Submitted:
18 February 2026
Posted:
19 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
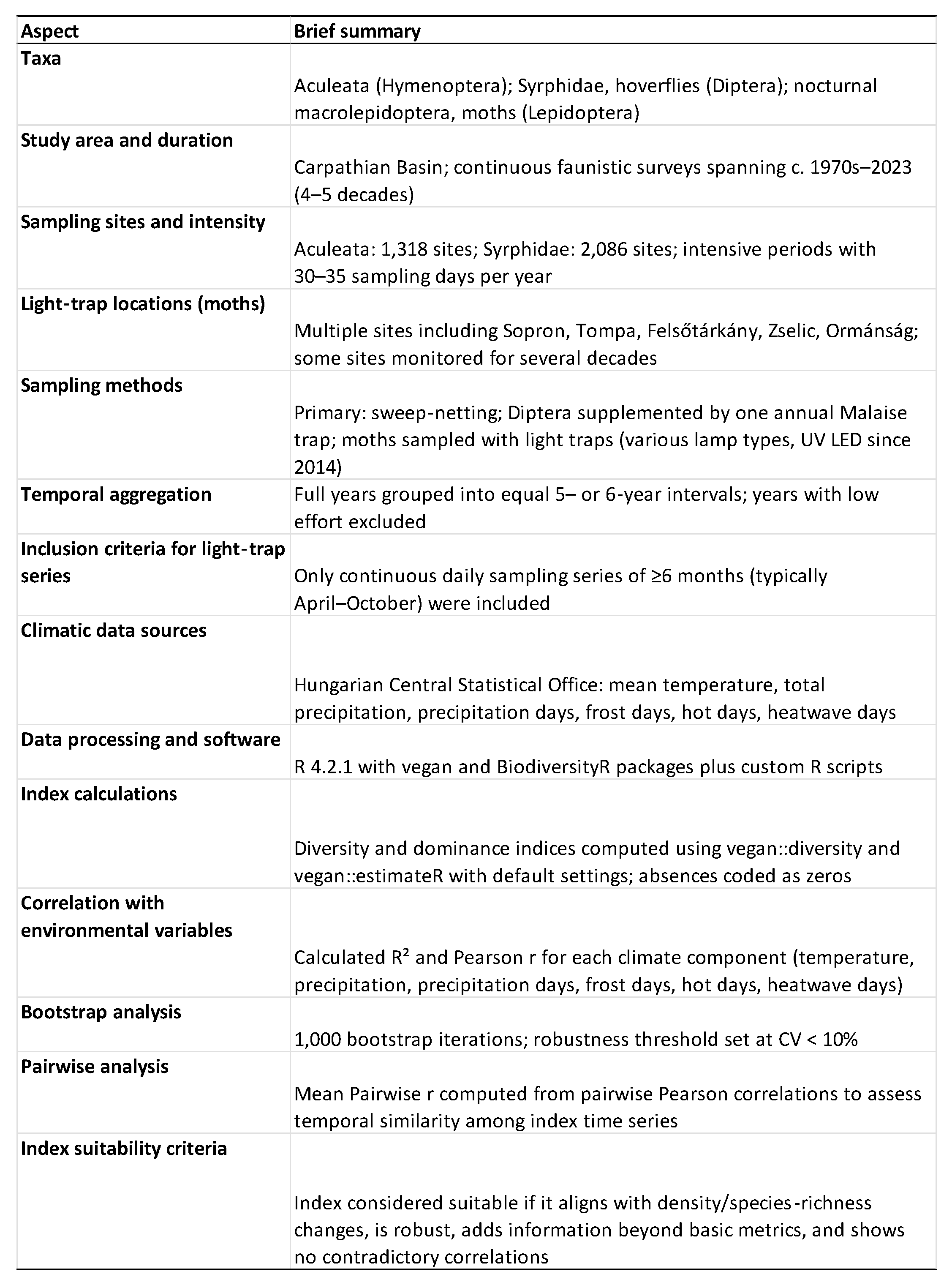
2. Materials and Methods
2.1. Hymenoptera, Diptera
2.2. Nocturnal Macrolepidoptera
2.3. Statistical Considerations in Ecological and Meteorological Data Collection
2.3.1. Data Preparation and Processing
2.3.2. Index Calculation
2.3.3. Correlation with Environmental Variables
2.3.4. Robustness and Pairwise Analysis
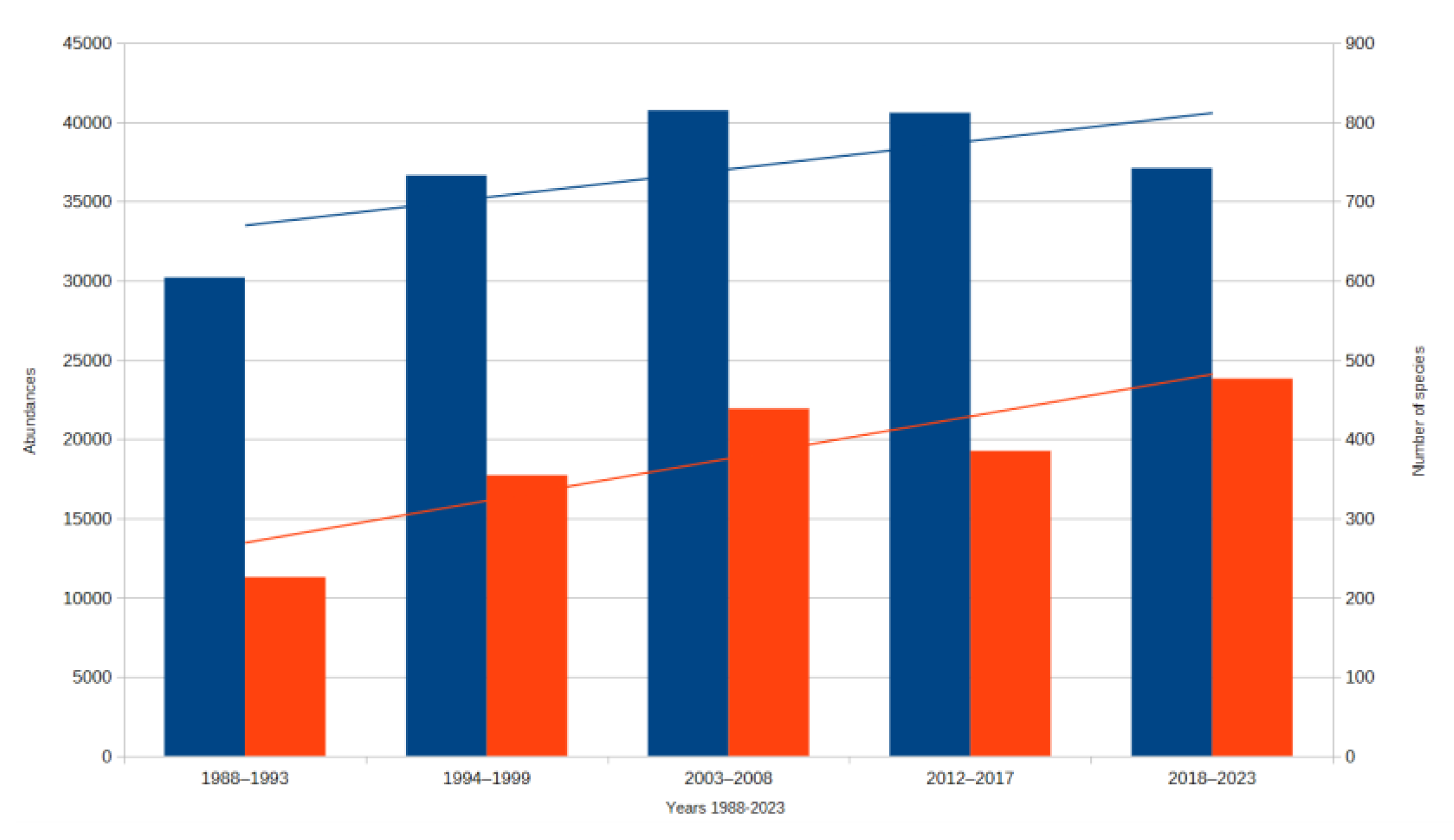
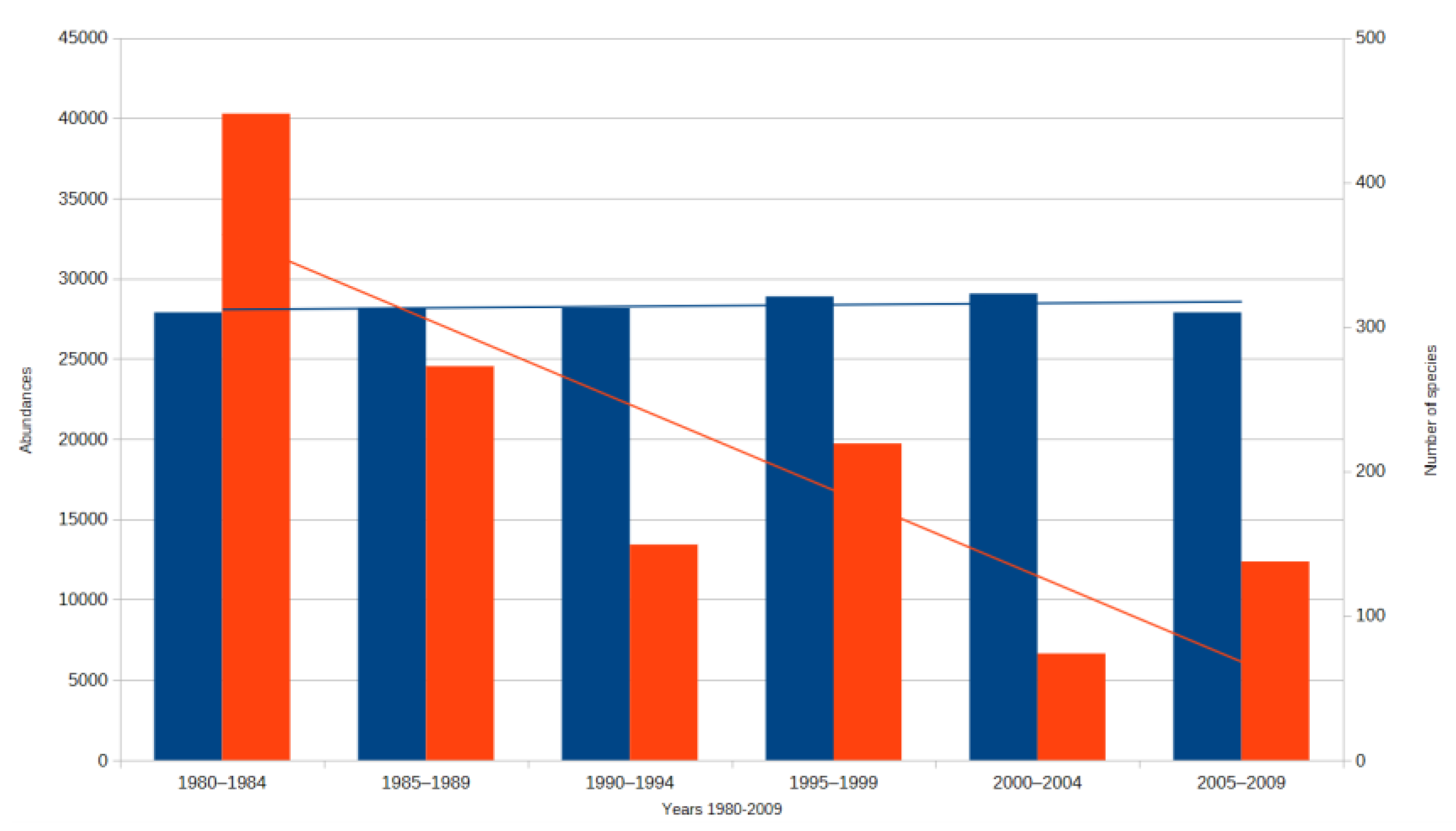
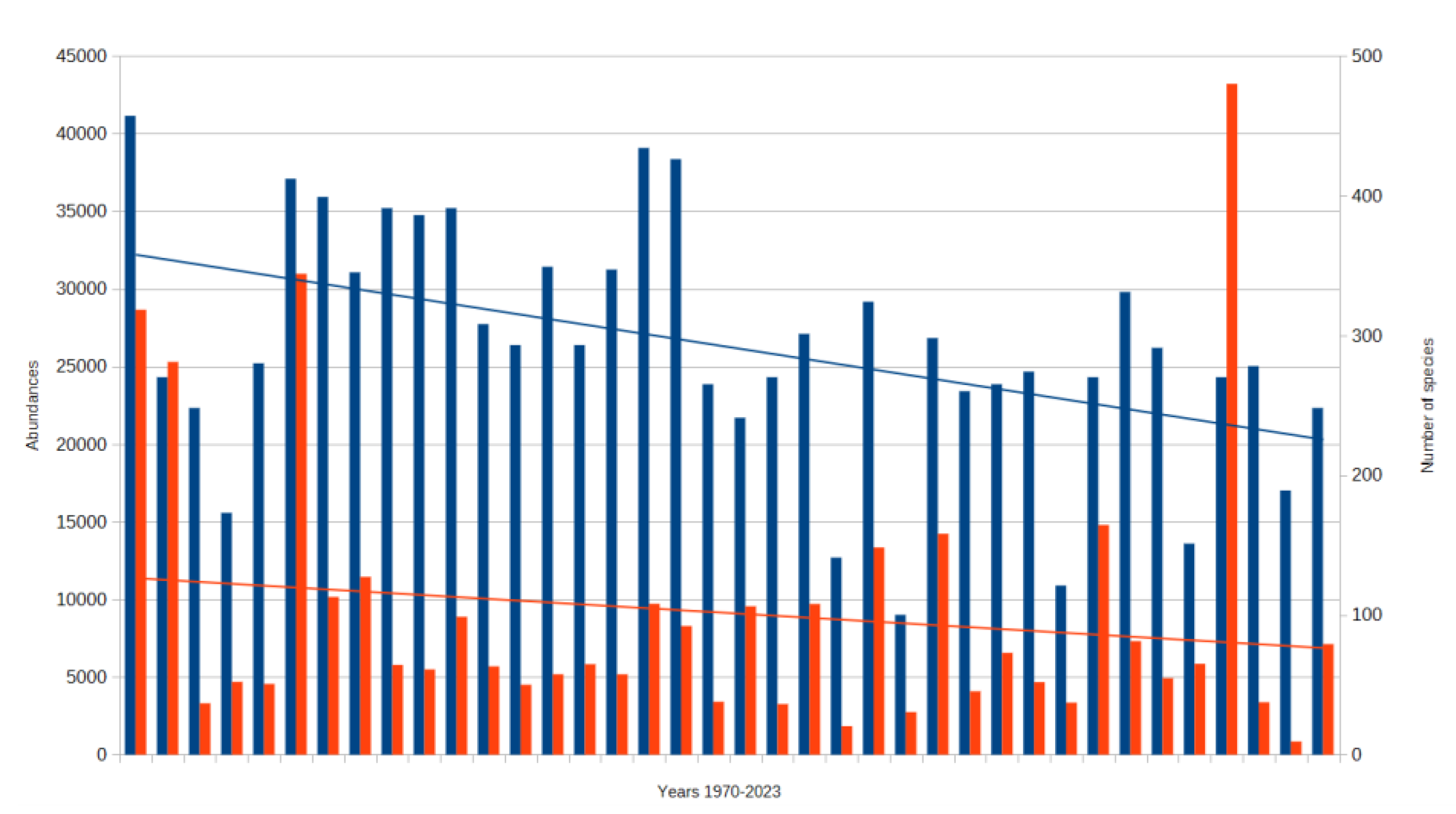
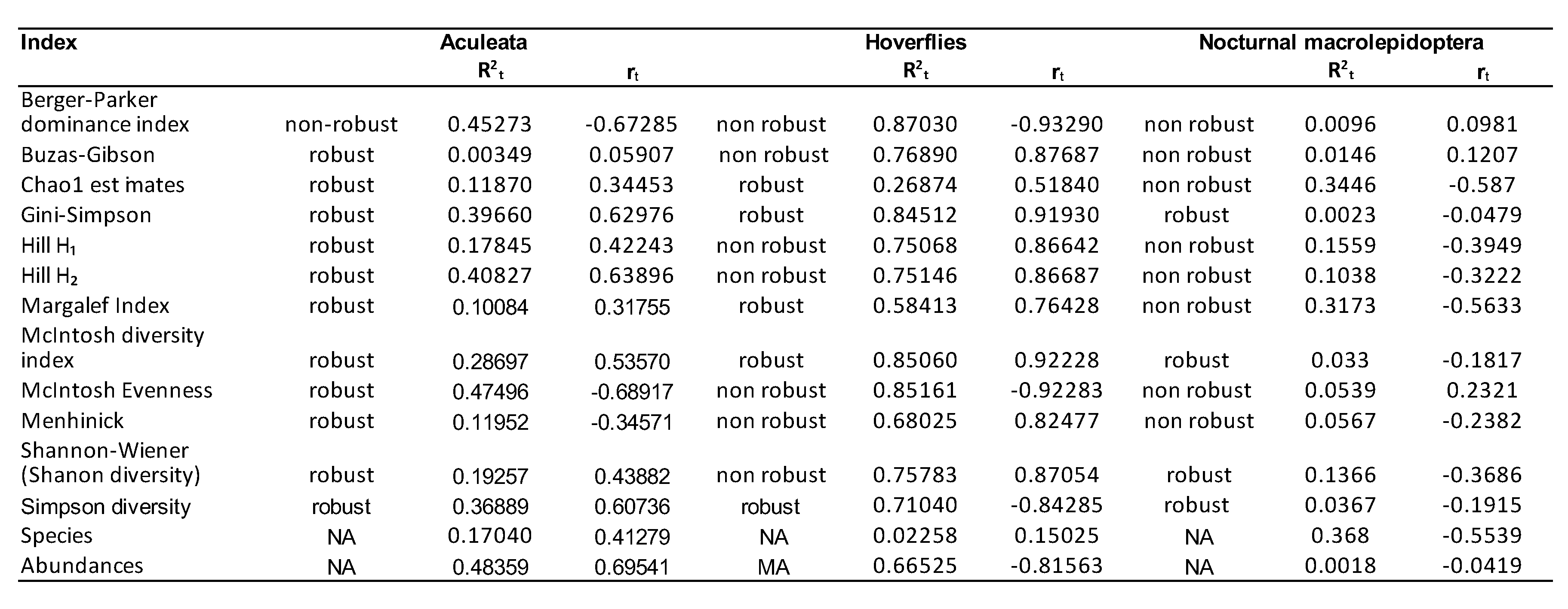
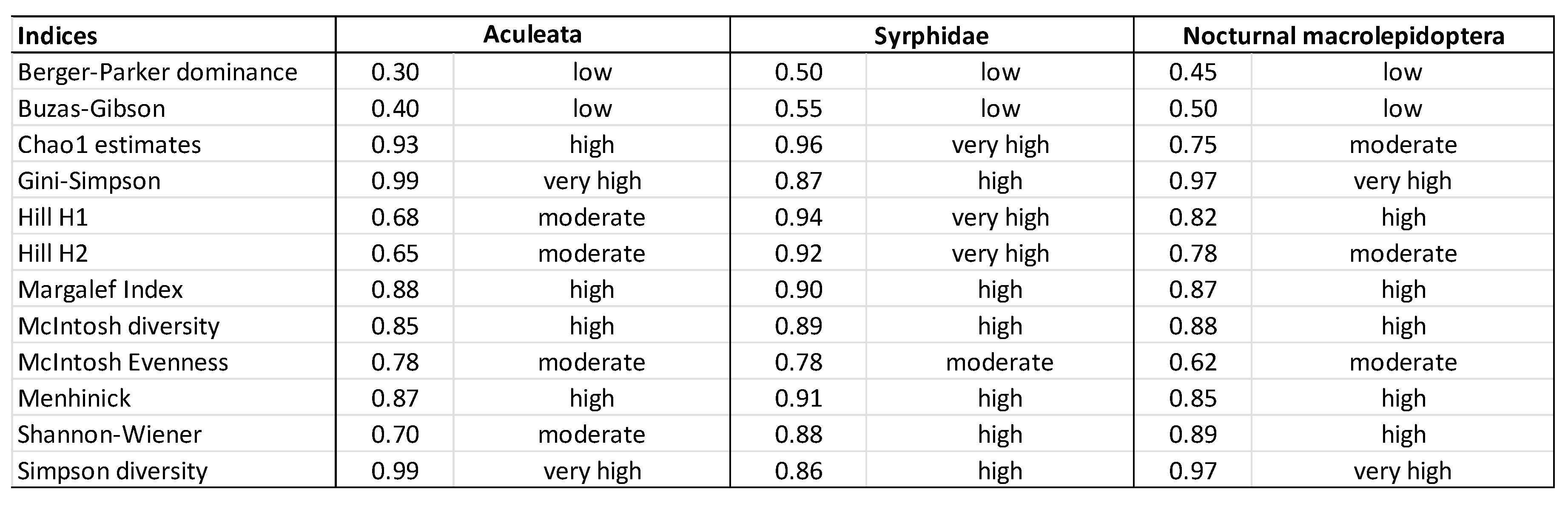
3. Results
3.1. Assessment of Three Types of Responses to Climate Change
3.1.1. Assesment of the Ecological Response of Thermophilic Groups for Climate Change
3.1.2. Assessment of the Ecological Response of Hygrophilous Groups for Climate Change
3.1.3. Assessment of the Ecological Response of Groups Exhibiting Low Sensitivity to Climate Change
3.2. Recommended Indices
3.2.1. Gini–Simpson Diversity
3.2.2. Simpson Diversity
3.2.3. McIntosh Diversity
3.2.4. McIntosh Evenness
3.3. Secondary Indices, Suitable for Specific Ecological Responses
3.3.1. Chao1 Species Richness Estimator
3.3.2. Margalef Index
3.3.3. Shannon–Wiener Diversity
3.3.4. Menhinick Index
3.4. Less Suitable Indicators in Tracking the Effects of Climate Change
3.4.1. Berger–Parker Dominance Index
3.4.2. Buzas–Gibson Index
3.4.3. Hill H1 (Shannon Equivalent)
3.4.4. Hill H2 (Inverse Simpson)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shannon, C. E. A Mathematical Theory of Communication. Bell System Tech. J. 1948, 27(3) 27(4), 379–423 623–656. [Google Scholar] [CrossRef]
- Simpson, E. H. Measurement of Diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Berger, W. H.; Parker, F. L. Diversity of planktonic foraminifera in deep-sea sediments. Science 1970, 168, 1345–1347. [Google Scholar] [CrossRef] [PubMed]
- Buzas, M. A.; Gibson, T. G. Species diversity: benthonic foraminifera in western North Atlantic. Science 1969, 163, 72–75. [Google Scholar] [CrossRef]
- Hill, M. O. Diversity and evenness: a unifying notation and its consequences. Ecology 1973, 54, 427–432. [Google Scholar] [CrossRef]
- Margalef, R. Information Theory in Ecology. General Systems 1958, 3, 36–71. [Google Scholar]
- McIntosh, R. P. An index of diversity and the concept of difference. Ecology 1967, 48, 392–404. [Google Scholar] [CrossRef]
- Chao, A. Nonparametric Estimation of the Number of Classes in a Population. Scandinavian Journal of Statistics 1984, 11(4), 265–270. [Google Scholar]
- Kitikidou, K.; Milios, E.; Stampoulidis, A.; Pipinis, E.; Radoglou, K. Using Biodiversity Indices Effectively: Considerations for Forest Management. Ecologies 2024, 5, 42–51. [Google Scholar] [CrossRef]
- Bashir, N. H., Meng, L.. Naeem, M., Chen, H. Biodiversity Assessment of Syrphid Flies (Diptera: Syrphidae) Within China. Diversity 2025, 17, 471. [CrossRef]
- Garcia, N.; Campos, J. C.; Alírio, J.; et al. Assessing spatial and temporal trends over time in potential species richness using satellite time-series and ecological niche models. Biodivers Conserv. 2024, 34, 429–446. [Google Scholar] [CrossRef]
- Willis, A. D.; Martin, B. D. Estimating Diversity in Networked Ecological Communities. Biostatistics 2020, 21, 1–17. [Google Scholar] [CrossRef]
- Roswell, M.; Dushoff, J.; Winfree, R. A Conceptual Guide to Measuring Species Diversity. Oikos 2021, 130, 321–338. [Google Scholar] [CrossRef]
- Nagendra, H. Opposite Trends in Response for the Shannon and Simpson Indices of Landscape Diversity. Appl. Geogr. 2002, 22(2), 175–186. [Google Scholar] [CrossRef]
- Morris, E.K.; Caruso, T.; Buscot, F.; Fischer, M.; Hancock, C.; Maier, T.S.; Meiners, T.; Müller, C.; Obermaier, E.; Prati, D.; Socher, S.A.; Sonnemann, I.; Wäschke, N.; Wubet, T.; Wurst, S.; Rillig, M.C. Choosing and using diversity indices: insights for ecological applications from the German Biodiversity Exploratories. Ecology and Evolution 2014, 4(18), 3514–3524. [Google Scholar] [CrossRef] [PubMed]
- Cosmulescu, S.; Stamin, F. D.; Răduțoiu, D.; Gheorghiu, N. C. Plant Diversity and Ecological Indices of Naturally Established Native Vegetation in Permanent Grassy Strips of Fruit Orchards in Southern Romania. Diversity 2025, 17, 494. [Google Scholar] [CrossRef]
- Stamin, F. D.; Cosmulescu, S. Assessing the Vegetation Diversity of Different Forest Ecosystems in Southern Romania Using Biodiversity Indices and Similarity Coefficients. Biology 2025, 14, 869. [Google Scholar] [CrossRef]
- Sánchez-Ochoa, D. J.; González, E. J.; Arizmendi, M. d. C.; Koleff, P.; Martell-Dubois, R.; Meave, J. A.; et al. Capturing Temporal Heterogeneity of Communities: A Temporal β-Diversity Based on Hill Numbers and Time Series Analysis. PLOS ONE 2025, 20(8), e0292574. [Google Scholar] [CrossRef]
- Buckland, S. T.; Yuan, Y.; Marcon, E. Measuring Temporal Trends in Biodiversity. In AStA Adv. Stat. Anal.; 2017. [Google Scholar] [CrossRef]
- Chung, H. I.; Choi, Y.; Biging, G. S.; Lee, W.-K.; Lee, D. K.; Chon, J.; Sung, H. C.; Lee, K.; Jeon, S.W. An Integrated Conceptual Approach for Biodiversity Risk Assessment: How Do Biodiversity Risk Patterns Respond to the Simultaneous Impacts of Climate Change and Urbanization? Land 2025, 14, 2374. [Google Scholar] [CrossRef]
- Guan, Y.; Liu, J.; Cui, W.; Chen, D.; Zhang, J.; Lu, H.; Maeda, E. E.; Zeng, Z.; Beck, H. E. Elevation Regulates the Response of Climate Heterogeneity to Climate Change. Geophys. Res. Lett. 2024, 51, e2024GL109483. [Google Scholar] [CrossRef]
- Twaróg, B. The Dynamics of Shannon Entropy in Analyzing Climate Variability for Modeling Temperature and Precipitation Uncertainty in Poland. Entropy 2025, 27, 398. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Fu, G. Impacts of Climate Change and Human Activities on Plant Species α-Diversity across the Tibetan Grasslands. Remote Sens. 2023, 15, 2947. [Google Scholar] [CrossRef]
- Supriatna, J. Biodiversity Indexes: Value and Evaluation Purposes. E3S Web of Conferences 2018, 48, 01001. [Google Scholar] [CrossRef]
- Gotelli, N. J.; Chao, A. Measuring and Estimating Species Richness, Species Diversity, and Biotic Similarity from Sampling Data. In Encyclopedia of Biodiversity, second edition; Levin, S.A., Ed.; Academic Press: New York, 2013; Volume 5, pp. 195–211. [Google Scholar]
- KSH (Central Statistical Office; Központi Statisztikai Hivatal. Magyarország és Budapest időjárásának adatai [Statisztikai tábla 15.1.1.37.]. KSH Statisztikai Adatgyűjtemény. Hungarian Central Statistical Office Meteorological data of Hungary and Budapest [Statistical Table 15.1.1.37]. KSH Statistical Database. n.d. Available online: https://www.ksh.hu/stadat_files/kor/hu/kor0037.html (accessed on 12. 12. 2025).
- Tóth, S. Egy természetrajzos muzeológus visszatekintése (Retrospective of a Natural History Museologist). A Veszprém Megyei Múzeumok Közleményei 1994, 19-20, 41–61. [Google Scholar]
- Józan, Z. A Zselic darázsfaunájának (Hymenoptera, Aculeata) állatföldrajzi és ökofaunisztikai vizsgálata (Zoologeographic and ecofaunistic study of the Aculeata fauna (Hymenoptera, Aculeata) of Zselic). Somogyi Múzeumok Közleményei 1992, 9, 279–292. [Google Scholar]
- Józan, Z. A Mecsek kaparódarázs faunájának (Hymenoptera, Sphecoidea) faunisztikai, állatföldrajzi és ökofunisztikai vizsgálata (Faunistical, zoogeographical and ecofaunistical investigation on the Sphecoids fauna of the Mecsek Montains (Hymenoptera, Sphecoidea). Nat. Som. 2002, 3, 45–56. [Google Scholar] [CrossRef]
- Tóth, S. A Bakonyvidék zengőlégy faunája (Diptera: Syrphidae). A Bakony természettudományi. kutatásainak eredményei 2001, 25, 1–448. [Google Scholar]
- Tóth, S. A Mecsek zengőlégy faunája (Diptera: Syrphidae). Acta Naturalia Pannonica 2008, 3, 9–12. [Google Scholar]
- Tóth, S. Magyarország zengőlégy faunája (Diptera, Syrphidae). Hoverflies of Hungary (Diptera, Syrphidae). e-Acta Nat. Pan 2011, Suppl., 5–408. [Google Scholar]
- Schmidt, P.; Ábrahám, L.; Farkas, S. () Repeated macromoth faunistic survey in Zselic after 40 years (Lepidoptera: Macrolepidoptera). Nat. Som. 2023, 40, 99–118. [Google Scholar] [CrossRef]
- Uherkovich, Á. Long-term monitoring of biodiversity by the study of butterflies and larger moths (Lepidoptera) in Sellye region (South Hungary, co. Baranya) in the years 1967–2022. Nat. Som. 2022, 9, 95–138. [Google Scholar]
- Hirka, A.; Szabóky, Cs.; Szőcs, L.; Csóka, Gy. 50 éves az Erdészeti Fénycsapda Hálózat (50 Years of the Forestry Light Trap Network). Erdészeti Lapok 2011, 146(12), 378–380. [Google Scholar]
- Haris, A.; Józan, Z.; Roller, L.; Šima, P.; Tóth, S. Changes in Population Densities and Species Richness of Pollinators in the Carpathian Basin during the Last 50 Years (Hymenoptera, Diptera, Lepidoptera). Diversity 2024, 16(6), 328. [Google Scholar] [CrossRef]
- Haris, A.; Józan, Z.; Schmidt, P.; Glemba, G.; Tomozii, B.; Csóka, G.; Hirka, A.; Šima, P.; Tóth, S. Climate Change Influences on Central European Insect Fauna over the Last 50 Years: Mediterranean Influx and Non-Native Species. Ecologies 2025, 6(1), 16. [Google Scholar] [CrossRef]
- Valtonen, A.; Hirka, A.; Szőcs, L.; Ayres, M. P.; Roininen, H.; Csóka, Gy. Long-term species loss and homogenization of moth-communities in Central-Europe. J. Anim. Ecol. 2017, 86, 730–738. [Google Scholar] [CrossRef]
- Gillespie, M.A.K.; Ims, R.A.; Schmidt, N.M.; Bollandsås, O.M.; Jepsen, J.U.; Høye, T.T. Status and Trends of Terrestrial Arthropod Abundance and Diversity in the North Atlantic Region of the Arctic. Insects 2020, 11, 174. [Google Scholar] [CrossRef] [PubMed]
- Gebert, L.; Habel, J.C.; Schmitt, T.; Drees, C.; Ulrich, W. Similar Temporal Patterns in Insect Richness, Abundance and Biomass across Major Habitat Types. Insects 2024, 15, 345. [Google Scholar] [CrossRef]
- Gebreegziabher, H. A Systematic Review of Insect Decline and Discovery: Trends, Drivers, and Conservation Strategies over the past Two Decades. Insects 2024, 15, 896. [Google Scholar] [CrossRef]
- R Core Team A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna; Available online: https://www.R-project.org (accessed on 01. 12. 2025).
- Oksanen, J.; Blanchet, F. G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M. H. H.; Szoecs, E.; Wagner, H. R package version 2.5-2; () Vegan: Community Ecology Package. 2018.
- Kindt, R.; Coe, R. Tree diversity analysis. A manual and software for common statistical methods for ecological and biodiversity studies; World Agroforestry Centre (ICRAF): Nairobi, 2005; p. pp. 196. [Google Scholar]
- Galiana, N.; Arnoldi, J.-F.; Mestre, F.; Rozenfeld, A.; Araújo, M. B. Power laws in species’ biotic interaction networks can be inferred from co-occurrence data. Nat. Ecol. Evol. 2023, 8, 209–217. [Google Scholar] [CrossRef]
- Wong, M. K. L.; Tsang, T. P. N.; Lewis, O. T.; Guénard, B. Trait-similarity and trait-hierarchy jointly determine fine-scale spatial associations of resident and invasive ant species. Ecography 2021, 44, 1–13. [Google Scholar] [CrossRef]
- Sandhu, H. S.; Shi, P.; Kuang, X.; Xue, F.; Ge, F. Applications of the Bootstrap to Insect Physiology. Florida Entomol. 2011, 94, 1036–1041. [Google Scholar] [CrossRef]
- Engel, E.; Ribeiro, A. L. P.; Lúcio, A. D. C.; Pasini, M. P. B.; Buzzatti, J. Z.; Rodrigues, F. T.; Cassol, L. O.; Godoy, W. A. C. The Co-occurrence Matrix and the Correlation Network of Phytophagous Insects Are Driven by Abiotic and Biotic Variables: the Case of Canola. Neotrop. Entomol. 2024, 53, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Bassi, M. I. E.; Staude, I. R. Insects decline with host plants but coextinctions may be limited. Proc. Natl. Acad. Sci. U.S.A 2024, 121, e2417408121. [Google Scholar] [CrossRef]
- Barendregt, A.; Zeegers, T.; van Steenis, W.; Jongejans, E. Forest hoverfly community collapse: Abundance and species richness drop over four decades. Insect Conserv Divers. 2022, 15, 510–521. [Google Scholar] [CrossRef]
- Szabó, S.; Árnyas, E.; Varga, Z. Long-term light trap study on the macro-moth (Lepidoptera: Macroheterocera) fauna of the Aggtelek National Park. Acta zool. Acad. Sci. Hung 2007, 53(3), 257–269. [Google Scholar]
- Varga, J.; Korompai, T.; Horokán, K.; Hirka, A.; Gáspár, C.; Kozma, P.; Csóka, G.; Csuzdi, C. Analysis of the Macrolepidoptera fauna in Répáshuta based on the catches of a light-trap between 2014–2019. Acta Univ. Esterházy Sect. Biol. 2022, 47, 59–75. [Google Scholar]
- Szentkirályi, F.; Leskó, K.; Kádár, F. Hosszú távú rovarmonitorozás a várgesztesi erdészeti fénycsapdával. 2. A nagylepke együttes diverzitási mintázatának változásai. (Long-term insect monitoring with forestry light trap of Várgesztes. 2. Changes of pattern of species diversity of Macrolepidopteran assemblages). Erdészeti Kutatások 2002-2004, 91, 131–143. [Google Scholar]
- Szentkirályi, F.; Leskó, K.; Kádár, F. Climatic effects on long-term fluctuations in species richness and abundance level of forest macrolepidopteran assesmblages in a Hungarian mountainous region. Carpth. J. Earth Environ. Sci. 2007, 2, 73–82. [Google Scholar]
- Végvári, Z.; Juhász, E.; Tóth, J. P.; Barta, Z.; Boldogh, S.; Szabó, S.; Varga, Z. Life-history traits and climatic responsiveness in noctuid moths. Oikos 2015, 124, 235–242. [Google Scholar] [CrossRef]
- Zografou, K.; Kati, V.; Grill, A.; Wilson, R.J.; Tzirkalli, E.; et al. Signals of Climate Change in Butterfly Communities in a Mediterranean Protected Area. PloS ONE 2014, 9(1), e87245. [Google Scholar] [CrossRef] [PubMed]
- Moreno, C. E.; Halffter, G. Assessing the completeness of bat biodiversity inventories using species accumulation curves. J. Appl. Ecol. 2000, 37, 149–158. [Google Scholar] [CrossRef]
- Willott, S. J. Species accumulation curves and the measure of sampling. J. Appl. Ecol. 2001, 38, 484–486. [Google Scholar] [CrossRef]
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




