Submitted:
19 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Background: Endophytic fungi are known for diverse bioactive compounds with immense potential for agriculture and medicinal applications. Coniochaeta dendrobiicola isolated from root of the Dedrobium longicornu was investigated for its antioxidant and metabolite composition. The present study compares the antioxidant properties, flavonoid and phenolic contents and metabolic profiles of broth and mycelium extracts. Methanolic broth and mycelium extracts were tested for antioxidant potential using DPPH, while total flavonoid and phenolic content were measured using a UV-VIS spectrophotometer High-resolution mass spectrometry (HRMS) revealed a markedly richer and more diverse metabolite profile in the broth extract compared to the mycelium fraction. The broth extract exhibited significantly higher antioxidant activity, flavonoid, and phenolic contents, correlating with the presence of diverse bioactive compounds, including indole derivatives, flavonoids, phenolic acids, quinoline derivatives, and antifungal metabolites. Notably, several indole-related and phenolic compounds detected predominantly in the broth are known for antioxidant, antimicrobial, and plant growth–promoting properties. These findings indicate that C. dendrobiicola actively secretes biologically relevant secondary metabolites into the extracellular medium, highlighting its potential for agricultural and pharmaceutical applications.
Keywords:
1. Introduction
2. Materials and Methods
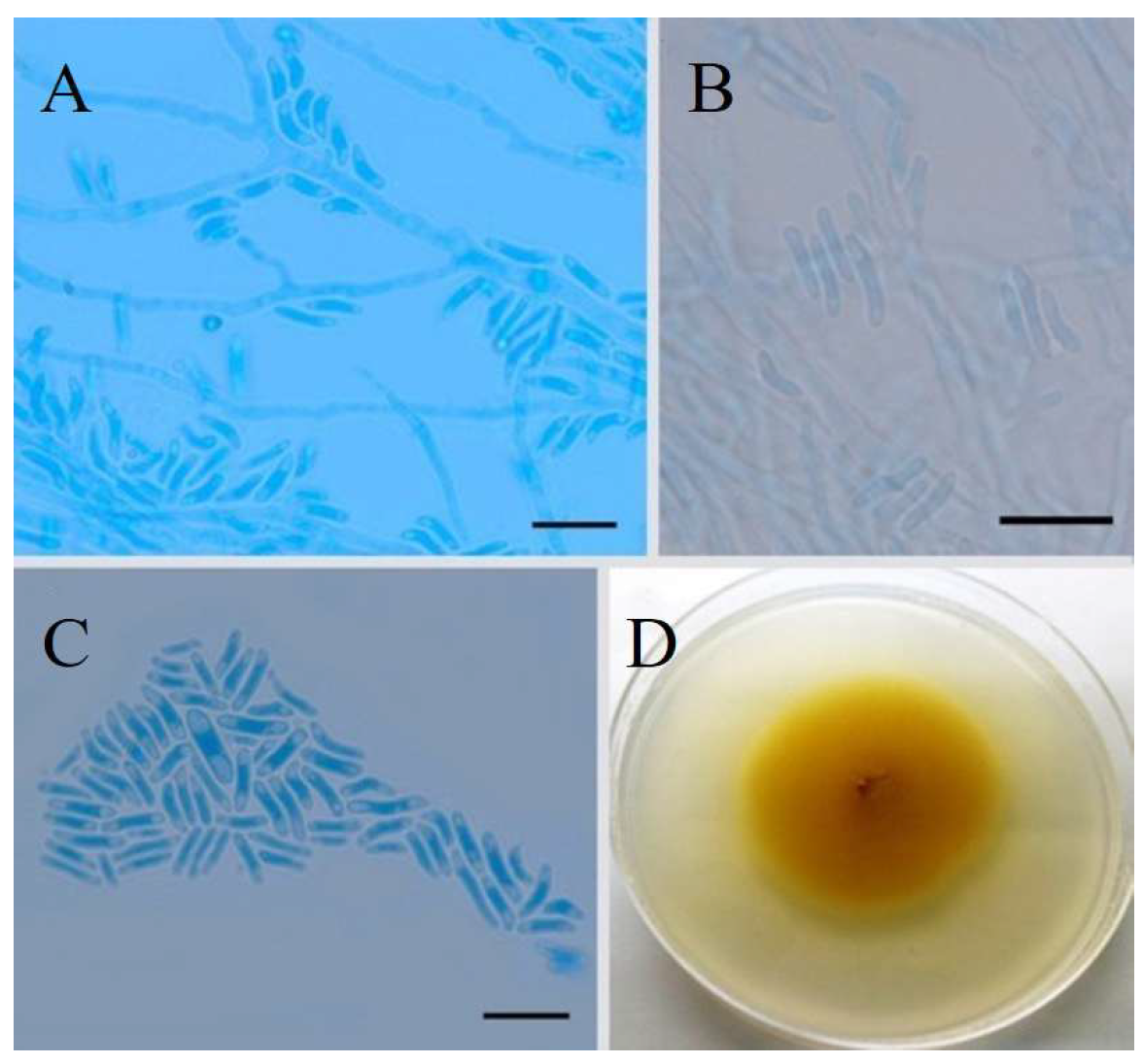
Fungal Extract Preparation
Estimation of Total Phenolic and Total Flavonoid Content of Fungal Extracts
Free Radical Scavenging Assay
High-Resolution Accurate Mass Spectrometry (HRMS) System Parameter
Statistical Analysis
3. Result
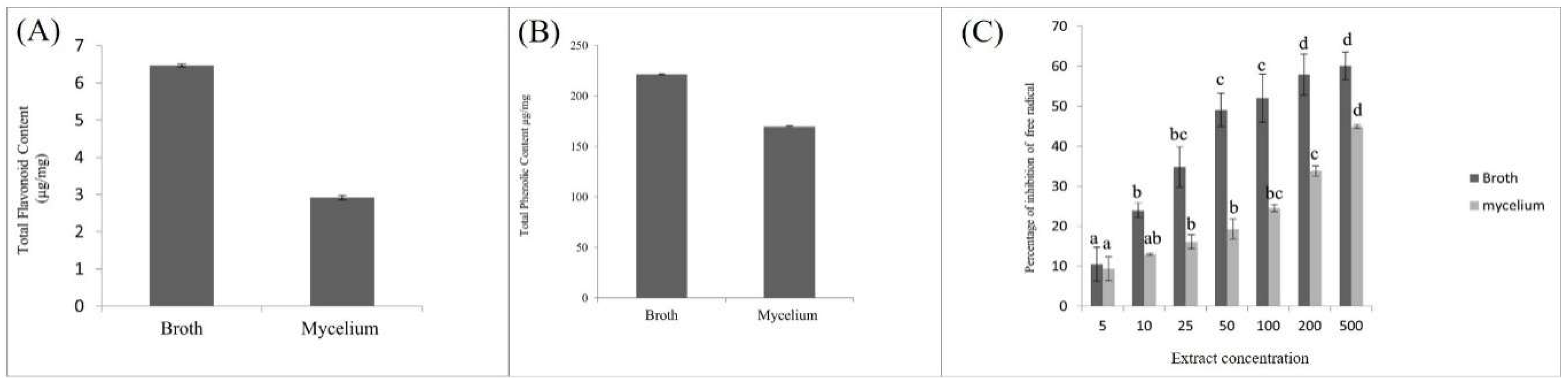
Total Phenolic Content, Total Flavonoid Content, and Antioxidant Activity of Broth and Mycelium
Comparative Antioxidant Activity of Fungal Mycelium and Broth Extract
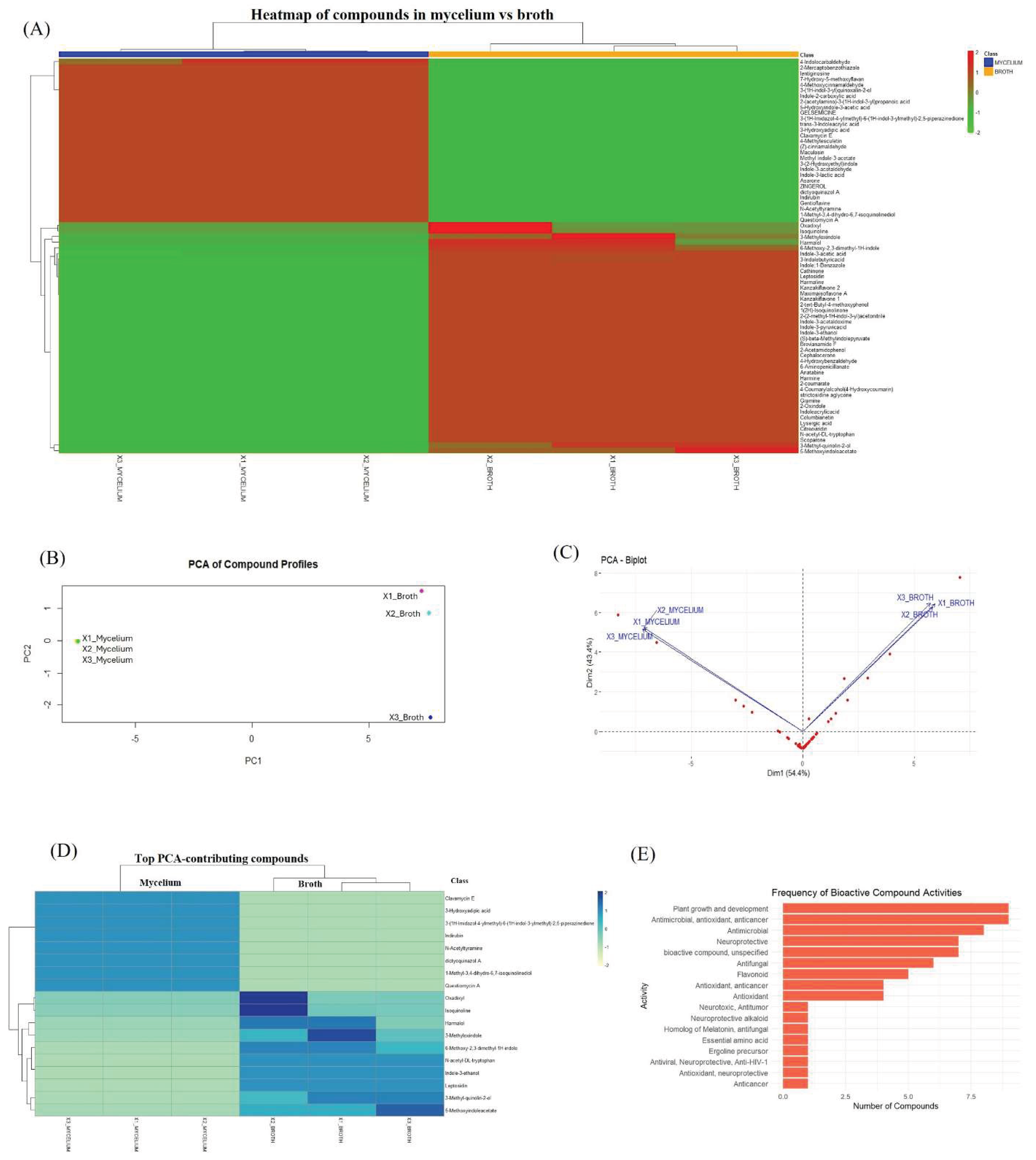
Comparative Analysis of Metabolites Produced by Intracellular Mycelium and Extracellular Broth
4. Discusssion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interests
References
- Alam, B.; Lǐ, J.; Gě, Q.; Khan, M.A.; Gōng, J.; Mehmood, S.; Yuán, Y.; Gǒng, W. Endophytic fungi: From symbiosis to secondary metabolite communications or vice versa? Front. Plant Sci. 2021, 12, 791033. [Google Scholar] [CrossRef]
- Baron, N.C.; Rigobelo, E.C. Endophytic fungi: A tool for plant growth promotion and sustainable agriculture. Mycology 2021, 13, 39–55. [Google Scholar] [CrossRef]
- Kashyap, A.K.; Dubey, S.K.; Shah, S.; Kumar, A. A short review on genes regulating biosynthesis of major secondary metabolites. In Phytochemical Genomics: Plant Metabolomics and Medicinal Plant Genomics; Elsevier: Amsterdam, The Netherlands, 2023; pp. 501–519. [Google Scholar]
- Pant, B.; Shah, S.; Shrestha, R.; Pandey, S.; Joshi, P.R. An overview on orchid endophytes. In Mycorrhiza—Nutrient Uptake, Biocontrol, Ecorestoration; Varma, A., Prasad, R., Tuteja, N., Eds.; Springer: Cham, Switzerland, 2017; pp. 435–453. [Google Scholar] [CrossRef]
- Phukhamsakda, C.; Nilsson, R.H.; Bhunjun, C.S.; et al. The numbers of fungi: Contributions from traditional taxonomic studies and challenges of metabarcoding. Fungal Divers. 2022, 114, 327–386. [Google Scholar] [CrossRef]
- Saini, H.P.; Meena, M.; Sahoo, A.; Mehta, T. A review on fungal endophytes of the family Fabaceae, their metabolic diversity and biological applications. Heliyon 2025, 11, e42153. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Okyere, S.K.; Wang, S.; et al. Endophytic fungi: An effective alternative source of plant-derived bioactive compounds for pharmacological studies. J. Fungi 2022, 8, 205. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Meshram, V.; Gupta, M.; et al. Fungal endophytes: Microfactories of novel bioactive compounds with therapeutic interventions. Biomolecules 2023, 13, 1038. [Google Scholar] [CrossRef]
- Shah, S.; Shah, B.; Sharma, R.; et al. Colonization with non-mycorrhizal culturable endophytic fungi enhances orchid growth and indole acetic acid production. BMC Microbiol. 2022, 22, 101. [Google Scholar] [CrossRef]
- Pant, B.; Joshi, P.R.; Maharjan, S.; et al. Comparative cytotoxic activity of wild harvested stems and in vitro-raised protocorms of Dendrobium chryseum. Adv. Pharmacol. Pharm. Sci. 2021, 2021, 8839728. [Google Scholar] [CrossRef]
- Li, T.; Yang, W.; Wu, S.; Selosse, M.A.; Gao, J. Progress and prospects of mycorrhizal fungal diversity in orchids. Front. Plant Sci. 2021, 12, 646325. [Google Scholar] [CrossRef]
- Garg, A.; Shah, S.; Patel, T.B.B.; Gautam, V.; Kumar, D. Contrasting effects of melatonin on Brassica juncea and Alternaria brassicae. Plant Stress 2025, 100845. [Google Scholar] [CrossRef]
- Miliauskas, G.; Venskutonis, P.R.; Van Beek, T.A. Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem. 2004, 85, 231–237. [Google Scholar] [CrossRef]
- Crous, P.W.; Carnegie, A.J.; Wingfield, M.J.; et al. Fungal Planet description sheets: 868–950. Persoonia 2019, 42, 291–473. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.H.; Xin, F.Z.; Xue, Y.; et al. Indole-3-propionic acid inhibits gut dysbiosis. Exp. Mol. Med. 2019, 51, 1–14. [Google Scholar] [CrossRef]
- Chua, R.W.; Song, K.P.; Ting, A.S.Y. Characterization of antimicrobial compounds from endophytic Fusarium incarnatum. Int. Microbiol. 2024, 27, 977–992. [Google Scholar] [CrossRef] [PubMed]
- Mehta, T.; Meena, M.; Nagda, A. Bioactive compounds of Curvularia species. Front. Microbiol. 2022, 13, 1069095. [Google Scholar] [CrossRef]
- Chen, M.; Liang, J.; Wang, Y.; et al. A new benzaldehyde from Aspergillus terreus. J. Zhejiang Univ. Sci. B 2022, 23, 230–240. [Google Scholar] [CrossRef]
- Reveglia, P.; Pacetti, A.; Masi, M.; et al. Phytotoxic metabolites produced by Diaporthe eres. Nat. Prod. Res. 2021, 35, 2872–2880. [Google Scholar] [CrossRef]
- Zang, X.; Wang, Y.; Han, C.; et al. 2-Acetamidophenol suppresses atherosclerosis. Mar. Drugs 2024, 22, 513. [Google Scholar] [CrossRef]
- Xue, M.; Hou, X.; Fu, J.; et al. Bioactive secondary metabolites from fungi triggered by epigenetic modifiers. J. Fungi 2023, 9, 172. [Google Scholar] [CrossRef]
- Xu, D.; Xue, M.; Shen, Z.; et al. Phytotoxic secondary metabolites from fungi. Toxins 2021, 13, 261. [Google Scholar] [CrossRef]
- Roager, H.M.; Licht, T.R. Microbial tryptophan catabolites in health and disease. Nat. Commun. 2018, 9, 3294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Du, H.F.; Liu, Y.F.; et al. Novel anti-inflammatory diketopiperazine alkaloids. Appl. Microbiol. Biotechnol. 2024, 108, 194. [Google Scholar] [CrossRef] [PubMed]
- Okano, T.; Kobayashi, N.; Izawa, K.; et al. Genes responsible for citreoviridin biosynthesis. Toxins 2020, 12, 125. [Google Scholar] [CrossRef] [PubMed]
- Auckloo, B.N.; Pan, C.; Akhter, N.; et al. Stress-driven discovery of cryptic antibiotics. Front. Microbiol. 2017, 8, 1450. [Google Scholar] [CrossRef]
- Shiratori, N.; Kobayashi, N.; Tulayakul, P.; et al. Occurrence of Penicillium brocae and P. citreonigrum. Toxins 2017, 9, 194. [Google Scholar] [CrossRef]
- Jastrzębski, M.K.; Kaczor, A.A.; Wróbel, T.M. Methods of lysergic acid synthesis. Molecules 2022, 27, 7322. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, X.; Nong, X.; et al. Oxindole alkaloids from Penicillium commune. Tetrahedron 2015, 71, 1640–1645. [Google Scholar] [CrossRef]
- Sharma, S.; Monga, Y.; Gupta, A.; Singh, S. 2-Oxindole and related heterocycles. RSC Adv. 2023, 13, 14249–14267. [Google Scholar] [CrossRef]
- Cabutaje, E.M.; Ueno, K.; Osaki-Oka, K.; et al. Phthalide derivatives and an oxindole compound from Pleurotus ostreatus. J. Pestic. Sci. 2023, 48, 156–167. [Google Scholar] [CrossRef]
- Fu, S.F.; Wei, J.Y.; Chen, H.W.; et al. Indole-3-acetic acid in fungal interactions. Plant Signal. Behav. 2015, 10, e1048052. [Google Scholar] [CrossRef]
- Kumar, P.; Lee, J.H.; Lee, J. Diverse roles of microbial indole compounds. Biol. Rev. 2021, 96, 2522–2545. [Google Scholar] [CrossRef]
- Tang, Z.; Wang, Y.; Yang, J.; et al. Flavonoid-producing endophytic fungi. PeerJ 2020, 8, e8978. [Google Scholar] [CrossRef] [PubMed]
- Kong, M.; Liang, J.; Ali, Q.; et al. 5-Methoxyindole affects Fusarium graminearum. Int. J. Mol. Sci. 2021, 22, 10991. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.R.; Shilts, T.; Ertürk, U.; et al. Indole derivatives produced by Colletotrichum acutatum. FEMS Microbiol. Lett. 2003, 226, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, J.; Budzyńska, B.; Kurach, Ł.; et al. Neuropsychopharmacological profiling of scoparone. Sci. Rep. 2022, 12, 822. [Google Scholar] [CrossRef]
- Liu, C.M.; Liang, X.; Yao, F.H.; Qi, S.H. Aromatic polyketides and isoquinoline alkaloids. Tetrahedron 2022, 112, 133067. [Google Scholar] [CrossRef]
- Shang, X.F.; Yang, C.J.; Morris-Natschke, S.L.; et al. Biologically active isoquinoline alkaloids. Med. Res. Rev. 2020, 40, 2212–2289. [Google Scholar] [CrossRef]
- Nord, C.; Levenfors, J.J.; Bjerketorp, J.; et al. Antibacterial isoquinoline alkaloids. Molecules 2019, 24, 4616. [Google Scholar] [CrossRef]
- Umashankar, P.; Nygård, Y. Filamentous fungi as emerging cell factories. Fungal Biol. Biotechnol. 2024, 11, 19. [Google Scholar] [CrossRef]
- Nag, S.; Chowdhury, C.; Das, N. Antioxidant potency of kanzakiflavone-2. J. Mol. Struct. 2021, 1231, 129928. [Google Scholar] [CrossRef]
- Murthy, M.S.R.; Rao, E.V. Maxima isoflavone. J. Phytochemistry 1985, 24, 767–768. [Google Scholar] [CrossRef]
- El Gendy, M.A.; Soshilov, A.A.; Denison, M.S.; El-Kadi, A.O.S. Harmaline and harmalol inhibit CYP1A1. Toxicol. Lett. 2012, 213, 306–313. [Google Scholar]
- Hu, Y.; Yu, X.; Yang, L.; et al. Antitumor effects of harmine. Front. Oncol. 2024, 14, 1382142. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.K.; Saxena, V.K.; Singh, K.V. A leptosidin glycoside. Phytochemistry 1981, 20, 2605. [Google Scholar] [CrossRef]
- Caturegli, P.; De Remigis, A.; Ferlito, M.; et al. Anatabine ameliorates autoimmune thyroiditis. Endocrinology 2012, 153, 4580–4587. [Google Scholar] [CrossRef]
- Boyce, G.R.; Gluck-Thaler, E.; Slot, J.C.; et al. Psychoactive alkaloids from cicada pathogens. Fungal Ecol. 2019, 41, 147–164. [Google Scholar] [CrossRef]
- Zhang, L.; Ge, Y.; Li, J.; et al. Simultaneous determination of columbianetin derivatives. J. Pharm. Biomed. Anal. 2018, 153, 221–231. [Google Scholar] [CrossRef]
- Robinson, M.; Riov, J.; Sharon, A. Indole-3-acetic acid biosynthesis in Colletotrichum gloeosporioides. Appl. Environ. Microbiol. 1998, 64, 5030–5032. [Google Scholar] [CrossRef]
- Peng, J.; Wang, X.; Wang, H.; et al. Advances in understanding grapevine downy mildew. Mol. Plant Pathol. 2024, 25, e13401. [Google Scholar] [CrossRef]
- Tang, J.; Li, Y.; Zhang, L.; et al. Biosynthetic pathways and functions of indole-3-acetic acid in microorganisms. Microorganisms 2023, 11, 2077. [Google Scholar] [CrossRef]
- King, H.D.; Langhärig, J.; Sanglier, J.J. Clavamycins. J. Antibiot. 1986, 39, 510–515. [Google Scholar] [CrossRef]
- Kozlovskiĭ, A.G.; Zhelifonova, V.P.; Antipova, T.V.; et al. Penicillium expansum producing xanthocillin X. Prikl. Biokhim. Mikrobiol. 2004, 40, 344–349. [Google Scholar] [PubMed]
- Pan, H.; Li, H.; Wu, S.; et al. De novo biosynthesis of N-acetyltyramine. Enzyme Microb. Technol. 2023, 162, 110149. [Google Scholar] [CrossRef] [PubMed]
- Karagianni, E.P.; Kontomina, E.; Davis, B.; et al. Xenobiotic metabolizing N-acetyltransferases in fungi. Sci. Rep. 2015, 5, 12900. [Google Scholar] [CrossRef] [PubMed]
- Correia, J.; Borges, A.; Simões, M.; Simões, L.C. Beyond penicillin: Filamentous fungi for drug discovery. Antibiotics 2023, 12, 1250. [Google Scholar] [CrossRef]
- Singh, V.K.; Kumar, A. Secondary metabolites from endophytic fungi. Symbiosis 2023, 1–15. [Google Scholar] [CrossRef]
- Wu, X.; Zou, K.; Liu, X.; et al. Intracellular and extracellular flavonoids from Aspergillus sp. Front. Microbiol. 2022, 13, 972294. [Google Scholar] [CrossRef]
- Ponnusamy, K.; Petchiammal, C.; Mohankumar, R.; Hopper, W. Antifungal activity of indirubin. J. Ethnopharmacol. 2010, 132, 349–354. [Google Scholar] [CrossRef]
- Lee, I.K.; Yun, B.S.; Han, G.; et al. Dictyoquinazols A–C. J. Nat. Prod. 2002, 65, 1769–1772. [Google Scholar] [CrossRef]
- Lizarme, Y.; Wangsahardja, J.; Marcolin, G.M.; et al. Synthesis and neuroprotective activity of dictyoquinazol A. Bioorg. Med. Chem. 2016, 24, 1480–1487. [Google Scholar] [CrossRef]
- Mao, Q.Q.; Xu, X.Y.; Cao, S.Y.; et al. Bioactive compounds of ginger. Foods 2019, 8, 185. [Google Scholar] [CrossRef]
- Bai, X.; Sheng, Y.; Tang, Z.; et al. Polyketides from Aspergillus. J. Fungi 2023, 9, 261. [Google Scholar] [CrossRef]
- Wang, P.; Ma, L.; Jin, J.; et al. Anti-aflatoxigenic mechanism of cinnamaldehyde. Sci. Rep. 2019, 9, 10499. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.L.; Liang, J.J.; Xue, Y.; et al. Gelsemium and its endophytic fungi. Curr. Top. Med. Chem. 2023, 23, 2452–2487. [Google Scholar] [CrossRef] [PubMed]
- Kumla, J.; Suwannarach, N.; Matsui, K.; Lumyong, S. Biosynthetic pathway of indole-3-acetic acid. PLoS ONE 2020, 15, e0227478. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T.; Odamaki, T.; Xiao, J.Z. Production of indole-3-lactic acid. Microorganisms 2019, 7, 340. [Google Scholar] [CrossRef]
- Que, Y.; Huang, D.; Gong, S.; et al. Indole-3-carboxylic acid from Lasiodiplodia pseudotheobromae. Front. Cell. Infect. Microbiol. 2022, 12, 898500. [Google Scholar] [CrossRef]
- Sun, P.; Huang, Y.; Yang, X.; et al. Role of indole derivatives in plant growth. Front. Plant Sci. 2023, 13, 1120613. [Google Scholar] [CrossRef]
- Ye, X.; Li, H.; Anjum, K.; et al. Dual role of indoles from intestinal microbiota. Front. Immunol. 2022, 13, 903526. [Google Scholar] [CrossRef]
- Paudel, B.; Maharjan, R.; Rajbhandari, P.; et al. Maculosin from Streptomyces sp. Pharm. Biol. 2021, 59, 933–936. [Google Scholar] [CrossRef]
- Morozumi, S. o-Methoxycinnamaldehyde from cinnamon. Appl. Environ. Microbiol. 1978, 36, 577–583. [Google Scholar] [CrossRef]
- Pina, J.; de Castro, C.S.; Delgado-Pinar, E.; Seixas de Melo, J.S. 4-Methylesculetin derivatives. J. Mol. Liq. 2019, 278, 616–626. [Google Scholar] [CrossRef]
- Pastuszak, I.; Molyneux, R.J.; James, L.F.; Elbein, A.D. Lentiginosine inhibits amyloglucosidase. Biochemistry 1990, 29, 1886–1891. [Google Scholar] [CrossRef] [PubMed]
- Machihara, K.; Tanaka, H.; Hayashi, Y.; et al. Questiomycin A stimulates sorafenib-induced cell death. Biochem. Biophys. Res. Commun. 2017, 492, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jia, Q.; Li, N.; et al. Recent developments of gramine. Molecules 2023, 28, 5695. [Google Scholar] [CrossRef]
- Tsivileva, O.M.; Koftin, O.V.; Evseeva, N.V. Coumarins as fungal metabolites. Antibiotics 2022, 11, 1156. [Google Scholar] [CrossRef]
- Fu, J.; Hu, L.; Shi, Z.; Sun, W.; Yue, D.; Wang, Y.; Ma, X.; Ren, Z.; Zuo, Z.; Peng, G.; Zhong, Z.; Deng, J.; Cao, S.; Hu, Y. Two metabolites isolated from endophytic fungus Coniochaeta sp. F-8 in Ageratina adenophora exhibit antioxidative activity and cytotoxicity. Nat. Prod. Res. 2021, 35, 2840–2848. [Google Scholar] [CrossRef]
- Chen, K.H.; Liao, H.L.; Arnold, A.E.; Korotkin, H.B.; Wu, S.H.; Matheny, P.B.; Lutzoni, F. Comparative transcriptomics of fungal endophytes in co-culture with their moss host Dicranum scoparium reveals fungal trophic lability and moss unchanged to slightly increased growth rates. New Phytol. 2022, 234, 1832–1847. [Google Scholar] [CrossRef]
- Nichols, N.N.; Mertens, J.A.; Frazer, S.E.; Hector, R.E. Growth of Coniochaeta Species on Acetate in Biomass Sugars. Fermentation 2022, 8, 721. [Google Scholar] [CrossRef]
| Compound Name | Enriched In | Nature of compound | Reported Source | Reported Activity | References |
|---|---|---|---|---|---|
| 3-Indolepropionic acid | Broth | Indole compound | Unreported from fungi | Antioxidant, neuroprotective | [15] |
| 3-Methyl-quinolin-2-ol | Broth | Isoquinoline | Fusarium incarnatum | Antifungal | [16] |
| 4-Hydroxybenzaldehyde | Broth | Phenolic compound | Curvularia, Diaporthe, Aspergillus | Antimicrobial | [17,18,19] |
| 2-Acetamidophenol | Broth | Phenolic compound | Penicillium chrysogenum, Aspergillus sp., Pseudomons chlororaphis | Antimicrobial, antioxidant, anticancer | [20] |
| N-acetyl-DL-tryptophan | Broth | Derivative of amino acid | Fungi, Bacteria | Essential amino acid | [21,22,23] |
| Brevianamide F | Broth | 2,5-diketopiperazine | Aspergillus, Penicillium | Antimicrobial | [24] |
| Citreoviridin | Broth | Mycotoxin | Penicillium, Aspergillus | Neurotoxic, Antitumor | [25,26,27] |
| Lysergic acid | Broth | Ergoline alkaloids | Claviceps purpurea, Periglandula spp. | Ergoline precursor | [28] |
| 2-Oxindole | Broth | Indole compound | Colletotrichum fragariae, Aspergillus versicolor, and Penicillium commune, plant and human gut | Antimicrobial, antioxidant, anticancer | [29,30,31] |
| Indole 3 ethanol | Broth | Indole compound | Endophytic, mycorrhizal fungi | Plant growth and development | [32,33,34] |
| Indole 3 butyric acid | Broth | Indole compound | Endophytic, mycorrhizal fungi | Plant growth and development | [32,33,34] |
| Indole 3 pyruvic acid | Broth | Indole compound | Endophytic, mycorrhizal fungi | Plant growth and development | [32,33,34] |
| 5-Methoxyindole | Broth | Indole compound | Unreported from fungi | Homolog of Melatonin, antifungal | [35] |
| Indole acrylic acid | Broth | Indole compound | Endophytic, mycorrhizal fungi | Plant growth and development | [32,33,34] |
| 3-Methyloxindole | Broth | Indole compound | Unreported from fungi | Bioactive compound, unspecified | [33,34,36] |
| Indole;1-Benzazole | Broth | Indole compound | Unreported from fungi | Bioactive compound, unspecified | [33,34,36] |
| 2-(2-methyl-1H-indol-3-yl)acetonitrile | Broth | Indole compound | Unreported from fungi | Bioactive compound, unspecified | |
| Scoparone | Broth | Phenolic compound | Plant derivatives | Neuroprotective | [37] |
| 5-Methoxyindoleacetate | Broth | Indole compound | Plant derivatives | Antioxidant | [34,35,37] |
| Indole-3-acetaldoxime | Broth | Indole compound | Plant derivatives | Antioxidant | [34,35,37] |
| 6-Methoxy-2,3-dimethyl-1H-indole | Broth | Indole compound | Plant derivatives | Antioxidant | [33,34,36] |
| Gramine | Broth | Indole alkaloid | Plant derivatives | Antimicrobial, antioxidant, anticancer | [33,34,36] |
| 1(2H)-Isoquinolinone | Broth | Isoquinolinone | Plant derivatives | Neuroprotective | [38,39,40] |
| 4-Coumarylalcohol(4-Hydroxycoumarin) | Broth | Coumarin | Plant derivatives | Antimicrobial, antioxidant, anticancer | [41] |
| Kanzakiflavone 1 | Broth | Flavonoid | Flavonoid | Flavonoid | [42] |
| Maximaisoflavone A | Broth | Flavonoid | Flavonoid | Flavonoid | [43] |
| Kanzakiflavone 2 | Broth | Flavonoid | Flavonoid | Flavonoid | [42] |
| 2- coumarate | Broth | Flavonoid | Flavonoid | Flavonoid | [42] |
| Isoquinoline | Broth | Alkaloid | Plant derivatives | Neuroprotective | [39,40,41] |
| Harmalol | Broth | Alkaloid | Plant derivatives | Antioxidant, anticancer | [44] |
| Harmaline | Broth | Alkaloid | Plant derivatives | Antioxidant, anticancer | [44] |
| Harmine | Broth | Alkaloid | Plant derivatives | Antioxidant, anticancer | [45] |
| Leptosidin | Broth | Flavonoid | Plant derivatives | Antioxidant, anticancer | [46] |
| Anatabine | Broth | Alkaloid | Plant derivatives | Neuroprotective | [47] |
| Cathinone | Broth | Alkaloid | Plant-mushroom derivatives | Neuroprotective | [48] |
| Columbianetin | Broth phenolic | furanocoumarin | Plant | Antifungal | [49] |
| Indole-3-acetic acid | Broth/ Mycelium | Indole compound | Endophytic, mycorrhizal fungi | Plant growth and development | [32,50] |
| Oxadixyl | Broth/Mycelium | Oxazolidinone Compound | Fungi | Antifungal | [51] |
| Methyl 2,3-dihydro-3-hydroxy-2-oxo-1H-indole-3-acetate | Mycelium | Indole compound | Unreported from fungi | Bioactive compound, unspecified | [52] |
| 1-Methyl-3,4-dihydro-6,7-isoquinolinediol | Mycelium | Isoquinoline | Plant derivatives | Neuroprotective | [39,40,41] |
| Clavamycin E | Mycelium | Clavams group of compounds | Fungi | Antimicrobial | [53] |
| Questiomycin A | Mycelium | Phenazine derivative | Penicillium expansum | Antimicrobial | [54] |
| N-Acetyltyramine | Mycelium Phenolic |
Tyramine alkaloid | Fungi (Aspergillus) | Antimicrobial, antioxidant, anticancer | [55,56] |
| Gentioflavine | Mycelium |
Alkaloid |
Fungi (Aspergillus endophytes) | Flavonoid | [57,58,59] |
| Indirubin | Mycelium | Indole compound | Wrightia tinctoria, Schizophyllum commune, Malassezia spp. | Anticancer | [60] |
| Dictyoquinazol A | Mycelium | Quinazolines compound | Dicyophora indusiata | Neuroprotective | [61,62] |
| Zingerol | Mycelium Phenolic |
Phenolic compound | Plant derivatives | Antimicrobial | [63] |
| Asarone | Mycelium | Phenylpropanoid | Aspergillus sp. | Antimicrobial | [64] |
| trans-3-Indoleacrylic acid | Mycelium | Indole compound | Lactiplantibacillus plantarum LPP95 | Antifungal | [65] |
| Gelsemicine | Mycelium | Indole compound | Gelsemium spp., Endophytic fungi | Neuroprotective alkaloid | [66] |
| 3-(1H-indol-3-yl)quinoxalin-2-ol | Mycelium | Indole compound | Fungi | Antimicrobial, antioxidant, anticancer | [31] |
| Indole-3-lactic acid | Mycelium | Indole compound | Ectomycorrhizal fungi | Plant growth and development | [67,68] |
| Indole-3-acetaldehyde | Mycelium | Indole compound | Ectomycorrhizal fungi | Plant growth and development | [67] |
| Indole-3-carboxylic acid | Mycelium | Indole compound | Lasiodiploda pseudotheobromae | Antifungal | [69] |
| 3-(2-Hydroxyethyl)indole | Mycelium | Indole compound | Sporormiella minimoides | Antiviral, Neuroprotective, Anti-HIV-1 | [70] |
| (S)-beta-Methylindolepyruvate | Mycelium | Indole compound | Unreported from fungi | Antimicrobial, antioxidant, anticancer | [24,71] |
| 4-Indolecarbaldehyde acid | Mycelium | Indole compound | Unreported from fungi | Bioactive compound, unspecified | [37,38,39] |
| 2-(acetylamino)-3-(1H-indol-3-yl)propanoic acid | Mycelium | Indole compound | Unreported from fungi | Bioactive compound, unspecified | |
| 5-Hydroxyindole-3-acetic acid | Mycelium | Indole compound | Endophytic, mycorrhizal fungi | Plant growth and development | [51] |
| Methyl indole-3-acetate | Mycelium | Indole compound | Endophytic, mycorrhizal fungi | Plant growth and development | [51] |
| 3-(1H-Imidazol-4-ylmethyl)-6-(1H-indol-3-ylmethyl)-2,5-piperazinedione | Mycelium | Indole compound | Unreported from fungi | Bioactive compound, unspecified | |
| Maculosin | Mycelium | dipeptide, pyrrolopyrazine | Bacterial derived | Antioxidant | [72] |
| 4-Methoxycinnamaldehyde | Mycelium | Phenolic compound | Plant derivatives | Antimicrobial | [73] |
| (Z)-cinnamaldehyde | Mycelium | Flavonoid | Plant derivatives | Antimicrobial | [73] |
| 4-Methylesculetin | Mycelium Phenolic |
Phenolic compound | Plant derivatives | Antimicrobial, antioxidant, anticancer | [74] |
| 7-Hydroxy-5-methoxyflavan | Mycelium | Flavonoid | Flavonoid | Antimicrobial, antioxidant, anticancer | [39,67] |
| lentiginosine | Mycelium | Alkaloid | Plant derivatives | Antifungal | [75] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




