Submitted:
19 February 2026
Posted:
24 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Inoculum and Nutrient Media
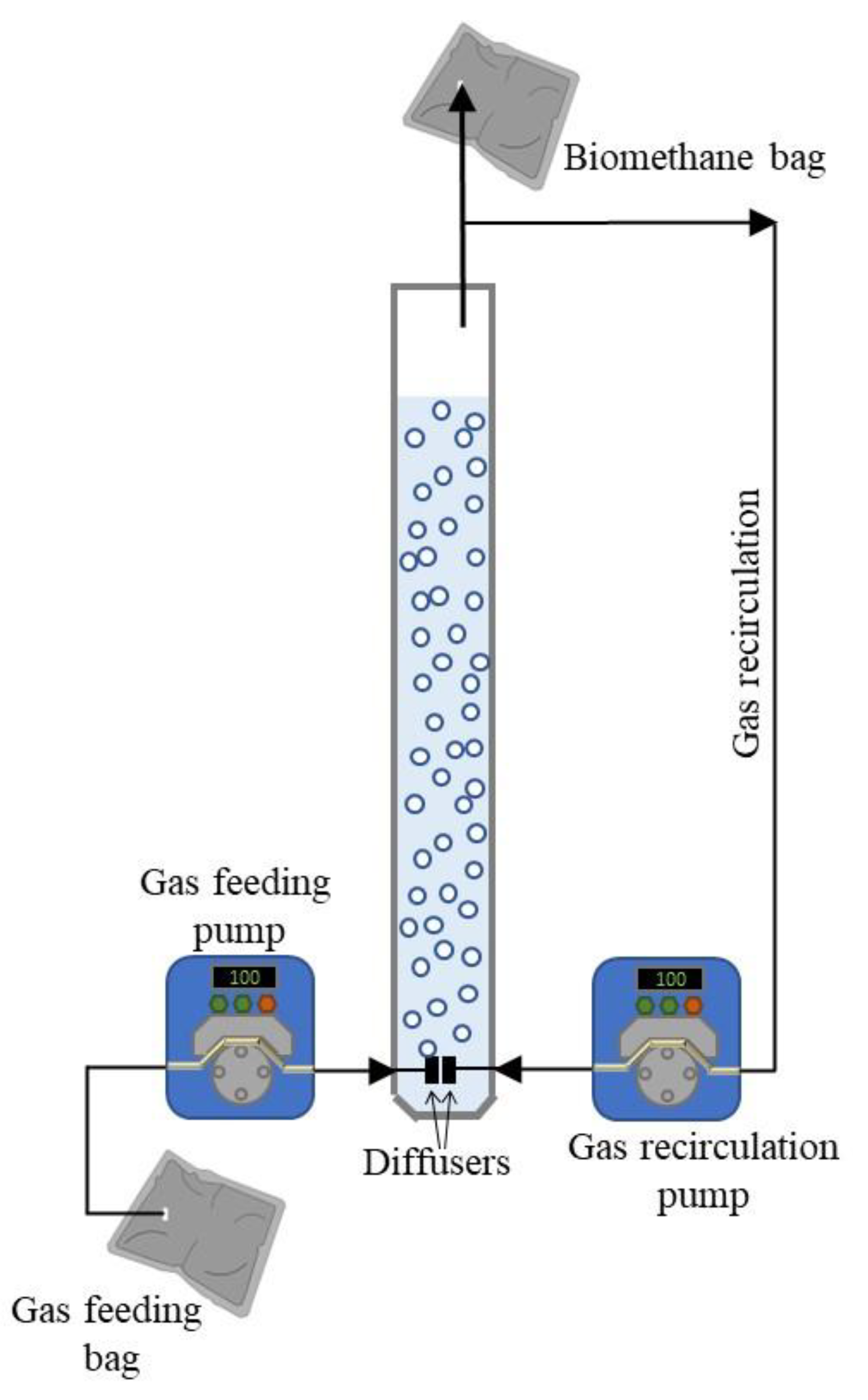
2.2. Experimental Setup and Operation
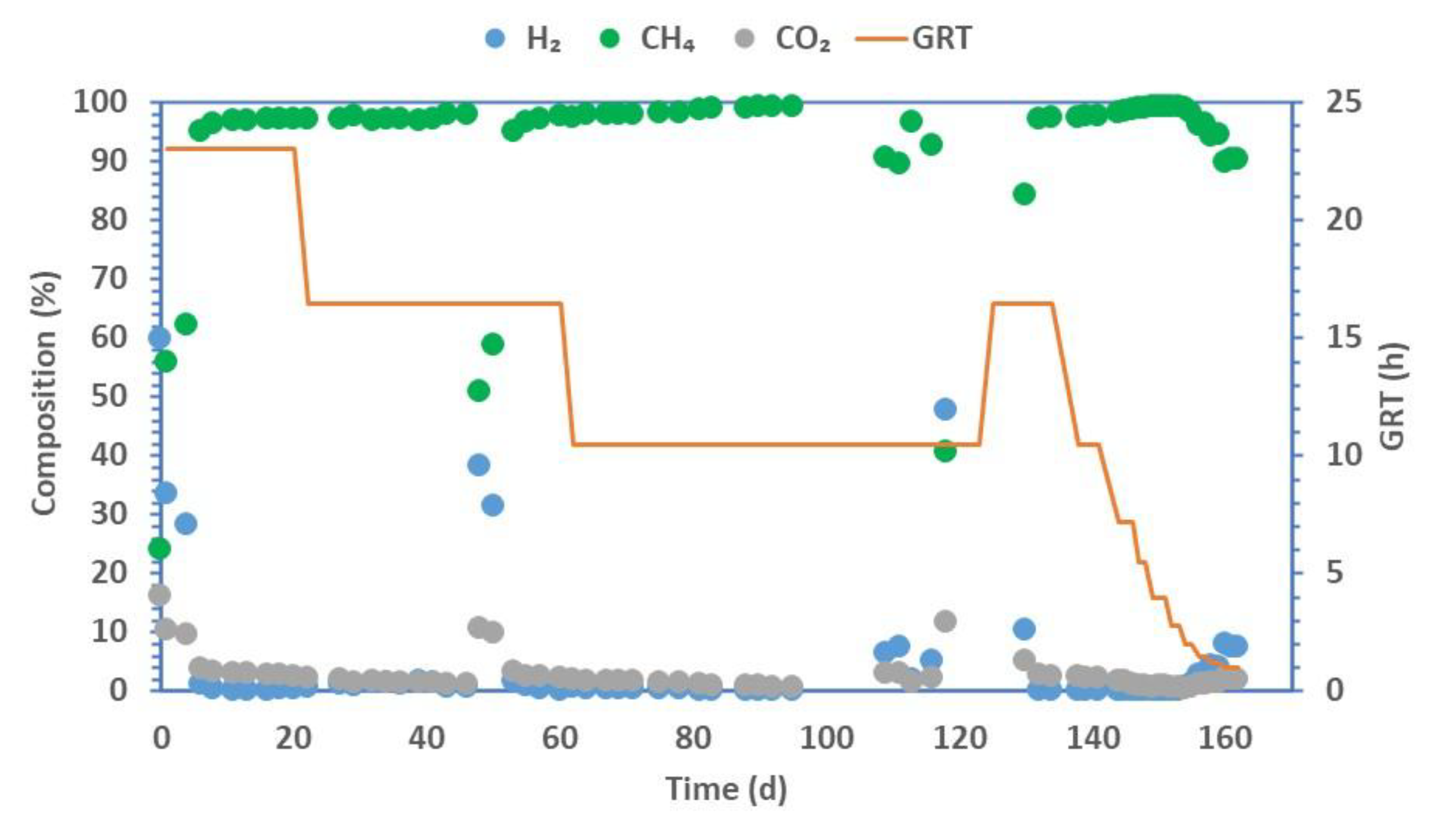
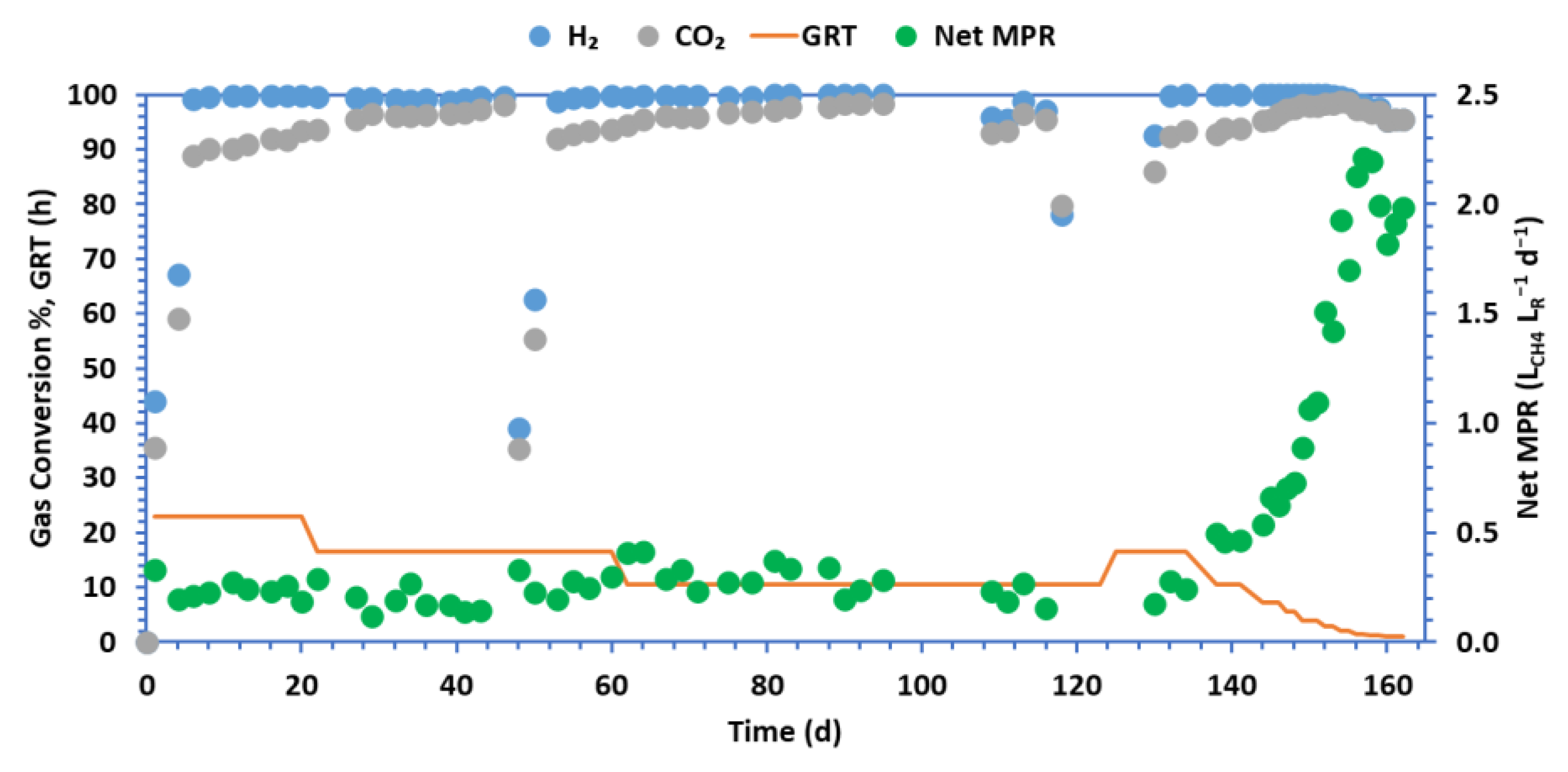
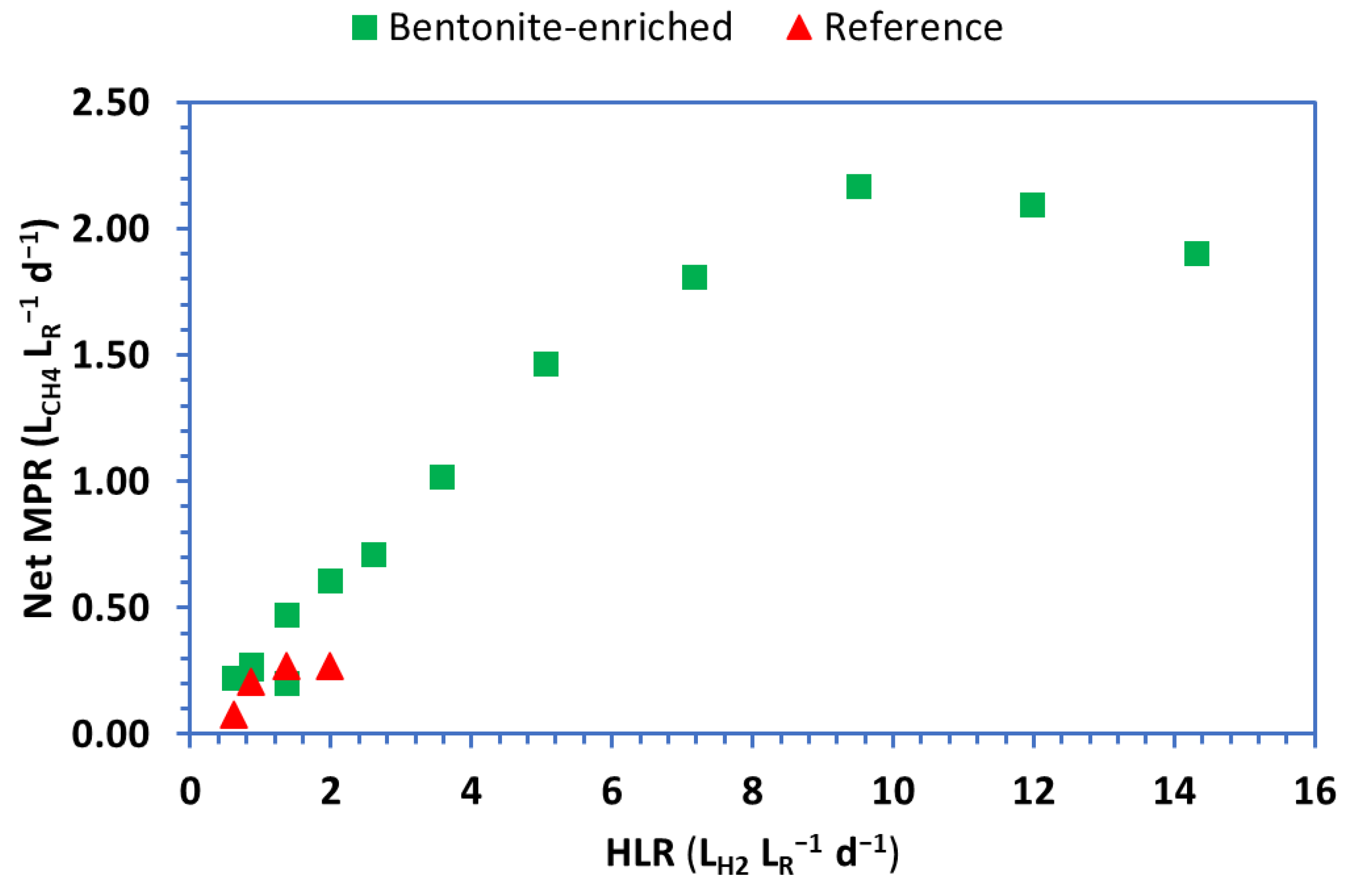
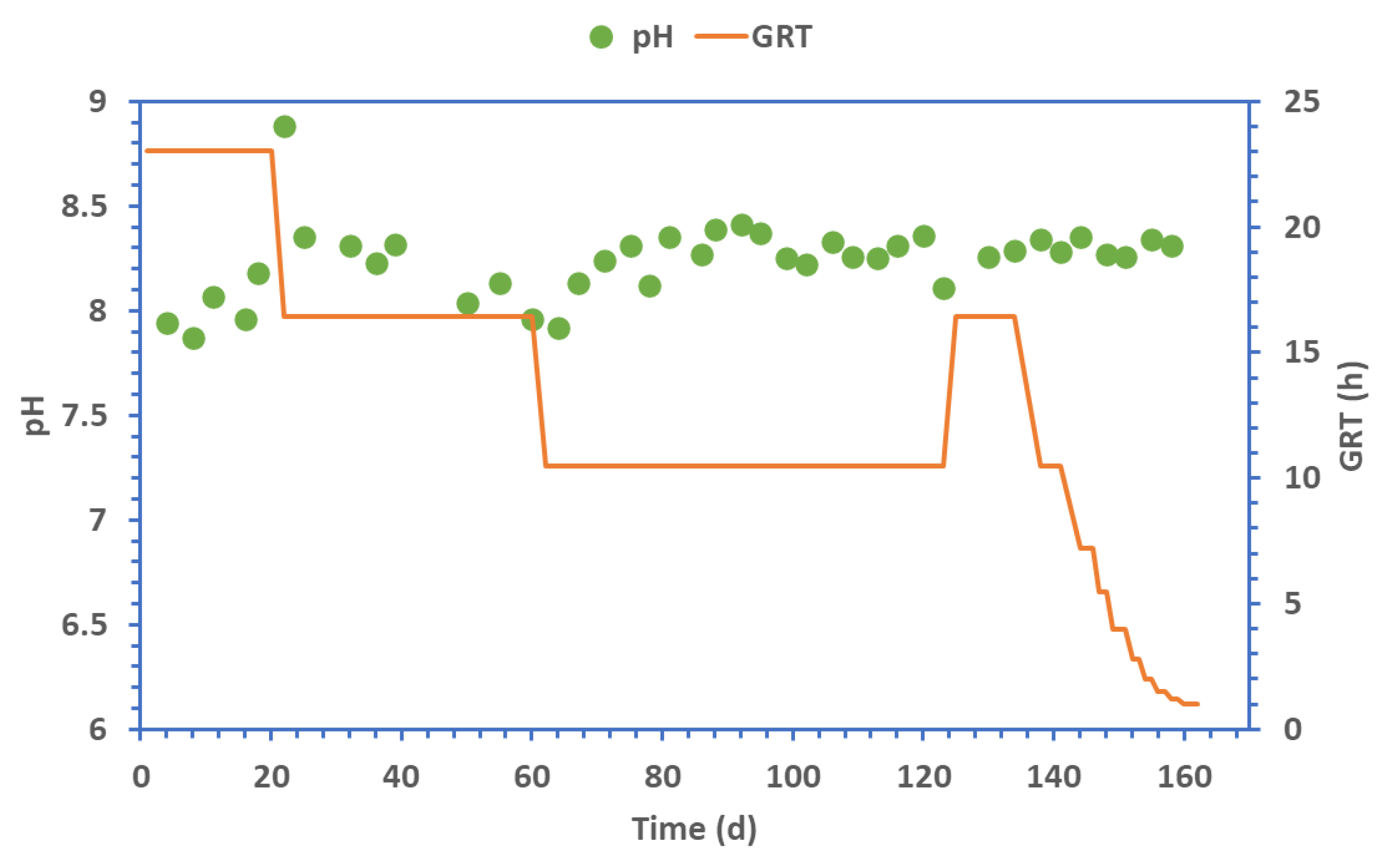
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BR | Bubble reactor |
| CH₄ | Methane |
| CLR | Carbon dioxide loading rate |
| CO2 | Carbon dioxide |
| COR | Carbon dioxide outlet rate |
| CSTR | Continuous stirred tank reactor |
| FOS/TAC | Ratio of volatile organic acids to total inorganic carbonate alkalinity |
| GHG | Greenhouse gas |
| GLR | Gas loading rate |
| GRT | Gas retention time (h) |
| H2 | Hydrogen |
| HLR | Hydrogen loading rate (L H2 LR−1 d−1) |
| HLR_in | Hydrogen inlet loading rate |
| HOR | Hydrogen outlet rate |
| kLA | Volumetric gas–liquid mass transfer coefficient |
| LR | Reactor working volume (L) |
| MLR | Methane loading rate |
| MPR | Methane production rate (L CH4 LR−1 d−1) |
| N2 | Nitrogen |
| NH3-N | Ammoniacal nitrogen |
| TBR | Trickle bed reactor |
| TS | Total solids |
| v/v | Volume per volume |
| VFAs | Volatile fatty acids |
| VS | Volatile solids |
Appendix A: Characterization of Bentonite and Digestate Used for Inoculation and Nutrient Supplement
| Parameter | Value |
| Dry matter (%) | 89 |
| pH dispersion 5% | 9.23 |
| Extractable Na (g kgDM−1) | 35 |
| Montmorillonate (g kgDM−1) | 800 |
| Particles <10 μm | 10 |
| Crystalline silica % | 3 |
| Specific surface absorption (mg 100g−1) | 300 |
| Disacidifying power (g kgDM−1) | 2.5 |
| Parameter | Value |
|---|---|
| pH | 7.78 ± 0.18 |
| Conductivity @25oC (mS cm−1) | 17.98 ± 0.23 |
| TS (%) | 3.29 ± 0.21 |
| VS (%) | 2.61 ± 0.50 |
| NH3-N (mg LR−1) | 1943 ± 32 |
| VFAs | |
| acetate (mg LR−1) | n.d. |
| propionate (mg LR−1) | n.d. |
| isobutyrate (mg LR−1) | n.d. |
| butyrate (mg LR−1) | n.d. |
| isovalerate (mg LR−1) | n.d. |
| valerate (mg LR−1) | n.d. |
| Total Alkalinity (g CaCO3 LR−1) | 10.9 |
| FOS/TAC | 0.178 |
| Potassium (%) | 2.07 |
| Aluminum (mg kgDM−1) | 1400 |
| Cadmium (mg kgDM−1) | 0.3 |
| Cobalt (mg kgDM−1) | 2.12 |
| Iron (mg kgDM−1) | 4680 |
| Copper (mg kgDM−1) | 391 |
| Manganese (mg kgDM−1) | 500 |
| Molybdenum (mg kgDM−1) | 8.75 |
| Nickel (mg kgDM−1) | 17.4 |
| Selenium (mg kgDM−1) | 0.95 |
| Tungsten (mg kgDM−1) | <0.500 |
| Tin (mg kgDM−1) | 1.29 |
| Zinc (mg kgDM−1) | 3.57 |
| Phosphorus (mg kgDM−1) | 3.01 |
Appendix B: Pressure Calculation
References
- Feickert Fenske, C.; Kirzeder, F.; Strübing, D.; Koch, K. Biogas Upgrading in a Pilot-Scale Trickle Bed Reactor – Long-Term Biological Methanation under Real Application Conditions. Bioresource Technology 2023, 376, 128868. [Google Scholar] [CrossRef]
- Charalambous, P.; Constantinou, D.; Samanides, C.G.; Vyrides, I. Enhancing Biogas Production from Cheese Whey Using Zero-Valent Iron: A Comparative Analysis of Batch and Semi-Continuous Operation Modes. Journal of Environmental Chemical Engineering 2023, 11, 111278. [Google Scholar] [CrossRef]
- Feickert Fenske, C.; Md, Y.; Strübing, D.; Koch, K. Preliminary Gas Flow Experiments Identify Improved Gas Flow Conditions in a Pilot-Scale Trickle Bed Reactor for H2 and CO2 Biological Methanation. Bioresource Technology 2023, 371, 128648. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Du, S.; Xie, L. Effects of pH on Ex-Situ Biomethanation with Hydrogenotrophic Methanogens under Thermophilic and Extreme-Thermophilic Conditions. Journal of Bioscience and Bioengineering 2021, 131, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Feickert Fenske, C.; Strübing, D.; Koch, K. Biological Methanation in Trickle Bed Reactors - a Critical Review. Bioresource Technology 2023, 385, 129383. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.; Cellamare, C.M.; Chiarini, L.; Tabacchioni, S.; Petta, L. Evaluation of the Controlled Hydrodynamic Cavitation as Gas Mass Transfer System for Ex-Situ Biological Hydrogen Methanation. Chemical Engineering Journal 2023, 471, 144475. [Google Scholar] [CrossRef]
- Agneessens, L.M.; Ottosen, L.D.M.; Andersen, M.; Berg Olesen, C.; Feilberg, A.; Kofoed, M.V.W. Parameters Affecting Acetate Concentrations during In-Situ Biological Hydrogen Methanation. Bioresource Technology 2018, 258, 33–40. [Google Scholar] [CrossRef]
- Ashraf, M.T.; Sieborg, M.U.; Yde, L.; Rhee, C.; Shin, S.G.; Triolo, J.M. Biomethanation in a Thermophilic Biotrickling Filter — pH Control and Lessons from Long-Term Operation. Bioresource Technology Reports 2020, 11, 100525. [Google Scholar] [CrossRef]
- Spyridonidis, A.; Vasiliadou, I.A.; Stathopoulou, P.; Tsiamis, A.; Tsiamis, G.; Stamatelatou, K. Enrichment of Microbial Consortium with Hydrogenotrophic Methanogens for Biological Biogas Upgrade to Biomethane in a Bubble Reactor under Mesophilic Conditions. Sustainability 2023, 15, 15247. [Google Scholar] [CrossRef]
- Francisco López, A.; Lago Rodríguez, T.; Faraji Abdolmaleki, S.; Galera Martínez, M.; Bello Bugallo, P.M. From Biogas to Biomethane: An In-Depth Review of Upgrading Technologies That Enhance Sustainability and Reduce Greenhouse Gas Emissions. Applied Sciences 2024, 14, 2342. [Google Scholar] [CrossRef]
- Kozak, M.; Köroğlu, E.O.; Cirik, K.; Zaimoğlu, Z. Evaluation of Ex-Situ Hydrogen Biomethanation at Mesophilic and Thermophilic Temperatures. International Journal of Hydrogen Energy 2022, 47, 15434–15441. [Google Scholar] [CrossRef]
- Angelidaki, I.; Treu, L.; Tsapekos, P.; Luo, G.; Campanaro, S.; Wenzel, H.; Kougias, P.G. Biogas Upgrading and Utilization: Current Status and Perspectives. Biotechnology Advances 2018, 36, 452–466. [Google Scholar] [CrossRef]
- Sposob, M.; Wahid, R.; Fischer, K. Ex-Situ Biological CO2 Methanation Using Trickle Bed Reactor: Review and Recent Advances. Rev Environ Sci Biotechnol 2021, 20, 1087–1102. [Google Scholar] [CrossRef]
- Huang, J.-H.; Fan, X.-L.; Li, R.; Sun, M.-T.; Zou, H.; Zhang, Y.-F.; Guo, R.-B.; Fu, S.-F. Biogas Upgrading by Biotrickling Filter: Effects of Temperature and Packing Materials. Chemical Engineering Journal 2024, 481, 148367. [Google Scholar] [CrossRef]
- Sposób, M. Optimization of Ex-Situ Biomethanation Process in Trickle Bed Reactor: The Impact of Slight H2/CO2 Ratio Adjustments and Different Packing Materials. Renewable Energy 2024, 222, 119971. [Google Scholar] [CrossRef]
- Rao, Y.; Chibwe, K.; Mantilla-Calderon, D.; Ling, F.; He, Z. Meta-Analysis of Biogas Upgrading to Renewable Natural Gas through Biological CO2 Conversion. Journal of Cleaner Production 2023, 426, 139128. [Google Scholar] [CrossRef]
- Mares, S.; Moreno-Andrade, I.; Quijano, G. Biological CH4 Production from H2/CO2 Streams: Influence of Trace Metals Concentration on the Hydrogenotrophic Process. Journal of Environmental Chemical Engineering 2023, 11, 109528. [Google Scholar] [CrossRef]
- Luo, G.; Angelidaki, I. Integrated Biogas Upgrading and Hydrogen Utilization in an Anaerobic Reactor Containing Enriched Hydrogenotrophic Methanogenic Culture. Biotech & Bioengineering 2012, 109, 2729–2736. [Google Scholar] [CrossRef]
- Kougias, P.G.; Treu, L.; Benavente, D.P.; Boe, K.; Campanaro, S.; Angelidaki, I. Ex-Situ Biogas Upgrading and Enhancement in Different Reactor Systems. Bioresource Technology 2017, 225, 429–437. [Google Scholar] [CrossRef]
- Ghofrani-Isfahani, P.; Tsapekos, P.; Peprah, M.; Kougias, P.; Zhu, X.; Kovalovszki, A.; Zervas, A.; Zha, X.; Jacobsen, C.S.; Angelidaki, I. Ex-Situ Biogas Upgrading in Thermophilic up-Flow Reactors: The Effect of Different Gas Diffusers and Gas Retention Times. Bioresource Technology 2021, 340, 125694. [Google Scholar] [CrossRef]
- Ebrahimian, F.; De Bernardini, N.; Tsapekos, P.; Treu, L.; Zhu, X.; Campanaro, S.; Karimi, K.; Angelidaki, I. Effect of Pressure on Biomethanation Process and Spatial Stratification of Microbial Communities in Trickle Bed Reactors under Decreasing Gas Retention Time. Bioresource Technology 2022, 361, 127701. [Google Scholar] [CrossRef]
- Kougias, P.G.; Tsapekos, P.; Treu, L.; Kostoula, M.; Campanaro, S.; Lyberatos, G.; Angelidaki, I. Biological CO2 Fixation in Up-Flow Reactors via Exogenous H2 Addition. Journal of Biotechnology 2020, 319, 1–7. [Google Scholar] [CrossRef]
- Ghofrani-Isfahani, P.; Tsapekos, P.; Peprah, M.; Kougias, P.; Zervas, A.; Zhu, X.; Yang, Z.; Jacobsen, C.S.; Angelidaki, I. Ex-Situ Biogas Upgrading in Thermophilic Trickle Bed Reactors Packed with Micro-Porous Packing Materials. Chemosphere 2022, 296, 133987. [Google Scholar] [CrossRef] [PubMed]
- Chatzis, A.; Orellana, E.; Gaspari, M.; Kontogiannopoulos, K.; Treu, L.; Zouboulis, A.; Kougias, P.G. Comparative Study on Packing Materials for Improved Biological Methanation in Trickle Bed Reactors. Bioresource Technology 2023, 385, 129456. [Google Scholar] [CrossRef] [PubMed]
- Spyridonidis, A.; Stamatelatou, K. Comparative Study of Mesophilic Biomethane Production in Ex Situ Trickling Bed and Bubble Reactors. Fermentation 2024, 10, 554. [Google Scholar] [CrossRef]
- Burkhardt, M.; Jordan, I.; Heinrich, S.; Behrens, J.; Ziesche, A.; Busch, G. Long Term and Demand-Oriented Biocatalytic Synthesis of Highly Concentrated Methane in a Trickle Bed Reactor. Applied Energy 2019, 240, 818–826. [Google Scholar] [CrossRef]
- Karyofyllidou, C.; Spyridonidis, A.; Diamantis, V.; Galiatsatos, I.; Tsiamis, G.; Stathopoulou, P.; Kosmadakis, I.; Eftaxias, A.; Stamatelatou, K. Mesophilic Trickle-Bed Reactors for Enhanced Ex Situ Biogas Upgrading at Short Gas Retention Times: Process Performance and Microbial Insights. Fermentation 2026, 12, 69. [Google Scholar] [CrossRef]
- Jensen, M.B.; Poulsen, S.; Jensen, B.; Feilberg, A.; Kofoed, M.V.W. Selecting Carrier Material for Efficient Biomethanation of Industrial Biogas-CO2 in a Trickle-Bed Reactor. Journal of CO2 Utilization 2021, 51, 101611. [Google Scholar] [CrossRef]
- Trejo-Aguilar, G.; Revah, S.; Lobo-Oehmichen, R. Hydrodynamic Characterization of a Trickle Bed Air Biofilter. Chemical Engineering Journal 2005, 113, 145–152. [Google Scholar] [CrossRef]
- Burkhardt, M.; Koschack, T.; Busch, G. Biocatalytic Methanation of Hydrogen and Carbon Dioxide in an Anaerobic Three-Phase System. Bioresource Technology 2015, 178, 330–333. [Google Scholar] [CrossRef]
- Ullrich, T.; Lindner, J.; Bär, K.; Mörs, F.; Graf, F.; Lemmer, A. Influence of Operating Pressure on the Biological Hydrogen Methanation in Trickle-Bed Reactors. Bioresource Technology 2018, 247, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Rittmann, B.E.; McCarty, P.L. Environmental Biotechnology: Principles and Applications; McGraw-Hill Education: New York, N.Y., 2018; ISBN 978-1-260-44059-1. [Google Scholar]
- Saini, S.; Tewari, S.; Dwivedi, J.; Sharma, V. Biofilm-Mediated Wastewater Treatment: A Comprehensive Review. Mater. Adv. 2023, 4, 1415–1443. [Google Scholar] [CrossRef]
- Zhao, T.; Chen, Y.; Yu, Q.; Shi, D.; Chai, H.; Li, L.; Ai, H.; Gu, L.; He, Q. Enhancement of Performance and Stability of Anaerobic Co-Digestion of Waste Activated Sludge and Kitchen Waste by Using Bentonite. PLoS ONE 2019, 14, e0218856. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.-M.; Sung, S.; Kang, S.; Kim, M.-S.; Kim, D.-H. Enrichment of Hydrogenotrophic Methanogens by Means of Gas Recycle and Its Application in Biogas Upgrading. Energy 2017, 135, 294–302. [Google Scholar] [CrossRef]
- Hu, F.; Zhang, S.; Liu, S.; Wan, L.; Gong, G.; Hu, T.; Wang, X.; Xu, L.; Xu, G.; Hu, Y. Alleviating Acid Inhibition via Bentonite Supplementation during Acidulated Swine Manure Anaerobic Digestion: Performance Enhancement and Microbial Mechanism Analysis. Chemosphere 2023, 313, 137577. [Google Scholar] [CrossRef]
- Tzenos, C.A.; Kalamaras, S.D.; Economou, E.-A.; Romanos, G.Em.; Veziri, C.M.; Mitsopoulos, A.; Menexes, G.C.; Sfetsas, T.; Kotsopoulos, T.A. The Multifunctional Effect of Porous Additives on the Alleviation of Ammonia and Sulfate Co-Inhibition in Anaerobic Digestion. Sustainability 2023, 15, 9994. [Google Scholar] [CrossRef]
- Odom, I.E. Smectite Clay Minerals: Properties and Uses. Phil. Trans. R. Soc. Lond. A 1984, 391–409. [Google Scholar] [CrossRef]
- Heller, H.; Keren, R. Rheology of Na-Rich Montmorillonite Suspension as Affected by Electrolyte Concentration and Shear Rate. Clays and clay miner. 2001, 49, 286–291. [Google Scholar] [CrossRef]
- Du, W.; Yang, Y.; Hu, L.; Chang, B.; Cao, G.; Nasir, M.; Lv, J. Combined Determination Analysis of Surface Properties Evolution towards Bentonite by pH Treatments. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2021, 626, 127067. [Google Scholar] [CrossRef]
- Zhang, S.; Tan, D.; Zhu, H.; Pei, H.; Shi, B. Rheological Behaviors of Na-Montmorillonite Considering Particle Interactions: A Molecular Dynamics Study. Journal of Rock Mechanics and Geotechnical Engineering 2025, 17, 4657–4671. [Google Scholar] [CrossRef]
- Alfaro, N.; Fdz-Polanco, M.; Fdz-Polanco, F.; Díaz, I. Evaluation of Process Performance, Energy Consumption and Microbiota Characterization in a Ceramic Membrane Bioreactor for Ex-Situ Biomethanation of H2 and CO2. Bioresource Technology 2018, 258, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Rachbauer, L.; Voitl, G.; Bochmann, G.; Fuchs, W. Biological Biogas Upgrading Capacity of a Hydrogenotrophic Community in a Trickle-Bed Reactor. Applied Energy 2016, 180, 483–490. [Google Scholar] [CrossRef]
- Lee, J.C.; Kim, J.H.; Chang, W.S.; Pak, D. Biological Conversion of CO2 to CH4 Using Hydrogenotrophic Methanogen in a Fixed Bed Reactor. J. Chem. Technol. Biotechnol. 2012, 87, 844–847. [Google Scholar] [CrossRef]
- Kamravamanesh, D.; Rinta Kanto, J.M.; Ali-Loytty, H.; Myllärinen, A.; Saalasti, M.; Rintala, J.; Kokko, M. Ex-Situ Biological Hydrogen Methanation in Trickle Bed Reactors: Integration into Biogas Production Facilities. Chemical Engineering Science 2023, 269, 118498. [Google Scholar] [CrossRef]
- Tsapekos, P.; Treu, L.; Campanaro, S.; Centurion, V.B.; Zhu, X.; Peprah, M.; Zhang, Z.; Kougias, P.G.; Angelidaki, I. Pilot-Scale Biomethanation in a Trickle Bed Reactor: Process Performance and Microbiome Functional Reconstruction. Energy Conversion and Management 2021, 244, 114491. [Google Scholar] [CrossRef]
- Thapa, A.; Park, J.-G.; Jun, H.-B. Enhanced Ex-Situ Biomethanation of Hydrogen and Carbon Dioxide in a Trickling Filter Bed Reactor. Biochemical Engineering Journal 2022, 179, 108311. [Google Scholar] [CrossRef]
- Bassani, I.; Kougias, P.G.; Treu, L.; Porté, H.; Campanaro, S.; Angelidaki, I. Optimization of Hydrogen Dispersion in Thermophilic Up-Flow Reactors for Ex Situ Biogas Upgrading. Bioresource Technology 2017, 234, 310–319. [Google Scholar] [CrossRef]
- Jønson, B.D.; Tsapekos, P.; Tahir Ashraf, M.; Jeppesen, M.; Ejbye Schmidt, J.; Bastidas-Oyanedel, J.-R. Pilot-Scale Study of Biomethanation in Biological Trickle Bed Reactors Converting Impure CO2 from a Full-Scale Biogas Plant. Bioresource Technology 2022, 365, 128160. [Google Scholar] [CrossRef]
- Sieborg, M.U.; Jønson, B.D.; Ashraf, M.T.; Yde, L.; Triolo, J.M. Biomethanation in a Thermophilic Biotrickling Filter Using Cattle Manure as Nutrient Media. Bioresource Technology Reports 2020, 9, 100391. [Google Scholar] [CrossRef]
- Porté, H.; Kougias, P.G.; Alfaro, N.; Treu, L.; Campanaro, S.; Angelidaki, I. Process Performance and Microbial Community Structure in Thermophilic Trickling Biofilter Reactors for Biogas Upgrading. Science of The Total Environment 2019, 655, 529–538. [Google Scholar] [CrossRef]
- Strübing, D.; Huber, B.; Lebuhn, M.; Drewes, J.E.; Koch, K. High Performance Biological Methanation in a Thermophilic Anaerobic Trickle Bed Reactor. Bioresource Technology 2017, 245, 1176–1183. [Google Scholar] [CrossRef]
- Vishnyakova, A.; Popova, N.; Artemiev, G.; Botchkova, E.; Litti, Y.; Safonov, A. Effect of Mineral Carriers on Biofilm Formation and Nitrogen Removal Activity by an Indigenous Anammox Community from Cold Groundwater Ecosystem Alone and Bioaugmented with Biomass from a “Warm” Anammox Reactor. Biology 2022, 11, 1421. [Google Scholar] [CrossRef]
- Wastewater Engineering: Treatment and Resource Recovery, Fifth Edition.; Tchobanoglous, G., Stensel, D.H., Tsuchihashi, R., Burton, F., Abu-Orf, M., Bowden, G., Pfrang, W., Metcalf, *!!! REPLACE !!!*, Eddy, Inc, Albert Einstein College of Medicine, Eds.; McGraw-Hill Education: New York, 2014; ISBN 978-0-07-340118-8. [Google Scholar]
| Parameters | Before bentonite addition | After bentonite addition |
|---|---|---|
| pH | 7.78 | 7.94 |
| Cond (mS cm−1 @25oC) | 17.98 | 16.74 |
| TS (%) | 3.29 | 21.93 |
| VS (%) | 2.61 | 2.30 |
| Phase | I | II | III | IV | V | VI | VII | VIII | IX | X | XI | XII | XIII |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GRT (h) | 23.02 | 16.44 | 10.46 | 16.44 | 10.46 | 7.19 | 5.48 | 3.97 | 2.81 | 1.98 | 1.49 | 1.19 | 0.99 |
| HLR (LH2 LR−1d−1) | 0.62 | 0.86 | 1.36 | 0.86 | 1.36 | 1.97 | 2.59 | 3.58 | 5.06 | 7.16 | 9.50 | 11.97 | 14.32 |
| CH4 (%) | 97.14 (0.06) | 97.14 (0.53) | 92.95 (3.48) | 97.31 (0.29) | 97.71 (0.15) | 98.43 (0.31) | 99.03 (0.07) | 99.22 (0.03) | 99.33 (0.11) | 98.58 (0.59) | 96.25 (0.05) | 94.50 (0.13) | 90.24 (0.33) |
| CH4R (%) | 92.24 (0.36) |
94.63 (0.15) | 91.15 (1.01) | 65.01 (7.05) | |||||||||
| H2 (%) | 0.25 (0.11) | 0.47 (0.36) | 4.85 (2.81) | 0.07 (0.10) | 0.02 (0.00) | 0.03 (0.05) | 0.00 (0.00) | 0.01 (0.01) | 0.06 (0.08) | 0.88 (0.51) | 2.67 (0.04) | 4.14 (0.11) | 7.67 (0.27) |
| H2R (%) | 4.92 (0.34) | 2.38 (0.16) | 4.93 (1.08) | 27.1 (5.61) | |||||||||
| CO2 (%) | 2.61 (0.13) | 2.39 (0.18) | 2.20 (0.68) | 2.62 (0.18) | 2.28 (0.15) | 1.54 (0.27) | 0.97 (0.07) | 0.77 (0.03) | 0.62 (0.03) | 0.54 (0.08) | 1.08 (0.01) | 1.36 (0.02) | 2.09 (0.06) |
| CO2R (%) | 2.84 (0.08) | 2.99 (0.06) | 3.92 (0.31) | 7.88 (1.46) | |||||||||
| ηH2 (%) | 99.81 (0.08) | 99.65 (0.27) | 97.17 (1.65) | 99.95 (0.08) | 99.99 (0.00) | 99.98 (0.03) | 100.0 (0.00) | 100.0 (0.00) | 99.96 (0.06) | 99.43 (0.32) | 98.26 (0.01) | 97.49 (0.12) | 95.49 (0.13) |
| ηH2R (%) | 94.81 (1.90) | 98.53 (0.2) | 96.53 (0.88) | 77.57 (5.96) | |||||||||
| ηCO2 (%) | 92.34 (0.92) | 93.33 (0.38) | 95.12 (1.50) | 92.89 (0.82) | 93.44 (0.58) | 95.85 (0.65) | 97.52 (0.17) | 97.99 (0.16 | 98.39 (0.06) | 98.65 (0.16) | 97.34 (0.01) | 96.88 (0.12) | 95.37 (0.11) |
| ηCO2R (%) | 92.49 (1.04) | 93.11 (1.37) | 89.83 (0.7) | 75.88 (5.9) | |||||||||
|
MPR (LCH4 LR−1d−1) |
0.48 (0.04) | 0.62 (0.03) | 0.74 (0.06) | 0.60 (0.03) | 1.01 (0.02) | 1.39 (0.06) | 1.75 (0.02) | 2.44 (0.11) | 3.48 (0.06) | 4.66 (0.16) | 5.95 (0.06) | 6.86 (0.14) | 7.60 (0.08) |
| MPRR | 0.37 (0) | 0.57 (0.03) | 0.73 (0.02) | 1.07 (0.06) | |||||||||
|
Net MPR (LCH4 LR−1d−1) |
0.23 (0.04) | 0.28 (0.03) | 0.20 (0.06) | 0.26 (0.03) | 0.47 (0.02) | 0.61 (0.06) | 0.71 (0.02) | 1.02 (0.11) | 1.46 (0.06) | 1.81 (0.16) | 2.17 (0.06) | 2.10 (0.14) | 1.91 (0.08) |
|
Net MPRR (LCH4 LR−1d−1) |
0.08 (0.04) | 0.21 (0.03) | 0.27 (0.02) | 0.27 (0.06) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




