Submitted:
13 February 2026
Posted:
27 February 2026
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Fundamentals of Single-Molecule Tracking
2.1. What is SMT?
2.2. Historical Context
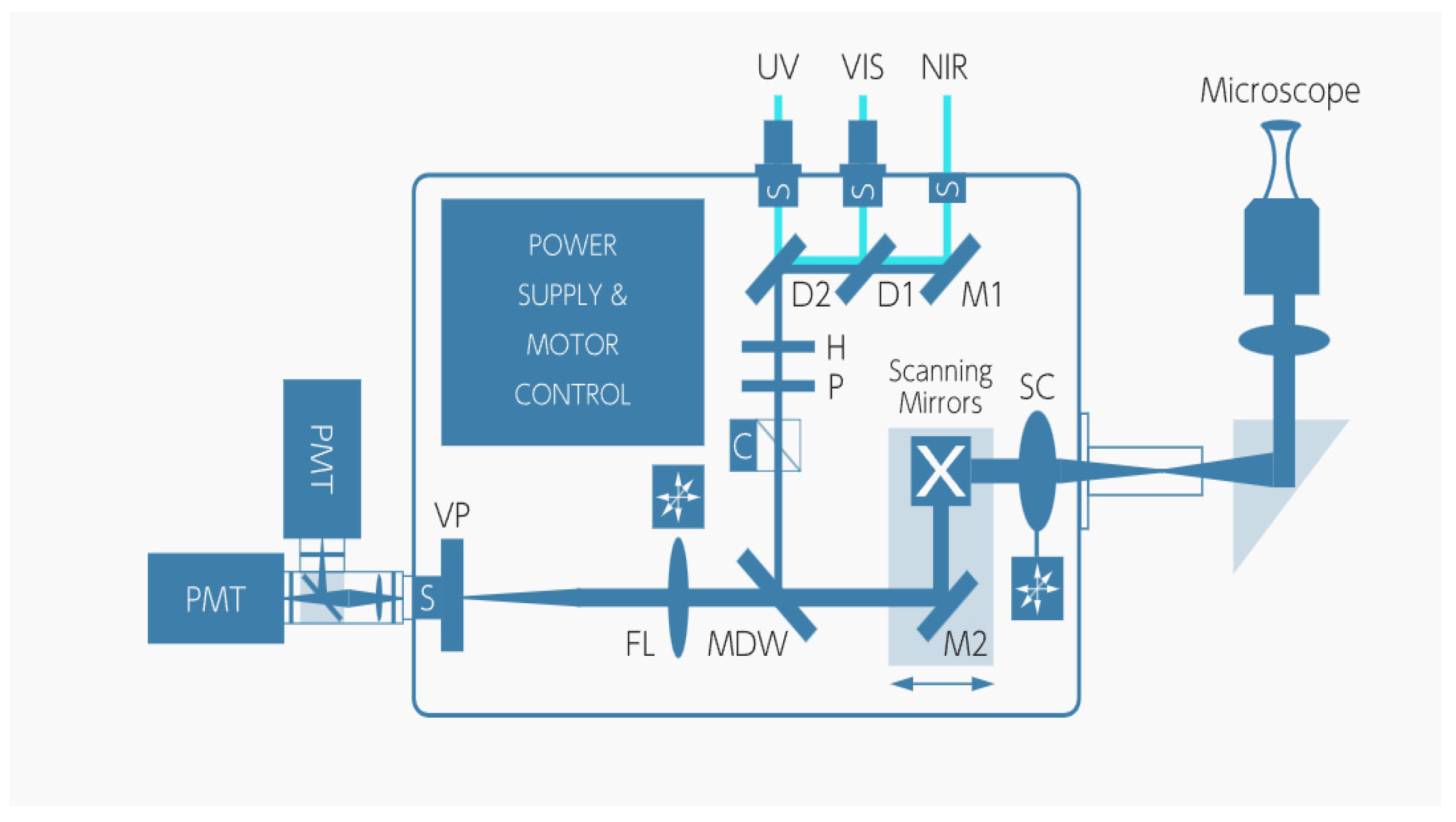
2.3. Key Techniques
2.4. Core Principles
- (i)
- Fluorescent Labeling
- (ii)
- High-Resolution Microscopy
- (iii)
- Two-Color Single-Molecule Tracking
- (iv)
- Tracking Algorithms
- (v)
- Machine Learning Integration
3. Some Challenges in Single-Molecule Tracking
4. Applications of Single-Molecule Tracking Techniques in Live Cells
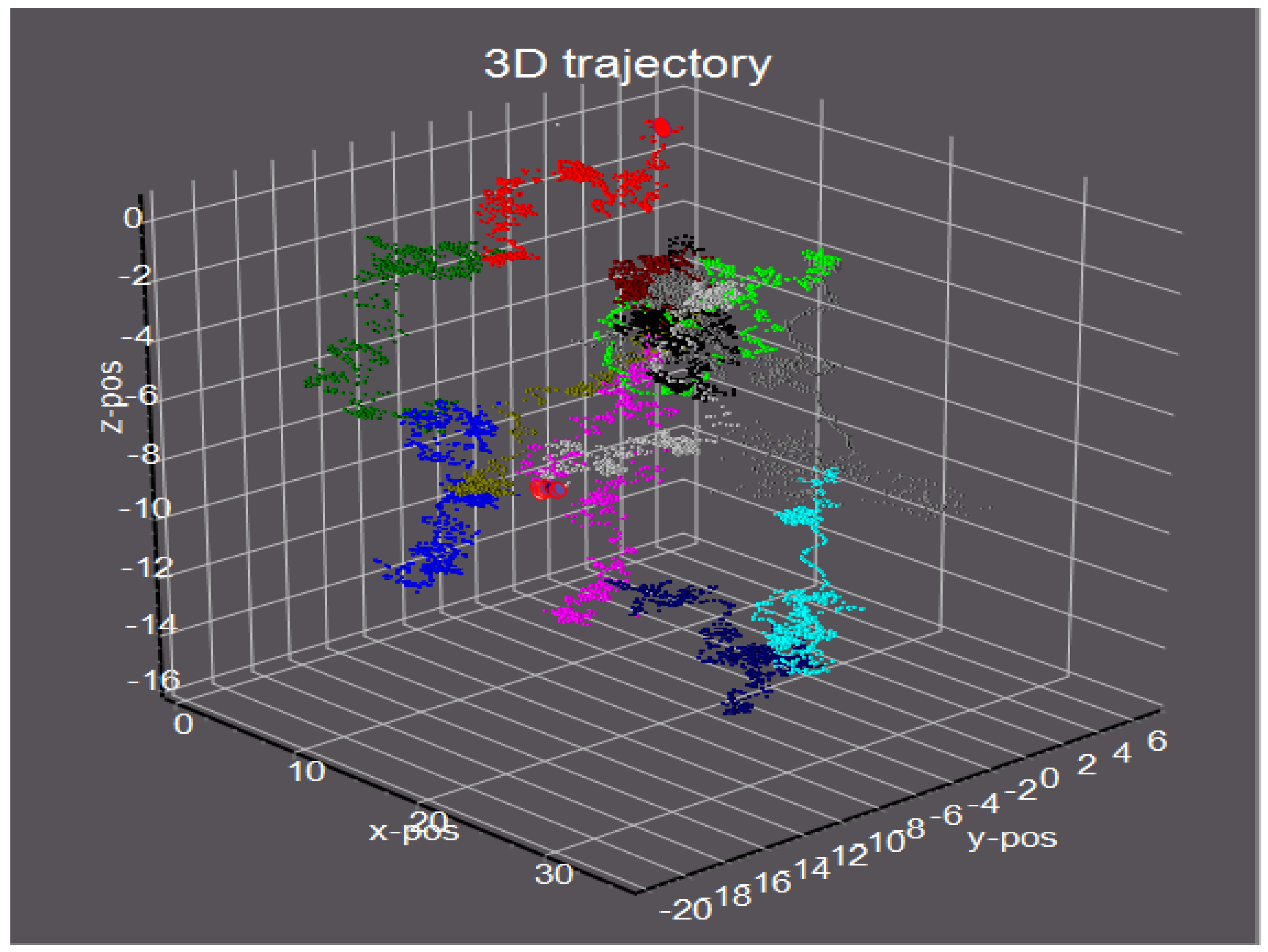
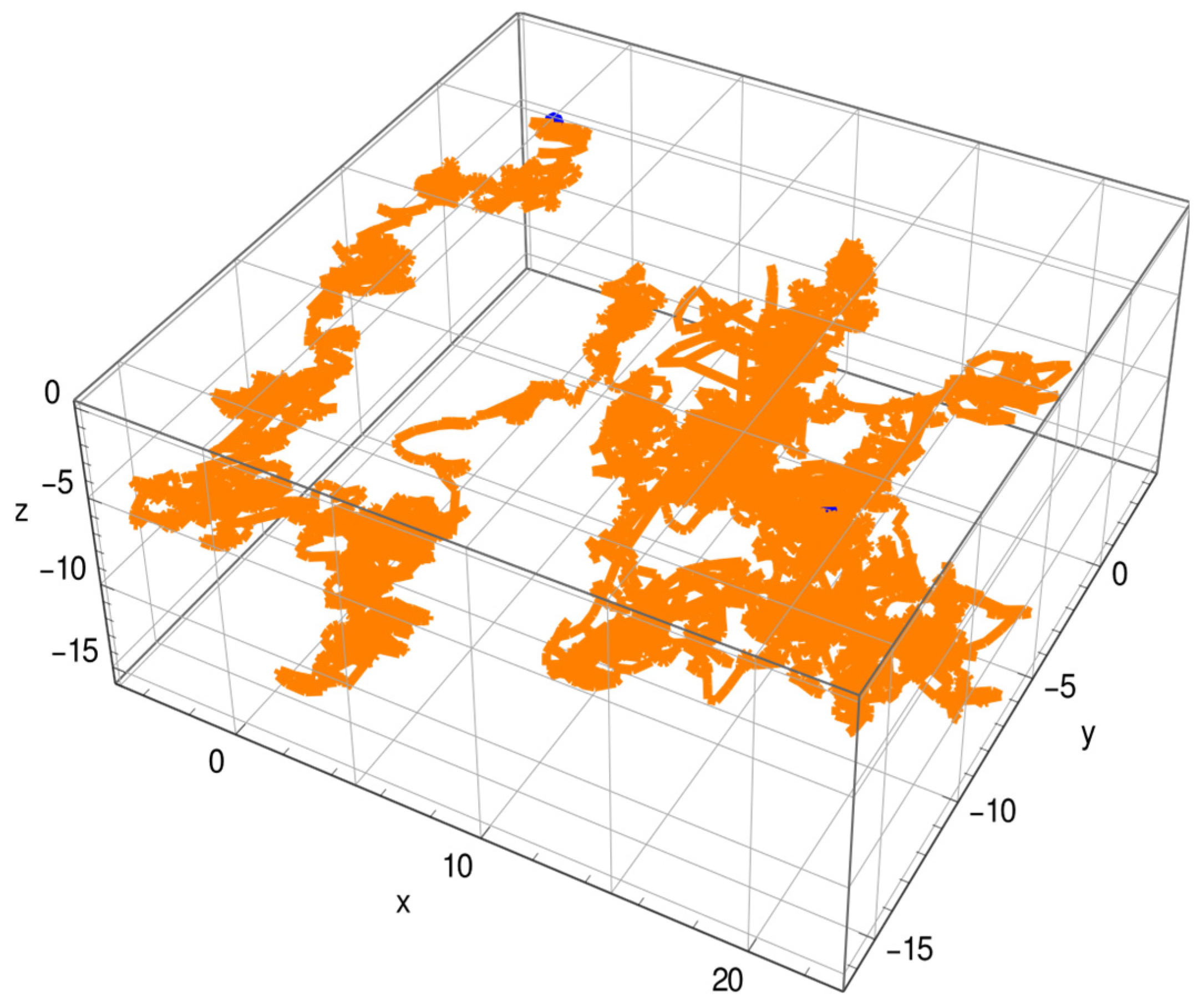
4.1. Protein Dynamics
4.2. Membrane Transport
4.3. Drug Discovery
4.4. RNA Dynamics
5. Present Demands in Single-Molecule Tracking
5.1. Photobleaching and Phototoxicity
5.2. Complexity of Cellular Systems
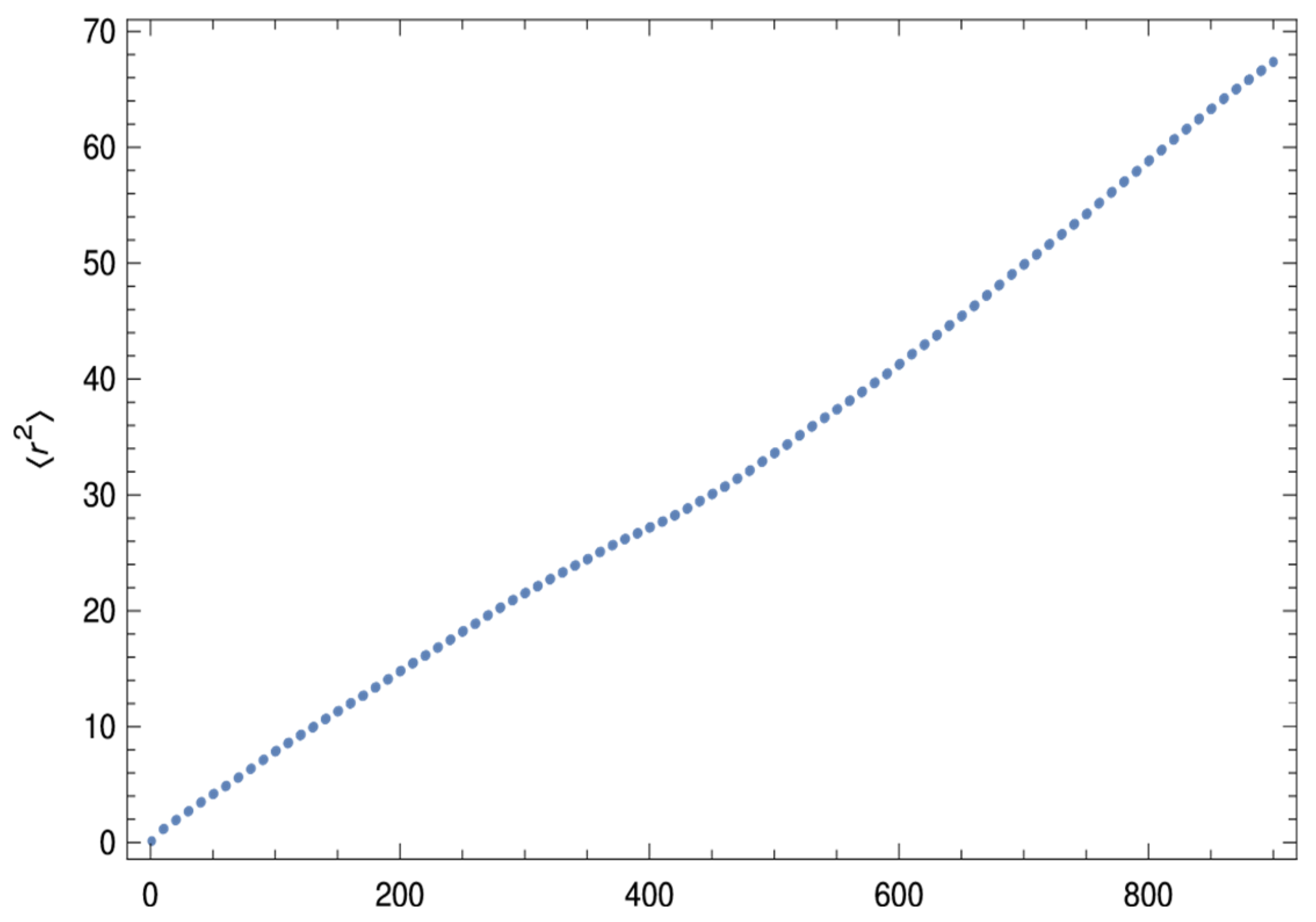
5.3. Data Analysis Bottlenecks
5.4. Plasmonic Platforms
6. Future Directions
6.1. Multi-Color SMT
6.2. Non-Labeling Methods
6.3. Integration with Systems Biology
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SMT | Single-Molecule Tracking |
| SPT | Single-Particle Tracking |
| SMLM | Single-Molecule Localization Microscopy |
| STORM | Stochastic Optical Reconstruction Microscopy |
| PALM | Photo-Activated Localization Microscopy |
| MinFlux | Minimal photon Flux |
| FRET | Förster Resonance Energy Transfer |
| TIRF | Total Internal Reflection Fluorescence microscopy |
| FLIM | Fluorescence Lifetime Microscopy |
| FCS | Fluorescence Correlation Spectroscopy |
| FCCS | Fluorescence Cross-Correlation Spectroscopy |
| SPSM-FCS | Single-Phase Single-Molecule Fluorescence Correlation Spectroscopy (autocorrelation and two-color (dual color) cross-correlation) |
| UV | Ultraviolet |
| IR | Infrared |
| NIR | Near-Infrared |
| KHz | Kilohertz, a unit of frequency, representing 1,000 cycles or oscillations per second |
| MSD | Mean Squared Displacement |
| D | Diffusion coefficient |
| 3D | Three dimensional |
| 2D | Two dimensional |
| EGFR | Epidermal Growth Factor Receptor |
| mCherry | Member of the mFruits family of monomeric red fluorescent proteins |
| eGFP | Enhanced Green Fluorescent Protein |
| DNA | Deoxyribonucleic Acid |
| RNA | Ribonucleic Acid |
| mRNA | Messenger Ribonucleic Acid |
References
- Földes-Papp, Z.; Baumann, G.; Kinjo, M.; Tamura, M. Single-Phase Single-Molecule Fluorescence Correlation Spectroscopy (SPSM-FCS). Distinguished article entry. In Encyclopedia of Medical Genomics & Proteomics; Fuchs, J., Podda, M. Editors, Eds.; Taylor & Francis, New York, USA, 2005, pp. 1-7.
- Moerner, W. E. Nobel Lecture: Single-molecule spectroscopy, imaging, and photocontrol. Reviews of Modern Physics 2015, 87, 1183-1212.
- Betzig, E.; Patterson, G. H.; Sougrat, R.; Lindwasser, O. W.; Olenych, S.; Bonifacino, J. S.; Davidson, M. W.; Lippincott-Schwartz, J.; Hess, H. F. Imaging intracellular fluorescent proteins at nanometer resolution. Science 2006, 313, 1642-1645.
- Moerner, W.E.; Kador, L. Optical detection and spectroscopy of single molecules in a solid. Physical Reviews Letters 1989, 62, 2535-2538.
- Dickson, R. M.; Cubitt, A. B.; Tsien, R. Y.; Moerner, W. E. On/Off blinking and switching behavior of single molecules of green fluorescent protein. Nature 1997, 388, 355–358.
- Betzig, E. Proposed Method for molecular optical imaging. Optics Letters 1995, 20, 237-239.
- Hell, S. W.; Wichmann, J. Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Optic Letters 1994, 19, 780-782.
- Klar, T. A.; Jakobs, S.; Dyba, M.; Egner, A.; Hell, S. Fluorescence microscopy with diffraction resolution barrier broken by stimulated emission. Proceedings of the National Academy of Sciences 2000, 97, 8206-8210.
- Földes-Papp, Z. Individual macromolecule motion in a crowded living cell. Current Pharmaceutical Biotechnology 2015, 16, 1-2.
- Földes-Papp, Z. Fluorescence Fluctuation Spectroscopic Approaches to the Study of a Single Molecule Diffusing in Solution and a Live Cell without Systemic Drift or Convection: A Theoretical Study. Current Pharmaceutical Biotechnology 2007, 8, 261-273.
- Földes-Papp, Z. Single-molecule time resolution in dilute liquids and live cells at the molecular scale: Constraints on the measurement time. American Journal of Translational Medicine 2021, 5, 154-165.
- Földes-Papp, Z. The thermodynamic signature of a single molecule or a single particle in dilute liquids and live cells: Single-Molecule Biophysics & Biochemistry based on the stochastic nature of diffusion. American Journal of Translational Medicine 2023, 7, 74-77.
- Földes-Papp, Z. Measurements of Single Molecules in Solution and Live Cells Over Longer Observation Times Than Those Currently Possible: The Meaningful Time. Current Pharmaceutical Biotechnology 2013, 14, 441-444.
- Baumann, G.; Földes-Papp, Z. Single-Molecule Tracking in Live Cell without Immobilization or without Hydrodynamic Flow by Simulations: Thermodynamic Jitter. Biophysica 2024, 3, 442-452.
- Baumann, G.; Földes-Papp, Z. Study on Single-Molecule Biophysics and Biochemistry in dilute liquids and live cells without immobilization or significant hydrodynamic flow: The thermodynamic Single-Molecule DEMON. Current Pharmaceutical Biotechnology 2022, 23, 1750-1757.
- Tang, W.; Fuentenebro Navas, D.; Vermeer, B.; Schmid S. Disentangling a Complex Biomolecular World with Singel-Molecule Resolution. Chimia 2025, 79, 770-779.
- F. Balzarotti, F.; Eilers, Y.; Gwosch, K. C.; Gynna, A. H.; Westphal, V.; Stefani, F. D.; Elf, J.; Hell, S. W. Nanometer resolution imaging and tracking of fluorescent molecules with minimal photon fluxes. Science 2017, 355, 606–612.
- Gwosch, G. K.; Pape, J. K.; Balzarotti, F.; Hoess, P.; Ellenberg, J.; Ries, J.; Hell, S. W. Minflux nanoscopy delivers 3d multicolor nanometer resolution in cells. Nature Methods 2020, 17, 217–224.
- Schultze, S.; Grubmüller, H. Bayesian MINIFLUX localization microscopy. Cornell University arXiv:2510.25654 [physics.comp-ph] 2025, (1-5).
- Driouchi, A.; Bretan, M.; Davis, B. J.; Heckert, A.; Seeger, M.; Silva, M. B.; Forrest, W.S.R.; Hsiung, J.; Tan, J.; Yang, H.; McSwiggen, D.T.; Song, L.; Sule, A.; Abaie, B.; Chen, H.; Chhun, B.; Conroy, B.; Elliott, L. A.; Gonzalez, E.; Ilkov, F.; Isaacs, J.; Labaria, G.; Lagana, M.; Larsen, D. D.; Margolin, B.; Nguyen, M. K.; Park, E.; Rine, J.; Tang, Y.; Vana, M.; Wilkey, A.; Zhang, Z.; Basham, S.; Ho, J. .J; Johnson, S.; Klammer, A. A.; Lin, K.; Darzacq, X.; Betzig, E.; Berman, R.T.; Anderson, D.J. Oblique line scan illumination enables expansive, accurate and sensitive single-protein measurements in solution and in living cells. Nature Methods 2025, 22, 559-568.
- Meroz, Y.; Sokolov, I.M.; Klafter, J. Subdiffusion of mixed origins: When ergodicity and nonergodicity coexist. Physical Review E 2010, 81, 0101001.
- Földes-Papp, Z.; Baumann, G.; Li, L.-C. Visualization of subdiffusive sites in a live single cell. Journal of Biological Methods 2021, 8, e142 (1-8).
- Földes-Papp, Z.; Baumann, G. Fluorescence molecule counting for single-molecule studies in crowded environment of living cells without and with broken ergodicity. Current Pharmaceutical Biotechnology 2011, 12, 824-833.
- Baumann, G.; Place, R. F.; Földes-Papp, Z. Meaningful interpretation of subdiffusive measurements in living cells (crowded environment) by fluorescence fluctuation microscopy. Current Pharmaceutical Biotechnology 2010, 11, 527-543.
- Catapano, C.; Dietz, M. S.; Kompa, J.; Jang, S.; Freund, P.; Johnsson, K.; Heilemann, M. Long-Term Single-Molecule Tracking in Living Cells using Weak-Affinity Protein Labeling. Angewandte Chemie International Edition 2025, 64, e202413117 (1-8).
- König, A. I.; Sorkin, R.; Alon, A.; Nachmias, D.; Dhara, K.; Brand, G.; Yifrach, O.; Arbely, E.; Roichman, Y.; Elia, N. Live cell single molecule tracking and localization microscopy of bioorthogonally labeled plasma membrane proteins. Nanoscale 2020, 12, 3236-3248.
- Sakuragi, S.; Kato, N.; Uchida; T.; Katagiri, T.; Enomoto, M.; Kato, R.; Yoshimura, H.; Oyama, C.; Katayama, I.; Chikuma, A.; Teramura, Y.; Bannai, H. Oligo DNA-based quantum dot (QD) single-particle tracking for multicolor single-molecule imaging. bioRxv 2025, preprint . [CrossRef]
- Ilievski, F.; Wikström, L.; Borg, A.; Volkov, I. L.; Brandis, G.; Johansson, M. Optimization of the genetic code expansion technology for intracellular labelling and single-molecule tracking of proteins in genomically re-coded E. coli. RSC Chemical Biology 2026, Advance Article online DOI: 10.1039/d5cb00221d.
- Bjerneld, E. J.; Földes-Papp; Z.; Käll, M.; Rigler, R. Single-molecule surface-enhanced Raman and fluorescence correlation spectroscopy of horseradish peroxidase. Journal of Physical Chemistry B 2002, 106, 1213-1218.
- Földes-Papp, Z.; Liao, S.-C.; Barbieri, B.; Gryczynski, K.; Luchowski, R.; Gryczynski, Z.; Gryczynski, I.; Borejdo, J.; You, T. Single actomyosin motor interactions in skeletal muscle. Biochimica et Biophysica Acta 2011, 1813, 856-866.
- Steves, M. A.; Xu, K. Direct microsecond wide-field single-molecule tracking and super-resolution mappimg via CCD vertical shift. Nature Communications 2025, 16, 10503 (1-19).
- Cabillic, M.; Forriere, H.; Bettarel, L.; Butler, C.; Neuhaus, A.; Idrissi, I.; Sambrano-Lopez, M. E.; Rossbroich, J.; Müller, L.-R.; Ries, J.; Grenci, G.; Viasnoff, V.; Levet, F.; Sibarita, J.-B.; Galland, R. In-depth single molecule localization microscopy using adaptive optics and single objective light-sheet microscopy. Nature Communications 2025, 16, 8362 (1-15).
- Ruprecht, V.; Brameshuber, M.; Schütz, G. J. Two-color single molecule tracking combined with photobleaching for the detection of rare molecular interactions in fluid biomembranes. Soft Matter 2010, 6, 568-581.
- Hänselmann. S.; Herten, D. P. Two-Color Single-Molecule Tracking in Live Cells. Methods Molecular Biology 2017, 1663, 127-138.
- Saavedra, L. A.; Barrantes, F. J. Temporal convolutional networks work as general feature extractions for single-particle diffusion analysis. Journal of Physics: Photonics 2025, 7, 025017 (1-15).
- Umney, O.; Slaney, H.; Williams, C. J. M.; Quirke P.; Peckham, M.; Curd, A. P. ClusterNet: Classifying Single-Molecule localization Microscopy Datasets with Graph-Based Deep Learning of Supracluster Structure. Small Science 2025, e202500255 (1-11).
- Pineda, J.; Masó-Orriols, S.; Masoliver, M.; Bertran, J.; Golsör, M.; Volpe, G.; Manzo C. Enhanced spatial clustering of single-molecule localizations with graph neural networks. Nature Communications 2025, 16, 9693 (1-17).
- Saguy, A.; Xiao, D.; Narayanasamy, K. K., Nakatani, Y.; Saliba, N.; Gagliano, G.; Gustavsson, A.-K.; Heilemann, M., Shechtman, Y. One-click reconstruction in single-molecule localization microscopy via experimental parameter-aware deep learning. npj Imaging 2025, 3, 61(1-10).
- Khater, I. M.; Nabi, I. R.; Hamarneh, G. HDCluster: High-Degree Graph Clustering for Robust Analysis of Single Molecule Localization Microscopy. bioRxiv [Preprint] 2025 (Oct 24), 2025.10.23.684134.
- Saxton, M. J.; Jacobson, K. Single-particle tracking: applications to membrane dynamics. Annual Review of Biophysics and Biomolecular Structure 1997, 26, 373-399.
- Jeon, J.-H.; Leijnse, N; Odderschede, L. B.; Metzler, R. Anomalous diffusion and power-law relaxation of the time averaged mean squared displacement in worm-like micellar solution. New Journal of Physics 2013, 15, 045011 (1-16).
- Ghosal, A.; Wang, Y.-H.; Nguyen, N.; Troyer, L.; Kim, S. Practical considerations for accurate estimation of diffusion parameters from single-particle tracking in living cells. The Journal of Physical Chemistry 2025, 163, 134202 (1-11).
- Baumann, G.; Gryczynski, I.; Földes-Papp, Z. Anomalous behavior in length distributions of 3D random Brownian walks (three dimensional random Brownian walks) and measured photon count rates within observation volumes of single-molecule trajectories in fluorescence fluctuation microscopy. Optics Express 2010, 18, 17883-17896.
- Lanza, A.; Martinez-Martin, I.; Tapia-Rojo, R.; Bo, S. Measuring irreversibility in stochastic systems by categorizing single-molecules displacements. Cornell University arXiv:2511.09183v1 [cond-mat.stat-mech] 2025, 1-12.
- Khrapak, S.; Khrapak, A. Stokes–Einstein Relation in Different Models of Water. Molecules 2024, 29, 5587. [CrossRef]
- Földes-Papp, Z.; Liao, S.-C. J.; You, T.; Barbieri, B. Reducing Background Contributions in Fluorescence Fluctuation Time-Traces for Single-Molecule Measurements in Solution. Current Pharmaceutical Biotechnology 2009, 10, 532-542.
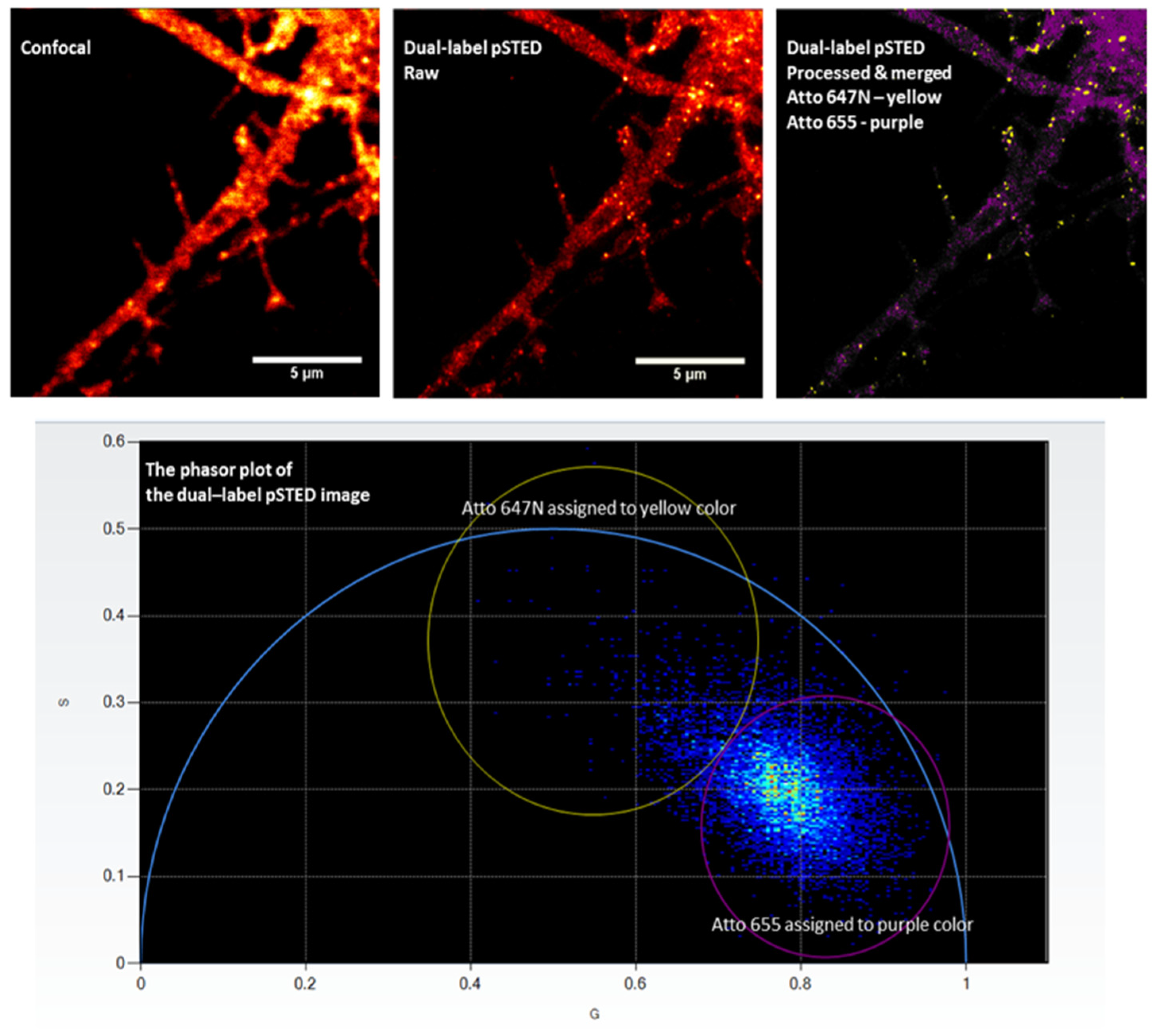
- Sun, Y.; Tortarolo, G.; Teng, K.-W.; Ishitsuka, Y.; Coskun, U. C.; Liao, S.-C. J.; Diaspro, A.; Vicidomini, G.; Selvin, P.R.; Barbieri, B. A novel pulsed STED microscopy method using FastFLIM and the phasor plots. In Proceedings SPIE 10069, Proceedings volume: Multiphoton Microscopy in the Biomedical Sciences XVII, San Francisco, California, United States of America, February 21, 2017; 100691C. [CrossRef]
- McSwiggen, D. T.; Liu, H.; Tan, R.; Puig, S. A.; Akella, L. B.; Berman, R.; Bretan, M.; Chen, H.; Darzacq, X.; Ford, K.; Godbey, R.; Gonzalez, E.; Hanuka, A.; Heckert, A.; Ho, J. J.; Johnson, S. L.; Kelso, R.; Klammer, A.; Krishnamurthy, R.; Li, J.; Lin, K.; Margolin, B.; McNamara, P.; Meyer, L.; Pierce, S. E.; Sule, A.; Stashko, C.; Tang, Y., Anderson, D. J.; Beck, H. P. A high-throughput platform for single-molecule tracking identifies drug interaction and cellular mechanisms. eLife 2023, 12, RP93183 (1-26).
- Liu, J.; Zhang, X.; Zhao, B.; Ling, H.; Li, Y.; Sun, K.; Chen, S.; Zhang, Y.; Zhai, T.; Zhang, Y.; Li, F.; Qian Liu, Q. In Situ Monitoring of Membrane Protein Dynamics Using High-Throughput Red-Light-Activated Single-Molecule Tracking. ACS Nano 2025, 19, 13466–13478.
- Watanabe, D., Hiroshima, M., Yasui, M.; Ueda, M. Single molecule tracking based drug screening. Nature Communication 2024, 15, 8975 (1-14).
- Park, J.; Sokolovska, N.; Cabriel, C.; Kobayashi, A.; Corsin, E.; Garcia-Fernandez, F.; Izeddin, I.; Miné-Hattab, J. Novel estimation of memory in molecular dynamics with extended and comprehensive single-molecule tracking software: FreeTrace. bioRxiv 2026, preprint (1-23), . [CrossRef]
- Zalejski, J.; Sun, J.; Sharma, A. Unravelling the Mystery inside Cells by Using Single-Molecule Fluorescence Imaging. Journal of Imaging 2023, 9, 192 (1-25).
- Park, C.; Jung, S.; Park, H. Single vesicle tracking for studying synaptic vesicle dynamics in small central synapses. Current Opinion in Neurobiology 2022,76, 102596.
- Graham, T. G. W.; Dugast-Darzacq, C.; Dailey, G. M.; Weng, B.; Anantakrishnan, S.; Darzacq, X.; Tjian, R. Single-molecule live imaging of subunit interactions and exchange within cellular regulatory complexes. Molecular Cell 2025, 85, 2854-2868.
- Bühler, B.; Sunbul, M. Single-Molecule RNA Imaging in Live Cells with an Avidity-Based Fluorescent Light-Up Aptamer biRhoBAST. Methods in Molecular Biology 2024; 2822, 87-100.
- Hou, S.; Exell, J.; Welsher, K. Real-time 3D single molecule tracking. Nature Communication 2020, 11, 3607.
- Si, D.; Dong, Z.; Xing, G.; Yao, L.; Lu, J.; Ma, M.; Yuan,Y.; Zhao, H.; Dong, B.; Li, C.; Yuan, L.; Chen, Z.; Wang, L. A bright and regenerative array tag with fluorogenic ligands for long-term live-cell nanoscopy and single-molecule tracking. Nature Communications 2026, 17, 576 (1-14).
- Place, R. F; Noonan, E. J.; Földes-Papp, Z.; Li, L.-C. Defining Features and Exploring Chemical Modifications to Manipulate RNAa Activity. Current Pharmaceutical Biotechnology 2010, 11, 518-526.
- Butler, C.; Saraceno G.E.; Kechkar, A.; Bénac, N.; Studer, V.; Dupuis, J.P.; Groc, L.; Galland, R.; Sibarita J.-B. Multi-Dimensional Spectral Single Molecule Localization Microscopy. Frontiers in Bioinformatics 2022, 2, 813494 (1-14).
- Rabiee, N.; Lan, X. Advancing Multicolor Super-Resolution Volume Imaging: Illuminating Complex Cellular Dynamics. Journal of the American Chemical Society Au 2025, 5, 2388–2419.
- Gebhardt, C. M. Single-molecule Tracking and Kinetic Analysis in Living Cells and Multicellular Organisms. Journal of Molecular Biology 2026, 438, 169308.
- Kim, K.; Jang, J.; Cho, J.; Ahn, Y.; Jeong, S.; Shin, J.; Yea, K.; Lee, W. J.; Seo, D. Reconciling a Kinetic Model for Dimerization of the EGFR Using Single-Molecule Tracking in Living Cells. The Journal of Physical Chemistry B 2025,129, 9567-957.
- Mondal, S.; Bonventre, E.; Hancock; W.O.; Rice, L.M. Single-molecule microtubule dynamics measurements reveal an intermediate state and clarify the role of nucleotide. Nature Communications 2026, 17, 559 (1-12).
- Földes-Papp, Z. What it means to measure a single molecule in a solution by fluorescence fluctuation spectroscopy. Experimental and Molecular Pathology 2006, 80, 209-218.
- Földes-Papp, Z; Rigler, R. Quantitative Two-Color Fluorescence Cross-Correlation Spectroscopy in the Analysis of Polymerase Chain Reaction. Biological Chemistry 2001, 382, 473-478.
- Földes-Papp, Z; Demel, U.; Tilz, G. P. Ultrasensitive detection and identification of fluorescent molecules by FCS: Impact for immunobiology. Proceedings of the National Academy of Sciences of the United States of America 2001, 98, 11509-11514.
- Földes-Papp, Z. ’True’ single-molecule molecule observations by fluorescence correlation spectroscopy and two-color fluorescence cross-correlation spectroscopy. Experimental and Molecular Pathology 2007, 82, 147-155.
- Földes-Papp, Z. How the molecule number is correctly quantified in two-color fluorescence cross-correlation spectroscopy: corrections for cross-talk and quenching in experiments. Current Pharmaceutical Biotechnology 2005, 6, 437-444.
- Muñoz-Gil, G.; Bachimanchi, H.; Pineda, J.; Midtvedt, B.; Fernández-Fernández, G.; Requena, B.; Ahsini, Y.; Asghar, S.; Bae, J.; Barrantes, F. J.; Bender, S. W. B.; Cabriel, C.; Conejero, J. A.; Escoto, M.; Feng, X.; Haidari, R.; Hatzakis, N. S.; Huang, Z.; Izeddin, I.; Jeong, H.; Jiang, Y.; Kæstel-Hansen, J.; Miné-Hattab, J.; Ni, R.; Park, J.; Qu, X.; Saavedra, L. A.; Sha, H.; Sokolovska, N.; Zhang, Y.; Volpe, G.; Lewenstein, M.; Metzler, R.; Krapf, D.; Volpe, G.; Manzo, C. Quantitative evaluation of methods to analyze motion changes in single-particle experiments. Nature Communication 2025, 16, 6749 (1-17).
- Niu, R.; Shao, J.; Wu, M.; Liu, C.; Chao, J. Single-Molecule Detection of Optical Signals Using DNA-Based Plasmonic Nanostructures. Biosensors 2025, 15, 398 (1-18). [CrossRef]
- Huang, X.; Shi, Q.; Lu, Y.; Li, B.; Ning, Y.; Feng, L.; Sojic, N.; Jiang, D.; Liu, B. Single-Molecule Electrochemiluminescence Imaging of Plasmonic Hot Spot Reactivity. Angewandte Chemie International Edition 2025, 64: e202508266.
- Wenger, J. UV plasmonics to enhance protein autofluorescence detection at the single molecule level. In Proceedings SPIE PC1358, Proceedings volume: UV and Higer Energy Photonics: From Materials to Applications 2025, San Diego, California, United States of America, September 19, 2025; PC135830A. [CrossRef]
- Nooteboom, S.W.; Lamberti, V.; Schellekens, T.; Zijlstra, P. Multiplexed and continuous single-molecule biosensing by spectral demixing of plasmon-enhanced fluorescence. Biosensors and Bioelectronics 2026, 298, 118384 (1-11).
- Ito, Y.; Sakata-Sogawa, K.; Tokunaga, M. Multi-color single-molecule tracking and subtrajectory analysis for quantification of spatiotemporal dynamics and kinetics upon T cell activation. Scientific Reports 2017, 7, 6994 (1-14).
- Fujita, H.; Zhong, C.; Arai, S.; Suzuki, M. Bright Dots and Smart Optical Microscopy to Probe Intracellular Events in Single Cells. Frontiers in Bioengineering and Biotechnology 2019, 6, 204 (1-7).
- Li, G.; Gong, C. High-Contrast Interferometric Imaging of Single-Molecule Dynamics on Optical Fibers. Cornell University arXiv:2510.10061v1 [physics.optics] 2025, 1-19.
- Dahal, L.; Walther, N.; Tjian, R.; Darzacq, X.; Graham, T. G. W. Single-molecule tracking (SMT): a window into live-cell transcription biochemistry. Biochemical Society Transactions 2023, 51, 557–569.
- Kamarulzaman, L.; Kim, S.; Hidaka, T.; Tsuchida, M.; Taniguchi, Y. Comprehensive molecular profiling of single-cell proteome via gel electrophoresis and 3D single-molecule imaging. bioRxiv 2026, preprint 1-30, . [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



