1. Introduction
Orofacial clefts, encompassing cleft lip, cleft alveolus, and cleft palate, represent among the most prevalent congenital craniofacial anomalies worldwide, with a reported incidence ranging from 1:500 to 1:1000 live births depending on ethnicity and geographic region [
1,
2]. These malformations frequently involve the alveolar process of the maxilla, resulting in discontinuity of the dental arch, impaired tooth eruption, and compromised maxillofacial growth [
2]. The functional and psychosocial burden of alveolar clefts is substantial, affecting mastication, speech, facial esthetics, and quality of life across childhood and adulthood [
3].
A defining characteristic of alveolar clefts is the absence or insufficiency of bone in the cleft region, which hinders normal dental development and orthodontic alignment [
4]. Hypodontia is common, particularly involving the maxillary lateral incisors adjacent to the cleft, further complicating oral rehabilitation [
3,
4]. Consequently, closure of the alveolar defect through alveolar bone grafting (ABG) has become an integral component of comprehensive cleft care, enabling canine eruption, orthodontic tooth movement, and subsequent prosthodontic rehabilitation [
2]. Secondary alveolar bone grafting, typically performed during the mixed dentition stage when the maxillary canine root is approximately two-thirds formed [
1,
5]. The iliac crest donor site, remains the gold standard due to its osteogenic, osteoinductive, and osteoconductive properties [
6,
7]. Despite its widespread use and generally favorable outcomes, ABG is not devoid of limitations [
4,
5,
6,
7,
8]. Post-grafting bone resorption is well documented, and a significant proportion of patients demonstrate inadequate bone volume at skeletal maturity, necessitating secondary or tertiary grafting procedures prior to definitive prosthetic rehabilitation [
4,
5,
6,
7,
8]. These limitations have direct implications for subsequent implant placement, as the quality and quantity of grafted bone are critical determinants of osseointegration and long-term implant survival [
9].
Dental implant–based rehabilitation has emerged as a preferred treatment modality in cleft patiens [
10] Compared with conventional fixed or removable prostheses, implants offer several advantages, including preservation of adjacent tooth structure, maintenance of alveolar bone through functional loading, and improved esthetic outcomes [
9,
10].
Nevertheless, implant therapy in cleft patients presents unique biological and technical challenges [
9]. Implants are frequently placed in previously grafted bone characterized by reduced volume, altered density, and scarred soft tissue envelopes, all of which may compromise primary stability and peri-implant tissue health [
10]. Furthermore, the prolonged interval between childhood grafting and adult implant placement may exacerbate graft resorption, occasionally requiring additional augmentation procedures such as guided bone regeneration at the time of implant insertion [
3,
4,
5].
Available evidence indicates that dental implants placed in grafted alveolar cleft regions demonstrate favorable survival outcomes, with most studies reporting survival rates above 90% during short to medium-term follow-up [
5,
6,
10]. However, interpretation of these results should be approached with caution, as the existing literature is largely based on retrospective designs, heterogeneous treatment protocols, and limited long-term follow-up data [
8,
9,
10]. While many authors support implant therapy as a reliable rehabilitative option, the overall quality of evidence remains limited.
The aim of this systematic review is to critically evaluate the existing clinical evidence regarding the survival and success rate of dental implants placed in grafted alveolar cleft regions in patients with cleft lip and/or palate. The review will synthesize all available evidence to evaluate the predictability and limitations of implant-supported rehabilitation in grafted alveolar cleft sites and to identify gaps in the current literature requiring further high-quality research.
2. Materials and Methods
2.1. Study Design and Reporting Guidelines
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) guidelines. The review protocol was developed a priori and followed a predefined research question, eligibility criteria, and methodological framework to minimize selection bias and enhance transparency.
2.2. Focused Question and PICO Framework
The review was structured using the Population, Intervention, Comparison, and Outcome (PICO) framework.
Population: Patients with congenital alveolar clefts associated with cleft lip and/or palate
Intervention: Placement of dental implants in previously grafted alveolar cleft sites
Comparison: Not mandatory; when available, comparisons between different grafting protocols, defect morphologies, or implant approaches were considered
Outcomes: Primary outcome was dental implant survival. Secondary outcomes included causes of implant loss and reported implant-related complications
The focused research question was:
What are the survival rates and reported causes of implant loss for dental implants placed in grafted alveolar cleft sites?
2.3. Eligibility Criteria
Inclusion Criteria
Studies were included if they met all of the following criteria:
Human clinical studies involving patients with alveolar clefts
Alveolar bone grafting performed prior to implant placement
Dental implants placed in the grafted cleft region
Implant survival reported as an outcome
Minimum follow-up of 12 months after implant placement
Prospective or retrospective cohort studies and case series more than 10 patients
Articles published in the English language
Exclusion Criteria
Studies were excluded if they met any of the following criteria:
Case reports or case series with fewer than 10 patients
Animal, in vitro, finite element, or cadaveric studies
Studies focusing exclusively on bone grafting or augmentation without implant survival outcomes
Studies reporting short-term osseointegration only, without survival data
Mixed populations in which cleft-specific implant outcomes could not be extracted
Reviews, editorials, technical notes, or conference abstracts
2.4. Information Sources and Search Strategy
A comprehensive electronic literature search was performed in the following databases:
PubMed/MEDLINE
Web of Science
Wiley Online Library
The search covered all records from database inception to the most recent available publications. The search strategy combined Medical Subject Headings (MeSH) and free-text terms related to alveolar clefts, bone grafting, and dental implants. They keywords used were: cleft, cleft lip, cleft palate, cleft alveolous, dental implants, dental implant.
Reference lists of included full-text articles were also manually screened to identify additional relevant studies.
2.5. Study Selection Process
All records retrieved from the electronic searches were imported into Rayyan systematic review software, where duplicate records were removed. Title and abstract screening was performed initially to exclude clearly irrelevant studies. Full-text articles of potentially eligible studies were then assessed independently against the predefined inclusion and exclusion criteria.
Discrepancies during full-text screening were resolved through discussion and consensus. Reasons for full-text exclusion were documented systematically and are presented in a dedicated exclusion table in accordance with PRISMA recommendations.
2.6. Data Extraction
Data extraction was performed using a standardized data extraction form. The following variables were collected from each included study:
Study design and year of publication
Number of patients and implants
Type of cleft deformity
Grafting technique and augmentation approach
Timing of implant placement relative to grafting
Duration of follow-up
Implant survival rate
Number and timing of implant failures
Reported causes of implant loss
Only complications explicitly associated with implant loss were recorded. If a study did not report the cause of implant failure, this was noted as “not specified.” Any discrepancies in data extraction were resolved by consensus.
2.7. Risk of Bias Assessment
The methodological quality of the included studies was assessed using an adapted Newcastle–Ottawa Scale (NOS) for observational studies. The assessment focused on three domains: selection of the study population, comparability of cohorts, and outcome assessment. Selection bias was evaluated based on the representativeness of the study sample and the adequacy of patient inclusion criteria. Comparability assessed the extent to which potential confounding factors (e.g., patient characteristics, grafting protocols, implant variables) were considered. Outcome assessment included the definition of implant failure, methods of outcome measurement, and adequacy of follow-up. Each study was categorized as having low, moderate, or high risk of bias according to its overall NOS-based methodological quality. Disagreements were resolved through discussion and consensus.
2.8. Statistical Analysis
Implant survival was analyzed at the implant level using data extracted from the included studies. The number of surviving implants was calculated from the reported number of implants placed and survival rates at the last follow-up. Pooled estimates were calculated using a random-effects meta-analysis of proportions with logit transformation, and results were back-transformed for interpretation. Heterogeneity was assessed using the I² statistic. Forest plots were constructed to present study-specific estimates and pooled effects. Prespecified subgroup analyses were performed according to implant timing (delayed, simultaneous, mixed) and use of guided bone regeneration (GBR reported vs not reported). Differences between subgroups were assessed using the Q test, with statistical significance set at p < 0.05.
For temporal comparison, included studies were categorized into an early era (1997–2008) and a modern era (2010–2026). Weighted pooled survival rates were calculated for each subgroup based on the total number of implants placed and surviving. Ninety-five percent confidence intervals (95% CIs) were computed using the Wald method for binomial proportions. Differences between survival proportions were assessed using a two-proportion Z-test, with statistical significance defined as p < 0.05. This analysis was conducted as a descriptive pooled comparison and did not account for inter-study heterogeneity.
3. Results
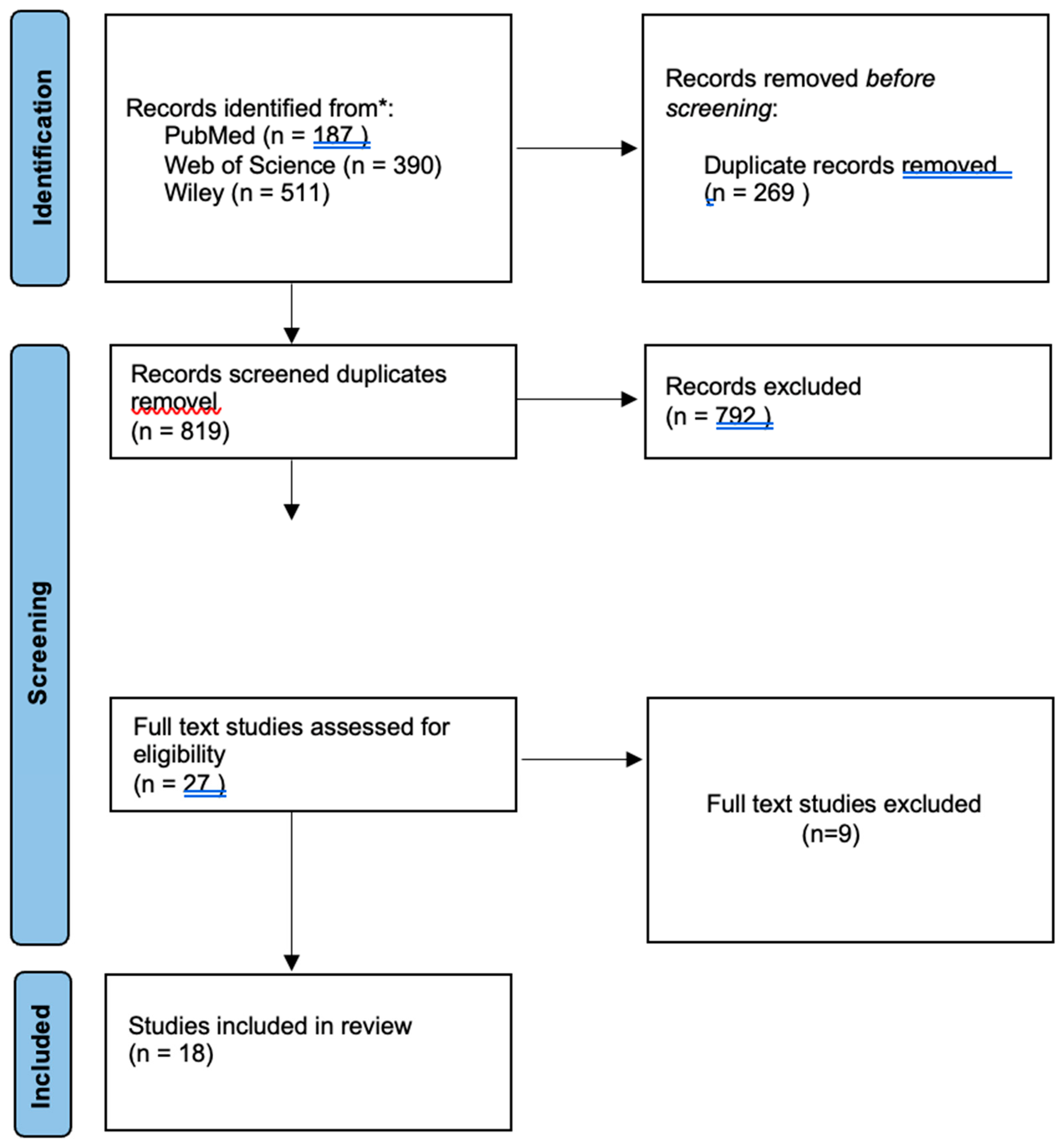
3.1. Study Selection
The electronic database search identified 1088 records through PubMed, Web of Science, Cochrrane Library and Wiley databases. After removal of duplicates, 819 unique records remained and were screened based on titles and abstracts. Following this initial screening, 792 records were excluded for not meeting the predefined inclusion criteria. A total of 27 full-text articles were assessed for eligibility. Of these, 9 studies were excluded after full-text review for the following reasons: non-clinical study design, absence of dental implant placement in grafted alveolar cleft sites, lack of implant survival or success outcomes, insufficient follow-up duration, small case series, mixed populations with non-separable cleft data, or outcomes not aligned with the objectives of the review (
Table 1). Ultimately, 18 studies fulfilled the inclusion criteria and were included in the qualitative synthesis of this systematic review (
Figure 1).
3.2. Overall Implant Survival
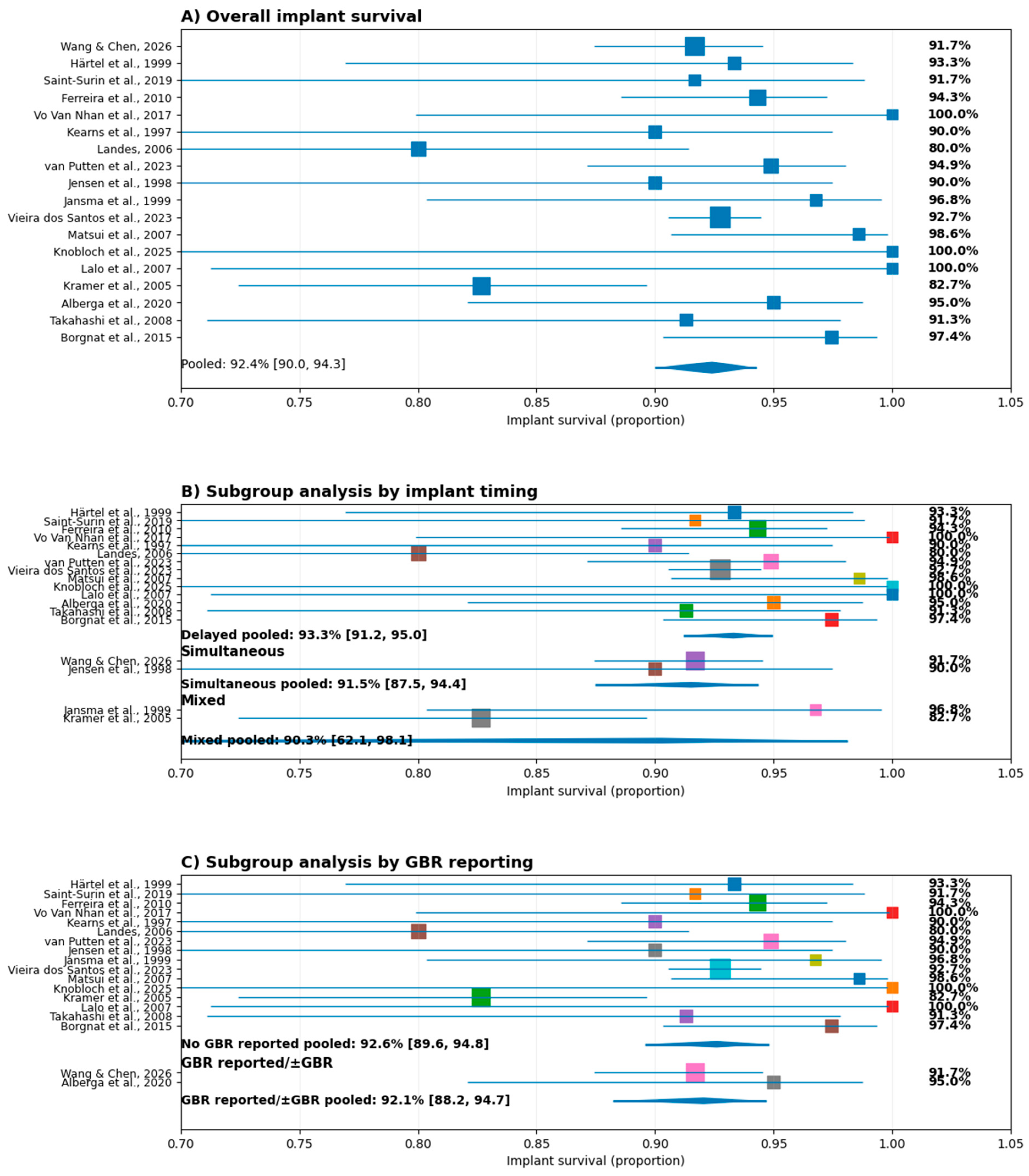
Across the included studies, implant survival in grafted alveolar cleft sites ranged from 80% to 100%, with the majority of contemporary studies reporting survival rates exceeding 90%.
A total of 18 studies were included in the quantitative synthesis of implant survival (
Figure 2A). The pooled survival rate was 92.4% (95% CI: 90.0–94.3), indicating a high probability of implant survival across diverse clinical settings and study designs. Individual study estimates ranged from 80.0% to 100%, with most studies reporting survival above 90%. Several investigations demonstrated survival rates of ≥98%, while a limited number reported lower values (80.0–82.7%). Despite this variability, confidence intervals largely overlapped, and the pooled estimate remained stable, suggesting limited heterogeneity and a consistent treatment effect across the included literature.
Early foundational studies from the late 1990s [
20,
21,
22,
23] demonstrated survival rates of approximately 90% following delayed implant placement in iliac crest–grafted clefts, with failures predominantly occurring during the early osseointegration phase. Subsequent investigations in the 2000s [
24,
25,
26,
27,
28,
29] reported comparable or improved outcomes, although survival rates as low as 80–82% were observed in cohorts involving short implants, simultaneous placement protocols, or compromised graft volume.
More recent studies (2015–2026) [
30,
31,
32,
33,
34,
35,
36,
37] consistently demonstrated survival rates between 91% and 100%, reflecting advances in surgical technique, grafting protocols, implant design, and prosthetic planning. Late implant loss was uncommon; most failures occurred within the first year after placement and were attributed to insufficient graft volume, inadequate primary stability, or early osseointegration failure. Peri-implantitis was reported as a cause of late failure in only a limited number of cases (
Table 2 and
Table 3).
The large retrospective cohort by Vieira dos Santos et al. [
35] reported a survival rate of 92.73% across 688 implants, reinforcing the reproducibility of favorable outcomes in cleft populations when appropriate surgical staging is employed. Similarly, morphology-guided augmentation strategies demonstrated survival rates of up to 98.3% at 12 months, suggesting that defect configuration may influence short-term implant stability.
When pooled by era, weighted implant survival increased from 91.2% (95% CI: 87.9%–94.5%) in early studies (1997–2008) to 94.2% (95% CI: 92.9%–95.5%) in modern studies (2010–2026), representing an absolute difference of 3.0% that reached statistical significance (two-proportion Z-test, p = 0.038).
Random-effects meta-analysis confirmed a pooled implant survival rate of 92.4% (95% CI: 90.0–94.3), with moderate heterogeneity across studies (I² = 33.5%) (
Figure 2A).
Subgroup Analysis by Implant Timing
Subgroup analysis according to implant placement timing (
Figure 2B) showed that delayed placement was the most frequently adopted approach (k = 14; n = 1,257) and yielded a pooled survival rate of 93.3% (95% CI: 91.2–95.0). Simultaneous placement was reported in fewer studies (k = 2; n = 260) and demonstrated a pooled survival of 91.5% (95% CI: 87.5–94.4).
Studies categorized as mixed timing showed a pooled survival of 90.3% (95% CI: 62.1–98.1), with wider confidence intervals reflecting the limited number of studies and smaller sample sizes. These studies were not included in direct subgroup comparisons due to methodological heterogeneity.
Overall, the overlapping confidence intervals and similar point estimates indicate that implant survival did not differ significantly between delayed and simultaneous placement strategies. The slightly higher point estimate observed for delayed placement is unlikely to be clinically meaningful.
Subgroup Analysis by GBR Reporting
Subgroup analysis based on the reporting or use of guided bone regeneration (GBR) is presented in
Figure 2C. Studies without GBR reporting constituted the majority of the dataset (k = 16) and demonstrated a pooled survival rate of 92.6% (95% CI: 89.6–94.8).
In contrast, studies explicitly reporting GBR use (k = 2) showed a pooled survival of 92.1% (95% CI: 88.2–94.7). The pooled estimates were nearly identical, and the confidence intervals showed substantial overlap, indicating no statistically significant subgroup effect. These findings suggest that the use or reporting of GBR did not materially influence implant survival outcomes in the analyzed studies.
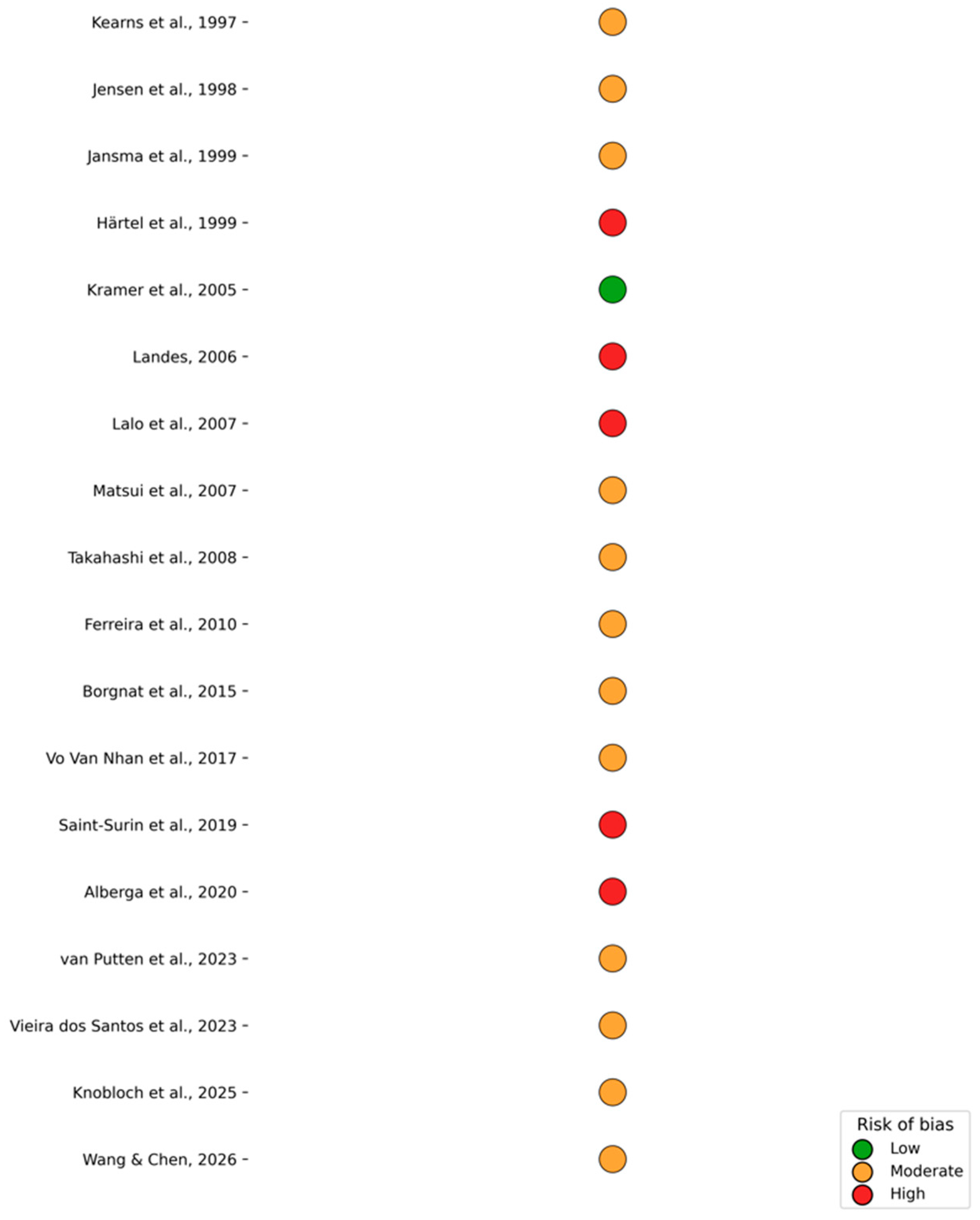
3.4. Risk of Bias Assessment
According to the Newcastle–Ottawa Scale, most included studies were judged to have a moderate risk of bias, primarily due to retrospective designs and limited adjustment for confounding factors. A small number of case series were classified as high risk, while only one study achieved a low-risk rating. Overall, the methodological quality of the evidence was considered acceptable for observational implant research (
Figure 3).
4. Discussion
The present systematic review demonstrates a pooled weighted implant survival of 94.2% in the modern era (2010–2026) compared to 91.2% in early-era studies (1997–2008), representing a statistically significant temporal improvement (p = 0.038). These findings confirm that implant-supported rehabilitation in grafted alveolar cleft sites has become increasingly predictable over time and likely reflect cumulative advances in surgical techniques, grafting protocols, implant design, and multidisciplinary treatment planning. The overall pooled survival of 92.4% further supports the reliability of implant therapy in this anatomically complex population, despite the biological challenges associated with grafted bone and scarred soft tissues.
These results align broadly with previously published systematic reviews while providing additional resolution through chronological stratification and explicit analysis of implant failure patterns. Pathak et al. [
9] reported a pooled survival rate of 93.5% irrespective of graft type, closely approximating the pooled value identified in the present review and reinforcing the predictability of implant therapy in grafted cleft sites. However, their meta-analysis did not stratify outcomes temporally or distinguish early from late implant loss, and the inclusion of esthetic and patient-reported outcomes broadened the scope but limited survival-specific interpretation. Similarly, Sales et al. [
5] identified a survival rate of approximately 93% across 483 implants with a mean follow-up of around 60 months, though their review highlighted significant heterogeneity and the limited availability of prospective studies—limitations that remain evident in the current evidence base.
Wang et al. [
3] reported a mean survival of 91.5% ± 4.77% with a mean follow-up of 54 months, a figure that closely mirrors the early-era pooled survival identified in this analysis. This parallel suggests that improvements observed in contemporary studies likely reflect technological and protocol advancements rather than differences in patient populations. Importantly, Wang et al. also emphasized the high rate of secondary or tertiary grafting, reported in up to 43.1% of cases, reinforcing the role of graft stability, maturation, and surgical staging as key determinants of implant prognosis. In contrast, Wermker et al. [
4] reported 5-year survival rates ranging from 80% to 96% (mean 88.6%), lower than more recent pooled estimates, likely due to the inclusion of earlier implant systems, machined surfaces, and less standardized grafting and timing protocols. Their review also highlighted the generally low methodological quality of the available literature, a concern that persists across cleft implant research.
The influence of graft maturation and timing is further supported by Mallick et al. [
6], who reported success rates between 95% and 100% when tertiary graft healing intervals of 3–6 months were respected. These findings support the hypothesis that optimized timing and controlled healing significantly contribute to improved outcomes observed in modern cohorts. Conversely, Vuletić et al. [
2] provided a narrative overview of grafting protocols and implant rehabilitation without pooled survival synthesis, and Guo et al. [
1] focused on secondary bone grafting techniques in children without evaluating implant survival, although both works underscore the foundational importance of graft quality prior to implant placement.
Across all reviews, including the present analysis, implant survival in grafted alveolar cleft sites consistently approaches outcomes observed in non-cleft implant populations. Modern survival rates, frequently exceeding 94%, now approximate general implant benchmarks reported in broader implant literature [
29,
30,
31,
32,
33,
34,
35,
36,
37]. Nevertheless, survival alone does not fully reflect treatment complexity. High rates of tertiary grafting and staged interventions indicate that favorable outcomes are often dependent on additional augmentation procedures and multidisciplinary coordination. Implant prognosis in cleft patients must therefore be interpreted within the context of surgical burden, orthodontic preparation, and prosthetic planning rather than survival metrics alone [
29,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39].
The pattern of implant failure identified across studies provides additional clinical insight. Failures occurred predominantly during the early osseointegration phase and were most frequently associated with insufficient graft volume, inadequate primary stability, or early biological failure, whereas late complications such as peri-implantitis were comparatively uncommon. This suggests that implant success in cleft patients is primarily determined by surgical and graft-related factors rather than long-term peri-implant tissue breakdown. Once osseointegration is achieved, implant behavior in grafted cleft bone appears similar to that observed in native maxillary bone.
Despite encouraging survival rates, the current evidence base remains methodologically limited. Consistent with prior reviews, the majority of included studies are retrospective case series with moderate-to-high risk of bias and heterogeneous reporting standards. Variability in follow-up duration, implant systems, grafting techniques, and definitions of survival versus success complicates direct comparison across studies. Earlier systematic reviews similarly reported pooled survival estimates between approximately 88% and 93% without stratification by treatment era or detailed analysis of failure timing [
3,
4,
5]. By incorporating chronological stratification, weighted pooled survival estimates, and statistical comparison between early and modern cohorts, the present study provides a more granular interpretation of temporal trends. Furthermore, the differentiation between early osseointegration failure and late biological complications offers clinically relevant insight not consistently addressed in previous analyses.
This review has several strengths. It integrates recent literature up to 2026, applies a structured methodological framework, and combines quantitative pooling with subgroup and temporal analyses, allowing a more comprehensive interpretation of implant outcomes in grafted cleft sites. The inclusion of failure timing and causes further enhances clinical relevance by identifying the early healing phase as the most critical period for implant prognosis.
However, several limitations must be acknowledged. The dominance of observational study designs, limited number of prospective investigations, and variability in outcome reporting restrict the strength of causal inference. Subgroup analyses were constrained by imbalances in study numbers, particularly for simultaneous implant placement and GBR reporting, and long-term data beyond 10 years remain scarce. In addition, patient-reported outcomes, esthetic assessments, and functional measures were inconsistently reported, limiting evaluation of treatment success beyond survival.
Future research should prioritize prospective multicenter studies with standardized definitions of implant survival and success, longer follow-up durations, and stratified analysis according to grafting protocols, implant timing, and defect morphology. Such studies are essential to refine clinical guidelines and further optimize implant-supported rehabilitation strategies in cleft populations.
5. Conclusions
Implant-supported rehabilitation in grafted alveolar cleft sites demonstrates high and predictable survival, particularly in contemporary clinical practice. Failures occur mainly during the early osseointegration phase and are most commonly related to insufficient graft volume or inadequate primary stability, highlighting the importance of careful surgical planning and graft assessment.
When appropriate bone reconstruction, healing, and multidisciplinary coordination are achieved, implant therapy represents a reliable component of comprehensive cleft care. Further prospective studies with standardized protocols and long-term follow-up are needed to strengthen evidence-based recommendations.
Supplementary Materials
The following supporting information is available through email from the corresponding author if asked.
Author Contributions
“Conceptualization, A.T and O.F..; methodology, A.M and A.G..; software, D.S..; validation, O.F and S.B.; formal analysis, A.T. and A.M .; investigation, A.T. and O.F..; resources, A.M. and A.T.; data curation, O.F..; writing—original draft preparation, . A.T and A.M.; writing—review and editing, O.F and S.B visualization, A.G and D.S.; supervision O.F. and B.S.; project administration O.F and A.G... All authors have read and agreed to the published version of the manuscript.”.
Funding
No funding to declare.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Available through email from the corresponding author if asked.
Acknowledgments
All authors had equal contribution on writing and preparing this manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Guo, J.; Li, C.; Zhang, Q.; et al. Secondary bone grafting for alveolar cleft in children with cleft lip or cleft lip and palate. Cochrane Database Syst. Rev. 2011, (6), CD008050. [CrossRef]
- Vuletić, M.; Knežević, P.; Jokić, D.; et al. Alveolar bone grafting in cleft patients: From bone defect to dental implants. Acta Stomatol. Croat. 2014, 48(4), 250–257. [CrossRef]
- Wang, F.; Wu, Y.; Zou, D.; Wang, G.; Kaigler, D. Clinical outcomes of dental implant therapy in alveolar cleft patients: A systematic review. Int. J. Oral Maxillofac. Implants 2014, 29(5), 1098–1105. [CrossRef]
- Wermker, K.; Jung, S.; Joos, U.; Kleinheinz, J. Dental implants in cleft lip, alveolus, and palate patients: A systematic review. Int. J. Oral Maxillofac. Implants 2014, 29(2), 384–390. [CrossRef]
- Sales, P.H.H.; Oliveira-Neto, O.B.; Torres, T.S.; de Lima, F.J.C. Effectiveness of dental implants placed in bone graft area of cleft patients. Int. J. Oral Maxillofac. Surg. 2019, 48(9), 1109–1114. [CrossRef]
- Mallick, R.; Pisulkar, S.K.; Reddy, S.G.; Jain, V. Assessing time gap between alveolar cleft repair and dental implant placement: A systematic review. J. Clin. Diagn. Res. 2022, 16(2), ZE28–ZE36. [CrossRef]
- Tai, C.C.; Sutherland, I.S.; McFadden, L. Prospective analysis of secondary alveolar bone grafting using computed tomography. J. Oral Maxillofac. Surg. 2000, 58(11), 1241–1250. [CrossRef] [PubMed]
- Feichtinger, M.; Mossböck, R.; Kärcher, H. Assessment of bone resorption after secondary alveolar bone grafting using three-dimensional computed tomography: A three-year study. Cleft Palate Craniofac. J. 2007, 44(2), 142–148. [CrossRef] [PubMed]
- Pathak, A.; Dhamande, M.M.; Sathe, S.; Gujjelwar, S. Effectiveness, esthetics, and success rate of dental implants in bone-grafted regions of cleft lip and palate patients: A systematic review and meta-analysis. Cureus2023, 15(11), e49101. [CrossRef] [PubMed]
- Howe, M.S.; Keys, W.; Richards, D. Long-term (10-year) dental implant survival: A systematic review and sensitivity meta-analysis. J. Dent. 2019, 84, 9–21. [CrossRef] [PubMed]
- Esper, L.A.; Sbrana, M.C.; Ribeiro, A.L.R.; et al. Dental implant rehabilitation in cleft lip and palate patients: A retrospective evaluation. Cleft Palate Craniofac. J. 2012, 49(2), 211–217.
- Dušková, M.; Leamerová, E.; Mayerová, D. Dental implants in patients with cleft lip and palate. J. Craniofac. Surg. 2007, 18(2), 409–415. [CrossRef] [PubMed]
- Dempf, R.; Teltzrow, T.; Kramer, F.J.; et al. Alveolar bone grafting in patients with complete clefts: A comparative study between secondary and tertiary bone grafting. Cleft Palate Craniofac. J. 2002, 39(1), 18–25. [CrossRef]
- Fiamengui Filho, J.F.; da Silva Dalben, G.; Santamaria, M. Jr. Aesthetic analysis of an implant-supported denture at the cleft area. Cleft Palate Craniofac. J. 2013, 50(5), 597–602. [CrossRef] [PubMed]
- Sogancı, O.; Altunok, E.C.; Aydın, C. Stress distribution in alveolar cleft bone grafts restored with dental implants: A finite element analysis. Cleft Palate Craniofac. J. 2016, 53(6), e180–e187.
- Dowgierd, K.; Kozakiewicz, M. Free flap reconstruction of alveolar clefts followed by dental implant placement: Clinical outcomes. J. Clin. Med. 2021, 10(15), 3303. [CrossRef]
- Green, M.A.; Padwa, B.L. Does timing of secondary alveolar bone grafting affect the need for additional bone augmentation prior to implant placement at cleft sites? J. Oral Maxillofac. Surg. 2021, 79(10), 2106–2115. [CrossRef]
- Green, M.A.; Flanagan, S.; Britt, M.C. Cortical block grafting successfully augments alveolar cleft sites for dental implant placement. J. Oral Maxillofac. Surg. 2024, 82(7), 820–827. [CrossRef]
- Cune, M.S.C.; Meijer, G.J.; Koole, R. Anterior tooth replacement with implants in grafted alveolar cleft sites: A case series. Clin. Oral Implants Res. 2004, 15(5), 616–624. [CrossRef]
- Kearns, G.; Perrott, D.H.; Sharma, A.; Kaban, L.B.; Vargervik, K. Placement of endosseous implants in grafted alveolar clefts. Cleft Palate Craniofac. J. 1997, 34(6), 520–525. [CrossRef] [PubMed]
- Jensen, J.; Sindet-Pedersen, S.; Enemark, H. Reconstruction of residual alveolar cleft defects with one-stage mandibular bone grafts and osseointegrated implants. J. Oral Maxillofac. Surg. 1998, 56(4), 460–466. [CrossRef]
- Jansma, J.; Raghoebar, G.M.; Batenburg, R.H.K.; Stellingsma, K.; van Oort, R.P. Bone grafting of cleft lip and palate patients for placement of endosseous implants. Cleft Palate Craniofac. J. 1999, 36(1), 67–72. [CrossRef] [PubMed]
- Härtel, J.; et al. Implant-supported prosthetic rehabilitation after alveolar cleft bone grafting: A clinical report. J. Craniomaxillofac. Surg. 1999, 27(6), 354–358. [CrossRef] [PubMed]
- Kramer, F.J.; Baethge, C.; Swennen, G.; et al. Dental implants in patients with orofacial clefts: A long-term follow-up study. Int. J. Oral Maxillofac. Surg. 2005, 34(7), 715–721. [CrossRef]
- Landes, C.A. Implant-borne prosthetic rehabilitation of bone-grafted cleft versus traumatic anterior maxillary defects. J. Oral Maxillofac. Surg. 2006, 64(2), 297–307. [CrossRef] [PubMed]
- Lalo, J.; Cheynet, F.; Wulfman, C.; et al. Prosthetic rehabilitation with dental implants in cleft lip and palate patients: A retrospective study. Rev. Stomatol. Chir. Maxillofac. 2007, 108(6), 413–418.
- Matsui, K.; Echigo, S.; Kimizuka, S.; et al. Long-term study of dental implants placed into alveolar cleft sites. Cleft Palate Craniofac. J. 2007, 44(4), 444–451. [CrossRef]
- Takahashi, T.; Sato, Y.; Yoda, N.; et al. Long-term follow-up of dental implants placed in a grafted alveolar cleft. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2008, 105(3), 297–302. [CrossRef]
- Ferreira, L.M.; Silva, G.C.; Garcia, I.R. Jr.; et al. Survival of dental implants in the cleft area: A retrospective study. Cleft Palate Craniofac. J. 2010, 47(6), 586–592. [CrossRef]
- Borgnat, S.; Rignon-Bret, C.; Wulfman, C.; et al. Implant-borne prosthetic rehabilitation in patients with cleft lip and palate: A retrospective study on 43 patients. Rev. Stomatol. Chir. Maxillofac. Chir. Orale 2015, 116(2), 80–86. [CrossRef]
- Vo Van Nhan; Pham Dang Hai; Nguyen Tien Dat; et al. A new technique in alveolar cleft bone grafting for dental implant placement in patients with cleft lip and palate. Cleft Palate Craniofac. J. 2017, 54(4), 417–424. [CrossRef]
- Saint-Surin, I.; Roman, T.; Touzet-Roumazeille, S.; Ferri, J.; Lauwers, L. Implant-borne rehabilitation for alveolar dental cleft: Retrospective analysis of thirty-nine cases. J. Stomatol. Oral Maxillofac. Surg. 2019, 120(6), 540–545. [CrossRef] [PubMed]
- Alberga, J.M.; Brouwer, J.; Vissink, A.; et al. Dental implant placement in alveolar cleft patients: A retrospective comparative study. Int. J. Oral Maxillofac. Surg. 2020, 49(6), 793–800. [CrossRef]
- van Putten, Y.; Rozeboom, A.V.J.; Strabbing, E.M.; Koudstaal, M.J.; Tahmaseb, A. A retrospective cohort study on dental implant survival in patients with grafted alveolar clefts. Clin. Oral Implants Res. 2023, 34(6), 618–626. [CrossRef]
- Vieira Dos Santos, C.C.; Gonçalves, M.G.; Costa, M.S.C.; et al. Survival rate of dental implants in the cleft area: A cross-sectional retrospective study. J. Craniofac. Surg. 2023, 34(7), 2146–2148. [CrossRef]
- Knobloch, L.A.; Larsen, P.; Gohel, A.; et al. Prospective cohort study to evaluate narrow diameter implants for the restoration of a missing maxillary lateral incisor in patients with a cleft palate: Five-year results. J. Prosthet. Dent. 2025, 134(5), 1738–1747. [CrossRef]
- Wang, L.; Chen, B. Vertical versus horizontal morphology of congenital alveolar cleft defects: A one-year comparative study on clinical, radiographic, and biochemical outcomes. J. Craniomaxillofac. Surg. 2026, 54(2), 104420. [CrossRef] [PubMed]
- Enache, M.; Didilescu, A.; Bud, E.; Bunta, O.; Feștilă, D.; Ghergie, M. Facial profile perception of young adults by different examiners. Rom. J. Leg. Med. 2023, 31, 23–29. [CrossRef]
- Topârcean, A.M.; Acatrinei, A.; Rusu, I.; Feștilă, D.; Câmpian, R.S.; Kelemen, B.; Dinu Ghergie, M.C. Genetic insights into skeletal malocclusion: The role of the FBN3 rs7351083 SNP in the Romanian population. Medicina 2024, 60, 1061. [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).