1. Introduction
Lumpy skin disease (LSD) is caused by the Lumpy Skin Disease Virus (LSDV) from the family Poxviridae, genus Capripoxvirus (CapV) the Neethling strain prototype[
1] characterized by fever, nodules on the skin, mucous membranes and internal organs, emaciation, enlarged lymph nodes, oedema of the skin, and sometimes death [
2]. The disease is a highly contagious transboundary infection that has a significant economic loss in the livestock industry as it can cause a temporary reduction in milk production, temporary or permanent sterility in bulls, damage to hides, and, occasionally, death [
3].
The lumpy skin disease virus was first reported in 1983 in the northwestern part of Ethiopia in the Amhara National Regional State surrounding the southwest part of Lake Tana [
4]. Because of the wide distribution of the disease and the size and structure of the cattle population in Ethiopia, LSDV is likely one of the most economically important livestock diseases in the country [
5]. The magnitude of LSDV occurrence varied across different agro-ecological zones of Ethiopia, ranging from 6.1% [
6] to 20.8% [
7].
Capripox viruses, responsible for Lumpy Skin Disease in cattle, Sheep pox, and Goat pox, cause significant socio-economic harm, necessitating the constant development of effective diagnostic tools to manage potential wide-scale outbreaks [
8]. Diagnosis of LSDV is the fundamental step for disease control, prevention, and eradication by implementing strategies like vaccination campaigns, controlling vectors, and establishing regional and global collaboration [
9]. The recommended diagnostic tools for LSDV are real-time Polymerase Chain Reaction (PCR), electron microscopy, sequencing, virus neutralization testing (VNT), Western blot, enzyme-linked immunosorbent assay, and immunofluorescence antibody testing (IFAT) [
10].
The advantages of pAb for developing tools for assay-specific target development and detection in basic research, diagnostics, and biomarker discovery have been extensively accepted [
11]. Thus, pAb can be harvested from mammals, despite the requirement of a large number of animals to undergo invasive procedures such as antigen injection and blood collection [
12]. However, pAbs can be produced from chickens using non-invasive protocols that result in higher yield, lower cross-reactivity with mammalian proteins, improved stability and resistance to degradation, cost-effectiveness, detection of conserved epitopes across species for targeting proteins with high sequence similarity, and use in a variety of ELISA formats [
13,
14]. Egg yolk antibodies provide higher yield per animal, the procedure of harvesting is ethical, and simple [
15], the reagents and extraction facelifts are cheap, purified antibodies have higher avidity and sensitivity with broader recognition of epitopes, and reduced interference in immunoassays [
16].
Chicken can transfer IgY from the plasma to egg yolks from the 14th–21st days following a single inoculation. High antibody titers can be achieved after 1-3 doses resulting in continuous IgY production. Chickenscan lay eggs continuously for around 72 weeks before their laying capacity declines, making IgY technology an abundant polyclonal antibody manufacturing process [
17].
Chickens are the best alternative animal for pAb production, helping to overcome the constraints of the conventional approach[
18]. In 1996, the European Center for the Validation of Alternative Methods (ECVAM) workshop proposed that chicken egg yolk antibody can be used instead of mammalian IgG for therapeutic use in medicine and biological research[
16,
19]. This is because egg yolk provides high-quality antibodies that can be easily produced in low-cost facilities [
20] without inducing animal damage or suffering [
21].
The distinct immunological properties of avian species offer advantages for producing diverse and highly specific pAb based on the principle that the greater the phylogenetic distance between the antigen donor and the antigen producer, the greater the potential for generating a strong antibody response [
22].
Nowadays, IgYtechnology is a new frontier for biological products such that the development of IgY-based immunoassay formats [
16]. For instance,IgY-based ELISA for the detection of Gentamicin residues in animal products was developed after immunizing chicken with Gentamicin conjugated with Bovine serum albumin (BSA) and the developed IgY-based indirect competitive ELISA was able to recover analyte from fresh milk, pork and chicken samples from 69.82% to 94.32% specificity[
23].On the other hand, both sensitive and specific sandwich ELISA was developed for the detection of toxigenic V. cholerae strains in clinical and environmental samples based on avian antibodies (IgY) targeting outer membrane protein (OmpW) and cytotoxin B (CtxB) antigens of V. cholera [
24]. Chicken egg-derived IgY antibodies were also used for the development of anti-Hepatitis B virus antigen (anti-HBsAg) pAb for application as capture agents within a sandwich ELISA [
25]. Moreover, IgY technologycan also be used for developing and evaluating avian immunoglobulins for the in vitro detection of biomolecules [
26].
1.1. Statement of the Problem
The need for the development of reliable and practical diagnostic immunoassay tools for the detection of bovine antibodies against LSDV is believed to be of paramount importance, helping researchers to improve serological surveillance [
11]. However, there was no single trial pretending to investigate the possibilities of using IgY for ELISA kit development in Ethiopia. Despite the high prevalence of LSDV in the country, the available diagnostic tools are expensive, and time-consuming, taking days to weeks to run a single test.
Currently, AHI is using virus isolation, VNT, and PCR for the detection of LSDV which is challenging to undergo serosurveillance [
27]. However, an enzyme-linked immunosorbent assay (ELISA) is recognized as an efficient diagnostic tool with less time-consuming and easier procedures than the viral neutralization test to measure antibody levels [
8,
28].
The development of a reliable and sensitive tool for LSDV necessitates the use of antigen-specific polyclonal antibodies (pAb). In this case, avian IgY generated from chicken egg yolkcan best substitute the mammalian IgG, which is commonly harvested from rabbits. Therefore, this research was conducted to fill the gap in large-scale LSDV serosurveillance by producing IgY, the first step in developing a faster, cost-effective alternative to available methods like virus isolation, VNT, and PCR, enabling more efficient testing and improved understanding of LSDV prevalence for better control strategies.
1.2. Objective of the Study
1.2.1. General Objective
To generate and characterize polyclonal IgY antibodies produced in chickens immunized with the lumpy skin disease virus (LSDV) vaccinal strain, and to evaluate their immunological properties for potential application in LSDV detection and research.
1.2.2. Specific Objectives
To isolate and purify LSDV-specific IgY antibodies from chicken egg yolk following immunization.
To evaluate the temporal antibody response and production kinetics of IgY against LSDV.
To determine the antigen-binding specificity and virus-neutralizing activity of egg yolk–derived IgY toward LSDV vaccinal strain antigens.
2. Materials and Methods
2.1. Study Site
The experimental work was conducted at the Animal Health Institute (AHI), Sebeta, Ethiopia, between January and May 2024. The institute provided the required facilities for cell culture, vaccinal strain propagation, virus titration, RT-PCR–based identity confirmation, virus neutralization assays, and for the production, purification, and quantification of egg yolk IgY antibodies.
Molecular weight characterization of the purified IgY was performed at the AU-PANVAC Molecular Biology Laboratory in Bishoftu, Ethiopia. Collaboration between the two laboratories enabled comprehensive analysis and confirmation of IgY production, purity, and molecular integrity.
2.2. Study Design
The experimental study was conducted from January to May 2024, for 42 days, to produce and evaluate specific immunoglobulin Y (IgY) antibodies against the lumpy skin disease virus vaccinal strain from chicken egg yolk for the development of an enzyme-linked immunosorbent assay. Virus adaptation and titration were performed, followed by the allocation of chickens (n=10) into two groups: 6 in the experimental group and 4 in the control group, randomly. The experimental group was immunized with two doses of the LSDV vaccinal strain, while the control group received a placebo (PBS). Two injections were administered on day 0 and day 15 into both sides of the pectoralis muscles [
21]).
2.3. Experimental Chicken
Ten healthy 52-week-old, the same batch, Bovans Brown layer chicks, were bought from a local farm in Sebeta town and managed in controlled environments for housing, inoculation, and sample collection. The chickens were allocated into two groups and housed separately in mesh wire enclosures. For a successful recovery from stress caused by transportation, feed, and environmental alterations, the chickens were given a week to acclimate before the trial commenced. Chickens were managed in a good state of welfare: they were healthy, comfortable, well-nourished, safe, able to express natural behaviors, and free from pain, fear, and distress, as per the OIE and NCC Animal Welfare Guidelines [
29].
2.4. Sample Size
The sample size was determined based on feasibility and consistency with previous exploratory IgY immunization and kinetic studies. A total of ten (n = 10) egg-laying chickens were included and allocated into an experimental group (n = 6) and a control group (n = 4). This sample size was considered adequate to evaluate temporal changes in IgY concentrations within and between groups under a repeated-measures experimental design.
2.5. Preparation, Adaptation, and Titration of Lumpy Skin Disease Virus Vaccine
Preparation and Adaptation of the Lumpy Skin Disease Virus Vaccinal Strain
To eliminate impurities, the Minimum Essential Medium (MEM) with L-glutamine was filtered via a 0.22 µm filter. The pH of the MEM was measured with a JENWAY™ 3310 pH meter and adjusted to 7.50, which is ideal for Vero cell culture, by carefully titrating with acidic buffer. The medium was thenenriched with 10% (v/v) Fetal Bovine Serum (FBS) to supply necessary growth factors, as well as 2% (v/v) streptomycin sulfate and Amphotericin B 250 µg/mL agents to avoid bacterial and fungal contamination [
30].
Vero cells were first seeded in T-75 culture flasks and allowed to reach confluency. Afterward, cells were trypsinized using a 2.5% trypsin enzyme solution to detach adherent cells, resulting in a suspension with a final concentration of 2 × 10⁵ cells/ml. The cell concentration was confirmed using the Countess® II Automated Cell Counter. The cell suspension was then combined with the complete medium prepared earlier.
Vero cells in complete media were incubated at 37 °C with 5% CO₂, 98% O₂, and about 95% humidity. The cells were inspected daily using an inverted microscope until they reached 60-80% confluence, which usually occurred within 2 to 3 days. The entire procedure was implemented according to the protocol of Sandell and Sakai (2011).
2.6. Titration of Lumpy Skin Disease Virus
A live LSDV vaccine with a titer of 3TCID50 was obtained from the National Veterinary Institute (NVI) and kept at -80 °C to ensure stability. To adapt the vaccinal virus, it was subjected to two sequential passages on Vero cell lines until optimal cytopathic effects (CPE) were observed. The LSDV vaccine, previously frozen at -80 °C after dilution with phosphate-buffered saline (PBS), was removed from the deep freezer and immediately thawed in a 37 °C water bath. The vaccine was then gently mixed to ensure homogeneity, seeded on Vero cells at the proper confluency (60–80%), and cultivated in a CO2 incubator for 7 days. The Vero cells used in this study were sourced from the Animal Health Institute Virus Isolation and Cell Culture Laboratory.
Considerable cytopathic effects, such as cell rounding, detachment, and disruption of the cell monolayer, were detected in Vero cells infected with the LSDV at the first and second passages, and freeze-thaw cycles were undertaken. The goal of freeze-thawing was to lyse the cells and release the virus using temperature changes.
The virus was freeze-thawed by taking it from the -80 °C deep freezer and placing it in a 37 °C water bath. The virus was heated until the ice melted entirely, under close monitoring. After being thawed, the sample was quickly returned to the -80 °C deep freezer to remain there for around 20 minutes until refrozen.
The freeze-thaw cycle was performed three times to produce fluctuations in temperature that disrupt cellular contents in the cytoplasm, allowing the virus to be released for efficient harvesting using a hypotonic burst to isolate virions from infected cultured cells [
31]. Then, the final suspension was collected in a Falcon tube and centrifuged at 3000 rpm and 4 °C for 10 minutes using a Thermo Scientific/Jouan CR4i refrigerated centrifuge to separate the virus from cellular debris.
The harvested virus was theninoculated into a fresh Vero cell line at about 60–80% confluence. Following the detection of optimal CPEon the 7th day [
2], a sample from the second passage was confirmed using RT-PCR to confirm the presence of LSDV. The viral titer was estimated using a tenfold serial dilution on 96-well micro-titration plates.
The median tissue culture infectious dose (TCID₅₀) of the viral stock was determined using the Reed–Muench method [
32]. Viral infectivity was calculated based on the proportion of positive cultures across serial dilutions. To ensure adequate immunogenicity, the vaccinal dose was set at 10³ TCID₅₀/mL.
2.7. Immunization of Chicken, Sample Collection, and IgY Extraction
Immunization and Sample Collection
Chickens in the experimental group were inoculated intramuscularly with a standardized dose of 10³ TCID₅₀/mL of LSDV diluted in phosphate-buffered saline (PBS). Using a 1 mL insulin syringe, the inoculum was divided into two equal volumes and administered into both sides of the pectoralis muscles of each chicken [
21]. Chickens in the control group received 1 mL of PBS without virus following the same injection protocol. A booster inoculation was administered to both experimental and control groups during the second week, using the same virus dose and placebo treatment, respectively.
Eggs and serum samples were collected weekly from day 7 to day 42 (
Table 1), with subsequent evaluation of IgY sourced from serum and egg yolk using VNT to assess the immune response to the vaccinal strain [
33,
34]. Daily visual inspections were performed to monitor forlesion development, and no visible pox lesions were found. Eggs collected were kept at +4 °C, and blood samples of 3 ml were drawn from each chicken’s brachial wing vein, kept at room temperature overnight, and serum was transferred into cryovials.
2.8. Extraction of Chicken IgY
Fresh, dark brown-shelled eggs were carefully broken, and the yolks were separated from the egg whites using a modified extraction protocol based on Polson (1980), as described by 1980), as described by [
35].
The procedure started with physically breaking the eggshells and gently transferring the yolks to an egg strainer, allowing the majority of the albumen (egg white) to drain away. The residual albumen on the vitelline membrane (yolk membrane) was removed by gently rolling the yolks over white paper. The vitelline membrane was thensliced with a lancet, and the yolk was transferred to a 50-mL Falcon tube. Each egg supplied roughly 15 ml of yolk (V1).
The yolk was blended with PBS in a volume twice that of the yolk, yielding a final volume of 45 ml. The mixture was vortexed before and after adding 3.5% PEG 6000 (w/v) to the total volume (V2). The solution was agitated on a thermo shaker incubator at 300 rpmfor 10 minutes, preceding centrifugation at 4 °C and 3500 rpm for 15 minutes. The supernatant was carefully decanted twice through a sterile tube using folded filter paper, producing a new volume (V3).
Afterward, 8.5% PEG 6000 (w/v) was added to the volume (V3), vortexed, and incubated on the thermo shaker at 300 rpm. Subsequently, the tubes were centrifuged at 4 °C for 15 minutes at 3500 rpm, and the supernatant was removed. The resulting pellet was vigorously vortexed and homogenized in 1 ml of PBS with a glass rod. PBS was thenadded to raise the total volume to 10 mL (V4).
The solution was subsequently blended with 12% PEG 6000 (w/v), shaken on a thermo shaker at 300 rpm for 10 minutes, thencentrifuged at 4 °C for 15 minutes at 3500 rpm. The supernatant was eliminated, and the pellet was resuspended and dissolved in 1.8 mL of PBS, vortexed, well-mixed with a glass rod, and transferred to a 2 mL cryovial for storage.
Lastly, the extracts were purified utilizing a Membra-Cel® dialysis membrane with a 14 kDa molecular weight cut-off (MWCO) to take away PEG 6000 and guarantee complete IgY purification against PBS.
2.9. Preparation of Dialysis Bag and Purification of the IgY Extract
The Membra-Cel
® dialysis membrane with a 14 kDa molecular weight cut-off (MWCO) was employed for the purification of IgY against PBS buffer and PEG. A 5 mM EDTA solution was utilized as the dialysate to facilitate the removal of low molecular mass compounds from the dialysis bag, following the protocol outlined by protocol outlined by [
17].
Dialysis tubing was cut into ten pieces, each 15 cm in length, and boiled in 1000 ml of 5 mM EDTA solution for 5 minutes using a hot plate. After boiling, the solution was decanted, and the dialysis bags were washed three times with distilled water. This procedure was repeated once more. At last, the dialysis bags were heated in distilled water for another 10 minutes before being cooled to 4 °C and stored until required.
The day after the dialysis bags were prepared, the tubing was cleaned with saline water, and the IgY extract was transferred into the bags, which were then tightly secured with thread. The firmly sealed dialysis bags were immersed in a beaker of 0.1% saline water and stirred overnight over a magnetic stirrer.
The saline solution was replaced with PBS the next morning, and dialysis was continued for three more hours. Following dialysis, the IgY extract was carefully removed from the beaker that held the dialysis tube using tweezers. The extract was then transferred from the bag to a 15-ml Falcon tube using a volumetric transfer pipette and stored at -20 °C until further examination.
2.10. Detection and Quantification of Antibodies
Virus neutralization Test
VNT was performed to evaluate the efficiency of IgY antibodies produced against LSDV and their ability to neutralize LSDV, preventing it from infecting the Vero cell line. This serological test was implemented to assess whetherchickens’IgYagainst LSDV prevents the infection ofthe Vero cell line, which enabled us to detect pAb in both serum and IgY.
2.11. Nano Drop Spectrophotometer
The NeoDotMicrovolume spectrophotometer was used to evaluate the concentration and purity of chicken IgY antibodies at an absorbance of 280 nm. In the beginning, 4 μL of IgY-free PBS was added to the bottom pedestal and loweredto the upper pedestal arm for 2-3 minutes to form a liquid column. Water was thenremoved from both pedestals using a dry, lint-free lab wipe. To perform a blank measurement, add 2 μL of PBS to the Peltier plate’s measurement pedestal. The blank sample was PBS, which served as a buffer solution for IgY extraction and purification.
After the blank measurement, clean the pedestal with a lint-free lab wipe and pipette 2 μL of the IgY sample onto the pedestal. The spectrophotometer arm was thenclosed, and all samples were evaluated, with subsequent cleaning, pipetting, and measurement between each sample analysis.
The purity of IgY was initially assessed by NanoDrop spectrophotometry using the A260/A280 absorbance ratio and UV spectrum profile, and further confirmed by SDS-PAGE.
2.12. IgY Confirmation by SDS-PAGE
Reduced SDS-PAGE gel electrophoresis was carried out to verify the purity and molecular weight of the purified chicken IgY antibodies, utilizing Invitrogen NuPAGENovexBis-Tris Mini Gels, the BenchMark™ Protein Ladder, and the XCellSureLock Mini Cell. The SDS-PAGE protocol is outlined in the NuPAGENovexBis-Tris Mini Gels product kit. The reduced IgY sample (10 μg) and 5 μL of MagicMark™ XP Western Protein Standard were loaded onto the NuPAGE mini-gel, and thenIgY migration was performed at 200 V in the separating gel for approximately 2 hours. The gel was stained with Simplyblue™ safestain (coomassie-based stain) to visualize protein bands and destained with a destaining solution to reveal the bands.
2.13. Data Management and Analysis
The data were organized, cleaned, and sorted using Microsoft Excel spreadsheets and analyzed with R-4.4.1. Linear regression was employed to observe trends in IgYconcentrations over six weeks, while repeated measures ANOVA was used to identify significant differences in mean IgY concentrations across ten experimental chickens over time. A linear mixed-effects model was applied to assess the log dilution trend during the virus neutralization test (VNT). Before conducting the repeated measures ANOVA, the normality of the data was verified to ensure valid comparisons, followed by a post hoc test to determine the specific weeks with significant differences. The assumptions for repeated measures ANOVA were tested and met, including sphericity (Mauchly’s Test: p = 0.121), normality (Shapiro-Wilk:p= 0.299 to 0.884), and Bartlett’s Test of Sphericity (p = 0.463). Homogeneity of variance and independence of observations were assumed to be met based on the study design.
2.14. Ethical Considerations
The use of experimental animals in this research was approved by the College of Veterinary Medicine and Animal Science UoG Research Ethics Review Committee (CVMASC-RERC) with reference: CVMASC/UoG/RERC/26/04/2023.
3. Results
3.1. Virus Titration
The initial titer of the LSDV vaccinal strain, determined using Vero cell culture, was 10⁻⁵ TCID₅₀/mL. To enhance the immunogenicity of the vaccinal strain and elicit a stronger immune response, the viral concentration was increased 100-fold, resulting in a final immunogenic titer of 10⁻³ TCID₅₀/mL.
3.2. Quantification of IgY Concentration
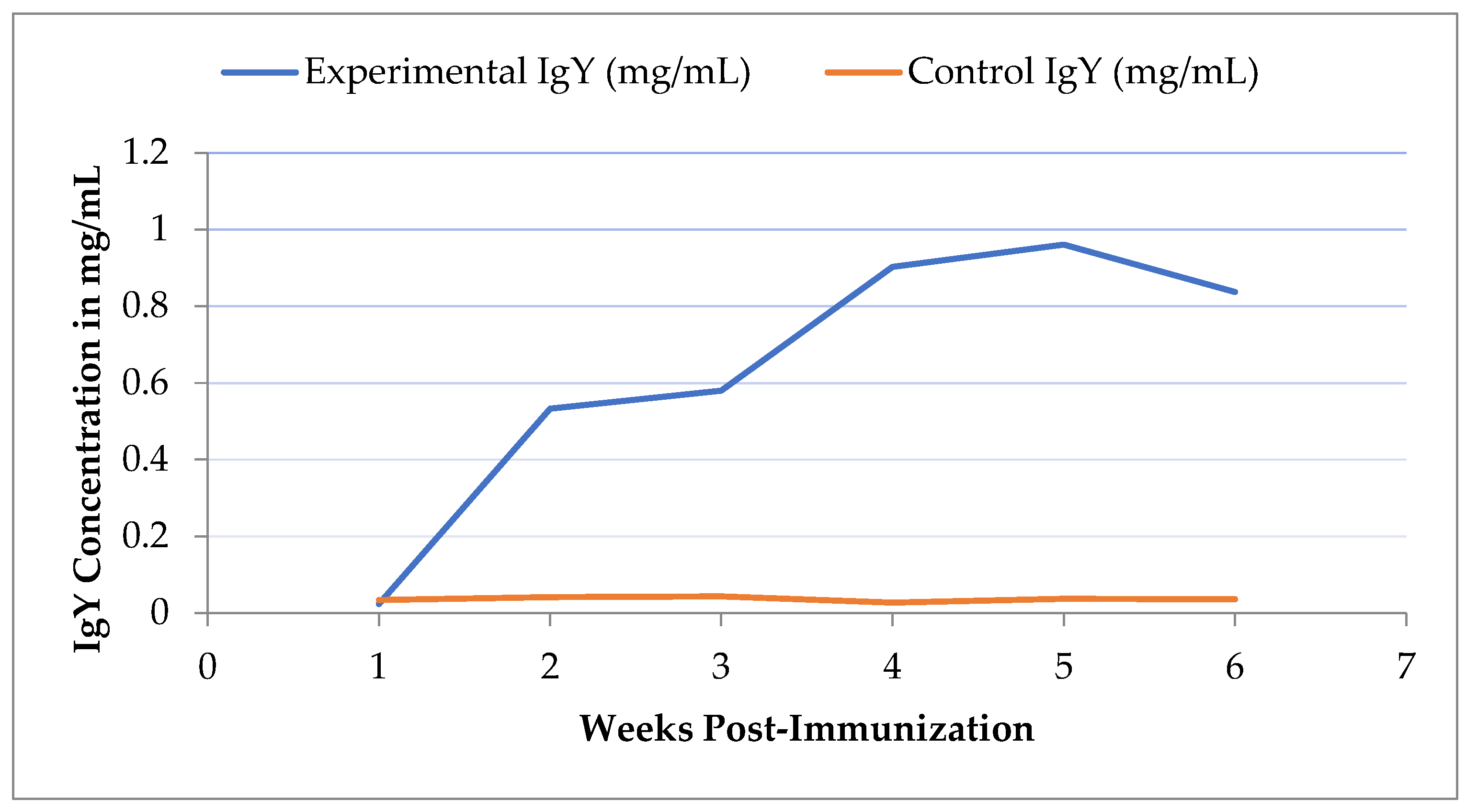
The NeoDot Microvolume spectrophotometer measured the concentration of egg yolk antibodies extracted weekly from individual chickens. The mean IgY concentrations (mg/mL) in the experimental group increased from 0.023 at Week 1 to 0.837 at Week 6, peaking at 0.961 mg/mL in Week 5 (
Table 2). In contrast, the control group exhibited consistently low IgY levels throughout the study period (
Table 3). The IgY production dynamics over the six weeks showed a statistically significant relationship between time (week) and IgY concentration (p < 0.024). The dynamics of IgY production in the experimental group showed three main trends: surging in the first two weeks and from the third to fourth week, reaching a steady state from the second to the third week, and declining after the fifth week (
Figure 1).
The mean IgY concentration from both the lumpy skin disease virus vaccinal strain-exposed (experimental) and non-exposed (placebo, PBS-treated) groups was analyzed using linear regression. The non-exposed group exhibited a slope of 0.001 (p = 0.301), indicating no significant change in IgY concentration, as the change was not significantly different from zero. In contrast, the experimental group showed a slope of 0.81 (p = 0.024), reflecting a significant increase in IgY concentration over time. These findings suggest that exposure to the lumpy skin disease virus vaccinal strain elicited a robust immune response in the experimental group, while the placebo group showed no measurable response.
The analysis result of repeated measures ANOVA indicated a significant effect of week on the mean concentration of IgY (p < 0.0001). The post hoc test confirmed significant differences in IgY concentration between each week.
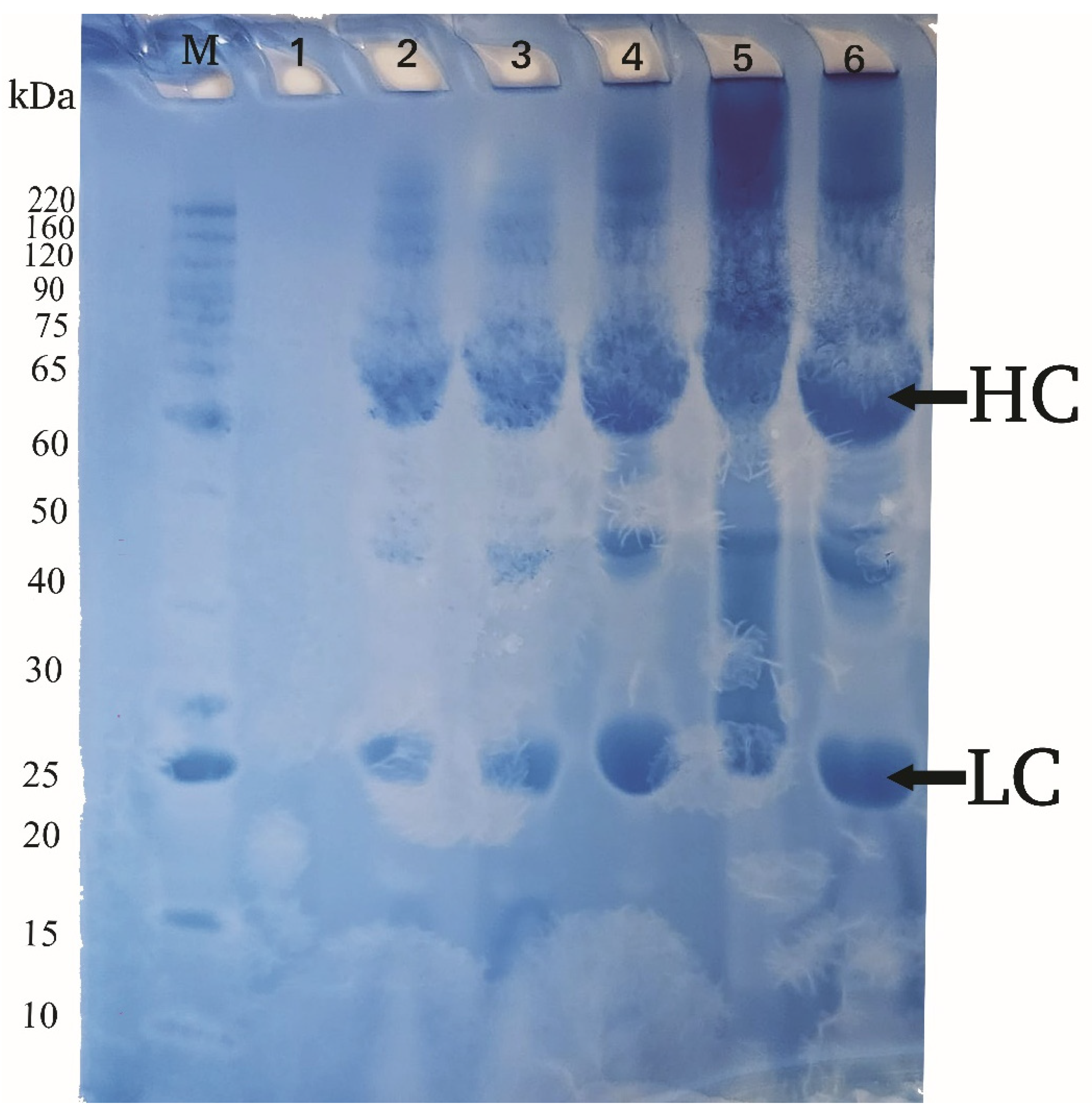
3.3. Confirmation of IgY Identity
The results from the SDS-PAGE analysis revealed two distinct protein bands corresponding to the heavy and light chains of chicken IgY. The heavy chain (HC) had an approximate molecular weight of 65 kDa, while the light chain (LC) was around 25 kDa. These values align with the expected total molecular weight of native IgY, approximately 180 kDa, confirming that the purified protein from the chicken egg yolk was IgY (
Figure 2).
Key: Lane M: Bench Mark™ Protein Ladder; Lane 1: IgY from egg yolk (Week 1); Lane 2: IgY from egg yolk (Week 2); Lane 3: IgY from egg yolk (Week 3); Lane 4: IgY from egg yolk (Week 4); Lane 5: IgY from egg yolk (Week 5); Lane 6: IgY from egg yolk (Week 6); HC: Heavy chain; LC: Light chain; kDa: Kilodalton.
3.4. Evaluating the Virus Neutralization Efficacy of IgY
In the Virus Neutralization Test (VNT) for Lumpy Skin Disease Virus (LSDV), IgY demonstrated effective neutralization at a dilution of 1:125 in two fold serial dilution. Virus neutralization tests (VNT) showed that egg yolk IgY began neutralizing activity in the second week, peaking in the fourth week, while serum IgY exhibited a delayed onset in the third week and declined earlier (
Figure 3. Egg yolk IgY consistently demonstrated stronger virus neutralization capacity than serum IgY.
The findings confirmed that egg yolk is a more effective and concentrated source of polyclonal IgY than serum. The VNT results, visualized with bar graphs, further demonstrated that egg yolk-derived IgY possesses higher affinity and specificity in virus neutralization compared to serum IgY.
4. Discussion
Immunoglobulin Y technology is a scalable production technology that started its foundation in 1980 [
36] and was rapidly adopted in developing countries and regions such as South America and India, as well as Japan, Germany, and other developed countries [
37]. This technology is highly relevant in immunodiagnostics, playing a crucial role in the development of immunoassays such as ELISA due to its specificity and sensitivity [
38]. Its relevance extends to disease control and serosurveillance, where IgY is potentially used for the detection, prevention, and treatment of human and animal infections [
39].
This study implemented the IgY extraction method using PEG as a precipitation agent according to modified[
40], Madera-Contreras, et al. (2022),and the purification method using the dialysis membrane based on the protocols of [
17]. It performed three experimental scenarios, including confirming IgY identity with SDS-PAGE, evaluating IgY’s neutralization capacity with VNT, and quantifying IgY concentration with the Nano Drop Spectrophotometer. The overall test result indicated that it is possible to generate IgY antibodies from chicken egg yolk by triggering the chicken’s immune system with immunization of LSDV viral antigen.
The original titer of the LSDV vaccinal strain for bovines, as determined by the National Veterinary Institute (NVI), was 3 TCID50/ml. After undergoing a second passage and subsequent titration, the titer was reduced to 0.001 TCID50/ml.
The SDS-PAGE profiling of IgY demonstrated that two immunoglobulin chains appeared on the gel with the heavy chain (HC) with 65 kDa, and the light chain (LC) with 25 kDa with increasing band intensity observed from the second week onward. Similar trends were reported by [
41] who studied the isolation of IgY antibodies from immunized chickens and provided a comparison between two chicken breeds. The result of this study is approximately aligned with the measurement of IgY molecular weight after the production of egg yolk antibodies specific to Nosemaceranae by [
42] which was 68 kDa and 27 kDa HC and the LC respectively. It has also a similar result to another recent finding of SDS PAGE result by [
43] from chicken IgY produced against H5 and H9 avian influenza viruses that scored 65 kDa and 26 kDa HC and light chain respectively. The SDS PAGE analysis of the IgY produced across 6 weeks indicated that egg yolk antibody is detectable as early as two weeks after inoculation of the priming dose. Preceding the initial detection from six-week IgY production profiling, the IgY band intensity has been increasing. This finding was comparable with the recent investigation conducted by [
44] on the production and evaluation of IgY against humanand simian rotaviruses emphasizing the increment of the antibody concentration over time. The invisible band of SDS PAGE results in the first week after inoculation of the virus is due to the MatAbtransferred to the egg in chickens being initially undetectable because the immunological timeline of chicken IgY antibody response is not evident on the 7th day [
45]. Additionally, the enhanced band intensity after the boosting immunization verifies that secondary antigen inoculating has significantly increased pAb levels in egg yolk by evoking the strongest and most rapid immune response, as confirmed by [
46]. The result of the study approved that both molecular and band intensities, as well as the sequential surge of IgY concentration, are aligned with current findings.
In the current study on the quantification of IgY, the mean concentration of chicken egg yolk antibodies varied over the study period, exhibiting an increasing trend. This suggested that boosting immunization over time can significantly increase the yield of IgY pAbs. The linear regression analysis has confirmed a significant increase inIgY concentration over six weeks, with distinct phases of boosting, steady state, and decline during the whole course of the study reflecting the nature of immune system workflow. The dynamics of IgY concentration change in this study is in agreement with reports made by [
47] on the study of monitoring the total amount of chicken IgY, against plant toxin ricin, and the Clostridium botulinum neurotoxins. The remarkable differences in the concentration of each week’sIgY product with wavingcyclic growth with periodic surges and a terminal decline pattern of IgY is coherentwith [
48] findings, which examined time-dependent variations in antibodytiter.
The VNT of IgY against LSDV results indicated that chicken egg yolk antibodies have a more robust neutralization capacity than serum IgY. This finding is in agreement with previous work by [
49] that compared egg yolk and serum antibody titers to four avian viruses.The analysis of the logarithm of dilution levels of the IgY to assess the neutralization potency of the antibody, using the linear mixed-effects model affirmed that IgY from yolk is much more potent than serum IgY significantly.The study noticed that the log dilution of egg yolk IgYwas more negative than values scored by dilution of the serumIgYindicating stronger virus-neutralizing capacity of the yolkIgY. This observation was parallel with the finding of [
13]who clearly stated that the IgY from the serum of chicken is not as potent as egg yolk IgY in neutralizing viral infections. The finding of this research is also aligned with the investigations of [
44]in their study about the development and assessment ofegg yolk IgY against humans and simian rotaviruses. This research’s finding is also consistent with the conclusion drawn by [
48] which stated that theIgY concentration in egg yolk is about 10 to 100 times higher than in the corresponding chicken serum that promoted the egg yolk effective source ofIgY antibodies for neutralizing the specific antigen.
The late beginning and early decline as well as lower peak percentage of neutralization in serum IgY, than egg yolkderived IgY of this research finding reflected the efficacy of egg yolk as a source of robust antibodies. These results support the observations made by. [
50]which highlighted the advantages of egg yolkIgY over serum antibodies while using immunotherapy for the prevention and treatment of terrestrial and aquatic animal diseases.
The results of this research were encouraging, however,various constraints inhibited to realizationof the desired objective. Initially, IgYpAb is extracted and purified for use as a primary antibody in the ELISA kit development procedure. However, it was not possible to harvest anti-chicken rabbit IgG for the secondary antibody due to the absence of a suitable laboratory animal. Additionally, there was no available enzyme (such as horseradish peroxidase or alkaline phosphatase) for conjugation with the secondary antibody and chromogenic substrate. The need for a highly advanced pAb purification laboratory with low-pressure chromatography was not met, leading to the use of a dialysis bag for this purpose. Finally, no commercially available enzyme-linked immunosorbent assay kit was found for comparison and serological profiling of the viral antigen, which hindered further validation of IgY by comparison with VNT results.
5. Conclusions and Recommendations
The findings of this study demonstrated that optimal IgY antibodies were successfully extracted from egg yolks starting from the second week post-immunization with the target antigen. This highlighted IgY as a viable alternative to mammalian antibodies for immunoassays and other immunoglobulin-based applications. A continuous supply of IgY was ensured with booster doses administered every 2 to 3 weeks. The study revealed that time (week) significantly affected IgY concentration, with levels increasing over the study period. Notably, IgY concentrations from immunized chickens significantly differed from those of non-immunized chickens, with the latter showing absent neutralization of the virus. IgY from egg yolk proved to be more potent and productive than serum-derived IgY, exhibiting significantly higher efficacy and antibody neutralization capacity.
In conclusion, egg yolk–derived IgY represents an efficient and practical alternative to mammalian antibodies for immunoassays and related applications. Booster immunizations administered at 2–3-week intervals are recommended to sustain high-potency IgY production, with optimal antibody levels detectable from the second week post-immunization. Egg yolk–derived IgY demonstrated superior potency and neutralization capacity compared with serum-derived IgY, supporting its preferential use. Further studies are warranted to evaluate the in vivo neutralizing efficacy of IgY for potential therapeutic and prophylactic applications.
Author Contributions
Fentahun Wondmnew and Demessa Negessu contributed equally to this work, including study design and manuscript drafting. They supervised the experimental animals and performed laboratory tests. Additionally, Fentahun Wondmnew analyzed the data, wrote, and prepared the manuscript. Saddam Mohammed and Anmaw Shite provided critical insights and valuable guidance throughout the study. All authors have reviewed and approved the final manuscript. They collectively accept responsibility for the integrity of the work and agree to address any questions related to its accuracy or reliability.
Funding
This research did not receive specific funding from any funding agency in the public, commercial, or not-for-profit sectors. However, the Animal Health Institute, Ethiopia, supported this study by purchasing experimental animals and providing access to the viral isolation laboratories. AU-PANVAC facilitated the molecular identification of IgY, and the Ministry of Labour and Skills provided financial assistance for personal expenses during the study.
Institutional Review Board Statement
The use of experimental animals in this research was approved by the College of Veterinary Medicine and Animal Science UoG Research Ethics Review Committee (CVMASC-RERC) with reference: CVMASC/UoG/RERC/26/04/2023.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Acknowledgments
The authors would like to thank the Animal Health Institute, Ethiopia, for their logistical support in procuring experimental animals and granting full access to their cell culture laboratory. We are also grateful to AU-PANVAC for their invaluable assistance in conducting the molecular identification of IgY. Special thanks to the Ministry of Labour and Skills for providing financial support in the form of pocket money during the research period. Additionally, we acknowledge the use of a Large Language Model (LLM), for assisting in refining the grammar and improving the clarity of the text during the preparation of the manuscript.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
ANOVA Analysis of Variance
CapV Capripoxvirus
CPE Cytopathic Effect
ELISA Enzyme-linked Immunosorbent Assay
Ig Immunoglobulin
LSD(V) Lumpy Skin Disease Virus
mAb Monoclonal Antibody
pAb Polyclonal Antibodies
PBS Phosphate-Buffer Saline
PEG, MW Molecular Weight of Polyethylene Glycol
SDS-PAGE Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis
TCID50 Tissue Culture Infectious Dose
VNT Virus Neutralization Test
WOAH World Organization for Animal Health
References
- Al-Salihi, KA. Lumpy Skin disease. Mirror Res Vet Sci Anim. 2014, 3(3), 6–23. [Google Scholar]
- WOAH. Lumpy Skin Disease. In Terrestrial Manual; 2023; pp. 128–33. [Google Scholar]
- Kiplagat, SK; Kitala, PM; Onono, JO; Beard, PM; Lyons, NA. Risk Factors for Outbreaks of Lumpy Skin Disease and the Economic Impact in Cattle Farms of Nakuru County, Kenya. Front Vet Sci 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Mebratu, GY; Kassa, B; Fikre, Y; Berhanu, B. Observation on the outbreak of lumpy skin disease in Ethiopia. Rev d”elevage Med Vet des pays Trop. 1984, 37(4), 395–9. [Google Scholar]
- Gari, G; Waret-Szkuta, A; Grosbois, V; Jacquiet, P; Roger, F. Risk factors associated with observed clinical lumpy skin disease in Ethiopia. Epidemiol Infect. 2010, 138(11), 1657–66. [Google Scholar] [CrossRef]
- Molla, B; Worku, Y; Shewaye, A; Mamo, A. Journal of Veterinary Medicine and Animal Health Prevalence of strongyle infection and associated risk factors in equine in Menz Keya Gerbil District, North-Eastern Ethiopia. 2015, 7(4), 117–21. Available online: http://www.academicjournals.org/JVMAH.
- Dubie, T; Hussen Abegaz, F; Dereje, B; Negash, W; Hamid, M. Seroprevalence and Associated Risk Factors of Lumpy Skin Disease of Cattle in Selected Districts of Afar Region, Ethiopia. Vet Med Res Reports 2022, Volume 13(August), 191–9. [Google Scholar] [CrossRef]
- Haegeman, A; De Vleeschauwer, A; De Leeuw, I; Vidanović, D; Šekler, M; Petrović, T; et al. Overview of diagnostic tools for Capripox virus infections. Prev Vet Med [Internet] Available from. 2020, 181(January), 104704. [Google Scholar] [CrossRef]
- Akther, M; Akter, SH; Sarker, S; Aleri, JW; Annandale, H; Abraham, S; et al. Global Burden of Lumpy Skin Disease, Outbreaks, and Future Challenges. MDPI 2023, 15, 1–29. [Google Scholar] [CrossRef]
- WOAH; World Organization organization of Animal Health. Lumpy Skin Disease. Technical Disease Card. 2022, pp. 1–5. Available online: https://www.woah.org/en/document/lumpy-skin-disease-technical-disease-card/.
- Ascoli, CA; Aggeler, B. Overlooked benefits of using polyclonal antibodies. Biotechniques [Internet] Available from. 2018, 65(3), 127–36. [Google Scholar] [CrossRef]
- Leenaars, M; Hendriksen, CFM. General Introduction: Protocols for. Ilar J [Internet] 2005, 46(3), 269–79. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15953834. [CrossRef]
- Kovacs-nolan, J; Mine, Y. Egg Yolk Antibodies for Passive Immunity; 2012; pp. 163–84. [Google Scholar]
- Müller, S; Schubert, A; Zajac, J; Dyck, T; Oelkrug, C. IgY antibodies in human nutrition for disease prevention. Nutr J [Internet] Available from. 2015, 14(1), 1–7. [Google Scholar] [CrossRef] [PubMed]
- Michael, A.; Meenatchisundaram, S.; Parameswari, G.; Subbraj, T.; Selvakumaran, R.; Ramalingam, S. Chicken egg yolk antibodies (IgY) as an alternative to mammalian antibodies. Indian J Sci Technol 2010, Vol. 3(4), 468–74. [Google Scholar] [CrossRef]
- Pereira, EPV; van Tilburg, MF; Florean, EOPT; Guedes, MIF. Egg yolk antibodies (IgY)and their applications in human and veterinary health: A review. Int Immunopharmacol [Internet] Available from. 2019, 73(May), 293–303. [Google Scholar] [CrossRef] [PubMed]
- Pauly, D; Chacana, PA; Calzado, EG; Brembs, B; Schade, R. Igy technology: Extraction of chicken antibodies from egg yolk by polyethylene glycol (PEG) precipitation. J Vis Exp. 2011, 51(51), 1–6. [Google Scholar]
- Narat, M. Production of Antibodies in Chickens. Food Technol Biotechnol. 2003, 41(3), 259–67. [Google Scholar]
- Wen, J; Zhao, S; He, D; Yang, Y; Li, Y; Zhu, S. Preparation and characterization of egg yolk immunoglobulin Y specific to influenza B virus. Antiviral Res. 2012, 93(1), 154–9. [Google Scholar] [CrossRef]
- Hau, J; Hendriksen, CFM. Refinement of polyclonal antibody production by combining oral immunization of chickens with harvest of antibodies from the egg yolk. ILAR J. 2005, 46(3), 294–9. [Google Scholar] [CrossRef]
- Schade, R; Staak, C; Hendriksen, C; Erhard, M; Hugl, H; Koch, G; et al. The production of avian (egg yolk) antibodies: IgY. The report and recommendations of ECVAM workshop 21; ATLA Alternatives to Laboratory Animals, 1996; pp. 925–34. [Google Scholar]
- Florida, SU. Florida State University Polyclonal Antibody Production Protocol - Rabbits; 2007. [Google Scholar]
- He, J; Hu, J; Thirumalai, D; Schade, R; Du, E; Zhang, X. Development of indirect competitive ELISA using egg yolk-derived immunoglobulin (IgY) for the detection of Gentamicin residues. J Environ Sci Heal - Part B Pestic Food Contam Agric Wastes 2016, 51(1), 8–13. [Google Scholar] [CrossRef]
- Bayat, M; Khabiri, A; Hemati, B. Development of IgY-Based Sandwich ELISA as a Robust Tool for Rapid Detection and Discrimination of Toxigenic Vibrio cholerae. Can J Infect Dis Med Microbiol 2018. [Google Scholar] [CrossRef]
- A’yun, RQ; Hakim, MD; Giri-Rachman, EA; Tan, MI; Niloperbowo, W. Anti-HBsAg IgY polyclonal antibodies potential as capture antibody for HBsAg Detection Kit development. Curr Res Biosci Biotechnol. 2024, 5(2), 1–4. [Google Scholar] [CrossRef]
- Karachaliou, CE; Vassilakopoulou, V; Livaniou, E. IgY technology: Methods for developing and evaluating avian immunoglobulins for the in vitro detection of biomolecules. World J Methodol. 2021, 11(5), 243–62. [Google Scholar] [CrossRef] [PubMed]
- Geletu, US; Musa, AA; Usmael, MA; Keno, MS. Molecular Detection and Isolation of Lumpy Skin Disease Virus during an Outbreak in West Hararghe Zone, Eastern Ethiopia. Vet Med Int. 2024, 2024(1), 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sthitmatee, N; Tankaew, P; Modethed, W; Rittipornlertrak, A; Muenthaisong, A; Apinda, N; et al. Development of in-house ELISA for detection of antibodies against lumpy skin disease virus in cattle and assessment of its performance using a bayesian approach. Heliyon [Internet] Available from. 2023, 9(2), e13499. [Google Scholar] [CrossRef] [PubMed]
- NCC. National Chicken Council Animal Welfare Guidelines. 2020, 1–19. [Google Scholar]
- Sandell, L; Sakai, D. Mammalian cell culture; Current Protocols in Essential Laboratory Techniques, 2011; Vol. 2011, pp. 1–32 p. [Google Scholar]
- Kong, BW; Foster, LK; Foster, DN. A method for the rapid isolation of virus from cultured cells. Biotechniques 2008, 44(1), 97–9. [Google Scholar] [CrossRef]
- Lei, C; Yang, J; Hu, J; Sun, X. On the Calculation of TCID50 for Quantitation of Virus Infectivity. Virol Sin. 2021, 36(1), 141–4. [Google Scholar] [CrossRef]
- Hamal, KR; Burgess, SC; Pevzner, IY; Erf, GF. Maternal antibody transfer from dams to their egg yolks, egg whites, and chicks in meat lines of chickens. Poult Sci. 2006, 85(8), 1364–72. [Google Scholar] [CrossRef]
- Morishita, TY. Poultry Blood Collection. In West Univ Heal Sci.; 2019; pp. 1–3. [Google Scholar]
- Madera-Contreras, A.M.; Solano-Texta, R.; Cisneros-Sarabia, A.C.; Bautista-Santos, I.B.; Vences-Velázquez, G.V.; Vences-Velázquez, A.V.; Cortés-Sarabia, KC. Optimized method for the extraction of contaminant-free IgY antibodies from egg yolk using PEG 6000. MethodsX [Internet] Available from. 2022, 9(October), 101874. [Google Scholar] [CrossRef]
- Polson, A; Barbara von Wechmar, M. Isolation of viral IgY antibdies from yolk of inmunized hens. Inmunol Comun. 1980, 9(5), 475–93. [Google Scholar]
- Zhang, X; ying; Vieira-pires, RS; Morgan, PM. IgY-Technology: Production and Application of Egg Yolk Antibodies; Springer: Cham, 2021; pp. 1–70. [Google Scholar]
- Yadoung, S; Ishimatsu, R; Xu, ZL; Sringarm, K; Pata, S; Thongkham, M; et al. Development of IgY-Based Indirect Competitive ELISA for the Detection of Fluoroquinolone Residues in Chicken and Pork Samples. Antibiotics 2022, 11(11). [Google Scholar] [CrossRef]
- Lee, L; Samardzic, K; Wallach, M; Frumkin, LR; Mochly-Rosen, D. Immunoglobulin Y for Potential Diagnostic and Therapeutic Applications in Infectious Diseases. Front Immunol. 2021, 12(June), 1–23. [Google Scholar] [CrossRef] [PubMed]
- Polson, A; von Wechmar, MB; van Regenmortel, MHV. Isolation of viral igy antibodies from yolks of immunized hens. Immunol Invest. 1980, 9(5), 475–93. [Google Scholar] [CrossRef] [PubMed]
- Amro, WA; Al-Qaisi, W; Al-Razem, F. Production and purification of IgY antibodies from chicken egg yolk. J Genet Eng Biotechnol [Internet] Available from. 2018, 16(1), 99–103. [Google Scholar] [CrossRef] [PubMed]
- Açık, MN; Karagülle, B; Yakut, S; Öztürk, Y; Kutlu, MA; Kalın, R; et al. Production, characterization and therapeutic efficacy of egg yolk antibodies specific to Nosema ceranae. PLoS One 2024, 19, 1–19. [Google Scholar] [CrossRef]
- Nahla, M; Abdelfattah, N; Ibrahim, M. Characterization of chicken IgY produced against H5 and H9 avian influenza viruses. Alexandria J Vet Sci. 2018, 59(1), 79. [Google Scholar] [CrossRef]
- Bentes, GA; Lanzarini, NM; Guimarães, JR; Heinemann, MB; Volotão E de, M; da Silva, A dos S; et al. Production and Evaluation of Chicken Egg Yolk Immunoglobulin (IgY) against Human and Simian Rotaviruses. Viruses 2022, 14(9), 1–10. [Google Scholar] [CrossRef]
- Ramakrishnan, S; Kappala, D. Avian infectious bronchitis virus. In Recent Adv Anim Virol; 2019; pp. 301–19. [Google Scholar]
- Agurto-Arteaga, A; Poma-Acevedo, A; Rios-Matos, D; Choque-Guevara, R; Montesinos-Millán, R; Montalván, Á; et al. Preclinical Assessment of IgY Antibodies Against Recombinant SARS-CoV-2 RBD Protein for Prophylaxis and Post-Infection Treatment of COVID-19. Front Immunol. 2022, 13(May), 1–12. [Google Scholar] [CrossRef]
- Pauly, D; Dorner, M; Zhang, X; Hlinak, A; Dorner, B; Schade, R. Monitoring of laying capacity, immunoglobulin Y concentration, and antibody titer development in chickens immunized with ricin and botulinum toxins over a two-year period. Poult Sci [Internet] Available from. 2009, 88(2), 281–90. [Google Scholar] [CrossRef]
- Schade, R; Calzado, EG; Sarmiento, R; Chacana, PA; Porankiewicz-asplund, J; Terzolo, HR. Scade Et Al 2015-Igy Tech. ATLA. 2005, 33, 129–54. [Google Scholar]
- Silim, AA; Venne, D. Comparison of Egg-Yolk and Serum Antibody Titers to Four Avian Viruses by Enzyme- Linked Immunosorbent Assay Using Paired Field Samples; American Association of Avian Pathologists Stable, 1989; Volume 33, 4, pp. 643–8. Available online: https://www.jstor.org/stable/1591138.
- Xu, Y; Li, X; Jin, L; Zhen, Y; Lu, Y; Li, S; et al. Application of chicken egg yolk immunoglobulins in the control of terrestrial and aquatic animal diseases: A review. Biotechnol Adv [Internet] Available from. 2011, 29(6), 860–8. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).