Submitted:
22 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract

Keywords:
1. Introduction
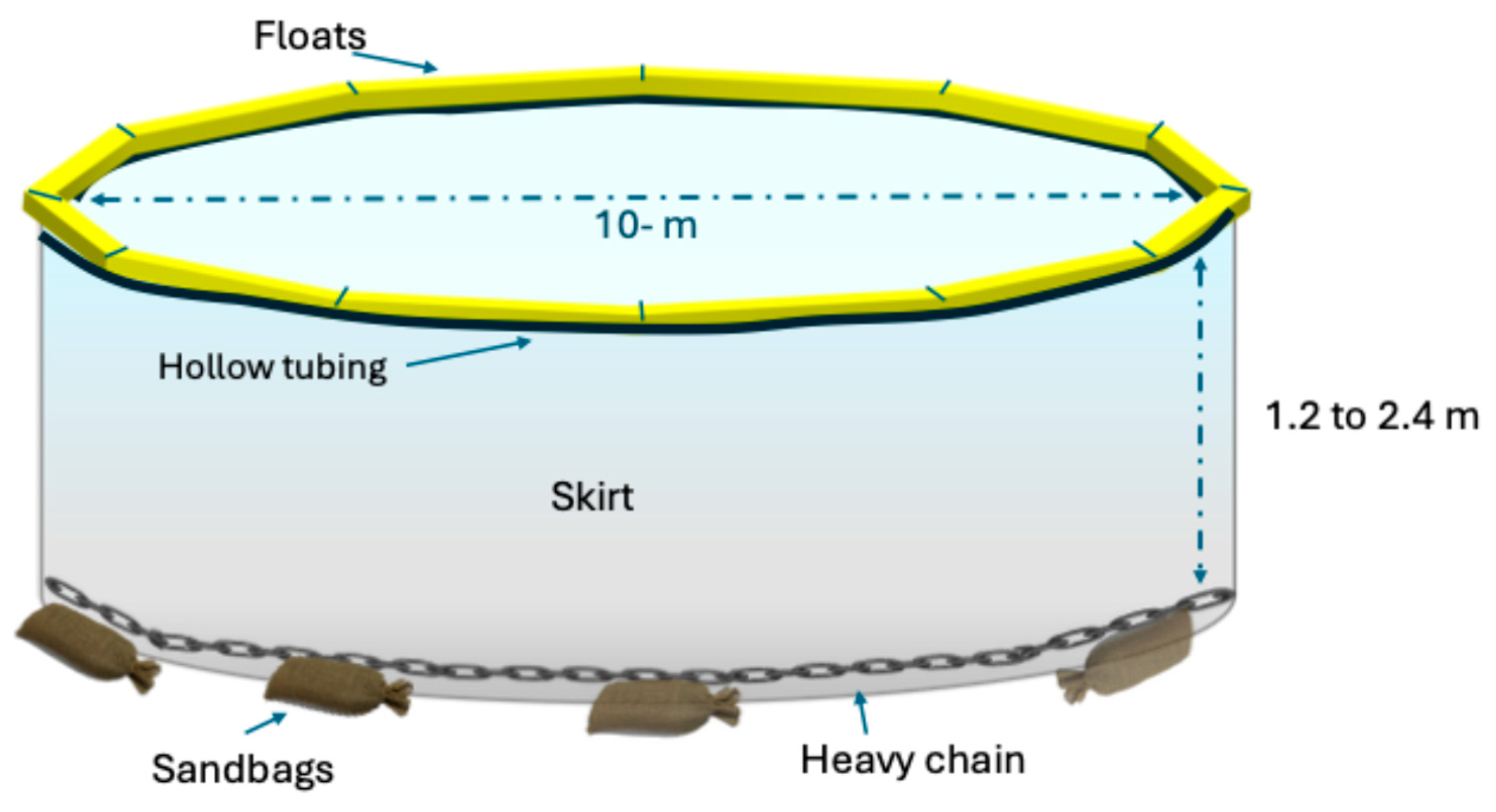
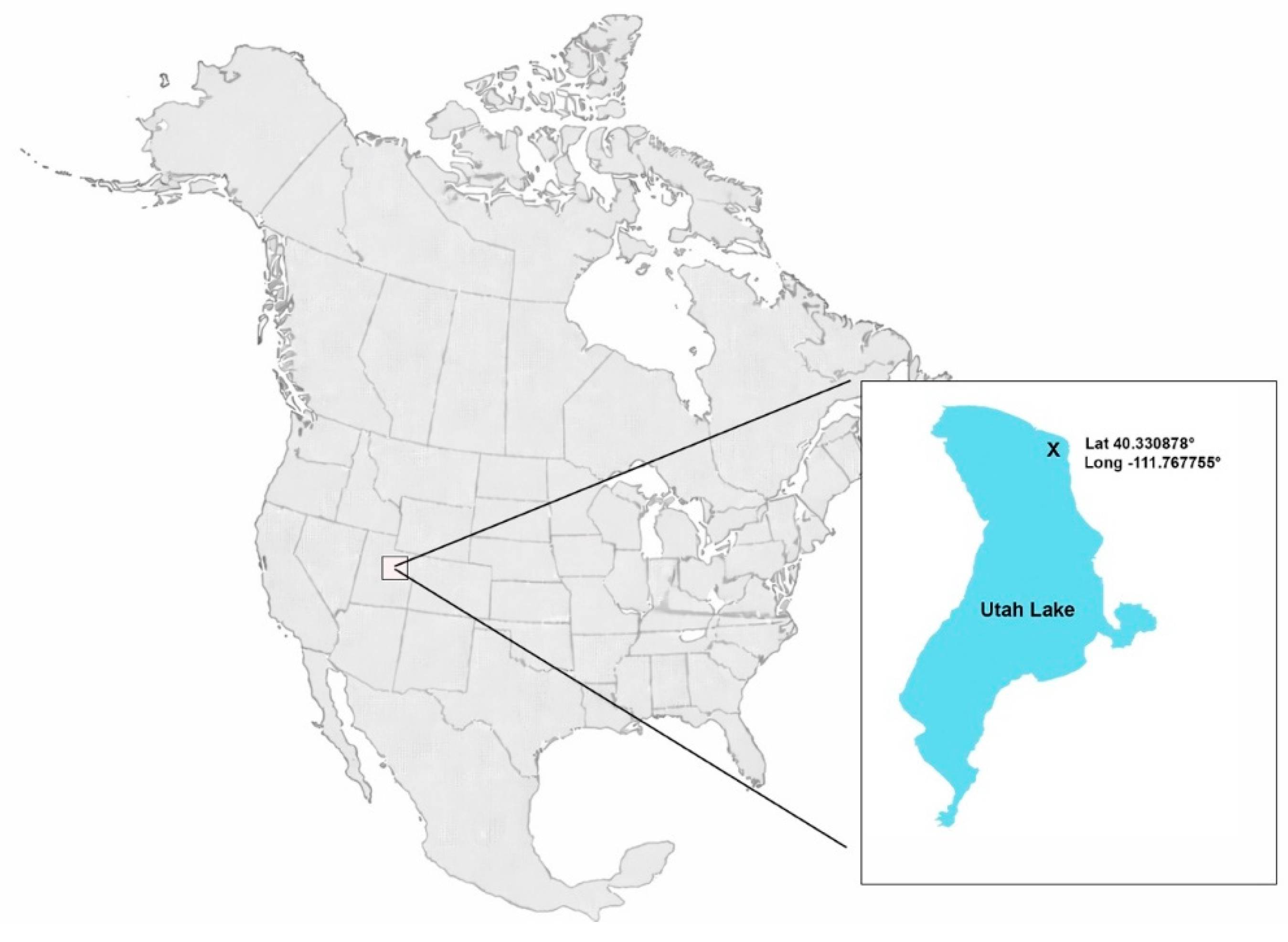
2. Materials and Methods
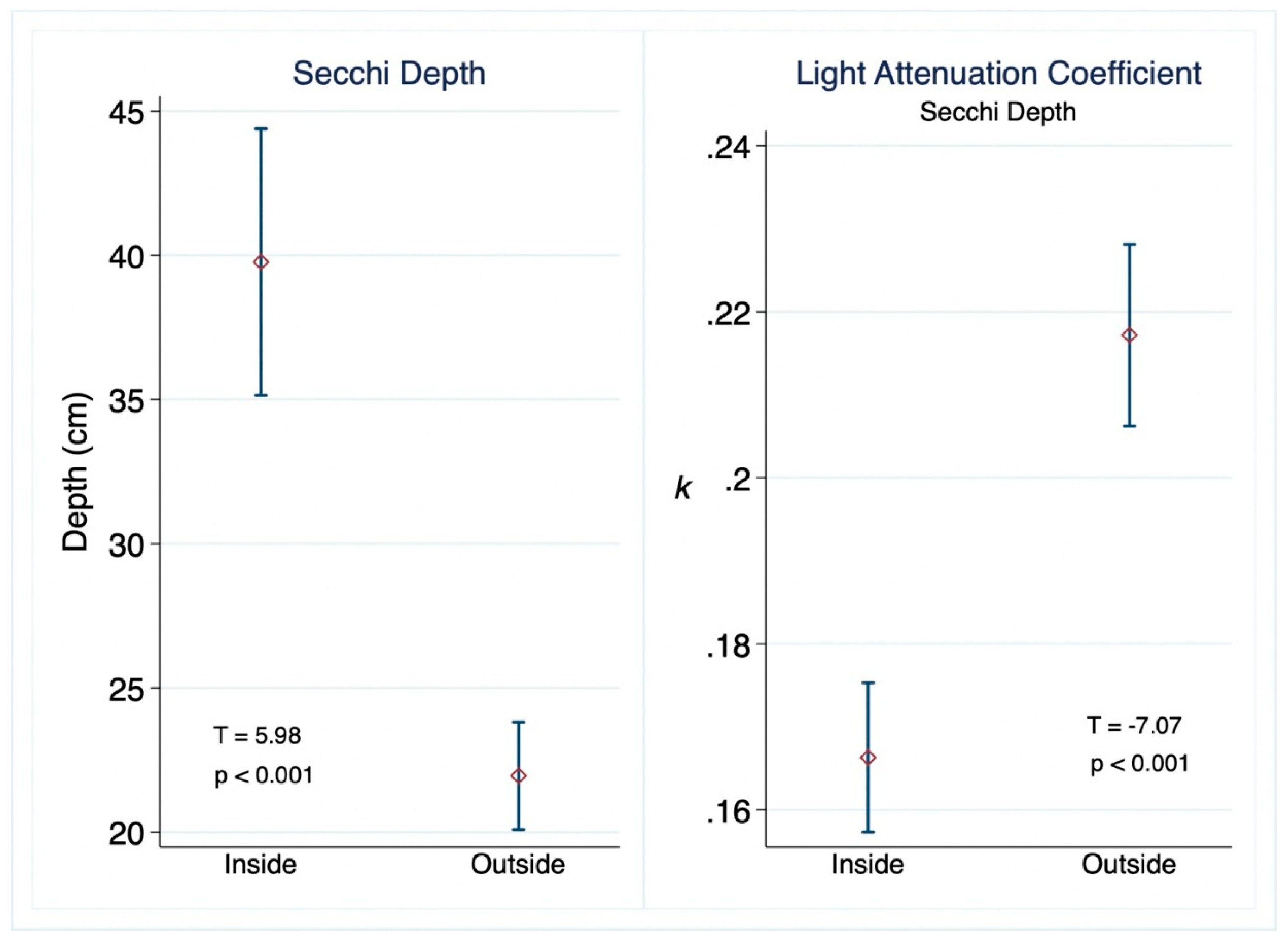
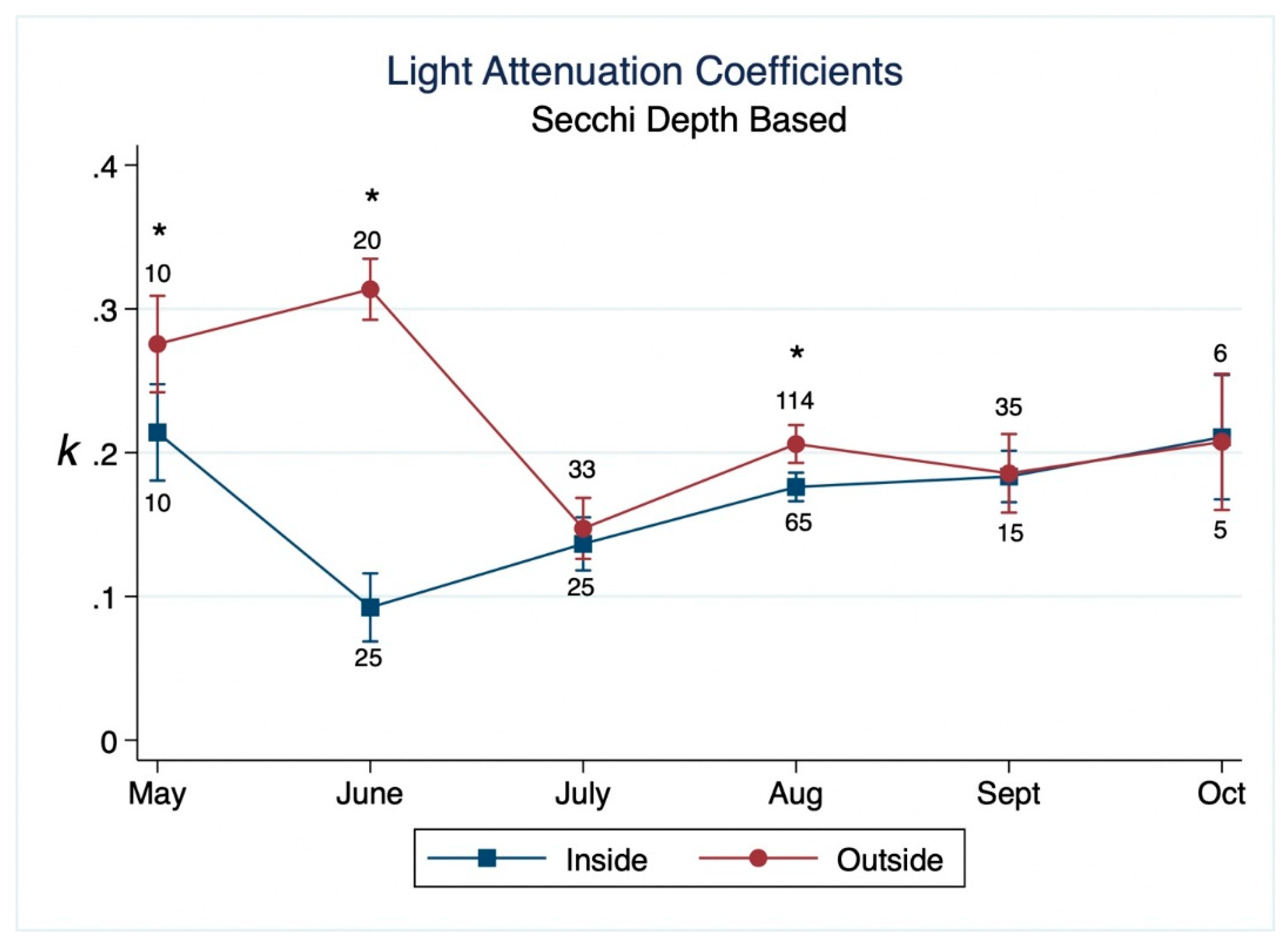
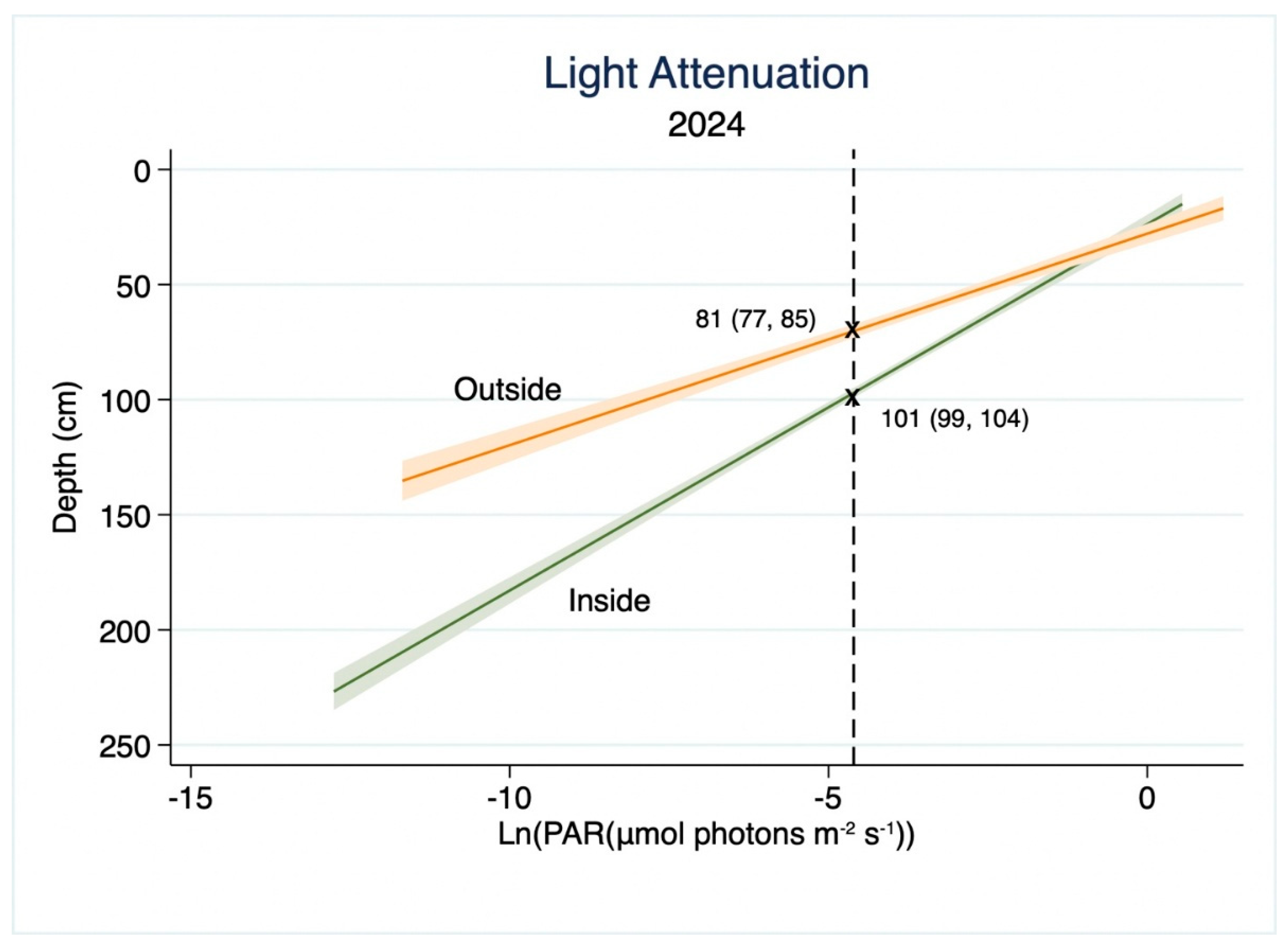
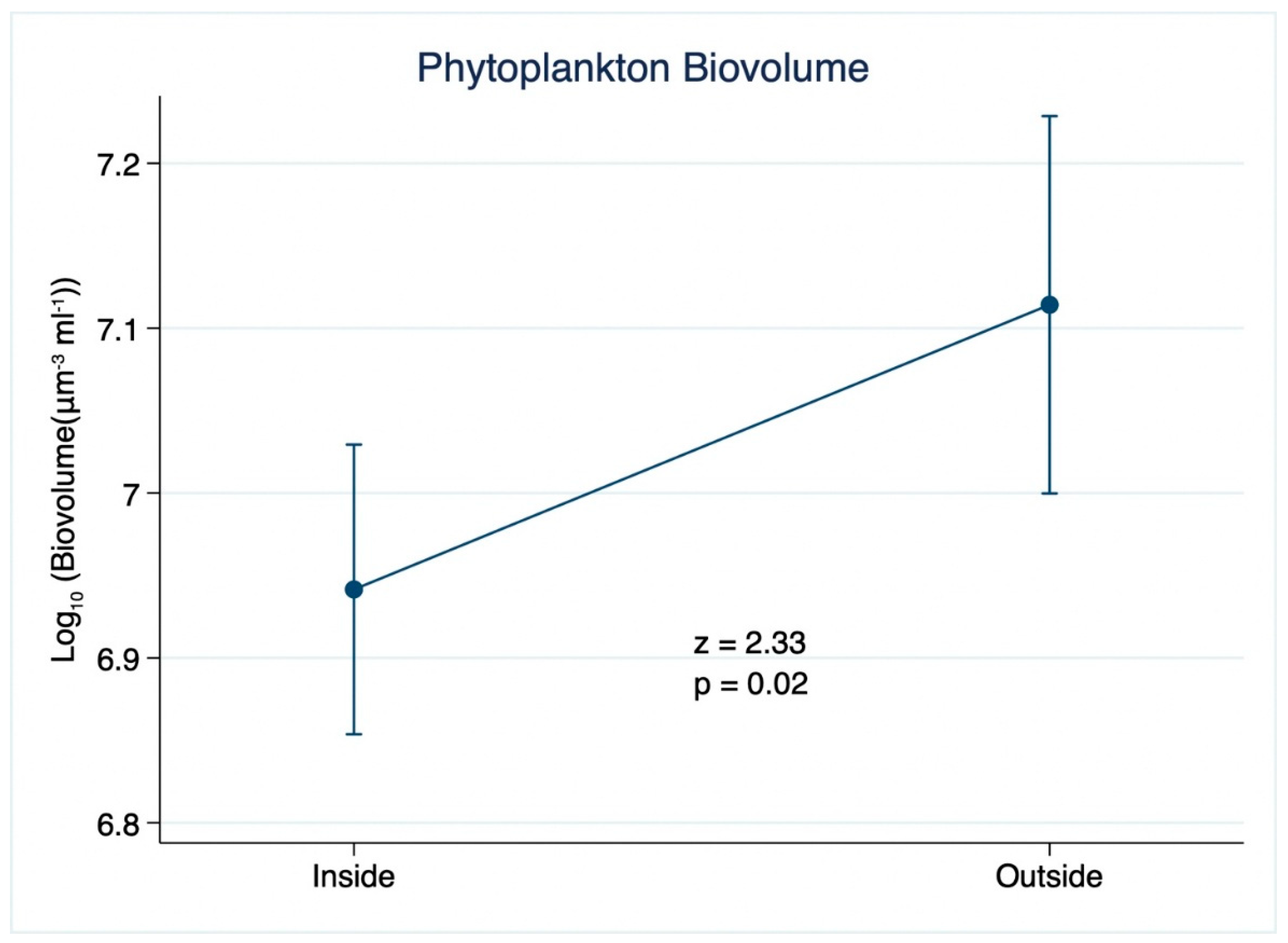
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Karlsson, J.; Byström, P.; Ask, J.; Ask, P.; Persson, L.; Jansson, M. Light limitation of nutrient-poor lake ecosystems. Nature 2009, 460, 506–509. [CrossRef]
- Kemp, W.M.; Boynton, W.R.; Adolf, J.E.; Boesch, D.F.; Boicourt, W.C.; Brush, G.; Cornwell, J.C.; et al. Eutrophication of Chesapeake Bay: historical trends and ecological interactions. Mar. Ecol. Prog. Ser. 2005, 303, 1–29. [CrossRef]
- Weiskerger, C.J.; Rowe, M.D.; Stow, C.A.; Stuart, D.; Johengen, T. Application of the Beer–Lambert model to attenuation of photosynthetically active radiation in a shallow, eutrophic lake. Water Resour. Res. 2018, 54, 8952–8962. [CrossRef]
- Medrano, E.A.; Uittenbogaard, R.E.; Pires, L.M.D.; van de Wiel, B.J.H.; Clercx, H.J.H. Coupling hydrodynamics and buoyancy regulation in Microcystis aeruginosa. Ecol. Model. 2013, 248, 41–56. [CrossRef]
- Whittington, J.; Sherman, B.; Green, D.; Oliver, R.L. Growth of Ceratium hirundinella in a subtropical reservoir. J. Plankton Res. 2000, 22, 1025–1045. [CrossRef]
- Babin, M.; Stramski, D.; Ferrari, G.M.; Claustre, H.; Bricaud, A.; Obolensky, G.; Hoepffner, N. Variations in light absorption coefficients in coastal waters. J. Geophys. Res. Oceans 2003, 108, 3211. [CrossRef]
- Bukata, R.P.; Jerome, J.H.; Kondratyev, K.Y.; Pozdnyakov, D.V. Optical Properties and Remote Sensing of Inland and Coastal Waters; CRC Press: Boca Raton, FL, USA, 1995.
- Effler, S.W.; Perkins, M.G.; O’Donnell, S.M.; Strait, C.M. Light attenuation in lakes. Inland Waters 2010, 1, 1–13.
- Kirk, J.T.O. Light and Photosynthesis in Aquatic Ecosystems, 3rd ed.; Cambridge Univ. Press: Cambridge, UK, 2011. [CrossRef]
- Morris, D.P.; Zagarese, H.; Williamson, C.E.; et al. Attenuation of solar UV radiation in lakes. Limnol. Oceanogr.1995, 40, 1381–1391. [CrossRef]
- Plummer, L.N.; Busenberg, E. Solubilities of calcite and aragonite. Geochim. Cosmochim. Acta 1982, 46, 1011–1040. [CrossRef]
- Wetzel, R.G. Limnology: Lake and River Ecosystems, 3rd ed.; Academic Press: San Diego, CA, USA, 2001.
- Robbins, L.L.; Blackwelder, P.L. Biochemical evidence for origin of whitings. Geology 1992, 20, 464–468.
- Richards, D.C.; Marshall, B.; Rushforth, S.; Wellard, B. Utah Lake mesocosm study. Zenodo 2025. [CrossRef]
- Taggart, J.B.; Ryan, R.L.; Williams, G.P.; et al. Historical phosphorus mass in Utah Lake. Water 2024, 16, 933. [CrossRef]
- Valek, R.A.; Tanner, K.B.; Taggart, J.B.; et al. ICP-OES detectable elements in Utah Lake. Water 2024, 16, 2170. [CrossRef]
- King, L.; Brahney, J.; Daly, S.; et al. Primary production modeling. Freshw. Sci. 2023, 42, 44–57. [CrossRef]
- Tanner, K.B.; Cardall, A.C.; Taggart, J.B.; Williams, G.P. Earth Observation investigation of algal blooms. Remote Sens. 2026, 18, 394. [CrossRef]
- Padial, A.A.; Thomaz, S.M. Prediction of light attenuation coefficient. Limnology 2008, 9, 143–151. [CrossRef]
- Ingle, J.D.; Crouch, S.R. Spectrochemical Analysis; Prentice Hall: Englewood Cliffs, NJ, USA, 1988.
- Hancke, K.; et al. Low light requirement for algae growth. J. Geophys. Res. Oceans 2018, 123, 985–1000. [CrossRef]
- Randelhoff, A.; et al. Arctic phytoplankton growth. Sci. Adv. 2020, 6, eabc2678. [CrossRef]
- Raven, J.A.; Kübler, J.E.; Beardall, J. Put out the light. J. Mar. Biol. Assoc. UK 2000, 80, 1–25. [CrossRef]
- StataCorp. Stata/IC 16.1 for Mac; StataCorp LLC: College Station, TX, USA, 2023.
- Mortensen, B.M.; Haber, M.J.; DeJong, J.T.; Caslake, L.F.; Nelson, D.C. Environmental factors on CaCO₃ precipitation. J. Appl. Microbiol. 2011, 111, 338–349. [CrossRef]
- Richards, D.C.; Marshall, B.; Rushforth, S.; Mickelsen, R.; Williams, G.; Miller, T. Effects of reduced turbation on food web function. Zenodo 2025. [CrossRef]
- Scheffer, M. Ecology of Shallow Lakes; Kluwer Academic: Dordrecht, Netherlands, 1998.
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic shifts in ecosystems. Nature 2001, 413, 591–596. [CrossRef]
- Edwards, K.F.; Thomas, M.K.; Klausmeier, C.A.; Litchman, E. Phytoplankton growth synthesis. Limnol. Oceanogr.2016, 61, 1232–1244. [CrossRef]
- Quinlan, R.; Filazzola, A.; Mahdiyan, O.; et al. Total phosphorus–chlorophyll relationships. Limnol. Oceanogr.2021, 66, 392–404. [CrossRef]
- Tammeorg, O.; Niemistö, J.; Möls, T.; et al. Wind-induced sediment resuspension. Aquat. Sci. 2013, 75, 559–570. [CrossRef]
- Tang, C.; Li, Y.; He, C.; Acharya, K. Sediment resuspension behavior. Sci. Total Environ. 2020, 708, 135131. [CrossRef]
- Zhang, S.; Yi, Q.; Buyang, S.; Cui, H.; Zhang, S. Bioavailable phosphorus enrichment. Sci. Total Environ. 2020, 710, 135672. [CrossRef]
- Schallenberg, M.; Burns, C.W. Sediment resuspension effects. Freshw. Biol. 2004, 49, 143–159. [CrossRef]
- Trochine, C.; Risholt, C.; Schou, M.; et al. Fish larvae diet in turbid lakes. Freshw. Biol. 2022, 67, 1601–1615. [CrossRef]
- Janetski, J.C. Utah Lake prehistory. Utah Hist. Q. 1990, 58, 5–31.
- Williams, G.P.; Nelson, S.; Rushforth, S.; et al. Human-driven trophic changes in Utah Lake. Water Air Soil Pollut.2023, 234, 218. [CrossRef]
- Richards, D.C.; Rushforth, S.; Marshall, B.; Williams, G.; Wellard, B. Utah Lake mesocosm study ecological section. Zenodo 2023. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



