Submitted:
12 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
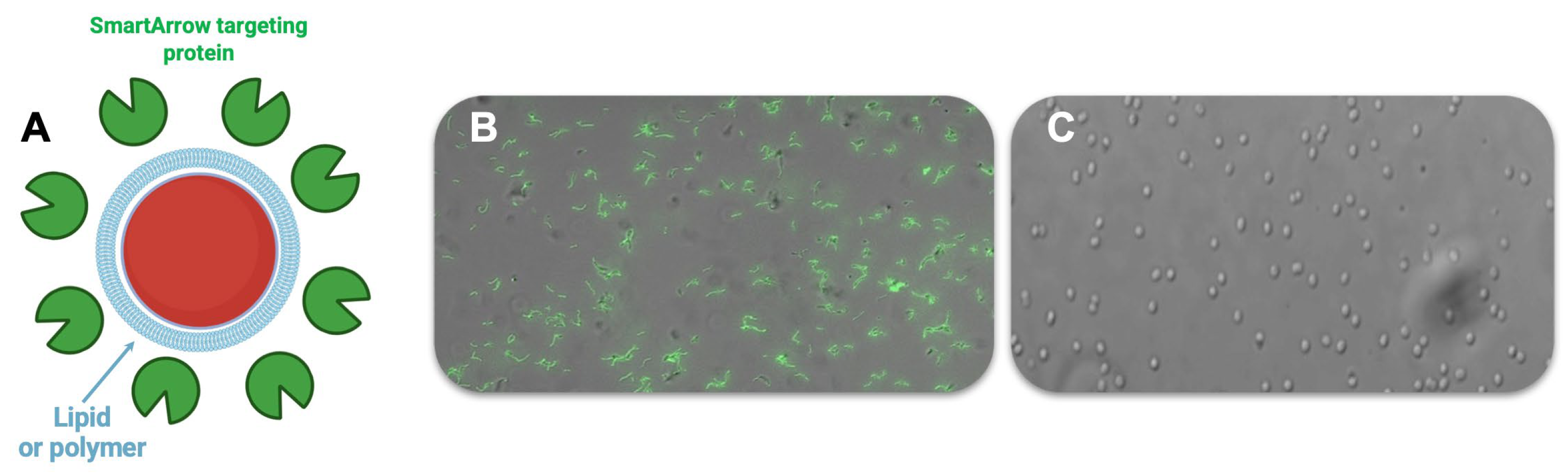
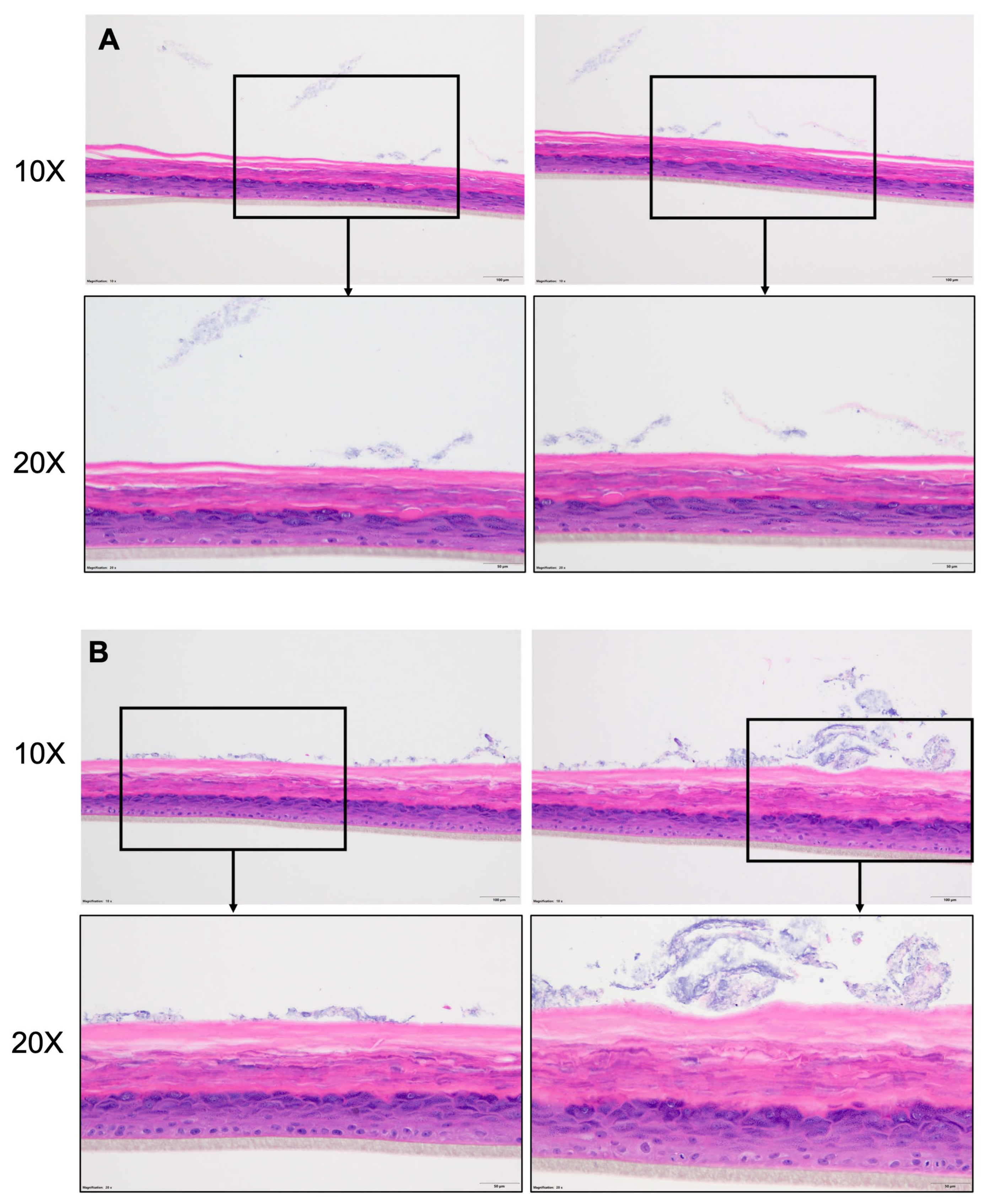
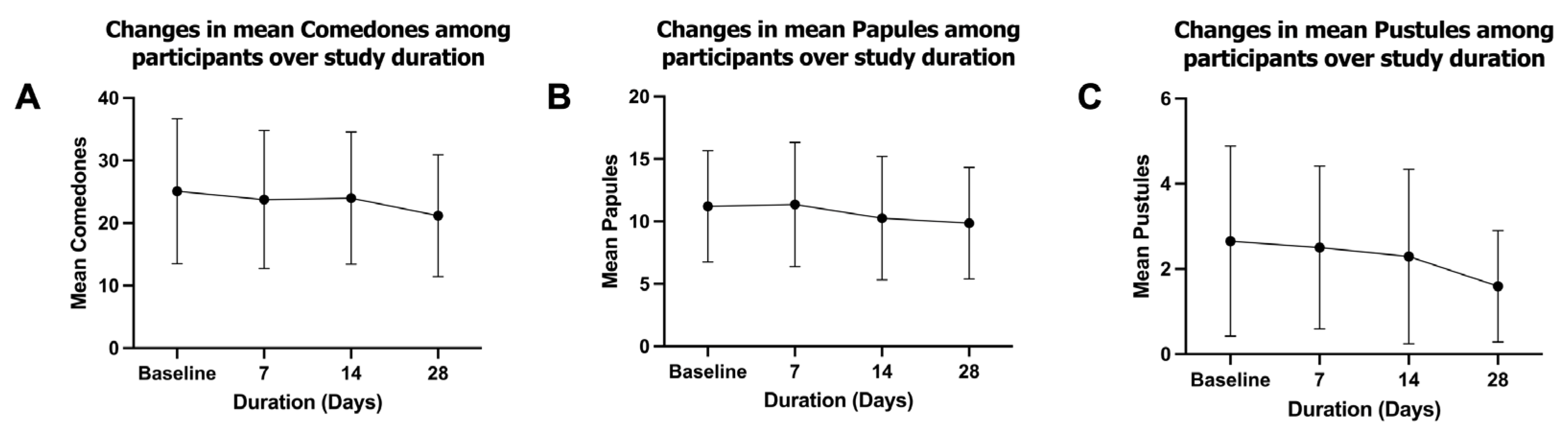
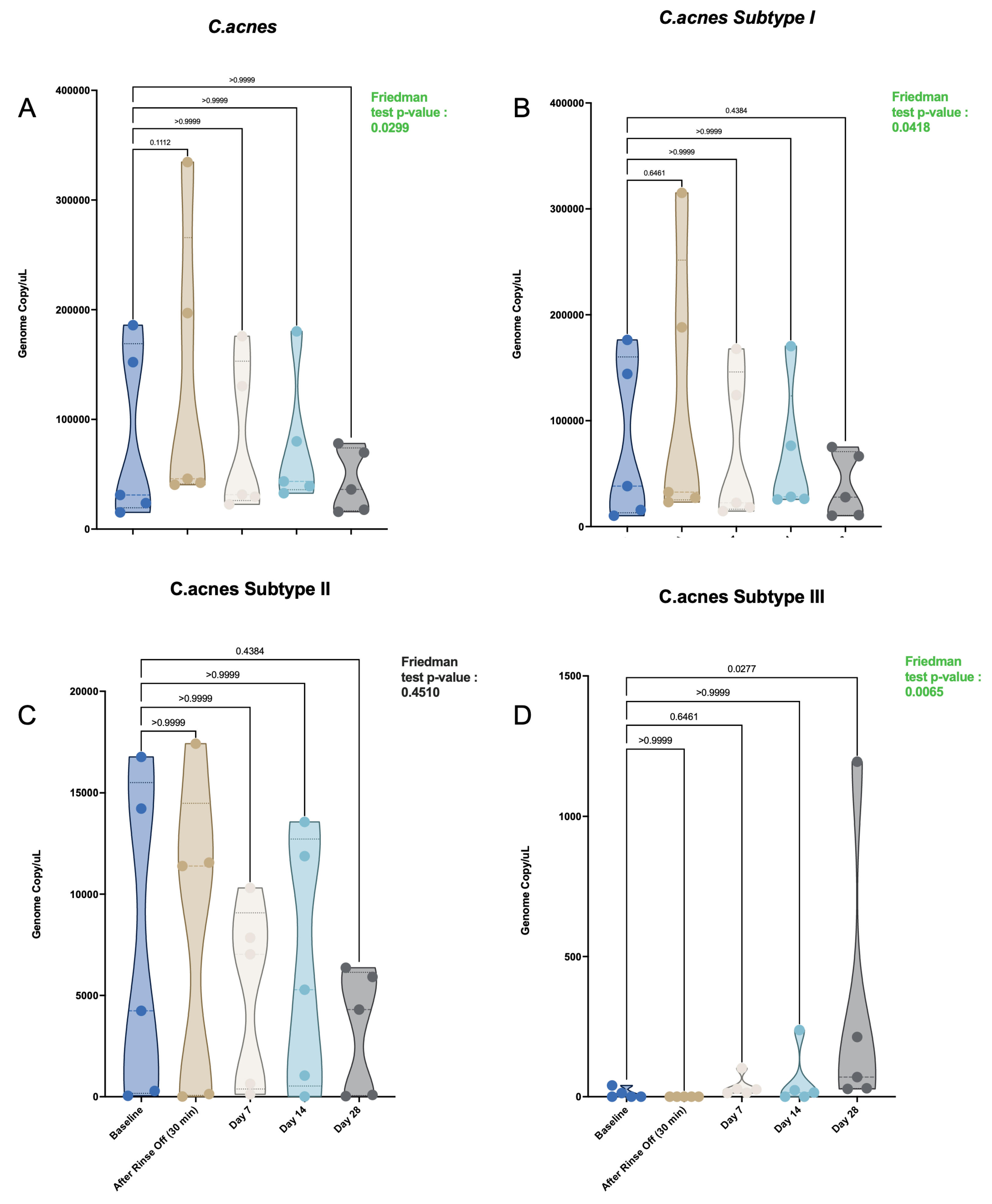
This review discusses the microbiology of acne vulgaris, a chronic inflammatory condition of the pilosebaceous unit that affects most adolescents and can persist into adulthood. The current standard of care consists largely of antibacterial interventions, based on the traditional view of Cutibacterium acnes as a pathogen. Alternative treatments are suggested by the "comedo switch" hypothesis, which attributes acne to aberrant differentiation of Lrig1+ sebaceous progenitor cells. While there is strong evidence to support this idea, it does not explain the efficacy of antibacterial interventions. We propose a unified mechanism wherein C. acnes phylotype IA1 can act as a trigger for the comedo switch. Unlike commensal strains, phylotype IA1 has high lipase activity, hydrolyzing sebum triglycerides into free fatty acids, specifically palmitic acid. This metabolite stimulates Lrig1+ progenitors, inducing inflammation and initial comedo formation. The review discusses C. acnes phylotypes, emphasizing known virulence factors of IA1, such as enhanced biofilm formation. We evaluate the efficacy and limitations of both old and new antibacterials and in this context present a novel material that disrupts C. acnes biofilms (SmartArrow™). In a trial of 34 subjects with mild-to-moderate acne, this non-antibiotic agent demonstrated a statistically significant reduction in inflammatory lesions and selectively depleted phylotype IA1 by 37%, without compromising microbiome diversity. Our findings support the causal role of C. acnes IA1 in acne inflammation as well as in the comedo switch, and suggest that biofilm disruption represents a promising therapeutic avenue.
Keywords:
1. Introduction
2. Cutibacterium Acnes
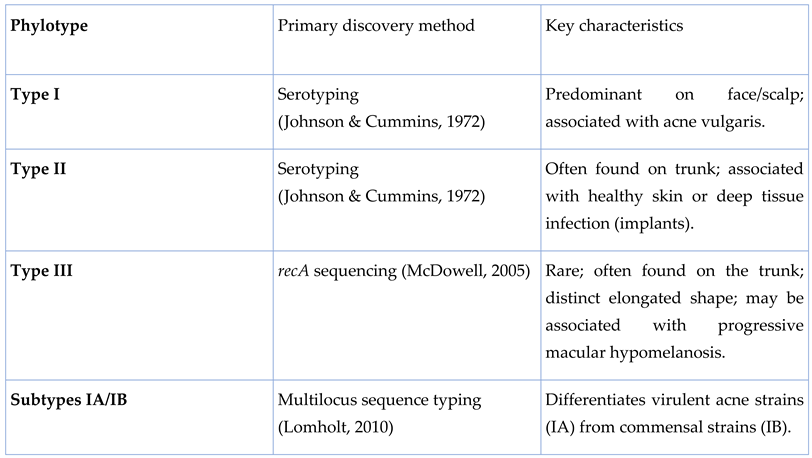 |
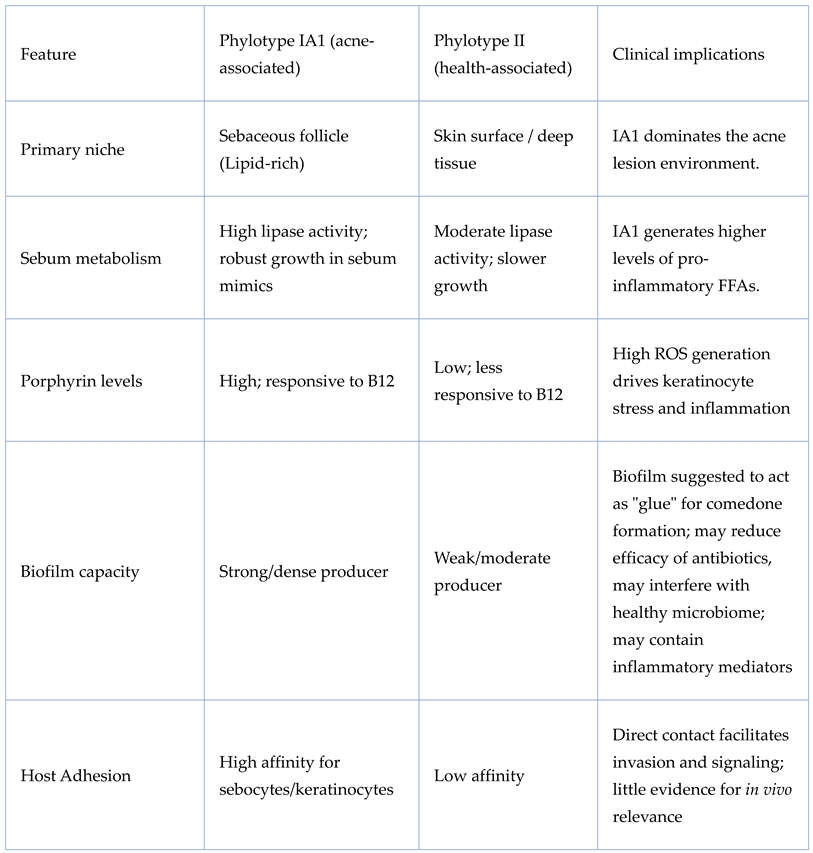 |
3. Cutibacterium Acnes and Comedogenesis
4. Antibacterial Interventions
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mohsin, N.; Hernandez, L.E.; Martin, M.R.; Does, A.V.; Nouri, K. Acne Treatment Review and Future Perspectives. Dermatologic Therapy 2022, 35, e15719. [Google Scholar] [CrossRef]
- Patangia, D.V.; Anthony Ryan, C.; Dempsey, E.; Paul Ross, R.; Stanton, C. Impact of Antibiotics on the Human Microbiome and Consequences for Host Health. Microbiologyopen 2022, 11, e1260. [Google Scholar] [CrossRef] [PubMed]
- Dessinioti, C.; Katsambas, A.D. The Role of Propionibacterium Acnes in Acne Pathogenesis: Facts and Controversies. Clinics in Dermatology 2010, 28, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Karoglan, A.; Gollnick, H.P.M. [Acne]. Hautarzt 2021, 72, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Clayton, R.W.; Göbel, K.; Niessen, C.M.; Paus, R.; Steensel, M.A.M.; Lim, X. Homeostasis of the Sebaceous Gland and Mechanisms of Acne Pathogenesis. Br J Dermatol 2019, 181, 677–690. [Google Scholar] [CrossRef]
- Van Steensel, M.A.M. The Genetics of Acne. Ann Hum Genet 2025, 89, 333–341. [Google Scholar] [CrossRef]
- Saurat, J.-H. Strategic Targets in Acne: The Comedone Switch in Question. Dermatology 2015, 231, 105–111. [Google Scholar] [CrossRef]
- Dagnelie, M.-A.; Corvec, S.; Saint-Jean, M.; Nguyen, J.-M.; Khammari, A.; Dréno, B. Cutibacterium Acnes Phylotypes Diversity Loss: A Trigger for Skin Inflammatory Process. Journal of the European Academy of Dermatology and Venereology 2019, 33, 2340–2348. [Google Scholar] [CrossRef]
- Scholz, C.F.P.; Kilian, M. The Natural History of Cutaneous Propionibacteria, and Reclassification of Selected Species within the Genus Propionibacterium to the Proposed Novel Genera Acidipropionibacterium Gen. Nov., Cutibacterium Gen. Nov. and Pseudopropionibacterium Gen. Nov. International Journal of Systematic and Evolutionary Microbiology 2016, 66, 4422–4432. [Google Scholar] [CrossRef]
- Johnson, J.L.; Cummins, C.S. Cell Wall Composition and Deoxyribonucleic Acid Similarities among the Anaerobic Coryneforms, Classical Propionibacteria, and Strains of Arachnia Propionica. J Bacteriol 1972, 109, 1047–1066. [Google Scholar] [CrossRef]
- Webster, G.F.; Cummins, C.S. Use of Bacteriophage Typing to Distinguish Propionibacterium Acne Types I and II. J Clin Microbiol 1978, 7, 84–90. [Google Scholar] [CrossRef]
- McDowell, A.; Valanne, S.; Ramage, G.; Tunney, M.M.; Glenn, J.V.; McLorinan, G.C.; Bhatia, A.; Maisonneuve, J.-F.; Lodes, M.; Persing, D.H.; et al. Propionibacterium Acnes Types I and II Represent Phylogenetically Distinct Groups. Journal of Clinical Microbiology 2005. [Google Scholar] [CrossRef] [PubMed]
- Lomholt, H.B.; Kilian, M. Population Genetic Analysis of Propionibacterium Acnes Identifies a Subpopulation and Epidemic Clones Associated with Acne. PLoS ONE 2010, 5, e12277. [Google Scholar] [CrossRef] [PubMed]
- Fitz-Gibbon, S.; Tomida, S.; Chiu, B.-H.; Nguyen, L.; Du, C.; Liu, M.; Elashoff, D.; Erfe, M.C.; Loncaric, A.; Kim, J.; et al. Propionibacterium Acnes Strain Populations in the Human Skin Microbiome Associated with Acne. J Invest Dermatol 2013, 133, 2152–2160. [Google Scholar] [CrossRef] [PubMed]
- Dreno, B.; Dekio, I.; Baldwin, H.; Demessant, A.L.; Dagnelie, M.-A.; Khammari, A.; Corvec, S. Acne Microbiome: From Phyla to Phylotypes. Journal of the European Academy of Dermatology and Venereology 2024, 38, 657–664. [Google Scholar] [CrossRef]
- Zhang, N.; Yuan, R.; Xin, K.Z.; Lu, Z.; Ma, Y. Antimicrobial Susceptibility, Biotypes and Phylotypes of Clinical Cutibacterium (Formerly Propionibacterium) Acnes Strains Isolated from Acne Patients: An Observational Study. Dermatol Ther (Heidelb) 2019, 9, 735–746. [Google Scholar] [CrossRef]
- Ponraj, D.S.; Lange, J.; Falstie-Jensen, T.; Jørgensen, N.P.; Ravn, C.; Poehlein, A.; Brüggemann, H. Amplicon-Based Next-Generation Sequencing as a Diagnostic Tool for the Detection of Phylotypes of Cutibacterium Acnes in Orthopedic Implant-Associated Infections. Front. Microbiol. 2022, 13, 866893. [Google Scholar] [CrossRef]
- McDowell, A.; McLaughlin, J.; Layton, A.M. Is Cutibacterium (Previously Propionibacterium) Acnes a Potential Pathogenic Factor in the Aetiology of the Skin Disease Progressive Macular Hypomelanosis? J Eur Acad Dermatol Venereol 2021, 35, 338–344. [Google Scholar] [CrossRef]
- Li, C.; Ravikrishnan, A.; Wijaya, I.; Naim, A.N.M.; Gounot, J.-S.; Wearne, S.; On, Y.Y.; Ho, E.X.P.; Ang, Q.Y.; Chia, M.; et al. Large-Scale Skin Metagenomics Reveals Extensive Prevalence, Coordination, and Functional Adaptation of Skin Microbiome Dermotypes across Body Sites. 2025, 2025.04.24.650393. [Google Scholar] [CrossRef]
- Valanne, S.; McDowell, A.; Ramage, G.; Tunney, M.M.; Einarsson, G.G.; O’Hagan, S.; Wisdom, G.B.; Fairley, D.; Bhatia, A.; Maisonneuve, J.-F.; et al. CAMP Factor Homologues in Propionibacterium Acnes: A New Protein Family Differentially Expressed by Types I and II. Microbiology (Reading) 2005, 151, 1369–1379. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, C.; Tao, J.; Zeng, W.; Zhu, Z.; Yao, C.; Shang, Y.; Tang, J.; Jin, T. Insights into Microbial Dysbiosis and Cutibacterium Acnes CAMP Factor Interactions in Acne Vulgaris. Microb Genom 2025, 11, 001449. [Google Scholar] [CrossRef]
- Nazipi, S.; Stødkilde, K.; Scavenius, C.; Brüggemann, H. The Skin Bacterium Propionibacterium Acnes Employs Two Variants of Hyaluronate Lyase with Distinct Properties. Microorganisms 2017, 5, 57. [Google Scholar] [CrossRef]
- Hajam, I.A.; Katiki, M.; McNally, R.; Lázaro-Díez, M.; Kolar, S.; Chatterjee, A.; Gonzalez, C.; Paulchakrabarti, M.; Choudhury, B.; Caldera, J.R.; et al. Functional Divergence of a Bacterial Enzyme Promotes Healthy or Acneic Skin. Nat Commun 2023, 14, 8061. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.; Gaspar, C.; Rolo, J.; Palmeira-de-Oliveira, R.; Teixeira, J.P.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, A. Comparative Efficacy of Essential Oils against Cutibacterium Acnes: Effect upon Strains from Phylotypes with Different Virulence Patterns. Microb Pathog 2025, 199, 107159. [Google Scholar] [CrossRef] [PubMed]
- Katsuta, Y.; Iida, T.; Inomata, S.; Denda, M. Unsaturated Fatty Acids Induce Calcium Influx into Keratinocytes and Cause Abnormal Differentiation of Epidermis. J Invest Dermatol 2005, 124, 1008–1013. [Google Scholar] [CrossRef]
- Johnsson, A.; Kjeldstad, B.; Melø, T.B. Fluorescence from Pilosebaceous Follicles. Arch Dermatol Res 1987, 279, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Emri, G.; Paragh, G.; Tósaki, Á.; Janka, E.; Kollár, S.; Hegedűs, C.; Gellén, E.; Horkay, I.; Koncz, G.; Remenyik, É. Ultraviolet Radiation-Mediated Development of Cutaneous Melanoma: An Update. J Photochem Photobiol B 2018, 185, 169–175. [Google Scholar] [CrossRef]
- DeForge, L.E.; Preston, A.M.; Takeuchi, E.; Kenney, J.; Boxer, L.A.; Remick, D.G. Regulation of Interleukin 8 Gene Expression by Oxidant Stress. J Biol Chem 1993, 268, 25568–25576. [Google Scholar] [CrossRef]
- Piquero-Casals, J.; Morgado-Carrasco, D.; Rozas-Muñoz, E.; Mir-Bonafé, J.F.; Trullàs, C.; Jourdan, E.; Piquero-Martin, J.; Zouboulis, C.C.; Krutmann, J. Sun Exposure, a Relevant Exposome Factor in Acne Patients and How Photoprotection Can Improve Outcomes. J Cosmet Dermatol 2023, 22, 1919–1928. [Google Scholar] [CrossRef]
- Kang, D.; Shi, B.; Erfe, M.C.; Craft, N.; Li, H. Vitamin B12 Modulates the Transcriptome of the Skin Microbiota in Acne Pathogenesis. Sci Transl Med 2015, 7, 293ra103. [Google Scholar] [CrossRef]
- Owen, J.J.; Youssef, R.M.; Altman, K. Vitamin B12–Induced Acneiform Eruption. Proc (Bayl Univ Med Cent) 38 91–93. [CrossRef] [PubMed]
- Reynolds, R.V.; Yeung, H.; Cheng, C.E.; Cook-Bolden, F.; Desai, S.R.; Druby, K.M.; Freeman, E.E.; Keri, J.E.; Stein Gold, L.F.; Tan, J.K.L.; et al. Guidelines of Care for the Management of Acne Vulgaris. J Am Acad Dermatol 2024, 90, 1006.e1–1006.e30. [Google Scholar] [CrossRef] [PubMed]
- Coenye, T.; Spittaels, K.-J.; Achermann, Y. The Role of Biofilm Formation in the Pathogenesis and Antimicrobial Susceptibility of Cutibacteriumacnes. Biofilm 2021, 4, 100063. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, I.; Sivori, F.; Truglio, M.; De Maio, F.; Lucantoni, F.; Cardinali, G.; Pontone, M.; Bernardi, T.; Sanguinetti, M.; Capitanio, B.; et al. Skin Dysbiosis and Cutibacterium Acnes Biofilm in Inflammatory Acne Lesions of Adolescents. Sci Rep 2022, 12, 21104. [Google Scholar] [CrossRef]
- Burkhart, C.G.; Burkhart, C.N. Expanding the Microcomedone Theory and Acne Therapeutics: Propionibacterium Acnes Biofilm Produces Biological Glue That Holds Corneocytes Together to Form Plug. J Am Acad Dermatol 2007, 57, 722–724. [Google Scholar] [CrossRef]
- Jahns, A.C.; Alexeyev, O.A. Three Dimensional Distribution of Propionibacterium Acnes Biofilms in Human Skin. Experimental Dermatology 2014, 23, 687–689. [Google Scholar] [CrossRef]
- Knutson, D.D. Ultrastructural Observations in Acne Vulgaris: The Normal Sebaceous Follicle and Acne Lesions. J Invest Dermatol 1974, 62, 288–307. [Google Scholar] [CrossRef]
- Byrd, A.L.; Belkaid, Y.; Segre, J.A. The Human Skin Microbiome. Nat Rev Microbiol 2018, 16, 143–155. [Google Scholar] [CrossRef]
- Choi, E.-J.; Lee, H.G.; Bae, I.-H.; Kim, W.; Park, J.; Lee, T.R.; Cho, E.-G. Propionibacterium Acnes-Derived Extracellular Vesicles Promote Acne-Like Phenotypes in Human Epidermis. J Invest Dermatol 2018, 138, 1371–1379. [Google Scholar] [CrossRef]
- Chudzik, A.; Migdał, P.; Paściak, M. Different Cutibacterium Acnes Phylotypes Release Distinct Extracellular Vesicles. Int J Mol Sci 2022, 23, 5797. [Google Scholar] [CrossRef]
- Cheung, C.T.; Lancien, U.; Corvec, S.; Mengeaud, V.; Mias, C.; Véziers, J.; Khammari, A.; Dréno, B. Pro-Inflammatory Activity of Cutibacterium Acnes Phylotype IA1 and Extracellular Vesicles: An in Vitro Study. Exp Dermatol 2024, 33, e15150. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Tan, A.Y.Q.; van Steensel, M.A.M.; Lim, X. Aberrant Wnt Signaling Induces Comedo-Like Changes in the Murine Upper Hair Follicle. Journal of Investigative Dermatology 2022, 142, 2603–2612.e6. [Google Scholar] [CrossRef] [PubMed]
- Veniaminova, N.A.; Jia, Y.Y.; Hartigan, A.M.; Huyge, T.J.; Tsai, S.-Y.; Grachtchouk, M.; Nakagawa, S.; Dlugosz, A.A.; Atwood, S.X.; Wong, S.Y. Distinct Mechanisms for Sebaceous Gland Self-Renewal and Regeneration Provide Durability in Response to Injury. Cell Reports 2023, 42. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.B.H.; Wei, S.; Tan, A.H.-M.; van Steensel, M.A.M.; Lim, X. Lrig1-Expressing Epidermal Progenitors Require SCD1 to Maintain the Dermal Papilla Niche. Sci Rep 2023, 13, 4027. [Google Scholar] [CrossRef]
- Sipilä, K.; Rognoni, E.; Jokinen, J.; Tewary, M.; Rudan, M.V.; Talvi, S.; Jokinen, V.; Dahlström, K.M.; Liakath-Ali, K.; Mobasseri, A.; et al. Embigin Is a Fibronectin Receptor That Affects Sebaceous Gland Differentiation and Metabolism. Developmental Cell 2022, 57, 1453–1465.e7. [Google Scholar] [CrossRef]
- Kurokawa, I.; Nakase, K. Recent Advances in Understanding and Managing Acne. F1000Res 2020, 9, F1000 Faculty Rev-792. [Google Scholar] [CrossRef]
- Sugihira, T.; Tamai, M.; Takagi, K.; Salcman, B.; Kobayashi, T.; Hobro, A.J.; Nakagawa, S.; Takeuchi, Y.; Ishikawa, E.; Maekawa, M.; et al. A Lipid-Driven, Microbe-Independent Mechanism of Acne via Lrig1+ Follicular Progenitor Cells 2025, 2025.08.30.673221. [CrossRef]
- Teder-Laving, M.; Kals, M.; Reigo, A.; Ehin, R.; Objärtel, T.; Vaht, M.; Nikopensius, T.; Metspalu, A.; Kingo, K. Genome-Wide Meta-Analysis Identifies Novel Loci Conferring Risk of Acne Vulgaris. Eur J Hum Genet 2024, 32, 1136–1143. [Google Scholar] [CrossRef]
- Kircik, L.H. Androgens and Acne: Perspectives on Clascoterone, the First Topical Androgen Receptor Antagonist. Expert Opin Pharmacother 2021, 22, 1801–1806. [Google Scholar] [CrossRef]
- Pearson, J.C.; Gillett, E.; Gadri, N.D.; Dionne, B. Tetracyclines, the Old and the New: A Narrative Review. CMI Communications 2025, 2, 105059. [Google Scholar] [CrossRef]
- Carr, M.; Chou, S.R.; Sun, J.; LaRock, D.L.; LaRock, C.N. Therapeutic Inhibition of Metalloproteases by Tetracyclines during Infection by Multi-Drug Resistant Pseudomonas 2025, 2025.02.14.638345. [CrossRef]
- Majeski, J.A.; Alexander, J.W. Evaluation of Tetracycline in the Neutrophil Chemotactic Response. J Lab Clin Med 1977, 90, 259–265. [Google Scholar] [PubMed]
- Vázquez-Laslop, N.; Mankin, A.S. How Macrolide Antibiotics Work. Trends in Biochemical Sciences 2018, 43, 668–684. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, P.; Ziesenitz, V.C.; Curtis, N.; Ritz, N. The Immunomodulatory Effects of Macrolides—A Systematic Review of the Underlying Mechanisms. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef]
- Dessinioti, C.; Katsambas, A. Antibiotics and Antimicrobial Resistance in Acne: Epidemiological Trends and Clinical Practice Considerations. Yale J Biol Med 2022, 95, 429–443. [Google Scholar]
- Walsh, T.R.; Efthimiou, J.; Dréno, B. Systematic Review of Antibiotic Resistance in Acne: An Increasing Topical and Oral Threat. The Lancet Infectious Diseases 2016, 16, e23–e33. [Google Scholar] [CrossRef]
- Lathakumari, R.H.; Vajravelu, L.K.; Satheesan, A.; Ravi, S.; Thulukanam, J. Antibiotics and the Gut Microbiome: Understanding the Impact on Human Health. Medicine in Microecology 2024, 20, 100106. [Google Scholar] [CrossRef]
- Dallo, M.; Patel, K.; Hebert, A.A. Topical Antibiotic Treatment in Dermatology. Antibiotics 2023, 12, 188. [Google Scholar] [CrossRef]
- Matin, T.; Patel, P.; Goodman, M.B. Benzoyl Peroxide. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Nacht, S.; Yeung, D.; Beasley, J.N.; Anjo, M.D.; Maibach, H.I. Benzoyl Peroxide: Percutaneous Penetration and Metabolic Disposition. J Am Acad Dermatol 1981, 4, 31–37. [Google Scholar] [CrossRef]
- Lam, M.; Hu, A.; Fleming, P.; Lynde, C.W. The Impact of Acne Treatment on Skin Bacterial Microbiota: A Systematic Review. J Cutan Med Surg 2022, 26, 93–97. [Google Scholar] [CrossRef]
- Weber, S.U.; Thiele, J.J.; Han, N.; Luu, C.; Valacchi, G.; Weber, S.; Packer, L. Topical Alpha-Tocotrienol Supplementation Inhibits Lipid Peroxidation but Fails to Mitigate Increased Transepidermal Water Loss after Benzoyl Peroxide Treatment of Human Skin. Free Radic Biol Med 2003, 34, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kuo, S.; Shu, M.; Yu, J.; Huang, S.; Dai, A.; Two, A.; Gallo, R.L.; Huang, C.-M. Staphylococcus Epidermidis in the Human Skin Microbiome Mediates Fermentation to Inhibit the Growth of Propionibacterium Acnes: Implications of Probiotics in Acne Vulgaris. Appl Microbiol Biotechnol 2014, 98, 411–424. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Guo, C.; Wang, Q.; Feng, C.; Duan, Z. A Pilot Study on the Efficacy of Topical Lotion Containing Anti-Acne Postbiotic in Subjects with Mild -to -Moderate Acne. Front Med (Lausanne) 2022, 9, 1064460. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Cogen, A.L.; Radek, K.A.; Park, H.J.; MacLeod, D.T.; Leichtle, A.; Ryan, A.F.; Di Nardo, A.; Gallo, R.L. Activation of TLR2 by a Small Molecule Produced by Staphylococcus Epidermidis Increases Antimicrobial Defense against Bacterial Skin Infections. J Invest Dermatol 2010, 130, 2211–2221. [Google Scholar] [CrossRef]
- Puebla-Barragan, S.; Reid, G. Probiotics in Cosmetic and Personal Care Products: Trends and Challenges. Molecules 2021, 26, 1249. [Google Scholar] [CrossRef]
- Karn, S.L.; Gangwar, M.; Kumar, R.; Bhartiya, S.K.; Nath, G. Phage Therapy: A Revolutionary Shift in the Management of Bacterial Infections, Pioneering New Horizons in Clinical Practice, and Reimagining the Arsenal against Microbial Pathogens. Front Med (Lausanne) 2023, 10, 1209782. [Google Scholar] [CrossRef]
- Harper, D.R. Introduction to Bacteriophages. In Bacteriophages; Springer: Cham, 2021; pp. 3–16. ISBN 978-3-319-41986-2. [Google Scholar]
- Mayorga-Ramos, A.; Carrera-Pacheco, S.E.; Barba-Ostria, C.; Guamán, L.P. Bacteriophage-Mediated Approaches for Biofilm Control. Front Cell Infect Microbiol 2024, 14, 1428637. [Google Scholar] [CrossRef]
- Golembo, M.; Puttagunta, S.; Rappo, U.; Weinstock, E.; Engelstein, R.; Gahali-Sass, I.; Moses, A.; Kario, E.; Ben-Dor Cohen, E.; Nicenboim, J.; et al. Development of a Topical Bacteriophage Gel Targeting Cutibacterium Acnes for Acne Prone Skin and Results of a Phase 1 Cosmetic Randomized Clinical Trial. Skin Health Dis 2022, 2, e93. [Google Scholar] [CrossRef]
- Verma, N.K.; Tan, S.J.; Chen, J.; Chen, H.; Ismail, M.H.; Rice, S.A.; Bifani, P.; Hariharan, S.; Paul, V.D.; Sriram, B.; et al. inPhocus: Current State and Challenges of Phage Research in Singapore. Phage (New Rochelle) 2022, 3, 6–11. [Google Scholar] [CrossRef]
- Fukaya-Shiba, A.; Ogata, A.; Kuribayashi, R.; Sakurai, A.; Suzuki, K.; Takadama, S.; Nishimura, J.; Uchiyama, J.; Ohge, H.; Takeuchi, T.; et al. Regulatory Considerations for Developing Phage Therapy Medicinal Products for the Treatment of Antimicrobial Resistant Bacterial Infections. Front Pharmacol 16 1713471. [CrossRef]
- Fenton, M.; Ross, P.; McAuliffe, O.; O’Mahony, J.; Coffey, A. Recombinant Bacteriophage Lysins as Antibacterials. Bioeng Bugs 2010, 1, 9–16. [Google Scholar] [CrossRef]
- Loeffler, J.M.; Nelson, D.; Fischetti, V.A. Rapid Killing of Streptococcus Pneumoniae with a Bacteriophage Cell Wall Hydrolase. Science 2001, 294, 2170–2172. [Google Scholar] [CrossRef]
- Lee, J.-H.; Hasnain, M.A.; Park, J.-H.; Choi, W.; Moon, G.-S. Comparative Antibacterial Activity of N-Terminal and C-Terminal Domains of a Recombinant Endolysin against Cutibacterium Acnes. Appl Environ Microbiol 2025, 91, e0116825. [Google Scholar] [CrossRef]
- Vestby, L.K.; Grønseth, T.; Simm, R.; Nesse, L.L. Bacterial Biofilm and Its Role in the Pathogenesis of Disease. Antibiotics (Basel) 2020, 9, 59. [Google Scholar] [CrossRef]
- Xiang, L.; Chen, R.; Wu, L.; Xu, A.; He, L.; Wang, J.; Lu, Y.; Xiao, R.; Liu, L.; Feng, Y.; et al. 49342 First FASN Inhibitor ASC40 to Treat Acne Vulgaris Patients: Final Results Form a Phase 2 Trial. Journal of the American Academy of Dermatology 2024, 91, AB217. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




