Submitted:
14 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design & Participants
2.2. Ethics
2.3. Statistical Analysis
3. Results
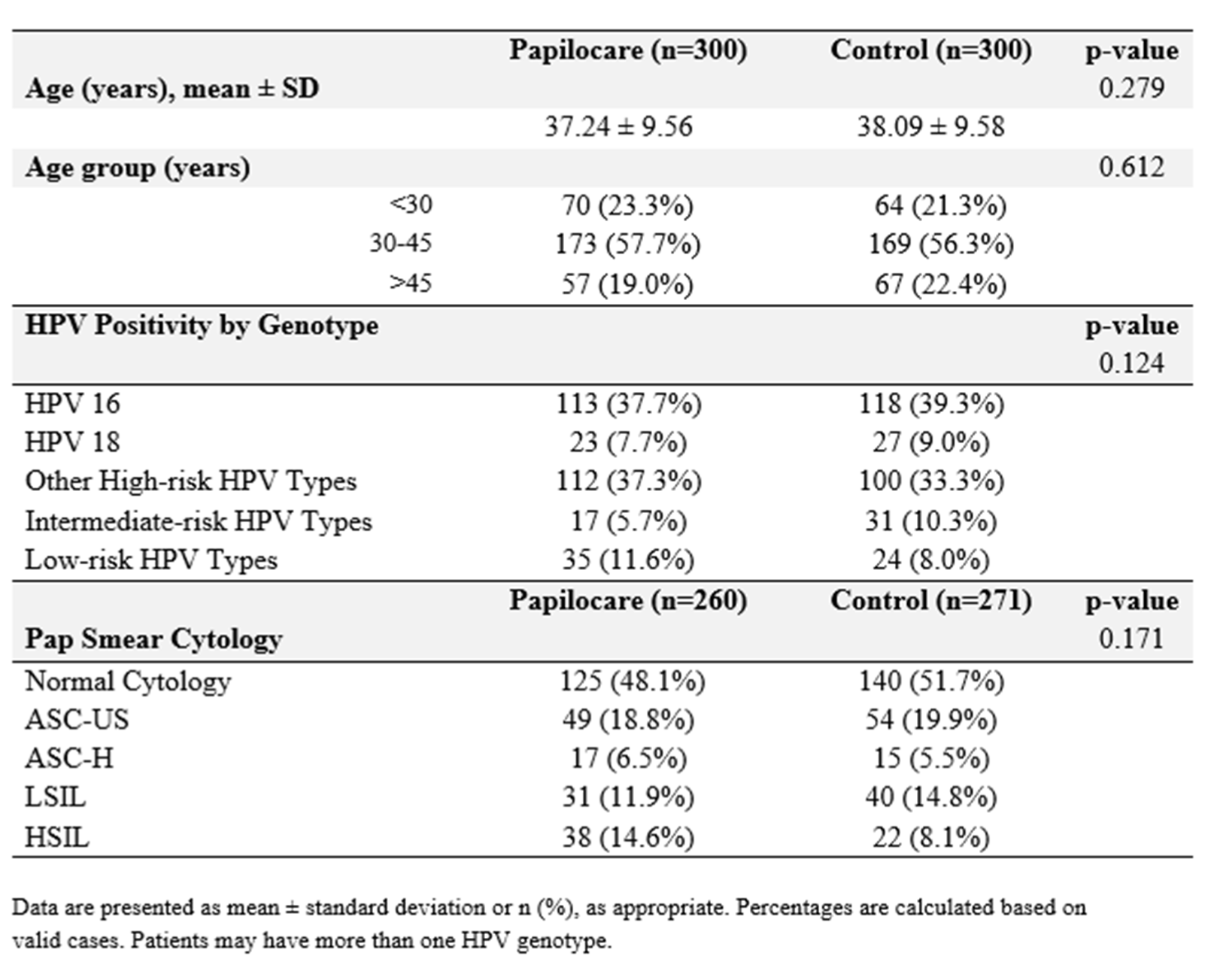
3.1. Study Population and Baseline Characteristics
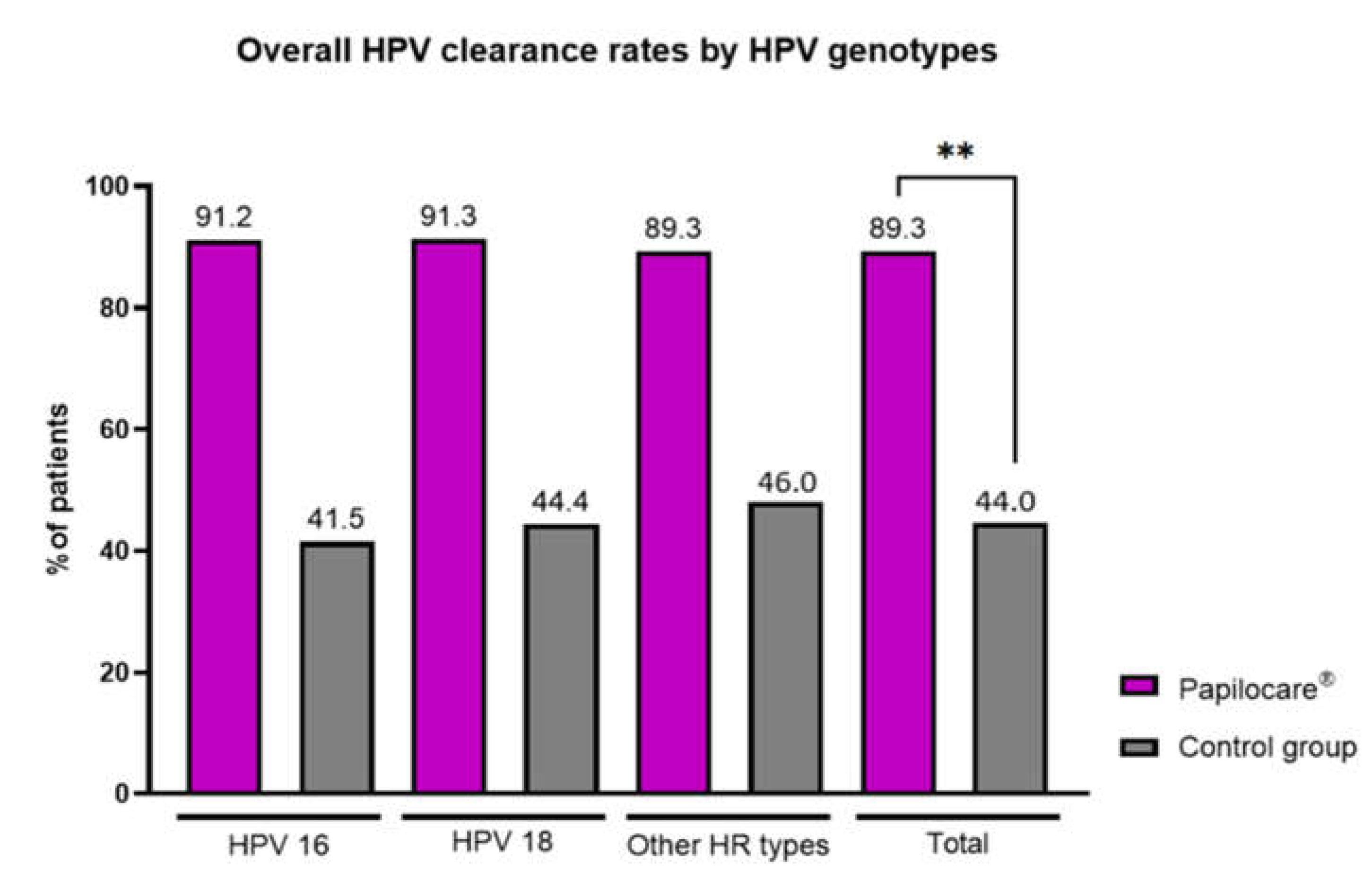
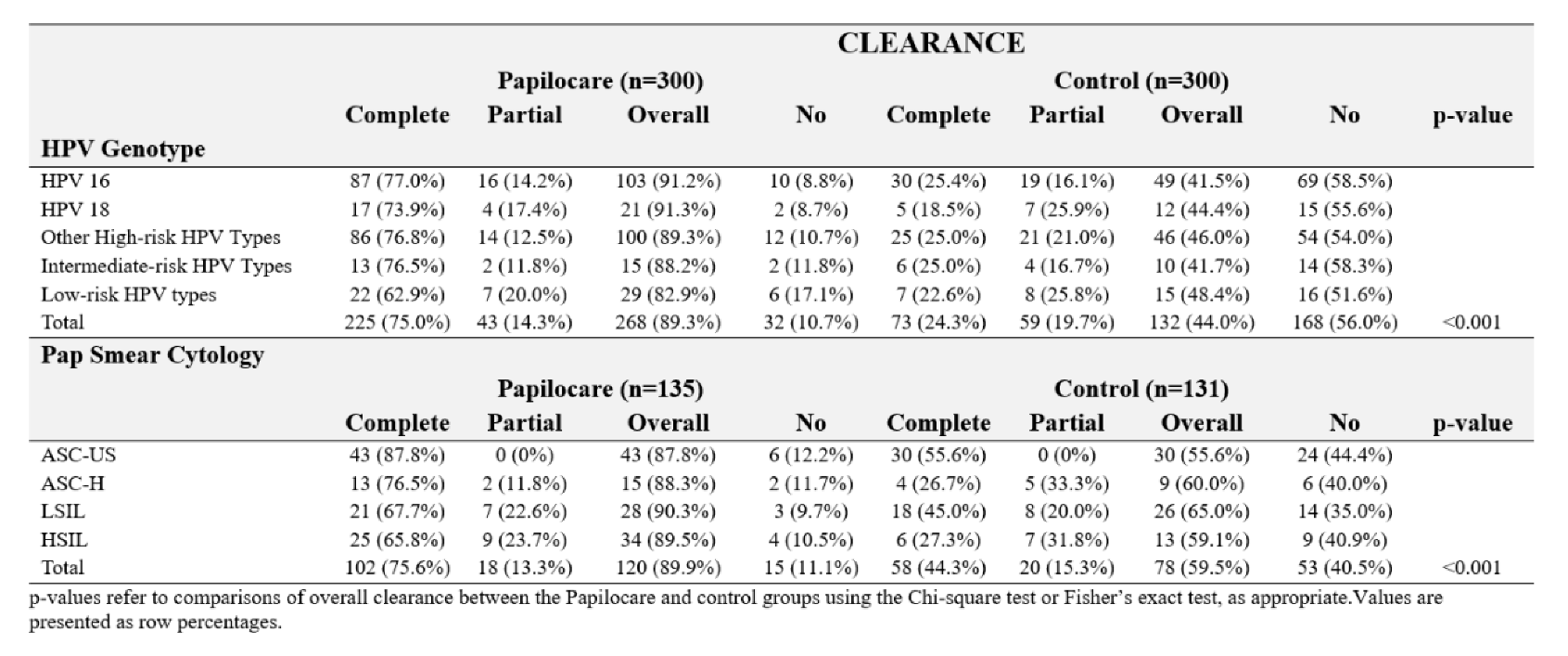
3.2. HPV Clearance Outcomes
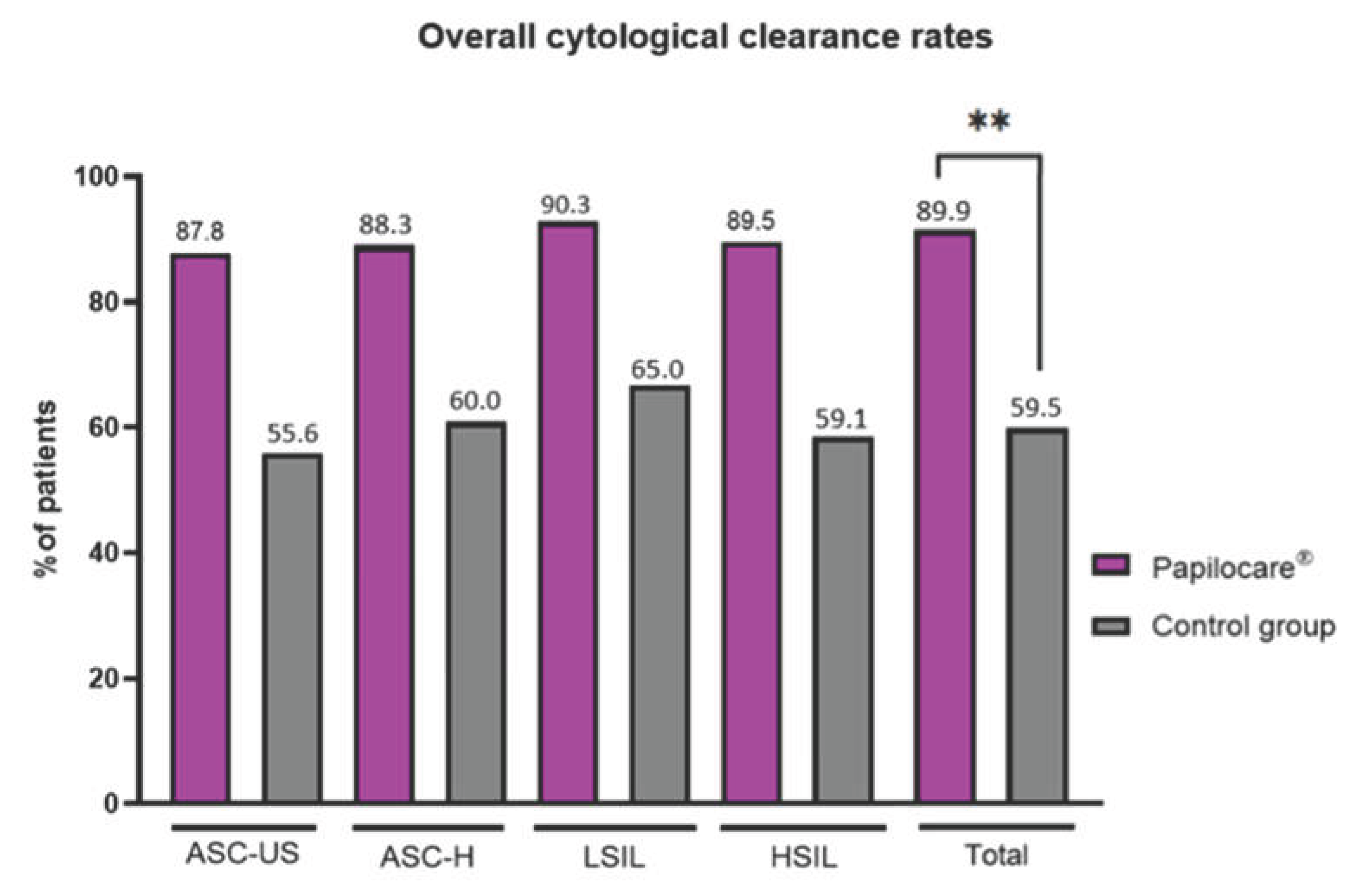
3.3. Cytological Clearance Outcomes
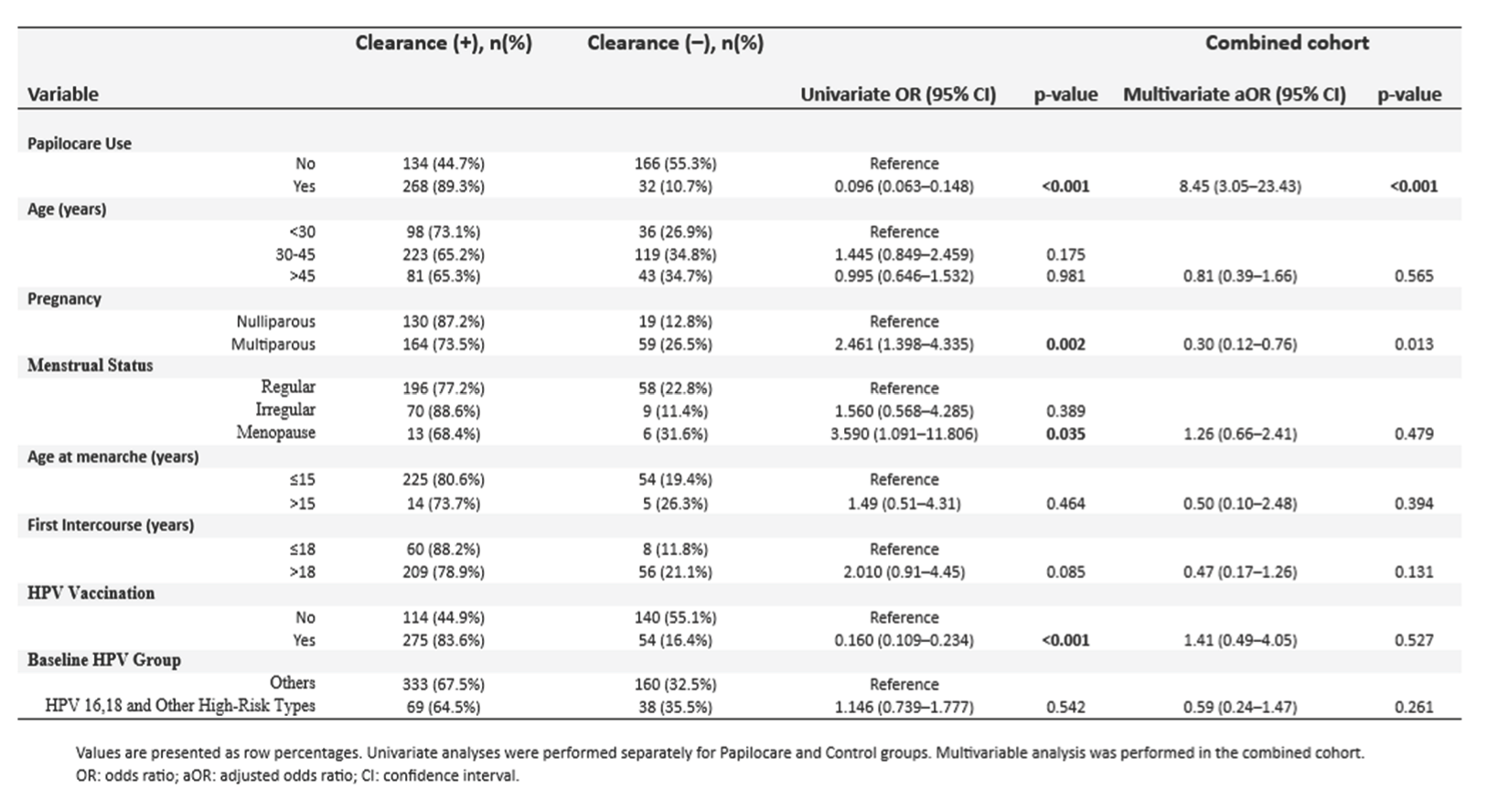
3.4. Multivariable Analysis
4. Discussion
4.1. Strengths and Limitations
4.1. Implications for Practice and Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HPV | Human Papillomavirus |
| LEEP | Loop electrosurgical excision procedure |
| ASC-US | Atypical squamous cells of undetermined significance |
| ASC-H | Atypical squamous cells—cannot exclude high-grade squamous intraepithelial lesion |
| LSIL | Low-grade squamous intraepithelial lesions |
| HSIL | Hifgh-grade squamous intraepithelial lesions |
| CIN | Cervical intraepithelial neoplasia |
References
- Plotzker, RE; Vaidya, A; Pokharel, U; Stier, EA. Sexually transmitted human papillomavirus: update in epidemiology, prevention, and management. Infect Dis Clin. 2023, 37(2), 289–310. [Google Scholar]
- He, S; Zhu, G; Zhou, Y; Yang, B; Wang, J; Wang, Z; et al. Predictive models for personalized precision medical intervention in spontaneous regression stages of cervical precancerous lesions. J Transl Med. 2024, 22(1), 686. [Google Scholar] [CrossRef]
- Ntuli, L; Mtshali, A; Mzobe, G; Liebenberg, LJP; Ngcapu, S. Role of immunity and vaginal microbiome in clearance and persistence of human papillomavirus infection. Front Cell Infect Microbiol. 2022, 12, 927131. [Google Scholar] [CrossRef] [PubMed]
- Cui, M; Wu, Y; Liu, Z; Liu, Y; Fan, L. Advances in the interrelated nature of vaginal microecology, HPV infection, and cervical lesions. Front Cell Infect Microbiol. 2025, 15, 1608195. [Google Scholar] [CrossRef]
- World Health Organization. Cervical cancer [Internet]. Fact Sheet. 2024. Available online: https://www.who.int/news-room/fact-sheets/detail/cervical-cancer.
- Bray, F; Laversanne, M; Sung, H; Ferlay, J; Siegel, RL; Soerjomataram, I; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024, 74(3), 229–63. [Google Scholar] [CrossRef]
- Osmani, V; Hörner, L; Nkurunziza, T; Rank, S; Tanaka, LF; Klug, SJ. Global prevalence of cervical human papillomavirus in women aged 50 years and older with normal cytology: a systematic review and meta-analysis. The Lancet Microbe [Internet] Available from. 2025, 6(1). [Google Scholar] [CrossRef] [PubMed]
- Wu, J; Jin, Q; Zhang, Y; Ji, Y; Li, J; Liu, X; et al. Global burden of cervical cancer: current estimates, temporal trend and future projections based on the GLOBOCAN 2022. J Natl Cancer Cent [Internet] 2025, 5(3), 322–9. Available online: https://www.sciencedirect.com/science/article/pii/S2667005425000134. [CrossRef]
- Kakotkin, V V; Semina, E V; Zadorkina, TG; Agapov, MA. Prevention strategies and early diagnosis of cervical cancer: current state and prospects. Diagnostics 2023, 13(4), 610. [Google Scholar] [CrossRef]
- O’Neill, AM; Dwyer, R. Primary prevention of cervical cancer in women: Human papillomavirus vaccine. Eur J Obstet Gynecol Reprod Biol. 2023, 281, 29–31. [Google Scholar] [CrossRef]
- Wirtz, C; Mohamed, Y; Engel, D; Sidibe, A; Holloway, M; Bloem, P; et al. Integrating HPV vaccination programs with enhanced cervical cancer screening and treatment, a systematic review. Vaccine 2022, 40, A116–23. [Google Scholar] [CrossRef]
- Welby, S; Rosillon, D; Feng, Y; Borys, D. Progression from human papillomavirus (HPV) infection to cervical lesion or clearance in women (18–25 years): natural history study in the control arm subjects of AS04-HPV-16/18 vaccine efficacy study in China between 2008 and 2016. Expert Rev Vaccines 2022, 21(3), 407–13. [Google Scholar] [CrossRef] [PubMed]
- Bosch, FX; Robles, C; Diaz, M; Arbyn, M; Baussano, I; Clavel, C; et al. HPV-FASTER: broadening the scope for prevention of HPV-related cancer. Nat Rev Clin Oncol. 2016, 13(2), 119–32. [Google Scholar] [CrossRef] [PubMed]
- Nugent, D; Apoola, A; Coleman, H; Gilmour, C; Lawton, MD; Nori, A; et al. British association for sexual health and HIV national guideline for the management of anogenital warts in adults (2024). Int J STD AIDS 2024, 35(7), 498–509. [Google Scholar] [CrossRef]
- He, Z; Lin, J; He, Y; Liu, S. Polysaccharide-Peptide from Trametes versicolor: The Potential Medicine for Colorectal Cancer Treatment. Biomedicines 2022, 10(11). [Google Scholar] [CrossRef]
- Jędrzejewski, T; Pawlikowska, M; Sobocińska, J; Wrotek, S. COVID-19 and Cancer Diseases-The Potential of Coriolus versicolor Mushroom to Combat Global Health Challenges. Int J Mol Sci 2023, 24(5). [Google Scholar] [CrossRef]
- Saleh, MH; Rashedi, I; Keating, A. Immunomodulatory Properties of Coriolus versicolor: The Role of Polysaccharopeptide. Front Immunol [Internet]. 2017, pp. 8–2017. Available online: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2017.01087.
- Serrano, L; López, AC; González, SP; Palacios, S; Dexeus, D; Centeno-Mediavilla, C; et al. Efficacy of a Coriolus versicolor–Based Vaginal Gel in Women With Human Papillomavirus–Dependent Cervical Lesions: The PALOMA Study. J Low Genit Tract Dis [Internet] 2021, 25(2). Available online: https://journals.lww.com/jlgtd/fulltext/2021/04000/efficacy_of_a_coriolus_versicolor_based_vaginal.9.aspx. [CrossRef]
- Cortés Bordoy, J; de Santiago García, J; Agenjo González, M; Dexeus Carter, D; Fiol Ruiz, G; García Ferreiro, C; et al. Effect of a Multi-Ingredient Coriolus-versicolor-Based Vaginal Gel in Women with HPV-Dependent Cervical Lesions: The Papilobs Real-Life Prospective Study. Cancers (Basel) 2023, 15(15). [Google Scholar] [CrossRef]
- Gil-Antuñano, SP; Serrano Cogollor, L; López Díaz, AC; González Rodríguez, SP; Dexeus Carter, D; Centeno Mediavilla, C; et al. Efficacy of a Coriolusversicolor-Based Vaginal Gel in Human Papillomavirus-Positive Women Older Than 40 Years: A Sub-Analysis of PALOMA Study. J Pers Med. 2022, 12(10), 1559. [Google Scholar] [CrossRef] [PubMed]
- Solomon, D; Davey, D; Kurman, R; Moriarty, A; O’Connor, D; Prey, M; et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA 2002, 287(16), 2114–9. [Google Scholar] [CrossRef]
- Perkins, RB; Castle, G.R.; Chelmow, PE; Einstein, D; Garcia, MH; F. 2019 ASCCP Risk-Based Management Consensus Guidelines for Abnormal Cervical Cancer Screening Tests and Cancer Precursors. Journal of Lower Genital Tract Disease 2020, 24. [Google Scholar] [CrossRef]
- Cruz, A; Pimentel, L; Rodríguez-Alcalá, LM; Fernandes, T; Pintado, M. Health benefits of edible mushrooms focused on Coriolus versicolor: A review. J Food Nutr Res. 2016, 4(12), 773–81. [Google Scholar]
- Criscuolo, AA; Sesti, F; Piccione, E; Mancino, P; Belloni, E; Gullo, C; et al. Therapeutic Efficacy of a Coriolus versicolor-Based Vaginal Gel in Women with Cervical Uterine High-Risk HPV Infection: A Retrospective Observational Study. Adv Ther. 2021, 38(2), 1202–11. [Google Scholar] [CrossRef] [PubMed]
- Gálvez, D. Treatment of Erythroplasia and Cervical HPV in a Young Patient with a Coriolus versicolor Vaginal Gel. Hum PAPILLOMAVIRUS 2023, 30, 30–2. [Google Scholar]
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





