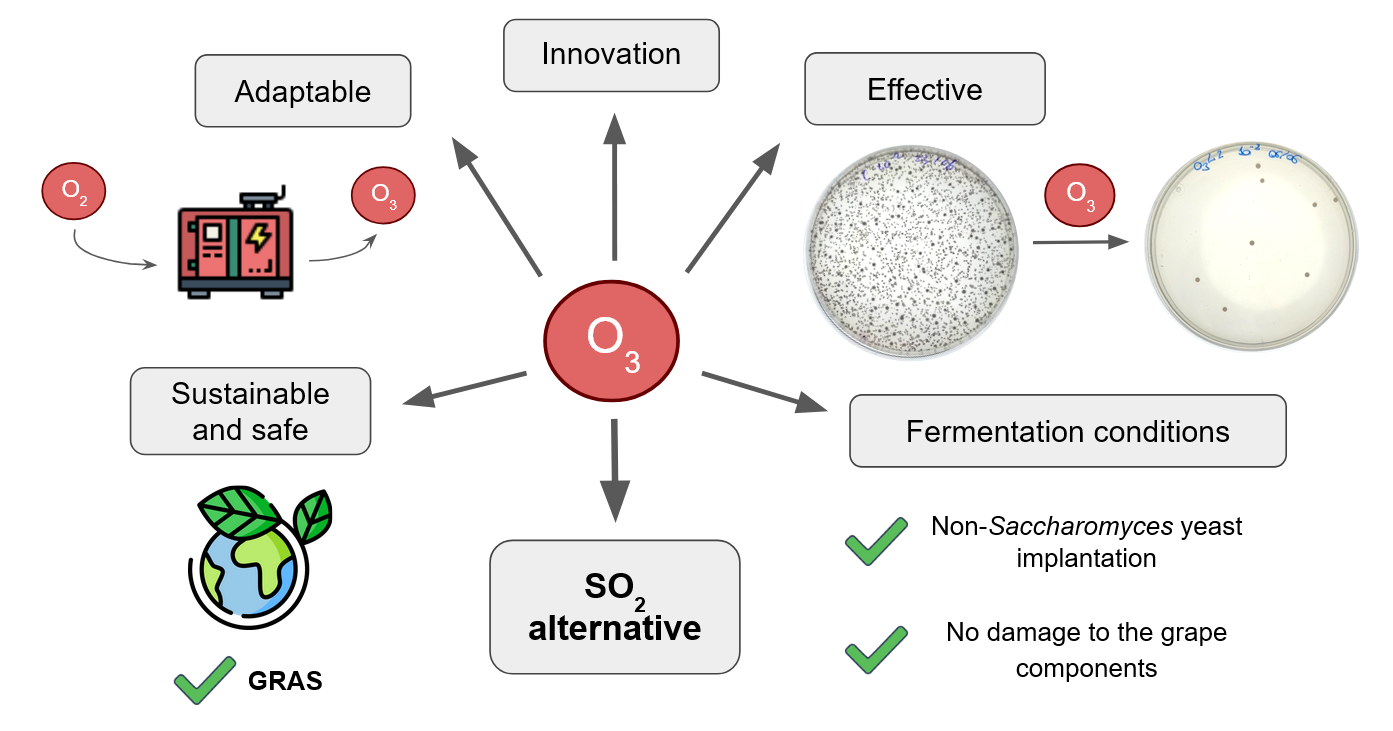
Climate change presents a challenge for global viticulture due to rising temperatures and water stress, which accelerate grape ripening, increase sugar levels, and reduce acidity. This compromises wine quality and microbial stability, resulting in higher re-liance on sulfur dioxide (SO₂). However, SO₂ can inhibit desirable fermentations, in-cluding those carried out by non-Saccharomyces yeasts, which are key biotechnological tools for climate adaptation due to their ability to modulate acidity, aroma, and etha-nol. Therefore, alternative disinfection methods are needed to control wild microbiota without hindering inoculated yeasts. This review critically analyzes ozone (O₃) as a non-thermal disinfection technology for winemaking. It examines the antimicrobial mechanism of ozone, its efficacy against wine-related microorganisms, its impact on the physicochemical and aromatic parameters of grapes, and its practical viability. Ozone effectively reduces spoilage-causing microbiota while preserving crucial grape compounds and providing a favorable environment for novel fermentation biotech-nologies. Compared to other emerging technologies and SO₂, ozone offers a balanced profile: effective disinfection, minimal residues, cost-effectiveness, and compatibility with sustainable winemaking. Ozone is emerging as a promising alternative to facili-tate controlled fermentations and improve wine quality among the current climatic and oenological challenges.