Submitted:
13 February 2026
Posted:
27 February 2026
You are already at the latest version
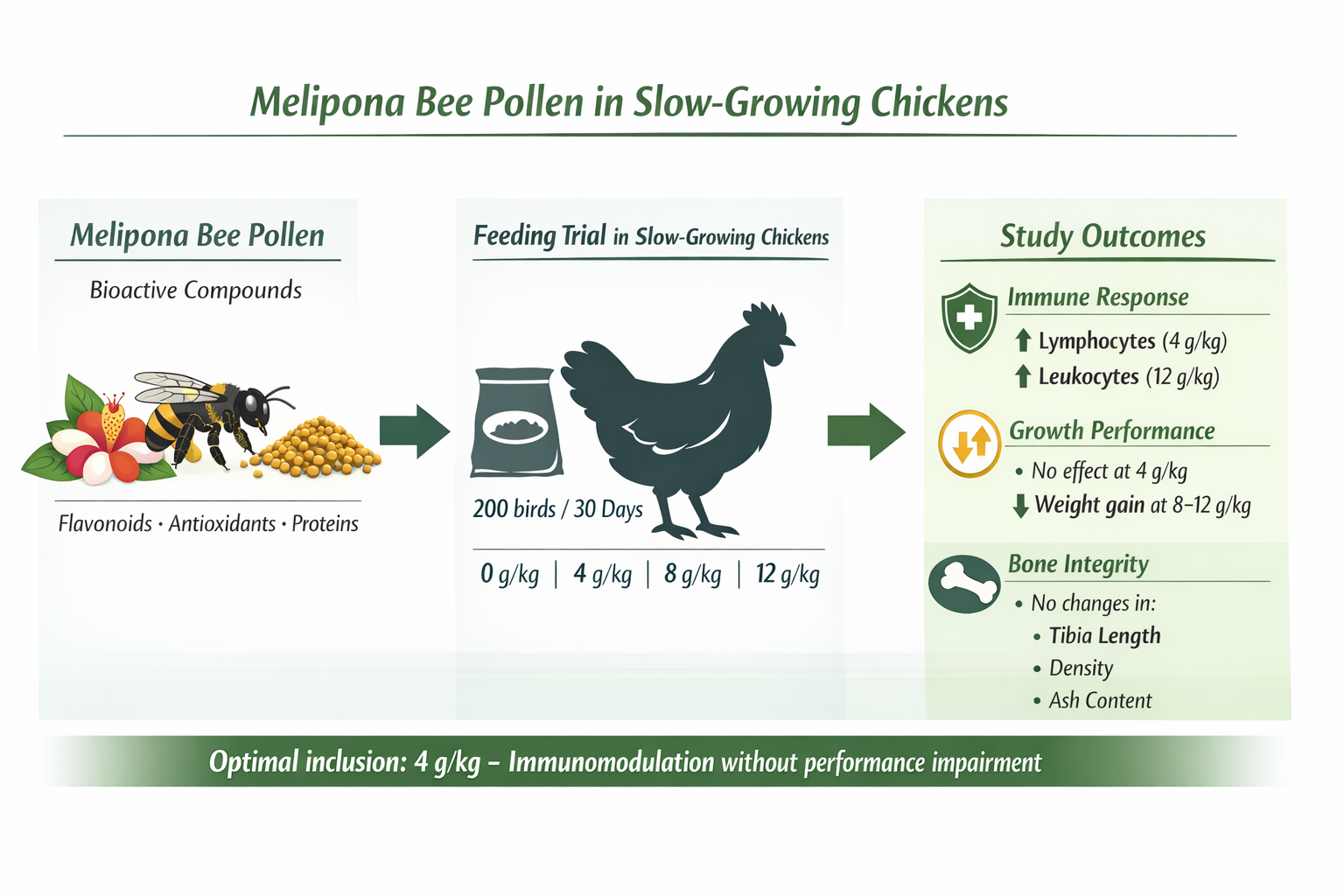
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Facilities, Poultry and Treatments
2.2. Productive Performance
2.3. Yield of Carcasses, Noble Cuts, Edible Viscera and Lymphoid Organs
2.4. Blood Analysis
2.5. Bone Histomorphometry
2.6. Statistical Procedures
3. Results
3.1. Growth Performance
3.2. Blood Parameters
3.3. Bone Histomorphometry
| Variables | TC | T1 | T2 | T3 | SEM | P-value |
|---|---|---|---|---|---|---|
| Length (mm) | 77.54 | 76.15 | 77.33 | 76.06 | 0.988 | 0.617 |
| Diameter (mm) | 6.15 | 6.07 | 6.08 | 5.78 | 0.151 | 0.356 |
| Weight (g) | 2.36 | 2.40 | 2.34 | 2.22 | 0.129 | 0.767 |
| Volume (cm3) | 2.40 | 2.50 | 2.50 | 2.40 | 0.149 | 0.928 |
| Density (g/cm3) | 1.00 | 0.97 | 0.94 | 0.93 | 0.047 | 0.689 |
| Ash (%) | 45.68 | 45.86 | 42.14 | 46.35 | 1.648 | 0.287 |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abd El-Aziz, A; Abo Ghanima, M; Mota-Rojas, D; Sherasiya, A; Ciani, F; El-Sabrout, K. Bee Products for Poultry and Rabbits: Current Challenges and Perspectives. Animals [Internet] 2023, 13(22), 3517. Available online: https://www.mdpi.com/2076-2615/13/22/3517. [CrossRef]
- El Ghouizi, A; Bakour, M; Laaroussi, H; Ousaaid, D; El Menyiy, N; Hano, C; et al. Bee Pollen as Functional Food: Insights into Its Composition and Therapeutic Properties. Antioxidants [Internet] 2023, 12(3), 557. Available online: https://www.mdpi.com/2076-3921/12/3/557. [CrossRef]
- Tsalsabilla, AT; Susilo, A; Amertaningtyas, D. Dietary Applications of Bee Pollen: Nutritional Composition, Health Benefits, and Applications. Asian J Food Res Nutr [Internet] 2025, 4(3), 758–66. Available online: https://journalajfrn.com/index.php/AJFRN/article/view/286. [CrossRef]
- Khalifa, SAM; Elashal, MH; Yosri, N; Du, M; Musharraf, SG; Nahar, L; et al. Bee Pollen: Current Status and Therapeutic Potential. Nutrients [Internet] 2021, 13(6), 1876. Available online: https://www.mdpi.com/2072-6643/13/6/1876. [CrossRef]
- Cavallero, A; Vidotto, F; Sbrana, C; Peres Fabbri, L; Petroni, G; Gabriele, M. Antioxidant-Rich Polyfloral Bee Pollen Exerts Antimicrobial Activity and Anti-Inflammatory Effect in A549 Lung Epithelial Cells by Modulating the NF-κB Pathway. Foods [Internet] 2025, 14(5), 802. Available online: https://www.mdpi.com/2304-8158/14/5/802. [CrossRef]
- Anjum, SI; Ullah, A; Gohar, F; Raza, G; Khan, MI; Hameed, M; et al. Bee pollen as a food and feed supplement and a therapeutic remedy: recent trends in nanotechnology. Front Nutr 2024, 11. [Google Scholar] [CrossRef]
- Urcan, AC; Criste, AD; Dezmirean, DS; Bobiș, O; Bonta, V; Burtescu, RF; et al. Enhancing Antioxidant and Antimicrobial Activities in Bee-Collected Pollen through Solid-State Fermentation: A Comparative Analysis of Bioactive Compounds. Antioxidants [Internet] 2024, 13(3), 292. Available online: https://www.mdpi.com/2076-3921/13/3/292. [CrossRef]
- Algethami, JS; El-Wahed, AAA; Elashal, MH; Ahmed, HR; Elshafiey, EH; Omar, EM; et al. Bee Pollen: Clinical Trials and Patent Applications. Nutrients [Internet] 2022, 14(14), 2858. Available online: https://www.mdpi.com/2072-6643/14/14/2858. [CrossRef]
- Pinheiro, SRF; Dourado, LRB; Silva, EP. Nutrição de aves caipiras criadas em sistema semiconfinado. In Nutrição de Não Ruminantes; Sakomura, NK, Silva, JH., Costa, FG., Fernandes, JB., Hauschild, L, Eds.; FUNEP: Jaboticabal, 2014; p. 678. [Google Scholar]
- Sakomura, NK; Rostagno, HS. Métodos de Pesquisa em Nutrição de Monogástricos, 2da ed.; Funep, 2016; p. 262 p. [Google Scholar]
- Yaeger, R. Atlas of Avian Hematology. Am J Trop Med Hyg [Internet] 1963, 12(1), 118. Available online: https://www.ajtmh.org/view/journals/tpmd/12/1/article-p118_a.xml. [CrossRef]
- Campbell, TW. Exotic Animal Hematology and Cytology; Wiley, 2015. [Google Scholar]
- Onasanya, GO; Oke, FO; Sanni, TM; Muhammad, AI. Parameters Influencing Haematological, Serum and Bio-Chemical References in Livestock Animals under Different Management Systems. Open J Vet Med [Internet] 2015, 05(08), 181–9. Available online: http://www.scirp.org/journal/doi.aspx?DOI=10.4236/ojvm.2015.58025. [CrossRef]
- Sánchez-Torres, L; Arredondo-Castro, M; Orozco-Benítez, G; Gutiérrez-Arenas, D; Carrillo-Beltrán, J; Lepe-Aguilar, R; et al. Variables hematológicas en aves deportivas, ganso común, pato doméstico, pato azteca, guajolote y pollo de engorda. In Abanico Agrofor; Jan 2021; p. 3. [Google Scholar]
- Aguilar, V. J; Zea, M. O; Vílchez, P. C. Rendimiento Productivo e Integridad Ósea de Pollos de Carne en Respuesta a Suplementación Dietaria con Cuatro Fuentes de Fitasa Comercial. Rev Investig Vet del Perú [Internet] 2018, 29(1), 169–79. Available online: https://revistas.gnbit.net/index.php/veterinaria/article/view/14078. [CrossRef]
- Li, QQ; Wang, K; Marcucci, MC; Sawaya, ACHF; Hu, L; Xue, XF; et al. Nutrient-rich bee pollen: A treasure trove of active natural metabolites. J Funct Foods [Internet] 2018, 49, 472–84. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1756464618304845. [CrossRef]
- Batista Rodrigues, R; Avelar Pucci, LE; Uczay, J; Molinari, M; Lazzari, R; Uczay, M. Pólen apícola como aditivo em dietas para frangos de corte. Nativa [Internet] 2018, 6(5), 551–6. Available online: https://periodicoscientificos.ufmt.br/ojs/index.php/nativa/article/view/5865. [CrossRef]
- Al-Kahtani, SN; Alaqil, AA; Abbas, AO. Modulation of Antioxidant Defense, Immune Response, and Growth Performance by Inclusion of Propolis and Bee Pollen into Broiler Diets. Animals [Internet] 2022, 12(13), 1658. Available online: https://www.mdpi.com/2076-2615/12/13/1658. [CrossRef]
- Haščík, P; Pavelková, A; Arpášová, H; Čuboň, J; Kačániová, M; Kunová, S. The effect of bee products and probiotics on meat performance of broiler chickens. J Microbiol Biotechnol food Sci [Internet] 2019, 9(1), 88–92. Available online: https://office2.jmbfs.org/index.php/JMBFS/article/view/8894. [CrossRef]
- Hashmi, MS; Haseik, P; Elimam, I; Garlik, J; Marek, B; Miroslava, K. Effects of Bee Pollen on the Technical and Allocative Efficiency of Meat Production of Ross 308 Broiler. Int J Poult Sci [Internet] 2012, 11(11), 689–95. Available online: https://www.scialert.net/abstract/?doi=ijps.2012.689.695. [CrossRef]
- Nemauluma, MFD; Ng’ambi, JW; Kolobe, SD; Malematja, E; Manyelo, TG; Chitura, T. Bee pollen an alternative to growth promoters for poultry production-A review. Appl Ecol Environ Res [Internet] 2022, 20(5), 3817–32. Available online: https://aloki.hu/pdf/2005_38173832.pdf. [CrossRef]
- Nemauluma, MFD; Manyelo, TG; Ng’ambi, JW; Kolobe, SD; Malematja, E. Effects of bee pollen inclusion on performance and carcass characteristics of broiler chickens. Poult Sci [Internet] 2023, 102(6), 102628. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0032579123001529. [CrossRef]
- Prakatur, I; Miskulin, M; Pavic, M; Marjanovic, K; Blazicevic, V; Miskulin, I; et al. Intestinal Morphology in Broiler Chickens Supplemented with Propolis and Bee Pollen. Animals [Internet] 2019, 9(6), 301. Available online: https://www.mdpi.com/2076-2615/9/6/301. [CrossRef]
- Sevim, B. Effects of supplemental bee pollen on performance, meat quality, serum constituents and immunity system in growing quails. S Afr J Anim Sci [Internet] 2022, 51(6), 745–51. Available online: https://www.ajol.info/index.php/sajas/article/view/224910. [CrossRef]
- Genova, JL; Rodrigues, RB; Martins, JS; Uczay, M; Henriques, JKS. Própolis e pólen apícola na nutrição de animais não ruminantes. Arch Zootec [Internet] 2020, 69(265), 124–31. Available online: http://www.uco.es/ucopress/az/index.php/az/article/view/5048. [CrossRef]
- Korani, S; Khalesi, N; Korani, M; Jamialahmadi, T; Sahebkar, A. Applications of honeybee-derived products in bone tissue engineering. Bone Reports [Internet] 2024, 20, 101740. Available online: https://linkinghub.elsevier.com/retrieve/pii/S235218722400007X. [CrossRef] [PubMed]
- Tomaszewska E, Knaga S, Dobrowolski P, Lamorski K, Jabłoński M, Tomczyk-Warunek A, et al. The effect of bee pollen on bone biomechanical strength and trabecular bone histomorphometry in tibia of young Japanese quail (Coturnix japonica). Loor JJ, editor. PLoS One [Internet]. 2020 Mar 18;15(3):e0230240. Available from: https://dx.plos.org/10.1371/journal.pone.0230240.
| Ingredient % | T control | T 1 | T 2 | T 3 |
|---|---|---|---|---|
| Fishmeal | 6.00 | 6.00 | 6.00 | 6.00 |
| Soy meal | 25.10 | 25.10 | 25.10 | 25.10 |
| Corn | 66.01 | 65.66 | 65.36 | 65.94 |
| Bee pollen | 0.00 | 0.40 | 0.80 | 1.20 |
| Calcium carbonate | 1.10 | 1.10 | 1.10 | 1.10 |
| Monocalcium phosphate | 0.80 | 0.80 | 0.80 | 0.80 |
| Choline chloride | 0.20 | 0.20 | 0.20 | 0.20 |
| Common salt | 0.20 | 0.20 | 0.20 | 0.20 |
| vit-min1 supplement | 0.10 | 0.10 | 0.10 | 0.10 |
| DL – methionine | 0.30 | 0.30 | 0.30 | 0.30 |
| L – lysine | 0.05 | 0.05 | 0.05 | 0.05 |
| Zinc bacitracina | 0.05 | 0.00 | 0.00 | 0.00 |
| Coccidiostat | 0.04 | 0.04 | 0.04 | 0.04 |
| Fungicab | 0.05 | 0.05 | 0.05 | 0.05 |
| Total | 100.00 | 100.00 | 100.00 | 100.00 |
| Food | Humidity1 | Dry matter1 | Mineral matter1 | Ethereal extract1 | Crude fiber2 | Crude protein2 | Nitrogen-free extract3 | Gross energy2 kcal/kg |
|---|---|---|---|---|---|---|---|---|
| Control treatment | 11.84 | 88.16 | 6.64 | 2.89 | 2.18 | 20.51 | 55.95 | 3844 |
| Treatment 1 | 11.90 | 88.10 | 6.52 | 1.96 | 2.31 | 21.04 | 56.27 | 3897 |
| Treatment 2 | 11.93 | 88.07 | 6.33 | 2.02 | 2.49 | 21.84 | 55.40 | 3879 |
| Treatment 3 | 11.92 | 88.08 | 6.08 | 1.98 | 2.30 | 21.17 | 56.55 | 3876 |
| Pollen | 14.34 | 85.66 | 5.45 | 2.80 | 0.45 | 45.55 | 31.40 | 5259 |
| Productive performance | TC | T1 | T2 | T3 | SEM | P-value |
|---|---|---|---|---|---|---|
| Initial weight (g) | 38.8 | 38.6 | 38.8 | 38.8 | 0.212 | 0.8795 |
| Final weight (g) | 780.2a | 772.2a | 713.4b | 704.0b | 11.707 | 0.0003 |
| Weight gain (g) | 741.2a | 733.4a | 674.8b | 665.2b | 11.707 | 0.0003 |
| Feed intake (g) | 1319.2 | 1344.4 | 1283.0 | 1300.0 | 24.042 | 0.3469 |
| Feed conversion (g/g) | 1.779b | 1,835ab | 1,904ab | 1.957a | 0.043 | 0.0494 |
| Variables | TC | T1 | T2 | T3 | SEM | P-value |
|---|---|---|---|---|---|---|
| Carcass (%) | 62.76 | 60.91 | 60.89 | 61.33 | 0.917 | 0.4494 |
| Breast (%) | 25.03 | 22.77 | 23.791 | 23.91 | 0.961 | 0.4527 |
| Thigh (%) | 15.92 | 16.71 | 16.94 | 15.99 | 0.504 | 0.4065 |
| Drumstick (%) | 17.53 | 17.22 | 17.47 | 17.79 | 0.487 | 0.8797 |
| Wing (%) | 14.99 | 15.11 | 15.34 | 15.05 | 0.300 | 0.8586 |
| Heart (%) | 0.87 | 0.89 | 0.96 | 0.91 | 0.023 | 0.0743 |
| Gizzard (%) | 3.57 | 3.93 | 3.93 | 3.94 | 0.183 | 0.4111 |
| Liver (%) | 3.39 | 3.39 | 3.19 | 3.20 | 0.104 | 0.3621 |
| Timo (%) | 0.61 | 0.64 | 0.59 | 0.78 | 0.086 | 0.4338 |
| Bursa (%) | 0.57 | 0.55 | 0.54 | 0.51 | 0.048 | 0.8767 |
| Spleen (%) | 0.23 | 0.22 | 0.24 | 0.23 | 0.018 | 0.9070 |
| Variables | TC | T1 | T2 | T3 | SEM | P-value |
|---|---|---|---|---|---|---|
| Hematocrit (g/dL) | 27.00 | 27.80 | 26.90 | 28.40 | 0.639 | 0.3315 |
| Hemoglobin (g/dL) | 8.91 | 9.18 | 8.88 | 9.37 | 0.211 | 0.3364 |
| Leukocytes (cells/mm3) | 44600b | 43000b | 37000b | 71100a | 2736.101 | <0.0001 |
| Heterophiles (%) | 37.40 | 28.10 | 38.50 | 40.20 | 4.045 | 0.1882 |
| Lymphocytes (%) | 52.50ab | 57.40a | 42.30b | 46.20ab | 3.359 | 0.0274 |
| Eosinophils (%) | 7.30ab | 9.70a | 11.30a | 4.40b | 1.228 | 0.0061 |
| Basophils (%) | 0.60 | 0.70 | 0.90 | 0.60 | 0.260 | 0.8275 |
| Monocytes (%) | 2.20b | 4.10ab | 7.00ab | 8.60a | 1.246 | 0.0099 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




