Submitted:
15 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
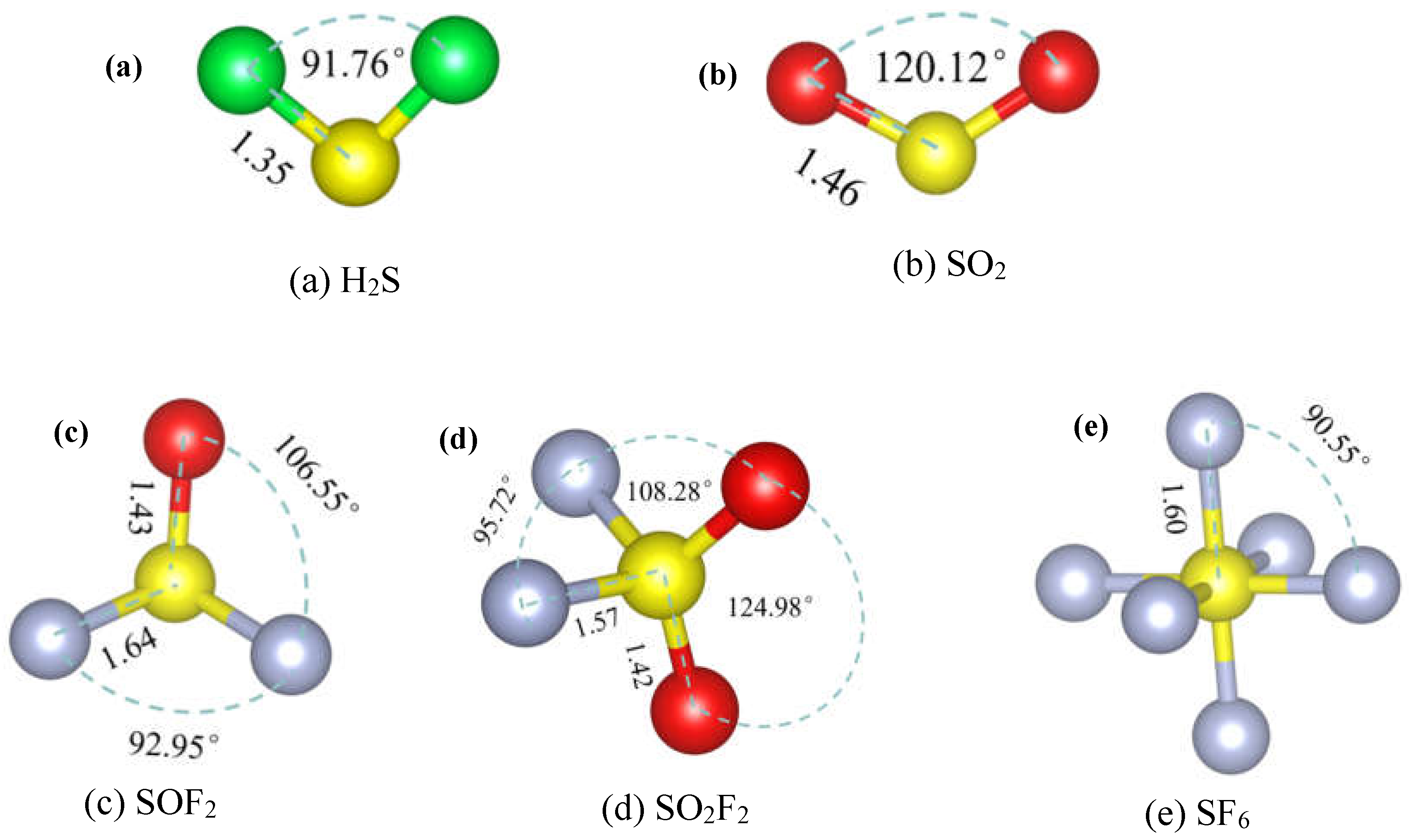
3. Results and Discussion
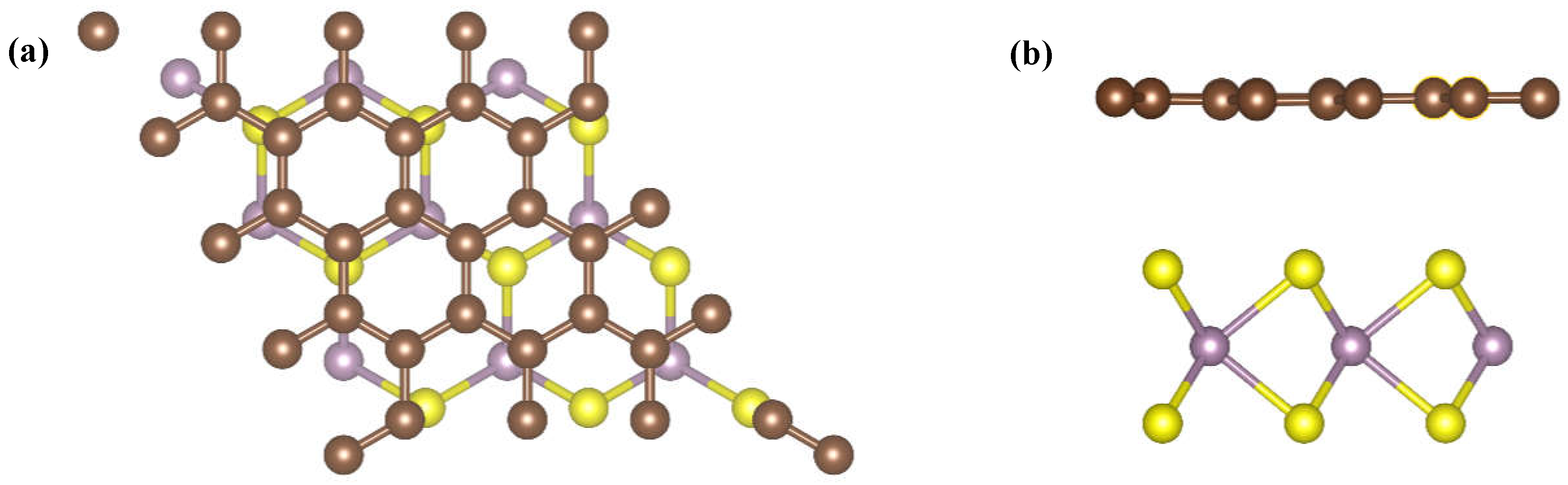
3.1. Adsorbing Characteristics of G/MoS2 Interface for Target Gases
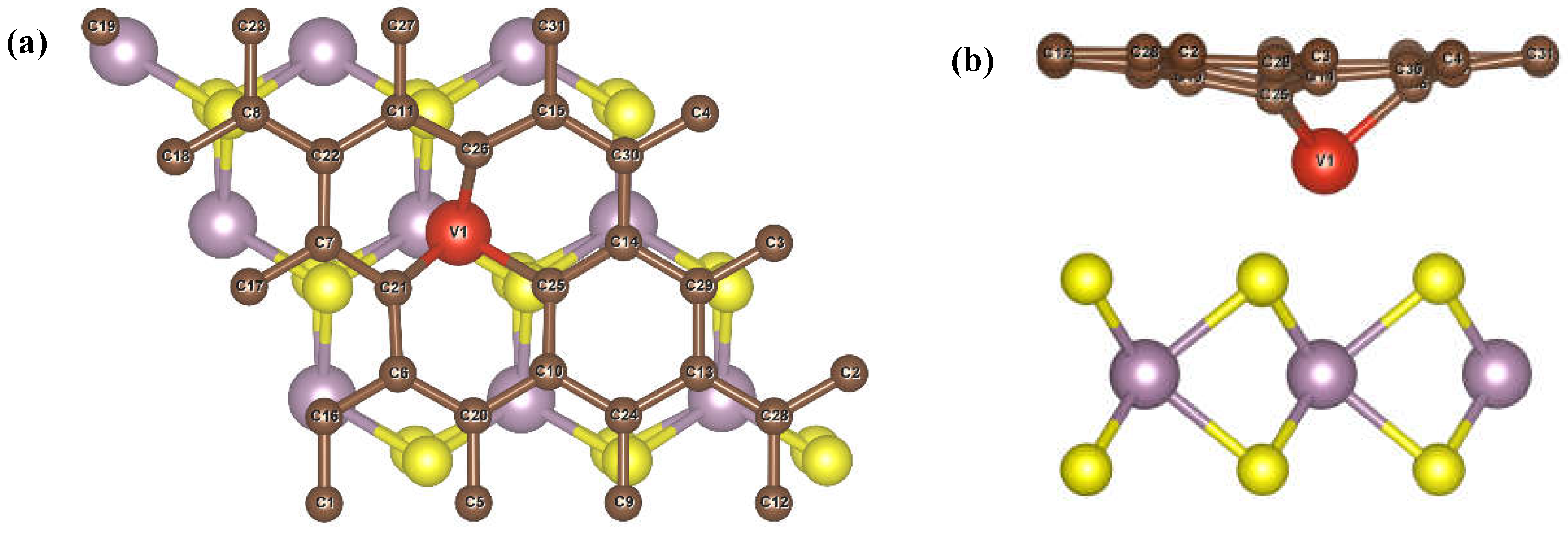
3.2. Adsorbing Characteristics of V-Doped G/MoS2 (GMV) for Target Gases
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, B.; et al. Analysis of the dielectric properties of R410A Gas as an alternative to SF6 for high-voltage applications. High Voltage 2019, 4, 41–48. [Google Scholar] [CrossRef]
- Tsai, W.-T. The decomposition products of sulfur hexafluoride (SF6): Reviews of environmental and health risk analysis. Journal of Fluorine Chemistry 2007, 128, 1345–1352. [Google Scholar] [CrossRef]
- Malik, N.; Qureshi, A. A Review of Electrical Breakdown in Mixtures of SF6 and Other Gases. IEEE Trans. Elect. Insul. 1979, EI-14, 1–13. [Google Scholar] [CrossRef]
- Xiao, S.; Zhang, X.; Tang, J.; Liu, S. A review on SF 6 substitute gases and research status of CF 3 I gases. Energy Reports 2018, 4, 486–496. [Google Scholar] [CrossRef]
- Vanbrunt, R. J. Production rates for oxyfluorides SOF2, SO2F2, and SOF4 in SF6 corona discharges. J. RES. NATL. BUR. STAN. 1985, 90, 229. [Google Scholar] [CrossRef]
- Dervos, C. T.; Vassiliou, P. Sulfur Hexafluoride (SF6 ): Global Environmental Effects and Toxic Byproduct Formation. Journal of the Air & Waste Management Association 2000, 50, 137–141. [Google Scholar]
- Dibeler, V. H.; Mohler, F. L. Dissociation of SF6, CF4, and SiF4 by electron impact. J. RES. NATL. BUR. STAN. 1948, 40, 25. [Google Scholar] [CrossRef]
- Zhong, L.; et al. Theoretical study of the chemical decomposition mechanism and model of Sulfur hexafluorid (SF6) under corona discharge. Journal of Fluorine Chemistry 2019, 220, 61–68. [Google Scholar] [CrossRef]
- Luo, C.; et al. Transition Metal (Co, V, W, Zr) Single-Atom Decorated Biphenylene for Enhancing the Sensing Performance of SF6 Decomposition Molecules. Langmuir 2024, 40, 9490–9500. [Google Scholar] [CrossRef]
- Zeng, F.; Li, H.; Cheng, H.; Tang, J.; Liu, Y. SF6 decomposition and insulation condition monitoring of GIE: A review. High Voltage 2021, 6, 955–966. [Google Scholar] [CrossRef]
- Zhang, W.; Gui, Y.; Yang, Y.; Tang, C. A hybrid nanogenerator based on wind energy harvesting for powering self-driven sensing systems. Journal of Cleaner Production 2023, 429, 139550. [Google Scholar] [CrossRef]
- Shao, Y.; et al. Core-sheath structured CNT@Ni-CNT fiber-based multifunctional fabric with high-sensitivity, wide-range strain sensing, and enhanced electromagnetic shielding absorption. Chemical Engineering Journal 2025, 512, 162358. [Google Scholar] [CrossRef]
- Wang, Y.; Gui, Y.; He, S.; Yang, J. Hybrid nanogenerator driven self-powered SO2F2 sensing system based on TiO2/Ni/C composites at room temperature. Sensors and Actuators B: Chemical 2023, 377, 133053. [Google Scholar] [CrossRef]
- Gui, Y.; Wang, Y.; He, S.; Yang, J. Self-powered smart agriculture real-time sensing device based on hybrid wind energy harvesting triboelectric-electromagnetic nanogenerator. Energy Conversion and Management 2022, 269, 116098. [Google Scholar] [CrossRef]
- Chen, Y.; et al. Integrated design and optimization of magnetically actuated self-sensing flexible gripper with fast response, high stability, and high payload-to-weight ratio. Sci. China Technol. Sci. 2025, 68, 1920206. [Google Scholar] [CrossRef]
- He, S.; Gui, Y.; Wang, Y.; Yang, J. A self-powered β-Ni(OH)2/MXene based ethanol sensor driven by an enhanced triboelectric nanogenerator based on β-Ni(OH)2@PVDF at room temperature. Nano Energy 2023, 107, 108132. [Google Scholar] [CrossRef]
- Ma, H.; et al. Humidity sensing characteristics of graphene and MoS2 as well as their heterostructures with different stacking configurations. [CrossRef]
- Cho, B.; et al. Chemical Sensing of 2D Graphene/MoS2 Heterostructure device. ACS Appl. Mater. Interfaces 2015, 7, 16775–16780. [Google Scholar] [CrossRef]
- Ghayyem, F.; Kiakojouri, A.; Frank, I.; Nadimi, E. Gas Sensing Properties of Graphene/MoS₂/Graphene Lateral Heterostructure: A First Principles Investigation. IEEE Sensors J. 2024, 24, 36334–36341. [Google Scholar] [CrossRef]
- Alzate-Carvajal, N.; Luican-Mayer, A. Functionalized Graphene Surfaces for Selective Gas Sensing. ACS Omega 2020, 5, 21320–21329. [Google Scholar] [CrossRef]
- Bag, A.; Lee, N.-E. Gas sensing with heterostructures based on two-dimensional nanostructured materials: a review. J. Mater. Chem. C 2019, 7, 13367–13383. [Google Scholar] [CrossRef]
- Baloglu, A. B.; et al. GRAPHENE/BN VAN DER WAALS HETEROSTRUCTURES FOR ENHANCED TOXIC GAS SENSING.
- Li, B.; Zhang, N.; Lei, Y.; Zhu, M.; Yang, H. Graphene/Chalcogenide Heterojunctions for Enhanced Electric-Field-Sensitive Dielectric Performance: Combining DFT and Experimental Study. Nanomaterials 2026, 16, 128. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; et al. van der Waals graphene/MoS2 heterostructures: tuning the electronic properties and Schottky barrier by applying a biaxial strain. Mater. Adv. 2022, 3, 624–631. [Google Scholar] [CrossRef]
- Foruzan, E.; Akmal, A. A. S.; Niayesh, K.; Lin, J.; Sharma, D. D. Comparative study on various dielectric barriers and their effect on breakdown voltage. High Voltage 2018, 3, 51–59. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. The Journal of Chemical Physics 2010, 132, 154104. [Google Scholar] [CrossRef]
- Hartmann, G.; Lee, M.; Hwang, G. S. Structural, electronic and adsorption properties of monolayer 2H-MoS2 on graphene substrates: A computational study. Inorganic Chemistry Communications 2019, 106, 135–138. [Google Scholar] [CrossRef]
- Wei, H.; Gui, Y.; Kang, J.; Wang, W.; Tang, C. A DFT Study on the Adsorption of H2S and SO2 on Ni Doped MoS2 Monolayer. Nanomaterials 2018, 8, 646. [Google Scholar] [CrossRef]
- Zou, Y.; et al. Charge Transfer in Graphene-MoS2 Vertical Heterostructures Tuned by Stacking Order and Substrate-Introduced Electric Field. ACS Appl. Mater. Interfaces 2024, 16, 30589–30597. [Google Scholar] [CrossRef]
- Cao, Q.; et al. Controllable Graphene/MoS2 Heterointerfaces by Perpendicular Surface Functionalization. Angew Chem Int Ed 2024, 63, e202415922. [Google Scholar] [CrossRef]
- Miao, H.; et al. DFT analysis of the sensitivity of graphene/MoS2 heterostructures toward H2CO. Vacuum 2023, 214, 112182. [Google Scholar] [CrossRef]
- Zhang, W.; et al. Effects of vertical strain and electrical field on electronic properties and Schottky contact of graphene/MoSe2 heterojunction. Journal of Physics and Chemistry of Solids 2021, 157, 110189. [Google Scholar] [CrossRef]
- Liu, X.; Li, Z. Electric Field and Strain Effect on Graphene-MoS2 Hybrid Structure: Ab Initio Calculations. J. Phys. Chem. Lett. 2015, 6, 3269–3275. [Google Scholar] [CrossRef]
- Pierucci, D.; et al. Band Alignment and Minigaps in Monolayer MoS2 -Graphene van der Waals Heterostructures. Nano Lett. 2016, 16, 4054–4061. [Google Scholar] [CrossRef]
- Li, B.; Zhou, Q.; Peng, R.; Liao, Y.; Zeng, W. Adsorption of SF6 decomposition gases (H2S, SO2, SOF2 and SO2F2) on Sc-doped MoS2 surface: A DFT study. Applied Surface Science 2021, 549, 149271. [Google Scholar] [CrossRef]
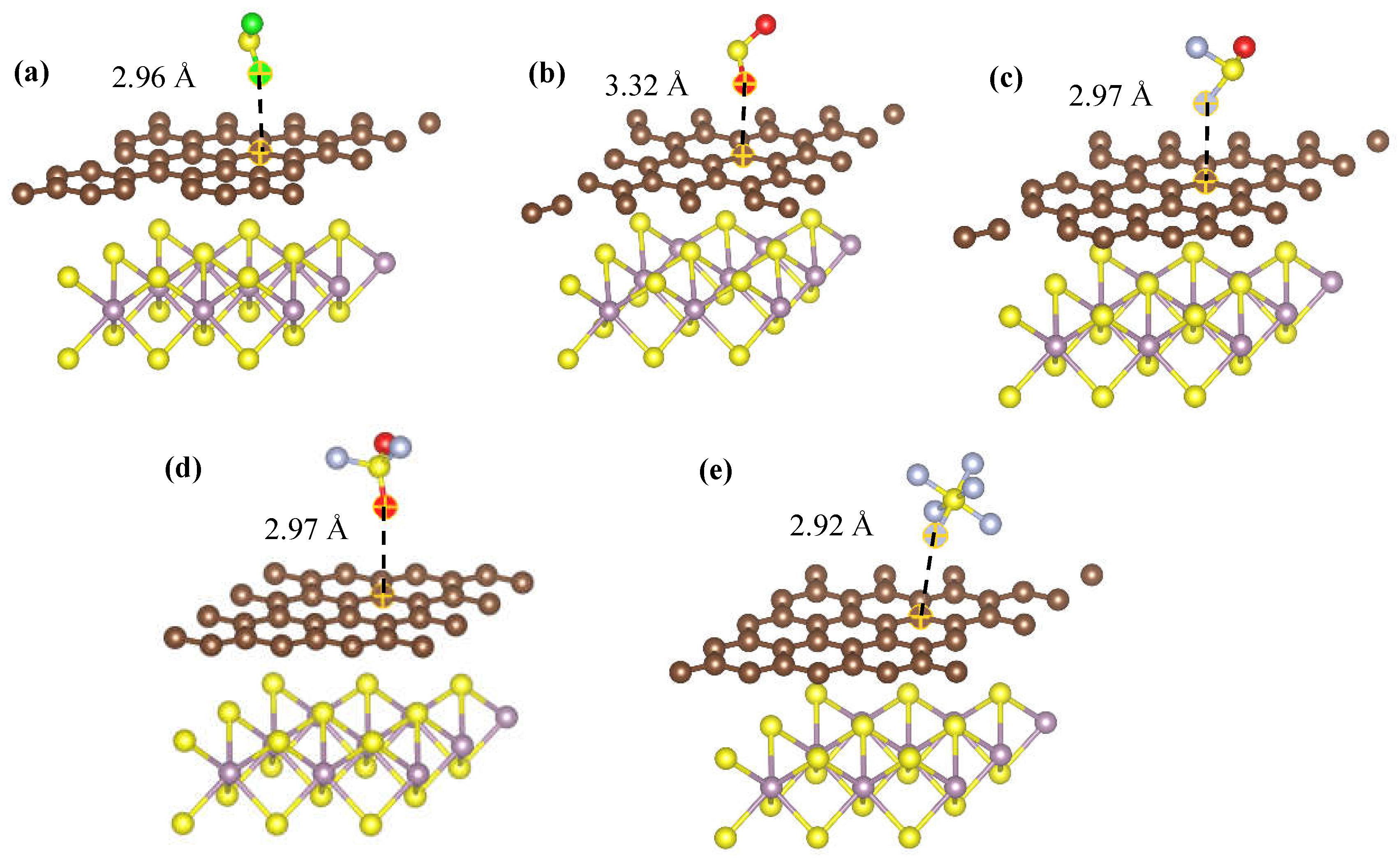
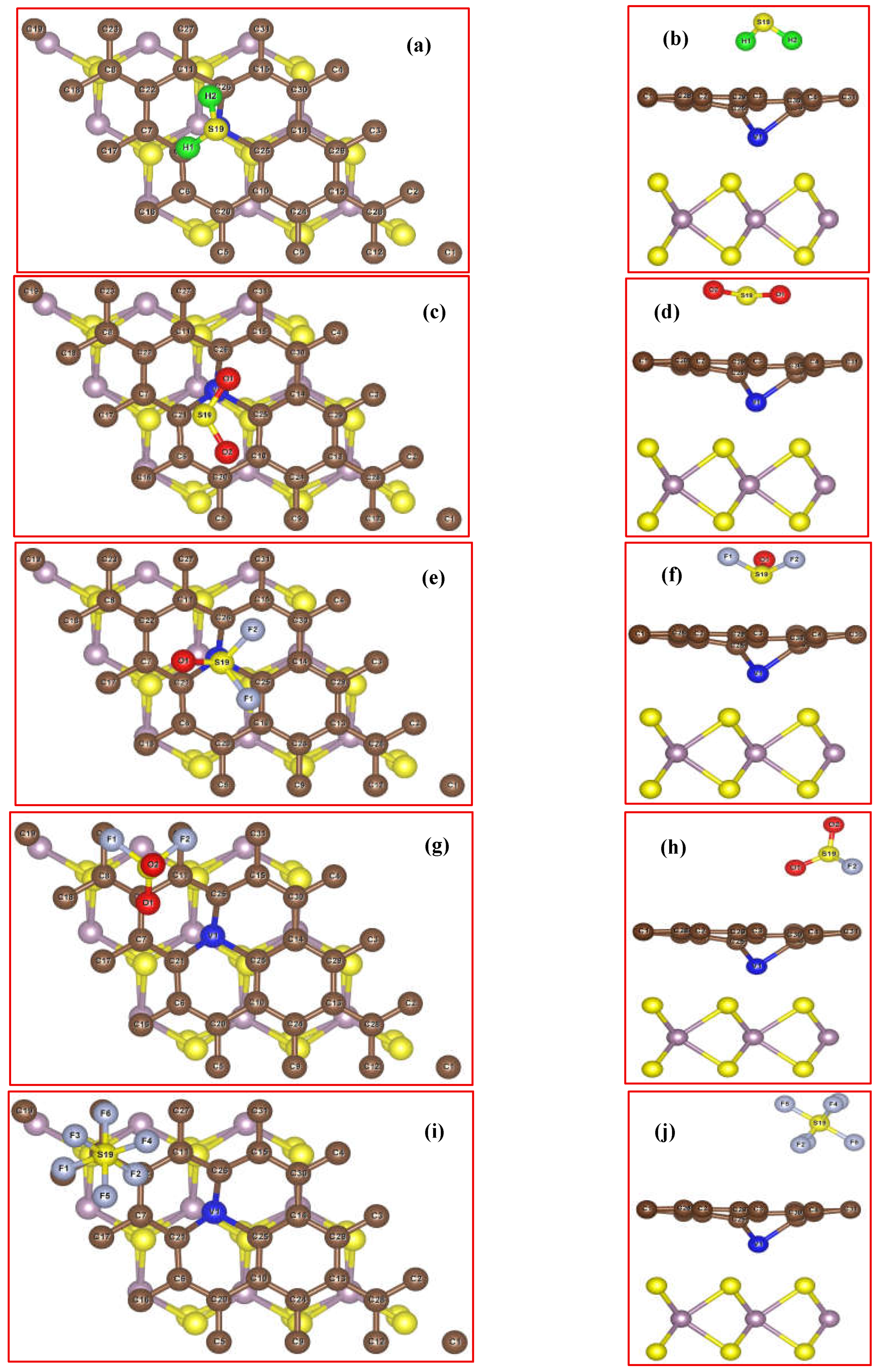
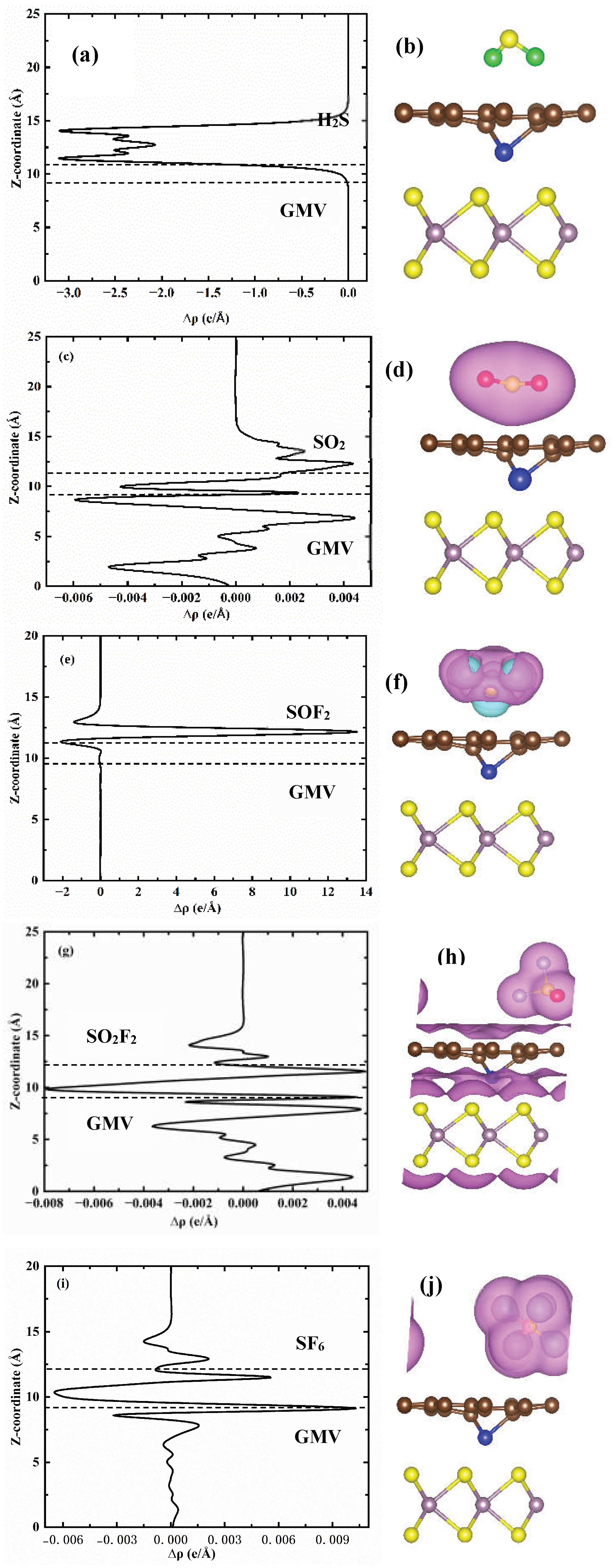
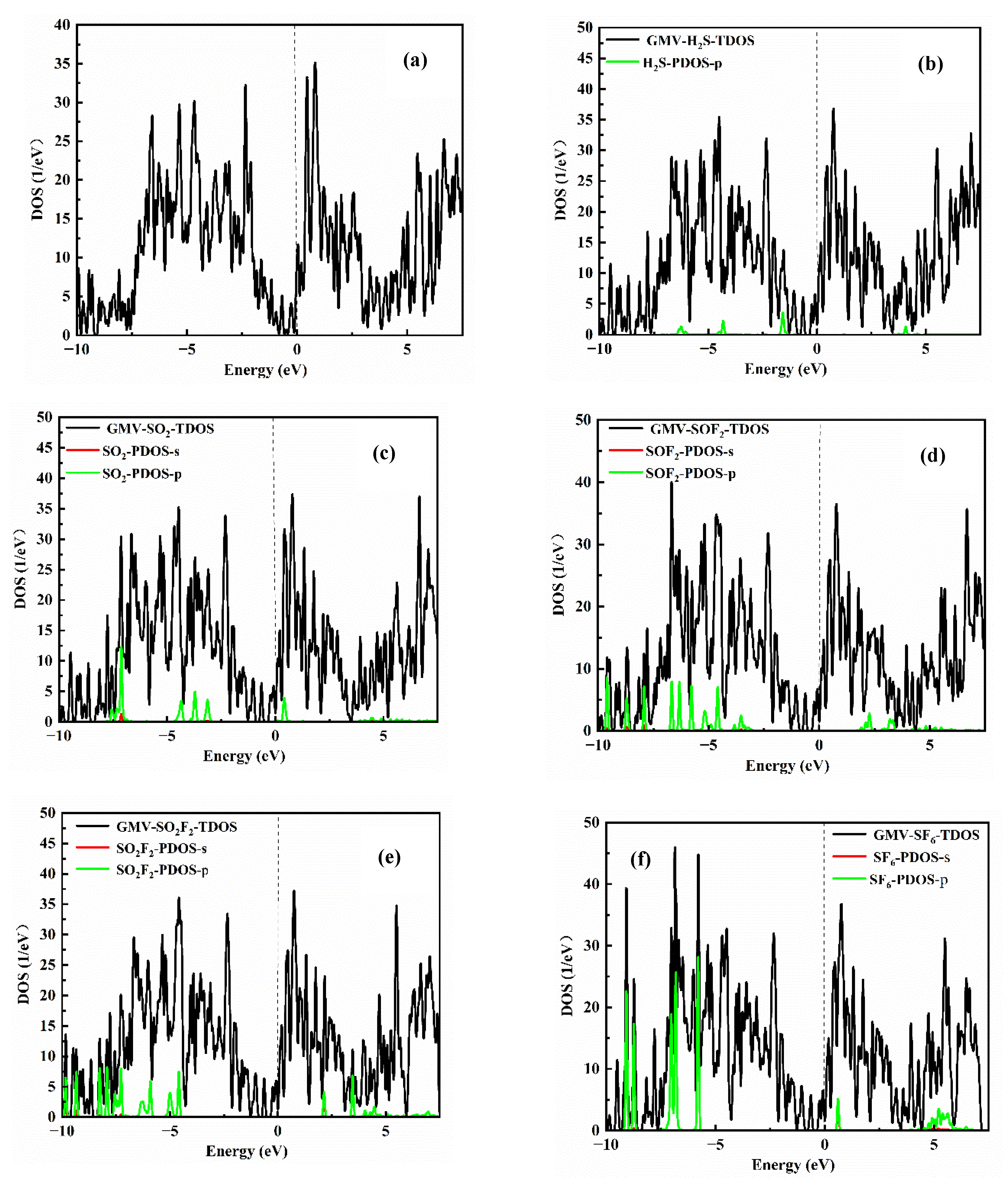
| Species | Ead/eV | D /Å | ∆Q/e |
|---|---|---|---|
| G/MoS2-H2S | -0.196 | 2.96 | -0.01 |
| G/MoS2-SO2 | -0.181 | 3.32 | -0.05 |
| G/MoS2-SOF2 | -0.141 | 2.97 | -0.01 |
| G/MoS2-SO2F2 | -0.164 | 2.97 | 0.02 |
| G/MoS2-SF6 | -0.241 | 2.92 | -0.02 |
| Species | Ead/eV | D /Å | ∆Q/e |
|---|---|---|---|
| GMV-H2S | -0.241 | 2.03 | 0.01 |
| GMV-SO2 | -0.380 | 2.34 | -0.06 |
| GMV-SOF2 | -0.317 | 2.18 | -0.027 |
| GMV-SO2F2 | -0.268 | 3.03 | -0.017 |
| GMV-SF6 | -0.224 | 2.85 | -0.021 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




