Submitted:
21 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
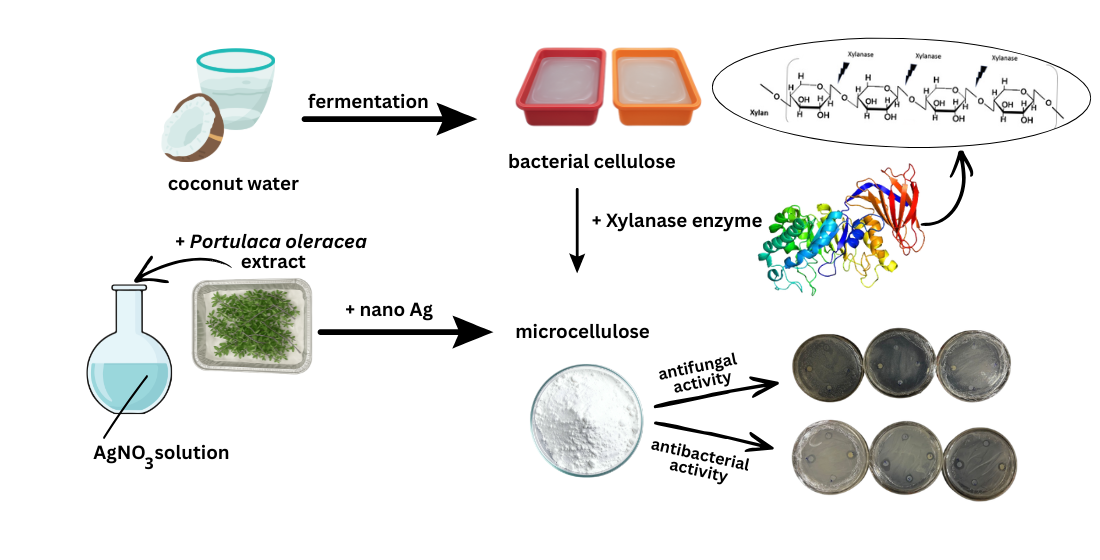
1. Introduction
2. Materials and Methods
2.1. Research Procedure
2.1.1. Cellulose Preparation
2.1.2. Preparation of Cellulose-Glycerol
2.1.3. Chitosan Application on Cellulose
2.1.4. Measurement of Physical Properties of Cellulose
2.1.6. Dried Cellulose Sheets
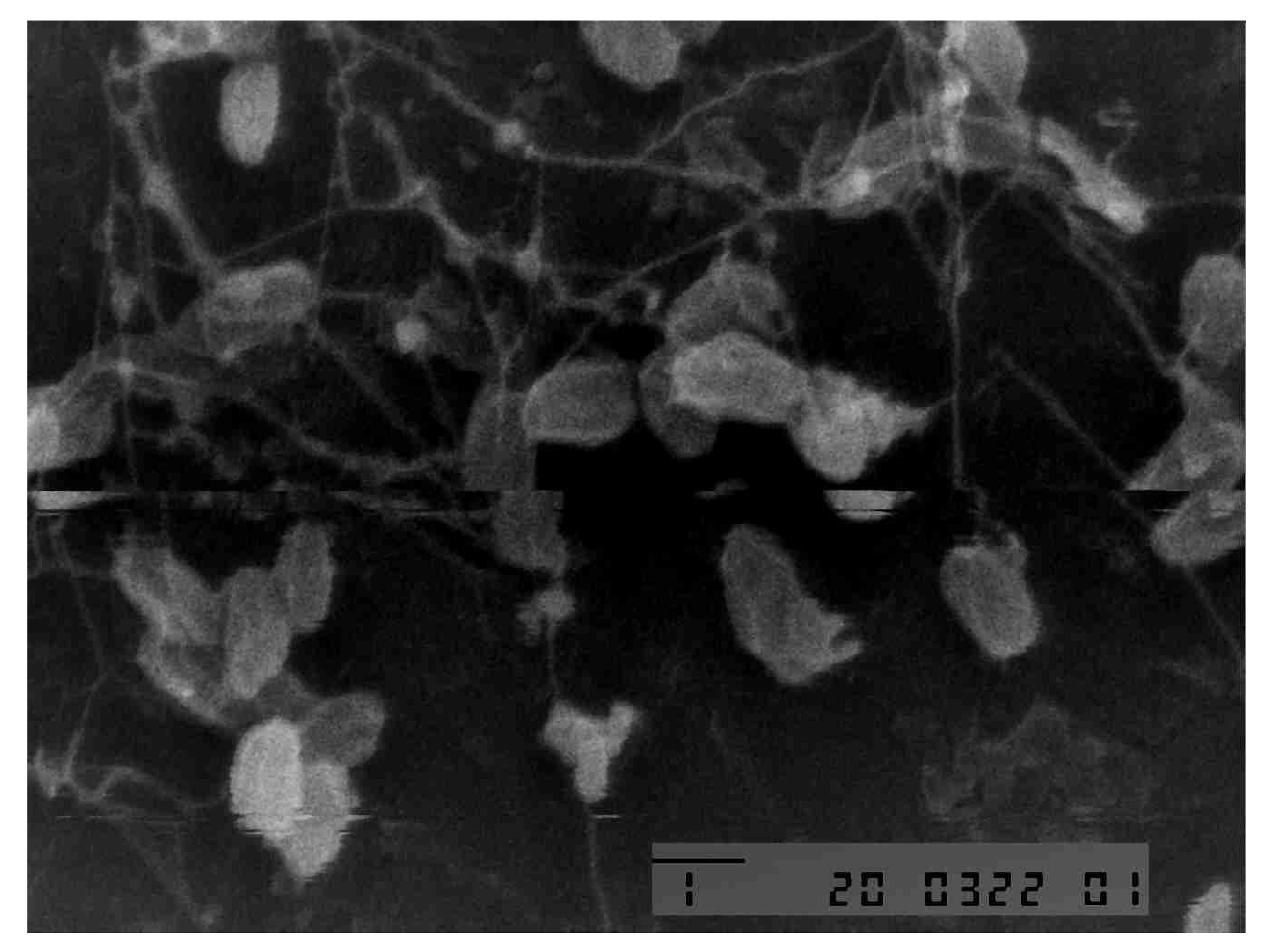
2.1.7. Microcellulose Size Analysis with SEM
2.1.8. Silver Nanoparticle Preparation
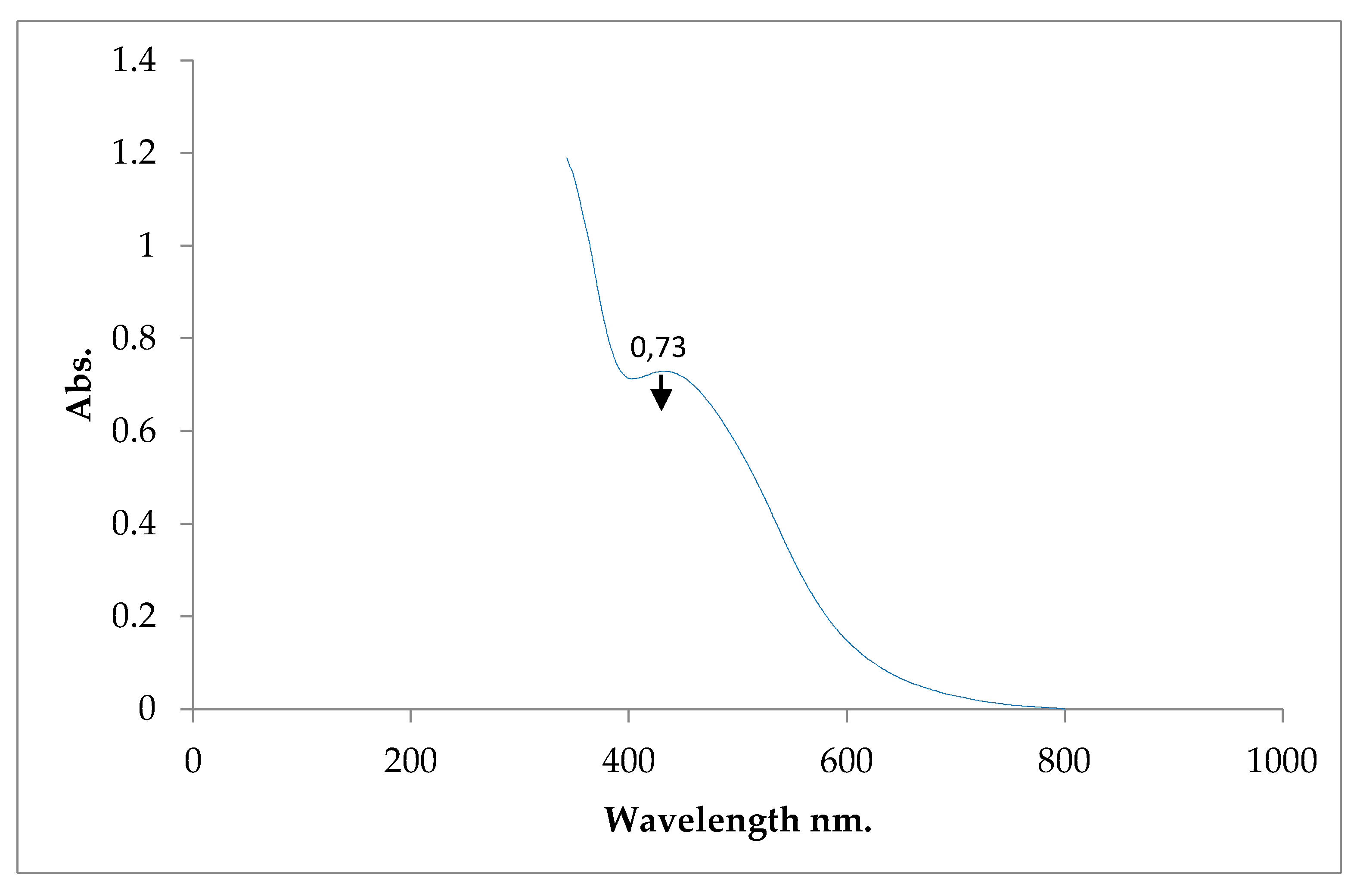
2.1.9. Wavelength Analysis of Silver Nanoparticles with UV Vis Spectrophotometry
2.1.10. Silver Nanoparticle Size Analysis with PSA
2.1.11. Deposition of Microcellulose in Silver Nanoparticles
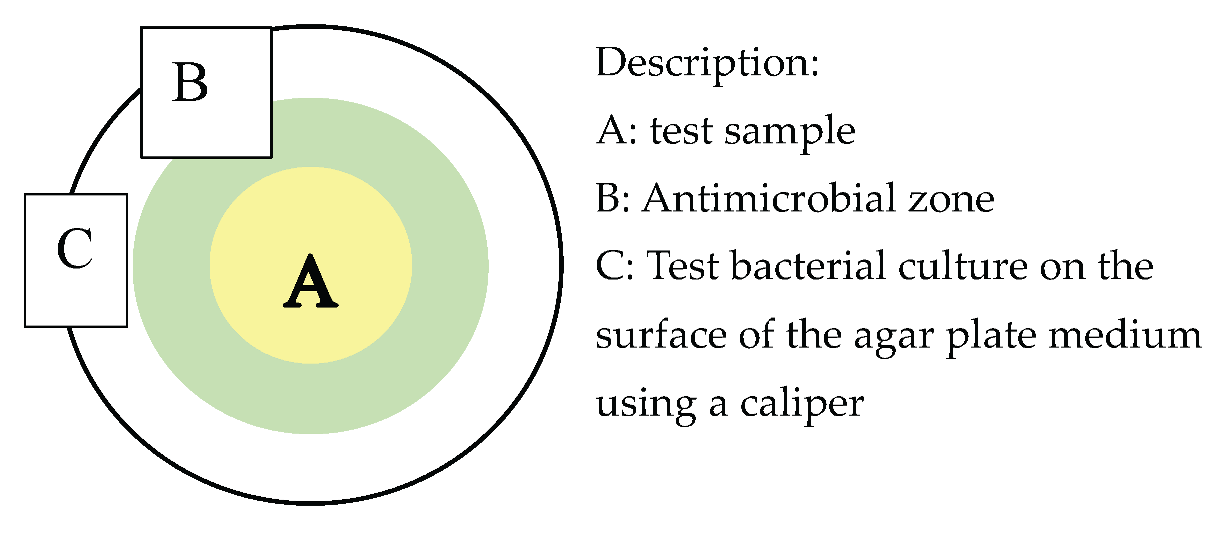
2.1.12. Antimicrobial Activity Test
2.2. Data Analysis Techniques
3. Results and Disscussion
3.1. Physical Properties of Bacterial Cellulose from Coconut Water
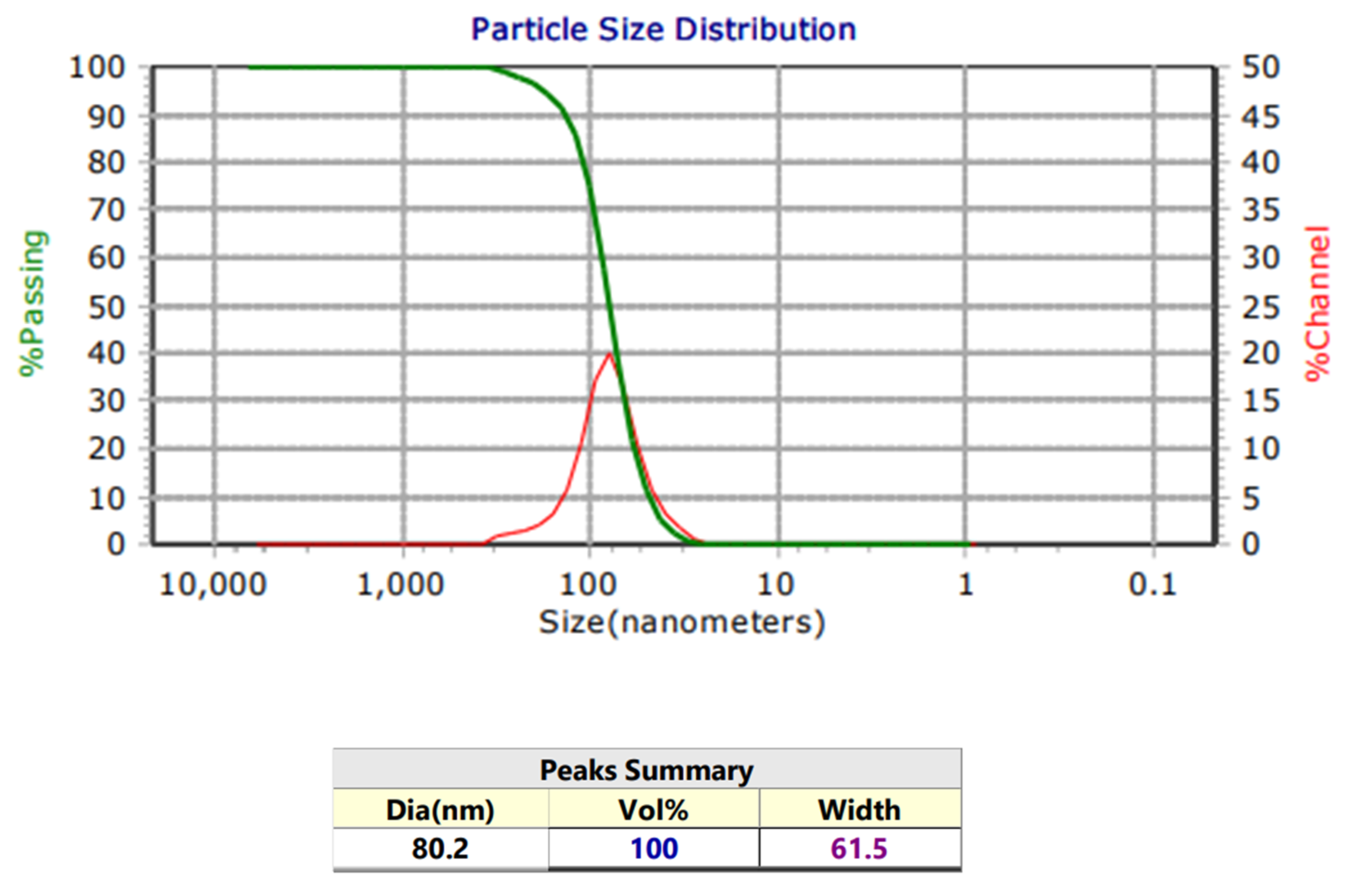
3.2. Microcellulose Character from Coconut Water
3.3. Biosynthesis of Silver Nanoparticles with Purslane Extract
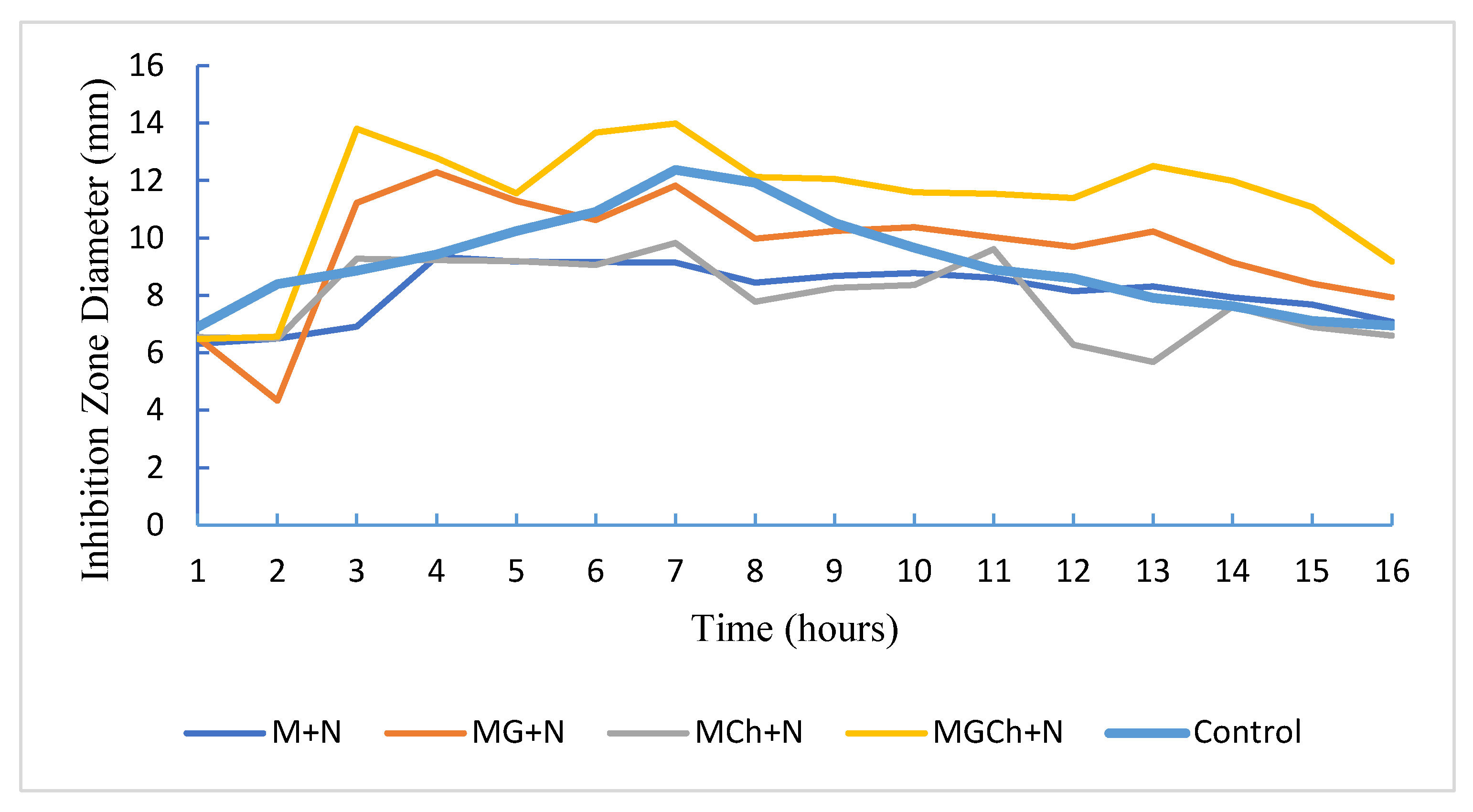
3.4. Antimicrobial Activity of Microcellulose
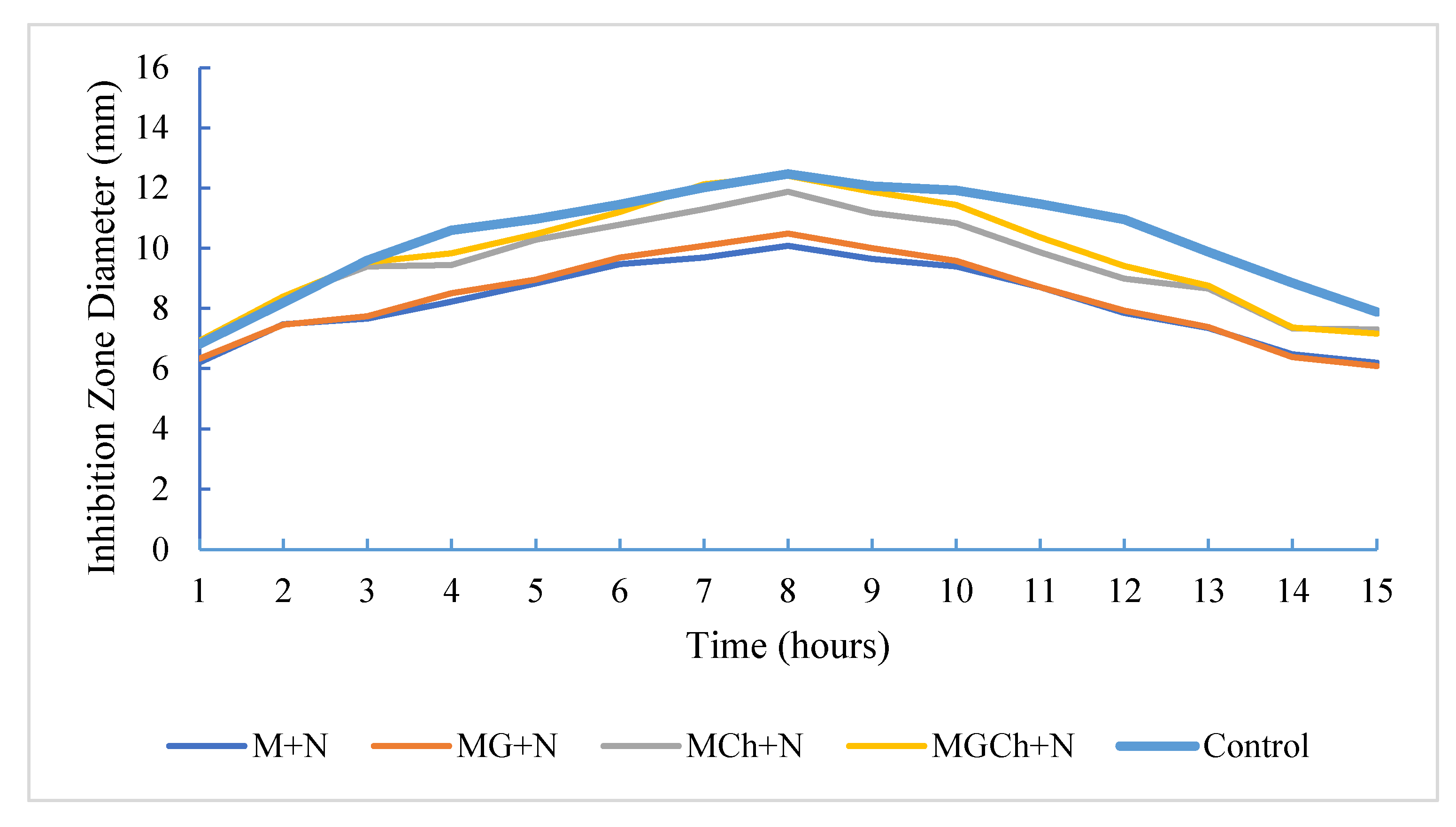
3.5. Antimicrobial Activity of Microcellulose Against S. epidermidis
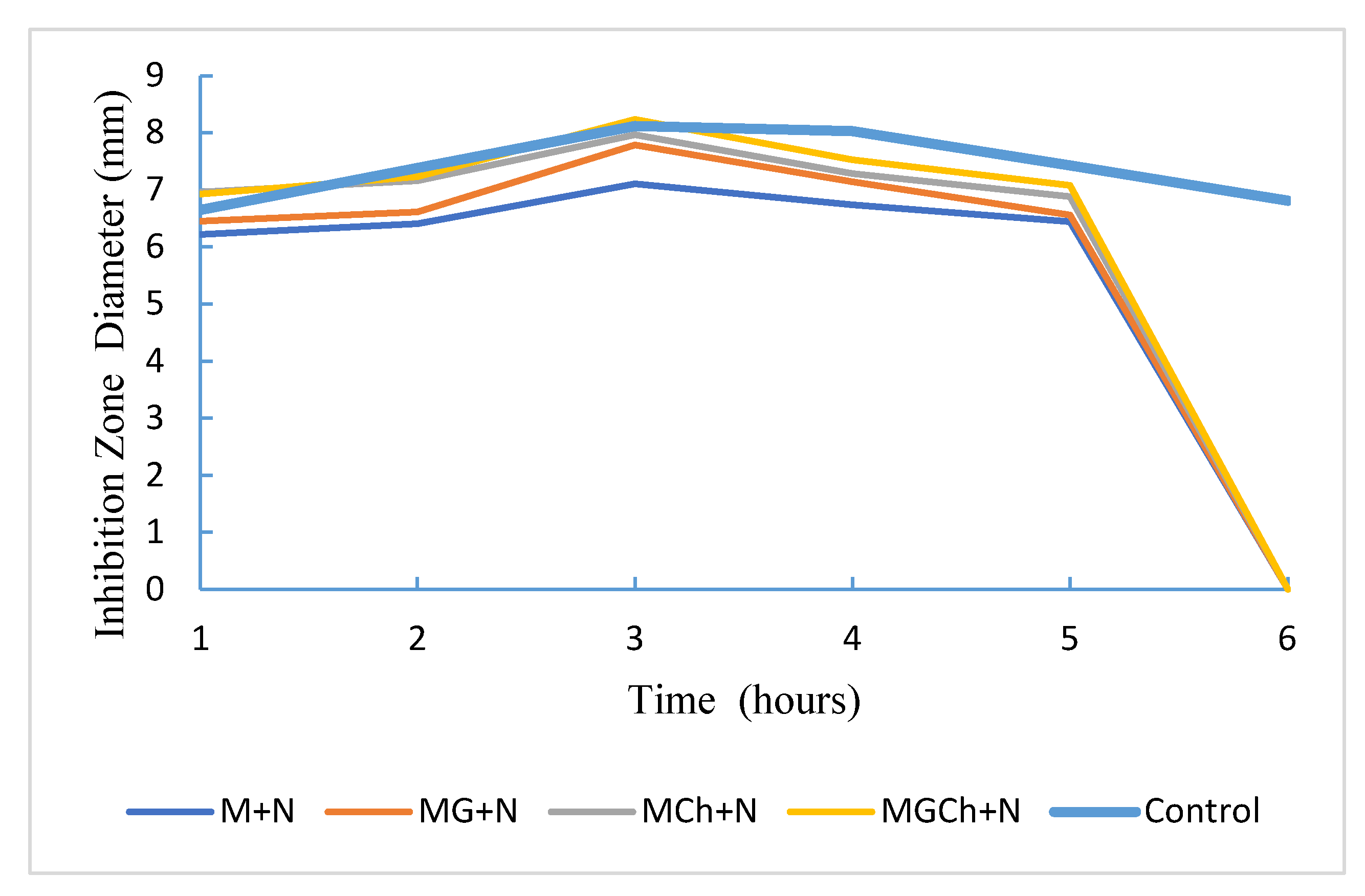
3.6. Antimicrobial Activity of Microcellulose in P. aeruginosa
| Treatment | Diameter of the barrier zone (mm) | Category |
|---|---|---|
| M+N | 10,083a | Strong |
| MG+N | 10.0497b | Strong |
| MCh+N | 11,883c | Strong |
| MGCh+N | 12,417p | Strong |
| Controls | 12.4663d | Strong |
3.7. Antimicrobial Activity of Microcellulose Against C. albicans
| Treatment | Diameter of the inhibition zone (mm) | Category |
|---|---|---|
| M+N | 7.1100a | Medium |
| MG+N | 7.7933a | Medium |
| MCh+N | 7.9733b | Medium |
| MGCh+N | 8.1200b | Medium |
| Controls | 8.2433b | Medium |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shi, S.; Wang, W.; Wang, F.; Yang, P.; Yang, H.; He, X.; Liao, X. Research Progress in Coconut Water: A Review of Nutritional Composition, Biological Activities, and Novel Processing Technologies. Foods 2025, 14, 1503. [Google Scholar] [CrossRef]
- Zhang, Y.; Kan, J.; Liu, X.; Song, F.; Zhu, K.; Li, N.; Zhang, Y. Chemical Components, Nutritional Value, Volatile Organic Compounds and Biological Activities In Vitro of Coconut (Cocos nucifera L.) Water with Different Maturities. Foods 2024, 13, 863. [Google Scholar] [CrossRef]
- Aba, R. P. M.; Luna, M. B. Z.; Villasis, J. C. Characterization of mature coconut (Cocos nucifera L.) water from different varieties. Food Humanit. 2024, 100248. [Google Scholar] [CrossRef]
- Selvaraju, V. P.; Gayathri; Ranpatabendi, T.; Fiore, A. Matured coconut water–nutritional profile and preservation methods. Coconut-Based Beverages Prod. 2025, 55–78. [Google Scholar] [CrossRef]
- Menshawy, M. N.; Abdel-Hamid, A. M.; El-Katatny, M. H. Maximizing cellulase and xylanase production from Novel Bacillus pumilus strain isolated from agricultural waste compost in Egypt and optimizing their activities. Discov. Appl. Sci. 2025, 7. [Google Scholar] [CrossRef]
- Rivas-Zúñiga, A.; Eceiza, A.; Fernández-d’Arlas, B. A mechano-enzymatic method to produce nano/microcellulose with ancestral endoglucanase: A comparative study. Nanoscale Adv. 2025, 7, 5625–5636. [Google Scholar] [CrossRef]
- Chen, M.; Li, Q.; Liu, C.; Meng, E.; Zhang, B. Microbial Degradation of Lignocellulose for Sustainable Biomass Utilization and Future Research Perspectives. Sustainability 2025, 17, 1–22. [Google Scholar] [CrossRef]
- Esfahani, S. N. M.; Rostami, S.; Amini, Z. Antibiotic Susceptibility Pattern of Nosocomial and Community-Acquired Pseudomonas aeruginosa in Isfahan: Prospective Multicenter Study. Kermanshah Univ Med Sci. 2024, 28, 1–7. [Google Scholar]
- Jurševics, K.; Rudenko, L.; Vetra, J.; Berzins, A. Adhesion and Colonization Intensity of Staphylococcus epidermidis, Pseudomonas aeruginosa, and Candida albicans on Smooth, Micro-Textured, and Macro-Textured Silicone Biomaterials. J. Funct. Biomater. 2025, 16, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Scalia, A. C.; Najmi, Z. Targeting Bacterial Biofilms on Medical Implants: Current and Emerging Approaches. antibiotics 2025, 14, 1–33. [Google Scholar] [CrossRef]
- Valentine, M.; Wilson, D.; Gresnigt, M. S.; Hube, B. Vaginal Candida albicans infections: host – pa thogen – microbiome inter actions. FEMS Microbiol. Rev. 2025, 49, 1–16. [Google Scholar] [CrossRef] [PubMed]
- An, C.; Liu, Y.; Zhang, X.; Li, J.; Chen, H. Iron dictates the growth, biofilm formation, and virulence of Pseudomonas aeruginosa in pulmonary infections. Front. Microbiol. 2026, 16, 1–12. [Google Scholar] [CrossRef]
- Hurley, J. Associations Between Candida and Staphylococcus aureus, Pseudomonas aeruginosa, and Acinetobacter Species as Ventilator-Associated Pneumonia Isolates in 84 Cohorts of ICU Patients. microorganisms 2025, 13, 1–18. [Google Scholar] [CrossRef]
- Gonzalez-pastor, R.; Carrera-Pacheco, S. E.; Zuniga-Miranda, J.; Rodriguez-Polit, C.; Mayorga-Ramos, A.; Guaman, L. P.; Barba-Ostria, C. Current Landscape of Methods to Evaluate Antimicrobial Activity of Natural Extracts. Molecules 2023, 28, 1–25. [Google Scholar] [CrossRef]
- Hulankova, R. Methods for Determination of Antimicrobial Activity of Essential Oils In Vitro—A Review. Plants 2024, 13, 1–23. [Google Scholar] [CrossRef]
- Benkova, M.; Soukup, O.; Marek, J. Antimicrobial susceptibility testing: currently used methods and devices and the near future in clinical practice. J. Appl. Microbiol. 2020, 129, 806–-822. [Google Scholar] [CrossRef] [PubMed]
- Kadeřábková, N.; Mahmood, A. J. S.; Mavridou, D. A. I. Antibiotic susceptibility testing using minimum inhibitory concentration ( MIC ) assays. Antimicrob. Resist. 2024, 2, 1–9. [Google Scholar] [CrossRef]
- Krapienis, M. G.; Lourenço, F. R. Agar diffusion microbiological assays for antibiotic potency estimation: Improvements in measurement and uncertainty analysis. J. Microbiol. Methods 2024, 216. [Google Scholar]
- Fei, S.; Fu, M.; Kang, J.; Luo, J.; Wang, Y.; Jia, J.; Liu, S.; Li, C. Enhancing bacterial cellulose production of Komagataeibacter nataicola through fermented coconut water by Saccharomyces cerevisiae: A metabonomics approach. Curr. Reserach Food Sci. 2024, 8, 1–9. [Google Scholar] [CrossRef]
- Phan, H. T.; Hoang, D. Q.; Nguyen, T.; Le, D. H. Nata de coco as an abundant bacterial cellulose resource to prepare aerogels for the removal of organic dyes in water. Bioresour. Technol. Reports 2023, 24. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, H.; Chen, Y.; Wang, J. Combined biosynthetic modulation and network fortification to improve nata de coco production. in Materials Today: Proceedings, 2025; pp. 123–134. [Google Scholar]
- Matos, J. M. S.; Evtuguin, D. V.; de Sousa, A. P. M.; Carvalho, M. G. V. S. Xylanase treatment of eucalypt kraft pulps: effect of carryover. Appl. Microbiol. Biotechnol. 2024, 108, 1–13. [Google Scholar] [CrossRef]
- Morán-Aguilar, M. G.; Calderón-Santoyo, M.; de Souza Oliveira, R. P.; Aguilar-Uscanga, M. G.; Domínguez, J. M. Enzyme mixtures rich in cellulase and xylanase applied to cellulose pulps for modification and hydrolysis. Materials (Basel). 2024, 18, 4968. [Google Scholar] [CrossRef] [PubMed]
- Munir. Production, characterization, and antimicrobial activity of polyhydroxyalkanoates synthesized by Bacillus species against skin pathogens. RSC Adv. 2025, 15, 35182–35200. [Google Scholar] [CrossRef] [PubMed]
- Skowron, K.; Budzyńska, A.; Wiktorczyk-Kapischke, N.; Warzonkoska, W.; Gospodarek-Komkowska, E.; Grudlewska-Buda, K. Assessment of antimicrobial efficacy in selected antibacterial cosmetics *. Pomeranian J Life Sci. 2024, 70, 74–82. [Google Scholar] [CrossRef]
- Akachat, B. Effect of glycerol concentration on the physicochemical properties of pectin films derived from Citrus limon waste. Foods 2025, 14, 1576. [Google Scholar]
- Karnwal, A. Advanced starch-based films for food packaging: Innovations in sustainability and functional properties. Food Chem. X 2025, 29, 1–21. [Google Scholar] [CrossRef]
- Li, J.; Chen, Z.; Zhu, H. Effect of glycerol plasticizer on the physical and mechanical properties of cellulose films extracted from plant fibres. Polymers (Basel). 2023, 15, 2458. [Google Scholar]
- Smith, A. R.; Gupta, B. Influence of glycerol plasticization on the structural, mechanical, and thermal properties of cellulose-based composites. J. Appl. Polym. Sci. 2025, 142, e54567. [Google Scholar]
- Darmenbayeva, A.; Zhussipnazarova, G.; Rajasekharan, R.; Massalimova, B.; Zharlykapova, R.; Nurlybayeva, A.; Mukazhanova, Z.; Aubakirova, G.; Begenova, B.; Manapova, S.; Bulekbayeva, K. Applications and Advantages of Cellulose – Chitosan Biocomposites: Sustainable Alternatives for Reducing Plastic Dependency. Polymers (Basel). 2025, 17, 1–15. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Z.; Wang, S.; Chai, X.; Li, K.; Feng, J. Bioinspired chitosan coatings enhanced with bacterial cellulose nanocrystals and apple polyphenols for preservation of perishable fruits. Food Chem. 2025, 410. [Google Scholar] [CrossRef]
- Carvalho, L. da S. C. de.; Brenes, R. G. R.; Grieco, M. A.; Bojorge, N.; Jr, N. B. Production of cellulose nano/microfibers through simultaneous milling and enzymatic hydrolysis with an optimized cocktail of cellulase/xylanase/LPMO. Ind. Crops Prod. 2024, 220. [Google Scholar]
- Benini, K. C. C. de C.; Yupanqui-Mendoza, S. L.; Arantes, V. Impact of enzymatic hydrolysis and drying on cellulose nanocrystal properties. Carbohydr. Res. 2025, 555. [Google Scholar]
- Rivas-Zuñiga, A.; Eceiza, A.; Fernandez-d’Arlas, B. A mechano-enzymatic method to produce nano / microcellulose with ancestral. Nanoscale Adv. 2025, 7, 5625–5636. [Google Scholar] [CrossRef]
- Guo, W.; Hui, L.; Song, F.; Qu, Y.; Wang, Q.; Zhang, Y.; Xin, J. A new strategy for biological enzyme bleaching: combined effects of laccase, xylanase, and mannanase in the bleaching of softwood kraft pulp – a synergistic effect of enzymes. Nord. Pulp Pap. Res. J. 2025, 40, 465–476. [Google Scholar] [CrossRef]
- De Andrades, D.; Sandrim, V. C.; De Andrades, D.; Sandrim, V. C. Agro-residue Valorization for Thermostable Xylanase Production by Aspergillus caespitosus and its Eco- friendly Application in Pulp Biobleaching Agro-residue Valorization for Thermostable Xylanase Production by Aspergillus caespitosus and its Eco- friendl. BioResources 2026, 21, 1690–1705. [Google Scholar]
- Abdel-rahman, M. A.; Alshallash, K. S.; Eid, A. M.; Hassan, S. E.; Salih, M.; Hamza, M. F.; Fouda, A. Exploring the Antimicrobial, Antioxidant, and Antiviral Potential of Eco-Friendly Synthesized Silver Nanoparticles Using Leaf Aqueous Extract of Portulaca oleracea L. Pharm. Artic. 2024, 17, 1–19. [Google Scholar] [CrossRef]
- Rodriguez-loya, J.; Lerma, M.; Gardea-torresdey, J. L. Dynamic Light Scattering and Its Application to Control Nanoparticle Aggregation in Colloidal Systems: A Review. micromachines Rev. 2024, 15, 1–21. [Google Scholar] [CrossRef]
- Jia, Z.; Li, J.; Gao, L.; Yang, D.; Kanaev, A. Dynamic Light Scattering: A Powerful Tool for In Situ Nanoparticle Sizing. colloids and interfaces 2023, 7, 1–18. [Google Scholar] [CrossRef]
- Iwani, M.; Wibowo, R. H.; Handayani, K. Classification of antibacterial activity based on inhibition zone diameter in agar diffusion assays. Adv. Healthc. Res. 2025, 3, 510–518. [Google Scholar]
- Wardani, N. P.; Primaharinastiti, R.; Poernomo, A. T.; Khatib, A. A comparative study of honey antimicrobial activity against MRSA and Candida albicans based on inhibition zone diameters. J. Farm. dan Ilmu Kefarmasian Indones. 2024, 12, 89–97. [Google Scholar]
- Karlo, J.; Vijay, A.; Phaneeswar, M. S.; Singh, S. P. Sensing the Bactericidal and Bacteriostatic Antimicrobial Mode of Action Using Raman Deuterium Stable Isotope Probing (DSIP) in Escherichia coli. ACS Omega 2024, 9, 23753–23760. [Google Scholar] [CrossRef]
- Liang, Y.; Heining, L.; Elsner, M.; Seidel, M. Flow cytometry for rapid analysis of bacteriostatic versus bactericidal effects in Legionella pneumophila disinfection. Anal. Bioanal. Chem 2025. [Google Scholar] [CrossRef]
- Dube, E.; Okuthe, G. E. Silver Nanoparticle-Based Antimicrobial Coatings: Sustainable Strategies for Microbial Contamination Control. Microbiol. Res. (Pavia). 2025, 16, 1–38. [Google Scholar] [CrossRef]
- Soleimani, S. Exploiting silver ions ’ antimicrobial properties for colorimetric detection of Salmonella via suppressed formation of Au @ Ag nanorods. Sci. Rep. 2025, 15, 1–13. [Google Scholar] [CrossRef]
- Casals, E.; Gusta, M. F.; Bastus, N.; Rello, J.; Puntes, V. Silver Nanoparticles and Antibiotics: A Promising Synergistic Approach to Multidrug-Resistant Infections. microorganisms 2025, 13, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Behera, S. S.; Majumdar, A. G.; Gullani, H.; Roy, A. L.; Dash, A. K.; Mohanty, P.S.; Ray, L. Green synthesis of silver-chitosan nanocomposite exhibits promising antibiofilm properties against pathogenic bacteria Escherichia coli and Staphylococcus aureus. The Micobe 2025, 6, 1–10. [Google Scholar] [CrossRef]
- Pastrana-alta, R. Y.; Huarote-garcia, E.; Egusquiza-Huamani, M. A.; Baena-Moncada, A. M. Antimicrobial activity of chitosan, alginate, pectin, and cellulose-based biopolymer composites with silver, copper oxide, and zinc oxide nanoparticles. RSC Adv. 2025, 15, 35807–35843. [Google Scholar] [CrossRef]
- Frippiat, T.; Art, T.; Delguste, C. Silver Nanoparticles as Antimicrobial Agents in Veterinary Medicine: Current Applications and Future Perspectives. Nanomaterials 2025, 15, 1–28. [Google Scholar] [CrossRef]
- Dybkova, S. Antimicrobial efficiency of ‘green’ silver nanoparticles: Influence of bacterial cell wall structure. Materials (Basel). 2025, 18, 4952. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A. S.; Batista, J. G. S.; Rodrigues, M. A. V.; Thipe, V. C.; Minarini, L. A. R.; Lopes, P. S.; Lugão, A. B. Advances in silver nanoparticles: a comprehensive review on their potential as antimicrobial agents and their mechanisms of action elucidated by proteomics. Front. Microbiol. 2024, 15. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Cao, Q.; Xiao, Q.; Wu, Y.; Ding, L.; Huang, H.; Li, Y.; Yang, J.; Meng, L. The progress and future of the treatment of Candida albicans infections based on nanotechnology. J. Nanobiotechnology 2024, 22, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Chandrakar, N.; Shukla, S. K.; Karley, D.; Upadhyay, N.; Nancharaiah, Y. V. Biogenic Silver Nanoparticles Exhibit Antifungal and Antibiofilm Activity Against Candida albicans via Intracellular ROS Production. J. Pathol. Microbiol. Immunol. 2025, 133, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Almotairy, A. R. Z. Novel synthesis and potential antifungal activity of silver nanospheres and nanotriangles against Candida spp. J. Appl. Microbiol. 2024, 127, 198–209. [Google Scholar]
| Parameter | Types of Bacterial Cellulose | |||
|---|---|---|---|---|
| C | CG | CCh | CGCh | |
| Wet weight | 178.89 g | 200.71 g | 135.86 g | 135.139 g |
| Dry weight | 3.616 g | 4.248 g | 8.05 g | 7.23 g |
| Transparency | Not transparent | Not transparent | Not transparent | Not transparent |
| Color | White | White | Brownish yellow | Brownish yellow |
| Consistency | Less elastic | Elastis | Less elastic | Elastis |
| Construction | Acidic | Acidic | Highly acidic | Highly acidic |
| Treatment | Diameter of the inhibition zone (mm) | Category |
|---|---|---|
| M+N | 9.1533a | Medium |
| MG+N | 11.8200b | Strong |
| MCh+N | 9.8400a | Medium |
| MGCh+N | 13.9967c | Strong |
| Controls | 12.3867bc | Strong |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




