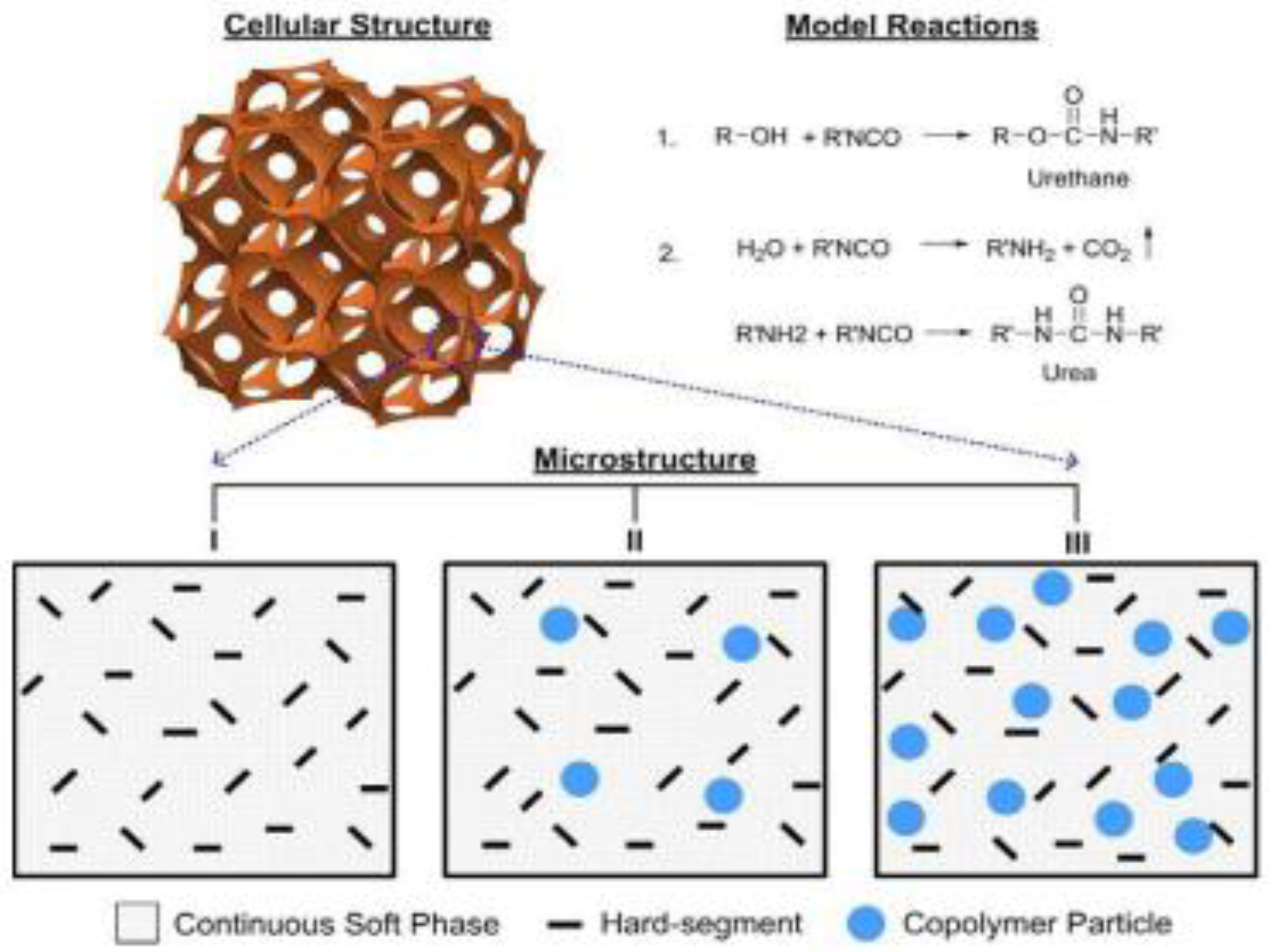
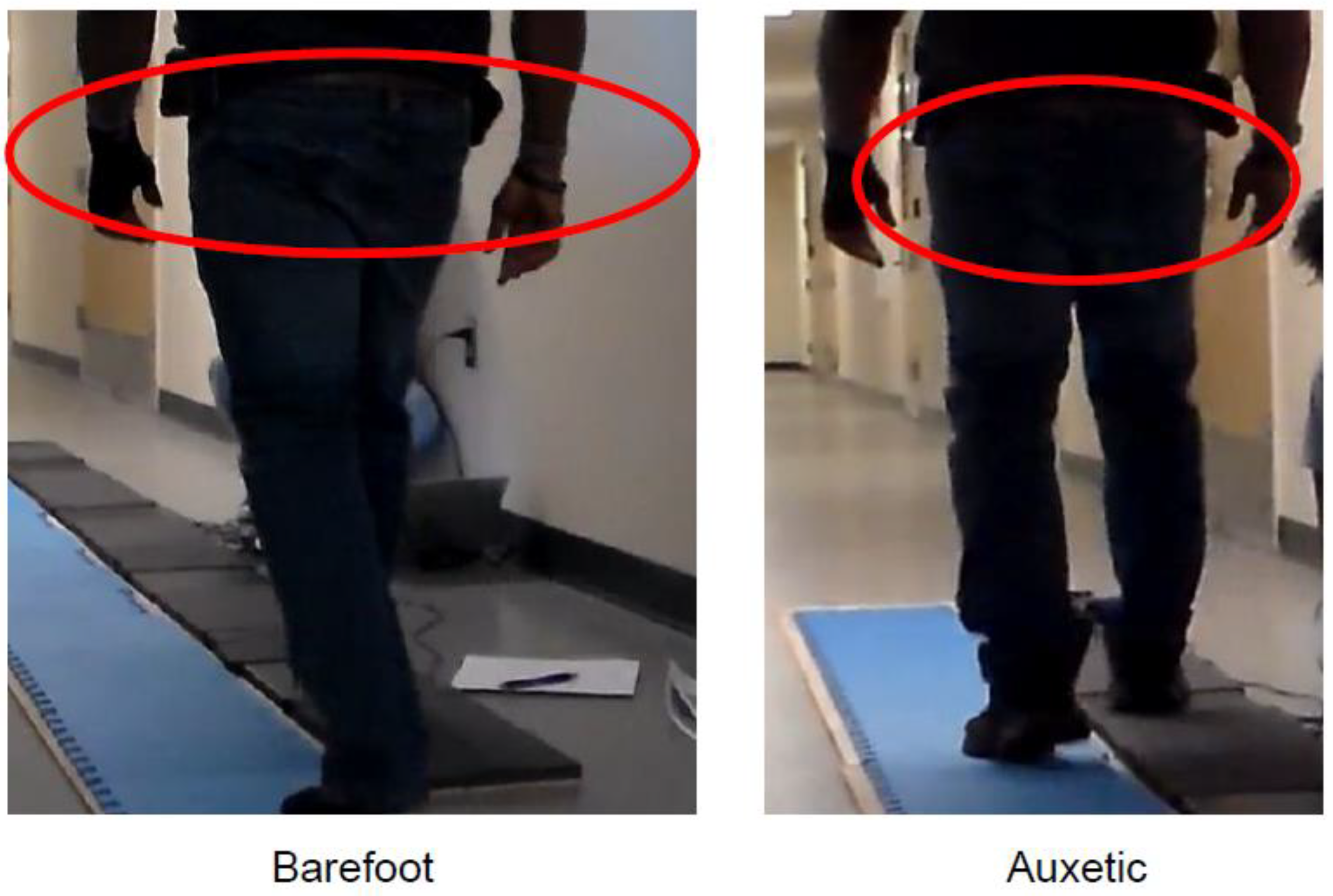
Appendix A
Table A2.
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects A, B, D, E, and F(with braces).
Table A2.
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects A, B, D, E, and F(with braces).
| GRFS data(In Newtons(N)); Subjects A, B, D, E, and F(with braces) |
| Group |
Cancer |
Diabetes |
Disease |
Control |
Injury |
| Barefoot |
771 |
918 |
817 |
689 |
912 |
| |
804 |
938 |
802 |
697 |
864 |
| |
800 |
943 |
466 |
698 |
896 |
| |
793 |
953 |
784 |
690 |
902 |
| |
761 |
923 |
811 |
667 |
892 |
| |
793 |
956 |
779 |
665 |
893 |
| |
|
|
|
|
|
| No auxetic |
764 |
963 |
712 |
709 |
794 |
| |
783 |
990 |
798 |
703 |
836 |
| |
798 |
963 |
765 |
432 |
951 |
| |
790 |
969 |
792 |
388 |
953 |
| |
767 |
929 |
787 |
610 |
965 |
| |
797 |
974 |
775 |
730 |
932 |
| |
|
|
|
|
|
| Auxetic |
796 |
957 |
777 |
774 |
944 |
| |
826 |
961 |
772 |
674 |
935 |
| |
793 |
939 |
756 |
729 |
981 |
| |
784 |
925 |
757 |
709 |
908 |
| |
790 |
955 |
572 |
738 |
883 |
| |
783 |
958 |
784 |
714 |
932 |
Table A2 presents the vertical ground reaction forces (GRFs) measured in Newtons (N) for Subjects A, B, D, E, and F (F used braces). Data is grouped into three conditions: barefoot walking, walking with standard insoles ("No auxetic"), and walking with experimental auxetic foam insoles ("Auxetic"). Subject C is excluded from this table to isolate specific neuropathic categories, with Subject A serving as the representative for diabetic neuropathy in this analysis.
Table A3.
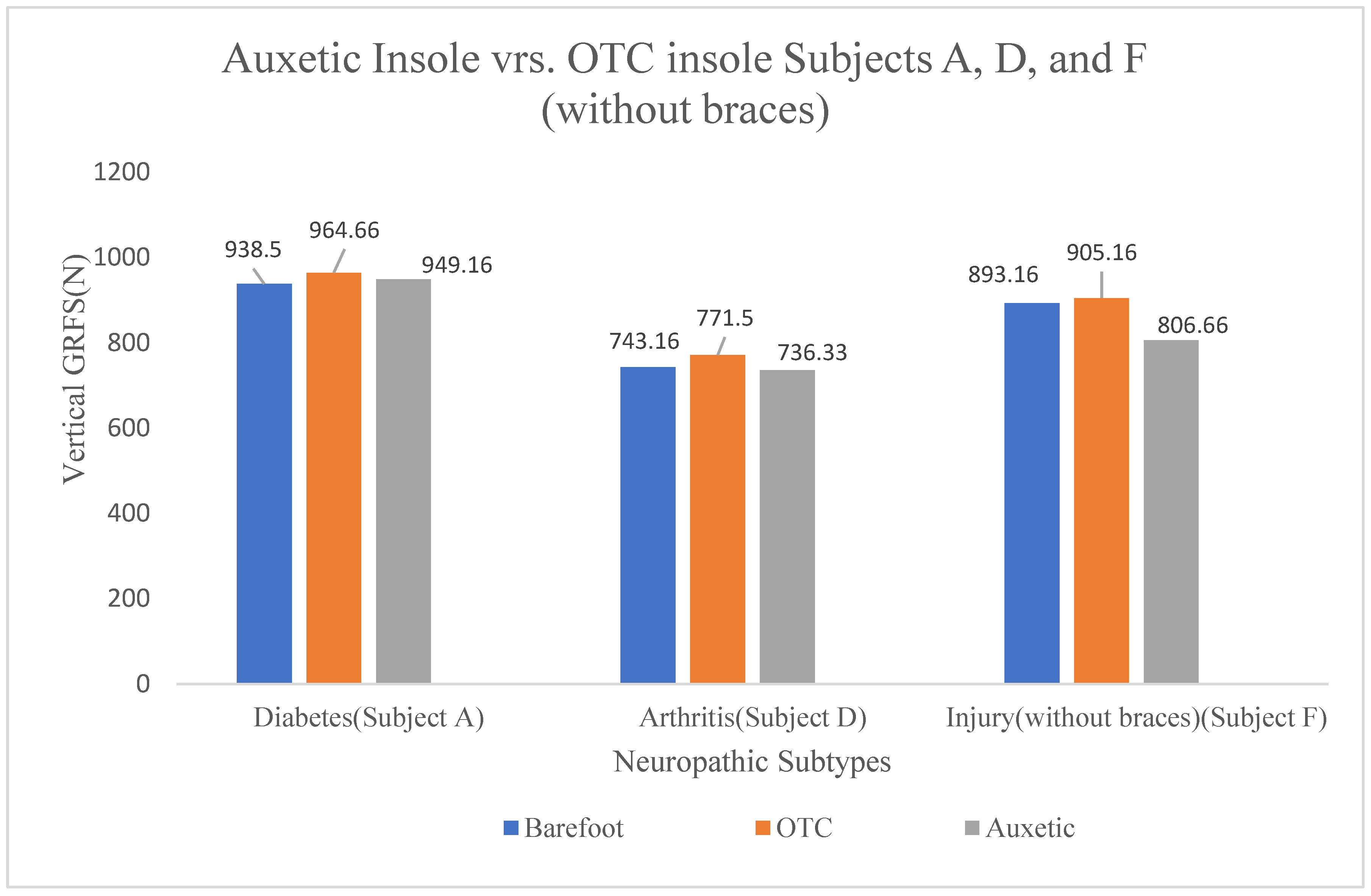
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects A, B, D, E, and F(without braces).
Table A3.
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects A, B, D, E, and F(without braces).
| GRFS data(In Newtons(N)); Subjects A, B, D, E, and F(without braces) |
| Group |
Cancer |
Diabetes |
Disease |
Control |
Injury |
| Barefoot |
771 |
918 |
817 |
689 |
912 |
| |
804 |
938 |
802 |
697 |
864 |
| |
800 |
943 |
466 |
698 |
896 |
| |
793 |
953 |
784 |
690 |
902 |
| |
761 |
923 |
811 |
667 |
892 |
| |
793 |
956 |
779 |
665 |
893 |
| |
|
|
|
|
|
| No auxetic |
764 |
963 |
712 |
709 |
794 |
| |
783 |
990 |
798 |
703 |
836 |
| |
798 |
963 |
765 |
432 |
951 |
| |
790 |
969 |
792 |
388 |
953 |
| |
767 |
929 |
787 |
610 |
965 |
| |
797 |
974 |
775 |
730 |
932 |
| |
|
|
|
|
|
| Auxetic |
796 |
957 |
777 |
774 |
807 |
| |
826 |
961 |
772 |
674 |
935 |
| |
793 |
939 |
756 |
729 |
631 |
| |
784 |
925 |
757 |
709 |
856 |
| |
790 |
955 |
572 |
738 |
779 |
| |
783 |
958 |
784 |
714 |
832 |
Table A3 presents the vertical ground reaction forces (GRFs) measured in Newtons (N) for subjects A, B, D, E, and F. This data specifically focuses on conditions where Subject F used no braces. The data is grouped into three conditions: barefoot walking, walking with standard insoles ("No auxetic"), and walking with experimental auxetic foam insoles ("Auxetic"). Subject C is excluded from this table to isolate specific neuropathic categories, with Subject A serving as the representative for diabetic neuropathy in this analysis.
Table A4.
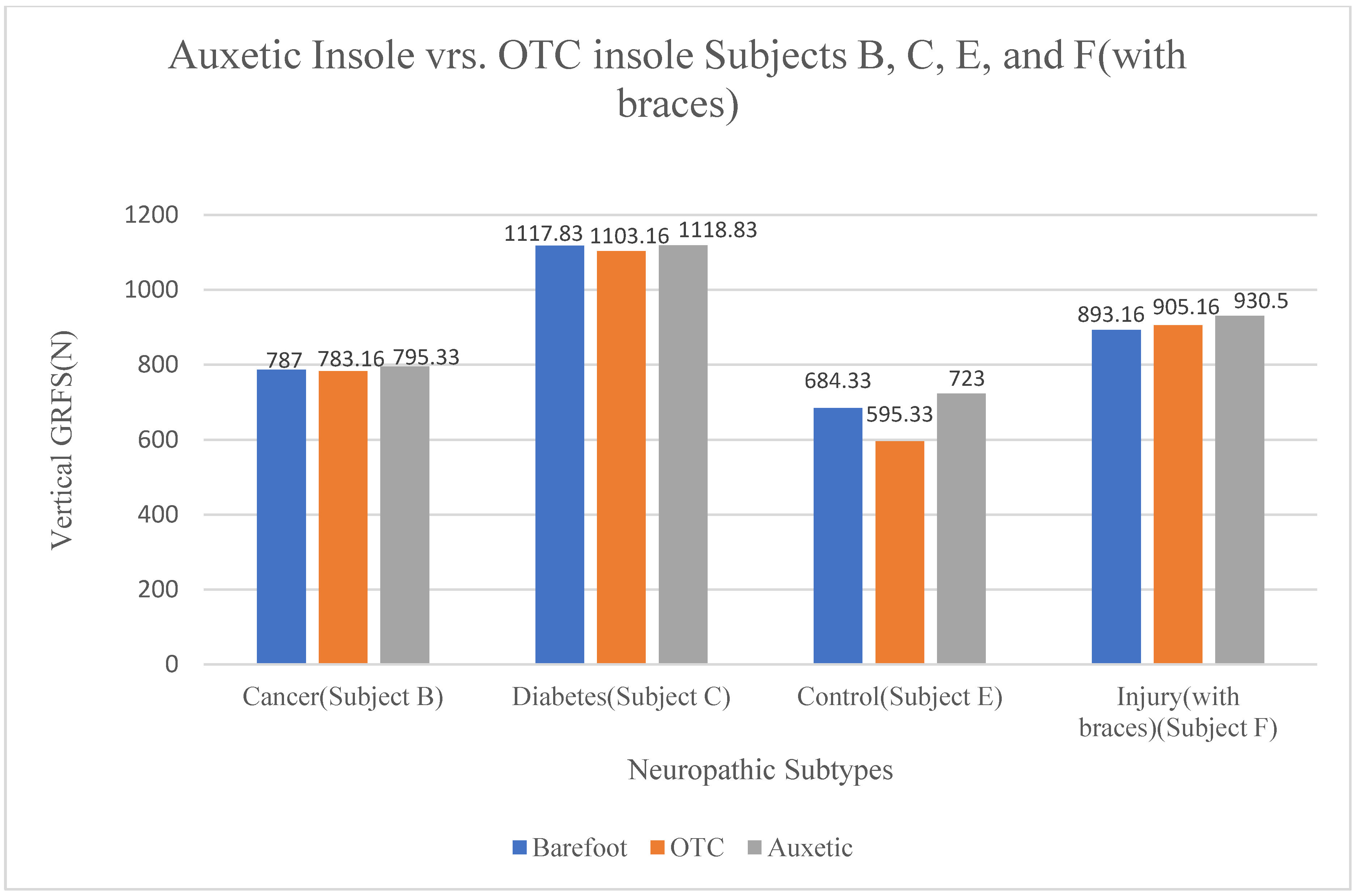
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects B, C, D, E, and F(with braces).
Table A4.
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects B, C, D, E, and F(with braces).
| GRFS data (In Newtons(N)); Subjects B, C, D, E, and F(with braces) |
| Group |
Cancer |
Diabetes |
Disease |
Control |
Injury |
| Barefoot |
771 |
1119 |
817 |
689 |
912 |
| |
804 |
1095 |
802 |
697 |
864 |
| |
800 |
1165 |
466 |
698 |
896 |
| |
793 |
1148 |
784 |
690 |
902 |
| |
761 |
1102 |
811 |
667 |
892 |
| |
793 |
1078 |
779 |
665 |
893 |
| |
|
|
|
|
|
| No auxetic |
764 |
1130 |
712 |
709 |
794 |
| |
783 |
1030 |
798 |
703 |
836 |
| |
798 |
1111 |
765 |
432 |
951 |
| |
790 |
1070 |
792 |
388 |
953 |
| |
767 |
1147 |
787 |
610 |
965 |
| |
797 |
1131 |
775 |
730 |
932 |
| |
|
|
|
|
|
| Auxetic |
796 |
1106 |
777 |
774 |
944 |
| |
826 |
1145 |
772 |
674 |
935 |
| |
793 |
1121 |
756 |
729 |
981 |
| |
784 |
1098 |
757 |
709 |
908 |
| |
790 |
1127 |
572 |
738 |
883 |
| |
783 |
1116 |
784 |
714 |
932 |
Table A4 presents the vertical ground reaction forces (GRFs) measured in Newtons (N) for Subjects B, C, D, E, and F (with braces). Data is grouped into three conditions: barefoot walking, walking with standard insoles ("No auxetic"), and walking with experimental auxetic foam insoles ("Auxetic"). Subject A is excluded from this table to isolate specific neuropathic categories, with Subject C serving as the representative for diabetic neuropathy in this analysis.
Table A5.
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects B, C, D, E, and F(without braces).
Table A5.
Vertical Ground Reaction Forces (GRFs) Data in Newtons (N) for Subjects B, C, D, E, and F(without braces).
| GRFS data (In Newtons(N)); Subjects B, C, D, E, and F(without braces) |
| Group |
Cancer |
Diabetes |
Disease |
Control |
Injury |
| Barefoot |
771 |
1119 |
817 |
689 |
912 |
| |
804 |
1095 |
802 |
697 |
864 |
| |
800 |
1165 |
466 |
698 |
896 |
| |
793 |
1148 |
784 |
690 |
902 |
| |
761 |
1102 |
811 |
667 |
892 |
| |
793 |
1078 |
779 |
665 |
893 |
| |
|
|
|
|
|
| No auxetic |
764 |
1130 |
712 |
709 |
794 |
| |
783 |
1030 |
798 |
703 |
836 |
| |
798 |
1111 |
765 |
432 |
951 |
| |
790 |
1070 |
792 |
388 |
953 |
| |
767 |
1147 |
787 |
610 |
965 |
| |
797 |
1131 |
775 |
730 |
932 |
| |
|
|
|
|
|
| Auxetic |
796 |
1106 |
777 |
774 |
807 |
| |
826 |
1145 |
772 |
674 |
935 |
| |
793 |
1121 |
756 |
729 |
631 |
| |
784 |
1098 |
757 |
709 |
856 |
| |
790 |
1127 |
572 |
738 |
779 |
| |
783 |
1116 |
784 |
714 |
832 |
Table A5 presents the vertical ground reaction forces (GRFs) measured in Newtons (N) for Subjects B, C, D, E, and F (without braces). Data is grouped into three conditions: barefoot walking, walking with standard insoles ("No auxetic"), and walking with experimental auxetic foam insoles ("Auxetic"). Subject A is excluded from this table to isolate specific neuropathic categories, with Subject C serving as the representative for diabetic neuropathy in this analysis.
Table A6.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects A, B, D, E, and F (braced condition).
Table A6.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects A, B, D, E, and F (braced condition).
| |
PARAMETER / C OMPARISON |
RESULT |
| KRUSKAL-WALLIS |
Observed Value |
192.254 |
| |
Critical Value |
5.991 |
| |
p |
<0.0001 |
| |
Degrees of Freedom |
2 |
| |
alpha |
0.05 |
| |
|
K |
| |
|
p<0.0001 |
| PAIRWISE COMPARISONS |
Insole vs. Neuropathy |
Significant* |
| |
p |
0.002 |
| |
|
p=0.002 |
| |
Insole vs. GRFS |
Significant* |
| |
p |
<0.0001 |
| |
|
p<0.0001 |
| |
Neuropathy vs. GRFS |
Significant* |
| |
p |
<0.0001 |
|
|
p<0.0001 |
Table A6 Results were significant (p < .0001) at the
α =0.05 level. Data from Subject C (diabetic neuropathy) were excluded to maintain a balanced assessment of various neuropathic etiologies, as diabetic neuropathy was already represented by Subject A
.
Table A7.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects A, B, D, E, and F (without braces).
Table A7.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects A, B, D, E, and F (without braces).
| |
PARAMETER / COMPARISON |
RESULT |
| KRUSKAL-WALLIS |
Observed Value |
192.254 |
| |
Critical Value |
5.991 |
| |
p |
<0.0001 |
| |
Degrees of Freedom |
2 |
| |
alpha |
0.05 |
| |
|
K |
| |
|
p<0.0001 |
| PAIRWISE COMPARISONS |
Insole vs. Neuropathy |
Significant* |
| |
p |
0.002 |
| |
|
p=0.002 |
| |
Insole vs. GRFS |
Significant* |
| |
p |
<0.0001 |
| |
|
p<0.0001 |
| |
Neuropathy vs. GRFS |
Significant* |
| |
p |
<0.0001 |
| |
|
p<0.0001 |
Table A7 Results were significant (p < .0001) at the
α = 0.05 level. Data from Subject C (diabetic neuropathy) were excluded to maintain a balanced assessment of various neuropathic etiologies, as diabetic neuropathy was already represented by Subject A.
Table A8.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects B,C, D, E, and F (braced condition).
Table A8.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects B,C, D, E, and F (braced condition).
| |
PARAMETER / COMPARISON |
RESULT |
| KRUSKAL-WALLIS |
Observed Value |
192.254 |
| |
Critical Value |
5.991 |
| |
p |
<0.0001 |
| |
Degrees of Freedom |
2 |
| |
alpha |
0.05 |
| |
|
K |
| |
|
p<0.0001 |
| PAIRWISE COMPARISONS |
Insole vs. Neuropathy |
Significant* |
| |
p |
0.002 |
| |
|
p=0.002 |
| |
Insole vs. GRFS |
Significant* |
| |
p |
<0.0001 |
| |
|
p<0.0001 |
| |
Neuropathy vs. GRFS |
Significant* |
| |
p |
<0.0001 |
| |
|
p<0.0001 |
Table A8 Results were significant (p < .0001) at the
α =0.05 level. Data from Subject A (diabetic neuropathy) were excluded to maintain a balanced assessment of various neuropathic etiologies, as diabetic neuropathy was already represented by Subject C.
Table A9.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects B,C, D, E, and F (without braces).
Table A9.
Kruskal-Wallis and Dunn’s post-hoc test results for Subjects B,C, D, E, and F (without braces).
| |
PARAMETER / COMPARISON |
RESULT |
| KRUSKAL-WALLIS |
Observed Value |
192.254 |
| |
Critical Value |
5.991 |
| |
p |
<0.0001 |
| |
Degrees of Freedom |
2 |
| |
alpha |
0.05 |
| |
|
K |
| |
|
p<0.0001 |
| PAIRWISE COMPARISONS |
Insole vs. Neuropathy |
Significant* |
| |
p |
0.002 |
| |
|
p=0.002 |
| |
Insole vs. GRFS |
Significant* |
| |
p |
<0.0001 |
| |
|
p<0.0001 |
| |
Neuropathy vs. GRFS |
Significant* |
| |
p |
<0.0001 |
| |
|
p<0.0001 |
Table A9 Results were significant (p < .0001) at the
α =0.05 level. Data from Subject A (diabetic neuropathy) were ex cluded to maintain a balanced assessment of various neuropathic etiologies, as diabetic neuropathy was already repre sented by Subject C.