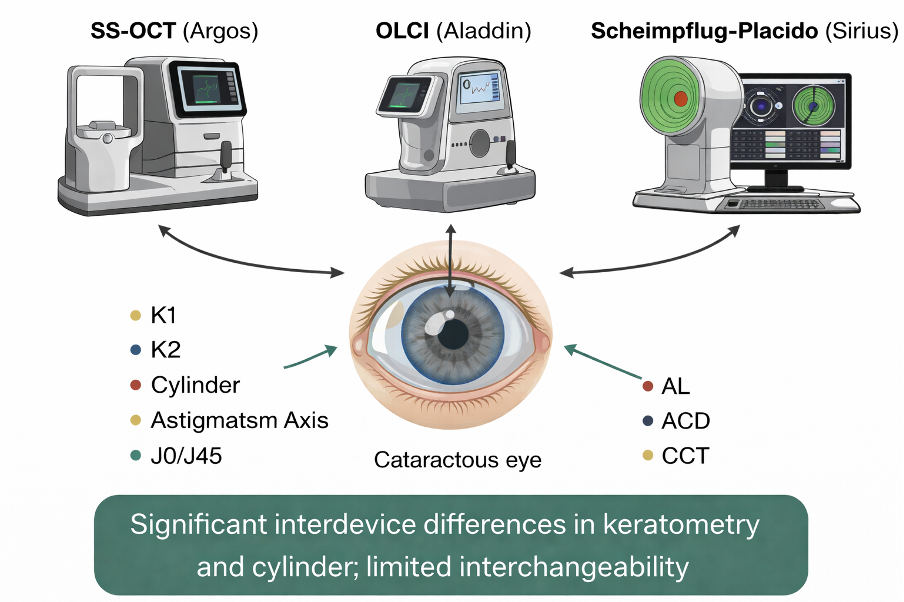
1. Introduction
The precise measurement of anterior segment parameters is paramount in contemporary cataract surgery, influencing the accuracy of intraocular lens power calculation, the final refractive outcome and the diagnosis and management of various corneal pathologies [
1].
A multitude of technologies have emerged to facilitate these measurements, each with its own set of advantages and limitations. Among these technologies, swept-source optical coherence tomography (SS-OCT) biometers have gained prominence due to their high-speed scanning capabilities and enhanced penetration through dense media, enabling comprehensive anterior segment imaging [
2]. The Argos biometer (MOVU Inc., Komaki, Japan), which employs SS-OCT technology, features a light source centered at 1050 nm. The narrow-bandwidth wavelength of this light source enhances the signal-to-noise ratio, enabling improved tissue penetration and image quality, even in the presence of dense cataracts [
3].
Optical low-coherence interferometry (OLCI) biometers, such as the Aladdin (Topcon, Tokyo, Japan), represent an older generation of optical biometers capable of non-invasively measuring axial length (AL), anterior chamber depth (ACD), pupillometry, corneal keratometry (K), and white-to-white topography (WTW). This biometer employs a 24-ring Placido disk reflection, with an approximate working distance of eight centimeters, to perform a precise keratometry and corneal topography assessment. However, the accuracy of measurements obtained from a OLCI biometer may be impacted by the presence of media opacities, such as hard cataracts [
4].
Corneal topographers employing combined Scheimpflug-Placido disk technology like the Schwind Sirius (SCHWIND eye-tech-solutions, GmbH, Kleinostheim) provide detailed information about corneal curvature, elevation, and thickness, which is essential for refractive surgery planning and the diagnosis of corneal ectasia. These devices provide valuable data that can be used for planning laser-assisted in situ keratomileusis (LASIK) treatments but also choosing a toric intraocular lens, correcting astigmatism and optimizing visual outcomes after cataract surgery and refractive procedures. [
5]
Persistent astigmatism post-cataract intervention can significantly impair visual quality and patient satisfaction. Approximately 40% of individuals undergoing cataract surgery exhibit corneal astigmatism exceeding 1 D, which can be addressed using a toric intraocular lens [
6]. As lenticular astigmatism is eliminated during cataract surgery, the measurement of total corneal astigmatism (anterior and posterior) remains critical in surgical planning. Measuring anterior corneal astigmatism requires combining various methods, such as keratometry, topography, Scheimpflug imaging, and OCT, to account for irregular astigmatism or ectatic disease that may not be apparent with a single technique. Accurately measuring posterior corneal astigmatism poses challenges, but techniques such as Scheimpflug imaging, OCT, and light-emitting diode-based devices, used in conjunction with theoretical formulas like the Barrett toric calculator, can help to compensate for this [
7].
Given the need for precision, this study aims to compare the measurements obtained across different devices, in order to determine the degree of agreement between them and to identify any potential sources of variability.
2. Materials and Methods
2.1. Study Design, Ethics, and Informed Consent
This retrospective, observational study, conducted at the Ophthalmology Department of “Victor Babes” University of Medicine and Pharmacy in Timisoara, Romania from January 2022 to June 2024, evaluated the measurements done to the patients undergoing successful cataract surgery. The study adhered to the principles of the Declaration of Helsinki, and the institutional ethics committee waived specific approval requirements due to its retrospective nature. Furthermore, the informed consent obtained from all participants for their surgical procedures explicitly included consent for the use of their data in research studies. Importantly, patient confidentiality was maintained throughout the study.
2.2. Patients and Procedure
Subjects included were those undergoing uneventful cataract surgery with intraocular lens (IOL) implantation. The exclusion criteria included ocular trauma, severe corneal or vitreous opacities, previous corneal surgery, retinal disease, and systemic disease affecting the eye. All patients were measured with the three devices in a single session, by the same examiner and faced no difficulties. The following parameters were obtained and compared: K1, K2 and cylinder on all devices. Corneal astigmatism was represented as vector components J0 and J45 calculated according to the following formulas [
8]
- ○
J0 = - (C/2) * cos(2*axis)
- ○
J45 = - (C/2) * sin(2*axis).
- ○
(C = cylinder or K2-K1, A = axis).
Central corneal thickness (CCT) was measured and compared between Aladdin and Schwind Sirius.
Additionally, white-to-white (WTW), lens thickness (LT) and anterior chamber depth (ACD) were measured using the two biometers, the SS-OCT Argos, and the OLCI Aladdin. All devices were calibrated prior to the measurement session.
2.3. Devices
The Argos SS-OCT device generates 2D OCT images at 3000 A-scans/s using 1060 nm light. It measures corneal curvature using a 1.3375 refractive index and a 2.2 mm ring of 16 LEDs combined with the OCT data. The corneal diameter is also determined from the OCT image, and the newer Argos 1.5 version uses this to calculate the white-to-white value. Additionally, the device measures central corneal thickness (CCT), anterior chamber depth (ACD), lens thickness (LT), and axial length (AL) using OCT data with varied refractive indices (cornea: 1.376; aqueous and vitreous humors: 1.336; lens: 1.410). However, the CCT measurement is not readily available on the report, like the other parameters.
The Aladdin biometer is based on OLCI, with an 820 mm superluminescent diode, for measuring AL, ACD, CCT, and LT. The keratometry (K) readings are calculated using a 24-ring Placido disk reflection, which captures data from approximately 1,024 reference points within a 3.0 mm (from 2.8 to 3.2 mm) optical zone on the corneal surface. The WTW distance is calculated from the corneal topography.
The Schwind Sirius topography instrument is an anterior segment analysis system that combines a rotating Scheimpflug camera and a Placido disc. It acquires 25 radial sections of the cornea and anterior chamber. The system provides data on the tangential and axial curvature of the anterior and posterior corneal surfaces, the overall refractive power of the cornea, corneal thickness mapping, and wavefront analysis. The corneal surfaces are examined using blue LED light. Measurements of the anterior corneal surface are obtained by combining Placido and Scheimpflug data, while measurements of other internal structures rely solely on Scheimpflug imaging.
2.4. Statistical Analysis
Data analysis was performed with JASP v0.19.3 (open-source software for statistical analysis, supported by the University of Amsterdam). Mean and standard deviation, median and interquartile range were used to present numerical variables, while frequency and percentages were used for nominal variables. The distribution of continuous variables was assessed using the Shapiro-Wilk Test for normality of numerical data distribution. For non-Gaussian distribution of data, we compared two groups using non-parametric Mann-Whitney U Test, respectively, more than two groups were compared using non-parametric Friedman Test. A p-value of less than 0.05 was considered statistically significant.
3. Results
In this study, a total of 170 eyes from 102 patients with uncomplicated cataract were evaluated. The mean age of the participants was 69.12 ± 10.26 years (ranging from 40 to 86 years). All eyes were successfully measured using the three devices, and no issues arose during the process (
Table 1).
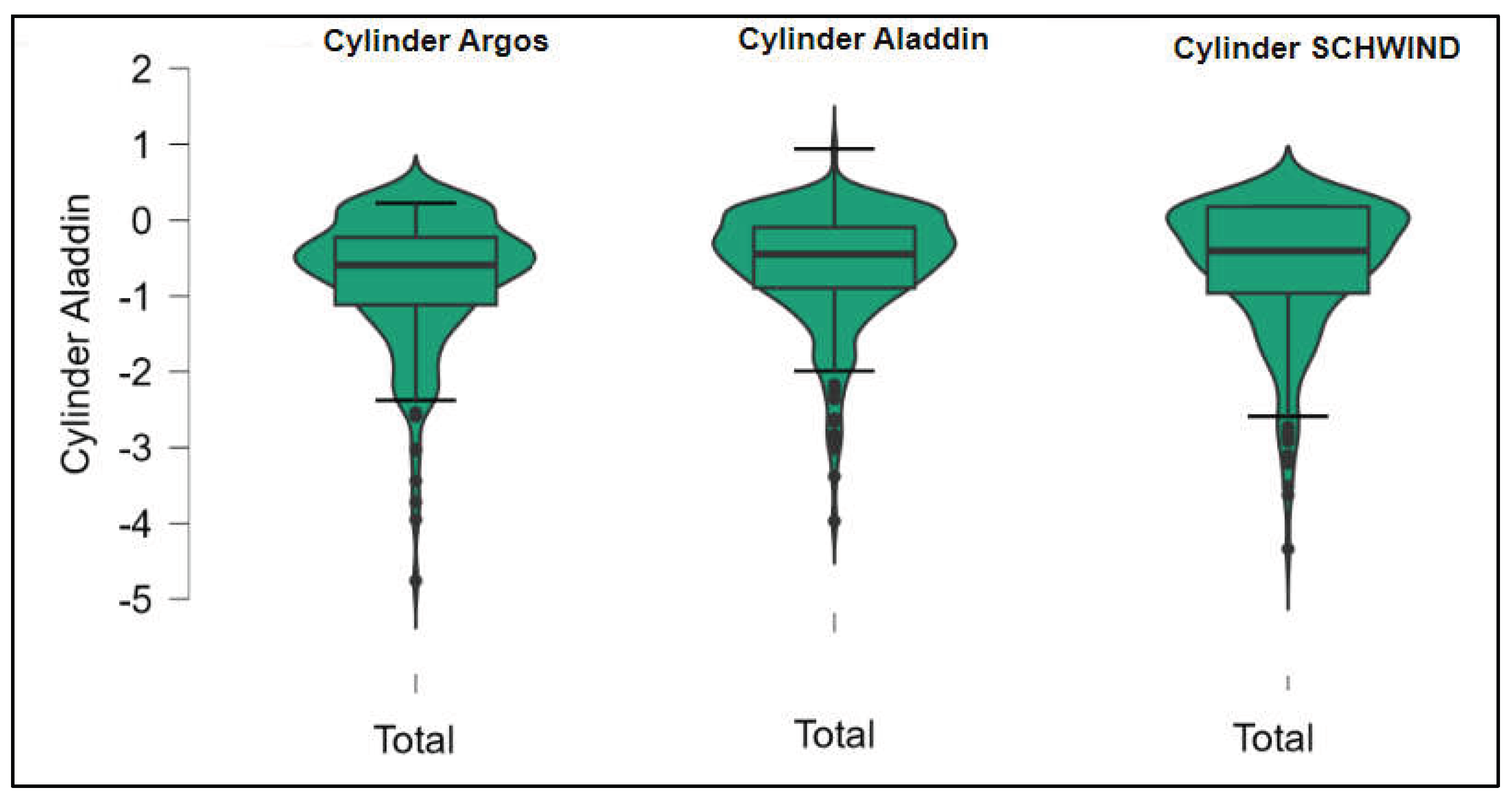
Further analysis revealed significant disparities in several key parameters across the devices, particularly in keratometry readings, cylinder, lens thickness, axial length, anterior chamber depth, and white-to-white measurements (p < 0.001 for all, except J0 and J45 components). Conversely, measurements of J0, J45, and central corneal thickness showed no statistically significant differences between the devices, suggesting a higher level of agreement for these specific parameters. The Supplemental
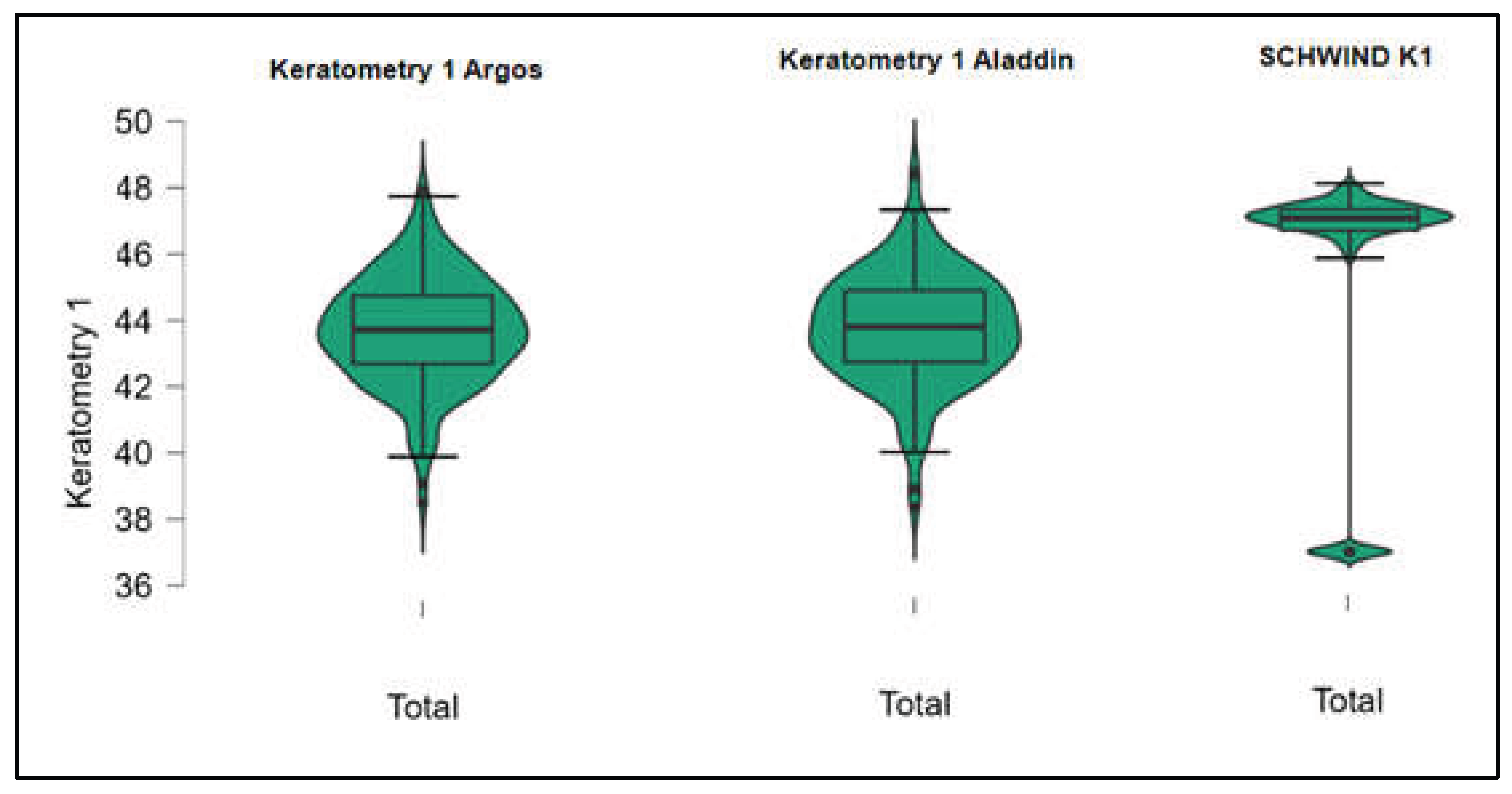
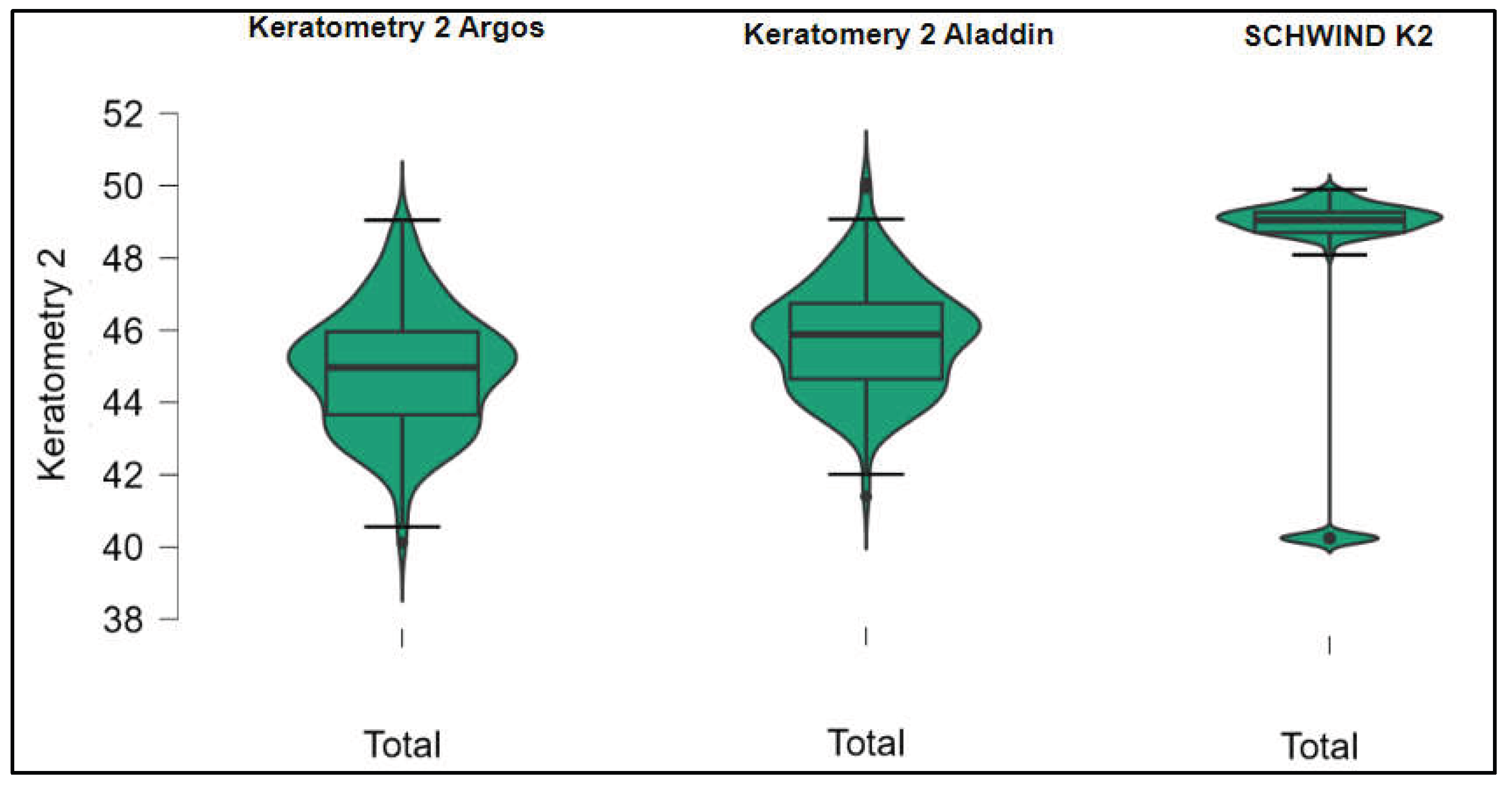
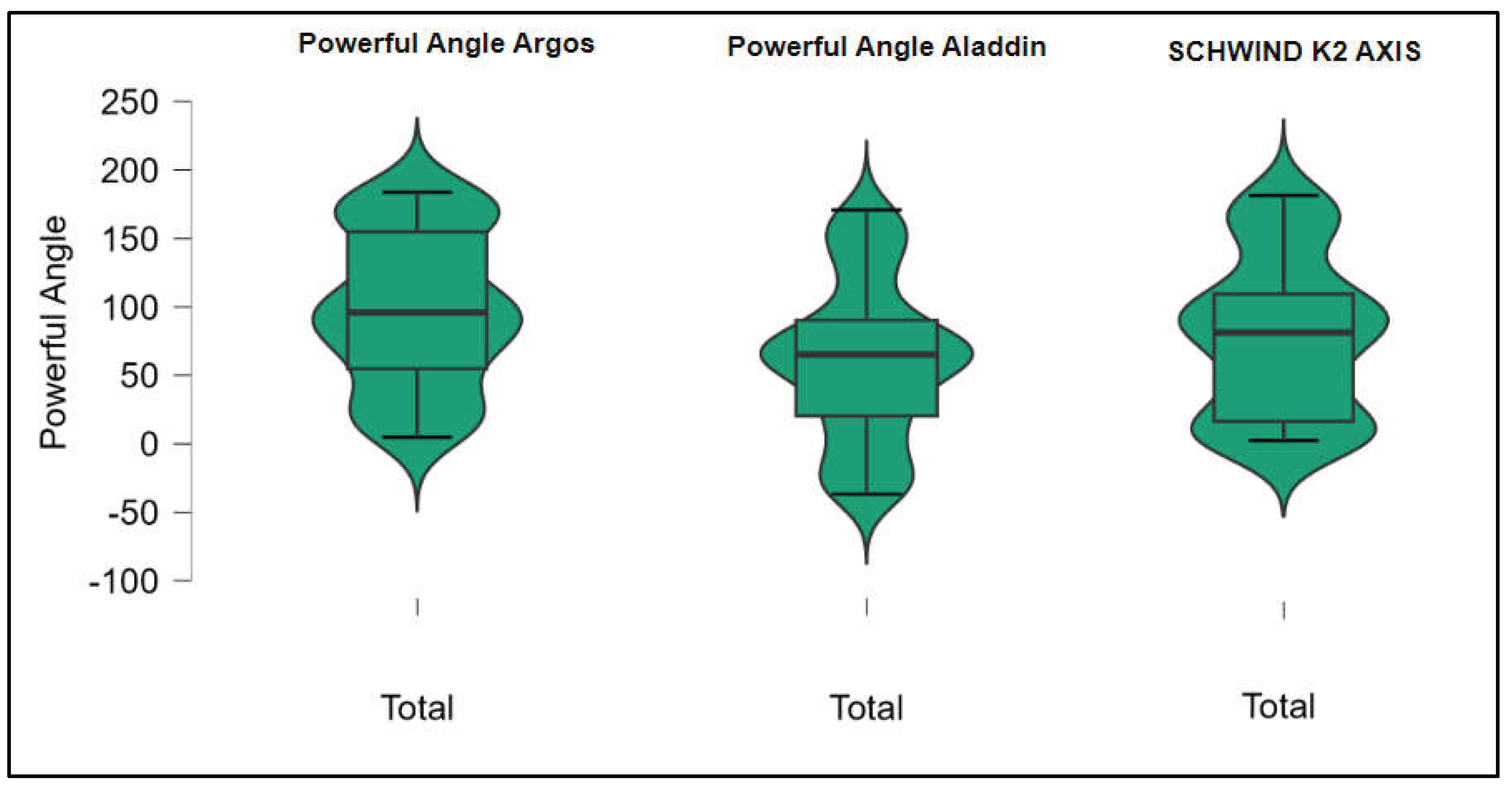
Figures S1–S4 show the difference in K1, K2, Powerful Angle and Cylinder measurements across the 3 devices (Friedman test). In the case of these variables, comparisons of measurements between 2 devices were continued (Mann-Whitney U test).
Figure 1.
Flat keratometry (K1) measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Figure 1.
Flat keratometry (K1) measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Figure 2.
Steep keratometry (K2) measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Figure 2.
Steep keratometry (K2) measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Figure 3.
Astigmatism axis (Powerful Angle) measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Figure 3.
Astigmatism axis (Powerful Angle) measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Figure 4.
Corneal cylinder measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Figure 4.
Corneal cylinder measurements across Argos SS-OCT, Aladdin OLCI, and Schwind Sirius (Friedman test).
Table 2.
Mann-Whitney U test comparison of keratometry and astigmatism between Argos SS-OCT and Aladdin OLCI biometers (W, z, p).
Table 2.
Mann-Whitney U test comparison of keratometry and astigmatism between Argos SS-OCT and Aladdin OLCI biometers (W, z, p).
Argos vs Alladin
|
W |
z |
p |
| Keratometry 1 |
8101 |
2.35 |
0.019* |
| Keratometry 2 |
10474.5 |
6.091 |
< 0.001* |
| Powerful Angle |
6994 |
0.656 |
0.512 |
| Cylinder |
4892 |
-3.796 |
< 0.001* |
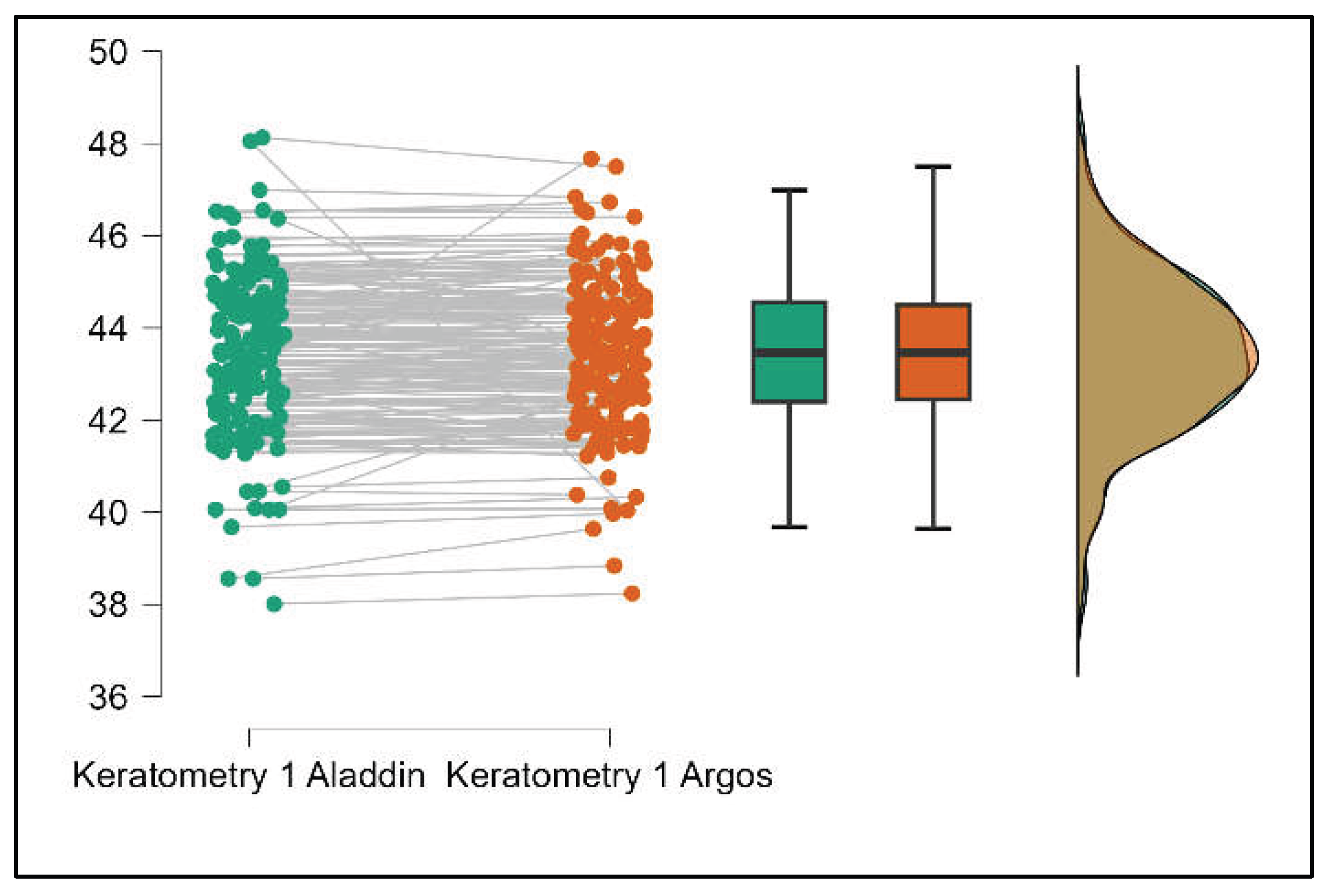
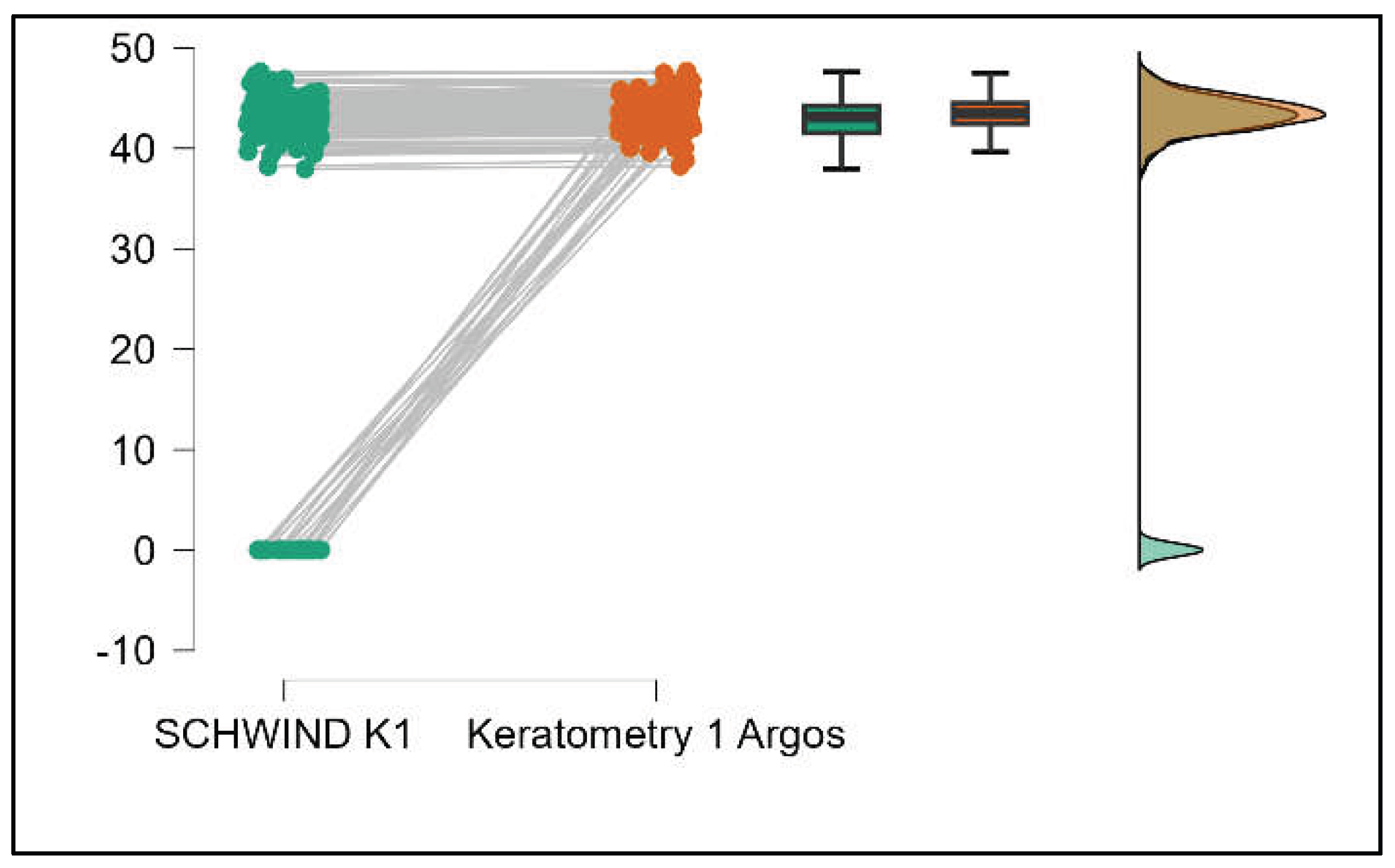
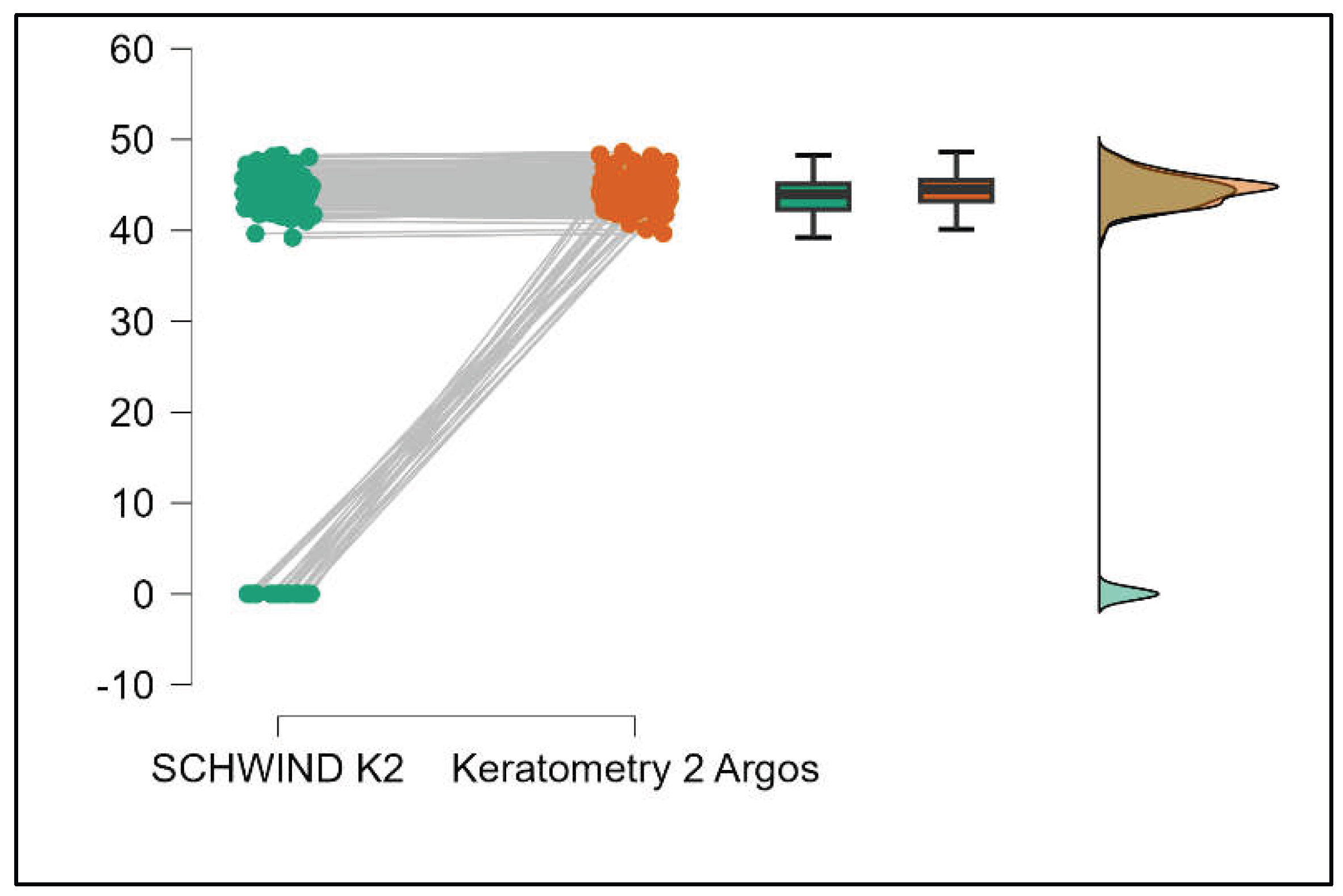
Values of K1 measured by Argos are significantly increased compared with Aladdin (p=0.019) as shown in Supplemental
Figure S5. Values of K2 measured by Argos also exhibited higher values than those obtained from Aladdin (p<0.001), as detailed in Supplemental
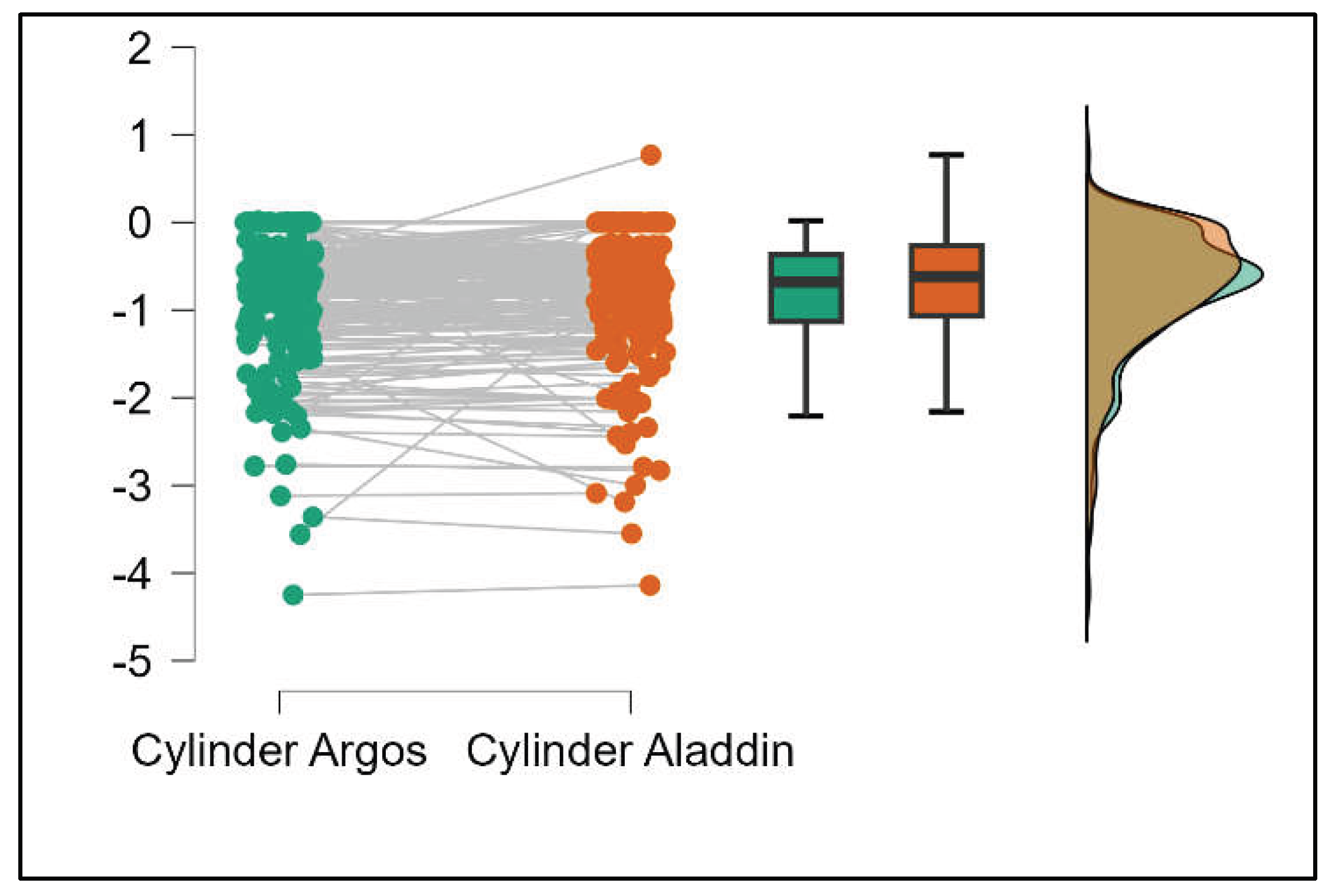
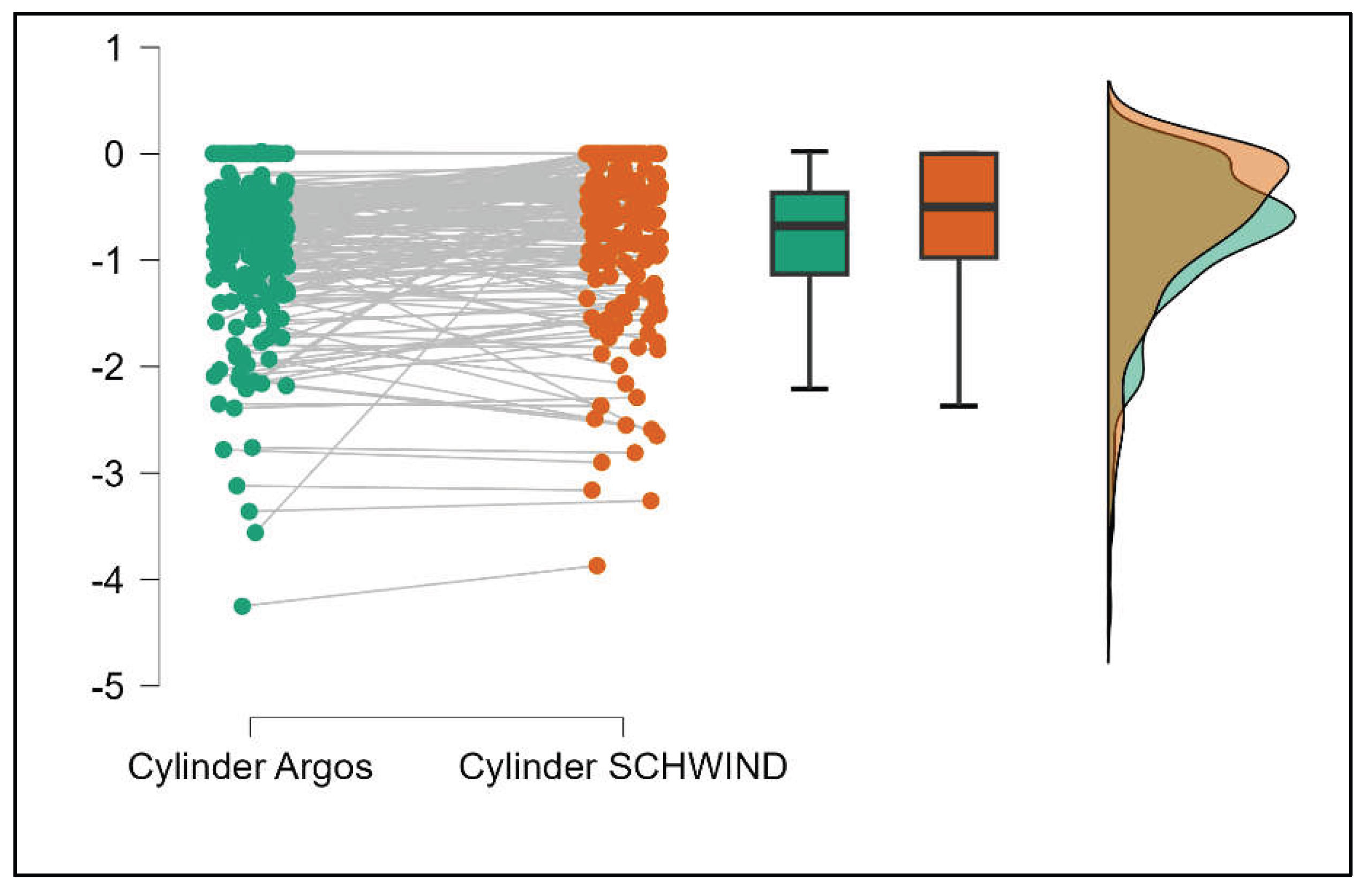
Figure S6. The Cylinder values from Argos were significantly lower than those from Aladdin (p<0.001) as shown in Supplemental
Figure S7.
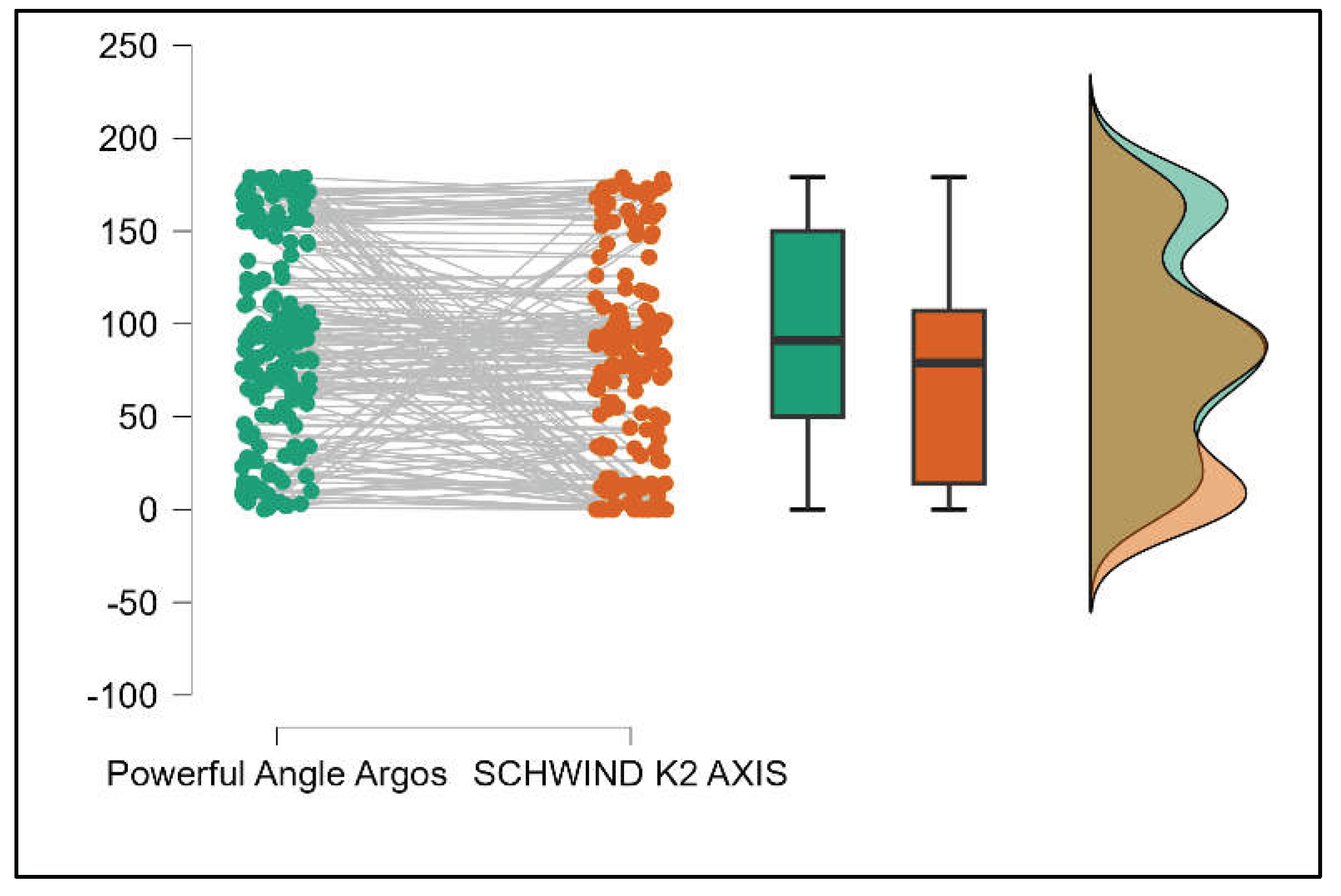
Conversely, the Powerful Angle demonstrated no significant difference between the Argos and Aladdin devices (p = 0.512), indicating a consistent measurement for this parameter across the two biometers.
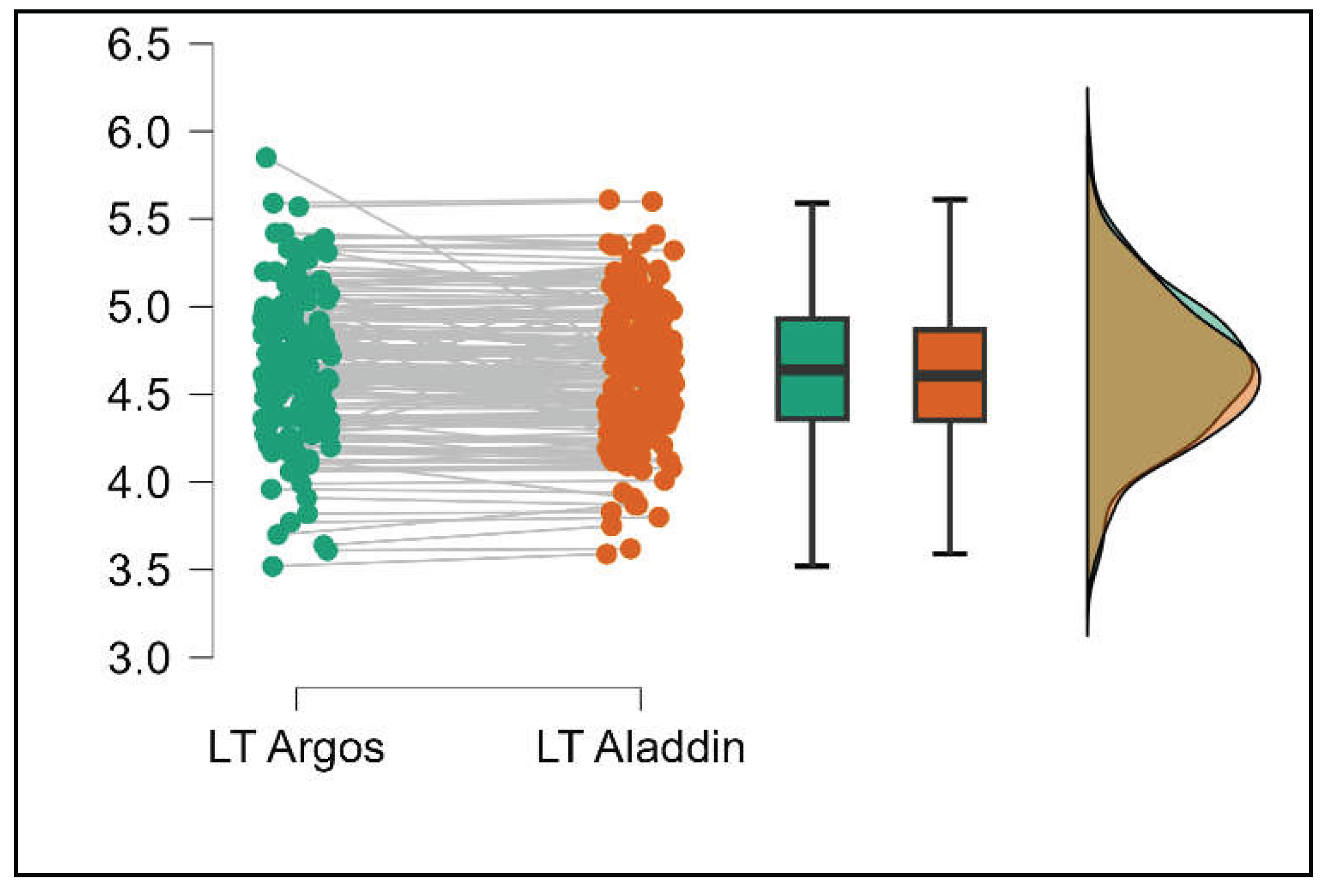
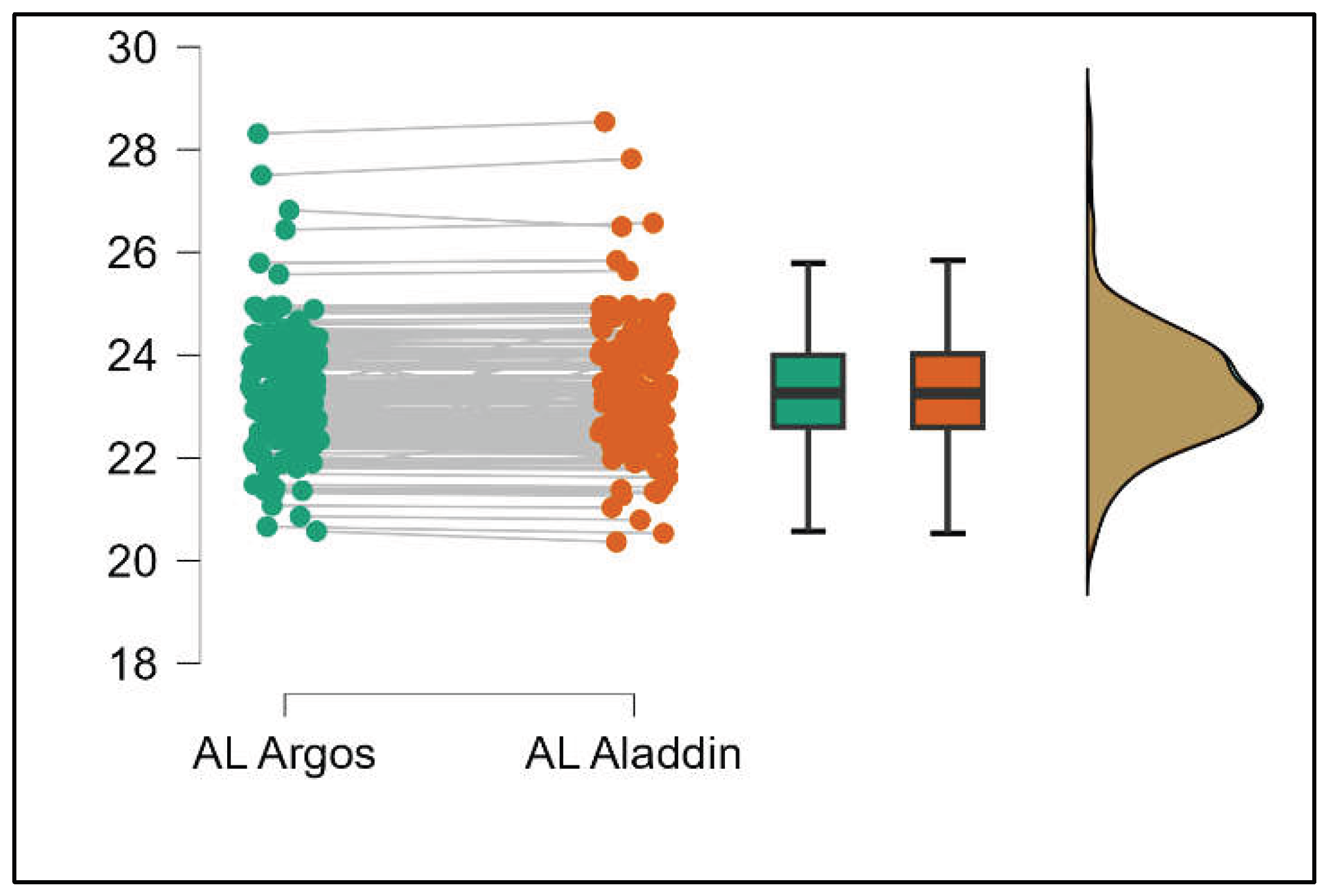
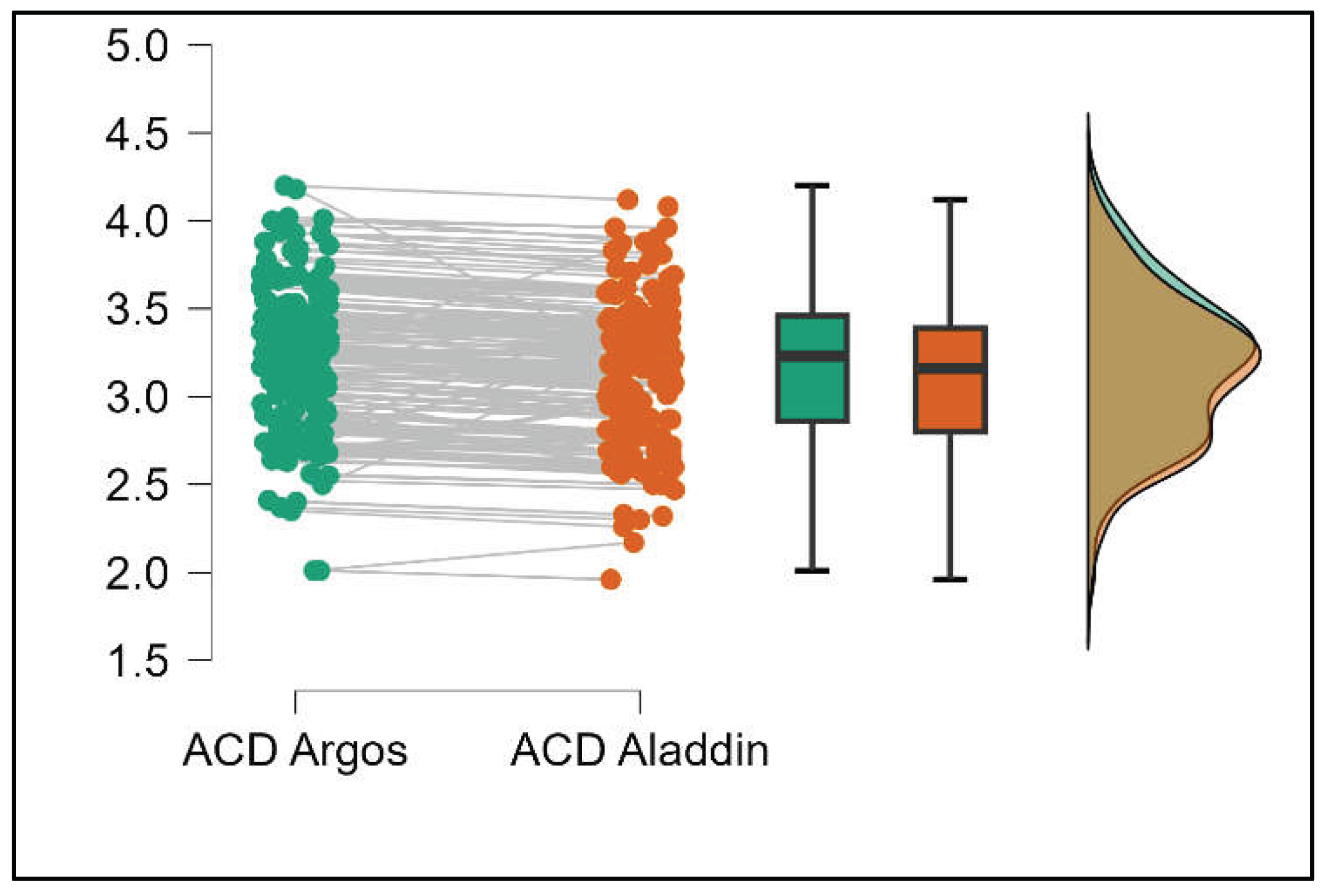
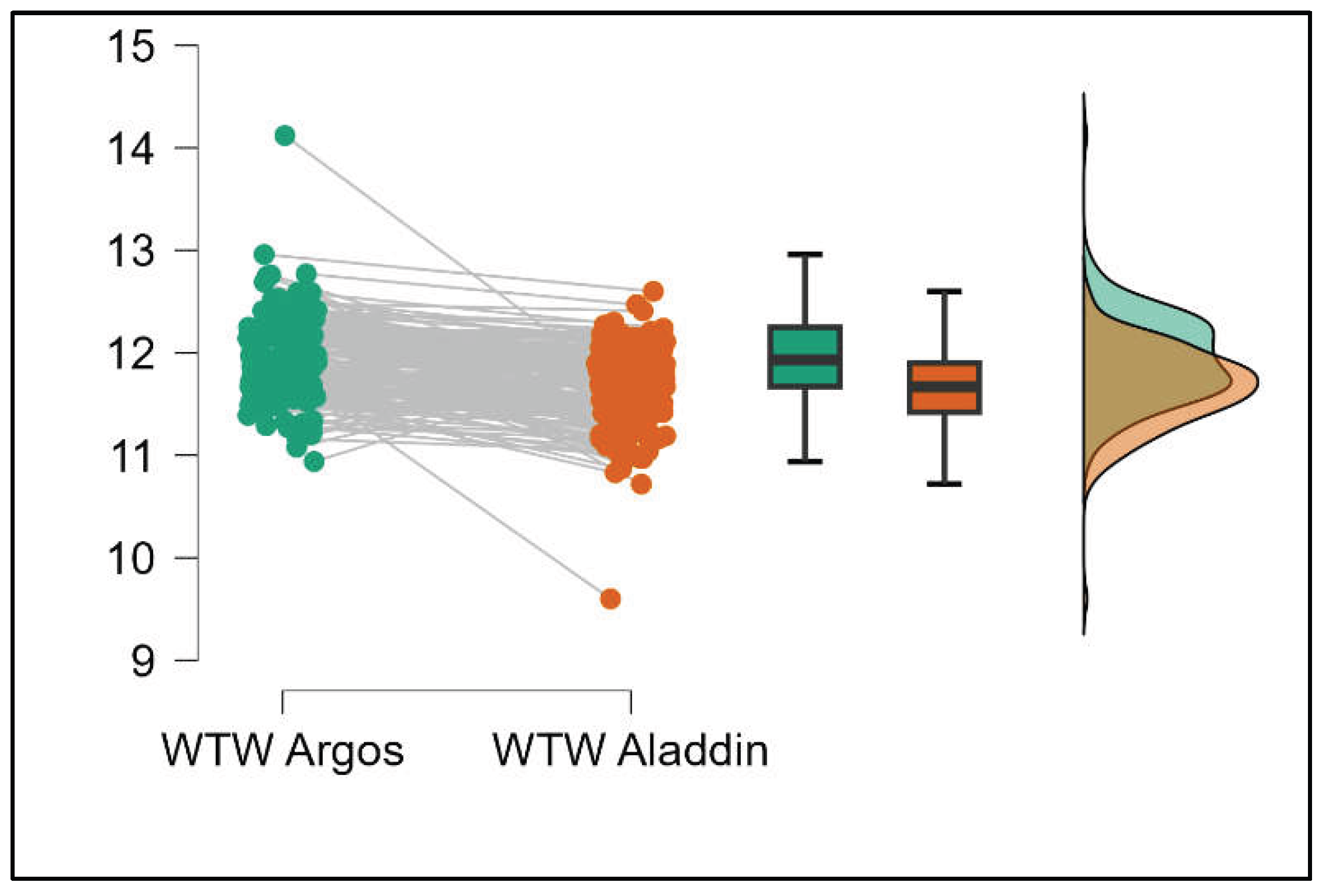
Similarly, the Argos device consistently yielded higher measurements for lens thickness (p<0.001), axial length (p<0.001), anterior chamber depth (p<0.001), and white-to-white (p<0.001) when compared to the Aladdin, as demonstrated in Supplemental
Figures S8–S11.
Figure 5.
Pairwise comparison of flat keratometry (K1) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 5.
Pairwise comparison of flat keratometry (K1) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 6.
Pairwise comparison of steep keratometry (K2) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 6.
Pairwise comparison of steep keratometry (K2) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 7.
Pairwise comparison of corneal cylinder between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 7.
Pairwise comparison of corneal cylinder between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 8.
Pairwise comparison of lens thickness (LT) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 8.
Pairwise comparison of lens thickness (LT) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 9.
Pairwise comparison of axial length (AL) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 9.
Pairwise comparison of axial length (AL) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 10.
Pairwise comparison of anterior chamber depth (ACD) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 10.
Pairwise comparison of anterior chamber depth (ACD) between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 11.
Pairwise comparison of white-to-white (WTW) distance between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Figure 11.
Pairwise comparison of white-to-white (WTW) distance between Argos SS-OCT and Aladdin OLCI biometers (Mann-Whitney U test).
Table 3 compares the values of keratometry readings, powerful angle, and cylinder between the Argos and Schwind devices. The analysis revealed that both K1 and K2 measurements from Argos were significantly higher than those from Schwind (Supplemental
Figures S12 and S13). The average Powerful Angle values are significantly lower in the case of Schwind compared to Argos (Mann-Whitney U test, p=0.003). This pattern of difference is further accentuated by the significantly lower cylinder values observed with Argos compared to Schwind (Mann-Whitney U test, p<0.001), as presented in Supplemental
Figure S14 and S15.
Figure 12.
Pairwise comparison of flat keratometry (K1) between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
Figure 12.
Pairwise comparison of flat keratometry (K1) between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
Figure 13.
Pairwise comparison of steep keratometry (K2) between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
Figure 13.
Pairwise comparison of steep keratometry (K2) between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
Figure 14.
Pairwise comparison of astigmatism axis (Powerful Angle) between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
Figure 14.
Pairwise comparison of astigmatism axis (Powerful Angle) between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
Figure 15.
Pairwise comparison of corneal cylinder between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
Figure 15.
Pairwise comparison of corneal cylinder between Argos SS-OCT and Schwind Sirius (Mann-Whitney U test).
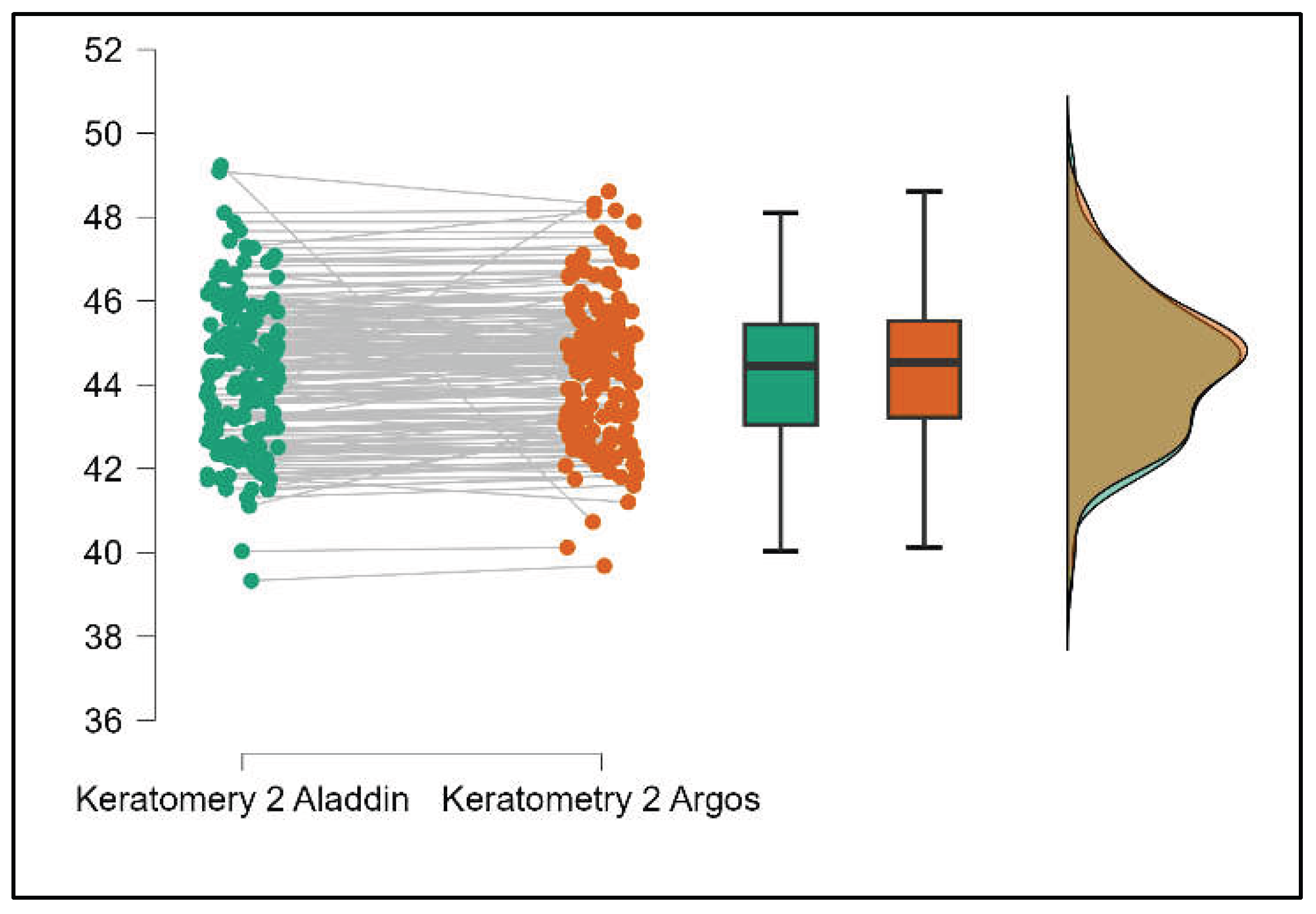
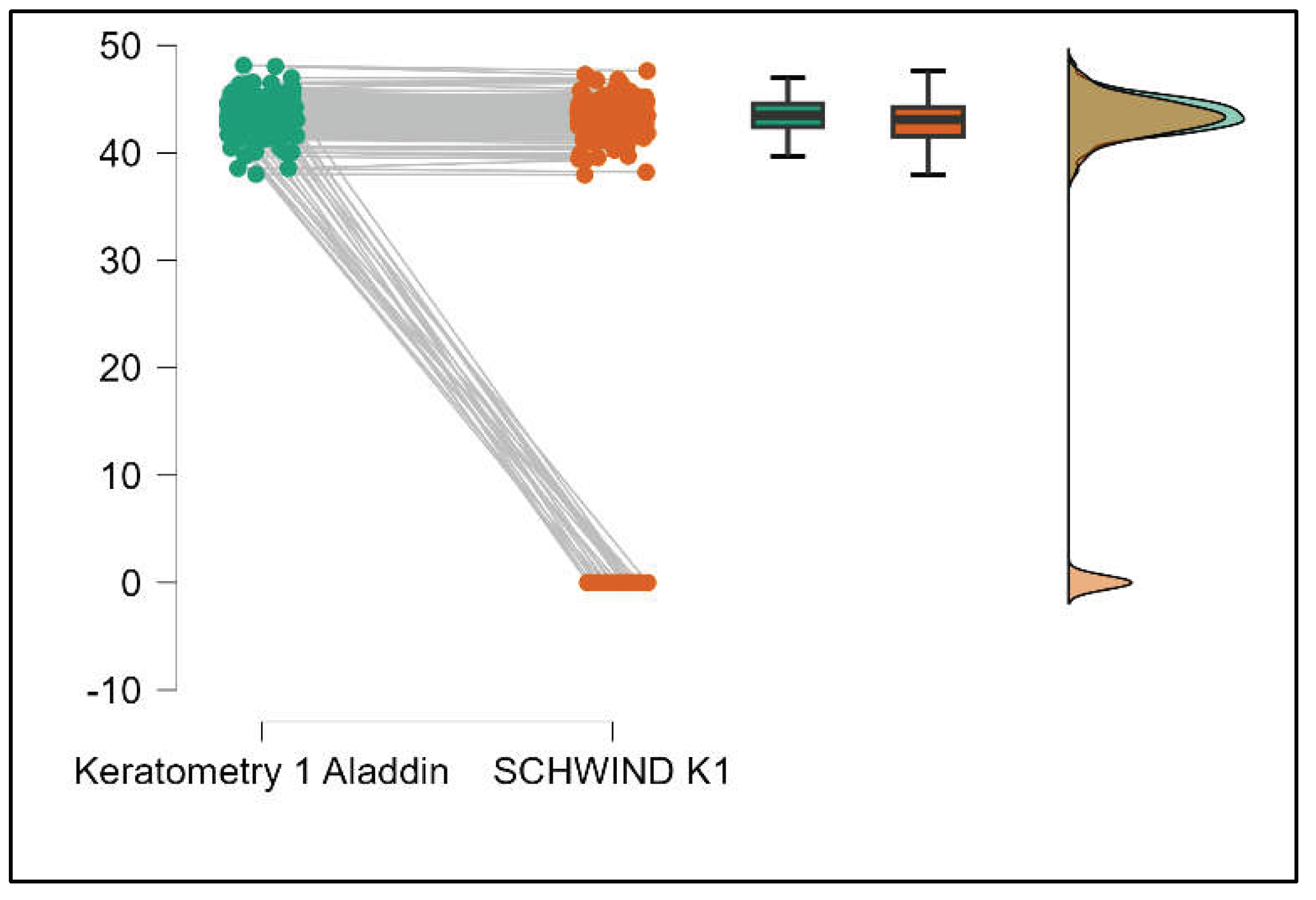
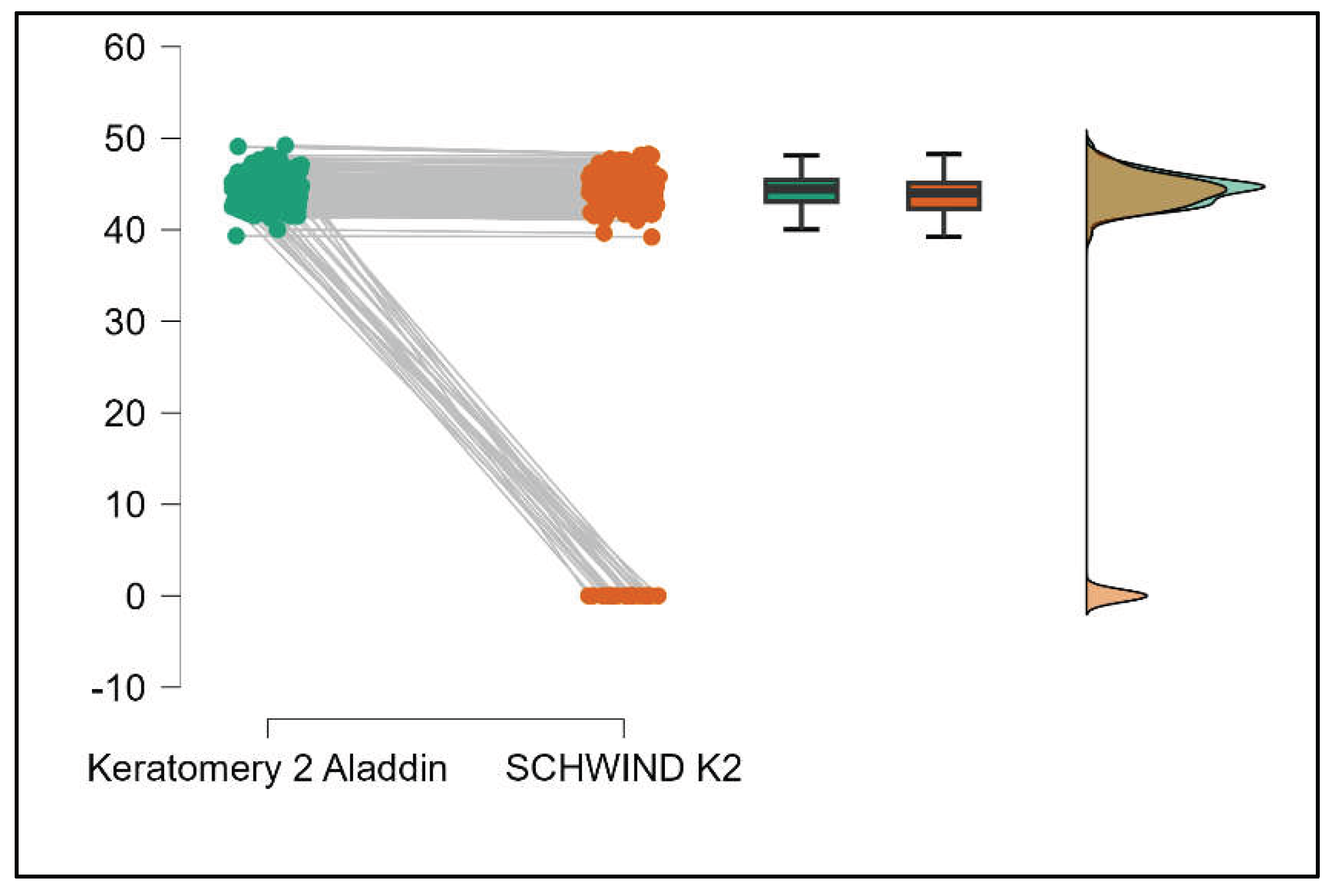
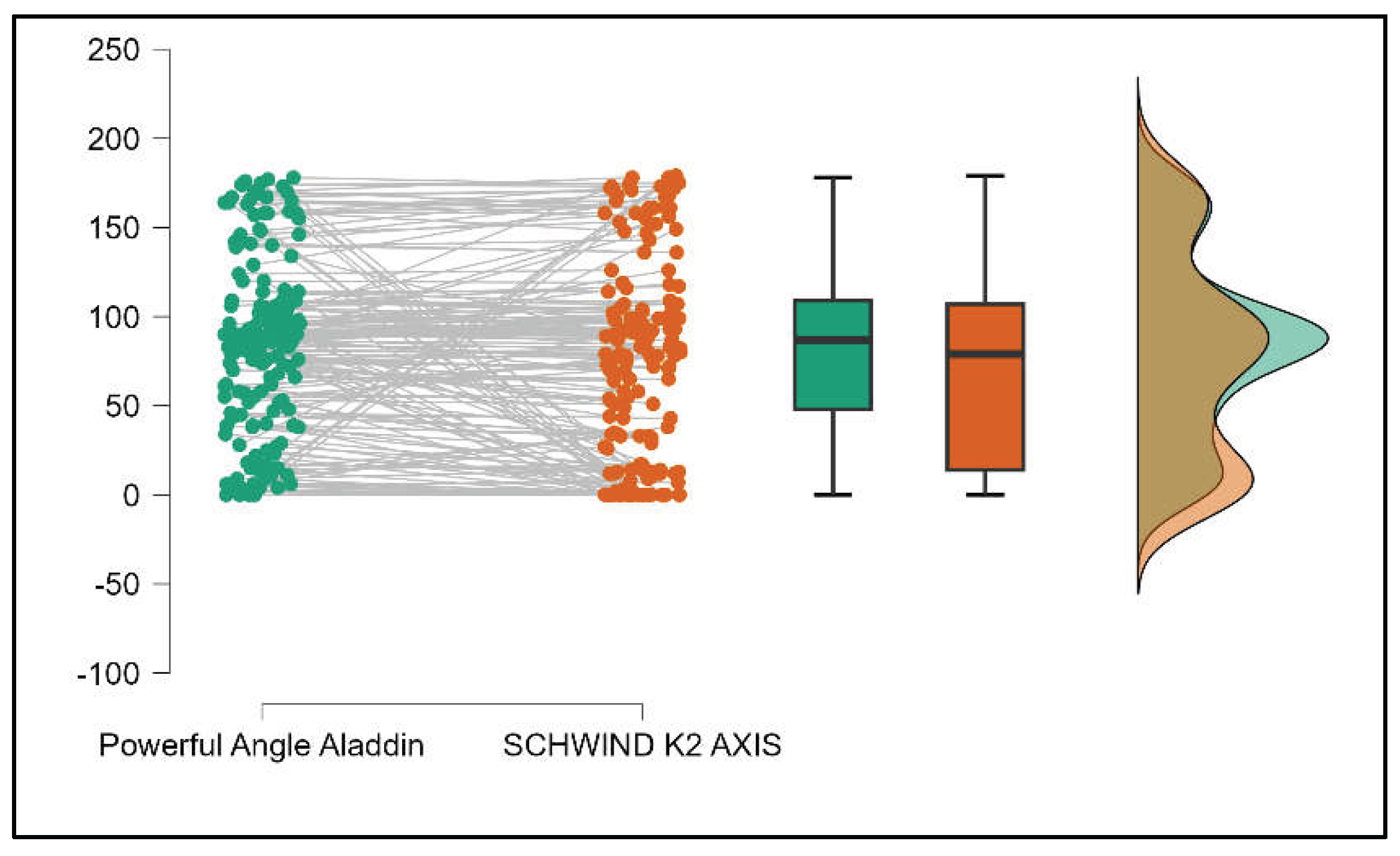
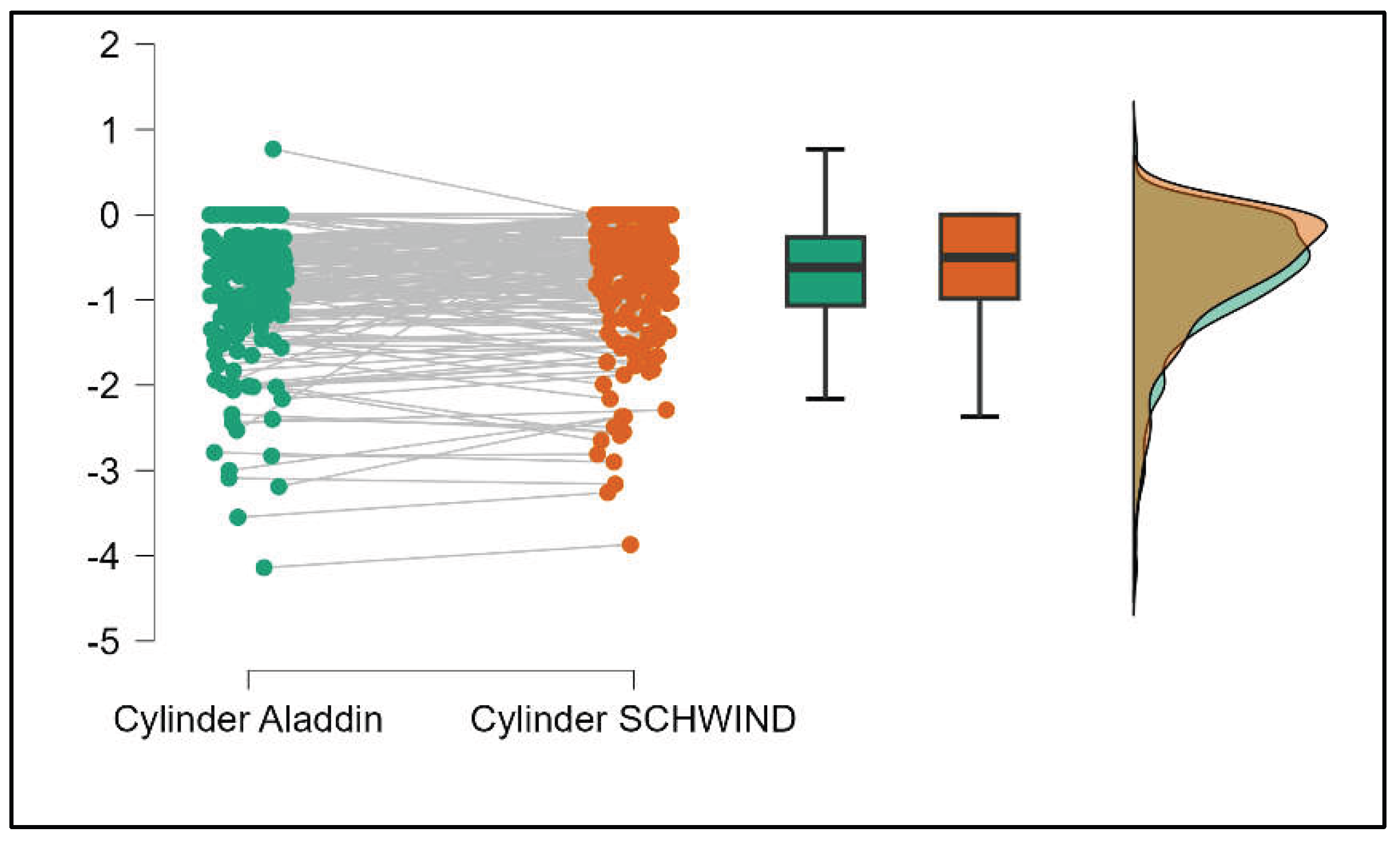
Table 4 shows statistically significant differences in K1, K2, Powerful Angle, and Cylinder measurements when comparing Aladdin and Schwind devices, with Aladdin consistently yielding higher values across these parameters (Supplemental
Figure 16,
Figure 17,
Figure 18 and
Figure 19). The average Powerful Angle values are significantly lower in the case of Schwind compared to Alladin (Mann-Whitney U test, p=0.009) and the average Cylinder values are significantly lower in the case of Schwind compared to Aladdin (Mann-Whitney U test, p=0.002).
4. Discussion
In cataract surgery, precise measurement of anterior segment parameters in conjunction with axial length is essential for accurate intraocular lens power calculation. Inaccurate measurements of axial length, keratometry, and anterior chamber depth have been identified as the primary sources of errors in intraocular lens power calculations. A 1 mm discrepancy in anterior chamber depth measurement results in postoperative refractive errors of approximately 1 D in myopic eyes, 1.5 D in emmetropic eyes, and 2.5 D in hypermetropic eyes. Likewise, a 0.1 D error in keratometry readings yields a corresponding refractive error of about 0.1 D [
9]. Importantly, recent comparative analyses of modern IOL power calculation formulas have shown that, despite advances in formula design, the accuracy of postoperative refractive outcomes remains highly dependent on the precision of the underlying biometric measurements, when using a toric intraocular lens, even minor inaccuracies in keratometric axis measurements can lead to incorrect axis orientation and suboptimal astigmatic correction [
10]. Given the critical importance of these parameters, evaluating the agreement between different biometry devices is essential for ensuring interchangeability and optimizing surgical planning, particularly in the context of advancing lens technologies and increasing patient expectations for precise refractive outcomes [
11].
In this study we assesed the agreement and interchangeability of biometric measurements obtained from three different machines, two optical biometers - Argos and Aladdin, and one Scheimpflug-Placido disk cornean topographer - Schwind Sirius. Our findings indicated statistically significant differences in various ocular parameters across these devices, challenging their direct interchangeability in some clinical contexts.
The statistically significant differences observed in keratometry values (K1, K2), powerful angle, cylinder, lens thickness, axial length, anterior chamber depth, and white-to-white measurements highlight the lack of interchangeability among the devices for these specific parameters, despite comparable central corneal thickness measurements [
12,
13]. This finding aligns with previous research indicating that while some parameters like central corneal thickness might show agreement, other anterior segment measurements and keratometric values often differ significantly between devices [
12,
14]. Specifically, previous studies have demonstrated that while modern biometers generally agree on axial length measurements, variations in keratometry and anterior chamber depth can lead to clinically relevant discrepancies in intraocular lens power calculations [
15]. For instance, studies comparing different swept-source optical coherence tomography biometers and Scheimpflug-based devices have consistently shown strong agreement in overall parameters, yet notable discrepancies in keratometry, necessitating careful consideration when selecting IOLs [
16]. Furthermore, some research has indicated that while optical biometers generally provide higher values for axial length and anterior chamber depth compared to ultrasound biometry, differences can still persist between various optical biometers for keratometry and corneal diameter [
17]. These differences underscore the critical need for clinicians to be aware of the specific characteristics and potential biases of each biometer used, especially when dealing with complex cases such as post-refractive surgery eyes or those with corneal opacities [
18].
The observed differences in keratometric measurements between the Argos, Aladdin, and Schwind Sirius devices can be attributed to the distinct underlying measurement principles and optical assumptions employed by each technology. The Argos SS-OCT biometer derives corneal curvature by integrating OCT-based anterior segment imaging with LED-based keratometry, whereas the Aladdin relies exclusively on Placido disk–based reflection over a predefined corneal zone. In contrast, the Schwind Sirius combines Placido-derived anterior surface data with Scheimpflug imaging, allowing for three-dimensional reconstruction of the cornea. These methodological differences likely account for the consistently higher keratometry values obtained with the optical biometers compared to the Scheimpflug-Placido system, a finding also reported in previous comparative studies [
12]. In line with our findings, [
19] reported excellent agreement between SS-OCT and OLCI biometers for axial length (ICC = 0.975), anterior chamber depth (ICC = 0.960), lens thickness (ICC = 0.951), and keratometry (K1 ICC = 0.921, K2 ICC = 0.927), but noted moderate to poor agreement for astigmatism axis, cylinder vectors J0 and J45, and white-to-white distance, emphasizing that interchangeability may be limited for parameters where precision is critical.
Despite statistically significant differences in keratometric magnitude and cylinder values, the absence of significant differences in the vector components J0 and J45 suggests a relatively preserved agreement in astigmatic orientation across devices. This finding is clinically relevant, as vector analysis has been shown to better represent astigmatic agreement than conventional cylinder and axis values alone [
8]. In addition, comparative analysis of refractive predictive accuracy showed that both SS-OCT and OLCI technologies deliver high refractive predictability in clinical practice, with more than 97% of eyes achieving postoperative spherical equivalent outcomes within ±1.00 D of target refraction and excellent uncorrected distance visual acuity regardless of biometer type, although subtle differences in lens thickness and anterior chamber depth prediction were observed [
20]. From a surgical planning perspective, this implies that while absolute keratometric values may not be interchangeable, the directional consistency of astigmatism remains largely comparable among devices, which may partially mitigate the clinical impact of measurement discrepancies when selecting toric intraocular lenses.
Measurements of axial length, anterior chamber depth, lens thickness, and white-to-white distance were consistently higher when obtained using the Argos SS-OCT biometer compared to the Aladdin OLCI device. These findings are consistent with previously reported data demonstrating systematic offsets between SS-OCT–based and OLCI-based biometers, likely related to differences in segmentation algorithms, refractive indices, and signal penetration depth [
11]. Given that modern intraocular lens power calculation formulas incorporate multiple biometric inputs, including lens thickness and anterior chamber depth, even small systematic differences may influence refractive prediction accuracy and should be considered when switching devices during preoperative assessment or postoperative follow-up [
21].
Central corneal thickness measurements showed no statistically significant differences between the Aladdin and Schwind Sirius devices, suggesting good agreement for this parameter across different imaging modalities. This observation supports previous reports indicating that CCT measurements tend to be more robust and less device-dependent than keratometric or anterior chamber parameters [
12]. As such, CCT values may be used interchangeably in routine clinical practice; however, caution remains warranted in cases requiring high precision, such as corneal refractive surgery or glaucoma risk assessment.
From a clinical perspective, the lack of full interchangeability among the evaluated devices highlights the importance of consistency in biometric assessment. Combining measurements obtained from different platforms may introduce cumulative errors in IOL power calculation, particularly in eyes undergoing toric or other premium IOL implantation or in patients with altered corneal anatomy [
7,
18]. Accordingly, clinicians should rely on a single, validated device throughout the preoperative workflow to minimize measurement-related variability and optimize refractive outcomes.
These findings support a “single-platform” approach to preoperative cataract assessment, particularly when refractive precision is critical. Although Argos and Aladdin produced similar mean keratometry values, statistically significant offsets in keratometry, cylinder, and especially anterior segment metrics used by modern formulas can compound into clinically relevant differences in lens selection, toric axis planning, and post-op refractive accuracy. Practically, clinicians should avoid mixing K/biometry inputs from different devices within the same calculation workflow; if device switching is unavoidable, consider re-deriving lens constants and repeating measurements to confirm stability, rather than assuming interchangeability.
4.1. Limitations
The limitations of this study include its retrospective design and restriction to eyes with uncomplicated cataract, which may limit generalizability to patients with corneal pathology or prior refractive surgery. Additionally, posterior corneal astigmatism was not directly compared across devices, an increasingly recognized contributor to total corneal power estimation. Future prospective studies incorporating postoperative refractive outcomes and agreement analyses such as Bland–Altman plots and intraclass correlation coefficients would further clarify the clinical relevance of the observed biometric differences.
5. Conclusions
In conclusion, significant differences were identified in multiple anterior segment and biometric parameters among the Argos SS-OCT biometer, Aladdin OLCI biometer, and Schwind Sirius Scheimpflug-Placido topographer. While certain parameters, such as central corneal thickness and astigmatic vector components, demonstrated acceptable agreement, others, including keratometry, axial length, anterior chamber depth, and white-to-white measurements, were not directly interchangeable. These findings emphasize the need for device-specific consistency in preoperative biometric evaluation to optimize intraocular lens power calculation and refractive outcomes in cataract surgery.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org.
Author Contributions
Conceptualization, L.A.B., C.R.; methodology, L.A.B., I.-I.Y., N.M., H.T.S.; software, M.M.; validation, I.-I.Y., N.M.; formal analysis, A.T., M.M.; investigation, L.A.B., I.-I.Y., N.M.; resources, C.R., H.T.S.; data curation, N.M., M.M.; writing—original draft preparation, L.A.B.; writing—review and editing, I.-I.Y., H.T.S., C.R.; visualization, M.M.; project administration, C.R.; supervision, C.R. All authors have read and agreed to the published version of the manuscript..
Funding
The article processing charge was paid by the Victor Babes University of Medicine and Pharmacy Timisoara.
Institutional Review Board Statement
The Local Commission of Ethics from the “Victor Babes” University of Medicine and Pharmacy from Timisoara, Romania operates under article 167 provisions of Law no. 95/2006, art. 28, chapter VIII of order 904/2006; with EU GCP Directives 2005/28/EC, International Conference of Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH); and with the Declaration of Helsinki—Recommendations Guiding Medical Doctors in Biomedical Research Involving Human Subjects.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study through the standard cataract surgery consent form, which included permission for the use of anonymized data in research.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
The authors used ChatGPT, an AI language model developed by OpenAI (San Francisco, CA, USA), to exclusively improve the manuscript’s language and readability. All the scientific content, interpretations, and conclusions are the original work of the authors.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| D |
Diopters |
| K |
Keratometry |
| LT |
Lens thickness |
| ACD |
Anterior Chamber Depth |
| WTW |
White-to-white |
| CCT |
Central corneal thickness |
| AL |
Axial Length |
| IOL |
Intraocular Lens |
| OLCI |
Optical Low-Coherence Interferometry |
| SS-OCT |
Swept-Source Optical Coherence Tomography |
References
- Hovanesian, J.A. Comparison of Preoperative Measurements with Intraoperative Aberrometry in Predicting Need for. Correction in Eyes with Low Astigmatism Undergoing Cataract Surgery. Clin. Ophthalmol. 2021, 15, 2189–2196. https://doi.org/10.2147/OPTH.S314618.
- Leung, C.K.; Weinreb, R.N. Anterior Chamber Angle Imaging with Optical Coherence Tomography. Eye 2011, 25, 261–267. [Google Scholar] [CrossRef]
- Huang, J.; Chen, H.; Li, Y.; Chen, Z.; Gao, R.; Yu, F.; Zhang, H.; Tang, X.; Wang, J. Comprehensive Comparison of Axial Length Measurement with Three Swept-Source OCT-Based Biometers and Partial Coherence Interferometry. J. Refract. Surg. 2019, 35, 115–120. [Google Scholar] [CrossRef]
- Kim, J.M.; Kang, M.S.; Jin, K.H. Comparison of Anterior Segment Measurements between Scheimpflug Camera and New Module of Optical Coherence Tomography. J. Korean Ophthalmol. Soc. 2018, 59, 613–621. [Google Scholar] [CrossRef]
- Khamar, P.; Shetty, R.; Narasimhan, R.; et al. Repeatability and Agreement of Corneal Topography Using Swept-Source OCT and Scheimpflug Technology. Indian J. Ophthalmol. 2023, 71, 168–174. [Google Scholar]
- Khan, M.I.; Muhtaseb, M. Keratometry in Cataract Surgery: Comparison between Optical Biometry and Standard Automated Keratometry. Clin. Exp. Ophthalmol. 2011, 39, 381–387. [Google Scholar] [CrossRef]
- Koch, D.D.; Ali, S.F.; Weikert, M.P.; Shirayama, M.; Jenkins, R.; Wang, L. Contribution of Posterior Corneal Astigmatism to Total Corneal Astigmatism. J. Cataract Refract. Surg. 2012, 38, 2080–2087. [Google Scholar] [CrossRef]
- Thibos, L.N.; Horner, D. Power Vector Analysis of the Optical Outcome of Refractive Surgery. J. Cataract Refract. Surg. 2001, 27, 80–85. [Google Scholar] [CrossRef]
- Olsen, T. Sources of Error in Intraocular Lens Power Calculation. J. Cataract Refract. Surg. 1992, 18, 125–129. [Google Scholar] [CrossRef]
- Lender, R.; Mirsky, D.; Greenberger, R.; et al. Evaluation of Three Biometric Devices: Ocular Parameters and Calculated Intraocular Lens Power. Sci. Rep. 2022, 12, 19478. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.Y.; Jun, I. Comparison of Anterior Segment Measurements with a New Multifunctional Unit and Five Other Devices. Korean J. Ophthalmol. 2022, 36, 338–349. [Google Scholar] [CrossRef]
- Telek, H.H.; Bilen, R.B.A.; Özdemir, Y.A.; et al. Comparison of Keratometric Values and Anterior Segment Pa-rameters Measured Using Scheimpflug Sirius Topography and Lenstar Biometry. Int. Ophthalmol. 2024, 44, 103. [Google Scholar] [CrossRef]
- Oh, R.; Oh, J.Y.; Choi, H.J.; Kim, M.K.; Yoon, C.H. Comparison of Ocular Biometric Measurements in Patients with Cataract Using Three Swept-Source Optical Coherence Tomography Devices. BMC Ophthalmol. 2021, 21, 62. [Google Scholar] [CrossRef]
- Özkan, H.; Ertunç, G.; Uğurlu, A.; et al. Comparison of Intraocular Lens Power and Biometry Measurements by Partial Coherence Interferometry, Swept-Source Optical Coherence Tomography, and Optical Low-Coherence Inter-ferometry. Res. Sq. Preprint 2023. [Google Scholar]
- Savini, G.; Hoffer, K.J.; Barboni, P.; Balducci, N.; Schiano-Lomoriello, D.; Ducoli, P. Accuracy of Optical Biometry Combined with Placido Disc Corneal Topography for Intraocular Lens Power Calculation. PLoS ONE 2017, 12, e0172634. [Google Scholar] [CrossRef]
- Yang, C.M.; Lim, D.H.; Kim, H.J.; Chung, T.-Y. Comparison of Two Swept-Source Optical Coherence Tomography Biometers and a Partial Coherence Interferometer. PLoS ONE 2019, 14, e0223114. [Google Scholar] [CrossRef]
- Yu, J.; Wen, D.; Zhao, J.; et al. Comprehensive Comparisons of Ocular Biometry: A Network-Based Big Data Analysis. Eye Vis. 2023, 10, 1. [Google Scholar] [CrossRef]
- Moon, J.Y.; Cho, S.C.; Kim, H.J.; Jun, R.M.; Han, K.E. Agreement between Two Swept-Source Optical Coherence Tomography Biometers and a Partial Coherence Interferometer. Korean J. Ophthalmol. 2022, 36, 326–337. [Google Scholar] [CrossRef]
- Munteanu, M.; Al Barri, L.; Stanca, S.; Mocanu, V.; Roşca, C.; Balica, N.C.; Stanca, H.T. Agreement in Biometric Parameters Between Swept-Source–Optical Coherence Tomography and Optical Low-Coherence Interferometry: Insights into Clinical Precision. J. Clin. Med. 2025, 14, 1407. [Google Scholar] [CrossRef]
- Al Barri, L.; Mercea, N.; Yasar, I.-I.; Munteanu, M.; Stanca, H.T. Evaluation of Refractive Predictive Accuracy in Intraocular Lens Power Calculations: A Comparative Study of Swept-Source Optical Coherence Tomography and Optical Low-Coherence Interferometry. J. Clin. Med. 2025, 14, 1201. [Google Scholar] [CrossRef]
- Yasar, I.-I.; Yasar, S.; Al Barri, L.; Dărăbuş, D.-M.; Tîrziu, A.-T.; Munteanu, M.; Stanca, H.T. Comparative Analysis of Intraocular Lens Power Calculation Formulas (Kane, Barrett Universal II, Hill–Radial Basis Function, and Ladas Super Formula): Which One Is More Accurate? J. Clin. Med. 2025, 14, 2443. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).