Submitted:
21 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Report
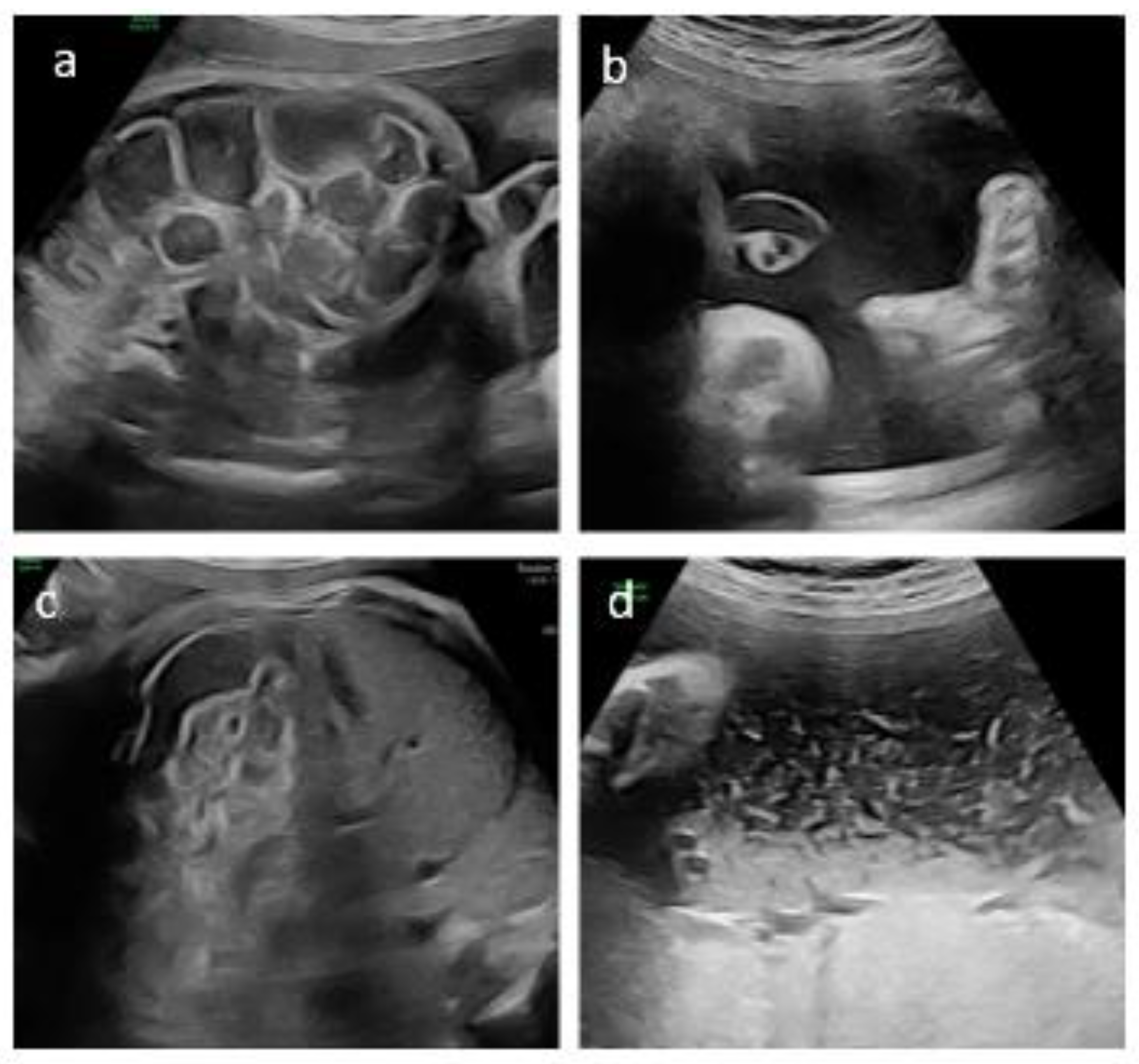
2.1. Prenatal Presentation and Diagnostic Evaluation
2.2. Prenatal Intervention
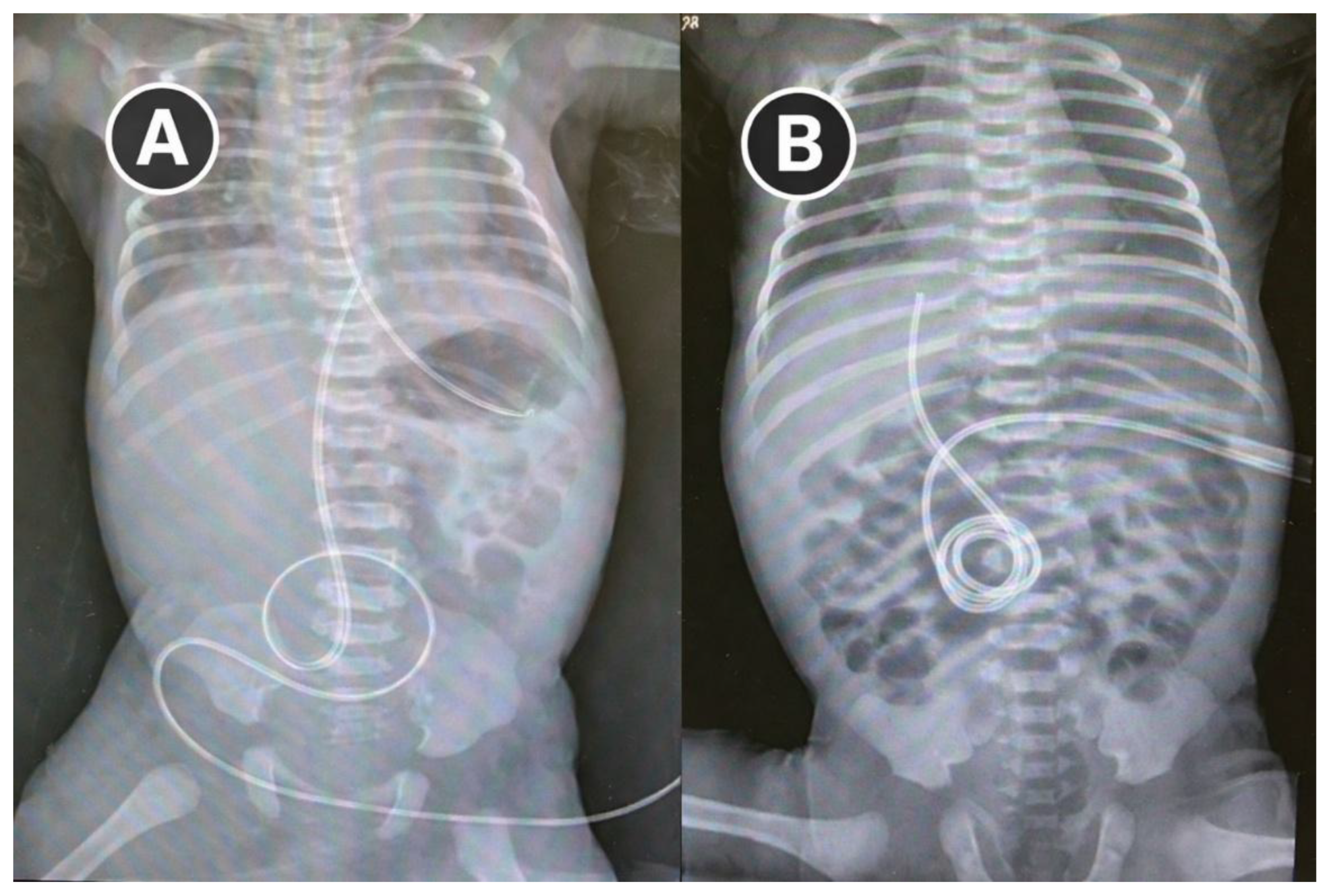
2.3. Neonatal Outcome
2.4. Pharmacokinetic Assessment
2.5. Psychological Assessment and Follow-Up of the Parental Couple
3. Materials and Methods
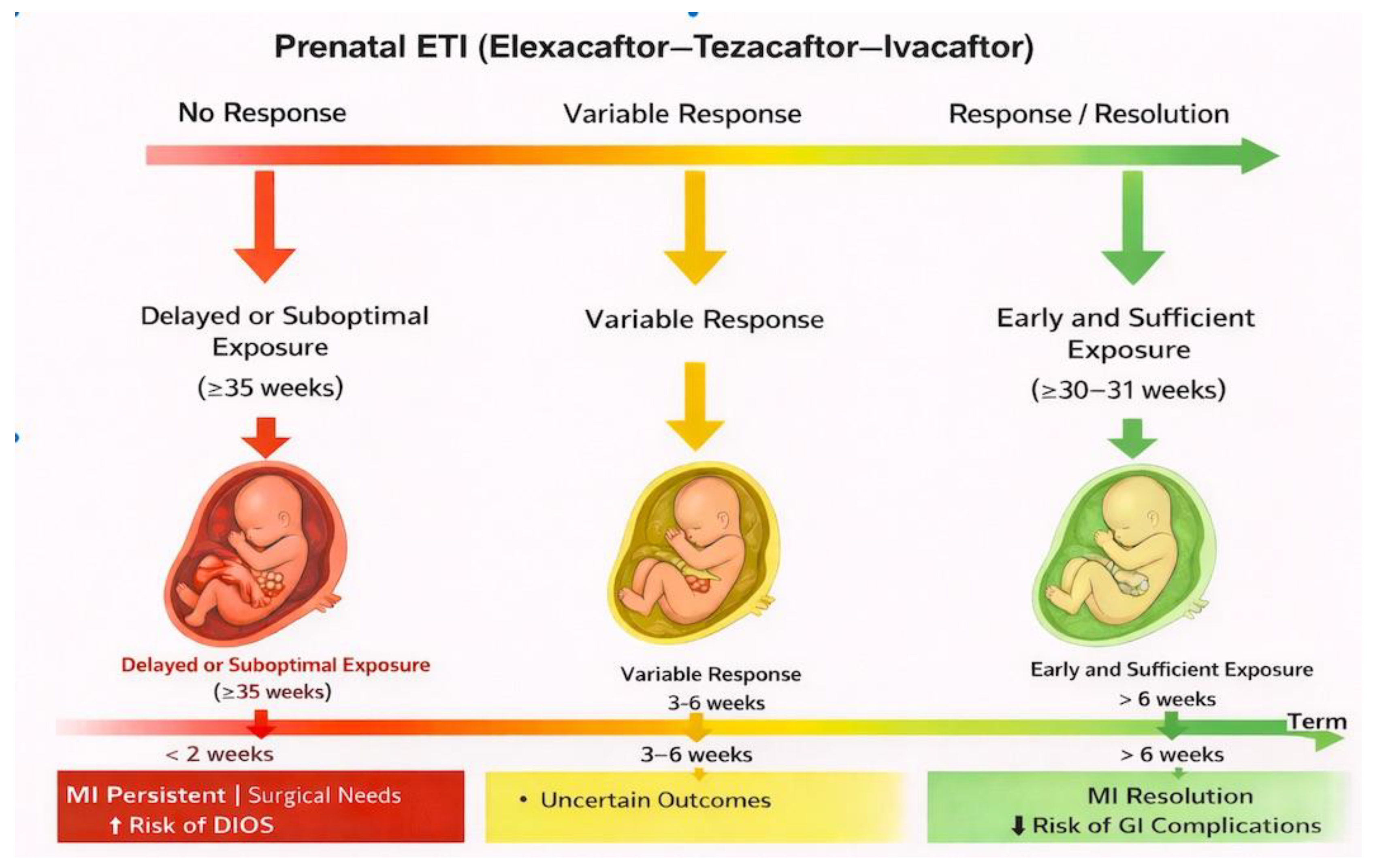
4. Results
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CF | Cystic fibrosis |
| DIOS | Distal ileal obstruction syndrome |
| ELX | Elexacaftor |
| ETI | Elexacaftor/tezacaftor/ivacaftor |
| IVA | Ivacaftor |
| LC–MS/MS | Liquid chromatography–tandem mass spectrometry |
| MI | Meconium ileus |
| pwCF | People with cystic fibrosis |
| QoL | Quality of life |
| TEZ | Tezacaftor |
| US | Ultrasound |
References
- Shteinberg, M.; Haq, I.J.; Polineni, D.; Davies, J.C. Cystic fibrosis. Lancet 2021, 397, 2195–2211. [Google Scholar] [CrossRef] [PubMed]
- Donos, M.A.; Țarcă, E.; Cojocaru, E.; Țarcă, V.; Butnariu, L.I.; Bernic, V.; Popovici, P.; Roșu, S.T.; Tîrnovanu, M.C.; Ionescu, N.S.; et al. Evolution and prognostic variables of cystic fibrosis in children and young adults: A narrative review. Diagnostics 2025, 15, 1940. [Google Scholar] [CrossRef] [PubMed]
- Sathe, M.; Houwen, R. Meconium ileus in cystic fibrosis. J. Cyst. Fibrosis 2017, 16, S32–S39. [Google Scholar] [CrossRef] [PubMed]
- Escobar, M.A.; Grosfeld, J.L.; Burdick, J.J.; Powell, R.L.; Jay, C.L.; Wait, A.D.; West, K.W.; Billmire, D.F.; Scherer, L.R.; Engum, S.A.; et al. Surgical considerations in cystic fibrosis: A 32-year evaluation of outcomes. Surger 2005, 138, 560–571. [Google Scholar] [CrossRef] [PubMed]
- Taylor-Cousar, J.L.; Jain, R. Maternal and fetal outcomes following elexacaftor–tezacaftor–ivacaftor use during pregnancy and lactation. J. Cyst. Fibros 2021, 20, 402–406. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Magaret, A.; Vu, P.T.; VanDalfsen, J.M.; Keller, A.; Wilson, A.; Putman, M.S.; Mayer-Hamblett, N.; Esther, C.R.; Taylor-Cousar, J.L. Prospectively evaluating maternal and fetal outcomes in the era of CFTR modulators: The MAYFLOWERS observational clinical trial study design. BMJ Open Respir. Res. 2022, 9, e001289. [Google Scholar] [CrossRef] [PubMed]
- Bonnel, A.S.; Bihouée, T.; Ribault, M.; Driessen, M.; Grèvent, D.; Foissac, F.; Truong, N.H.; Benhamida, M.; Arnouat, B.; Borghese, R.; et al. First real-world study of fetal therapy with CFTR modulators in cystic fibrosis: Report from the MODUL-CF study. J. Cyst. Fibros. 2025, 24, 457–465. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Kaftrio. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/kaftrio](https://www.ema.europa.eu/en/medicines/human/EPAR/kaftrio (accessed on 18 February 2026).
- Mancini, A.; Simeoli, R.; Cristiani, L.; Cairoli, S.; Ciciriello, F.; Boni, A.; Alghisi, F.; Rossi, C.; Antonetti, G.; Vici, C.D.; et al. Development and validation of a new LC–MS/MS method for simultaneous quantification of ivacaftor, tezacaftor and elexacaftor plasma levels in pediatric cystic fibrosis patients. Pharmaceuticals 2025, 18, 1028. [Google Scholar] [CrossRef] [PubMed]
- Quittner, A.L.; Abbott, J.; Georgiopoulos, A.M.; Goldbeck, L.; Smith, B.; Hempstead, S.E.; Marshall, B.; Sabadosa, K.A.; Elborn, S. International Committee on Mental Health in Cystic Fibrosis: Consensus statements for screening and treating depression and anxiety. Thorax 2016, 71, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.; Löwe, B. A brief measure for assessing generalized anxiety disorder: The GAD-7. Arch. Intern. Med. 2006, 166, 1092–1097. [Google Scholar] [CrossRef] [PubMed]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B. The PHQ-9: Validity of a brief depression severity measure. J. Gen. Intern. Med 2001, 16, 606–613. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. *Diagnostic and Statistical Manual of Mental Disorders (DSM-5), 5th ed.; American Psychiatric Publishing: Washington, DC, USA, 2013. [Google Scholar]
- Copeland, D.R.; St Peter, S.D.; Sharp, S.W.; Islam, S.; Cuenca, A.; Tolleson, J.S.; Dassinger, M.S.; Little, D.C.; Jackson, R.J.; Kokoska, E.R.; et al. Diminishing role of contrast enema in simple meconium ileus. *. Pediatr. Surg 2009, 44, 2130–2132. [Google Scholar] [CrossRef] [PubMed]
- Szentpetery, S.; Foil, K.; Hendrix, S.; Gray, S.; Mingora, C.; Head, B.; Johnson, D.; Flume, P.A. A case report of CFTR modulator administration via carrier mother to treat meconium ileus in a F508del homozygous fetus. J. Cyst. Fibros. 2022, 21, 721–724. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Montes, E.; Salcedo Lobato, E.; Galindo Izquierdo, A.; García Alcázar, D.; Villalain González, C.; Moral-Pumarega, M.T.; Bustos Lozano, G.; Luna-Paredes, C. Prenatal CFTR modulator therapy: A promising way to change the impact of cystic fibrosis. Fetal Diagn. Ther. 2023, 50, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Fortner, C.N.; Seguin, J.M.; Kay, D.M. Normal pancreatic function and false-negative CF newborn screen in a child born to a mother taking CFTR modulator therapy during pregnancy. *J. Cyst. Fibros. 2021, 20, 835–836. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, A.; Martiniano, S.L.; Sagel, S.D.; Zaretsky, M.V.; Zemanick, E.T.; Hoppe, J.E. Outcomes of prenatal use of elexacaftor/tezacaftor/ivacaftor in carrier mothers to treat meconium ileus in fetuses with cystic fibrosis. J. Cyst. Fibros. 2025, 24, 466–468. [Google Scholar] [CrossRef] [PubMed]
- Szentpetery, S.; Riva, D.; Blumenfeld, Y.J.; Engelhardt, J.F.; Luna-Paredes, C.; Milla, C.; Schneider-Futschik, E.K.; Sheffield, J.S.; Darrah, R.; Davies, J.C.; et al. PRenatal mOdulator treatment to PrEvent CF complicaTions (PROTECT) workshop report. J. Cyst. Fibros. 2025, 24, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Destoop, M.; Brantner, C.; Wilms, E.B.; Tytgat, S.; Peels, B.; van der Graaf, R.; Liem, T.B.Y.; de Winter-de Groot, K.M. CFTR modulator therapy via carrier mother to treat meconium ileus in a F508del homozygous fetus: Insights from an unsuccessful case. J. Cyst. Fibros. 2025, 24, 476–478. [Google Scholar] [CrossRef] [PubMed]
- Hong, E.; Hebert, M.F.; Fay, E.; Turner, G.; Eshaghian, P.; Trimble, A.; Chung, P.S.; Rao, A.P.; Beringer, P.M. Predicting maternal and fetal exposures of elexacaftor–tezacaftor–ivacaftor during pregnancy through physiologically based pharmacokinetic models. Clin. Pharmacol. Ther. 2025, 118, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Kowalik, A.; Roberts, E.; Harris, A.H.; Sund, M.; Wird, S.; Kvist, O.; Hjelte, L. Clinical outcomes of two infants with cystic fibrosis, including presence of the vas deferens, born to a woman with cystic fibrosis taking CFTR modulators during both pregnancies. J. Cyst. Fibros2024 23, 1027–1030. [CrossRef] [PubMed]
| Sample Type | Elexacaftor (ELX) | Tezacaftor (TEZ) |
Ivacaftor (IVA) |
|---|---|---|---|
| Mother plasma concentration 12 days post ETI initiation (µg/mL) | 8.59 | 0.95 | 0.75 |
| Plasma concentration from cord blood (Vein) (µg/mL) | 3.03 | 2.33 | 0.44 |
| Plasma concentration from cord blood (Artery) (µg/mL) | 2.88 | 2.38 | 0.43 |
| Amniotic Fluid (µg/mL) | 0.18 | 0.74 | 0.10 |
| Cord-to-maternal plasma concentration ratio | 0.34 | 2.48 | 0.58 |
| Author (Year) | Maternal status | GA at ETI initiation | Duration of prenatal exposure | Fetal MI outcome | Pancreatic status at birth | Sweat chloride | Postnatal ETI exposure |
|---|---|---|---|---|---|---|---|
| Szentpetery et al. (2022) | Healthy carrier | 32 weeks | 27 days | Complete in utero resolution | Pancreatic sufficient (2 wks) | Borderline | Continued during breastfeeding |
| Gómez-Montes et al. (2023) | Healthy carrier | 31 weeks | ~8 weeks | Resolution before birth | Pancreatic insufficient | Positive | Not specified |
| Fortner et al. (2019) | Mother with CF | Early pregnancy (continuous) | Entire gestation | No MI reported | Transient pancreatic sufficiency | False-negative NBS | Continued |
| PROTECT workshop (2024) | Multicenter cases | ~30–32 weeks (median) | Mean ~6 weeks | Resolution in most; shortest exposure → poorest outcomes | Variable | Variable | Variable |
| MODUL-CF (2024) | Multicenter cohort | Median 30 weeks | Median 5 weeks | MI resolution within ~2 weeks | Not uniformly preserved | Variable | Not systematic |
| Metcalf et al. (2025) | Mixed (series) | Variable | Variable | Early/longest exposure → full resolution; shortest → surgery | Variable | Variable | Variable |
| Destoop et al. (2025) |
Healthy carrier | 27 weeks | 11 weeks | No resolution | Pancreatic insufficient | Positive | Discontinued postpartum |
| Present case (Italy) | Healthy carrier | 33 weeks | 3 weeks | In utero stabilization; no surgery | Initially sufficient → PI at 1 month | Positive | Discontinued postpartum |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





