The presence of vanadium, even at low contents, has a positive effect on highly alloyed Cr-Mo steels, because in the process of solidification, the V
6C
5 carbide crystals are formed from the melt, blocking the further growth of primary austenite dendrites, thus enabling the creation of a fine-grained structure [
19,
20,
21]. Vanadium, as a distinctly carbide-forming element, not only forms grains of V
6C
5 carbide, but affects the morphology of M
7C
3 carbide, as well. It also reduces the stability of the austenite and generally refines the structure of the metal matrix. Similarly to iron, vanadium replaces chromium in the lattice of M
7C
3 carbide, which leads to an increase in the chromium content in the metal matrix and to a higher degree of hardenability of austenite.
3.1. Microstructural Analysis
The microstructure of a sample I, containing 1.8% C and 0.5% V is shown in
Figure 1, where one can see the martensite crystals and a carbide network around the grains of the metal matrix, formed by the eutectic carbide M
7C
3. It has precipitated in the form of lamellae, plates and rosettes. The M
7C
3 carbide, in the two-dimensional space appears in the form of strips. A large number of those strips are usually grouped into bundles, having the same spatial orientation, thus in the two-dimensional space those bundles of strips appear resembling the lamellae. In
Figure 1, the points where the EDS analysis was performed are marked from 1 to 5. For each of those points the measurements were repeated three times, and
Table 3 provides the mean value of those three measurements. Maximum experimental deviations were ± 2.0%.
Chemical analysis was carried out for points 1, 2 and 5 of the metal matrix, and for points 3 and 4 of the carbide network. The analysis of the metal matrix indicates that it is dominated by martensite and residual austenite, but the increased contents of carbon and vanadium indicate the presence of finely dispersed vanadium carbides in the metal matrix.
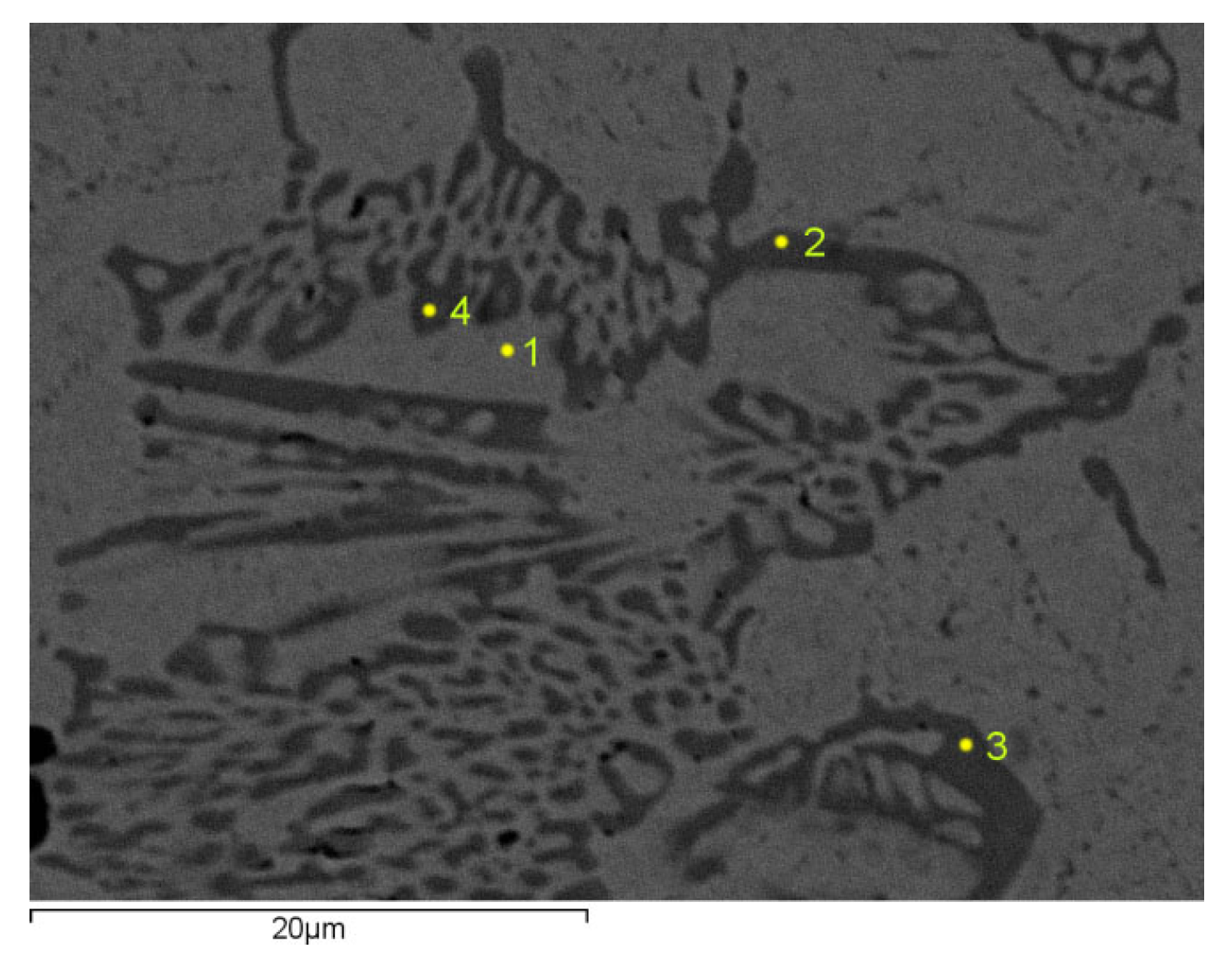
Figure 2 shows the microstructure of sample number II with 1.8% C and 1% V. The chemical analysis of the metal matrix was performed at point 1, and at points 2, 3 and 4, a carbide analysis was performed.
Table 3 shows the chemical composition of the tested phases of sample I, and
Table 4 shows the chemical analysis of the examined phases of sample II.
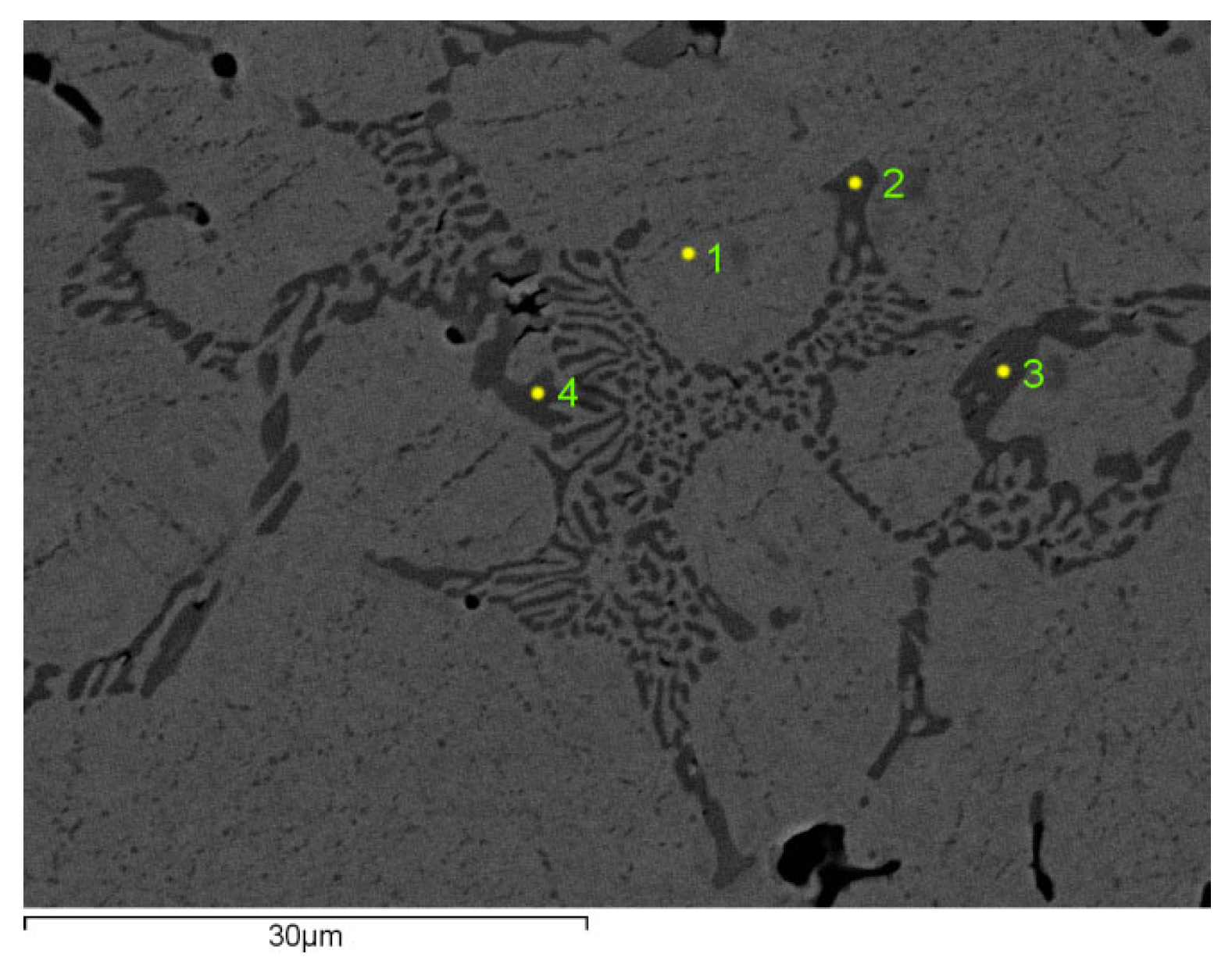
Figure 3 shows the microstructure of sample number III and
Figure 4 of sample number IV, with 1.8% C and 2% V, respectively. For sample number III, points 2, 3 and 4 in the figure indicate the places where the chemical analysis of the carbide was performed, and point 1 shows the place where the chemical analysis of the metal matrix was performed. Chemical composition of the tested phases of sample III is presented in
Table 4.
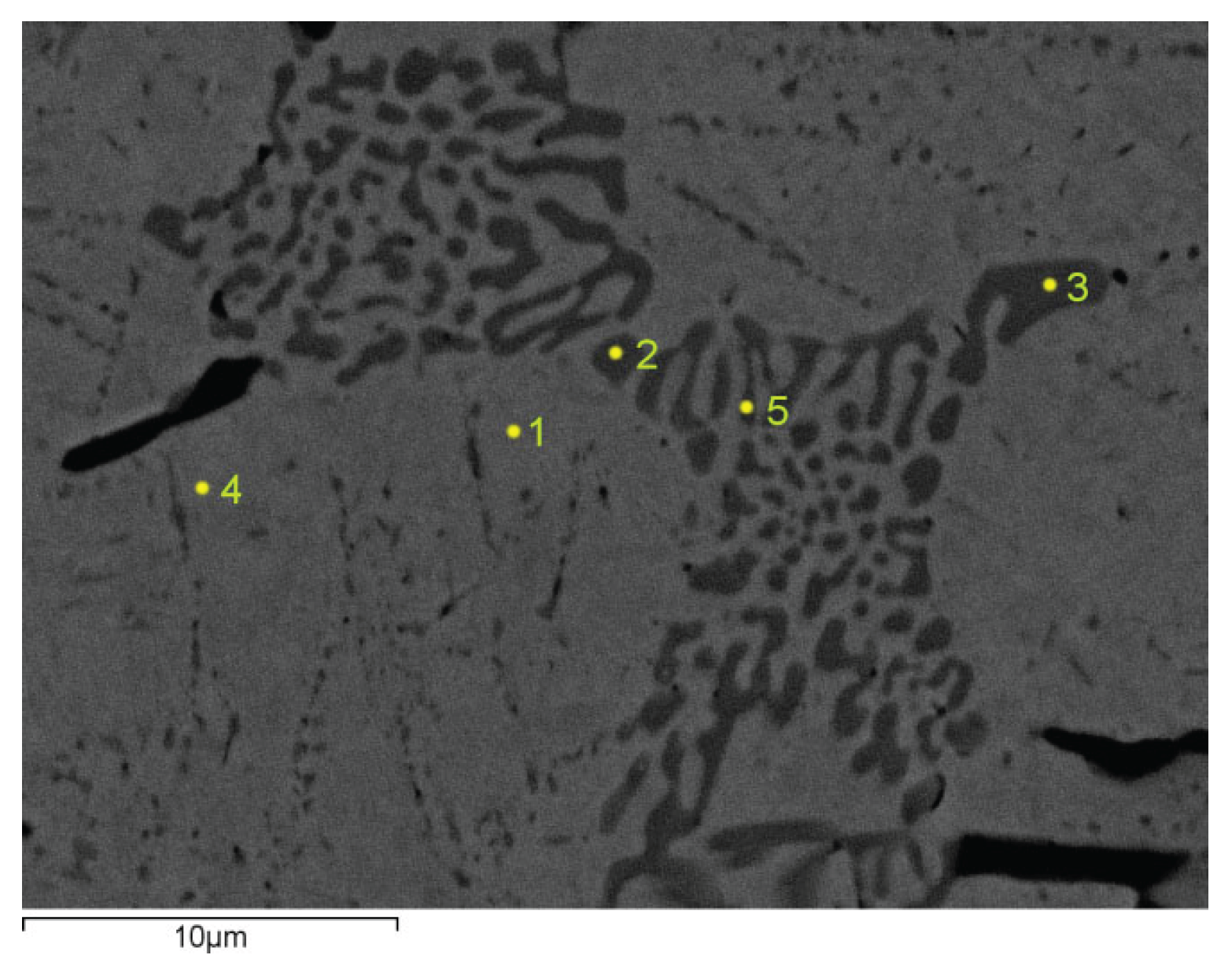
Figure 4 shows the microstructure of sample number IV with 1.8% C and 3% V. The points marked with 2, 3 and 5 show the test points on the carbide grid, while the points 1 and 4 show the test points on the metal matrix.
Table 5 shows the chemical analysis of the examined phases of sample IV.
Table 5.
Chemical composition of the tested phases of sample III, wt %.
Table 5.
Chemical composition of the tested phases of sample III, wt %.
| Point # |
C |
Si |
V |
Cr |
Fe |
Ni |
Mo |
| 1 |
3.56 |
0.54 |
2.10 |
10.81 |
81.43 |
0.43 |
1.14 |
| 2 |
14.88 |
- |
12.16 |
37.61 |
32.25 |
- |
3.10 |
| 3 |
15.42 |
- |
13.26 |
39.58 |
27.71 |
- |
4.02 |
| 4 |
14.78 |
- |
12.91 |
39.77 |
29.07 |
- |
3.48 |
Table 6.
Chemical composition of the tested phases of sample IV, wt %.
Table 6.
Chemical composition of the tested phases of sample IV, wt %.
| Point # |
C |
Si |
V |
Cr |
Fe |
Ni |
Mo |
| 1 |
8.77 |
0.55 |
1.35 |
9.78 |
78.48 |
0.43 |
0.64 |
| 2 |
15.43 |
- |
9.95 |
33.23 |
38.74 |
- |
2.65 |
| 3 |
15.89 |
- |
12.23 |
38.50 |
30.63 |
- |
2.75 |
| 4 |
3.08 |
0.55 |
1.62 |
10.57 |
83.40 |
- |
0.78 |
| 5 |
5.50 |
0.38 |
5.41 |
23.27 |
64.05 |
- |
1.38 |
As with the previous examples, there is a significantly higher content of chromium, vanadium and iron in the carbide network compared to the amount of these elements in the metal matrix. Based on the examined microstructures, it can be unambiguously concluded that it is M7C3 carbide.
The black spots and fields near the carbide mesh that can be observed in the microstructure photographs, shown in
Figures 1 to 4, represent the pits that were created during the grinding and polishing of the samples. Due to the higher hardness of the carbide, compared to the metal matrix, the metal filings during the grinding created indentations that can be seen as black spots in the photos.
Figure 5 shows the microstructure of sample IV with maps of the distribution of elements present in the tested steel samples, Fe, C, Cr, Mo, Ni, V and Si. The carbide network and the metal matrix can be seen from the microstructure shown. The EDS analysis of the distribution map shows a significantly higher content of chromium, vanadium and carbon in the carbide network compared to the amount of these elements in the metal matrix. Iron can be observed to the greatest extent in the metal matrix, but the presence of other elements can also be found in smaller concentrations. With an increase in the percentage of vanadium in the steel, 0.5-3%, the radial arrangement of the carbides becomes increasingly dominant. Vanadium forms carbide V
6C
5 with carbon.
Vanadium, similar to molybdenum, is partly distributed between the phases present in the steel. In the carbide network, vanadium with chromium forms the CrV carbide. With the increase of the vanadium content in the steel up to 3%, there is also the precipitation of pure vanadium in the carbide network.
Chromium reacts with carbon to form hard carbides of the Cr7C3 and Cr23C6 types, which affect the structure of the steel’s matrix. Carbides of the type Cr7C3 and Cr23C6 can also contain iron, so that the interstitial phases (Cr,Fe)7C3 and (Cr,Fe)23C6 are formed. In the highly alloyed Cr-Mo steels, molybdenum is distributed between the carbide and the metal matrix.
In addition to SEM-EDS analysis, the samples were examined by XRD analysis. The diffractometric analysis was carried out to determine the phases present in the examined samples.
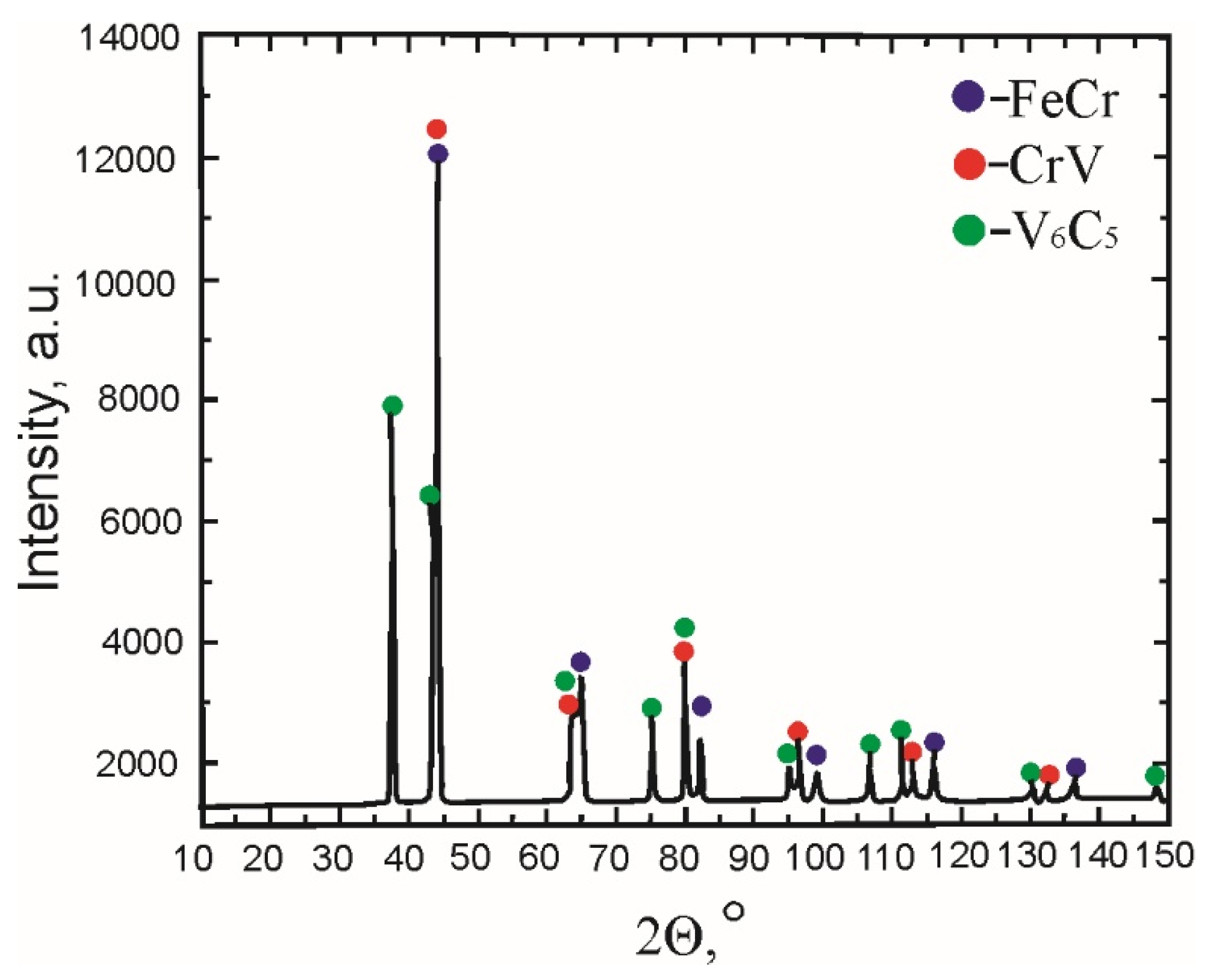
Figure 6 shows the XRD diffractogram of tested sample IV (3% V). The identified phases are: Fe-Cr (blue circles), CrV (red circles) and V
6C
5 (green circles).
Table 7 presents the results obtained by XRD analysis.
By applying the Rietveld method of the peak refinement, three phases were identified in the sample, namely FeCr, CrV and V6C5. The FeCr phase was compared to the literature values (a = b = c = 2.872(3) Å). The obtained lattice parameters for the FeCr phase were a = b = c = 2.8654(3) Å. The disorder in the lattice parameters of the detected FeCr phase originates from the elements Mo, Mn, Si and Ni, which were also detected in this phase in a small percentage.
The second phase determined in sample IV is CrV with cubic crystal lattice and space group Im m, with lattice parameters a = b = c = 2.918986(1) Å. The mentioned phase was compared to the literature values of the lattice parameters (a = b = c = 2.919 Å); obviously the lattice parameters match each other.
The third phase determined in sample IV (3% V) is V6C5 with trigonal crystal lattice and space group P3112. The lattice parameters of V6C5 were compared to the literature values (a = b = 5.09 Å and c =14.4 Å). The experimentally determined lattice parameters of sample IV coincide with those values.
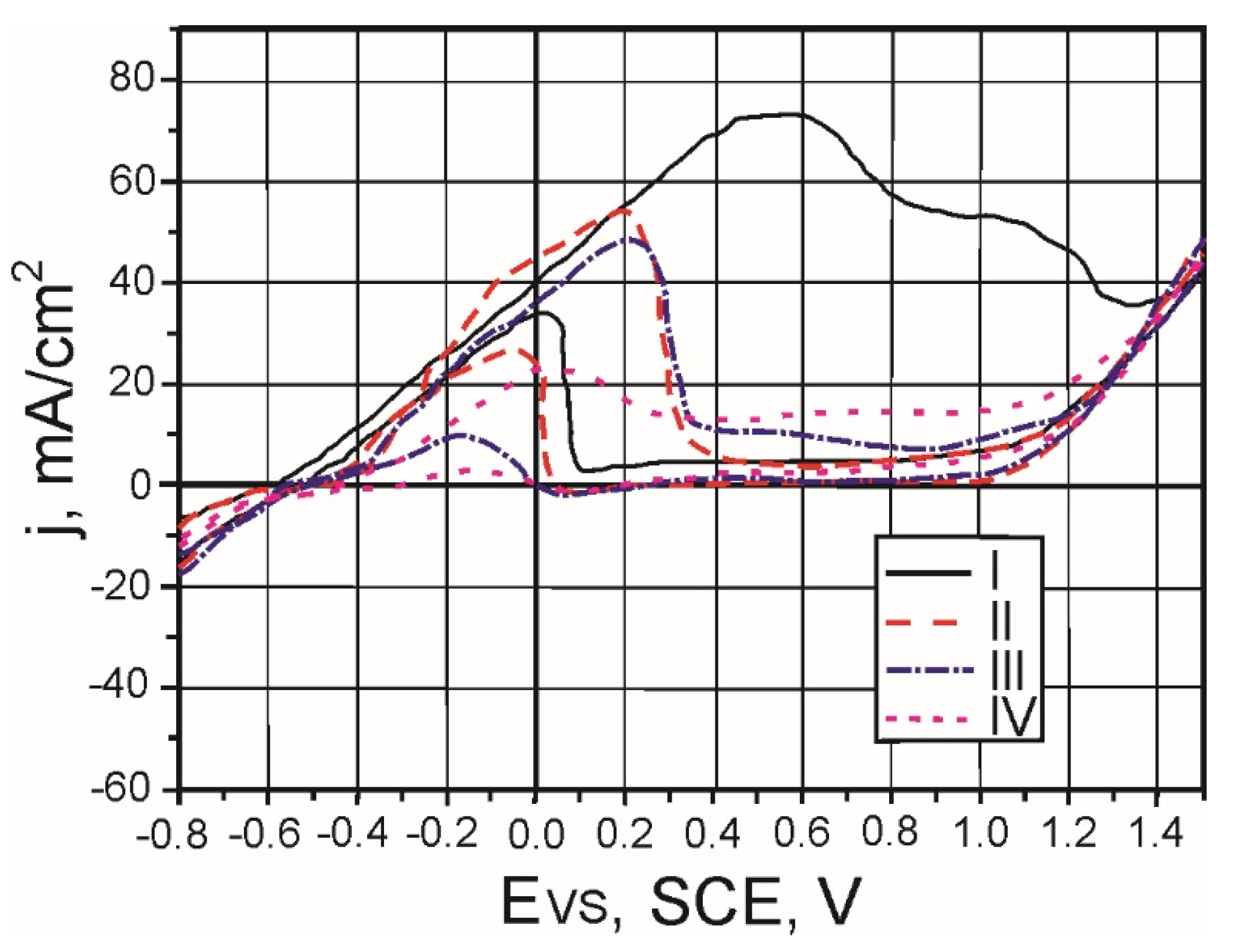
3.2. Determination of Friction Coefficient and Wear Rate
The friction coefficient was determined as the average value along the sliding path, i.e., time of contact.
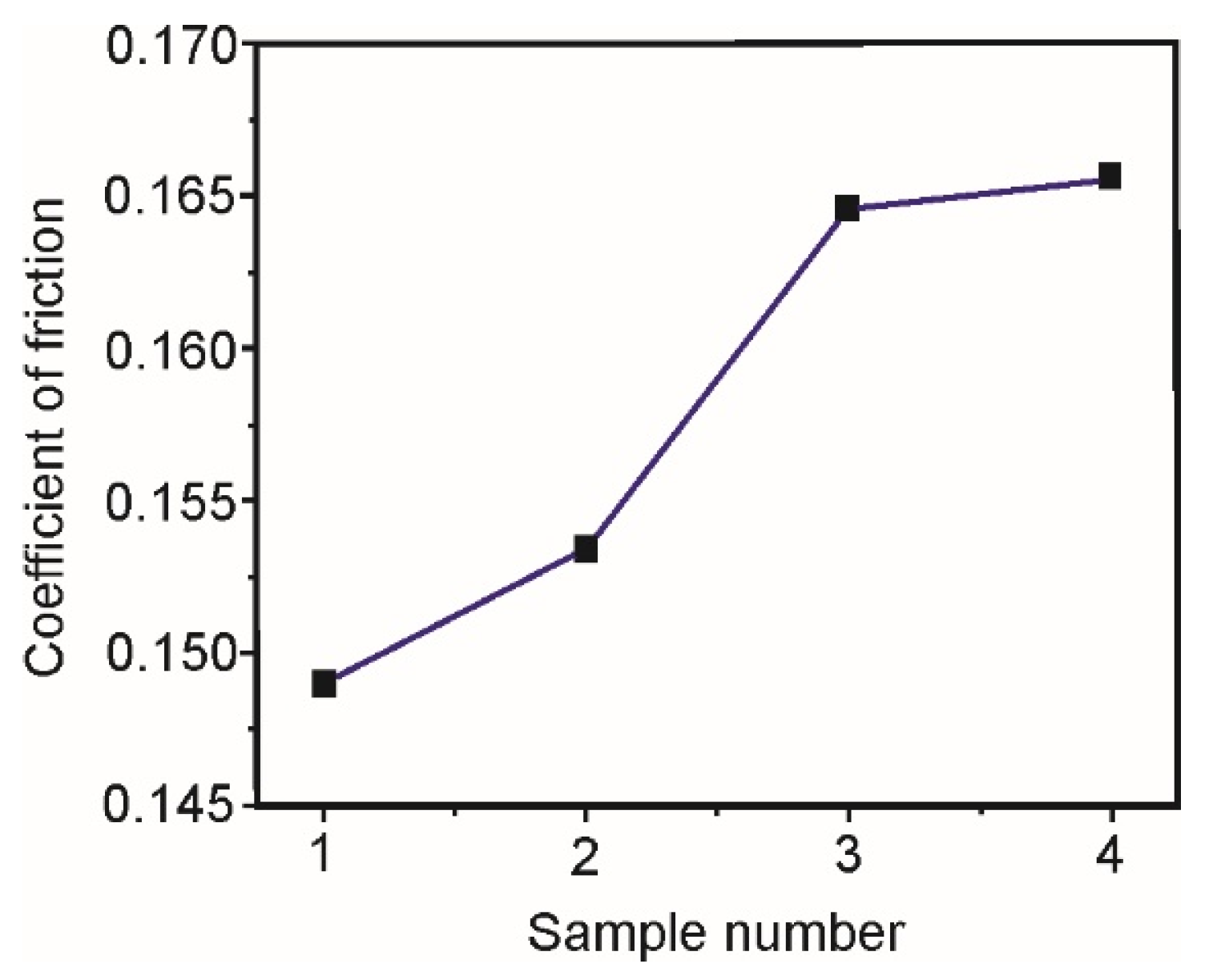
Table 8 shows values of the coefficients of friction of five measurements, for all the samples. In addition to the mean values of the friction coefficients, for each sample are shown values of the standard deviation, which are also shown on diagram in
Figure 7.
From the diagram in
Figure 7 one can see that the coefficient of friction increases linearly with the increase of the vanadium content in the samples. The highest coefficient of friction has sample IV with 3% V.
During the experimental testing of the wear process, the roughness of the contact surfaces, as well as the wear of the ball of the tribometer device, were neglected. That is why the hardness of the ball material should be higher than of the tested material. Regardless of the fulfillment of this condition, when the hard materials are tested, ball wear is inevitable. In this particular case, the preliminary tests showed that the ball made of Al2O3, sapphire and hard metal, wears during the test. After analyzing the wear of the ball of these three materials, it was concluded that the tests should continue with a ball made of hard metal, because it has the most favorable surface geometry, i.e., the smallest roughness. Dimples were observed on the surface of the balls made of Al2O3 and sapphire, which were the result of wear. The dimples’ diameter was up to 10 μm. The existence of dimples on the ball allows accumulation of wear products and formation of stickers on the ball itself. When tested with a hard metal ball, there were no stickers on the ball’s surface. Those stickers would affect both the value of the coefficient of friction and the value of wear.
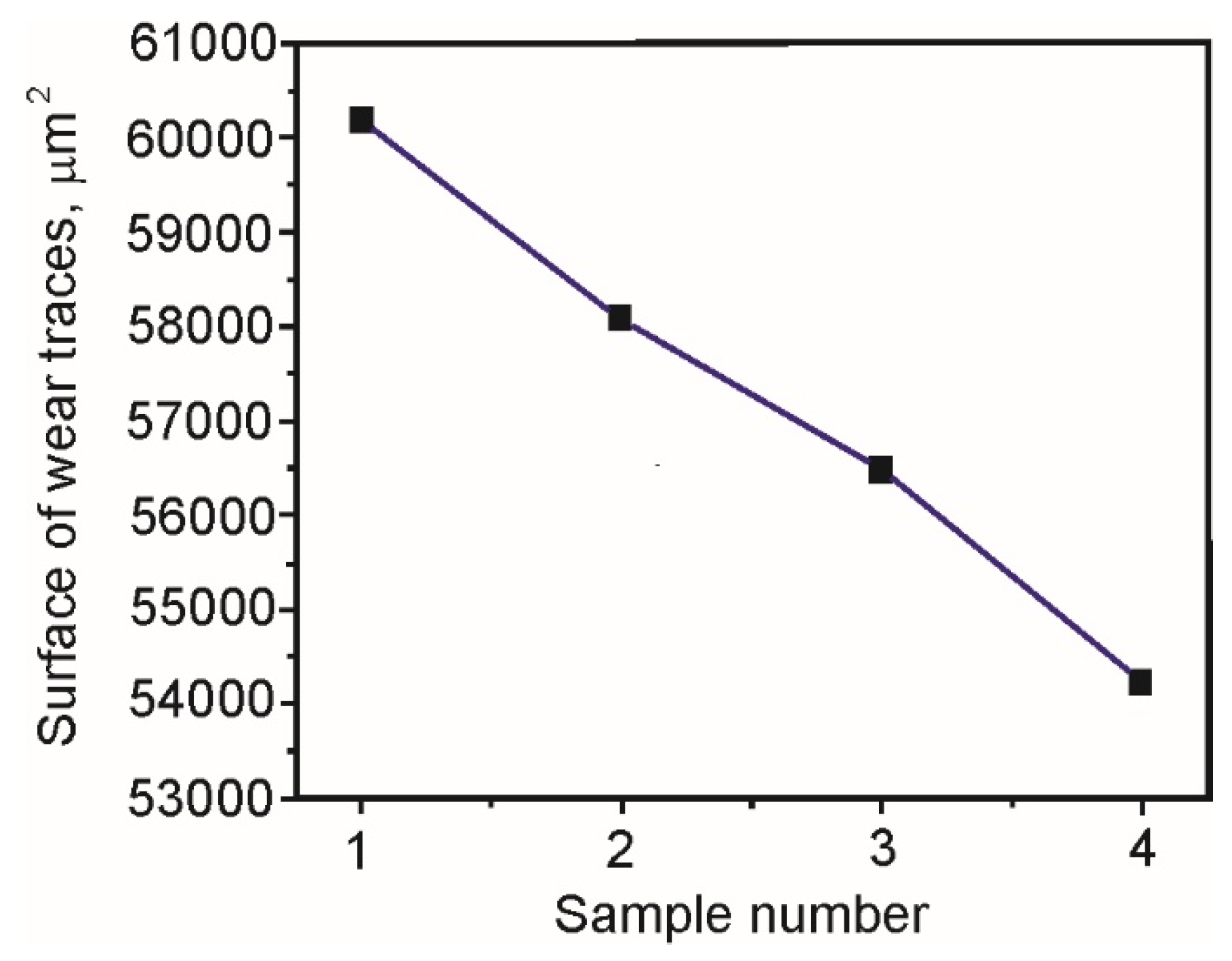
Table 9 presents the measured values of surfaces and lengths of the resulting wear traces, while
Figure 8 shows the average values of wear traces’ surfaces for all four samples.
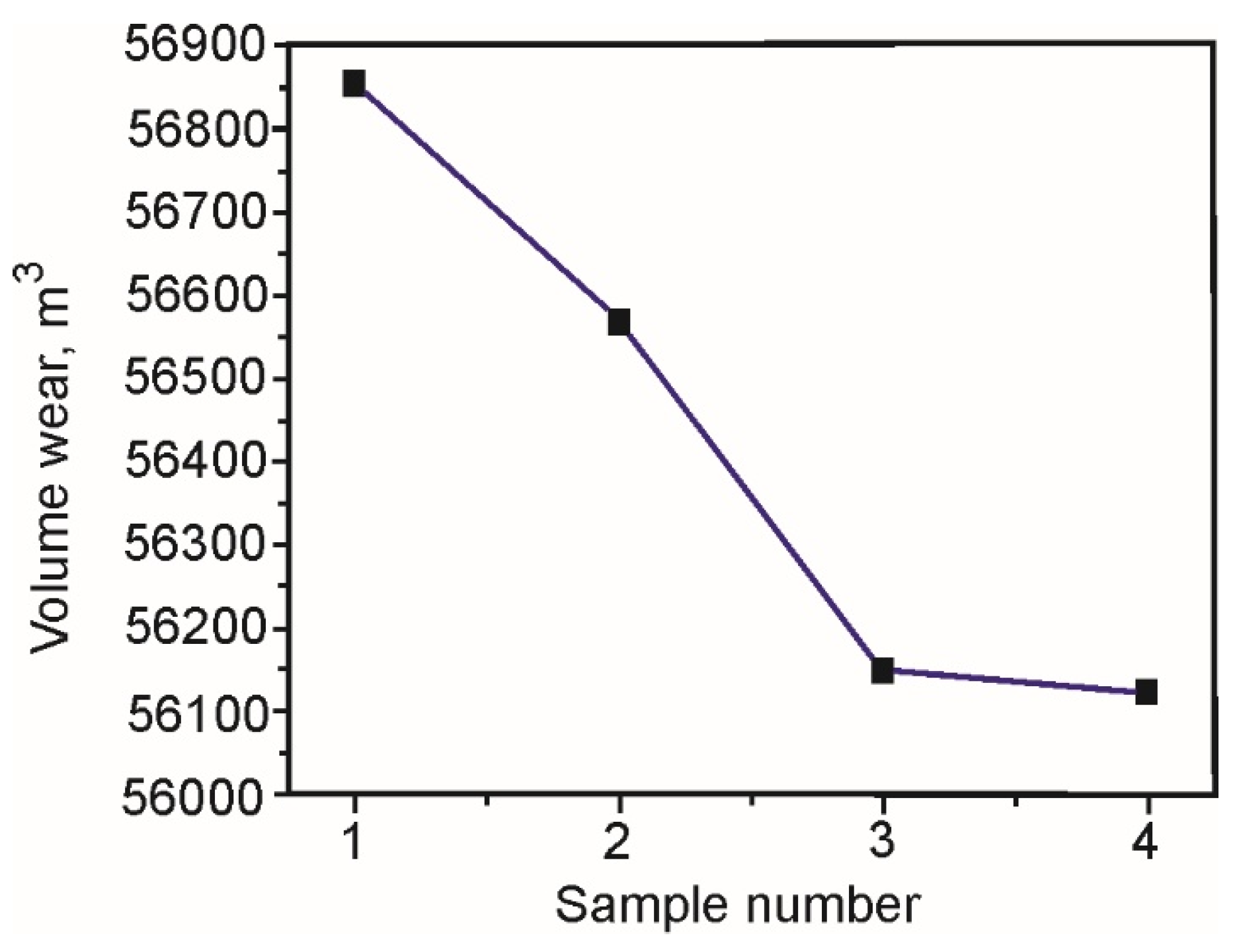
Based on those measured values, and knowing the ball’s diameter, one can calculate the volume of the worn material, which is presented in
Table 10, while
Figure 9 shows the diagram of the wear volume average values, for all four samples.
Based on the obtained wear volume values, it can be observed that the wear volume of sample IV is the smallest and that the deviations in the measured values are the smallest. The wear mechanism of all the tested samples is similar, which is indicated by the relatively small oscillations in the friction coefficients’ values.
On the photographs of the wear traces, at some places on the contact surface of the tested material, the presence of a tribological layer can be seen,
Figure 10.
The resulting tribological layer is a mixture of the material of the ball, the tested material and their oxides. Due to the temperature generation in the thin contact layer, the wear products are joining, thus forming a kind of protective layer on the surface of the tested material.
The parallel marks observed around these zones clearly indicate abrasive wear. The assumption about the existence of ball material in the structure was confirmed by presence of the ball wear, clearly visible in the given images of ball appearance after the testing,
Figure 11.
Table 10.
Results of measuring the hardness of alloy steel II.
Table 10.
Results of measuring the hardness of alloy steel II.
| Phase |
Measurement point # |
Average value |
| 1 |
2 |
3 |
4 |
|
| Carbide network |
1714.75 |
1907.89 |
1657.91 |
2056.83 |
1834.34 |
| Metal matrix |
1408.31 |
1550.95 |
1410.79 |
1423.67 |
1448.43 |
The hardness test was performed at several points in the carbide mesh and the metal matrix so that the mean value was taken as the representative one. Hardness was measured according to the standard procedure using the Vickers method on sample II with 1 wt.% V. The difference in the hardness of the tested samples is insignificant, and for that reason there was no need to determine the hardness of all other samples. The obtained results are shown in
Table 10.
From the presented data can be seen that the carbide network made by vanadium carbides has a higher hardness than the metal matrix itself.