1.1. Introduction
Colorectal cancer represents a considerable health burden worldwide, being the third most common cancer type and the second-leading cause of cancer related death [
1]. The standard of care for patients with non-metastatic colorectal cancer is surgical resection with clinical and histopathologic staging informing the recommendation for adjuvant systemic therapy [
2,
3]. Currently, there are no validated markers that can reliably guide chemotherapy selection in patients for whom histopathologic staging indicates systemic therapy may be beneficial [
4,
5]. Further, toxicity of systemic therapy is of great concern to clinicians and patients alike [
6]. Despite recent enthusiasm for a small number of patients that are candidates for immunotherapy [
7,
8] treatment is largely empiric, with “guideline concordant care” driving clinical recommendations and being considered the gold standard [
9]. While guideline concordant care can include precision/personalized medicine, a one-size-fits all approach of empiric guidelines leaves a lot to be desired in terms of individual patient benefit and patient expectations [
10,
11,
12]. As physicians and researchers, we need to ask ourselves - is standard of care enough?
In this commentary we describe a hypothetical patient journey for a patient with colon cancer manifesting with common difficulties that hinder the ability to provide personalized medicine.
1.2. The Patient Journey
We describe a clinically grounded hypothetical journey of a patient with colorectal cancer. Our hypothetical patient is a 55-year-old female who has just undergone her first screening colonoscopy. She has no family history of colon cancer but unfortunately has a 3 cm polyp in her right colon biopsied that appears to be cancerous. She is called 5 business days later and informed that she has invasive colonic adenocarcinoma [
1,
13]. She is also informed that she has been referred to her local university hospital colon cancer multidisciplinary clinic (MDC) [
14]. She is extremely concerned but somewhat comforted by the reputed academic prowess of the university and the rapid referral for her problem [
15]. She arrives for an early morning appointment at which time she has a full medical history taken and physical examination performed by a mid-level provider or a junior level surgery or medicine resident (doctor in training). Following her history and physical exam she is quickly shuttled to the lab for a full panel of electrolytes, complete blood count, and “tumor markers”. From there she is transferred for a CT examination of her chest, abdomen and pelvis. Rapidly, she has completed what is currently considered an appropriate patient intake [
14,
16,
17] . At this point she will return for an afternoon appointment to discuss her case. Our patient is considering a litany of questions regarding her cancer stage [
18], her surgery and whether or not she will require chemotherapy and radiation [
19]. Her questions have been gleaned from Google searches [
20] and well-meaning advice from family, friends and acquaintances who have had experience with colon cancer or at least hold personal opinions on medical procedures [
21].
At a planned lunch, a large group of physicians, mid-level providers, residents, interns, medical students and other assorted ancillary personnel are reviewing and discussing the cases to be seen in clinic that afternoon. From her intake it is has been discovered that our patient has no evidence of metastatic or regional disease on her axial imaging, no family history, no mitigating medical issues, she is eligible for no study protocols and has no abnormal tumor markers detected in her pathology review to suggest she is a candidate for immunotherapy. It is therefore recommended that she have upfront surgery via a minimally invasive technique that promises limited pain and a rapid discharge from the hospital [
22,
23]. Having returned to the clinic she is greeted by the provider she met that morning along with a more senior physician who will be her surgeon. The surgeon engages in a brief introduction, explains the plan of care and asks if our patient has any questions. The patient begins by asking, “what stage cancer do I have?” [
18] This is a common question and, in many ways, demonstrates the lack of pre-clinic preparation patients receive as they go through the initial intake process.” [
15,
24,
25]. In the best of circumstances, her stage question leads into a discussion of the tumor, lymph nodes and metastatic disease (TNM) staging system and the need for surgery to remove and further evaluate her tumor. The risks and benefits of the surgery along with brief mention of the complications will be followed by signing of a surgical consent and she will be placed on the surgery schedule. At some point in this process the patient will likely hear that she will be receiving “guideline concordant care” [
14,
16,
17].
It is not clear if most patients have knowledge of what this means [
26]. This phrase has undoubtedly been mentioned at the physician lunch while reviewing pathology slides and CT scans. Within a few days to weeks from this visit our patient will return to the hospital to undergo surgical segmental resection of a portion of her colon. Presumably the surgery goes well and is also guideline concordant [
27]. At this point our patient has her clinical staging completed [
28]. For the sake of this commentary, we will assume that she has had no unexpected metastases seen during her operative intervention, nor visible evidence of T4 disease (cancer penetrating the wall of the colon) detected. Based on her preoperative testing, she has stage 1-3 colon cancer pending her formal pathology evaluation. As previously mentioned, she has no family history or other contributory factors that would be considered “high risk” for systemic recurrence. At this point she is again told that she will be recommended to undergo guideline concordant care once her pathology is known that might include chemotherapy. Our patient does well from a surgical standpoint and is told that within 72 hours she will be discharged to complete her recovery at home [
29].
In this hypothetical scenario, our patient will have her case reviewed at a multidisciplinary tumor board and a discussion of her next steps in care will take place. Once again, her plan of care should be guideline concordant. For the purposes of this hypothetical case- our patient’s pathology has returned as a T2, N0, MX moderately differentiated adenocarcinoma. Based on the existing guidelines [
14] for early-stage disease she will be recommended to have surveillance, whereas more advanced disease would elicit a recommendation for adjuvant therapy [
30,
31]. In this scenario the patient will be informed that her chance of survival with surgery alone is quite high (around 90%) [
32]. Unfortunately, there is a small, but real chance her cancer can recur and therefore ongoing surveillance is important.
1.3. Recurrence, Shortcomings and Potential Paths to Improvement
The above hypothetical patient presentation leaves us to consider whether or not we have done enough? It has clearly been demonstrated that multidisciplinary clinics are very efficient in seeing patients and getting them into treatment [
10] and while this appears to cover all the bases, it is certainly not without issues [
9,
33,
34,
35]. In terms of providing personalized care, undoubtedly there is a need for improvements [
10,
36]. It is known, even in early stage patients, that a low, but real risk of metastasis exists [
37]. The incidence of recurrent disease is higher in stage 3 and 4 colorectal cancer patients and in patients who develop colorectal cancer at a younger age [
38,
39,
40]. A recent publication from Denmark highlights some difficulties in recommending the best next step in treatment for colon cancer patients with nonmetastatic disease. This study examined a large number of patients with stage 1-3 disease and noted recurrence rates of more than 16% in stage 1 patients at 1 year, decreasing to around 7% at 3 years post-operatively. For stage 2 patients the rates were approximately 22% at 1 year dropping to around 12% at 3 years and finally for stage 3 patients the rates were 35.5% and 24.6% at 1 and 3 years respectively [
39]. While lower rates are found in other studies [
41,
42] there is still a small percentage, but large corresponding number of early stage colon cancer patients who develop metastatic disease that might benefit from systemic therapies. Similarly not all patients with stage 3 disease go on to develop metastases and therefore will not benefit from standard systemic therapies as guidelines recommend [
42,
43,
44]. When we examine current guidelines as they exist, there is a limited number of options for adjuvant therapy (essentially combinations of standard chemotherapeutic agents: capecitabine; 5 fluorouracil and oxaliplatin) [
14,
28]. The recommendations are anchored primarily in the MOSAIC Trial [
45,
46]
In this trial a total of 1123 patients were randomized and the rate of disease-free survival at three years was 78.2 percent (95 percent confidence interval, 75.6 to 80.7) in the group given 5 FU/Leucovorin plus oxaliplatin, and 72.9 percent (95 percent confidence interval, 70.2 to 75.7) in the 5 FU/Leucovorin group (P=0.002). While this is highly significant and the toxicity can generally be managed [
6,
47]
recent work suggests that patients may be more enthusiastic about increased survival from adjuvant chemotherapy than clinicians [
48]
. More recently it has been shown, amongst more aged patients, the expectation of prolonged survival needs to be higher for them to accept the risks of adjuvant therapy [
49]
. The real-world benefits of increased survival balanced against long term toxicity (peripheral neuropathy, depression and poor sleep quality) may not be appreciated by patients, particularly as symptoms may commence after treatment is completed [
50,
51,
52]
. Thus, we note that the with use of current guidelines, there is potential to under treat early-stage colon cancer patients and over treat a percentage of more advanced stage patients for whom surgery alone would be sufficient. This again leads us to ask,” is current practice enough”? Improvements in selecting patients for receipt of adjuvant therapy as well as candidacy for up front small molecule inhibitors, immunotherapy or even more efficient surveillance definitely need to be addressed in a “guideline concordant” approach, suggesting we need new data.
1.4. Colorectal Cancer in the ‘Omics Era
Genomic profiling has undeniably had a transformative effect in the field of oncology. The proportion of cancer patients receiving genomically guided treatment and the number benefiting from it has been increasing over the past two decades. One pan-cancer study reported that the percentage of patients benefiting from such treatments increased from 0.7% in 2006 to 4.9% in 2018 [
53], while another 2024 study reported 31.6% of tumors carry a biomarker considered predictive in standard-of-care [
54]. A recent CRC-specific study of 575 patients reported a rich landscape of potentially clinically actionable genomic findings, including many well-known driver mutations. Ultimately, they suggested that
51% of patients could theoretically be eligible for clinical actionability based on genomic findings, while 49% could be enrolled in clinical trials of investigational therapies. Nonetheless, this does not represent real-world benefit, due to the current entrenchment of more traditional treatment paradigms. This fact is not without justification, since clinical actionability of a mutation does not guarantee successful treatment. One recent pan-cancer meta-analysis of approximately fifty-five thousand tumors reported that of the 15% of patients who received a genomically guided treatment, the objective response rate was only 25%. While genomically guided treatment has and will continue to be a driver of improved cancer care, it is only one piece of the required treatment armamentarium, and crucial elements are still missing. One such missing element is the transition of functional precision medicine approaches into standard clinical care [
55]
.
1.5. From Classical to Modern Functional Precision Medicine
Functional precision medicine has existed in some form since the mid-1900s [
56] and exposes patient-derived tissues to drugs in an attempt to predict clinical response, ex vivo [
57]. It encompasses traditional approaches including traditional 2D monolayer and clonogenic drug sensitivity assays, as well as more contemporary methods including patient-derived xenografts (PDX), patient-derived organoids, and emerging microphysiologic (e.g., microfluidic or tumor-on-chip) systems.
Early methods such as clonogenic assays seemed initially promising but were ultimately later abandoned [
58] for various reasons, including long assay times, prohibitive biomass requirements, limited testing scope, growth artifacts and irreproducibility [
59]. Other highlighted issues included low evaluation rates [
58], labor-intensiveness, [
58], prohibitive assay times, lack of scalability [
60,
61] and inability to correlate with clinical response [
62]. While PDX models have provided valuable insights into tumor biology and therapeutic response, they are associated with limitations including months-long engraftment times, presence of murine components, lack of a functional human immune system and clonal evolution that may alter microenvironmental interactions and genomic fidelity relative to the tumor of origin. They are also resource-intensive and relatively low throughput compared to ex vivo systems [
63,
64,
65,
66]. Microphysiologic systems offer improved biomimicry compared to traditional models, however, these platforms remain largely experimental, technically complex, and relatively low throughput, which may limit clinical deployment in the short-term [
67,
68,
69,
70]. Despite the existence of some commercial tests [
56] no methods are widely accepted by the medical community [
63].
In recent years the field has been transitioning toward alternative models can exhibit multifactorial biomimicry of an individual’s tumor. 3D patient-derived tumor organoids (PDTOs) have been demonstrated to maintain features of the original tumor including intratumoral heterogeneity, architecture, and polyclonality, and are thus regarded as a possible approach to overcoming shortcomings of traditional methods [
67]. Organoids have been shown to retain histological features, hotspot mutations and receptor status of the tumor-of-origin [
71] and dose response studies have demonstrated organoids can mirror the sensitivity profile of a patient [
72,
73]. In colorectal cancer specifically, clinical studies have shown wide-ranging success in clinical correlation studies [
74].
Organoid assays are not without their own challenges. Differences in sample storage, media composition, cell procurement, and experimental protocols all represent sources of potential experimental variability [
75], however it is believed that industrial developments like automation and miniaturization will be key in addressing many challenges associated with traditional assays [
76,
77,
78,
79,
80] and will facilitate clinically compatible turnaround times which are necessary for widespread adoption of any assay [
81]. Ultimately the success of PDTO-based drug screening in personalized cancer care will require rapid turnaround from tumor sampling to treatment recommendations in order to guide clinical decisions [
82,
83]. The specific promise of organoids in clinical care has been evidenced and strengthened by the NIH’s recent announcement of their Standardized Organoid Modeling (SOM) Center. The goal of the center is the development of standardized organoids and associated protocols for deployment in biological and medicinal research [
84].
Across the field, standardized prospective performance metrics, validation frameworks, regulatory pathways, and workflow integration into multidisciplinary oncology practice remain areas of active development. Our own approach described below represents one highly automated strategy with potential to play a role in this evolving landscape. Like other platforms, it will require prospective validation and demonstration of reproducibility and clinical utility before broader adoption can be realized, however we believe preliminary evidence offers hope for our own or similar technologies to realize clinical benefits.
1.6. Clinical Correlation and Resistant Subclone Identification in a Clinical CRC Study
In a recent publication, we demonstrated association between ex vivo testing of patient derived tumor models and patient clinical response to treatment in a retrospective clinical study incorporating a cohort of 21 colorectal cancer patients treated with neoadjuvant standard-of-care therapies. While our cohort was modestly sized, and follow-up work including prospective validation and independent replication of outcomes are required to confirm and quantify clinical utility, our initial findings were both encouraging and hypothesis-generating. Patient response to treatment was modeled using patient-derived tumor organoids (PDTOs) and our proprietary MOSgen™ system and then tested for correlation with actual clinical outcomes. Ex vivo testing was associated with clinical response using binary patient response categorization with 83% accuracy, and disease-free survival (DFS) also correlated with in-assay measured responses. Furthermore we were able to identify patients whose PDTOs demonstrated higher response to alternative standard-of-care therapies than to the one they received in their clinical treatment, inspiring the preliminary hypothesis that such patients might have benefited from a non-empiric treatment selection paradigm [
85].
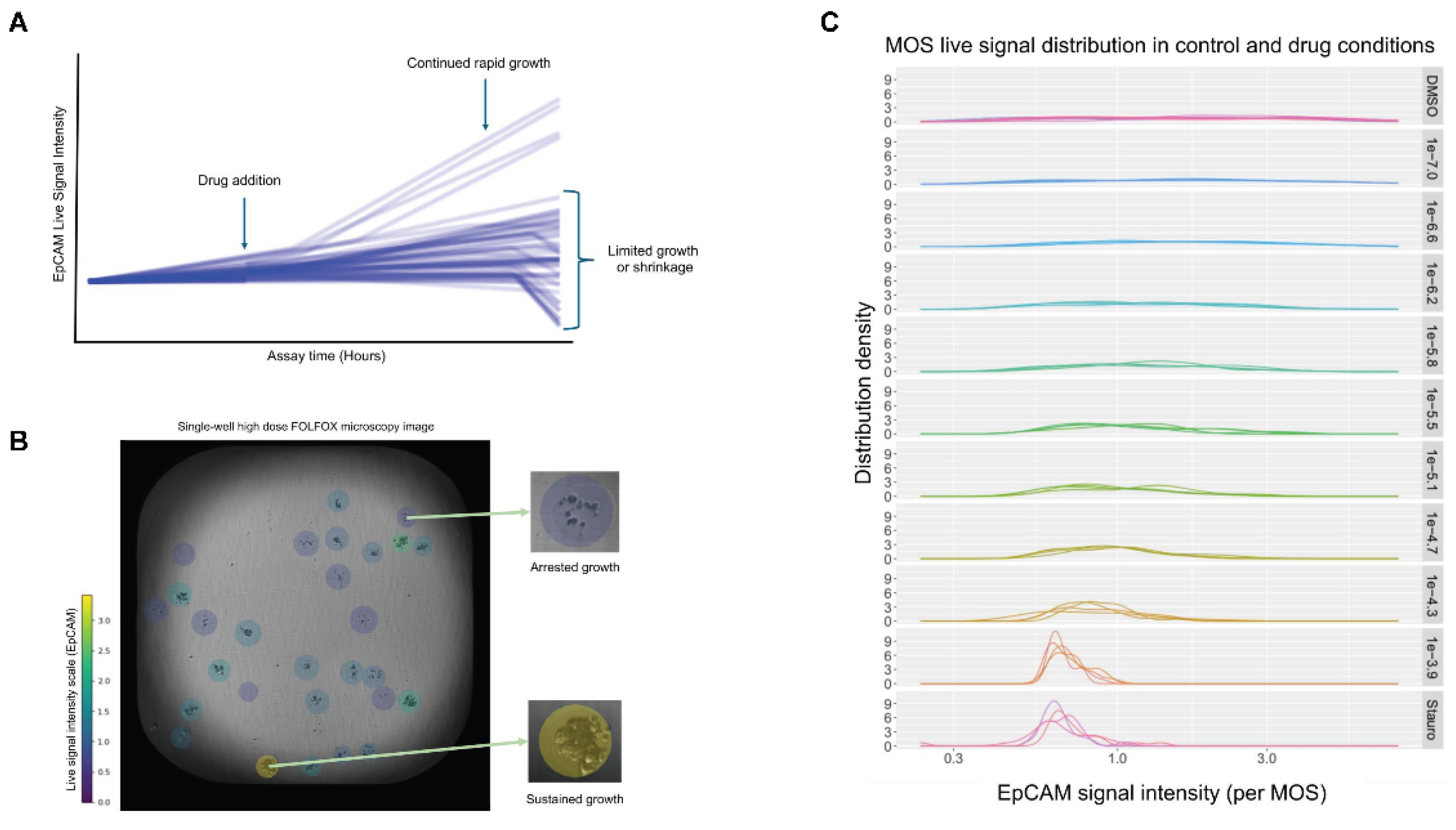
The assay used in the aforementioned study utilized brightfield microscopy imaging of approximately 40 individual patient-derived models (MOS
®® droplets) per well in 384 well plates with wells spanning multiple drugs, and 9-point concentration gradients. Fluorescently labeled epithelial cell adhesion molecule (EpCAM) was used as a marker of tumor cell viability with signal intensity measured longitudinally across a one-week period. While clinical correlation was based upon summation of well-level EpCAM signals, we also utilized machine learning techniques to automatically identify all individual MOS droplets per-well and subsequently generated per-MOS-droplet viability measures. This provided the ability to detect differential growth and response rates for each individual patient MOS model within each well of a plate, possibly corresponding to the well-documented heterogeneity of cancer cells in-vivo (
Figure 1a). By combining per-MOS-droplet EpCAM signal measurements with a colored intensity gradient and per-well brightfield microscopy images we were further able to visualize the morphology of MOS models demonstrating heterogeneous responses to drug doses, with some outlier MOS models demonstrating vigorous sustained growth despite high-dose drug treatment while others appeared arrested in growth and condensed in terms of cellular morphology (
Figure 1b). EpCAM signal density distributions per-well were also profiled and demonstrated wider distributions corresponding to higher overall levels of growth heterogeneity in vehicle or lower drug dose containing wells, while heterogeneity decreased with high drug doses, and high viability being observed only in isolated outlier MOS models. We hypothesize that such outliers might represent drug-resistant subclonal populations within the patient tumor, however this interpretation remains hypothetical since follow-up orthogonal validation was not part of the study (
Figure 1c). While this analysis was exploratory and the clinical study was retrospective, it suggests that with further work, there is the potential for technologies like ours to accurately predict patient treatment sensitivities in a prospective setting and further, to identify clinically relevant, heterogeneous intra-patient drug responses. The latter would require follow-up validation work to generate evidence supporting the clinical relevance of differential drug responses ex vivo. This could include comparative ‘omic profiling of outlier MOS models to their drug responsive counterparts for identification of known or novel drivers of resistance, or isolation of non-responsive MOS models followed by secondary or combination treatment response profiling. Such efforts could offer the prospect of identifying sub-clonal drug resistance, and even potentially determining alternative, accompanying or second-line agents that might evoke cell death in initially resistant subpopulations of cells from a single patient assay, early in a patient’s clinical journey. Further, identification of such subclones could introduce the possibility of more efficacious treatments in all stages of colorectal cancer. Such isolation of identified subclones would offer a focused alternative to traditionally employed pan tumor assessment [
86,
87], enabling genetic analysis of the most problematic cancer-cell populations. While the potential seems high, whether this strategy ultimately translates into improved clinical outcomes will require prospective evaluation in appropriately designed studies.
1.7. Conclusions
There have been advances in the care of colorectal cancer patients, including ERAS (enhanced recovery after surgery) and Minimally Invasive Surgery (MIS) with the promise of reduced pain and earlier hospital discharge [
88,
89]. There have been advances in our ability to measure minimal residual disease MRD in colon cancer [
90,
91]. More recently, there have been exciting advances in immunotherapy for appropriately selected patients [
84]. Unfortunately, guideline concordant therapy for non-metastatic colon cancer revolves around systemic therapy recommendations based on data that is two decades old [
88]. At the same time, the aged, frail population continues to increase [
92], and more young patients are presenting with aggressive colon cancers than ever before [
93]. It is our belief that current paradigms of colon cancer treatment may benefit from the near-term incorporation of personalized medicine approaches. The work of Nors et al. [
40] has demonstrated that recurrence rates for non-metastatic colorectal cancer, particularly at 1 to 2 years, are much higher than previously quoted. Such findings help to explain unexpected disease recurrence in early-stage disease as well as treatment failure in patients receiving standard of care systemic therapy for more advanced disease. Our preliminary work in MOS models, created from primary colon cancers, suggest problematic subclones that do not respond to standard of care systemic therapy may be present and identifiable in ex vivo models (
Figure 1b-c). Identification of treatment resistant cell niches may offer the potential for fastidious use of ‘omics by focusing investigation on problematic subclones rather than addressing potentially billions of cells in a primary tumor. Further, this work could enable improved diagnostics for a wide population of cancer patients. Ultimately, the role of functional precision medicine in colorectal cancer will be determined by prospective validation and thoughtful integration alongside existing standards of care, and continued investigation will help define its contribution to more individualized clinical care.
Author Contributions
CCB, GRO, HS, and WRS wrote the article. MRS performed the analysis in the original, referenced study. All authors read and edited the article.
Funding
No grants supported this work.
Institutional Review Board Statement
Not applicable. The original study received approval by the local medical ethics committee (S-136/2021 and S-708/2019).
Informed Consent Statement
Not applicable. For the original study all patients provided written informed consent for data and tissue collection and analysis.
Data Availability Statement
No new data are associated with this article.
Conflicts of Interest
All authors are employees of Xilis, Inc.
References
- Bray, F; Laversanne, M; Sung, H; Ferlay, J; Siegel, RL; Soerjomataram, I; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024, 74(3), 229–63. [Google Scholar] [CrossRef]
- Shah, MA; Renfro, LA; Allegra, CJ; André, T; de Gramont, A; Schmoll, H-J; et al. Impact of Patient Factors on Recurrence Risk and Time Dependency of Oxaliplatin Benefit in Patients With Colon Cancer: Analysis From Modern-Era Adjuvant Studies in the Adjuvant Colon Cancer End Points (ACCENT) Database. J Clin Oncol. 2016, 34(8), 843–53. [Google Scholar] [CrossRef]
- Sargent, D; Sobrero, A; Grothey, A; O’Connell, MJ; Buyse, M; Andre, T; et al. Evidence for cure by adjuvant therapy in colon cancer: observations based on individual patient data from 20,898 patients on 18 randomized trials. J Clin Oncol. 2009, 27(6), 872–7. [Google Scholar] [CrossRef]
- Koncina, E; Haan, S; Rauh, S; Letellier, E. Prognostic and Predictive Molecular Biomarkers for Colorectal Cancer: Updates and Challenges. Cancers (Basel) 2020, 12(2), 319. [Google Scholar] [CrossRef] [PubMed]
- Punt, CJA; Koopman, M; Vermeulen, L. From tumour heterogeneity to advances in precision treatment of colorectal cancer. Nat Rev Clin Oncol 2017, 14(4), 235–46. [Google Scholar] [CrossRef]
- Grothey, A; Sobrero, AF; Shields, AF; Yoshino, T; Paul, J; Taieb, J; et al. Duration of Adjuvant Chemotherapy for Stage III Colon Cancer. N Engl J Med. 2018, 378(13), 1177–88. [Google Scholar] [CrossRef]
- Ganesh, K; Stadler, ZK; Cercek, A; Mendelsohn, RB; Shia, J; Segal, NH; et al. Immunotherapy in colorectal cancer: rationale, challenges and potential. Nat Rev Gastroenterol Hepatol 2019, 16(6), 361–75. [Google Scholar] [CrossRef] [PubMed]
- Chalabi, M; Verschoor, YL; Tan, PB; Balduzzi, S; Van Lent, AU; Grootscholten, C; et al. Neoadjuvant Immunotherapy in Locally Advanced Mismatch Repair-Deficient Colon Cancer. N Engl J Med. 2024, 390(21), 1949–58. [Google Scholar] [CrossRef]
- Robinson, TP; Kaiser, K; Lark, M; Ruedinger, B; Robb, BW; Morgan, T; et al. NCCN guideline concordance in colon and rectal cancer patients within a comprehensive health system. The American Journal of Surgery 2025, 240, 116114. [Google Scholar] [CrossRef] [PubMed]
- Tsimberidou, AM; Sireci, A; Dumanois, R; Pritchard, D. Strategies to Address the Clinical Practice Gaps Affecting the Implementation of Personalized Medicine in Cancer Care. JCO Oncol Pract. 2024, 20(6), 761–6. [Google Scholar] [CrossRef]
- Cornetta, K; Brown, CG. Balancing Personalized Medicine and Personalized Care: Academic Medicine. 2013, 88(3), 309–13. [Google Scholar]
- Panozzo, S; Collins, A; McLachlan, S-A; Lau, R; Le, B; Duffy, M; et al. Scope of Practice, Role Legitimacy, and Role Potential for Cancer Care Coordinators. Asia Pac J Oncol Nurs. 2019, 6(4), 356–62. [Google Scholar] [CrossRef]
- Sundling, KE; Zhang, R; Matkowskyj, KA. Pathologic Features of Primary Colon, Rectal, and Anal Malignancies. Cancer Treat Res. 2016, 168, 309–30. [Google Scholar] [PubMed]
- Benson, AB; Venook, AP; Al-Hawary, MM; Cederquist, L; Chen, Y-J; Ciombor, KK; et al. NCCN Guidelines Insights: Colon Cancer, Version 2.2018. J Natl Compr Canc Netw. 2018, 16(4), 359–69. [Google Scholar] [CrossRef] [PubMed]
- Chimonas, S; Lipitz-Snyderman, A; Kuperman, G. Cancer patients’ messages about radiology/pathology reports: Insights for AI. J Clin Oncol. 2025, 43((16_) suppl, 1570–1570. [Google Scholar] [CrossRef]
- National Quality Forum: National voluntary consensus... - Google Scholar [Internet]. Available online: https://scholar.google.com/scholar_lookup?title=National%20Voluntary%20Consensus%20Standards%20for%20Quality%20of%20Cancer%20Care:%20A%20concensus%20report&publication_year=2009& (accessed on 24 January 2026).
- D’Amico, TA; Bandini, LAM; Balch, A; Benson, AB; Edge, SB; Fitzgerald, CL; et al. Quality Measurement in Cancer Care: A Review and Endorsement of High-Impact Measures and Concepts. Journal of the National Comprehensive Cancer Network 2020, 18(3), 250–9. [Google Scholar] [CrossRef]
- Gattellari, M; Butow, PN; Tattersall, MH; Dunn, SM; MacLeod, CA. Misunderstanding in cancer patients: why shoot the messenger? Ann Oncol 1999, 10(1), 39–46. [Google Scholar] [CrossRef]
- Sarpatwari, A; Lee, SB; Zakoul, H; Tekle, W; Freedman, A; Belitkar, S; et al. Patient Perceptions of and Experiences with Risk Evaluation and Mitigation Strategies. Clin Pharmacol Ther 2026, 119(1), 267–75. [Google Scholar] [CrossRef]
- de Arruda RM, Ayoub IA, Nunes R, de Azevedo RS, Nunes M do PT. Consulting “Dr. Google”: how the digital search for internet health information influences doctor-patient relationship. Cad Saude Publica 41(7), e00153623. [CrossRef]
- Mathes, BM; Norr, AM; Allan, NP; Albanese, BJ; Schmidt, NB. Cyberchondria: Overlap with health anxiety and unique relations with impairment, quality of life, and service utilization. Psychiatry Res. 2018, 261, 204–11. [Google Scholar] [CrossRef]
- Kaiser, AM. Evolution and future of laparoscopic colorectal surgery. World J Gastroenterol 2014, 20(41), 15119–24. [Google Scholar] [CrossRef]
- Chen, E; Chen, L; Zhang, W; Zhou, W. Advanced innovations in reducing anastomotic leak: a review of emerging biomaterial applications in colorectal surgery. Int J Colorectal Dis. 2025, 40(1), 210. [Google Scholar] [CrossRef] [PubMed]
- Leeftink, AG; Bikker, IA; Vliegen, IMH; Boucherie, RJ. Multi-disciplinary planning in health care: a review. Health Syst (Basingstoke) 9(2), 95–118. [CrossRef]
- Winters, DA; Soukup, T; Sevdalis, N; Green, JSA; Lamb, BW. The cancer multidisciplinary team meeting: in need of change? History, challenges and future perspectives. BJU Int. 2021, 128(3), 271–9. [Google Scholar] [CrossRef]
- Taiwong, A; Uppor, W; Vibulchai, N. Concordance in the healthcare context: A concept analysis. Belitung Nursing Journal 2024, 10(3), 252–60. [Google Scholar] [CrossRef]
- Chan, K; Palis, BE; Cotler, JH; Janczewski, LM; Weigel, RJ; Bentrem, DJ; et al. Association of commission on cancer accreditation with receipt of guideline-concordant care and survival among patients with colon cancer. World J Surg 2025, 49(1), 34–45. [Google Scholar] [CrossRef]
- Emile, SH; Horesh, N; Garoufalia, Z; Dourado, J; Rogers, P; Salama, E; et al. Accuracy of Clinical Staging of Localized Colon Cancer: A National Cancer Database Cohort Analysis. Ann Surg Oncol. 2024, 31(10), 6461–9. [Google Scholar]
- Emmanuel, A; Chohda, E; Botfield, C; Ellul, J. Accelerated discharge within 72 hours of colorectal cancer resection using simple discharge criteria. Ann R Coll Surg Engl. 2018, 100(1), 52–6. [Google Scholar] [CrossRef]
- Gögenur, M; Rosen, AW; Iveson, T; Kerr, RS; Saunders, MP; Cassidy, J; et al. Time From Colorectal Cancer Surgery to Adjuvant Chemotherapy: Post Hoc Analysis of the SCOT Randomized Clinical Trial. JAMA Surg 2024, 159(8), 865–71. [Google Scholar] [CrossRef] [PubMed]
- Baxter, NN; Kennedy, EB; Bergsland, E; Berlin, J; George, TJ; Gill, S; et al. Adjuvant Therapy for Stage II Colon Cancer: ASCO Guideline Update. J Clin Oncol. 2022, 40(8), 892–910. [Google Scholar] [CrossRef] [PubMed]
- Xu, B; Yu, L; Zhao, L-Z; Ma, D-W. Prognostic factors in the patients with T2N0M0 colorectal cancer. World J Surg Oncol 2016, 14, 76. [Google Scholar] [CrossRef]
- Dunn, C; Halpern, MT; Sapkaroski, D; Gibbs, P. Measuring the quality of care in metastatic colorectal cancer: a scoping review of quality indicators. JNCI Cancer Spectr 2024, 8(5), kae073. [Google Scholar] [CrossRef]
- Kehl, KL; Lathan, CS; Johnson, BE; Schrag, D. Race, Poverty, and Initial Implementation of Precision Medicine for Lung Cancer. J Natl Cancer Inst. 2019, 111(4), 431–4. [Google Scholar] [CrossRef]
- Nogueira, LM; May, FP; Yabroff, KR; Siegel, RL. Racial Disparities in Receipt of Guideline-Concordant Care for Early-Onset Colorectal Cancer in the United States. J Clin Oncol. 2024, 42(12), 1368–77. [Google Scholar] [CrossRef]
- The tradeoffs: Personalized treatment vs. one-size-fits-all standards [Internet]. Available online: https://phrma.org/blog/the-tradeoffs-personalized-treatment-vs-one-size-fits-all-standards (accessed on 21 January 2026).
- Riihimäki, M; Hemminki, A; Sundquist, J; Hemminki, K. Patterns of metastasis in colon and rectal cancer. Sci Rep. 2016, 6(1), 29765. [Google Scholar] [CrossRef]
- Balboa-Barreiro, V; Pértega-Díaz, S; García-Rodríguez, T; González-Martín, C; Pardeiro-Pértega, R; Yáñez-González-Dopeso, L; et al. Colorectal cancer recurrence and its impact on survival after curative surgery: An analysis based on multistate models. Digestive and Liver Disease 2024, 56(7), 1229–36. [Google Scholar] [CrossRef] [PubMed]
- Nors, J; Iversen, LH; Erichsen, R; Gotschalck, KA; Andersen, CL. Incidence of Recurrence and Time to Recurrence in Stage I to III Colorectal Cancer: A Nationwide Danish Cohort Study. JAMA Oncol. 2024, 10(1), 54–62. [Google Scholar] [CrossRef] [PubMed]
- Nors, J; Gotschalck, KA; Erichsen, R; Andersen, CL. Risk of recurrence in early-onset versus late-onset non-metastatic colorectal cancer, 2004–2019: a nationwide cohort study. The Lancet Regional Health – Europe [Internet]. 1 Dec 2024, p. 47. Available online: https://www.thelancet.com/journals/lanepe/article/PIIS2666-7762(24)00260-6/fulltext.
- Ferreira Pinto, P; Peyroteo, M; Baía, C; Marques, M; Cardoso, MJ; Videira, JF; et al. Early-Stage Colon Cancer Surveillance: Pattern and Timing of Recurrence and the Role of 5-Year Surveillance. GE Port J Gastroenterol 2024, 32(2), 109–17. [Google Scholar] [CrossRef]
- Cancer of the Colon and Rectum - Cancer Stat Facts [Internet]. Available online: https://seer.cancer.gov/statfacts/html/colorect.html (accessed on 21 January 2026).
- Nappi, A; Berretta, M; Romano, C; Tafuto, S; Cassata, A; Casaretti, R; et al. Metastatic Colorectal Cancer: Role of Target Therapies and Future Perspectives. Curr Cancer Drug Targets 2018, 18(5), 421–9. [Google Scholar] [CrossRef]
- Tsilimigras, DI; Ntanasis-Stathopoulos, I; Pawlik, TM. Molecular Mechanisms of Colorectal Liver Metastases. Cells 2023, 12(12), 1657. [Google Scholar] [CrossRef] [PubMed]
- André, T; Boni, C; Mounedji-Boudiaf, L; Navarro, M; Tabernero, J; Hickish, T; et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004, 350(23), 2343–51. [Google Scholar] [CrossRef] [PubMed]
- André, T; Boni, C; Mounedji-Boudiaf, L; Navarro, M; Tabernero, J; Hickish, T; et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004, 350(23), 2343–51. [Google Scholar] [CrossRef] [PubMed]
- Oxaliplatin-based therapy: strategies to prevent or minimize neurotoxicity - WCRJ [Internet]. Available online: https://www.wcrj.net/article/232 (accessed on 23 January 2026).
- Narita, Y; Taniguchi, H; Komori, K; Kimura, K; Kinoshita, T; Komori, A; et al. Differences in attitude toward adjuvant chemotherapy between colorectal cancer survivors and the medical staff of Japanese hospitals. Int J Clin Oncol 2015, 20(4), 755–60. [Google Scholar] [CrossRef] [PubMed]
- Wakkerman, FC; Post, CCB; Mens, JWM; Jürgenliemk-Schulz, IM; Koppe, FLA; Haverkort, MAD; et al. Age-related differences in patient preferences for adjuvant chemotherapy for high-risk endometrial cancer. Maturitas [Internet]. 2026 Jan 1; 204. Available online: https://www.maturitas.org/article/S0378-5122(25)00613-9/fulltext (accessed on 23 January 2026).
- Park, SB; Lin, CSY; Krishnan, AV; Goldstein, D; Friedlander, ML; Kiernan, MC. Long-term neuropathy after oxaliplatin treatment: challenging the dictum of reversibility. Oncologist 2011, 16(5), 708–16. [Google Scholar] [CrossRef]
- Choi, J; Kong, K; Mozaffar, T; Holcombe, RF. Delayed oxaliplatin-associated neurotoxicity following adjuvant chemotherapy for stage III colon cancer. Anticancer Drugs 2006, 17(1), 103–5. [Google Scholar] [CrossRef]
- Tofthagen, C; Donovan, KA; Morgan, MA; Shibata, D. Oxaliplatin-Induced Peripheral Neuropathy’s Effects on Health-Related Quality of Life of Colorectal Cancer Survivors. Support Care Cancer 2013, 21(12), 3307–13. [Google Scholar] [CrossRef]
- Marquart, J; Chen, EY; Prasad, V. Estimation of the Percentage of US Patients With Cancer Who Benefit From Genome-Driven Oncology. JAMA Oncol 2018, 4(8), 1093–8. [Google Scholar] [CrossRef]
- Suehnholz, SP; Nissan, MH; Zhang, H; Kundra, R; Nandakumar, S; Lu, C; et al. Quantifying the Expanding Landscape of Clinical Actionability for Patients with Cancer. Cancer Discov. 2024, 14(1), 49–65. [Google Scholar] [CrossRef]
- Jan, Y-H; Tan, KT; Chen, S-J; Yip, TTC; Lu, CT; Lam, AK-Y. Comprehensive assessment of actionable genomic alterations in primary colorectal carcinoma using targeted next-generation sequencing. Br J Cancer 2022, 127(7), 1304–11. [Google Scholar] [CrossRef]
- Napoli, GC; Figg, WD; Chau, CH. Functional drug screening in the era of precision medicine. Front Med (Lausanne) 2022, 9, 912641. [Google Scholar] [CrossRef]
- Letai, A. Functional precision cancer medicine—moving beyond pure genomics. News@nat,Com 2017, 23, 1028–35. [Google Scholar] [CrossRef] [PubMed]
- Su, Y. Cancer Chemosensitivity Testing: Review. J Cancer Ther. 2014, 05(07), 672–9. [Google Scholar] [CrossRef]
- Weisenthal, LM; Lippman, ME. Clonogenic and nonclonogenic in vitro chemosensitivity assays. Cancer Treat Rep. 1985, 69(6), 615–32. [Google Scholar]
- Crystal, AS; Shaw, AT; Sequist, LV; Friboulet, L; Niederst, MJ; Lockerman, EL; et al. Patient-derived models of acquired resistance can identify effective drug combinations for cancer. Science 2014, 346(6216), 1480–6. [Google Scholar] [CrossRef]
- Friedman, AA; Letai, A; Fisher, DE; Flaherty, KT. Precision medicine for cancer with next-generation functional diagnostics. Nat Rev Cancer 2015, 15(12), 747–56. [Google Scholar] [CrossRef]
- Morand du Puch, CB; Vanderstraete, M; Giraud, S; Lautrette, C; Christou, N; Mathonnet, M. Benefits of functional assays in personalized cancer medicine: more than just a proof-of-concept. Theranostics 2021, 11(19), 9538–56. [Google Scholar] [CrossRef]
- Tentler, JJ; Tan, AC; Weekes, CD; Jimeno, A; Leong, S; Pitts, TM; et al. Patient-derived tumour xenografts as models for oncology drug development. Nat Rev Clin Oncol. 2012, 9(6), 338–50. [Google Scholar] [CrossRef]
- Hidalgo, M; Amant, F; Biankin, AV; Budinská, E; Byrne, AT; Caldas, C; et al. Patient-derived xenograft models: an emerging platform for translational cancer research. Cancer Discov. 2014, 4(9), 998–1013. [Google Scholar] [CrossRef] [PubMed]
- Byrne, AT; Alférez, DG; Amant, F; Annibali, D; Arribas, J; Biankin, AV; et al. Interrogating open issues in cancer precision medicine with patient-derived xenografts. Nat Rev Cancer 2017, 17(4), 254–68. [Google Scholar] [CrossRef]
- Ben-David, U; Ha, G; Tseng, Y-Y; Greenwald, NF; Oh, C; Shih, J; et al. Patient-derived xenografts undergo mouse-specific tumor evolution. Nat Genet. 2017, 49(11), 1567–75. [Google Scholar] [CrossRef]
- Bhatia, SN; Ingber, DE. Microfluidic organs-on-chips. Nat Biotechnol 2014, 32(8), 760–72. [Google Scholar] [CrossRef] [PubMed]
- Low, LA; Mummery, C; Berridge, BR; Austin, CP; Tagle, DA. Organs-on-chips: into the next decade. Nat Rev Drug Discov. 2021, 20(5), 345–61. [Google Scholar] [CrossRef]
- Zhang, B; Radisic, M. Organ-on-a-chip devices advance to market. Lab Chip 2017, 17(14), 2395–420. [Google Scholar] [CrossRef]
- Oliver, GR; Barnett, CC; Hightower, KE; Kang, Y; Baljevic, M. Ex vivo treatment response prediction in multiple myeloma: Assay formats, clinical correlation, and future directions. Cancers (Basel) [Internet] 2026, 18(3). [Google Scholar] [CrossRef]
- Önder, CE; Ziegler, T; Becker, R; Brucker, S; Hartkopf, A; Engler, T; et al. Advancing cancer therapy predictions with patient-derived organoid models of metastatic breast cancer. Cancers (Basel) [Internet]. Available from. 1 July 2023, 15. [CrossRef]
- Pan, B; Zhao, D; Liu, Y; Li, N; Song, C; Li, N; et al. Breast cancer organoids from malignant pleural effusion-derived tumor cells as an individualized medicine platform. In Vitro Cell Dev Biol Anim 2021, 57(5), 510–8. [Google Scholar] [CrossRef]
- Choi, W; Kim, YH; Woo, SM; Yu, Y; Lee, MR; Lee, WJ; et al. Establishment of patient-derived organoids using ascitic or pleural fluid from cancer patients. Cancer Research and Treatment: Official Journal of Korean Cancer Association 2023, 55(4), 1077–86. [Google Scholar] [CrossRef]
- Sakshaug, BC; Folkesson, E; Haukaas, TH; Visnes, T; Flobak, Å. Systematic review: predictive value of organoids in colorectal cancer. Sci Rep. 2023, 13(1), 18124. [Google Scholar] [CrossRef] [PubMed]
- Tong, L; Cui, W; Zhang, B; Fonseca, P; Zhao, Q; Zhang, P; et al. Patient-derived organoids in precision cancer medicine. Med (N Y) 2024, 5(11), 1351–77. [Google Scholar] [CrossRef] [PubMed]
- Taurin, S; Alzahrani, R; Aloraibi, S; Ashi, L; Alharmi, R; Hassani, N. Patient-derived tumor organoids: A preclinical platform for personalized cancer therapy. Transl Oncol. 2025, 51(102226), 102226. [Google Scholar] [CrossRef]
- Zhao, Z; Chen, X; Dowbaj, AM; Sljukic, A; Bratlie, K; Lin, L; et al. Organoids. Nat Rev Methods Primers [Internet] Available from. 2022, 2(1). [Google Scholar] [CrossRef]
- Wensink, GE; Elias, SG; Mullenders, J; Koopman, M; Boj, SF; Kranenburg, OW; et al. Patient-derived organoids as a predictive biomarker for treatment response in cancer patients. NPJ Precis Oncol 2021, 5(1), 30. [Google Scholar] [CrossRef]
- Jiang, S; Zhao, H; Zhang, W; Wang, J; Liu, Y; Cao, Y; et al. An automated organoid platform with inter-organoid homogeneity and inter-patient heterogeneity. Cell Rep Med. 2020, 1(9), 100161. [Google Scholar] [CrossRef]
- Yang, C; Yang, L; Feng, Y; Song, X; Bai, S; Zhang, S; et al. Modeling methods of different tumor organoids and their application in tumor drug resistance research. Canc Drug Resist 2025, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Foo, MA; You, M; Chan, SL; Sethi, G; Bonney, GK; Yong, W-P; et al. Clinical translation of patient-derived tumour organoids- bottlenecks and strategies. Biomark Res. 2022, 10(1), 10. [Google Scholar] [CrossRef] [PubMed]
- Yang, H; Li, J; Wang, Z; Khutsishvili, D; Tang, J; Zhu, Y; et al. Bridging the organoid translational gap: integrating standardization and micropatterning for drug screening in clinical and pharmaceutical medicine. Life Med 2024, 3(2), lnae016. [Google Scholar] [CrossRef] [PubMed]
- Xiang, D; He, A; Zhou, R; Wang, Y; Xiao, X; Gong, T; et al. Building consensus on the application of organoid-based drug sensitivity testing in cancer precision medicine and drug development. Theranostics 2024, 14(8), 3300–16. [Google Scholar] [CrossRef]
- NIH establishes nation’s first dedicated organoid development center to reduce reliance on animal modeling [Internet]. National Institutes of Health (NIH). Available online: https://www.nih.gov/news-events/news-releases/nih-establishes-nations-first-dedicated-organoid-development-center-reduce-reliance-animal-modeling (accessed on 8 October 2025).
- Gobits, R; Schleußner, N; Oliver, GR; Rutenberg Schoenberg, M; de Jesus Domingues, AM; Ramkumar, P; et al. Functional precision medicine using MicroOrganoSpheres for treatment response prediction in advanced colorectal cancer. JCO Precis Oncol 2026, 10, e2500501. [Google Scholar] [CrossRef]
- Nakamura, Y; Watanabe, J; Akazawa, N; Hirata, K; Kataoka, K; Yokota, M; et al. ctDNA-based molecular residual disease and survival in resectable colorectal cancer. Nat Med. 2024, 30(11), 3272–83. [Google Scholar] [CrossRef]
- Tie, J; Cohen, JD; Lahouel, K; Lo, SN; Wang, Y; Kosmider, S; et al. Circulating Tumor DNA Analysis Guiding Adjuvant Therapy in Stage II Colon Cancer. N Engl J Med. 2022, 386(24), 2261–72. [Google Scholar] [CrossRef]
- Fan, Q; Fu, Z; Xiong, D. Advantages and prospects of robotic surgery for colorectal cancer. Intelligent Surgery 2025, 8, 1–7. [Google Scholar] [CrossRef]
- Gustafsson, UO; Rockall, TA; Wexner, S; How, KY; Emile, S; Marchuk, A; et al. Guidelines for perioperative care in elective colorectal surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations 2025. Surgery 2025, 184, 109397. [Google Scholar] [CrossRef] [PubMed]
- Venook, AP. Colorectal Cancer Surveillance With Circulating Tumor DNA Assay. JAMA Netw Open 2022, 5(3), e221100. [Google Scholar] [CrossRef]
- Reinert, T; Henriksen, TV; Christensen, E; Sharma, S; Salari, R; Sethi, H; et al. Analysis of Plasma Cell-Free DNA by Ultradeep Sequencing in Patients With Stages I to III Colorectal Cancer. JAMA Oncol 2019, 5(8), 1124–31. [Google Scholar] [CrossRef]
- Kim, DH; Rockwood, K. Frailty in Older Adults. N Engl J Med 2024, 391(6), 538–48. [Google Scholar] [CrossRef] [PubMed]
- Sung, H; Siegel, RL; Laversanne, M; Jiang, C; Morgan, E; Zahwe, M; et al. Colorectal cancer incidence trends in younger versus older adults: an analysis of population-based cancer registry data. The Lancet Oncology 2025, 26(1), 51–63. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).