Submitted:
13 February 2026
Posted:
13 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Section
2.1. Chemical Reagents
2.2. Synthesis of Pristine SrTiO3 and Al-Doped SrTiO3
2.3. Photoelectrode Fabrication
2.4. Characterization
2.5. Photoelectrochemical (PEC) Measurements
3. Results and Discussion
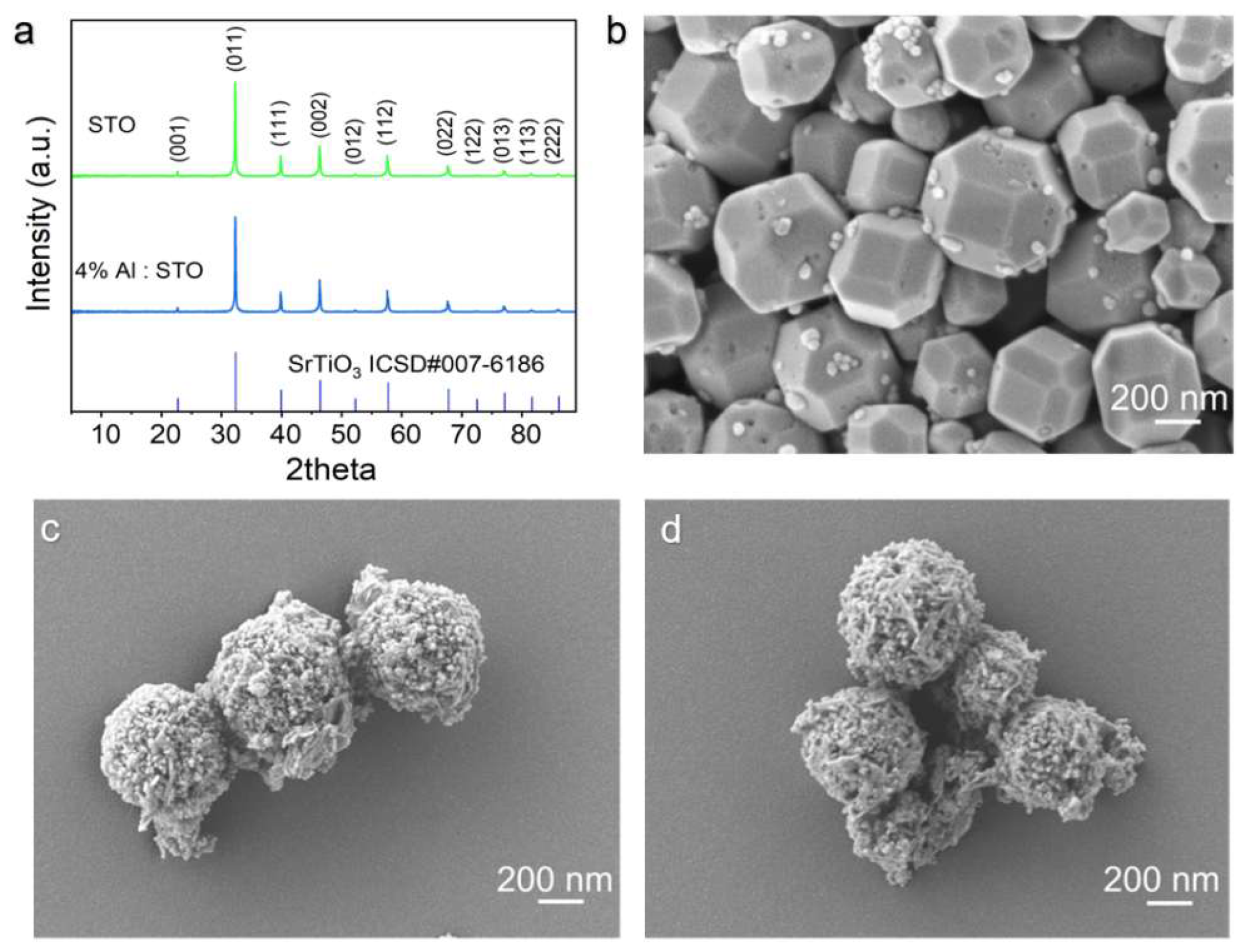
3.1. Structural and Morphological Characteristics
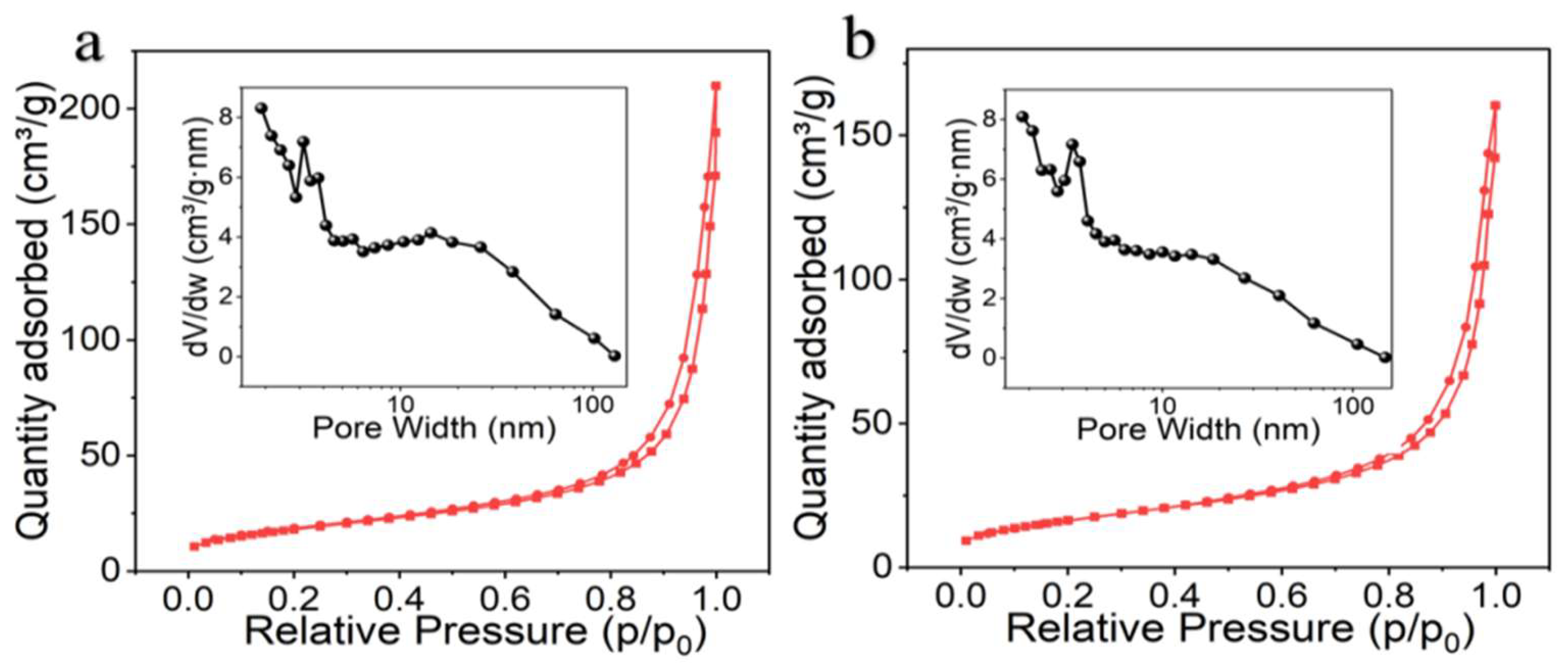
3.2. Textural Properties
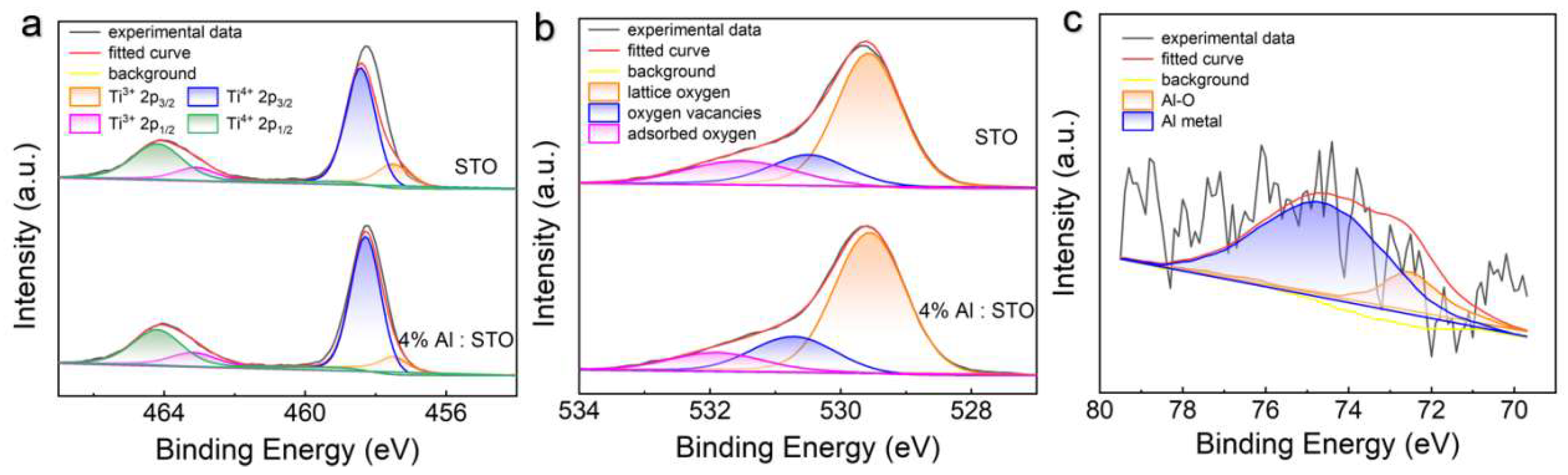
3.3. Chemical States and Defect Evolution
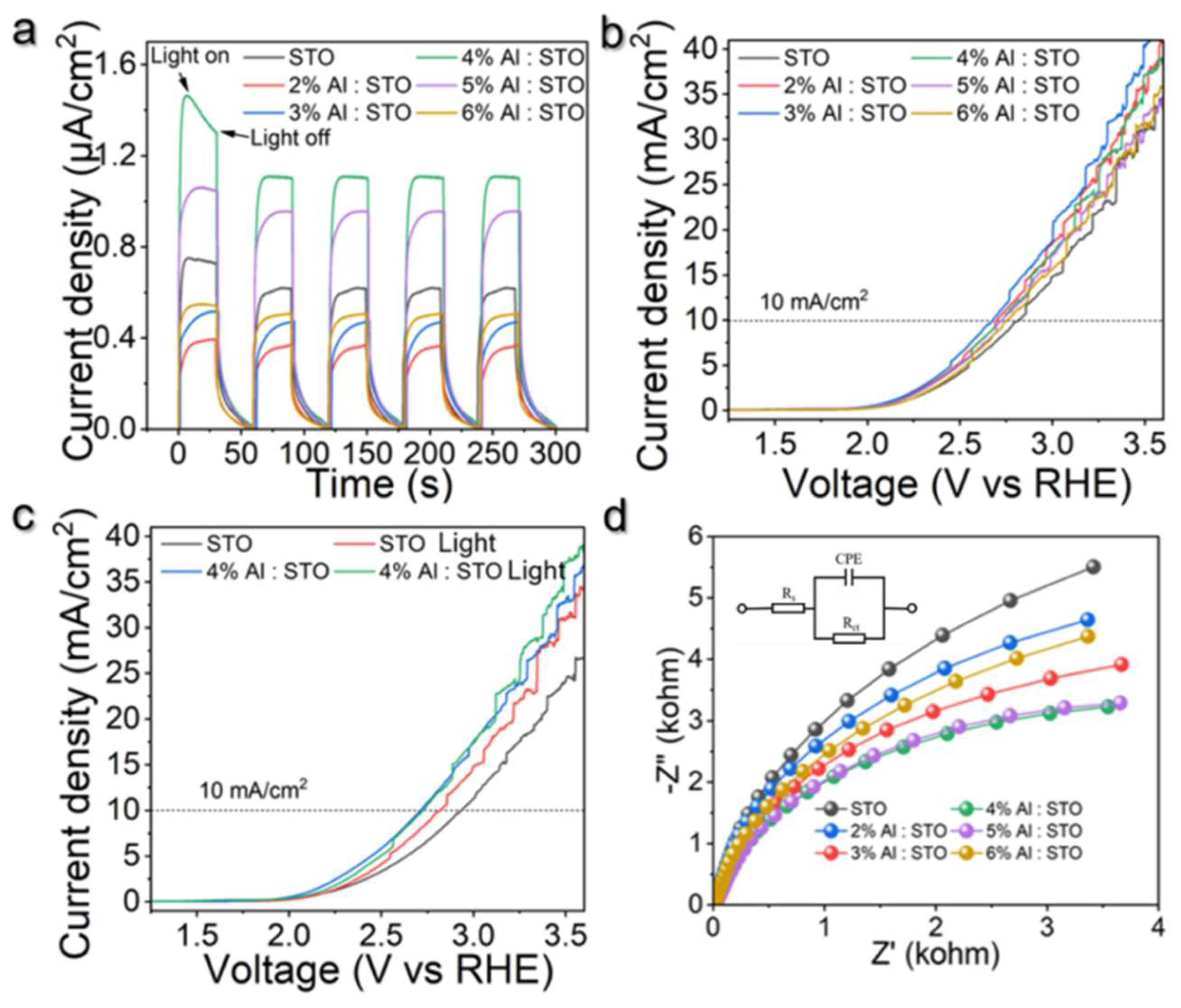
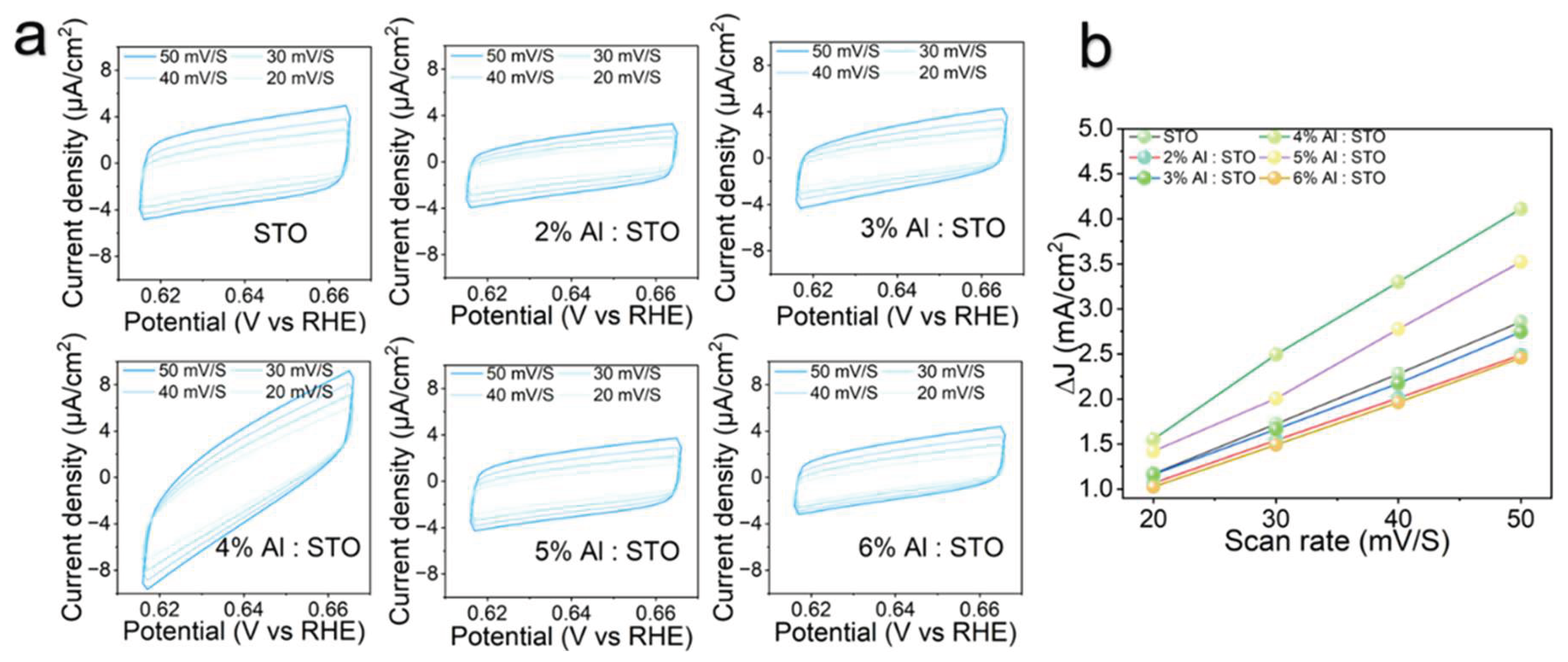
3.4. Photoelectrochemical Performance and Charge-Transfer Kinetics
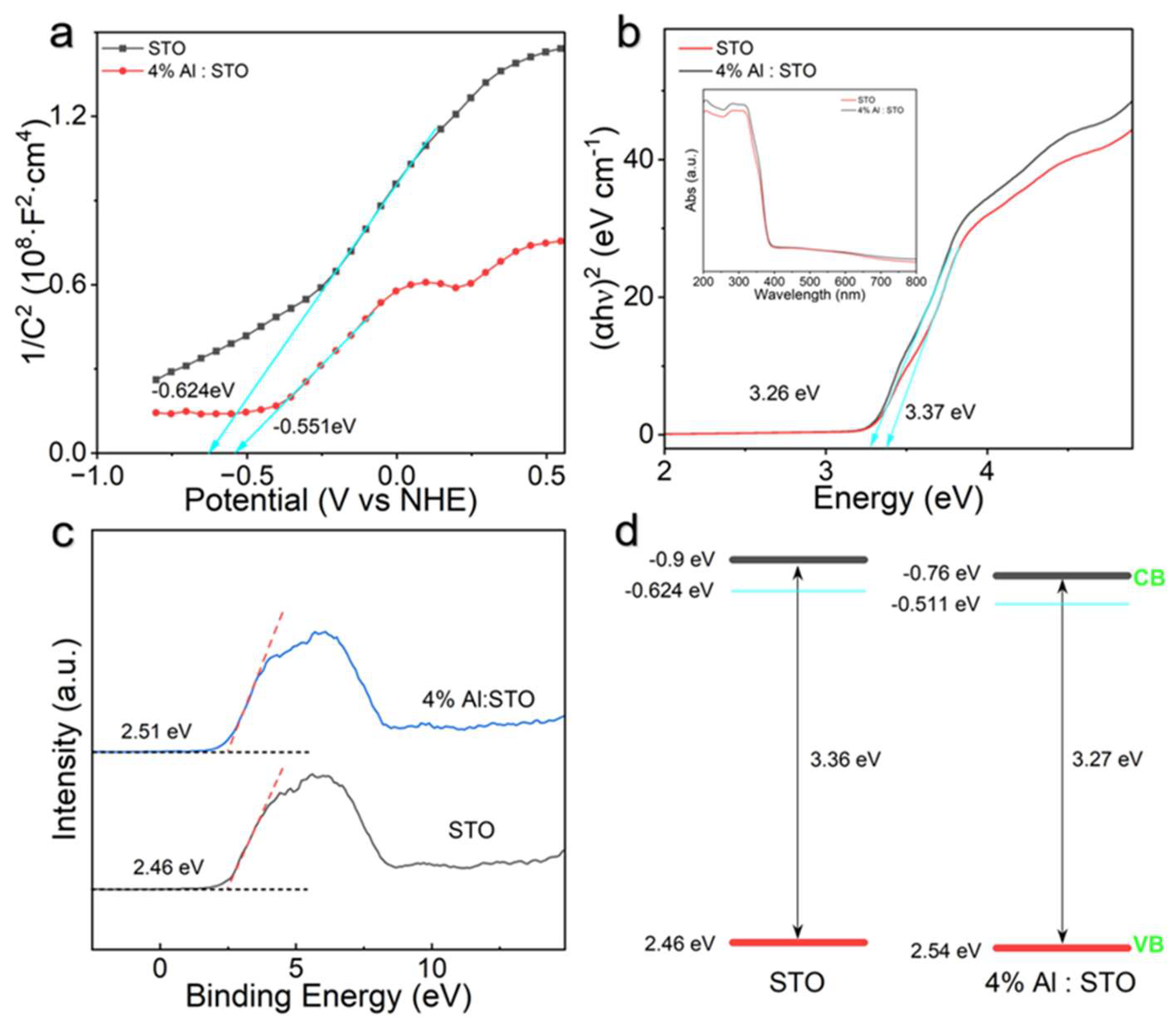
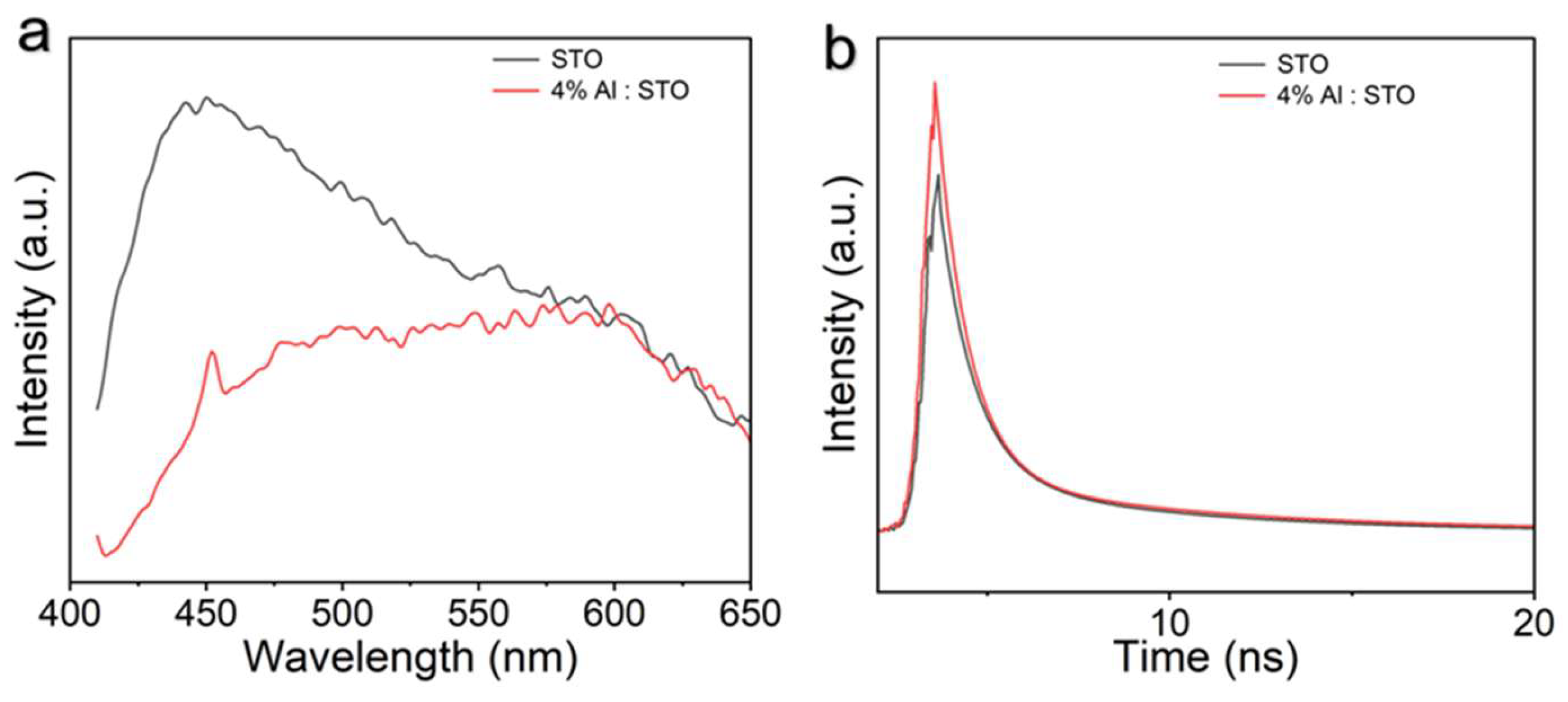
3.5. Band Structure and Charge Recombination
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Masoumi, Z.; Tayebi, M.; Kolaei, M.; Lee, B.-K. Efficient and stable core-shell α–Fe2O3/WS2/WOx photoanode for oxygen evolution reaction to enhance photoelectrochemical water splitting. App. Catal. B: Environ. 2022, 313, 121447. [Google Scholar] [CrossRef]
- Duan, W.J.; Han, J.C.; Yang, S.; Wang, Z.M.; Yu, Q.B.; Zhan, Y.Q. Understanding CO2 adsorption in layered double oxides synthesized by slag through kinetic and modelling techniques. Energy 2024, 297, 131303. [Google Scholar] [CrossRef]
- Joy, R.; Meena, B.; Kumar, M.; Jospeh, M.; Joseph, S.; Subrahmanyam, C.; Haridas, S. Exploring the role of ZnS as passivation layer on SrTiO3/Bi2S3 heterojunction photoanode for improved solar water splitting. Catal. Today 2024, 433, 114669. [Google Scholar] [CrossRef]
- Wang, W.; Xu, M.; Xu, X.; Zhou, W.; Shao, Z. Perovskite Oxide Based Electrodes for High-Performance Photoelectrochemical Water Splitting. Angew. Chem. Int. Ed. 2019, 59(1), 136–152. [Google Scholar] [CrossRef]
- Avcıoǧlu, C.; Avcıoǧlu, S.; Bekheet, M.F.; Gurlo, A. Photocatalytic Overall Water Splitting by SrTiO3: Progress Report and Design Strategies. ACS Appl. Energy Mater. 2023, 6(3), 1134–1154. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, H.; Xue, L.; Sun, J.; Wang, X.; Xiong, P.; Zhu, J. Recent advances in the heteroatom doping of perovskite oxides for efficient electrocatalytic reactions. Nanoscale 2021, 13(47), 19840–19856. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Li, S.; Zhong, S.; Zhou, X.; Ge, Y.; Luo, J.; Zhou, X.; Zhou, X.; Zhong, Y. Al-SrTiO3 decorated with non-noble metal co-catalyst NC-W2N for boosting photocatalytic overall water splitting via enhancing interfacial redox activity and charge separation. J. Alloy. Compd. 2023, 947, 169515. [Google Scholar] [CrossRef]
- Lv, S.; Pei, M.; Liu, Y.; Si, Z.; Wu, X.; Ran, R.; Weng, D.; Kang, F. A strategy to construct a highly active CoxP/SrTiO3(Al) catalyst to boost the photocatalytic overall water splitting reactions. Nanoscale 2022, 14(6), 2427–2433. [Google Scholar] [CrossRef]
- Wang, Q.; Hisatomi, T.; Ma, S.S.K.; Li, Y.; Domen, K. Core/Shell Structured La- and Rh-Codoped SrTiO3 as a Hydrogen Evolution Photocatalyst in Z-Scheme Overall Water Splitting under Visible Light Irradiation. Chem. Mater. 2014, 26(14), 4144–4150. [Google Scholar] [CrossRef]
- Cao, D.; Zhang, J.; Wang, A.; Yu, X.; Mi, B. Fabrication of Cr-doped SrTiO3/Ti-doped α-Fe2O3 photoanodes with enhanced photoelectrochemical properties. J. Mater. Sci. Technol. 2020, 56, 189–195. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Z.-H.; Li, W.; Ding, P.C.; Wang, M.M.; Tang, Y.Y.; Lin, H.Y.; Peng, Y.; Wang, M.Y.; Zheng, Z.; Yang, S.; Dai, S.; Zhang, X.; Liu, P.F.; Yang, H.G. Al3+-Dependent Anisotropic Facet Tailoring on SrTiO3 Single Crystal for Photocatalytic Overall Water Splitting. Angew. Chem. Int. Ed. 2025, 64(30), p. e202508114. [Google Scholar] [CrossRef]
- Qin, Y.L.; Fang, F.; Xie, Z.Z.; Lin, H.W.; Zhang, K.; Yu, X.; Chang, K. La,Al-Codoped SrTiO3 as a Photocatalyst in Overall Water Splitting: Significant Surface Engineering Effects on Defect Engineering. ACS Catal. 2021, 11(18), 11429–11439. [Google Scholar] [CrossRef]
- Li, R.; Takata, T.; Zhang, B.; Feng, C.; Wu, Q.; Cui, C.; Zhang, Z.; Domen, K.; Li, Y. Criteria for Efficient Photocatalytic Water Splitting Revealed by Studying Carrier Dynamics in a Model Al-doped SrTiO3 Photocatalyst. Angew. Chem. Int. Ed. 2023, 62(49), p. e202313537. [Google Scholar] [CrossRef]
- Sakata, Y.; Miyoshi, Y.; Maeda, T.; Ishikiriyama, K.; Yamazaki, Y.; Imamura, H.; Ham, Y.; Hisatomi, T.; Kubota, J.; Yamakata, A.; Domen, K. Photocatalytic property of metal ion added SrTiO3 to Overall H2O splitting. Appl. Catal. A-Gen. 2016, 521, 227–232. [Google Scholar] [CrossRef]
- Ham, Y.; Hisatomi, T.; Goto, Y.; Moriya, Y.; Sakata, Y.; Yamakata, A.; Kubota, J.; Domen, K. Flux-mediated doping of SrTiO3 photocatalysts for efficient overall water splitting. J. Mater. Chem. A 2016, 4(8), 3027–3033. [Google Scholar] [CrossRef]
- Zhao, Z.; Goncalves, R.V.; Barman, S.K.; Willard, E.J.; Byle, E.; Perry, R.; Wu, Z.; Huda, M.N.; Moulé, A.J.; Osterloh, F.E. Electronic structure basis for enhanced overall water splitting photocatalysis with aluminum doped SrTiO3 in natural sunlight. Energ. Environ. Sci 2019, 12(4), 1385–1395. [Google Scholar] [CrossRef]
- Su, Z.Y.; Fang, F.; Li, X.; Han, W.J.; Liu, X.L.; Chang, K. Synergistic surface oxygen defect and bulk Ti3+ defect engineering on SrTiO3 for enhancing photocatalytic overall water splitting. J. Colloid Interface Sci. 2022, 626, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Zhao, Y.; Li, A.; Wang, S.; Wang, Z.; Yang, J.; Wang, Y.; Liu, T.; Chen, R.; Zhu, J.; Fan, F.; Li, R.; Li, C. Enhancing charge separation on high symmetry SrTiO3 exposed with anisotropic facets for photocatalytic water splitting. Energ. Environ. Sci 2016, 9(7), 2463–2469. [Google Scholar] [CrossRef]
- Tan, H.; Zhao, Z.; Zhu, W.-B.; Coker, E.N.; Li, B.; Zheng, M.; Yu, W.; Fan, H.; Sun, Z. Oxygen Vacancy Enhanced Photocatalytic Activity of Pervoskite SrTiO3. ACS Appl. Mater. Interfaces 2014, 6(21), 19184–19190. [Google Scholar] [CrossRef]
- Zhu, D.D.; Zhou, Q.X. Nitrogen doped g-C3N4 with the extremely narrow band gap for excellent photocatalytic activities under visible light. Appl. Catal. B: Environ. 2021, 281, 119474. [Google Scholar] [CrossRef]
- Ran, X.; Bai, Y.; Zeng, H.; Zhang, J.; Fu, H.; An, X.; Yang, X. Manipulating Oxygen Vacancy in SrTiO3 Nanoparticles to Achieve Enhanced Photoelectrochemical Performance in Water Splitting. ACS Appl. Nano Mater. 2024, 7(23), 27543–27554. [Google Scholar] [CrossRef]
- Wong, C.P.P.; Lai, C.W.; Lee, K.M.; Pan, G.T.; Huang, C.M.; Yang, T.C.K.; Juan, J.C. Enhanced Conductivity Boosts the Cathodic Performance of Aluminium-Doped SrTiO3 in Rechargeable Alkaline Zinc Battery. J. Electrochem. Soc. 2021, 168(8), p. 080530. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, L.; Liu, B.; Wang, X.; Wang, T.; Xiao, X.; Wang, S.; Huang, W. Engineering BiVO4 and Oxygen Evolution Cocatalyst Interfaces with Rapid Hole Extraction for Photoelectrochemical Water Splitting. ACS Catal. 2023, 13(9), 5938–5948. [Google Scholar] [CrossRef]
- Mo, G.L.; Jin, Y.K.; Deng, Y.J.; Zhang, C.H.; Liu, J.B.; Niu, Q.Y.; Gao, X.Y.; Bi, Y.B.; Chen, H.Y.; Li, P. Band-Gap Engineering of Isoreticular Hydrogen-Bonded Organic Frameworks for Boosting Photocatalytic Hydrogen Evolution. In Angew. Chem. Int. Ed.; 2026. [Google Scholar]
- Luo, Y.; Deng, B.; Pu, Y.; Liu, A.; Wang, J.; Ma, K.; Gao, F.; Gao, B.; Zou, W.; Dong, L. Interfacial coupling effects in g-C3N4/SrTiO3 nanocomposites with enhanced H2 evolution under visible light irradiation. App. Catal. B: Environ. 2019, 247, 1–9. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y.; Ni, D.; Chen, Z.; Wang, X.; Bu, Y.; Ao, J.P. Improvement of BiVO4 Photoanode Performance During Water Photo-Oxidation Using Rh-Doped SrTiO3 Perovskite as a Co-Catalyst. Adv. Funct. Mater. 2019, 29(32), p. 1902101. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



