1. Introduction
Endometrial cancer is the most common gynecologic malignancy in developed countries and is primarily managed with surgical staging [
1]. Laparoscopic surgery has become the preferred approach compared with laparotomy due to reduced postoperative morbidity, shorter hospital stay, and faster recovery [
2].
Laparoscopic surgery offers particular advantages for patients with endometrial cancer, who frequently present with comorbidities such as obesity and diabetes that adversely affect wound healing [
3,
4]. However, vaginal cuff healing after laparoscopic hysterectomy may be delayed compared with open abdominal hysterectomy. This delay has been attributed to the use of electrocautery during colpotomy and laparoscopic magnification, which may increase thermal tissue injury and result in smaller-than-optimal suture bites [
5].
Complete vaginal cuff healing is essential to prevent local complications, including infection and dehiscence, particularly in patients who require adjuvant radiotherapy [RT]. Current guidelines recommend initiating adjuvant RT within 6 to 8 weeks after hysterectomy, as delays beyond 9 weeks are associated with poorer oncologic outcomes [
6,
7]. Therefore, factors nfluencing early vaginal cuff healing represent a clinically relevant concern in minimally invasive gynecologic oncology.
Immunonutrition (IMN), containing arginine, glutamine, omega-3 fatty acids, and nucleotides, has been shown to modulate immune responses, reduce postoperative inflammation, and decrease infectious complications in patients undergoing major abdominal and oncologic surgery. IMN also promotes collagen synthesis and connective tissue growth, suggesting a potential role in wound healing [
8]. Based on this evidence IMN is recommended by the European Society for Clinical Nutrition and Metabolism [ESPEN] for patients undergoing major gastrointestinal or head and neck surgery. IMNhas also been proposed to support surgical wound healing; however, evidence regarding its role in gynecologic oncology—particularly following laparoscopic surgery—remains limited.
The primary objective of this prospective observational cohort study was to evaluate the effect of postoperative IMNon vaginal cuff healing in well-nourished patients undergoing laparoscopic surgery for endometrial cancer. The secondary objective was to assess postoperative complications occurring within 30 days.
2. Materials and Methods
This prospective observational cohort study was conducted at the Department of Gynecologic Oncology, Adana City Hospital, University of Health Sciences, between January 2024 and October 2025. The study protocol was approved by the institutional ethics committee [approval no. 07/12/2023-2981], and written informed consent was obtained from all participants in accordance with the Declaration of Helsinki, including consent for the publication of anonymized clinical images.
Eligible patients underwent elective laparoscopic surgery for biopsy-proven cancer or endometrial intraepithelial neoplasia. Patients with suspected endometrial pathology without preoperative biopsy were also included. The sample size was determined by consecutively enrolling all patients who underwent laparoscopic surgery at the institution during the study period.
Postoperative oral immune-modulating diets (IMDs) were incorporated into institutional postoperative care and offered to all eligible patients. Patients who received IMDs constituted the immunonutrition (IMN) group, whereas patients who were managed with a standard postoperative diet constituted the control group. Exclusion criteria included chronic immunosuppressive conditions, prior pelvic radiotherapy, severe hepatic or renal disease, conversion to laparotomy, vaginal route cuff closure, withdrawal of consent, or failure to attend scheduled postoperative follow-up.
2.1. Surgery
All procedures were performed by three experienced gynecologic oncologists following a standardized protocol. Surgical staging consisted of laparoscopic hysterectomy with or without bilateral salpingo-oophorectomy, with sentinel lymph node mapping and lymphadenectomy when indicated.
A circular laparoscopic colpotomy was performed using monopolar energy [50 W]. Vaginal cuff closure was completed laparoscopically using the same absorbable barbed suture in a continuous running fashion, with two to three back bites to secure the proximal end. This technique was applied uniformly to minimize procedural variability. Operative time, blood loss, and complications were recorded prospectively. Vaginal cuff length and uterine weight were measured intraoperatively.
2.2. Postoperative İnterventions
All patients in both cohorts were managed according to a standardized Enhanced Recovery After Surgery [ERAS] protocol. The protocol included early mobilization, standardized multimodal analgesia, and routine prophylaxis against thromboembolism and infection.
Measures for surgical site infection prevention were implemented in accordance with ERAS principles and included standardized perioperative antibiotic prophylaxis, strict postoperative glycemic control targeting blood glucose levels below 180 mg/dL, preoperative vaginal preparation with povidone–iodine, meticulous surgical technique, maintenance of normothermia using an under-body forced-air warming device, and early postoperative recovery strategies. All patients were instructed to maintain pelvic rest for six weeks following surgery.
2.3. Enteral Nutritional Supplementation
The IMDs regimen consisted of two commercially available enteral formulas: Impact® and Resource®. Each 237 mL serving of Impact® provided 4.2 g of arginine, 430 mg of nucleotides, and 1.1 g of omega-3 fatty acids [eicosapentaenoic and docosahexaenoic acids], yielding approximately 237 kcal per serving. Each 237 mL serving of Resource® provided 5 g of glutamine and approximately 20 kcal.
Patients in the IMN group consumed one serving of Impact® and three servings of Resource® daily for seven postoperative days [total daily volume 948 mL], starting within 24 hours after surgery. The prescribed supplementation was intended to provide immune-modulating substrates rather than full caloric replacement and was administered in addition to the standard postoperative hospital diet. The control group received standard postoperative diet alone.
2.4. Compliance and Tolerance
Adherence to the immune-modulating diet protocol was assessed during scheduled postoperative follow-up visits. Compliance was defined as the proportion of the prescribed regimen completed during the 7-day intervention period and was calculated based on daily intake diaries and verification by package counts.
Compliance was calculated using the following formula:Compliance [%] = [number of days on which both supplements were consumed / 7] × 100.
Tolerance was evaluated by recording any interruptions or discontinuations of İmmune-modulating diets , together with the documented reasons for non-use, including gastrointestinal adverse effects or palatability-related complaints.
2.5. Outcome Assesments
Baseline patient characteristics and comorbidities, were recorded. Preoperative nutritional status was evaluated using the Nutritional Risk Screening 2002 [NRS-2002] [
8]. Patients with an NRS score of ≥3 were defined as being at nutritional risk. Surgical site infection was assessed according to Centers for Disease Control and Prevention [CDC] criteria [
9]. Wound evaluations were performed daily during hospitalization and at scheduled postoperative follow-up visits for up to 30 days. Infections requiring medical or surgical intervention were considered clinically significant.
2.6. Assessment of Vaginal Cuff Healing
Vaginal cuff healing was assessed at postoperative weeks 4 and 6 by a single blinded surgeon [BAT] using validated criteria [
10,
11]. Complete vaginal cuff healing was defined as complete mucosal approximation without separation, exposed suture material, or granulation tissue. Incomplete healing included any deviation from these criteria. Patients exhibiting incomplete healing at follow-up were reassessed at two-week intervals until complete healing was observed.
Vaginal cuff bleeding was defined as any postoperative bleeding originating from the vaginal cuff, either observed during examination or reported by the patient. Vaginal cuff hematoma was defined as any blood collection at the vaginal cuff detected by transvaginal ultrasonography. Vaginal cuff infection was diagnosed based on clinical signs and symptoms such as purulent vaginal discharge, induration, erythema, or tenderness of the vaginal cuff. Cuff dehiscence was defined as a full-thickness separation of the vaginal epithelium.
2.7. Statistical Analyses
Statistical analyses were performed using IBM SPSS version 26.0 [IBM Corp., Armonk, NY, USA]. Continuous variables were compared using the t-test or Mann–Whitney U test. Categorical variables were analyzed using the chi-square or Fisher’s exact test. Logistic regression analysis was performed to identify factors associated with complete vaginal cuff healing at postoperative week 4. Results are presented as odds ratios with 95% confidence intervals. A p-value <0.05 was considered statistically significant.
3. Results
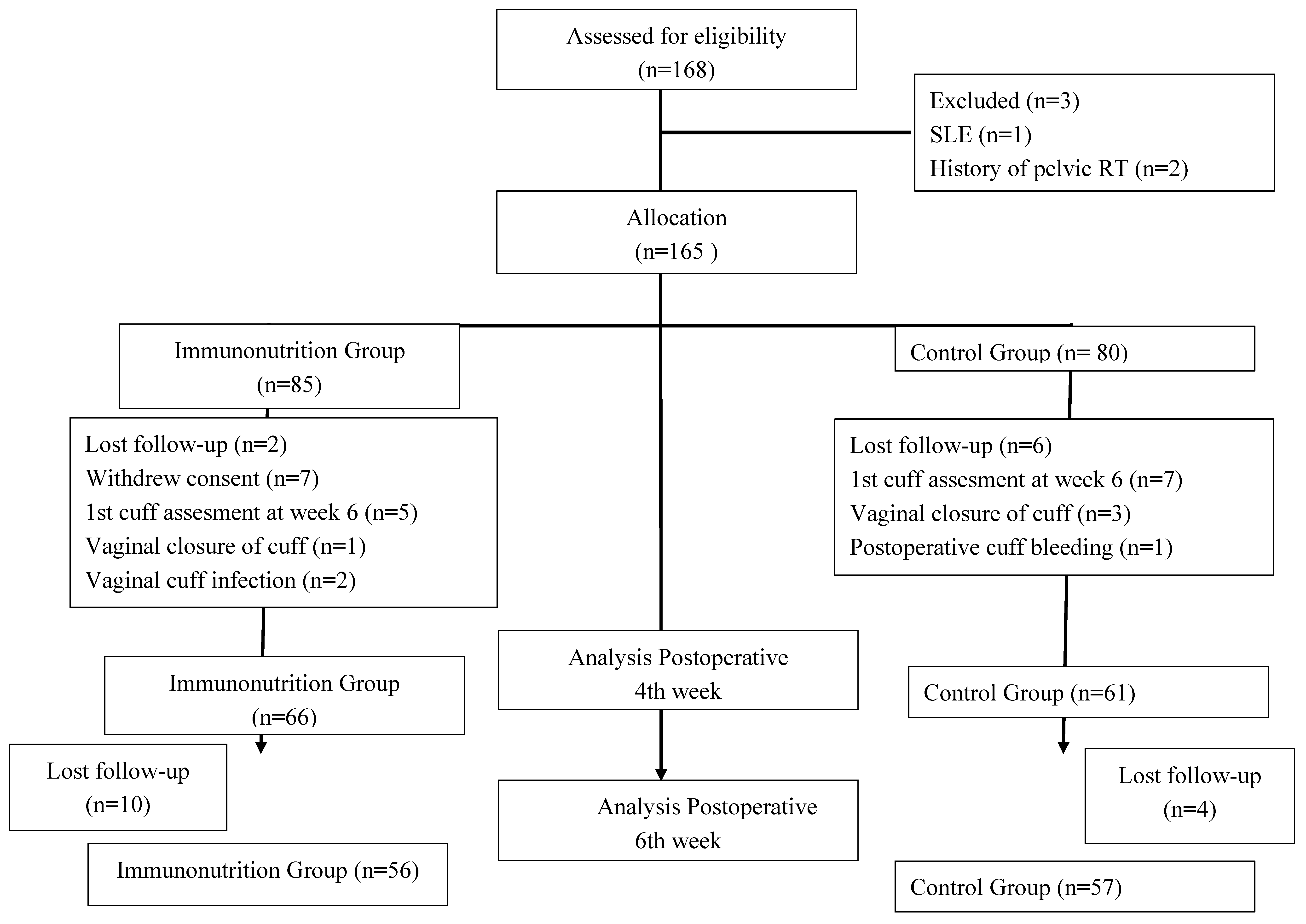
The study flowchart and protocol variations are presented in
Figure 1. Initially, vaginal cuff evaluations were planned for 6th and 8th week after operation. However, the high rate of complete vaginal cuff healing observed at 6th week prompted a protocol modification, and the first postoperative assessment was rescheduled to postoperative 4th week in order to better capture early wound-healing differences. Consequently, 12 patients who had already undergone their first vaginal cuff evaluation at 6th week were excluded from the final analysis to ensure methodological consistency.
After exclusions, 127 patients were included in the 4th week analysis (61 control and 66 immunonutrition). For 6th week evaluation, 113 patients were available. Baseline demographic and clinical characteristics of the study population are summarized in
Table 1.
Baseline characteristics were comparable between groups. There were no significant differences in age, body mass index, smoking status, menopausal status, comorbidities, American Society of Anesthesiologists classification, or nutritional status.
In the control group, seven patients had a history of breast or thyroid cancer, whereas in the IMN group, five patients had previously been treated for breast, thyroid, or colorectal cancer. All prior malignancies were clinically stable and no patient had an active malignancy other than the current endometrial pathology at the time of enrollment.
Surgical and pathological characteristics are presented in
Table 2. Surgical and pathological characteristics were also similar between groups, including operative time, vaginal cuff closure time, cuff length, uterine weight, surgical procedure, histological subtype, and FIGO stage.
3.1. Postoperative Morbidity
Postoperative complications are summarized in
Table 3. No significant differences were observed between the groups regarding febrile morbidity, readmission rates, or surgical site infection. In the IMNgroup, one patient developed pneumonia during postoperative hospitalization and received antibiotic treatment. In the control group, two patients developed urinary tract infections after discharge; one had a urine culture positive for
Pseudomonas aeruginosa, while the other exhibited culture findings consistent with contamination. Both were managed with outpatient antibiotic therapy. Superficial incisional surgical site infection occurred in two patients in each group and were treated on an outpatient basis with antibiotics. The mean length of hospitalization did not differ significantly between the Control and IMNgroups [2.51 ± 2.02 vs. 2.32 ± 1.37 days, p = 0.79].
Regarding vaginal cuff–related complications, one patient in the control group developed a vaginal cuff hematoma on postoperative 4th day and required hospitalization for conservative management. In the IMN group, one patient was hospitalized on postoperative 7th day for vaginal cuff bleeding and underwent bleeding control with suturing under anesthesia. Additionally, two patients in the IMN group developed infectious vaginal cuff complications: one presented with a vaginal cuff abscess on postoperative day 10 and was hospitalized for antibiotic treatment and drainage, while another required hospitalization for vaginal cuff infection and received antibiotic therapy. Patients who developed vaginal cuff complications were excluded from the final analyses for vaginal cuff healing.
3.2. Vaginal Cuff Healing
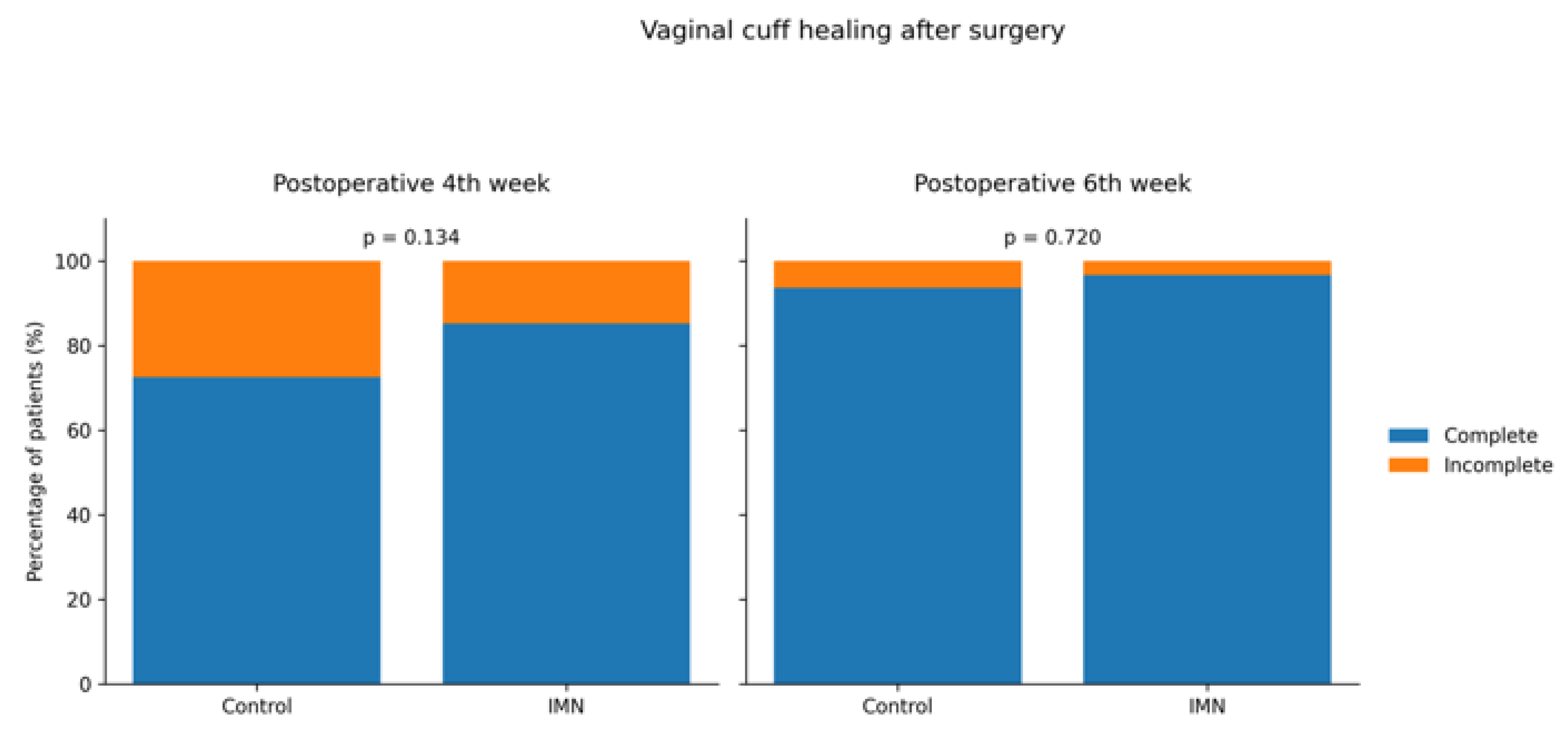
At postoperative 4th week, complete vaginal cuff healing was observed in 74.6% of patients in the control group and 86.2% in the IMN group [p = 0.17]. By postoperative 6th week, complete vaginal cuff healing rates increased to 93.8% in the control group and 96.7% in the IMN group, with no statistically significant difference between groups [p = 0.72]. Graphical comparisons of vaginal cuff healing at postoperative weeks 4 and 6 are presented in
Figure 2, and detailed numerical data are provided in Supplementary Table 1.
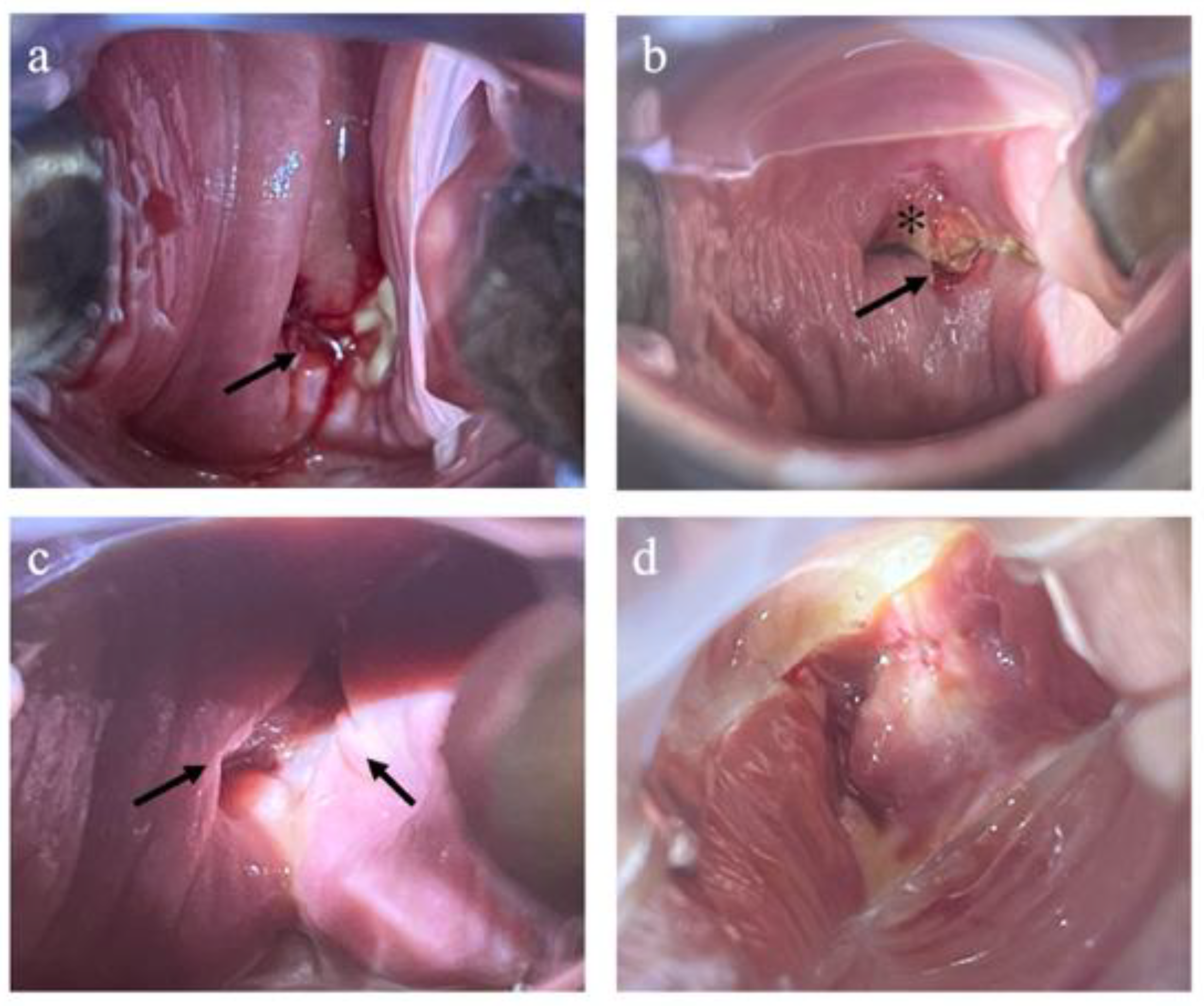
In the IMN group, one patient demonstrated partial vaginal cuff dehiscence at the postoperative 4th week evaluation. As there was no evidence of evisceration and the patient remained asymptomatic, with benign final pathology, expectant management was adopted. At the postoperative 6th week follow-up, a reduction in the size of the dehiscence was observed. Representative examples of vaginal cuff assessment are shown in
Figure 3.
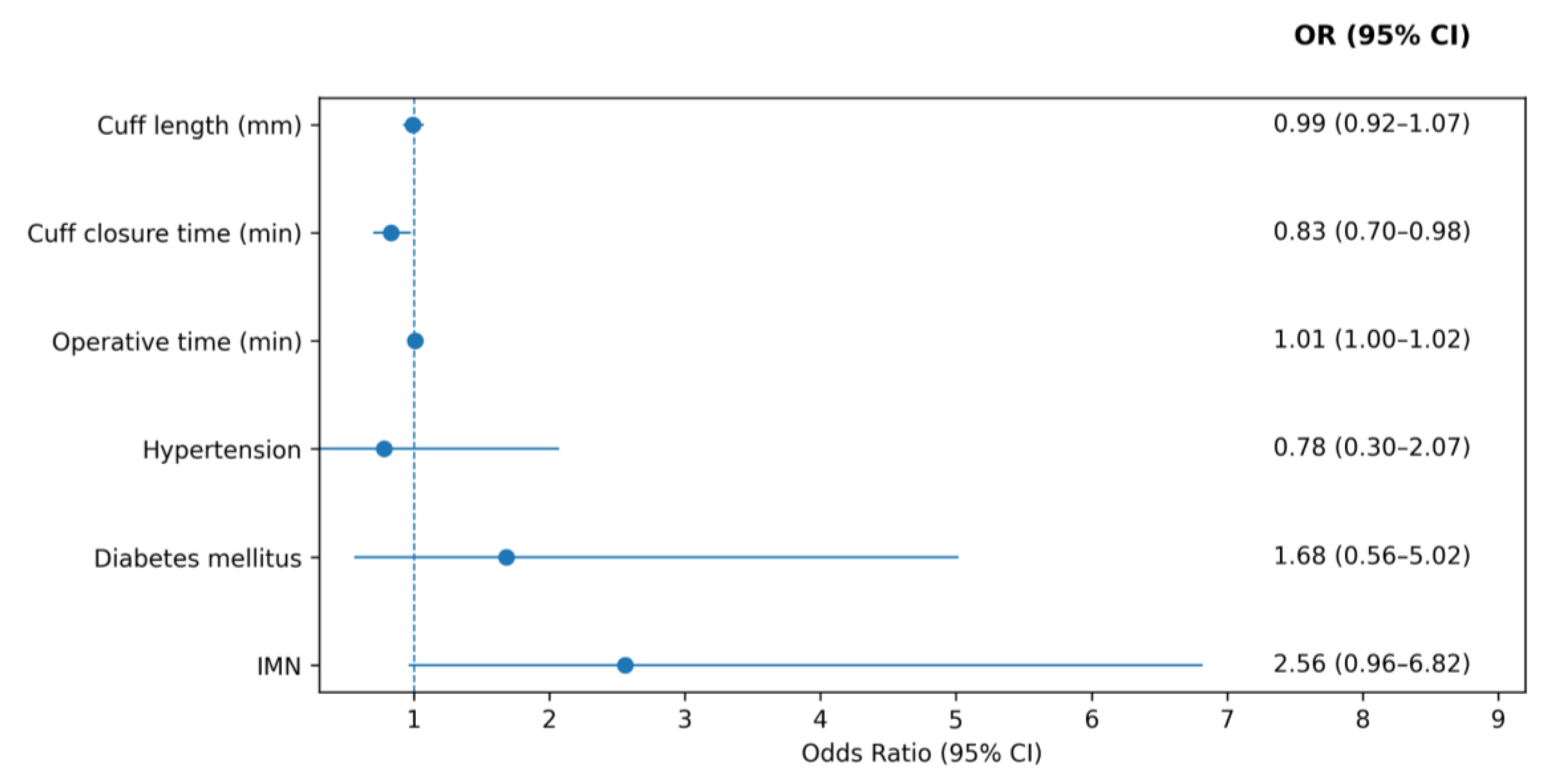
Univariable analyses assessed the association of postoperative IMN use and vaginal cuff closure time with complete vaginal cuff healing at the postoperative 4th week; neither variable showed a statistically significant association. In multivariable logistic regression analysis, postoperative IMN was associated with higher likelihood of complete vaginal cuff healing at the postoperative 4th week [odds ratio [OR] 2.56, 95% confidence interval [CI] 0.96–6.82], but did not achieve statistical significance [p = 0.06]. Longer vaginal cuff closure time was independently associated with a lower probability of complete early vaginal cuff healing [p = 0.02] Supplementary Figure 1].
3.3. Compliance and Tolerance
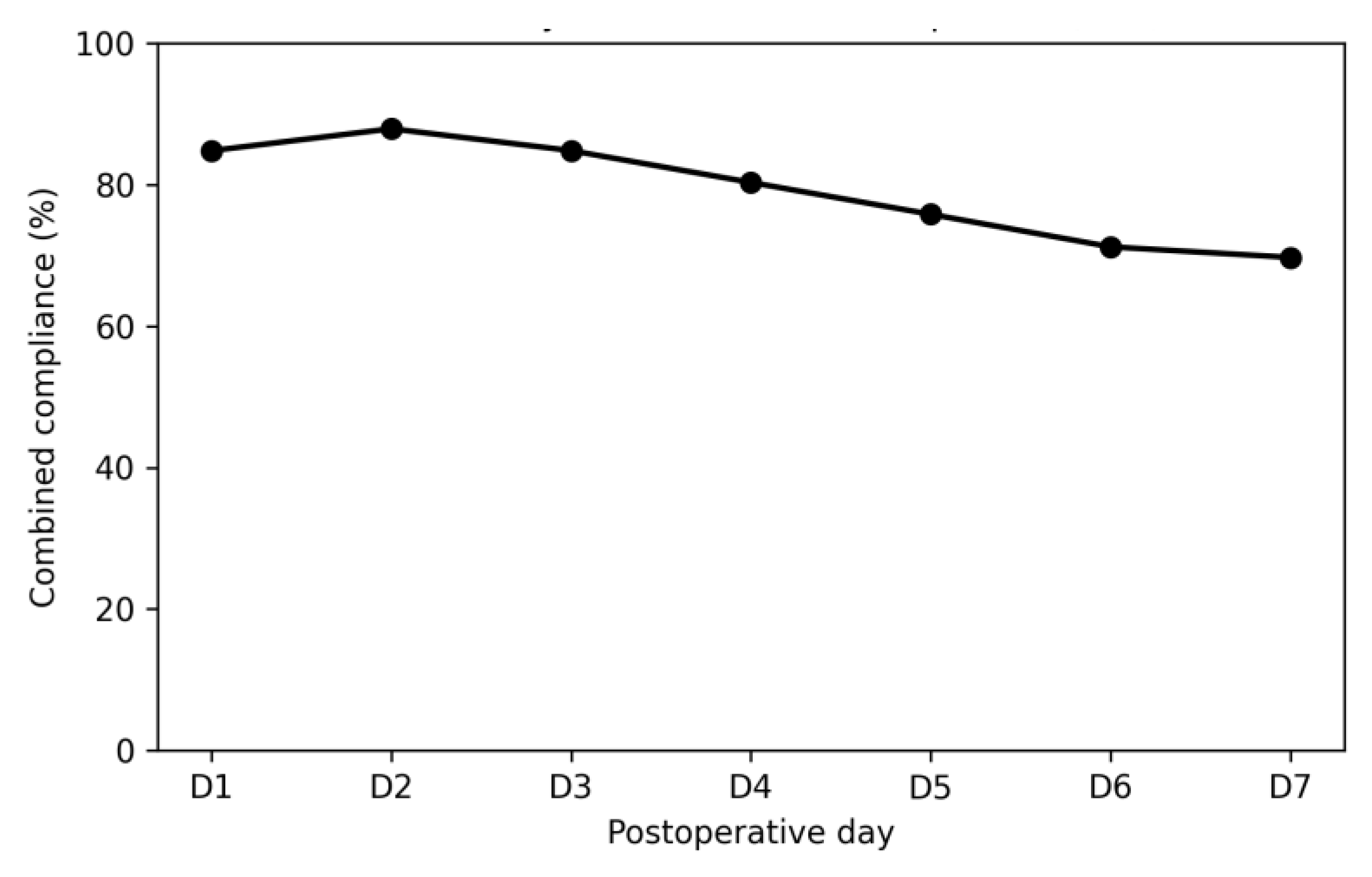
In the IMN group, the median combined compliance over postoperative days 1 to 7 was 100% [IQR 71–100], while the mean combined compliance was 79.2±35.0%. A total of 48 out of 66 patients [72.7%] achieved good compliance [≥80%], and 43 out of 66 patients [65.2%] completed the full prescribed regimen [100%]. Combined compliance at the day level declined from 84.8% on the first postoperative day to 69.7% on the 7th postoperative day, indicating reduced adherence during the later postoperative period [Supplementary Figure 2].
IMDs were well tolerated, with 93.9% of patients completing the regimen Incomplete intake of the prescribed regimen was observed in 6.1% of patients due to mild intolerance [diarrhea in one patient and taste aversion in three patients], and no severe adverse events were observed.
4. Discussion
In this prospective cohort study, patients who received immune-modulating diets (IMDs) had a higher rate of complete vaginal cuff healing at the 4th postoperative week than those on a standard diet; however, this difference did not reach statistical significance. By the 6th postoperative week, rates of complete vaginal cuff healing were high and comparable between the groups. The incidence of postoperative complications within 30 days was also similar.
Immunonutrition (IMN) has been demonstrated to modulate immune and inflammatory responses, enhance collagen synthesis, and reduce postoperative infectious complications in patients undergoing majör oncologic surgery [
6,
15]. In alignment with this evidence, international guidelines recommend perioperative IMN for selected high-risk surgical populations [
7]. However, evidence regarding the role of IMN in gynecologic oncology, particularly after laparoscopic surgery, remains limited.
In the present study, the daily IMN dose was consistent with regimens reported in the literature; however, supplementation was limited to the postoperative period due to clinical considerations. The elective laparoscopic approach and brief preoperative admission period in this cohort precluded standardized preoperative supplementation. As highlighted in the Enhanced Recovery After Surgery literature, barriers to routine preoperative IMN in gynecologic oncology include limited preoperative patient contact, infrastructural constraints, cost considerations, and reduced patient compliance [
16,
17]. Evidence from six meta-analyses suggests that perioperative IMN provides the greatest benefit in surgical oncology, while postoperative administration alone confers a slightly reduced but comparable effect, and preoperative-only supplementation yields less benefit [
18]. However, current guidelines provide limited direction regarding the optimal timing, duration, and dosage of IMN [
19]. The absence of standardized recommendations regarding the optimal timing, duration, and dosage of IMN may partly explain heterogeneity across published studies and the lack of statistically significant findings in the present cohort.
Both study groups exhibited high rates of complete vaginal cuff healing at early postoperative time points. Previous cohorts have reported complete healing rates of approximately 80% at 6th weeks and over 95% at 8th weeks following open hysterectomy [
13]. In contrast, the present cohort included patients with comorbidities known to impair wound healing, yet still demonstrated high rates of complete healing at the 4th and 6th postoperative weeks [
14]. These results do not support concerns that laparoscopic colpotomy with electrosurgical energy inherently compromises vaginal cuff healing [
20]. Instead, they suggest that surgeon experience and adherence to standardized, full-thickness vaginal cuff closure techniques may mitigate potential risks associated with energy use and magnification.
Vaginal cuff closure time emerged as an independent factor associated with early vaginal cuff healing. A longer closure duration may reflect increased technical difficulty, friable tissue quality, or greater local tissue trauma during suturing, all of which may impair microvascular perfusion and delay the early phases of wound repair. In contrast, cuff length was not independently associated with early healing outcomes, indicating that surgical technique and efficiency may be more influential than anatomical dimensions alone.
Although vaginal cuff healing was classified as complete according to the predefined clinical criteria in this study, it is important to recognize that complete mucosal healing at early postoperative assessment does not eliminate the risk of subsequent vaginal cuff dehiscence [
21]. Since dehiscence can occur several months after hysterectomy, studies with extended follow-up are necessary to determine whether IMN confers protection against late vaginal cuff complications, particularly dehiscence, which is a rare but clinically significant adverse outcome following laparoscopic staging for gynecologic malignancies.
Postoperative compliance with IMDs was high, with approximately 88% of patients consuming at least 75% of the planned IMN dose. This finding contrasts with previously reported compliance rates, where adherence to IMN, particularly postoperatively, has been substantially lower. A recent narrative review of IMN within ERAS protocols for gynecologic oncology noted that postoperative compliance declined from nearly 78% in the preoperative period to approximately 28% after surgery, primarily due to nausea, anorexia, and impaired oral intake [
19]. The high compliance observed in this cohort may be attributable to the minimally invasive surgical approach, which is associated with faster gastrointestinal recovery, earlier resumption of oral intake, and a lower incidence of postoperative nausea and vomiting. Similar improvements in tolerance and adherence have been reported in laparoscopic colorectal surgery when IMNis combined with minimally invasive surgical techniques [
22].
Postoperative morbidity did not differ between the study groups. Although perioperative IMN has been shown to reduce hospital and intensive care unit length of stay in surgical populations requiring prolonged postoperative care, such as patients undergoing gastrointestinal, head and neck, or ovarian cancer surgery, these benefits may be less apparent in the context of laparoscopic staging for gynecologic malignancy, where postoperative recovery is inherently rapid, incisions are small, and hospital stay is already brief [
23,
25].
Several limitations warrant consideration. The non-randomized design may have introduced selection bias, despite comparable baseline characteristics between groups. The relatively small sample size may have reduced the statistical power to detect modest differences. IMN was administered only in the postoperative period, which prevented evaluation of potential benefits from preoperative or perioperative supplementation. Although multiple surgeons participated, the vaginal cuff closure technique and suture material were standardized to reduce inter-operator variability.
5. Conclusions
Postoperative IMN was not independently associated with early vaginal cuff healing following laparoscopic surgical staging for endometrial cancer. Surgical factors, particularly cuff closure time, appeared to be the primary determinants of vaginal cuff healing. The incidence of postoperative complications within 30 days was similar between patients receiving IMN and those managed with a standard postoperative diet. Further randomized studies evaluating standardized perioperative IMN protocols, with extended follow-up for late vaginal cuff complications, are recommended.
Author Contributions
SB: Project development, Data collection and management, Data analysis, Manuscript writing/editing BAT: Data collection and management EK: Data collection and management OU: Data collection and management MAN: Project development, Manuscript editing SSO: Manuscript editing TY: Data collection SYŞ: Data collection.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the guidelines of the Helsinki Declaration. The study was approved by the ethics committee of the University of Health Science Adana City Training and Research Hospital (07 December 2023, Decision No: 2981).
Informed Consent Statement
Written informed consent was obtained from all participants.
The authors affirm that human research participants provided informed consent for publication of the images in Figure 3.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request. Conflicts of Interest: The authors declare no conflicts of interest.
Acknowledgments
The authors acknowledge the contribution of the clinical staff at the Gynecologic Oncology Outpatient Department, particularly Emine Kurtuluş, Badel Altuntaş, and Tuğçe Çörekli for their assistance in patient management and follow-up.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CDC |
Centers for Disease Control and Prevention |
| ESPEN |
European Society for Clinical Nutrition and Metabolism |
| ERAS |
Enhanced Recovery After Surgery |
| NRS |
Nutritional Risk Screening |
Appendix A
Table A1.
The incidence of complete vaginal cuff wound healing at 4th and 6th weeks after operation.
Table A1.
The incidence of complete vaginal cuff wound healing at 4th and 6th weeks after operation.
Cuff healing
(Postoperative 4th week) |
Control n (%)
|
IMN n (%)
|
P value |
| Total |
46 (74) |
57(86) |
0.13 |
| Subtotal |
15 (25) |
9 (13) |
| Total patients |
61 (100%) |
66(100) |
Cuff healing
(Po 6th week) |
|
|
|
| Total |
53 (93) |
54 (96) |
0.72 |
| Subtotal |
4 (6) |
2 (3) |
| Total patients |
57 |
56 |
Appendix B
Figure A1.
Multivariable logistic regression analysis of factors associated with complete vaginal cuff healing at postoperative 4th week.
Figure A1.
Multivariable logistic regression analysis of factors associated with complete vaginal cuff healing at postoperative 4th week.
Figure A2.
Day-level combined compliance with postoperative IMDs.
Figure A2.
Day-level combined compliance with postoperative IMDs.
References
- Siegel, RL; Miller, KD; Fuchs, HE; Jemal, A. Cancer statistics, 2023. CA Cancer J Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef]
- Walker, JL; Piedmonte, MR; Spirtos, NM; et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic Oncology Group LAP2 Study. J Clin Oncol. 2009, 27, 5331–5336. [Google Scholar] [CrossRef] [PubMed]
- Uccella, S; Ceccaroni, M; Cromi, A; et al. Vaginal cuff closure after minimally invasive hysterectomy: a randomized controlled trial. Am J Obstet Gynecol. 2018, 218, 500.e1–500.e10. [Google Scholar] [CrossRef]
- Crosbie, EJ; Hoskins, PJ; et al. The impact of adjuvant radiotherapy timing on survival outcomes in endometrial cancer. Gynecol Oncol. 2012, 125, 79–85. [Google Scholar]
- Abu-Rustum, N; Campos, SM; Amarnath, S; et al. NCCN Guidelines® Insights: Uterine Neoplasms, Version 3.2025. J Natl Compr Canc Netw. 2025, 23, 284–291. [Google Scholar] [CrossRef]
- Bharadwaj, S; Trivax, B; Tandon, P; Alkam, B; Hanouneh, I; Steiger, E. Should perioperative immunonutrition for elective surgery be the current standard of care? Gastroenterol Rep (Oxf) 2016, 4, 87–95. [Google Scholar] [CrossRef]
- Weimann, A; Braga, M; Carli, F; et al. ESPEN practical guideline: Clinical nutrition in surgery. Clin Nutr. 2021, 40, 4745–4761. [Google Scholar] [CrossRef]
- Kondrup, J; Rasmussen, HH; Hamberg, O; Stanga, Z; ESPEN Working Group. Nutritional Risk Screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr. 2003, 22, 321–336. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). National Healthcare Safety Network (NHSN) Patient Safety Component Manual: Chapter 17. Surgical Site Infection (SSI) Event. CDC: Atlanta (GA), 2023. Available online: https://www.cdc.gov/nhsn/pdfs/pscmanual/17pscnosinfdef_current.pdf (accessed on 24 January 2026).
- Tinsopharat, C; Paiwattananupant, K; Lekskul, N; et al. Vaginal stump characteristics after total abdominal hysterectomy using electrosurgery versus scalpel or scissors for vaginal incision: a randomized controlled trial. J Med Assoc Thai 2016, 99, 1277–1282. [Google Scholar] [PubMed]
- Watcharaprapapong, J; Treetampinich, C; Na Ayudhya, NI; Paiwattananupant, K; Chinthakanan, O. Incidence of complete vaginal cuff wound healing at sixth and eighth week after total abdominal hysterectomy. J Obstet Gynaecol Res. 2019, 45, 399–404. [Google Scholar] [CrossRef]
- Guo, S; DiPietro, LA. Critical review: factors affecting wound healing. J Dent Res. 2010, 89, 219–229. [Google Scholar] [CrossRef]
- Martin, P; Nunan, R. Cellular and molecular mechanisms of repair in acute and chronic wounds. Br J Dermatol 2015, 173, 370–378. [Google Scholar] [CrossRef]
- Falanga, V. Wound healing and its impairment in the diabetic foot. Lancet 2005, 366, 1736–1743. [Google Scholar] [CrossRef]
- Shen, J; Dai, S; Li, Z; et al. Effect of enteral immunonutrition in patients undergoing surgery for gastrointestinal cancer: an updated systematic review and meta-analysis. Front Nutr. 2022, 9, 941975. [Google Scholar] [CrossRef]
- Lygizos, V; Haidopoulos, D; Vlachos, DE; et al. Immunonutrition in ERAS protocol for patients with gynecologic cancer: a narrative review of the literature. Life (Basel) 2025, 15, 487. [Google Scholar] [CrossRef] [PubMed]
- Oodit, R; Biccard, BM; Panieri, E; et al. Guidelines for perioperative care in elective abdominal and pelvic surgery at primary and secondary hospitals in low- and middle-income countries: ERAS Society recommendations. World J Surg. 2022, 46, 1826–1843. [Google Scholar] [CrossRef]
- Kavalukas, S; McClave, SA. Immunonutrition vs standard nutrition for patients with cancer. Nutr Clin Pract. 2023, 38, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Ho, C; Mohd Yusof, BN; Abdul Majid, H; Daud, ZAM. Effect of perioperative immunonutrition intervention among gynecological cancer patients: a systematic review. Clin Nutr ESPEN 2024, 64, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Moya, P; Miranda, E; Soriano-Irigaray, L; et al. Perioperative immunonutrition in normo-nourished patients undergoing laparoscopic colorectal resection. Surg Endosc. 2016, 30, 4946–4953. [Google Scholar] [CrossRef]
- Nezhat, C; Kennedy Burns, M; Wood, M; et al. Vaginal cuff dehiscence and evisceration: a review. Obstet Gynecol. 2018, 132, 972–985. [Google Scholar] [CrossRef]
- Wiebe, E; Covens, A; Thomas, G. Vaginal vault dehiscence and increased use of vaginal vault brachytherapy: what are the implications? Int J Gynecol Cancer 2012, 22, 1611–1616. [Google Scholar] [CrossRef] [PubMed]
- Ferrero, A; Vassallo, D; Geuna, M; et al. Immunonutrition in ovarian cancer: clinical and immunological impact. J Gynecol Oncol. 2022, 33, e77. [Google Scholar] [CrossRef] [PubMed]
- Chapman, JS; Roddy, E; Westhoff, G; et al. Postoperative enteral immunonutrition for gynecologic oncology patients undergoing laparotomy decreases wound complications. Gynecol Oncol. 2015, 137, 523–528. [Google Scholar] [CrossRef]
- Matsui, R; Sagawa, M; Sano, A; et al. Impact of perioperative immunonutrition on postoperative outcomes for patients undergoing head and neck or gastrointestinal cancer surgeries: a systematic review and meta-analysis of randomized controlled trials. Ann Surg. 2024, 279, 419–428. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).