Submitted:
17 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
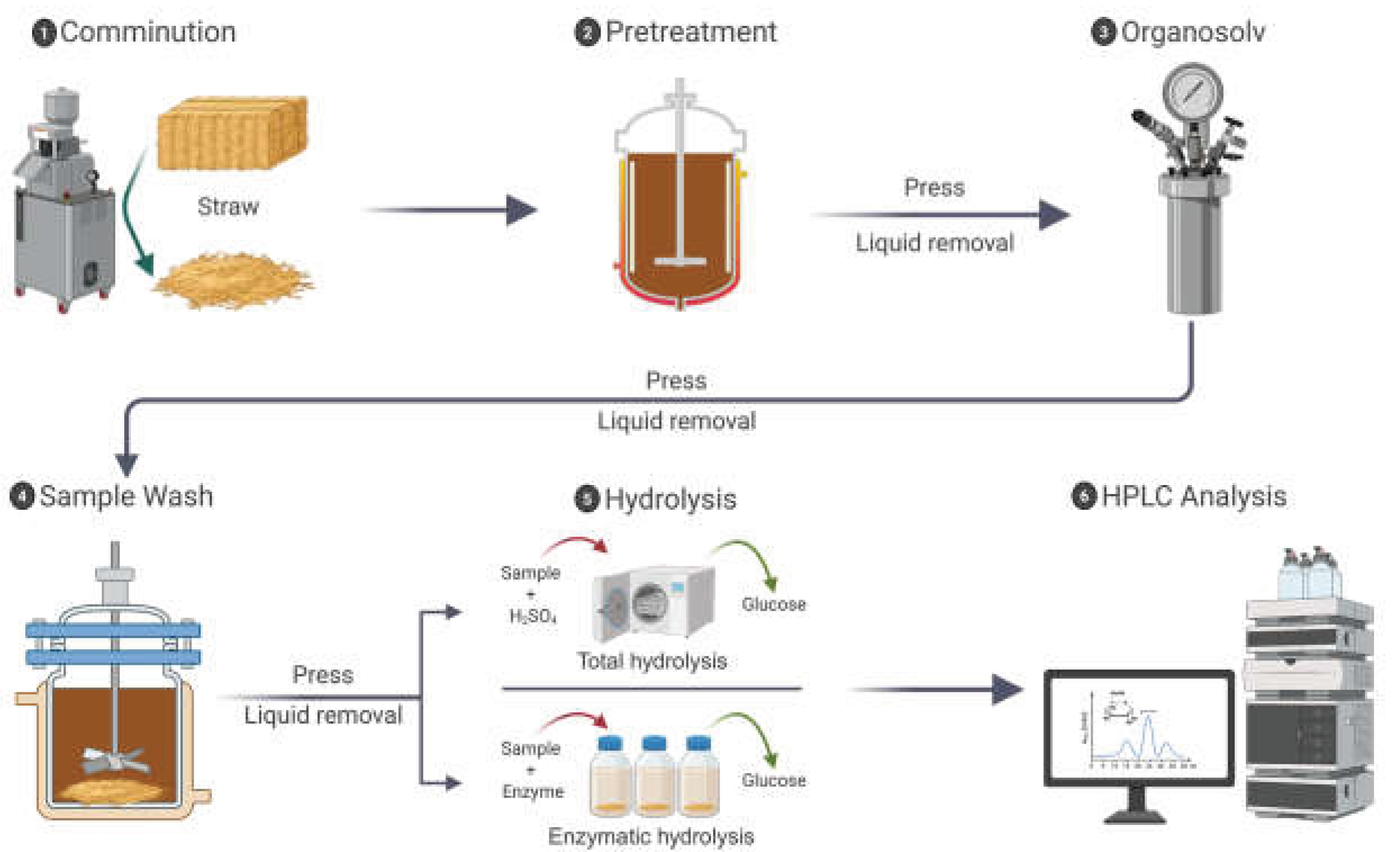
2.1. Experimental Workflow
2.2. Raw Material and Comminution
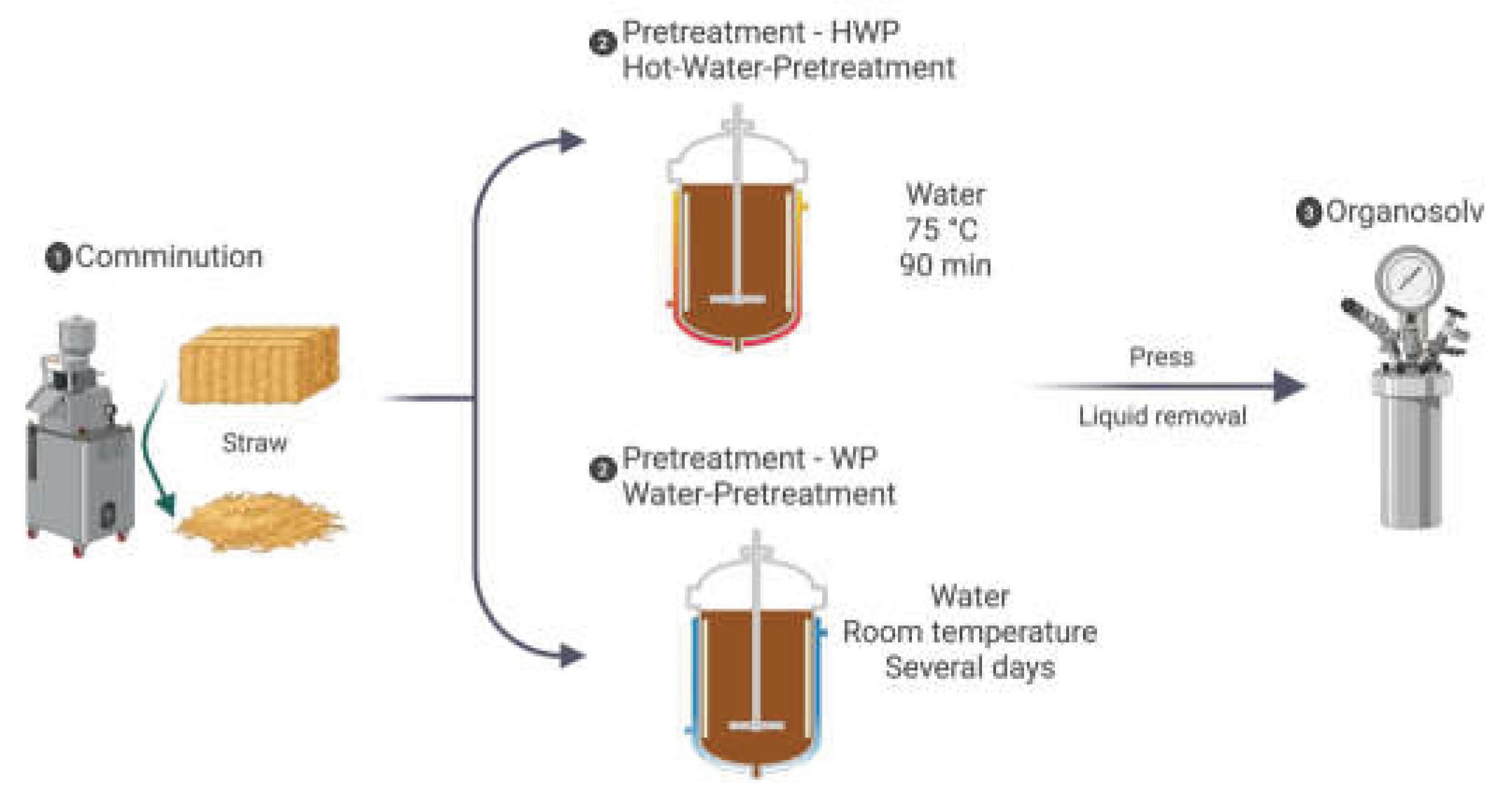
2.3. Pretreatment
2.4. Organosolv Extraction
2.5. Sample Wash
2.6. Hydrolysis
2.7. HPLC Analysis
2.8. Press and Liquid Removal
2.9. Measurement of Dry Matter Content
2.10. DoE and Simulation Model
3. Results and Discussion
3.1. Parameter Optimization for Organosolv Extraction
3.1.1. Evaluation of the Effect of Different Pretreatment Catalysts on Enzymatic Hydrolysis
3.1.2. Evaluation of the Effect of Liquor Ratios on Enzymatic Hydrolysis
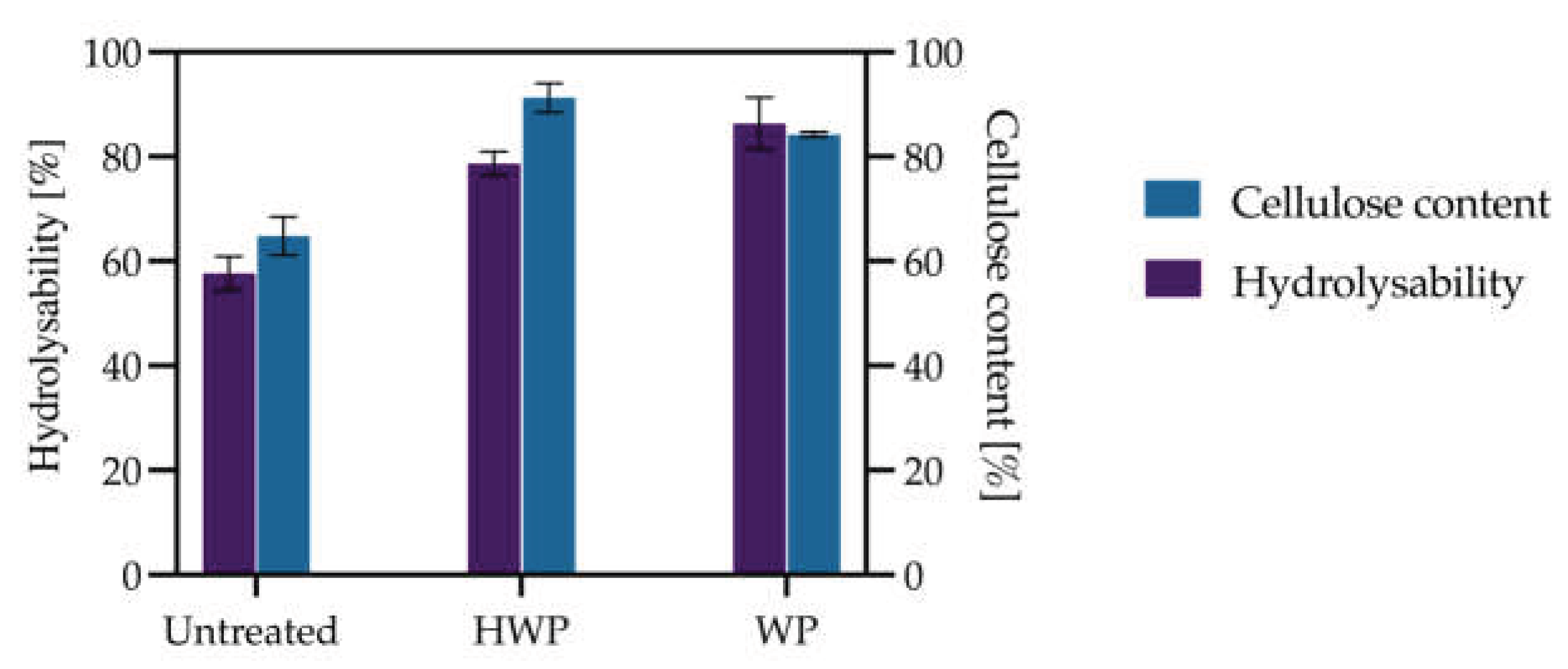
3.1.3. Evaluation of Pretreatment Impact on Organosolv Efficiency and Enzymatic Hydrolysis
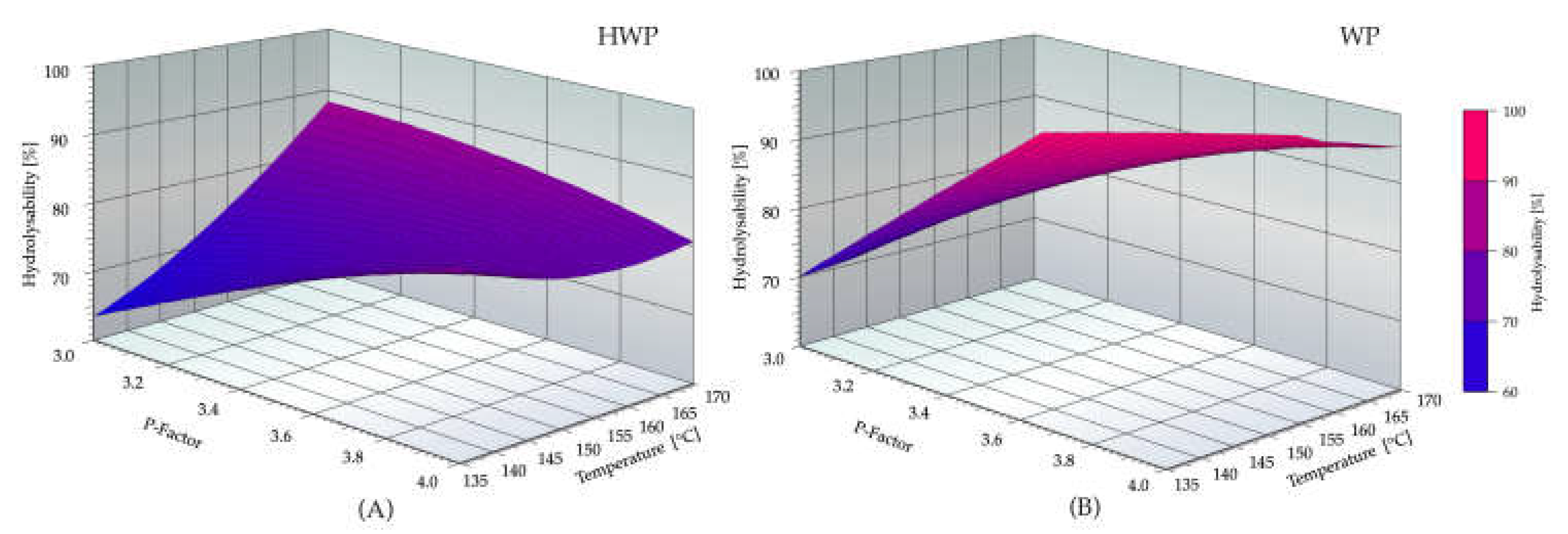
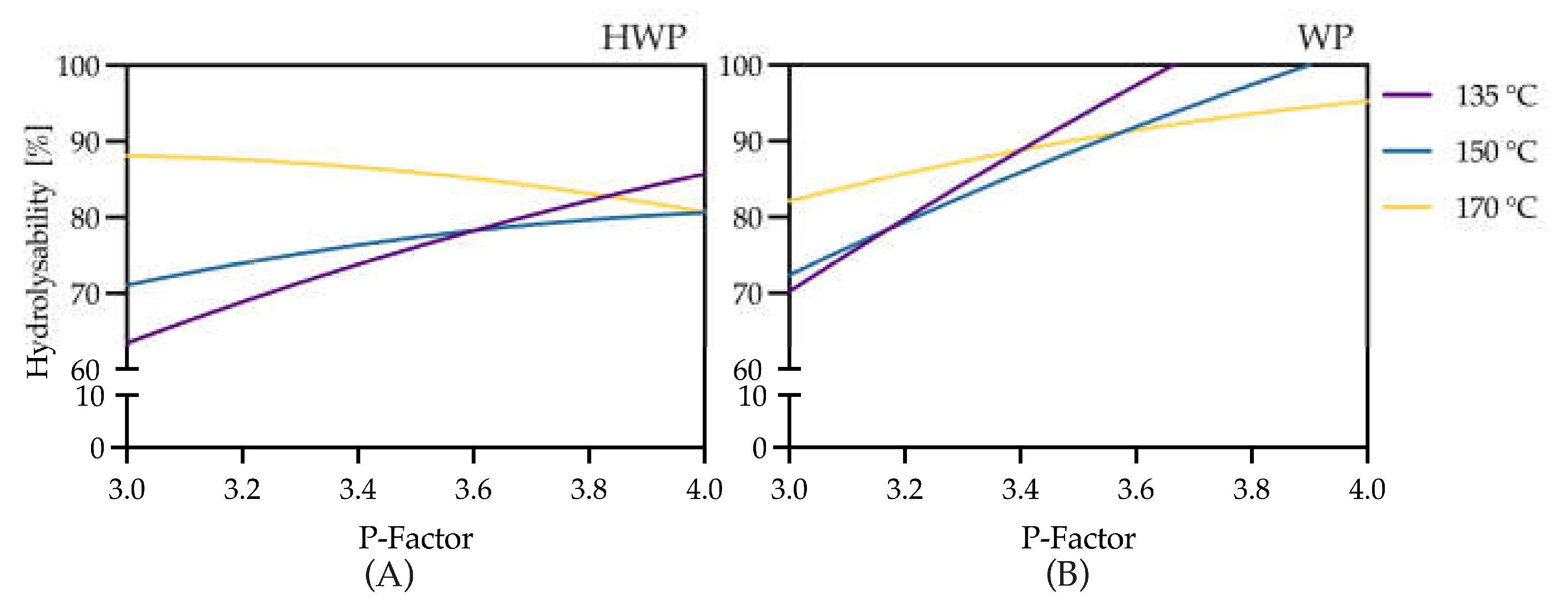
3.2. Evaluation of Organosolv Severity (Temperature and P-Factor) on Enzymatic Hydrolysis of Pretreated Straw
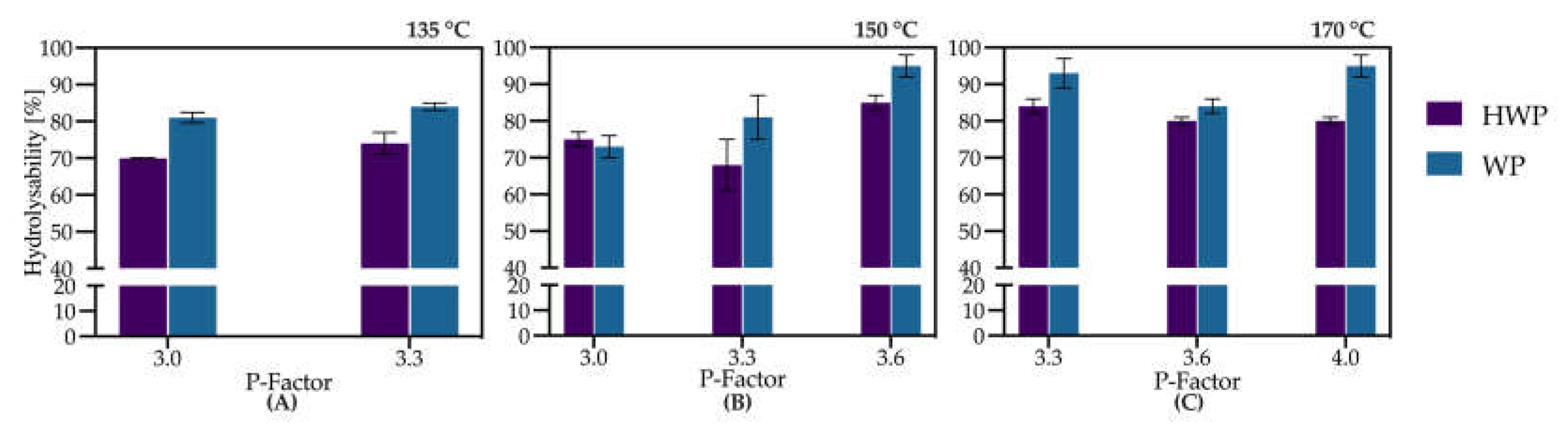
3.3. Model-Based Optimization of Organosolv Extraction Severity
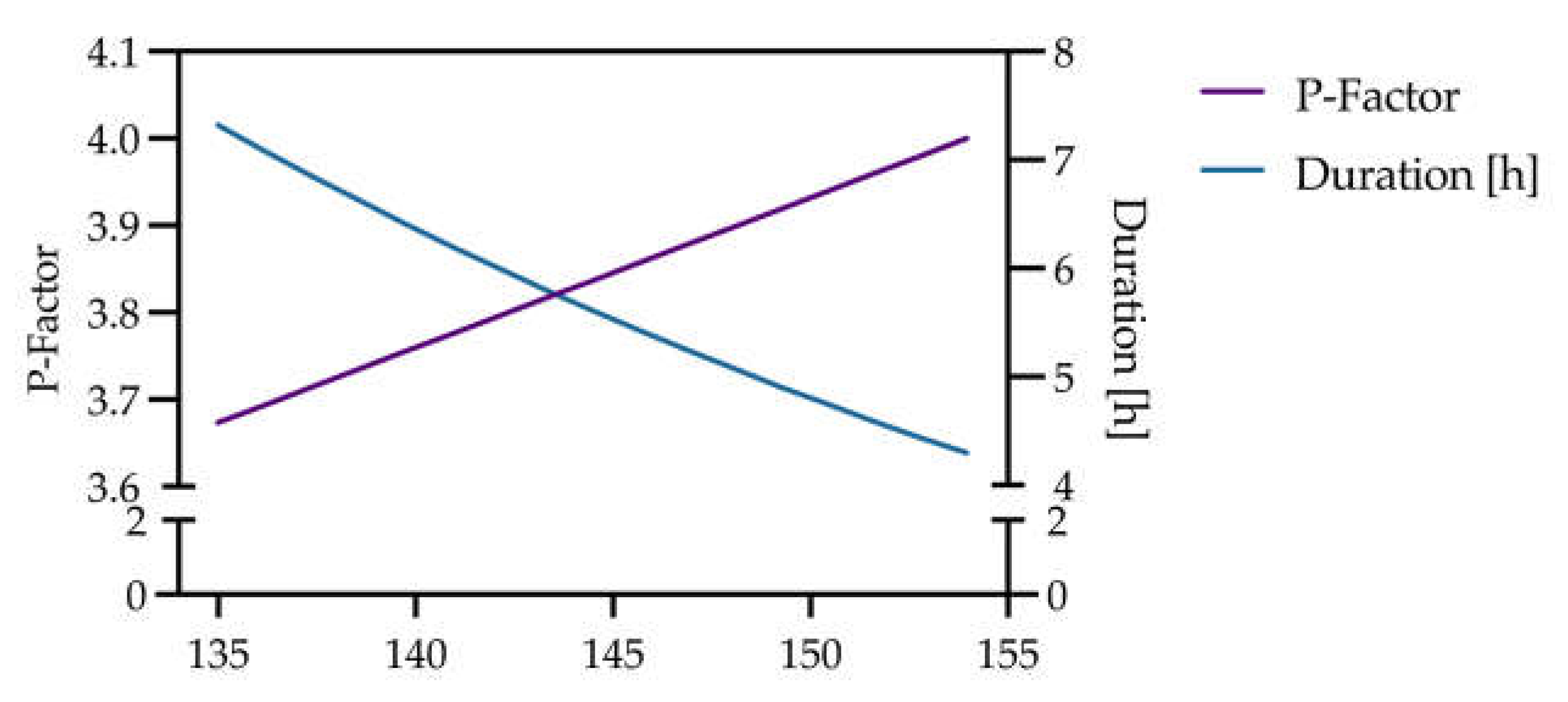
3.4. Trade-Off Between Extraction Temperature, P-Factor, and Residence Time
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baral, N.R.; Sundstrom, E.R.; Das, L.; Gladden, J.; Eudes, A.; Mortimer, J.C.; Singer, S.W.; Mukhopadhyay, A.; Scown, C.D. Approaches for More Efficient Biological Conversion of Lignocellulosic Feedstocks to Biofuels and Bioproducts. ACS Sustainable Chem. Eng. 2019, 7, 9062–9079. [Google Scholar] [CrossRef]
- Sharma, S.; Tsai, M.-L.; Sharma, V.; Sun, P.-P.; Nargotra, P.; Bajaj, B.K.; Chen, C.-W.; Dong, C.-D. Environment Friendly Pretreatment Approaches for the Bioconversion of Lignocellulosic Biomass into Biofuels and Value-Added Products. Environments 2022, 10, 6. [Google Scholar] [CrossRef]
- Tufail, T.; Saeed, F.; Afzaal, M.; Ain, H.B.U.; Gilani, S.A.; Hussain, M.; Anjum, F.M. Wheat Straw: A Natural Remedy against Different Maladies. Food Science & Nutrition 2021, 9, 2335–2344. [Google Scholar] [CrossRef]
- Ćilerdžić, J.; Galić, M.; Vukojević, J.; Brčeski, I.; Stajić, M. Potential of Selected Fungal Species to Degrade Wheat Straw, the Most Abundant Plant Raw Material in Europe. BMC Plant Biol 2017, 17, 249. [Google Scholar] [CrossRef]
- Kovačević, Z.; Bischof, S.; Bilandžija, N.; Krička, T. Conversion of Waste Agricultural Biomass from Straw into Useful Bioproducts—Wheat Fibers and Biofuels. Sustainability 2024, 16, 4739. [Google Scholar] [CrossRef]
- Reichel, R.; Wei, J.; Islam, M.S.; Schmid, C.; Wissel, H.; Schröder, P.; Schloter, M.; Brüggemann, N. Potential of Wheat Straw, Spruce Sawdust, and Lignin as High Organic Carbon Soil Amendments to Improve Agricultural Nitrogen Retention Capacity: An Incubation Study. Front. Plant Sci. 2018, 9, 900. [Google Scholar] [CrossRef] [PubMed]
- Carvalheiro, F.; Silva-Fernandes, T.; Duarte, L.C.; Gírio, F.M. Wheat Straw Autohydrolysis: Process Optimization and Products Characterization. Appl Biochem Biotechnol 2009, 153, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Yu, H.; Huang, Y. Structure and Morphology of Cellulose in Wheat Straw. Cellulose 2005, 12, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Hernández, C.; Escamilla-Alvarado, C.; Sánchez, A.; Alarcón, E.; Ziarelli, F.; Musule, R.; Valdez-Vazquez, I. Wheat Straw, Corn Stover, Sugarcane, and Agave Biomasses: Chemical Properties, Availability, and Cellulosic-bioethanol Production Potential in Mexico. Biofuels Bioprod Bioref 2019, 13, 1143–1159. [Google Scholar] [CrossRef]
- Lynd, L.R.; Weimer, P.J.; Van Zyl, W.H.; Pretorius, I.S. Microbial Cellulose Utilization: Fundamentals and Biotechnology. Microbiol Mol Biol Rev 2002, 66, 506–577. [Google Scholar] [CrossRef]
- Himmel, M.E.; Ding, S.-Y.; Johnson, D.K.; Adney, W.S.; Nimlos, M.R.; Brady, J.W.; Foust, T.D. Biomass Recalcitrance: Engineering Plants and Enzymes for Biofuels Production. Science 2007, 315, 804–807. [Google Scholar] [CrossRef]
- Ragauskas, A.J.; Williams, C.K.; Davison, B.H.; Britovsek, G.; Cairney, J.; Eckert, C.A.; Frederick, W.J.; Hallett, J.P.; Leak, D.J.; Liotta, C.L.; et al. The Path Forward for Biofuels and Biomaterials. Science 2006, 311, 484–489. [Google Scholar] [CrossRef]
- Deng, Z.; Xia, A.; Liao, Q.; Zhu, X.; Huang, Y.; Fu, Q. Laccase Pretreatment of Wheat Straw: Effects of the Physicochemical Characteristics and the Kinetics of Enzymatic Hydrolysis. Biotechnol Biofuels 2019, 12, 159. [Google Scholar] [CrossRef]
- Huang, C.; Jiang, X.; Shen, X.; Hu, J.; Tang, W.; Wu, X.; Ragauskas, A.; Jameel, H.; Meng, X.; Yong, Q. Lignin-Enzyme Interaction: A Roadblock for Efficient Enzymatic Hydrolysis of Lignocellulosics. Renewable and Sustainable Energy Reviews 2022, 154, 111822. [Google Scholar] [CrossRef]
- Paës, G.; Navarro, D.; Benoit, Y.; Blanquet, S.; Chabbert, B.; Chaussepied, B.; Coutinho, P.M.; Durand, S.; Grigoriev, I.V.; Haon, M.; et al. Tracking of Enzymatic Biomass Deconstruction by Fungal Secretomes Highlights Markers of Lignocellulose Recalcitrance. Biotechnol Biofuels 2019, 12, 76. [Google Scholar] [CrossRef] [PubMed]
- Galbe, M.; Wallberg, O. Pretreatment for Biorefineries: A Review of Common Methods for Efficient Utilisation of Lignocellulosic Materials. Biotechnol Biofuels 2019, 12, 294. [Google Scholar] [CrossRef]
- Prasad, A.; Sotenko, M.; Blenkinsopp, T.; Coles, S.R. Life Cycle Assessment of Lignocellulosic Biomass Pretreatment Methods in Biofuel Production. Int J Life Cycle Assess 2016, 21, 44–50. [Google Scholar] [CrossRef]
- Den, W.; Sharma, V.K.; Lee, M.; Nadadur, G.; Varma, R.S. Lignocellulosic Biomass Transformations via Greener Oxidative Pretreatment Processes: Access to Energy and Value-Added Chemicals. Front. Chem. 2018, 6, 141. [Google Scholar] [CrossRef]
- Huijgen, W.J.J.; Reith, J.H.; Den Uil, H. Pretreatment and Fractionation of Wheat Straw by an Acetone-Based Organosolv Process. Ind. Eng. Chem. Res. 2010, 49, 10132–10140. [Google Scholar] [CrossRef]
- Smit, A.; Huijgen, W. Effective Fractionation of Lignocellulose in Herbaceous Biomass and Hardwood Using a Mild Acetone Organosolv Process. Green Chem. 2017, 19, 5505–5514. [Google Scholar] [CrossRef]
- Constant, S.; Basset, C.; Dumas, C.; Di Renzo, F.; Robitzer, M.; Barakat, A.; Quignard, F. Reactive Organosolv Lignin Extraction from Wheat Straw: Influence of Lewis Acid Catalysts on Structural and Chemical Properties of Lignins. Industrial Crops and Products 2015, 65, 180–189. [Google Scholar] [CrossRef]
- Serna-Loaiza, S.; Adamcyk, J.; Beisl, S.; Miltner, M.; Friedl, A. Sequential Pretreatment of Wheat Straw: Liquid Hot Water Followed by Organosolv for the Production of Hemicellulosic Sugars, Lignin, and a Cellulose-Enriched Pulp. Waste Biomass Valor 2022, 13, 4771–4784. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Vicente, A.A.; Teixeira, J.A. Kinetic Modeling of Enzymatic Saccharification Using Wheat Straw Pretreated under Autohydrolysis and Organosolv Process. Industrial Crops and Products 2012, 36, 100–107. [Google Scholar] [CrossRef]
- Zhang, X.; Yu, G.; Feng, X.; Li, Z.; Li, B.; Cui, Q. Ammonia-Ethanol-Water Pretreatment of Wheat Straw for Facilitating Enzymatic Saccharification Integrated with the Preparation of Submicron Lignin Spheres. BioRes 2020, 15, 5087–5109. [Google Scholar] [CrossRef]
- Nair, L.G.; Agrawal, K.; Verma, P. Organosolv Pretreatment: An in-Depth Purview of Mechanics of the System. Bioresour. Bioprocess. 2023, 10, 50. [Google Scholar] [CrossRef]
- Overend, R.P. Fractionation of Lignocellulosics by Steam-Aqueous Pretreatments. Phil. Trans. R. Soc. Lond. A 1987, 321, 523–536. [Google Scholar] [CrossRef]
- Calhoun, J.M.; Yorston, F.H.; Maass, O. THE EFFECT OF SULPHITE LIQUOR COMPOSITION ON THE RATE OF DELIGNIFICATION OF SPRUCE WOOD AND YIELD OF PULP. Can. J. Res. 1937, 15b, 457–474. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Hedegaard Thomsen, M.; Trajano, H.L. (Eds.) Hydrothermal Processing in Biorefineries; Springer International Publishing: Cham, 2017; ISBN 978-3-319-56456-2. [Google Scholar]
- Montané, D.; Overend, R.P.; Chornet, E. Kinetic Models for Non-homogeneous Complex Systems with a Time-dependent Rate Constant. Can J Chem Eng 1998, 76, 58–68. [Google Scholar] [CrossRef]
- Vaidya, A.A.; Murton, K.D.; Smith, D.A.; Dedual, G. A Review on Organosolv Pretreatment of Softwood with a Focus on Enzymatic Hydrolysis of Cellulose. Biomass Conv. Bioref. 2022, 12, 5427–5442. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass. Laboratory analytical procedure 2008, 1617, 1–16. [Google Scholar]
- Guo, T.; Bode, J.; Kuka, K.; Tippkötter, N. Enhancing Single-Cell Protein Yield Through Grass-Based Substrates: A Study of Lolium Perenne and Kluyveromyces Marxianus. Fermentation 2025, 11, 266. [Google Scholar] [CrossRef]
- Franz, R.G. Comparisons of pKa and Log P Values of Some Carboxylic and Phosphonic Acids: Synthesis and Measurement. AAPS PharmSci 2001, 3, 1–13. [Google Scholar] [CrossRef]
- Zheng, J. IUPAC/Dissociation-Constants: V1.0 2022.
- Yasuda, S.; Murase, N. Chemical Structures of Sulfuric Acid Lignin. Part XII. Reaction of Lignin Models with Carbohydrates in 72% H2SO4. Holzforschung 1995, 49, 418–422. [Google Scholar] [CrossRef]
- Jasiukaitytė-Grojzdek, E.; Huš, M.; Grilc, M.; Likozar, B. Acid-Catalysed α-O-4 Aryl-Ether Bond Cleavage in Methanol/(Aqueous) Ethanol: Understanding Depolymerisation of a Lignin Model Compound during Organosolv Pretreatment. Sci Rep 2020, 10, 11037. [Google Scholar] [CrossRef]
- Kalogiannis, K.G.; Matsakas, L.; Aspden, J.; Lappas, A.A.; Rova, U.; Christakopoulos, P. Acid Assisted Organosolv Delignification of Beechwood and Pulp Conversion towards High Concentrated Cellulosic Ethanol via High Gravity Enzymatic Hydrolysis and Fermentation. Molecules 2018, 23, 1647. [Google Scholar] [CrossRef]
- Amarasekara, A.S.; Wiredu, B. A Comparison of Dilute Aqueous P-Toluenesulfonic and Sulfuric Acid Pretreatments and Saccharification of Corn Stover at Moderate Temperatures and Pressures. Bioresource Technology 2012, 125, 114–118. [Google Scholar] [CrossRef]
- Goldmann, W.M.; Ahola, J.; Mikola, M.; Tanskanen, J. Solubility and Fractionation of Indulin AT Kraft Lignin in Ethanol-Water Media. Separation and Purification Technology 2019, 209, 826–832. [Google Scholar] [CrossRef]
- Santos, T.M.; Rigual, V.; Domínguez, J.C.; Alonso, M.V.; Oliet, M.; Rodriguez, F. Fractionation of Pinus Radiata by Ethanol-Based Organosolv Process. Biomass Conv. Bioref. 2024, 14, 451–464. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, K. Low-Temperature Alkali-Anthraquinone Pretreatment of Wheat Straw: Gradual Dissolution of Lignin and Carbohydrate Retention in Pretreated Solids. BioRes 2017, 12, 8550–8564. [Google Scholar] [CrossRef]
- Cachet, N.; Benjelloun-Mlayah, B. Comparison of Organic Acid-Based Organosolv Lignins Extracted from the Residues of Five Annual Crops. BioRes 2021, 16, 7966–7990. [Google Scholar] [CrossRef]
- Obama, P.; Ricochon, G.; Muniglia, L.; Brosse, N. Combination of Enzymatic Hydrolysis and Ethanol Organosolv Pretreatments: Effect on Lignin Structures, Delignification Yields and Cellulose-to-Glucose Conversion. Bioresource Technology 2012, 112, 156–163. [Google Scholar] [CrossRef]
- Sun, F.F.; Zhao, X.; Hong, J.; Tang, Y.; Wang, L.; Sun, H.; Li, X.; Hu, J. Industrially Relevant Hydrolyzability and Fermentability of Sugarcane Bagasse Improved Effectively by Glycerol Organosolv Pretreatment. Biotechnol Biofuels 2016, 9, 59. [Google Scholar] [CrossRef]
- Sun, T.-S.; Wang, K.; Yang, G.; Yang, H.-Y.; Xu, F. Hydrothermal Treatment and Enzymatic Saccharification of Corncobs. BioResources 2014, 9, 3000–3013. [Google Scholar] [CrossRef]
- Chen, H.; Li, X.; Yu, S.; Qin, Y.; Qu, Y.; Zhao, J. Potassium Permanganate Assisted Organosolv Pretreatment Enhances Enzymatic Hydrolysis of Corn Stover. GCB Bioenergy 2021, 13, 665–678. [Google Scholar] [CrossRef]
- Li, Z.; Jiang, Z.; Fei, B.; Pan, X.; Cai, Z.; Liu, X.; Yu, Y. Ethanol Organosolv Pretreatment of Bamboo for Efficient Enzymatic Saccharification. BioRes 2012, 7, 3452–3462. [Google Scholar] [CrossRef]
- Wolf, M.; Berger, F.; Hanstein, S.; Weidenkaff, A.; Endreß, H.-U.; Oestreich, A.M.; Ebrahimi, M.; Czermak, P. Hot-Water Hemicellulose Extraction from Fruit Processing Residues. ACS Omega 2022, 7, 13436–13447. [Google Scholar] [CrossRef]
- Zhang, X.; Du, L.; Li, S. STEPWISE EXTRACTION OF HEMICELLULOSES WITH WATER AND ALKALI FROM LARCH WOOD AND THEIR SUGAR COMPOSITIONS. WR 2024, 68, 638–649. [Google Scholar] [CrossRef]
- Raghavendran, V.; Nitsos, C.; Matsakas, L.; Rova, U.; Christakopoulos, P.; Olsson, L. A Comparative Study of the Enzymatic Hydrolysis of Batch Organosolv-Pretreated Birch and Spruce Biomass. AMB Expr 2018, 8, 114. [Google Scholar] [CrossRef] [PubMed]
- Ilanidis, D.; Stagge, S.; Jönsson, L.J.; Martín, C. Hydrothermal Pretreatment of Wheat Straw: Effects of Temperature and Acidity on Byproduct Formation and Inhibition of Enzymatic Hydrolysis and Ethanolic Fermentation. Agronomy 2021, 11, 487. [Google Scholar] [CrossRef]
- Chu, Q.; Tong, W.; Chen, J.; Wu, S.; Jin, Y.; Hu, J.; Song, K. Organosolv Pretreatment Assisted by Carbocation Scavenger to Mitigate Surface Barrier Effect of Lignin for Improving Biomass Saccharification and Utilization. Biotechnol Biofuels 2021, 14, 136. [Google Scholar] [CrossRef] [PubMed]
- Kellock, M.; Maaheimo, H.; Marjamaa, K.; Rahikainen, J.; Zhang, H.; Holopainen-Mantila, U.; Ralph, J.; Tamminen, T.; Felby, C.; Kruus, K. Effect of Hydrothermal Pretreatment Severity on Lignin Inhibition in Enzymatic Hydrolysis. Bioresource Technology 2019, 280, 303–312. [Google Scholar] [CrossRef]
- Momoh, O.L.Y.; Ouki, S. Development of a Novel Fractal-like Kinetic Model for Elucidating the Effect of Particle Size on the Mechanism of Hydrolysis and Biogas Yield from Ligno-Cellulosic Biomass. Renewable Energy 2018, 118, 71–83. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, L.; Liu, D. Biomass Recalcitrance. Part I: The Chemical Compositions and Physical Structures Affecting the Enzymatic Hydrolysis of Lignocellulose. Biofuels Bioprod Bioref 2012, 6, 465–482. [Google Scholar] [CrossRef]

| Wash Steps | Wash solution | Liquor ratio* |
|---|---|---|
| 1 | 60% (w/w) ethanol–water solution | 1:5 |
| 2 | Water | 1:20 |
| 3 | Water | 1:10 |
| Catalyst type | pKa | CCatalyst [%-DM] | P-Factor | Glucose yield |
|---|---|---|---|---|
| Phosphoric acid | 2.15 [33] | 20 | 3.4 | 20 ± 6.7% |
| Lactic acid | 3.86 [33] | 50 | 3.4 | 41 ± 1.1% |
| Acetic acid | 4.76 [33] | 50 | 3.4 | 23 ± 3.7% |
| Sulfuric acid | -3.0 [33] | 5 | 3.9 | 83 ± 2.4% |
| PTSA | -2.8 [34] | 5 | 3.9 | 81 ± 6.2% |
| Without catalyst | - | - | 3.9 | 22 ± 1.5% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




