Submitted:
18 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. Cell Starvation and Activation of Jurkat Cells
2.4. Drug Treatment Conditions
2.5. Cell Viability Assay
2.6. Assessment of Cell Death Mechanisms
2.7. Protein Extraction and Western Blotting
2.8. Statistical Analysis
3. Results
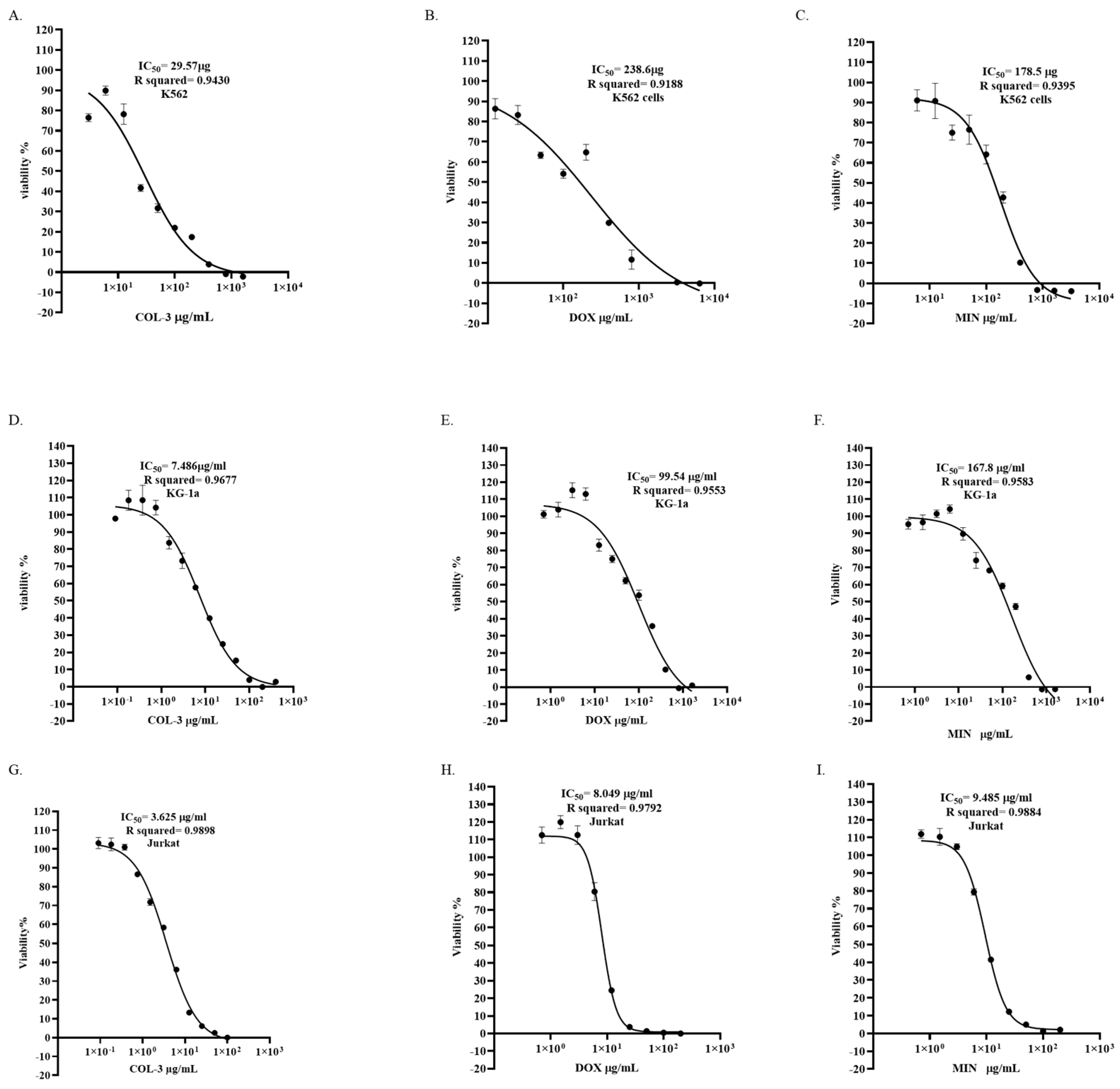
3.1. Cytotoxic Effects of Tetracycline Analogues in Leukemia Cell Lines
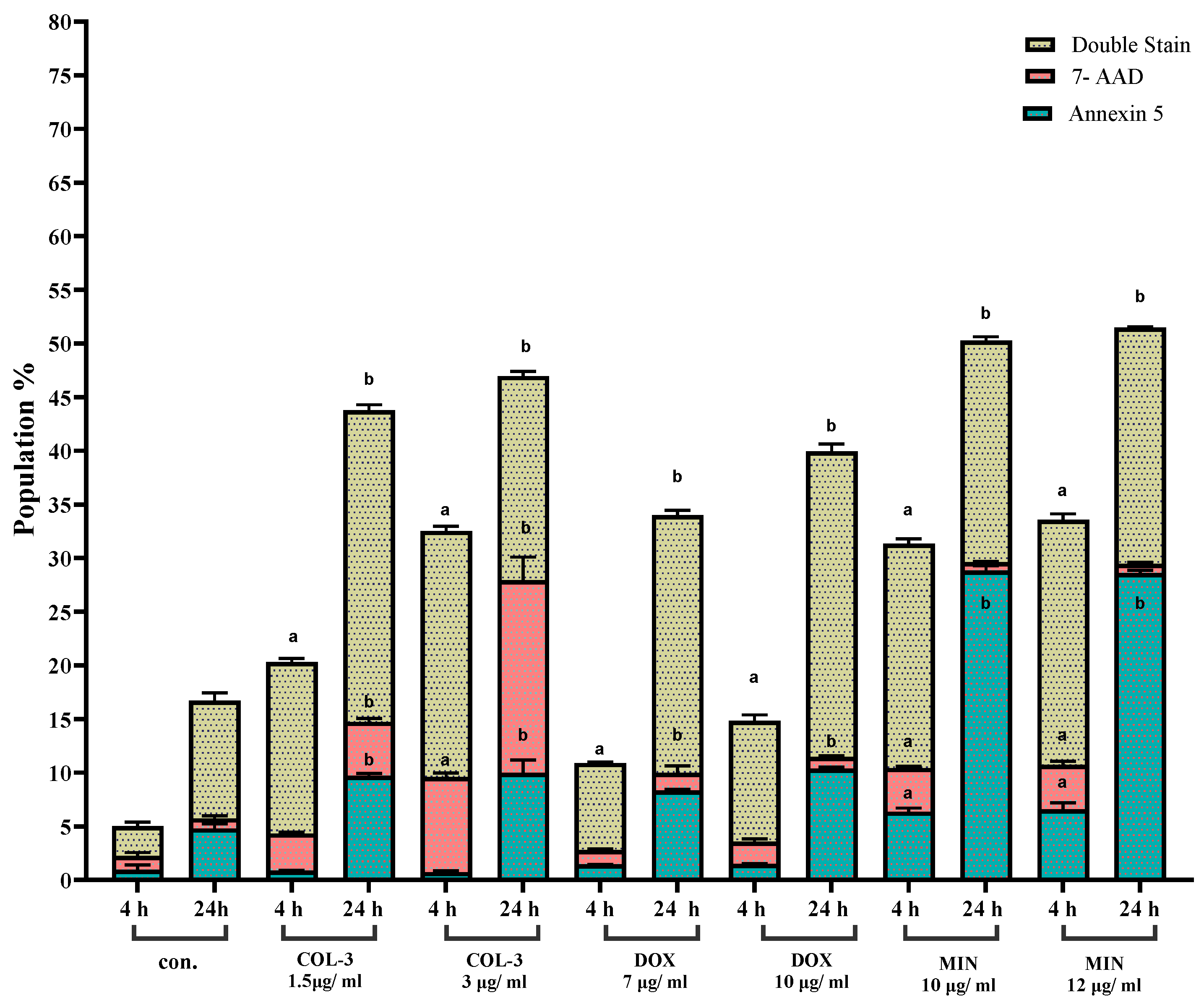
3.2. Tetracycline Analogues Induce Apoptosis with Cell Line-Specific Features
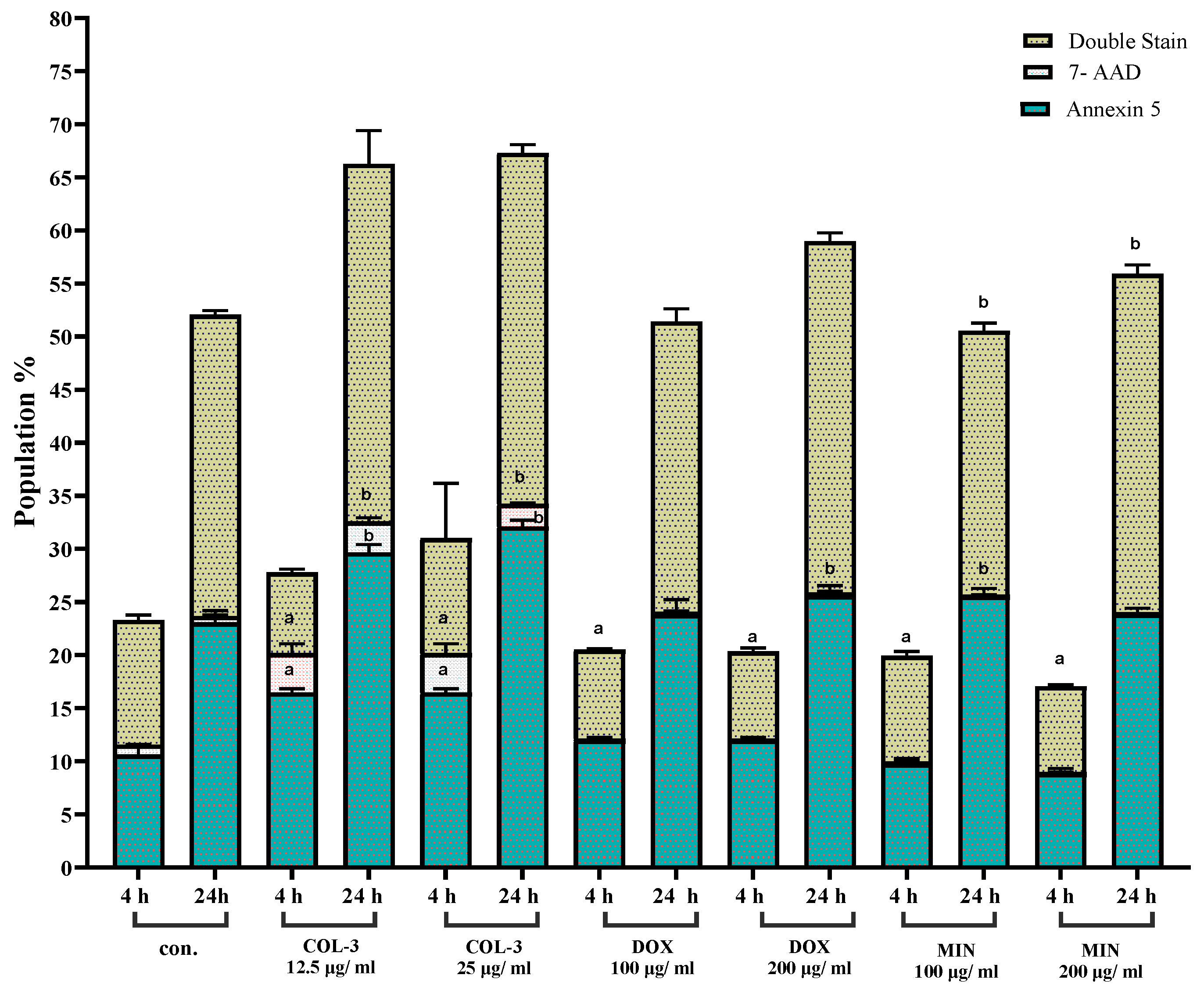
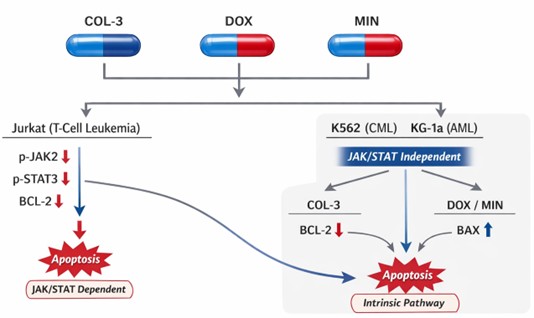
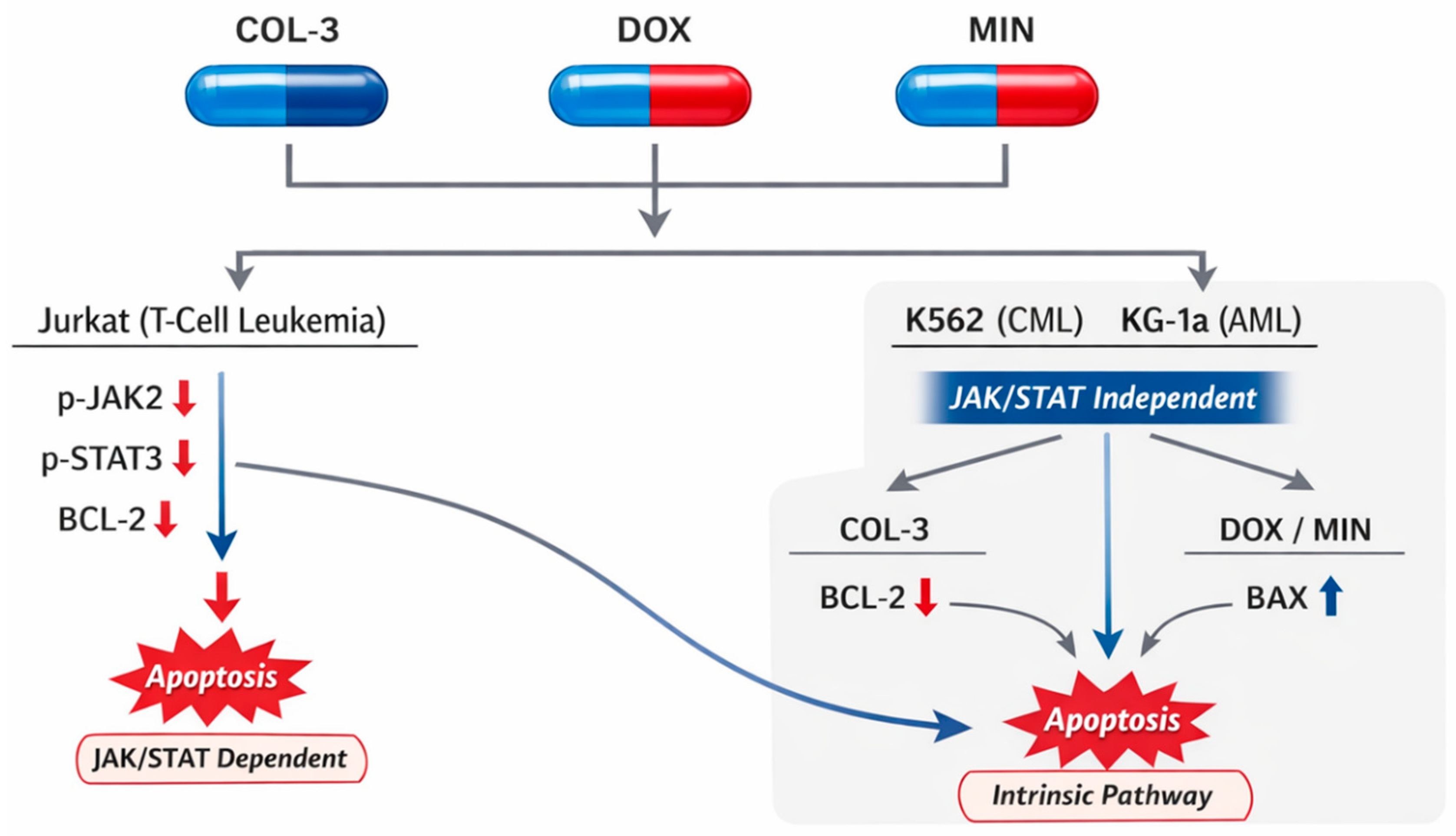
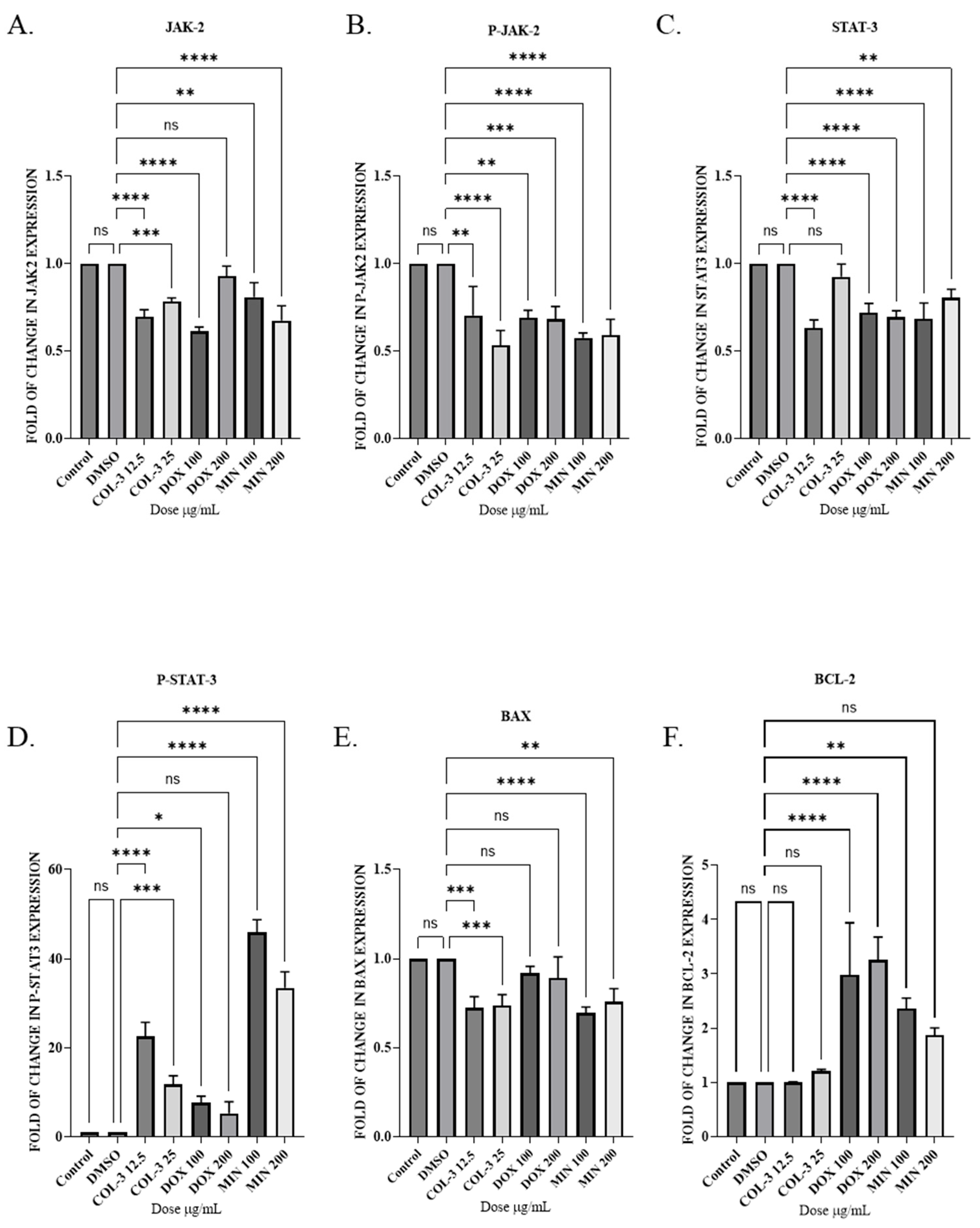
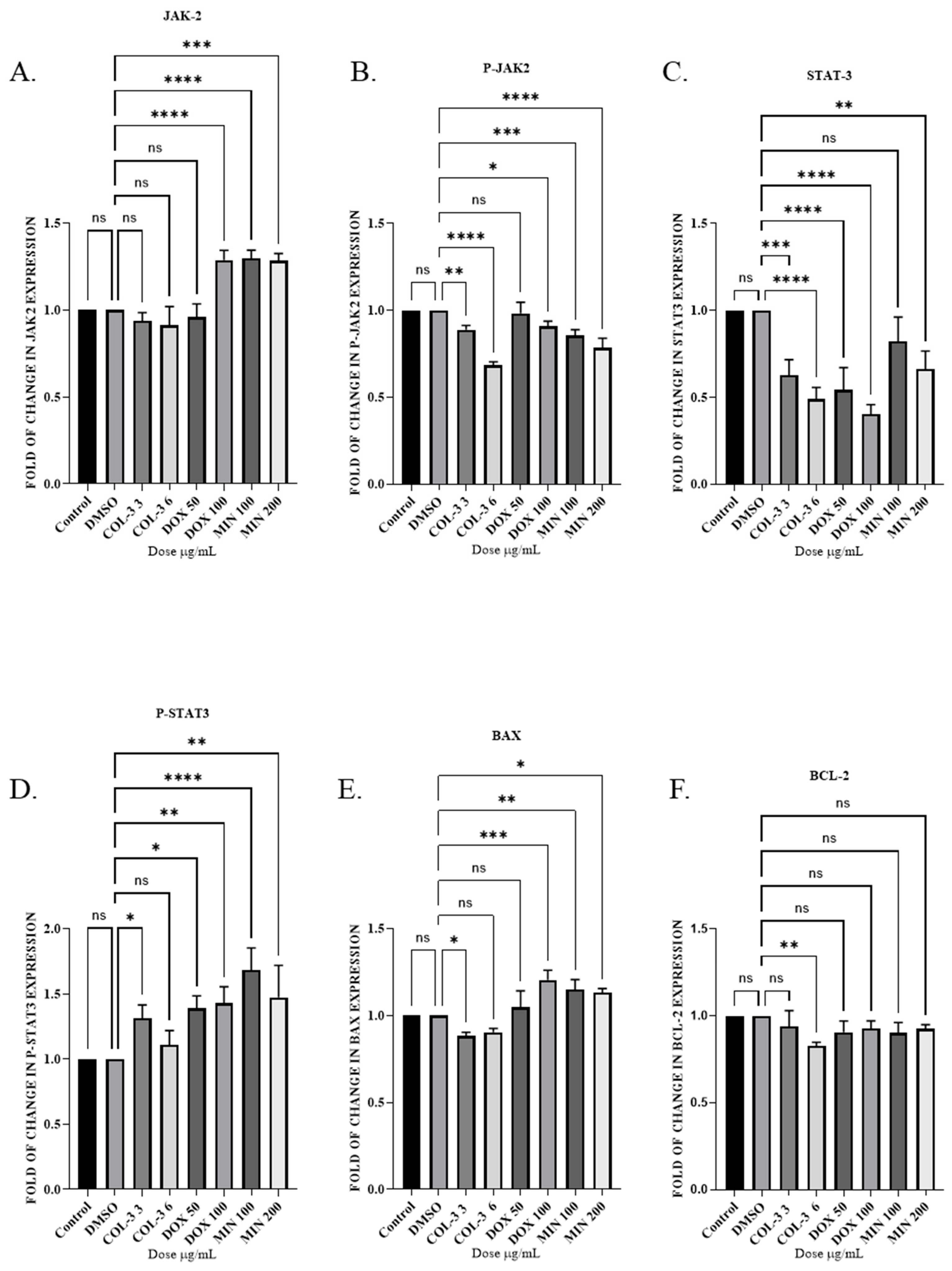
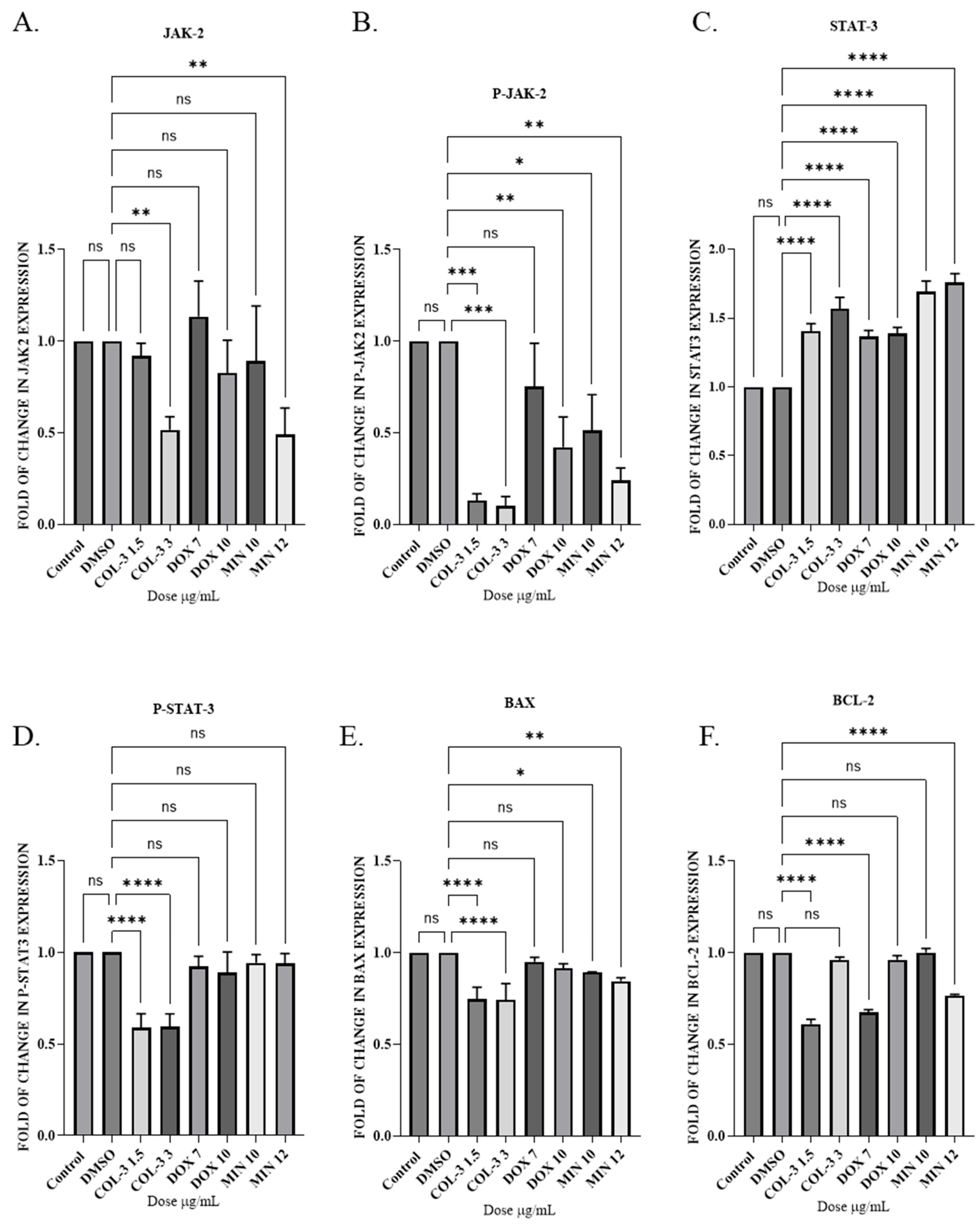
4. Effects on the JAK2/STAT3 Pathway and BCL-2 Family Proteins
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| COL-3 | tetracycline-3 |
| DOX | doxycycline |
| MIN | minocycline |
References
- Chukhlovin, A.B. Drug Repurposing in Leukemia Treatment and Hematopoietic Stem Cell Transplantation. CTT 2019, 8, 12–19. [CrossRef]
- Langedijk, J.; Mantel-Teeuwisse, A.K.; Slijkerman, D.S.; Schutjens, M.-H.D.B. Drug Repositioning and Repurposing: Terminology and Definitions in Literature. Drug Discovery Today 2015, 20, 1027–1034. [CrossRef]
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; Doig, A.; Guilliams, T.; Latimer, J.; McNamee, C.; et al. Drug Repurposing: Progress, Challenges and Recommendations. Nat Rev Drug Discov 2019, 18, 41–58. [CrossRef]
- Damery, E.; Solimando, D.A.; Waddell, J.A. Arsenic Trioxide and Tretinoin (AsO/ATRA) for Acute Promyelocytic Leukemia (APL). Hosp Pharm 2016, 51, 628–632. [CrossRef]
- Eisenstein, E.L.; Hill, K.D.; Wood, N.; Kirchner, J.L.; Anstrom, K.J.; Granger, C.B.; Rao, S.V.; Baldwin, H.S.; Jacobs, J.P.; Jacobs, M.L.; et al. Evaluating Registry-Based Trial Economics: Results from the STRESS Clinical Trial. Contemporary Clinical Trials Communications 2024, 38, 101257. [CrossRef]
- Nehme, R.; Hallal, R.; El Dor, M.; Kobeissy, F.; Gouilleux, F.; Mazurier, F.; Zibara, K. Repurposing of Acriflavine to Target Chronic Myeloid Leukemia Treatment. CMC 2021, 28, 2218–2233. [CrossRef]
- Pelcovits, A.; Niroula, R. Acute Myeloid Leukemia: A Review. R I Med J (2013) 2020, 103, 38–40.
- SEER data base; National Cancer Institute Acute Myeloid Leukemia—Cancer Stat Facts Available online: https://seer.cancer.gov/statfacts/html/amyl.html (accessed on 5 July 2024).
- Möricke, A.; Reiter, A.; Zimmermann, M.; Gadner, H.; Stanulla, M.; Dördelmann, M.; Löning, L.; Beier, R.; Ludwig, W.-D.; Ratei, R.; et al. Risk-Adjusted Therapy of Acute Lymphoblastic Leukemia Can Decrease Treatment Burden and Improve Survival: Treatment Results of 2169 Unselected Pediatric and Adolescent Patients Enrolled in the Trial ALL-BFM 95. Blood 2008, 111, 4477–4489. [CrossRef]
- Puumala, S.E.; Ross, J.A.; Aplenc, R.; Spector, L.G. Epidemiology of Childhood Acute Myeloid Leukemia. Pediatric Blood & Cancer 2013, 60, 728–733. [CrossRef]
- Seibel, N.L. Treatment of Acute Lymphoblastic Leukemia in Children and Adolescents: Peaks and Pitfalls. Hematology 2008, 2008, 374–380. [CrossRef]
- Cortes, J.E.; Gambacorti-Passerini, C.; Deininger, M.W.; Mauro, M.J.; Chuah, C.; Kim, D.-W.; Dyagil, I.; Glushko, N.; Milojkovic, D.; Le Coutre, P.; et al. Bosutinib Versus Imatinib for Newly Diagnosed Chronic Myeloid Leukemia: Results From the Randomized BFORE Trial. JCO 2018, 36, 231–237. [CrossRef]
- Copland, M.; Slade, D.; Byrne, J.; Brock, K.; De Lavallade, H.; Craddock, C.; Clark, R.; Smith, M.; Bishop, R.; Milojkovic, D.; et al. FLAG-IDA and Ponatinib in Patients with Blast Phase Chronic Myeloid Leukaemia: Results from the Phase I/II UK Trials Acceleration Programme Matchpoint Trial. Blood 2019, 134, 497–497. [CrossRef]
- Jabbour, E.J.; Faderl, S.; Kantarjian, H.M. Adult Acute Lymphoblastic Leukemia. Mayo Clinic Proceedings 2005, 80, 1517–1527. [CrossRef]
- Martinelli, G.; Boissel, N.; Chevallier, P.; Ottmann, O.; Gökbuget, N.; Topp, M.S.; Fielding, A.K.; Rambaldi, A.; Ritchie, E.K.; Papayannidis, C.; et al. Complete Hematologic and Molecular Response in Adult Patients With Relapsed/Refractory Philadelphia Chromosome–Positive B-Precursor Acute Lymphoblastic Leukemia Following Treatment With Blinatumomab: Results From a Phase II, Single-Arm, Multicenter Study. JCO 2017, 35, 1795–1802. [CrossRef]
- Molina, J.C.; Carraway, H.E. Treatment of Relapsed Acute Lymphocytic Leukemia in Adult Patients. Curr. Treat. Options in Oncol. 2024, 25, 993–1010. [CrossRef]
- Van Weelderen, R.E.; Klein, K.; Harrison, C.J.; Jiang, Y.; Abrahamsson, J.; Arad-Cohen, N.; Bart-Delabesse, E.; Buldini, B.; De Moerloose, B.; Dworzak, M.N.; et al. Measurable Residual Disease and Fusion Partner Independently Predict Survival and Relapse Risk in Childhood KMT2A -Rearranged Acute Myeloid Leukemia: A Study by the International Berlin-Frankfurt-Münster Study Group. JCO 2023, 41, 2963–2974. [CrossRef]
- Liang, D.; Wang, Q.; Zhang, W.; Tang, H.; Song, C.; Yan, Z.; Liang, Y.; Wang, H. JAK/STAT in Leukemia: A Clinical Update. Mol Cancer 2024, 23, 25. [CrossRef]
- Lin, M.; Nebral, K.; Gertzen, C.G.W.; Ganmore, I.; Haas, O.A.; Bhatia, S.; Fischer, U.; Kuhlen, M.; Gohlke, H.; Izraeli, S.; et al. JAK2 p.G571S in B-Cell Precursor Acute Lymphoblastic Leukemia: A Synergizing Germline Susceptibility. Leukemia 2019, 33, 2331–2335. [CrossRef]
- Liu, K.; Wu, Z.; Chu, J.; Yang, L.; Wang, N. Promoter Methylation and Expression of SOCS3 Affect the Clinical Outcome of Pediatric Acute Lymphoblastic Leukemia by JAK/STAT Pathway. Biomedicine & Pharmacotherapy 2019, 115, 108913. [CrossRef]
- Chhipa, R.; Singh, S.; Surve, S.; Vijayakumar, M.; Bhat, M. Doxycycline Potentiates Antitumor Effect of Cyclophosphamide in Mice. Toxicology and Applied Pharmacology 2005, 202, 268–277. [CrossRef]
- Sagar, J.; Sales, K.; Dijk, S.; Taanman, J.; Seifalian, A.; Winslet, M. Does Doxycycline Work in Synergy with Cisplatin and Oxaliplatin in Colorectal Cancer? World J Surg Onc 2009, 7, 2. [CrossRef]
- Dijk, S.N.; Protasoni, M.; Elpidorou, M.; Kroon, A.M.; Taanman, J.-W. Mitochondria as Target to Inhibit Proliferation and Induce Apoptosis of Cancer Cells: The Effects of Doxycycline and Gemcitabine. Sci Rep 2020, 10, 4363. [CrossRef]
- Zhu, C.; Yan, X.; Yu, A.; Wang, Y. Doxycycline Synergizes with Doxorubicin to Inhibit the Proliferation of Castration-Resistant Prostate Cancer Cells. ABBS 2017, 49, 999–1007. [CrossRef]
- Li, J.; Qin, Y.; Zhao, C.; Zhang, Z.; Zhou, Z. Tetracycline Antibiotics: Potential Anticancer Drugs. European Journal of Pharmacology 2023, 956, 175949. [CrossRef]
- Song, H.; Fares, M.; Maguire, K.R.; Sidén, Å.; Potácová, Z. Cytotoxic Effects of Tetracycline Analogues (Doxycycline, Minocycline and COL-3) in Acute Myeloid Leukemia HL-60 Cells. PLoS ONE 2014, 9, e114457. [CrossRef]
- Fares, M.; Abedi-Valugerdi, M.; Hassan, M.; Potácová, Z. DNA Damage, Lysosomal Degradation and Bcl-xL Deamidation in Doxycycline- and Minocycline-Induced Cell Death in the K562 Leukemic Cell Line. Biochemical and Biophysical Research Communications 2015, 463, 268–274. [CrossRef]
- Onoda, T.; Ono, T.; Dhar, D.K.; Yamanoi, A.; Nagasue, N. Tetracycline Analogues (Doxycycline and COL-3) Induce Caspase-dependent and -independent Apoptosis in Human Colon Cancer Cells. Intl Journal of Cancer 2006, 118, 1309–1315. [CrossRef]
- Ruiz-Moreno, C.; Velez-Pardo, C.; Jimenez-Del-Rio, M. Minocycline Induces Apoptosis in Acute Lymphoblastic Leukemia Jurkat Cells. Toxicology in Vitro 2018, 50, 336–346. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



