophysiological basis while addressing their limitations as standalone predictors.
5. Conclusion
This study reveals that the inflammatory biomarkers SII and SIRI are significantly associated with mortality in UGIB patients, although their ability to independently predict outcomes remains limited. These easily calculated indices derived from routine blood tests may provide complementary information in the risk assessment of UGIB patients. The modest but independent association of SIRI with mortality after adjusting for known risk factors suggests it may capture a distinct aspect of risk related to the inflammatory response. Prospective studies with larger patient groups are needed to confirm these results, to examine changes in inflammatory biomarkers during UGIB and to investigate the possible integration of these markers into clinical evaluations. The ultimate goal remains to enhance our ability to identify high-risk patients early and tailor management strategies accordingly, potentially improving outcomes in this life-threatening condition.
All stages of this research were conducted solely by the author without any external assistance. The freely available OpenAI GPT-4o tool was used in a highly limited manner. Its use was restricted to minor adjustments such as paragraph formatting, section separation, standardization of reference formatting, suggesting synonyms for no more than ten words, and correcting a few typographical errors.
Author Contributions
Conceptualization, F.Z.A., C.C. and V.A.; Methodology, F.Z.A., C.C., D.A., A.U. and V.A.; Software, F.Z.A.; Validation, F.Z.A., C.C. and D.A.; Formal analysis, F.Z.A. and D.A.; Investigation, F.Z.A., C.C. and V.A.; Resources, F.Z.A.; Data curation, D.A., A.U. and V.A.; writing—original draft preparation, F.Z.A., C.C. and V.A.; Writing—review and editing, F.Z.A., C.C., A.U. and V.A.; Visualization, F.Z.A. and D.A.; Supervision, F.Z.A. and A.U.
Funding
This research received no external funding.
Institutional Review Board Statement
This study was approved by the Interventional Clinical Research Ethics Com-mittee of Haran University (approval no: 76244175-050.04-393103, date: November 29, 2024). The study was conducted in accordance with the principles of the Declaration of Helsinki.
Informed Consent Statement
Written informed consent could not be obtained because the study was planned retrospectively.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author(s).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lau, LHS; Sung, JJY. Treatment of upper gastrointestinal bleeding in 2020: New techniques and outcomes. Dig Endosc. 2021, 33(1), 83–94. [Google Scholar] [CrossRef]
- Antunes, C; Tian, C; Copelin, EL, II. Upper Gastrointestinal Bleeding. In StatPearls; StatPearls Publishing: Treasure Island (FL), 17 August 2024. [Google Scholar]
- Radaelli, F; Rocchetto, S; Piagnani, A; Savino, A; Di Paolo, D; Scardino, G; et al. Scoring systems for risk stratification in upper and lower gastrointestinal bleeding. Best Pract Res Clin Gastroenterol. 2023, 67, 101871. [Google Scholar] [CrossRef]
- Abougergi, MS; Travis, AC; Saltzman, JR. The in-hospital mortality rate for upper GI hemorrhage has decreased over 2 decades in the United States: a nationwide analysis. Gastrointest Endosc. 2015, 81(4), 882–8.e1. [Google Scholar] [CrossRef]
- Twum-Barimah, E; Abdelgadir, I; Gordon, M; Akobeng, AK. Systematic review with meta-analysis: the efficacy of tranexamic acid in upper gastrointestinal bleeding. Aliment Pharmacol Ther. 2020, 51(11), 1004–1013. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, SA; Jang, S. Pathophysiology and Management of Variceal Bleeding. Drugs 2021, 81(6), 647–667. [Google Scholar] [CrossRef]
- Wilkins, T; Wheeler, B; Carpenter, M. Upper Gastrointestinal Bleeding in Adults: Evaluation and Management [published correction appears in Am Fam Physician. 2021 Jan 15;103(2):70.]. Am Fam Physician 2020, 101(5), 294–300. [Google Scholar]
- Raţiu, I; Lupuşoru, R; Popescu, A; Sporea, I; Goldiş, A; Dănilă, M; et al. Acute gastrointestinal bleeding: A comparison between variceal and nonvariceal gastrointestinal bleeding. Medicine (Baltimore) 2022, 101(45), e31543. [Google Scholar] [CrossRef]
- Garcia-Tsao, G; Abraldes, JG; Berzigotti, A; Bosch, J. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases [published correction appears in Hepatology. 2017 Jul;66(1):304. doi: 10.1002/hep.29169.]. Hepatology 2017, 65, 310–335. [Google Scholar] [CrossRef] [PubMed]
- Baba, Y; Nakagawa, S; Toihata, T; Harada, K; Iwatsuki, M; Hayashi, H; et al. Pan-immune-inflammation Value and Prognosis in Patients With Esophageal Cancer. Ann Surg Open 2021, 3(1), e113. [Google Scholar] [CrossRef]
- Song, CY; Meng, YL; Liu, B; Yan, L; Shang, PZ; Jia, ZF; et al. [Correlation analysis of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio and central cervical lymph node metastasis of papillary thyroid microcarcinoma]. Zhonghua Zhong Liu Za Zhi 2021, 43, 944–948, Chinese. [Google Scholar] [CrossRef]
- Hu, X; Tian, T; Zhang, X; Sun, Q; Chen, Y; Jiang, W. Neutrophil-to-lymphocyte and hypopharyngeal cancer prognosis: System review and meta-analysis. Head Neck 2023, 45(2), 492–502. [Google Scholar] [CrossRef]
- Heymann, WR. The neutrophil-to-lymphocyte ratio in cutaneous oncology: Simply elegant. J Am Acad Dermatol 2022, 86(3), 533–534. [Google Scholar] [CrossRef] [PubMed]
- Wang, H; Nie, H; Bu, G; Tong, X; Bai, X. Systemic immune-inflammation index (SII) and the risk of all-cause, cardiovascular, and cardio-cerebrovascular mortality in the general population. Eur J Med Res. 2023, 28(1), 575. [Google Scholar] [CrossRef]
- Huang, H; Liu, Q; Zhu, L; Zhang, Y; Lu, X; Wu, Y; et al. Prognostic Value of Preoperative Systemic Immune-Inflammation Index in Patients with Cervical Cancer. Sci Rep. 2019, 9(1), 3284. [Google Scholar] [CrossRef]
- Wang, RH; Wen, WX; Jiang, ZP; Du, ZP; Ma, ZH; Lu, AL; et al. The clinical value of neutrophil-to-lymphocyte ratio (NLR), systemic immune-inflammation index (SII), platelet-to-lymphocyte ratio (PLR) and systemic inflammation response index (SIRI) for predicting the occurrence and severity of pneumonia in patients with intracerebral hemorrhage. Front Immunol. 2023, 14, 1115031. [Google Scholar] [CrossRef]
- Schietroma, M; Romano, L; Schiavi, D; Pessia, B; Mattei, A; Fiasca, F; et al. Systemic inflammation response index (SIRI) as predictor of anastomotic leakage after total gastrectomy for gastric cancer. Surg Oncol. 2022, 43, 101791. [Google Scholar] [CrossRef]
- Kulakli, S; Oguz, ID; Aksan, B. Could Blood Cell-Based Inflammatory Markers Be Used to Monitor Response to Biologic Therapy in Psoriasis? Med Bull Sisli Etfal Hosp. 2023, 57(4), 536–542. [Google Scholar] [CrossRef]
- Ö, Bozan; Atiş, ŞE. Relationship of the CRP/albumin ratio and the systemic immune-inflammation index with Forrest classification in patients with gastrointestinal bleeding. J Health Sci Med. 2022, 5(2), 482–486. [Google Scholar] [CrossRef]
- Long, B; Gottlieb, M. Emergency medicine updates: Upper gastrointestinal bleeding. Am J Emerg Med 2024, 81, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Almadi, MA; Almutairdi, A; Alruzug, IM; Aldarsouny, TA; Semaan, T; Aldaher, MK; et al. Upper gastrointestinal bleeding: Causes and patient outcomes. Saudi J Gastroenterol 2021, 27(1), 20–27. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y; Shu, J; Mu, J; He, Q; Chen, F; Hu, Y; et al. Clinical analysis of 99 children with Henoch-Schönlein purpura complicated with overt gastrointestinal bleeding. Clin Rheumatol 2022, 41(12), 3783–3790. [Google Scholar] [CrossRef]
- Lau, JYW; Yu, Y; Tang, RSY; Chan, HCH; Yip, HC; Chan, SM; et al. Timing of Endoscopy for Acute Upper Gastrointestinal Bleeding. N Engl J Med. 2020, 382(14), 1299–1308. [Google Scholar] [CrossRef]
- Mandrekar, JN. Receiver operating characteristic curve in diagnostic test assessment. J Thorac Oncol. 2010, 5(9), 1315–1316. [Google Scholar] [CrossRef] [PubMed]
- Hess, JJ; Wallenstein, J; Ackerman, JD; Akhter, M; Ander, D; Keadey, MT; et al. Scribe Impacts on Provider Experience, Operations, and Teaching in an Academic Emergency Medicine Practice. West J Emerg Med 2015, 16(5), 602–10. [Google Scholar] [CrossRef]
- Dertli, R; Toka, B; Asıl, M; Kayar, Y; Karakarcayıldız, A; Göktepe, MH; et al. Can neutrophil-lymphocyte ratio predict mortality in acute non-variceal upper gastrointestinal bleeding? Ulus Travma Acil Cerrahi Derg 2022, 28(5), 626–633. [Google Scholar] [CrossRef]
- Bjarnason, I; Scarpignato, C; Holmgren, E; Olszewski, M; Rainsford, KD; Lanas, A. Mechanisms of Damage to the Gastrointestinal Tract From Nonsteroidal Anti-Inflammatory Drugs. Gastroenterology 2018, 154(3), 500–514. [Google Scholar] [CrossRef]
- Lanas, A; Dumonceau, JM; Hunt, RH; Fujishiro, M; Scheiman, JM; Gralnek, IM; Campbell, HE; Rostom, A; Villanueva, C; Sung, JJY; et al. Non-variceal upper gastrointestinal bleeding. Nat Rev Dis Primers 2018, 4, 18020. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).