Submitted:
21 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Preparation of Stromal Vascular Fractions (SVFs)
2.3. In Vitro IL-10 Treatment of SVFs
2.4. In Vivo IL-10 Injection into Adipose Tissue
2.5. RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction (Q-PCR)
2.6. Western Immunoblot Analysis
2.7. Kupffer Cell Purification
2.8. Flow Cytometry Analysis
2.9. Insulin Treatment
2.10. Plasma DPP4 Activity
2.11. Serum Alanine Aminotransferase (ALT) Assay
2.12. Enzyme-Linked Immunosorbent Assay (ELISA)
2.13. Intraperitoneal Glucose Tolerance Test (IPGTT)
2.14. Statistical Analysis
3. Results
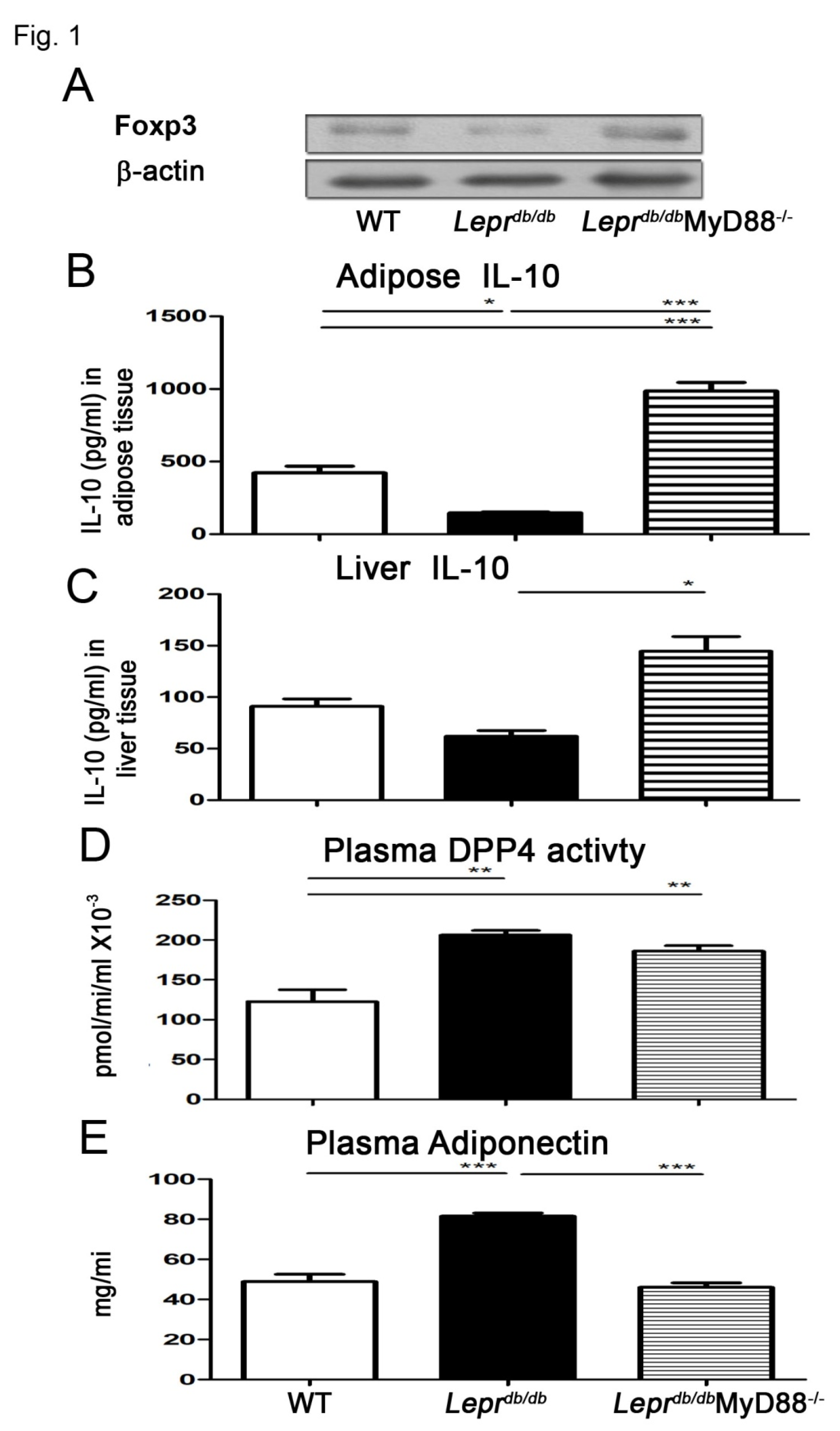
3.1. Leprdb/dbMyD88−/− Mice Exhibited Increased IL-10 Levels and Foxp3 Expression in Adipose Tissue SVFs and Reduced Circulating Adiponectin and DPP4 Activity
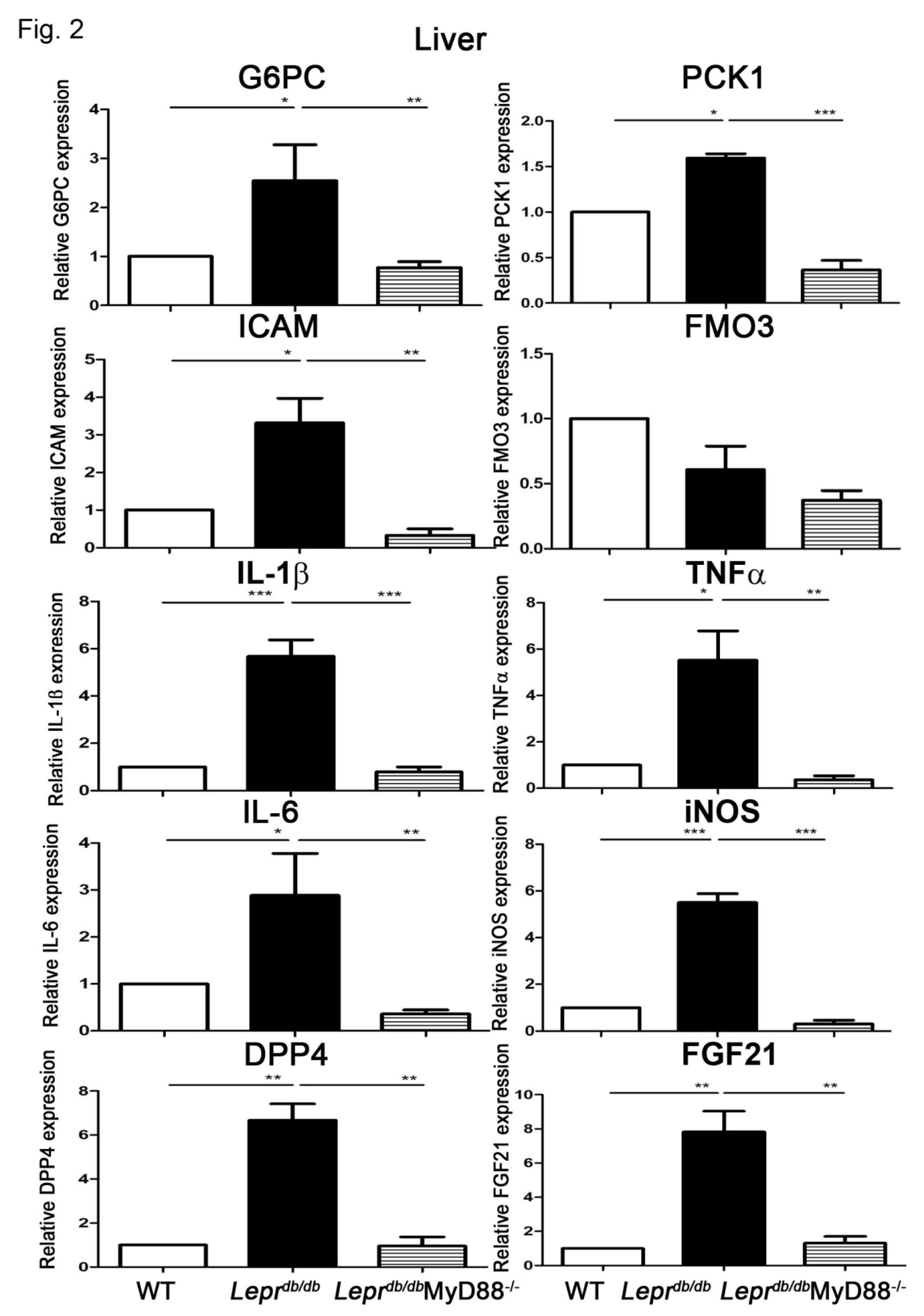
3.2. Leprdb/dbMyD88−/− Mice Displayed Reduced Hepatic Inflammatory and Gluconeogenic Gene Expression
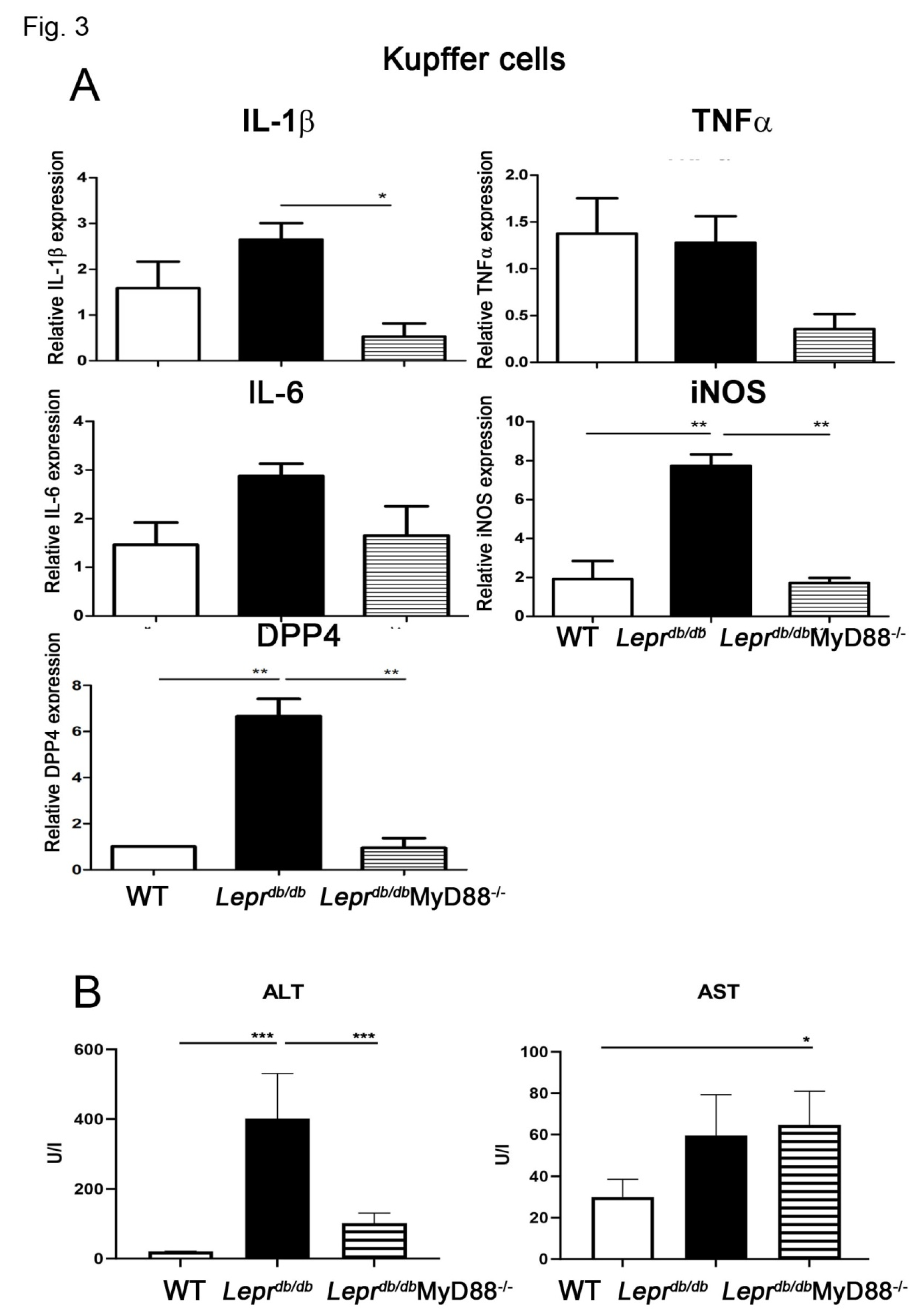
3.3. MyD88 Deficiency Reduced Inflammatory Gene Expression in Kupffer Cells and Improved Liver Function
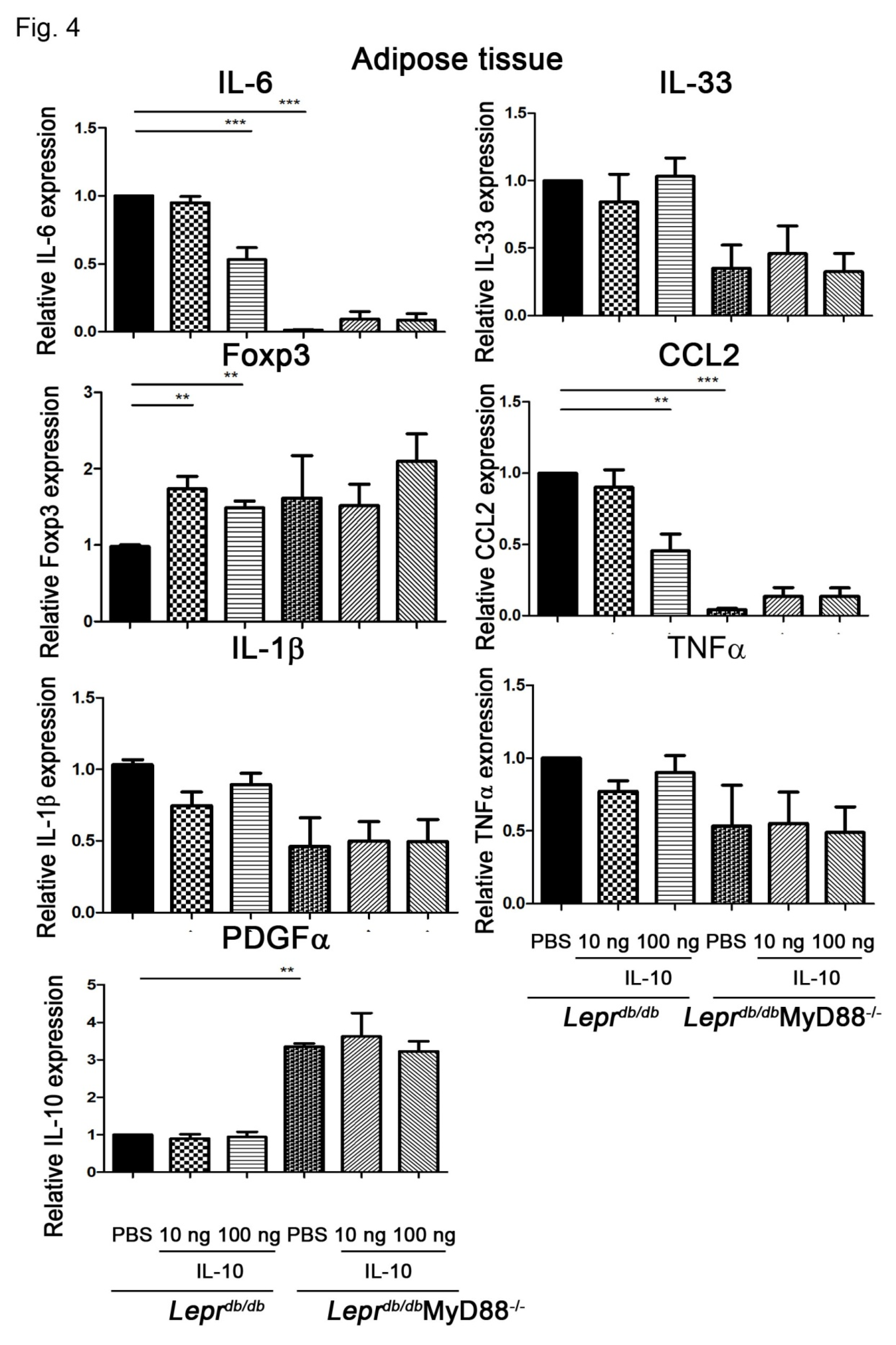
3.4. SVFs from Leprdb/dbMyD88−/− Mice Exhibited Reduced Inflammatory Cytokines and Increased PDGFα Expression and IL-10 Treatment Recapitulated These Effects
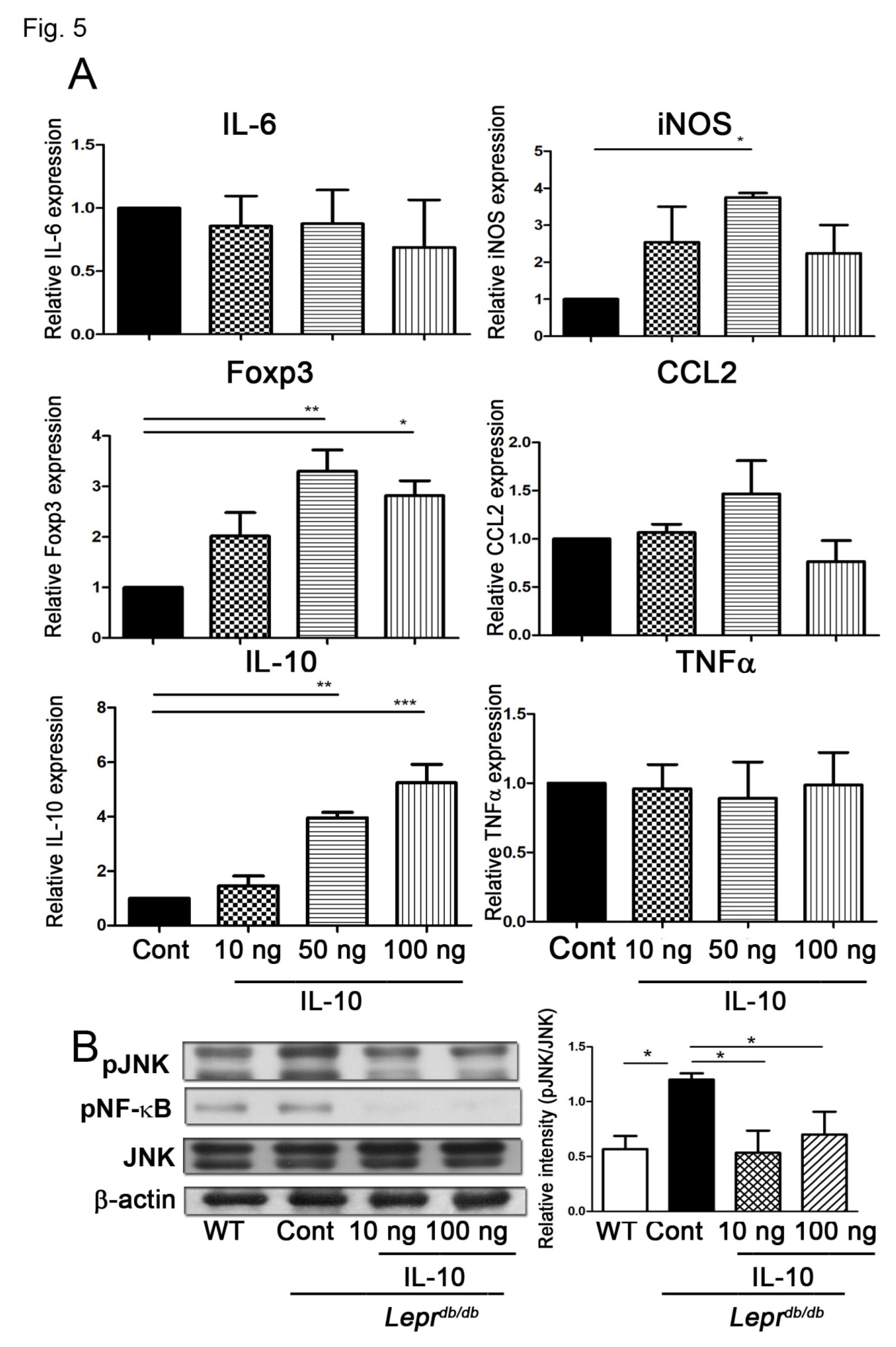
3.5. IL-10 Injection Enhanced Foxp3 and IL-10 Expression and Suppressed JNK and NF-κB Signaling in Adipose SVFs
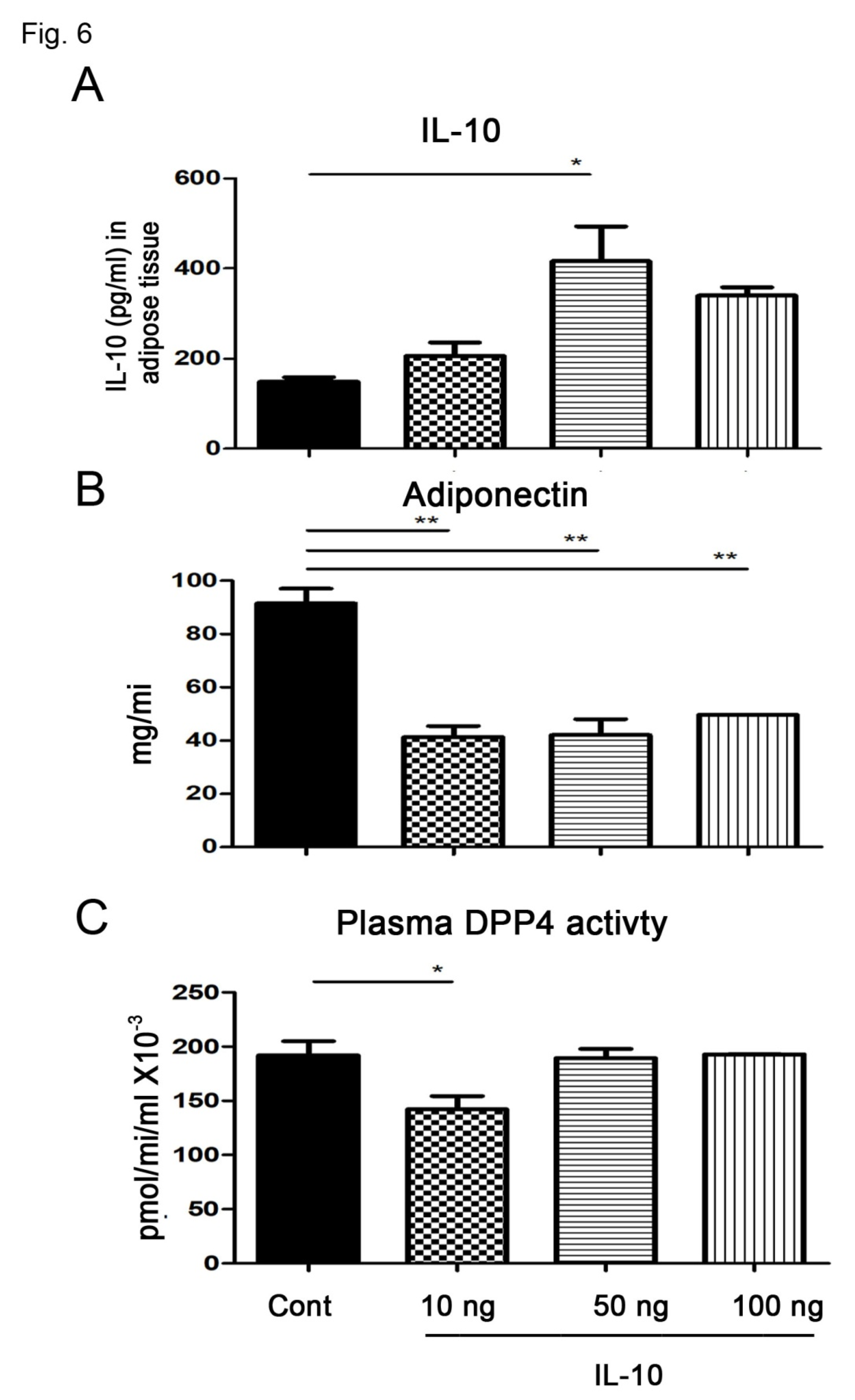
3.6. IL-10 Injection Reduced Circulating Adiponectin and DPP4 Activity
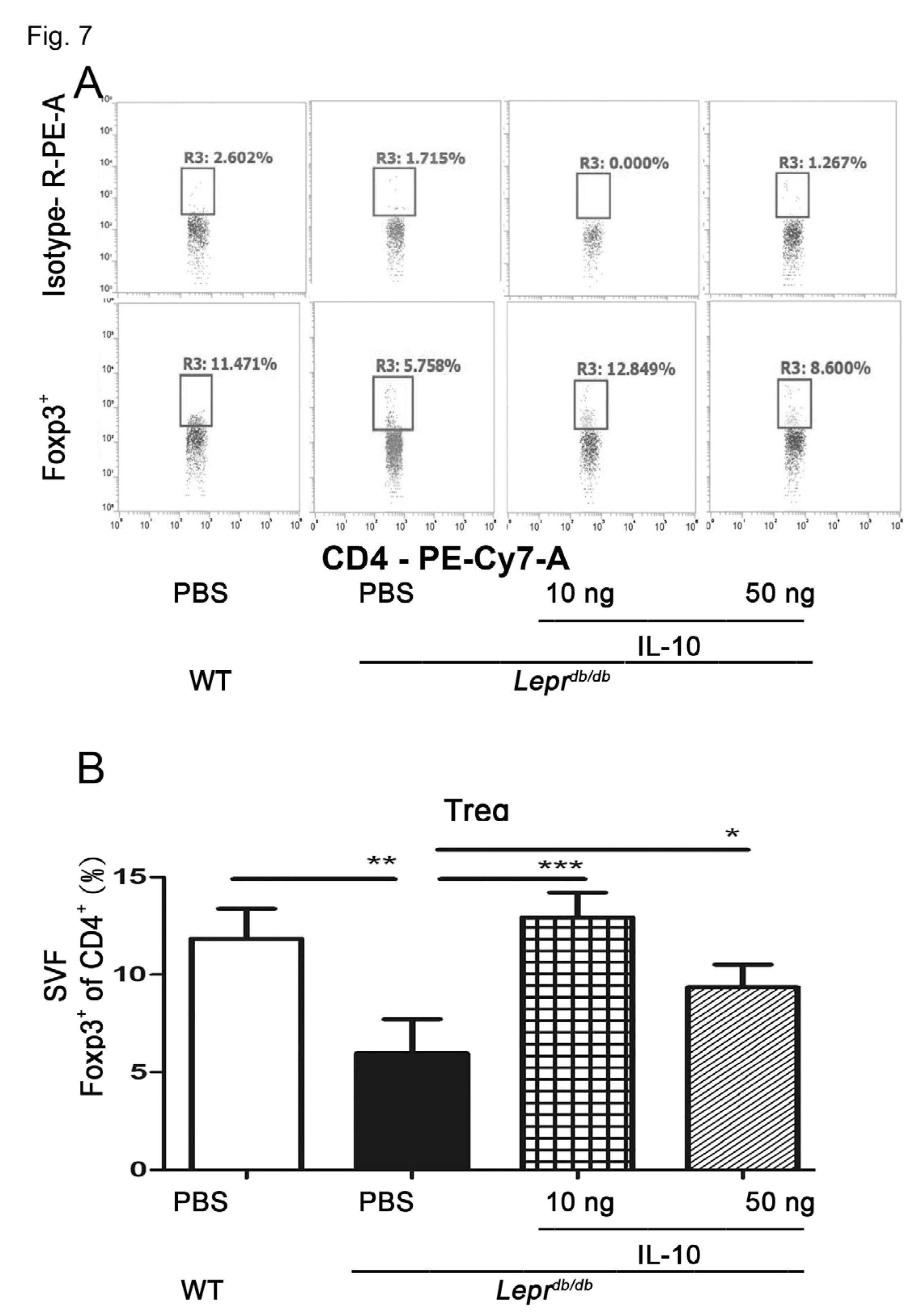
3.7. IL-10 Promoted CD4+ Regulatory T-Cell Accumulation in Adipose Tissue
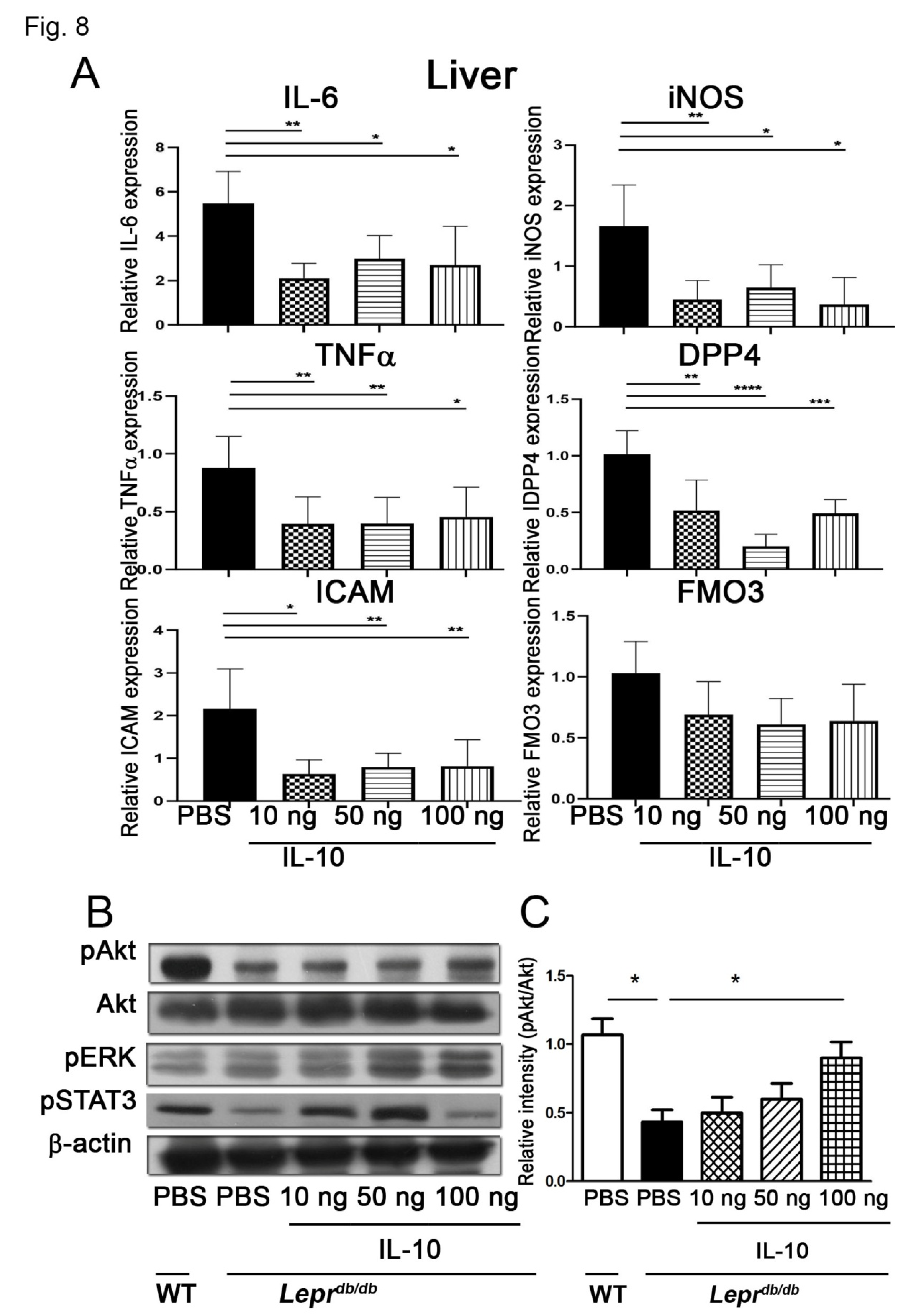
3.8. IL-10 Reduced ICAM, TNF-α, IL-6, DPP4, and iNOS mRNA Expression and Increased pAkt and pERK Levels in the Liver
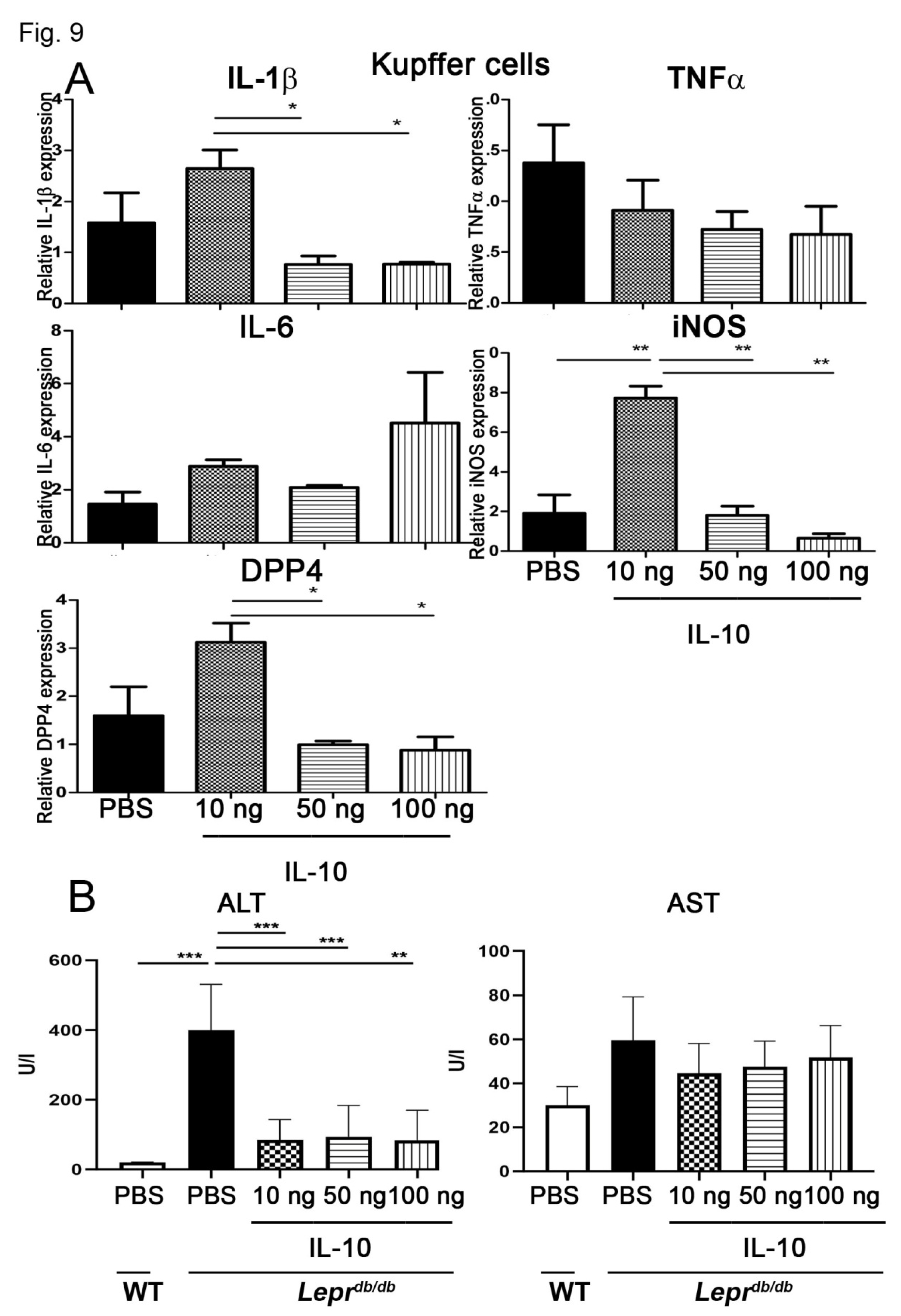
3.9. IL-10 Reduced Inflammatory Gene Expression in Kupffer Cells and Improved Liver Function
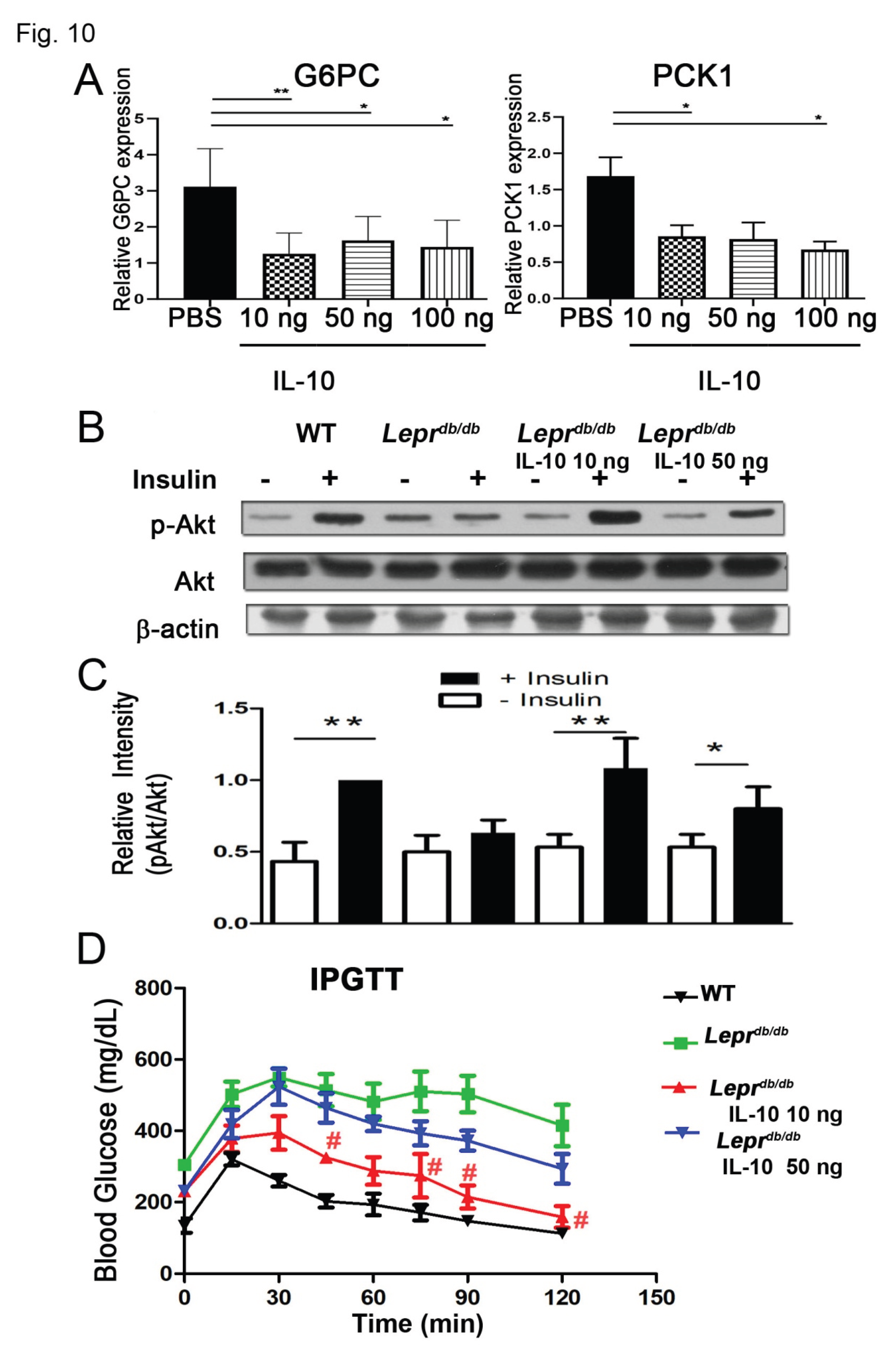
3.10. IL-10 Reduced Hepatic Gluconeogenic Gene Expression
3.11. IL-10 Enhanced Insulin Sensitivity and Improved Glucose Tolerance in Diabetic Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviation
| DM | Diabetes mellitus |
| DKA | Diabetic ketoacidosis |
| SVFs | Stromal vascular fractions |
| PCK1 | Cytosolic form of Phosphoenolpyruvate carboxykinase |
| G6PC | Glucose 6-phosphatase catalytic subunits |
| TNF-α | Tumor Necrosis Factor-α |
| IL-1β | Interleukin-1β |
| IL-33 | Interleukin-33 |
| CCL2 | Monocyte chemoattractant protein-1 |
| iNOS | Inducible nitric oxide synthase |
| DPP4 | Dipeptidyl peptidase-4 |
| ALT | Plasma alanine aminotransferase |
| IL-10 | Interleukin-10 |
| Foxp3 | Forkhead box protein P3 |
| MSC | Mesenchymal stem cells |
| FGF21 | Fibroblast growth factor 21 |
| ATM | Adipose tissue macrophage |
| NATM | Non-adipose tissue macrophage |
| PDGFα | Platelet-derived growth factor |
| STAT3 | signal transducer and activator of transcription 3 |
| mTOR | mammalian target of tapamycin |
| WAT | white adipose tissue |
| MyD88 | Myeloid differentiation factor 88 |
References
- Sulen, A; Aouadi, M. Fed Macrophages Hit the Liver’s Sweet Spot with IL-10. Molecular cell. 2020, 79(1), 1–3. [Google Scholar] [CrossRef] [PubMed]
- Huh, JY; Park, YJ; Ham, M; Kim, JB. Crosstalk between adipocytes and immune cells in adipose tissue inflammation and metabolic dysregulation in obesity. Molecules and cells 2014, 37(5), 365–71. [Google Scholar] [CrossRef] [PubMed]
- Dollet, L; Lundell, LS; Chibalin, AV; Pendergrast, LA; Pillon, NJ; Lansbury, EL; et al. Exercise-induced crosstalk between immune cells and adipocytes in humans: Role of oncostatin-M. Cell Rep Med. 2024, 5(1), 101348. [Google Scholar] [CrossRef] [PubMed]
- Tian, J; Zhao, Y; Wang, L; Li, L. Role of TLR4/MyD88/NF-kappaB signaling in heart and liver-related complications in a rat model of type 2 diabetes mellitus. J Int Med Res. 2021, 49(3), 300060521997590. [Google Scholar] [CrossRef] [PubMed]
- Degirmenci, I; Ozbayer, C; Kebapci, MN; Kurt, H; Colak, E; Gunes, HV. Common variants of genes encoding TLR4 and TLR4 pathway members TIRAP and IRAK1 are effective on MCP1, IL6, IL1beta, and TNFalpha levels in type 2 diabetes and insulin resistance. Inflamm Res. 2019, 68(9), 801–14. [Google Scholar] [CrossRef]
- Boonstra, A; Rajsbaum, R; Holman, M; Marques, R; Asselin-Paturel, C; Pereira, JP; et al. Macrophages and myeloid dendritic cells, but not plasmacytoid dendritic cells, produce IL-10 in response to MyD88- and TRIF-dependent TLR signals, and TLR-independent signals. J Immunol. 2006, 177(11), 7551–8. [Google Scholar] [CrossRef] [PubMed]
- Cintra, DE; Pauli, JR; Araujo, EP; Moraes, JC; de Souza, CT; Milanski, M; et al. Interleukin-10 is a protective factor against diet-induced insulin resistance in liver. J Hepatol. 2008, 48(4), 628–37. [Google Scholar] [CrossRef] [PubMed]
- Toda, G; Soeda, K; Okazaki, Y; Kobayashi, N; Masuda, Y; Arakawa, N; et al. Insulin- and Lipopolysaccharide-Mediated Signaling in Adipose Tissue Macrophages Regulates Postprandial Glycemia through Akt-mTOR Activation. Molecular cell. 2020, 79(1), 43–53 e4. [Google Scholar] [CrossRef] [PubMed]
- Lee, YS; Wollam, J; Olefsky, JM. An Integrated View of Immunometabolism. Cell. 2018, 172(1-2), 22–40. [Google Scholar] [CrossRef] [PubMed]
- Sakai, N; Van Sweringen, HL; Schuster, R; Blanchard, J; Burns, JM; Tevar, AD; et al. Receptor activator of nuclear factor-kappaB ligand (RANKL) protects against hepatic ischemia/reperfusion injury in mice. Hepatology 2012, 55(3), 888–97. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P; Santner-Nanan, B; Hu, M; Skarratt, K; Lee, CH; Stormon, M; et al. IL-10 Potentiates Differentiation of Human Induced Regulatory T Cells via STAT3 and Foxo1. J Immunol. 2015, 195(8), 3665–74. [Google Scholar] [CrossRef] [PubMed]
- Hosoi, T; Yokoyama, S; Matsuo, S; Akira, S; Ozawa, K. Myeloid differentiation factor 88 (MyD88)-deficiency increases risk of diabetes in mice. PLoS One 2010, 5(9). [Google Scholar] [CrossRef] [PubMed]
- Couper, KN; Blount, DG; Riley, EM. IL-10: the master regulator of immunity to infection. J Immunol. 2008, 180(9), 5771–7. [Google Scholar] [CrossRef] [PubMed]
- Robert, S; Gysemans, C; Takiishi, T; Korf, H; Spagnuolo, I; Sebastiani, G; et al. Oral delivery of glutamic acid decarboxylase (GAD)-65 and IL10 by Lactococcus lactis reverses diabetes in recent-onset NOD mice. Diabetes 2014, 63(8), 2876–87. [Google Scholar] [CrossRef] [PubMed]
- Thompson, MA; Ohnuma, K; Abe, M; Morimoto, C; Dang, NH. CD26/dipeptidyl peptidase IV as a novel therapeutic target for cancer and immune disorders. Mini Rev Med Chem. 2007, 7(3), 253–73. [Google Scholar] [CrossRef] [PubMed]
- Rochette, L; Zeller, M; Cottin, Y; Vergely, C. Diabetes, oxidative stress and therapeutic strategies. Biochimica et biophysica acta 2014, 1840(9), 2709–29. [Google Scholar] [CrossRef]
- Chen, LW; Chen, PH; Yen, JH. Inhibiting adipose tissue M1 cytokine expression decreases DPP4 activity and insulin resistance in a type 2 diabetes mellitus mouse model. PLoS One 2021, 16(5), e0252153. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, JP; Richards, AA; Hickman, IJ; Macdonald, GA; Prins, JB. Adiponectin--a key adipokine in the metabolic syndrome. Diabetes, obesity & metabolism 2006, 8(3), 264–80. [Google Scholar]
- Iwabu, M; Yamauchi, T; Okada-Iwabu, M; Sato, K; Nakagawa, T; Funata, M; et al. Adiponectin and AdipoR1 regulate PGC-1alpha and mitochondria by Ca(2+) and AMPK/SIRT1. Nature 2010, 464(7293), 1313–9. [Google Scholar] [CrossRef] [PubMed]
- Maeda, N; Shimomura, I; Kishida, K; Nishizawa, H; Matsuda, M; Nagaretani, H; et al. Diet-induced insulin resistance in mice lacking adiponectin/ACRP30. Nat Med. 2002, 8(7), 731–7. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



