Submitted:
14 February 2026
Posted:
17 February 2026
You are already at the latest version
Abstract
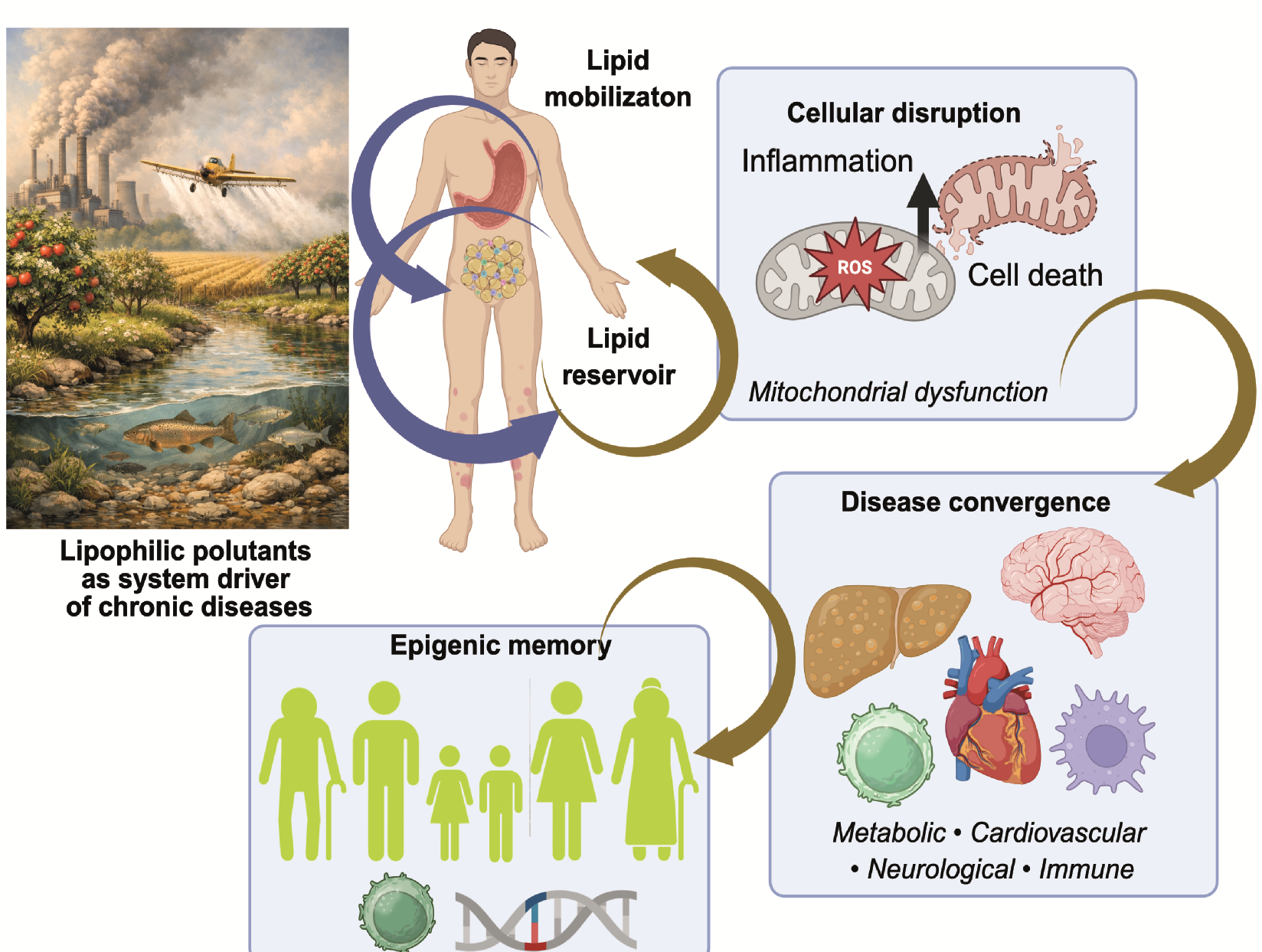
Keywords:
1. Introduction
2. Methods
3. Results
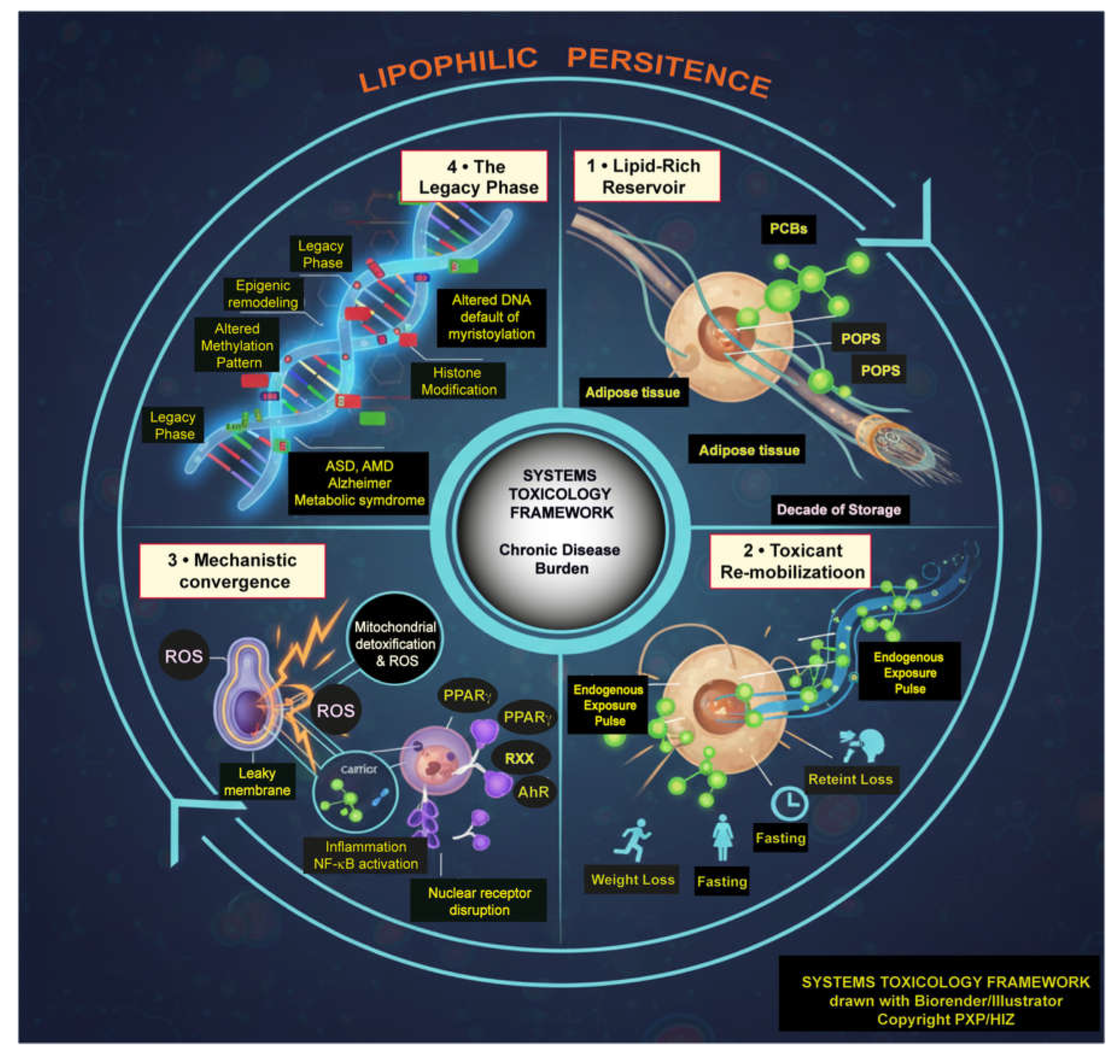
4. Discussion
5. Conclusion
Author Contributions
Funding
Acknowledgments
Competing Interests
Abbreviations
References
- Zeliger, HI. Toxic effects of chemical mixtures. Arch Environ Health 2003, 58, 23–9. [Google Scholar] [CrossRef]
- Lee, YM; Jacobs, DR, Jr.; Lee, DH. Persistent Organic Pollutants and Type 2 Diabetes: A Critical Review of Review Articles. Front Endocrinol (Lausanne) 2018, 9, 712. [Google Scholar] [CrossRef] [PubMed]
- Lind, PM; Lee, DH; Jacobs, DR; Salihovic, S; van Bavel, B; Wolff, MS; Lind, L. Circulating levels of persistent organic pollutants are related to retrospective assessment of life-time weight change. Chemosphere 2013, 90, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Rainey, NE; Saric, A; Leberre, A; Dewailly, E; Slomianny, C; Vial, G; Zeliger, HI; Petit, PX. Synergistic cellular effects including mitochondrial destabilization, autophagy and apoptosis following low-level exposure to a mixture of lipophilic persistent organic pollutants. Sci Rep 2017, 7, 4728. [Google Scholar] [CrossRef]
- Zeliger, HI. Lipophilic chemical exposure as a cause of cardiovascular disease. Interdiscip Toxicol 2013, 6, 55–62. [Google Scholar] [CrossRef]
- Zeliger, HI. Exposure to lipophilic chemicals as a cause of neurological impairments, neurodevelopmental disorders and neurodegenerative diseases. Interdiscip Toxicol 2013, 6, 103–10. [Google Scholar] [CrossRef]
- Lolescu, BM; Furdui-Linta, AV; Ilie, CA; Sturza, A; Zara, F; Muntean, DM; Blidisel, A; Cretu, OM. Adipose tissue as target of environmental toxicants: focus on mitochondrial dysfunction and oxidative inflammation in metabolic dysfunction-associated steatotic liver disease. Mol Cell Biochem 2025, 480, 2863–2879. [Google Scholar] [CrossRef] [PubMed]
- Zeliger, HI. Co-morditities of environmental diseases: A common cause. Interdiscip Toxicol 2014, 7, 117–22. [Google Scholar] [CrossRef]
- Heindel, JJ. Endocrine disruptors and the obesity epidemic. Toxicol Sci 2003, 76, 247–9. [Google Scholar] [CrossRef]
- Heindel, JJ; Newbold, R; Schug, TT. Endocrine disruptors and obesity. Nat Rev Endocrinol 2015, 11, 653–61. [Google Scholar] [CrossRef]
- Grandjean, P; Landrigan, PJ. Developmental neurotoxicity of industrial chemicals. Lancet 2006, 368, 2167–78. [Google Scholar] [CrossRef]
- Ing, C; Bellinger, DC. Long-term cognitive and behavioral outcomes following early exposure to general anesthetics. Curr Opin Anaesthesiol 2022, 35, 442–447. [Google Scholar] [CrossRef]
- Choi, MA; Rose, S; Langouet, S. Per- and polyfluoroalkyl substances as potentiators of hepatotoxicity in an exposome framework: Current challenges of environmental toxicology. Toxicology 2025, 515, 154167. [Google Scholar] [CrossRef]
- Lolescu, BM; Furdui-Linta, AV; Ilie, CA; Sturza, A; Zara, F; Muntean, DM; Blidisel, A; Cretu, OM. Correction to: Adipose tissue as target of environmental toxicants: focus on mitochondrial dysfunction and oxidative inflammation in metabolic dysfunction-associated steatotic liver disease. Mol Cell Biochem 2025, 480, 3927. [Google Scholar] [CrossRef]
- Grandjean, P; Kishi, R; Kogevinas, M. International Society for Environmental E Prevention of Developmental Neurotoxicity. Epidemiology 2017, 28, 157–158. [Google Scholar] [CrossRef] [PubMed]
- Forns, J; Stigum, H; Hoyer, BB; Sioen, I; Sovcikova, E; Nowack, N; Lopez-Espinosa, MJ; Guxens, M; Ibarluzea, J; Torrent, M; Wittsiepe, J; Govarts, E; Trnovec, T; Chevrier, C; Toft, G; Vrijheid, M; Iszatt, N; Eggesbo, M. Prenatal and postnatal exposure to persistent organic pollutants and attention-deficit and hyperactivity disorder: a pooled analysis of seven European birth cohort studies. Int J Epidemiol 2018, 47, 1082–1097. [Google Scholar] [CrossRef]
- Hamzavi, SF; Elahi Vahed, I; Samadi Shams, A; Nozari, F; Gamzeh Latava, B; Mardukhi, S; Sabaghi, B; Hosseini, ZS; Masoumi Shahr, EBZ; Ahrari, S; Keshavarzian, A; Rahmanian, M. Association between polychlorinated biphenyls and hypertension risk: a systematic review and meta-analysis. Front Cardiovasc Med 2025, 12, 1529431. [Google Scholar] [CrossRef]
- Lind, PM; Salihovic, S; Stubleski, J; Karrman, A; Lind, L. Association of Exposure to Persistent Organic Pollutants With Mortality Risk: An Analysis of Data From the Prospective Investigation of Vasculature in Uppsala Seniors (PIVUS) Study. JAMA Netw Open 2019, 2, e193070. [Google Scholar] [CrossRef]
- Zeliger, HI. Lipophilic chemical exposure as a cause of type 2 diabetes (T2D). Rev Environ Health 2013, 28, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Sjoberg Lind, Y; Lind, PM; Salihovic, S; van Bavel, B; Lind, L. Circulating levels of persistent organic pollutants (POPs) are associated with left ventricular systolic and diastolic dysfunction in the elderly. Environ Res 2013, 123, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Roos, V; Ronn, M; Salihovic, S; Lind, L; van Bavel, B; Kullberg, J; Johansson, L; Ahlstrom, H; Lind, PM. Circulating levels of persistent organic pollutants in relation to visceral and subcutaneous adipose tissue by abdominal MRI. Obesity (Silver Spring) 2013, 21, 413–8. [Google Scholar] [CrossRef]
- Reddy, SR; Bangeppagari, M; Lee, SJ. Immune-Epigenetic Effects of Environmental Pollutants: Mechanisms, Biomarkers, and Transgenerational Impact. Curr Issues Mol Biol 2025, 47. [Google Scholar] [CrossRef]
- Zeliger, HI; Lipinski, B. Physiochemical basis of human degenerative disease. Interdiscip Toxicol 2015, 8, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Zeliger, HI. Predicting disease onset in clinically healthy people. Interdiscip Toxicol 2016, 9, 39–54. [Google Scholar] [CrossRef] [PubMed]
- zeliger, HI. Oxidative stress indes (OSI). condensed questionnaire. Eur. J. Med. Sci 2020, 2, 163. [Google Scholar] [CrossRef]
- Lind, PM; van Bavel, B; Salihovic, S; Lind, L. Circulating levels of persistent organic pollutants (POPs) and carotid atherosclerosis in the elderly. Environ Health Perspect 2012, 120, 38–43. [Google Scholar] [CrossRef]
- Lind, PM; Salihovic, S; van Bavel, B; Lind, L. Circulating levels of perfluoroalkyl substances (PFASs) and carotid artery atherosclerosis. Environ Res 2017, 152, 157–164. [Google Scholar] [CrossRef]
- Lee, YM; Bae, SG; Lee, SH; Jacobs, DR, Jr.; Lee, DH. Persistent organic pollutants and hyperuricemia in the U.S. general population. Atherosclerosis 2013, 230, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P; Harari, R; Barr, DB; Debes, F. Pesticide exposure and stunting as independent predictors of neurobehavioral deficits in Ecuadorian school children. Pediatrics 2006, 117, e546-56. [Google Scholar] [CrossRef]
- Le Magueresse-Battistoni, B; Vidal, H; Naville, D. Lifelong consumption of low-dosed food pollutants and metabolic health. J Epidemiol Community Health 2015, 69, 512–5. [Google Scholar] [CrossRef]
- Zeliger, HI. Air pollution, obesity and disease. Euro J Med and Health Sci 2024, 6, 2093. [Google Scholar] [CrossRef]
- Le Magueresse-Battistoni, B; Multigner, L; Beausoleil, C; Rousselle, C. Effects of bisphenol A on metabolism and evidences of a mode of action mediated through endocrine disruption. Mol Cell Endocrinol 2018, 475, 74–91. [Google Scholar] [CrossRef]
- Pienavani, T. Imperfection. A natural history; MIT press: Cambridge Massachusetts, USA, 2022. [Google Scholar]
- Lind, L; Zethelius, B; Salihovic, S; van Bavel, B; Lind, PM. Circulating levels of perfluoroalkyl substances and prevalent diabetes in the elderly. Diabetologia 2014, 57, 473–9. [Google Scholar] [CrossRef]
- Bolognesi, G; Bacalini, MG; Pirazzini, C; Garagnani, P; Giuliani, C. Evolutionary Implications of Environmental Toxicant Exposure. Biomedicines 2022, 10. [Google Scholar] [CrossRef]
- Lind, L; Salihovic, S; Lampa, E; Lind, PM. Mixture effects of 30 environmental contaminants on incident metabolic syndrome-A prospective study. Environ Int 2017, 107, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Le Magueresse-Battistoni, B; Vidal, H; Naville, D. Environmental Pollutants and Metabolic Disorders: The Multi-Exposure Scenario of Life. Front Endocrinol (Lausanne) 2018, 9, 582. [Google Scholar] [CrossRef] [PubMed]
- Pezzatti, J; Boccard, J; Codesido, S; Gagnebin, Y; Joshi, A; Picard, D; Gonzalez-Ruiz, V; Rudaz, S. Implementation of liquid chromatography-high resolution mass spectrometry methods for untargeted metabolomic analyses of biological samples: A tutorial. Anal Chim Acta 2020, 1105, 28–44. [Google Scholar] [CrossRef] [PubMed]
- Ma, X; Gui, T; Yang, S; Jin, S; Qiao, J; Xie, Y; Wang, J; Ouyang, W; Song, G; Yi, X; Liu, C; Pan, M; Liu, W; Qiao, N; Dai, Y; Tao, Y; Xu, J; Yin, T; Fang, H; Mi, J; Chen, SJ. Metabolic biomarkers for predicting onset and severity of CAR-T therapy-induced cytokine release syndrome in multiple myeloma. In Front Med; 2025. [Google Scholar] [CrossRef]

| Pollutant Class |
Primary Target | Key Mechanisms & Health Outcomes | Refs |
|---|---|---|---|
| PFAS | Adipose tissue, Liver, CNS, Vascular system | Linked to Metabolic Syndrome, Type 2 Diabetes, and NAFLD/NASH. Strongly associated with hypertension, coronary artery disease, and stroke. Connected to neurodevelopmental disorders (ASD, ADHD). |
[1,2,3,5,7,10,11,14,15,16] |
| PCBs | Adipose tissue, Myelin sheaths, CNS | Associated with impaired cognitive function, reduced executive functioning, and neurodevelopmental disorders. Disrupts neuronal differentiation and synaptogenesis. Linked to hypertension. |
[7,11,15,16,17,18] |
| OCPs (Pesticides) | Adipose tissue, CNS | Readily integrate into cell membranes and neural lipid structures. Linked to neurodevelopmental impairments and cognitive deficits. |
[7,11,15,16] |
| PAHs | Vascular tissue, Adipose tissue | Mechanistically linked to atherogenesis, oxidized LDL formation, and plaque instability. Contributes to cardiovascular events and vascular inflammation. |
[2,19] |
| Phthalates & Bisphenols | Endocrine organs, CNS | Act as endocrine disruptors altering estrogen receptor-dependent metabolic pathways. Associated with ADHD, cognitive impairment, and behavioral dysfunction. |
[7,10,11,15,16] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




