Submitted:
12 February 2026
Posted:
13 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
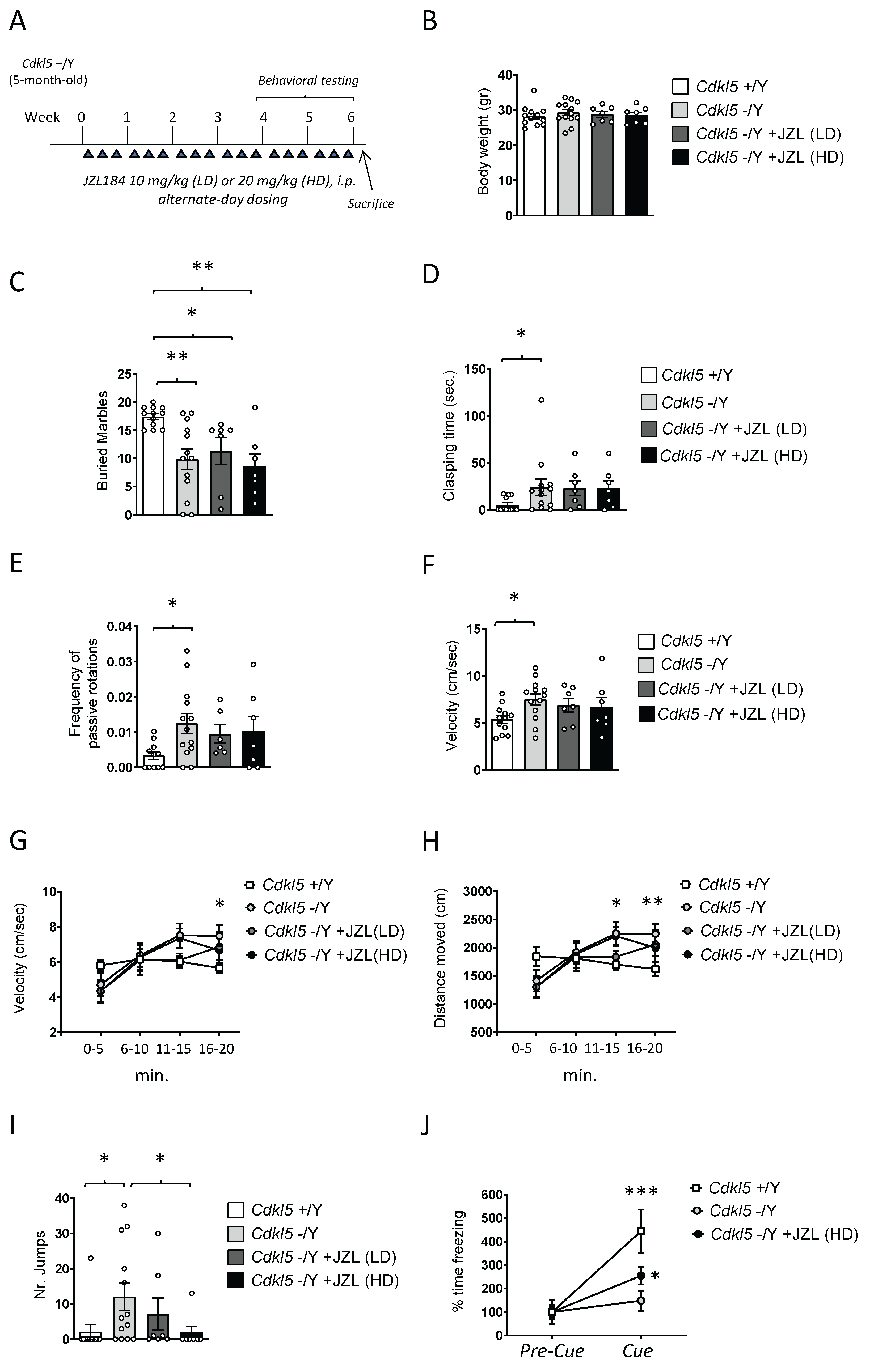
2.1. JZL184 Fails to Rescue Core Behavioral Deficits, with Only Mild Improvements at the Higher Dose, in Cdkl5 KO Mice
2.1.1. Rationale for Dosing Regimen and Treatment Schedule
2.1.2. Behavioral Assessment
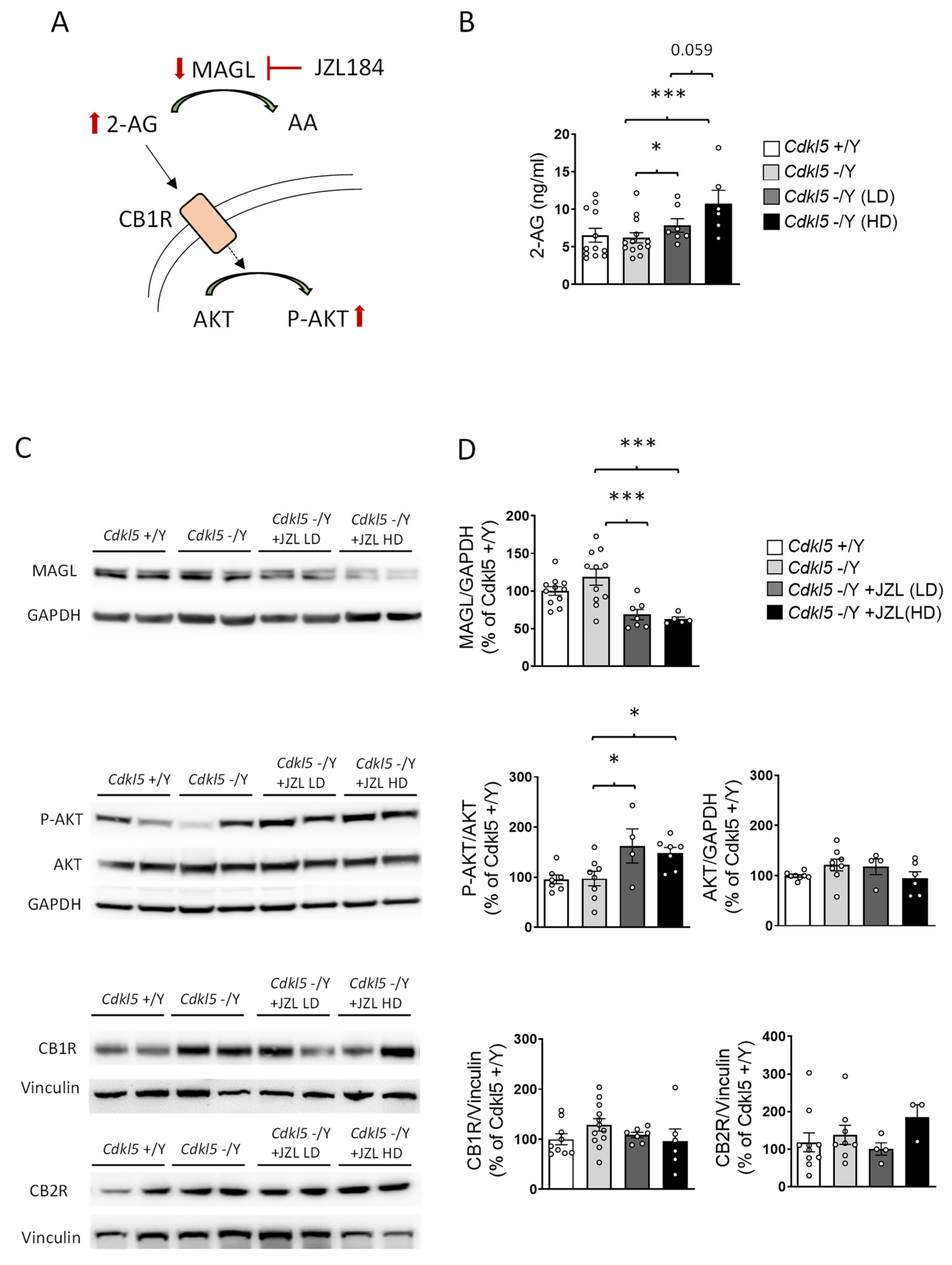
2.2. JZL184 Effectively Inhibits MAGL Activity and Increases 2-AG Levels in Cdkl5 KO Mice
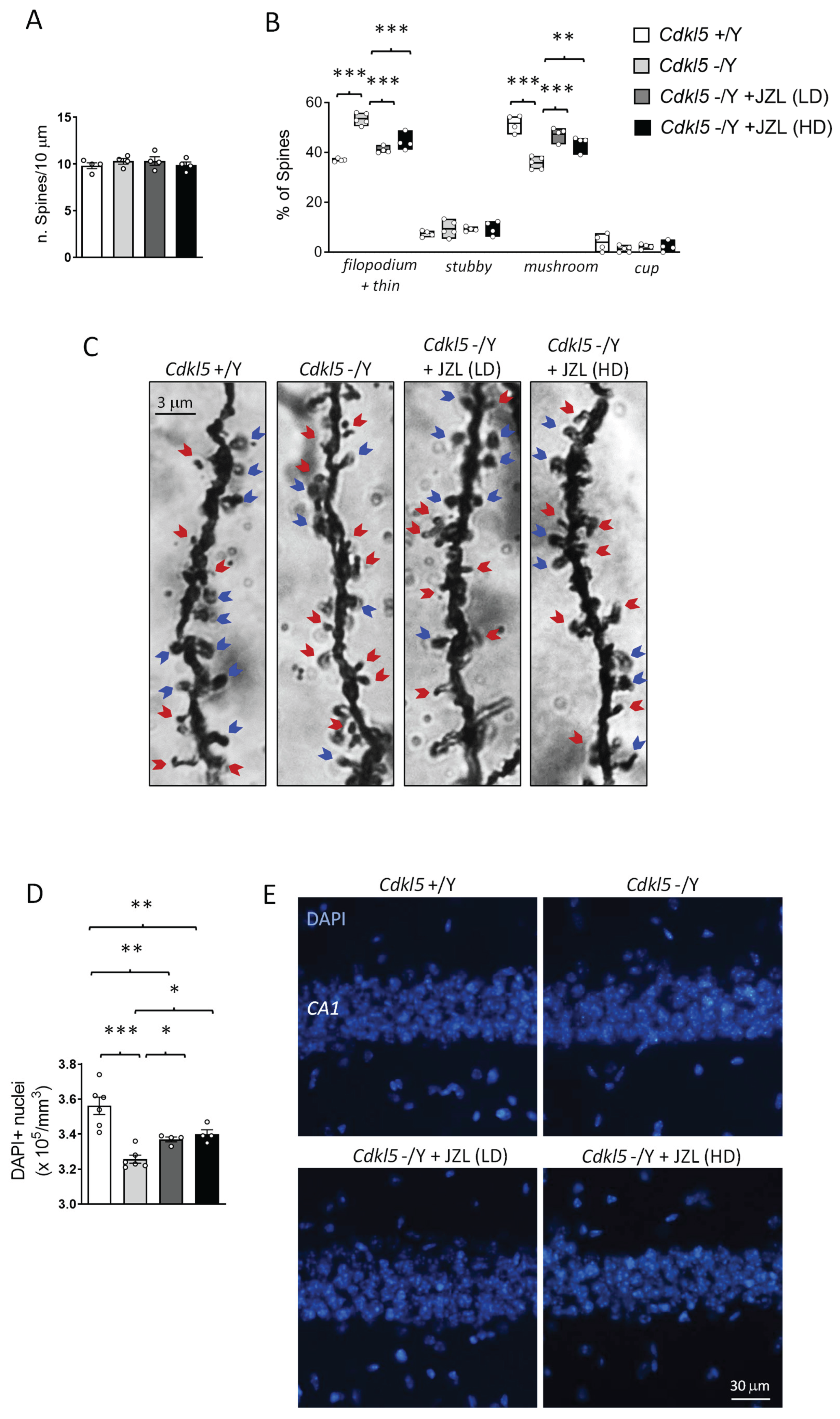
2.3. JZL184 Partial Rescues Cortical Spine Maturation and Hippocampal Neuronal Survival in Cdkl5 KO Mice
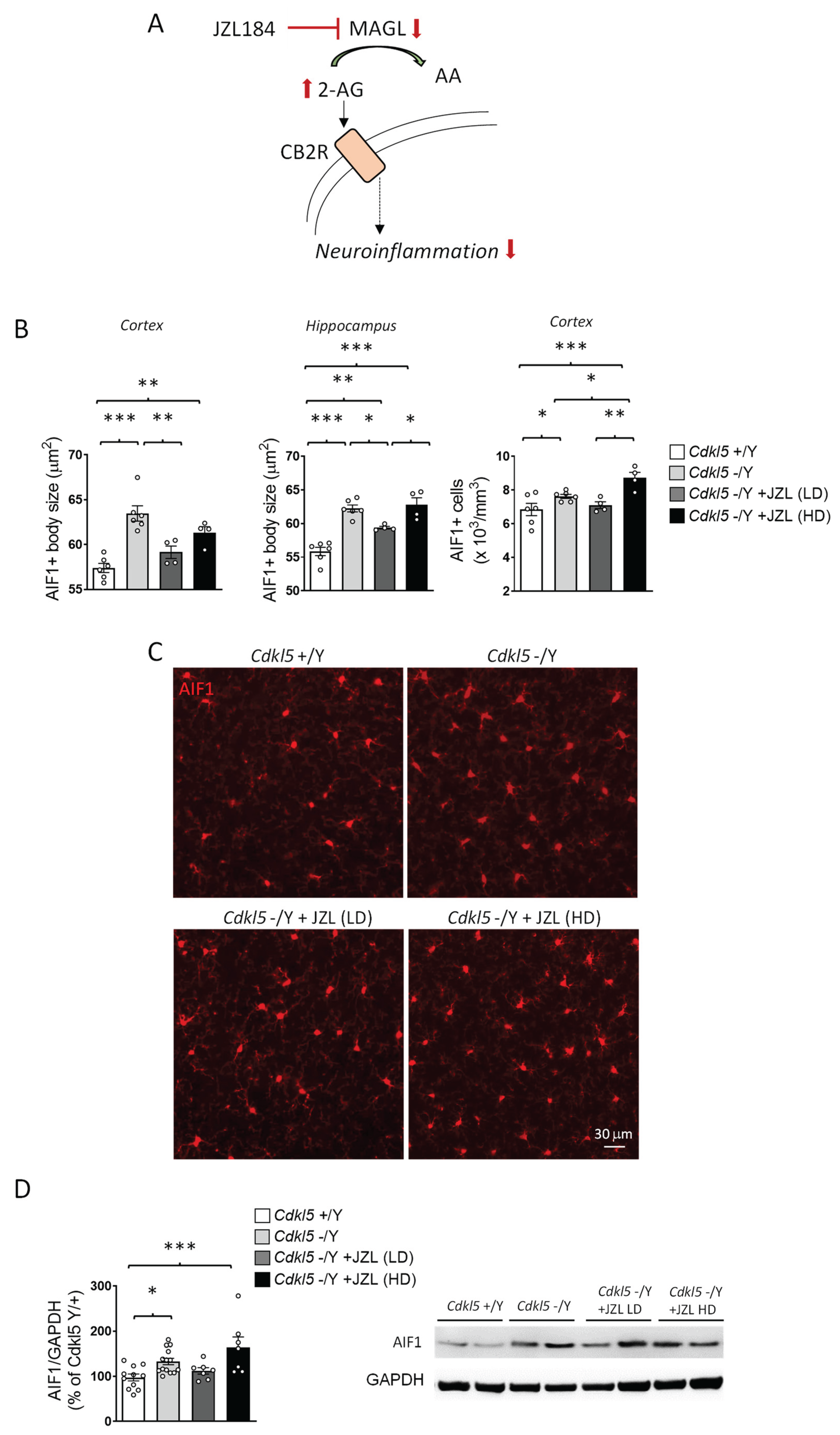
2.4. JZL184 Exerts Dose-Dependent and Only Partial Effects on Microglial Activation in Cdkl5 KO Mice
3. Discussion
4. Materials and Methods
4.1. Mouse Colony
4.2. JZL184 Treatment
4.3. Quantification of 2-Arachidonoylglycerol (2-AG) in Mouse Plasma
4.4. Behavioral Assays
4.4.1. Marble Burying Test
4.4.2. Hindlimb Clasping
4.4.3. Accelerating Rotarod
4.4.4. Open Field Test
4.4.5. Passive Avoidance
4.4.6. Fear Conditioning
4.4.7. Von Frey Filament Test
4.5. Histology and Immunohistochemistry
4.5.1. Immunofluorescence Staining
4.5.2. Golgi Staining
4.6. Measurements
4.6.1. Cell Density
4.6.2. Morphometric Microglial Cell Analysis
4.6.3. Dendritic Spine Number and Morphology
4.7. Western Blotting
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fehr, S.; Wilson, M.; Downs, J.; Williams, S.; Murgia, A.; Sartori, S.; Vecchi, M.; Ho, G.; Polli, R.; Psoni, S.; et al. The CDKL5 disorder is an independent clinical entity associated with early-onset encephalopathy. Eur J Hum Genet 2013, 21, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Demarest, S.; Pestana-Knight, E.M.; Olson, H.E.; Downs, J.; Marsh, E.D.; Kaufmann, W.E.; Partridge, C.A.; Leonard, H.; Gwadry-Sridhar, F.; Frame, K.E.; et al. Severity Assessment in CDKL5 Deficiency Disorder. Pediatr Neurol 2019, 97, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Demarest, S.T.; Olson, H.E.; Moss, A.; Pestana-Knight, E.; Zhang, X.; Parikh, S.; Swanson, L.C.; Riley, K.D.; Bazin, G.A.; Angione, K.; et al. CDKL5 deficiency disorder: Relationship between genotype, epilepsy, cortical visual impairment, and development. Epilepsia 2019, 60, 1733–1742. [Google Scholar] [CrossRef] [PubMed]
- Mangatt, M.; Wong, K.; Anderson, B.; Epstein, A.; Hodgetts, S.; Leonard, H.; Downs, J. Prevalence and onset of comorbidities in the CDKL5 disorder differ from Rett syndrome. Orphanet J Rare Dis 2016, 11, 39. [Google Scholar] [CrossRef]
- Van Bergen, N.J.; Massey, S.; Quigley, A.; Rollo, B.; Harris, A.R.; Kapsa, R.M.I.; Christodoulou, J. CDKL5 deficiency disorder: molecular insights and mechanisms of pathogenicity to fast-track therapeutic development. Biochem Soc Trans 2022, 50, 1207–1224. [Google Scholar] [CrossRef]
- Sun, X.; Wang, T. Research progress on the pathogenesis of CDKL5 pathogenic variants and related encephalopathy. Eur J Pediatr 2023, 182, 3049–3056. [Google Scholar] [CrossRef]
- Amendola, E.; Zhan, Y.; Mattucci, C.; Castroflorio, E.; Calcagno, E.; Fuchs, C.; Lonetti, G.; Silingardi, D.; Vyssotski, A.L.; Farley, D.; et al. Mapping pathological phenotypes in a mouse model of CDKL5 disorder. PLoS One 2014, 9, e91613. [Google Scholar] [CrossRef]
- Fuchs, C.; Gennaccaro, L.; Trazzi, S.; Bastianini, S.; Bettini, S.; Lo Martire, V.; Ren, E.; Medici, G.; Zoccoli, G.; Rimondini, R.; et al. Heterozygous CDKL5 Knockout Female Mice Are a Valuable Animal Model for CDKL5 Disorder. Neural Plast 2018, 2018, 9726950. [Google Scholar] [CrossRef]
- Lupori, L.; Sagona, G.; Fuchs, C.; Mazziotti, R.; Stefanov, A.; Putignano, E.; Napoli, D.; Strettoi, E.; Ciani, E.; Pizzorusso, T. Site-specific abnormalities in the visual system of a mouse model of CDKL5 deficiency disorder. Hum Mol Genet 2019, 28, 2851–2861. [Google Scholar] [CrossRef]
- Pizzo, R.; Gurgone, A.; Castroflorio, E.; Amendola, E.; Gross, C.; Sassoe-Pognetto, M.; Giustetto, M. Lack of Cdkl5 Disrupts the Organization of Excitatory and Inhibitory Synapses and Parvalbumin Interneurons in the Primary Visual Cortex. Front Cell Neurosci 2016, 10, 261. [Google Scholar] [CrossRef]
- Leonard, H.; Downs, J.; Benke, T.A.; Swanson, L.; Olson, H.; Demarest, S. CDKL5 deficiency disorder: clinical features, diagnosis, and management. Lancet Neurol 2022, 21, 563–576. [Google Scholar] [CrossRef] [PubMed]
- Dale, T.; Downs, J.; Olson, H.; Bergin, A.M.; Smith, S.; Leonard, H. Cannabis for refractory epilepsy in children: A review focusing on CDKL5 Deficiency Disorder. Epilepsy Res 2019, 151, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Kosenkov, A.M.; Mal’tseva, V.N.; Maiorov, S.A.; Gaidin, S.G. The role of the endocannabinoid system in the pathogenesis and treatment of epilepsy. Rev Neurosci 2025, 36, 351–370. [Google Scholar] [CrossRef] [PubMed]
- Lowe, H.; Toyang, N.; Steele, B.; Bryant, J.; Ngwa, W. The Endocannabinoid System: A Potential Target for the Treatment of Various Diseases. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Maccarrone, M.; Di Marzo, V.; Gertsch, J.; Grether, U.; Howlett, A.C.; Hua, T.; Makriyannis, A.; Piomelli, D.; Ueda, N.; van der Stelt, M. Goods and Bads of the Endocannabinoid System as a Therapeutic Target: Lessons Learned after 30 Years. Pharmacol Rev 2023, 75, 885–958. [Google Scholar] [CrossRef]
- Rodrigues, R.J.; Marques, J.M.; Kofalvi, A. Cannabis, Endocannabinoids and Brain Development: From Embryogenesis to Adolescence. Cells 2024, 13. [Google Scholar] [CrossRef]
- Aguado, T.; Palazuelos, J.; Monory, K.; Stella, N.; Cravatt, B.; Lutz, B.; Marsicano, G.; Kokaia, Z.; Guzman, M.; Galve-Roperh, I. The endocannabinoid system promotes astroglial differentiation by acting on neural progenitor cells. J Neurosci 2006, 26, 1551–1561. [Google Scholar] [CrossRef]
- Aguado, T.; Romero, E.; Monory, K.; Palazuelos, J.; Sendtner, M.; Marsicano, G.; Lutz, B.; Guzman, M.; Galve-Roperh, I. The CB1 cannabinoid receptor mediates excitotoxicity-induced neural progenitor proliferation and neurogenesis. J Biol Chem 2007, 282, 23892–23898. [Google Scholar] [CrossRef]
- Berghuis, P.; Rajnicek, A.M.; Morozov, Y.M.; Ross, R.A.; Mulder, J.; Urban, G.M.; Monory, K.; Marsicano, G.; Matteoli, M.; Canty, A.; et al. Hardwiring the brain: endocannabinoids shape neuronal connectivity. Science 2007, 316, 1212–1216. [Google Scholar] [CrossRef]
- Li, X.; Yennawar, M.; Wiest, A.; O’Brien, W.T.; Babrowicz, B.; White, R.S.; Talos, D.M.; Jensen, F.E. Cannabidiol attenuates seizure susceptibility and behavioural deficits in adult CDKL5(R59X) knock-in mice. Eur J Neurosci 2024, 59, 3337–3352. [Google Scholar] [CrossRef]
- Perulli, M.; Bianchetti, M.; Pantalone, G.; Quintiliani, M.; Gambardella, M.L.; Picilli, M.; Marini, C.; Cesaroni, E.; Battaglia, D.I. Real-world efficacy and safety of cannabidiol in developmental and epileptic encephalopathies. Epilepsia Open 2025. [Google Scholar] [CrossRef] [PubMed]
- Massey, S.; Quigley, A.; Rochfort, S.; Christodoulou, J.; Van Bergen, N.J. Cannabinoids and Genetic Epilepsy Models: A Review with Focus on CDKL5 Deficiency Disorder. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Bononi, G.; Poli, G.; Rizzolio, F.; Tuccinardi, T.; Macchia, M.; Minutolo, F.; Granchi, C. An updated patent review of monoacylglycerol lipase (MAGL) inhibitors (2018-present). Expert Opin Ther Pat 2021, 31, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Long, J.Z.; Nomura, D.K.; Cravatt, B.F. Characterization of monoacylglycerol lipase inhibition reveals differences in central and peripheral endocannabinoid metabolism. Chem Biol 2009, 16, 744–753. [Google Scholar] [CrossRef]
- Long, J.Z.; Li, W.; Booker, L.; Burston, J.J.; Kinsey, S.G.; Schlosburg, J.E.; Pavon, F.J.; Serrano, A.M.; Selley, D.E.; Parsons, L.H.; et al. Selective blockade of 2-arachidonoylglycerol hydrolysis produces cannabinoid behavioral effects. Nat Chem Biol 2009, 5, 37–44. [Google Scholar] [CrossRef]
- Schlosburg, J.E.; Blankman, J.L.; Long, J.Z.; Nomura, D.K.; Pan, B.; Kinsey, S.G.; Nguyen, P.T.; Ramesh, D.; Booker, L.; Burston, J.J.; et al. Chronic monoacylglycerol lipase blockade causes functional antagonism of the endocannabinoid system. Nat Neurosci 2010, 13, 1113–1119. [Google Scholar] [CrossRef]
- Gennaccaro, L.; Fuchs, C.; Loi, M.; Pizzo, R.; Alvente, S.; Berteotti, C.; Lupori, L.; Sagona, G.; Galvani, G.; Gurgone, A.; et al. Age-Related Cognitive and Motor Decline in a Mouse Model of CDKL5 Deficiency Disorder is Associated with Increased Neuronal Senescence and Death. Aging Dis 2021, 12, 764–785. [Google Scholar] [CrossRef]
- Trent, S.; Abdullah, M.H.; Parwana, K.; Valdivieso, M.A.; Hassan, Z.; Muller, C.P. Fear conditioning: Insights into learning, memory and extinction and its relevance to clinical disorders. Prog Neuropsychopharmacol Biol Psychiatry 2025, 138, 111310. [Google Scholar] [CrossRef]
- Wang, I.T.; Allen, M.; Goffin, D.; Zhu, X.; Fairless, A.H.; Brodkin, E.S.; Siegel, S.J.; Marsh, E.D.; Blendy, J.A.; Zhou, Z. Loss of CDKL5 disrupts kinome profile and event-related potentials leading to autistic-like phenotypes in mice. Proc Natl Acad Sci U S A 2012, 109, 21516–21521. [Google Scholar] [CrossRef]
- Ma, M.; Bai, J.; Ling, Y.; Chang, W.; Xie, G.; Li, R.; Wang, G.; Tao, K. Monoacylglycerol lipase inhibitor JZL184 regulates apoptosis and migration of colorectal cancer cells. Mol Med Rep 2016, 13, 2850–2856. [Google Scholar] [CrossRef]
- Hu, H.Y.; Kruijssen, D.L.H.; Frias, C.P.; Rozsa, B.; Hoogenraad, C.C.; Wierenga, C.J. Endocannabinoid Signaling Mediates Local Dendritic Coordination between Excitatory and Inhibitory Synapses. Cell Rep 2019, 27, 666–675 e665. [Google Scholar] [CrossRef] [PubMed]
- Della Sala, G.; Putignano, E.; Chelini, G.; Melani, R.; Calcagno, E.; Michele Ratto, G.; Amendola, E.; Gross, C.T.; Giustetto, M.; Pizzorusso, T. Dendritic Spine Instability in a Mouse Model of CDKL5 Disorder Is Rescued by Insulin-like Growth Factor 1. Biol Psychiatry 2016, 80, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Trazzi, S.; Fuchs, C.; Viggiano, R.; De Franceschi, M.; Valli, E.; Jedynak, P.; Hansen, F.K.; Perini, G.; Rimondini, R.; Kurz, T.; et al. HDAC4: a key factor underlying brain developmental alterations in CDKL5 disorder. Hum Mol Genet 2016, 25, 3887–3907. [Google Scholar] [CrossRef] [PubMed]
- Rusconi, L.; Salvatoni, L.; Giudici, L.; Bertani, I.; Kilstrup-Nielsen, C.; Broccoli, V.; Landsberger, N. CDKL5 expression is modulated during neuronal development and its subcellular distribution is tightly regulated by the C-terminal tail. J Biol Chem 2008, 283, 30101–30111. [Google Scholar] [CrossRef]
- van der Stelt, M.; Di Marzo, V. Cannabinoid receptors and their role in neuroprotection. Neuromolecular Med 2005, 7, 37–50. [Google Scholar] [CrossRef]
- Tang, S.; Wang, I.J.; Yue, C.; Takano, H.; Terzic, B.; Pance, K.; Lee, J.Y.; Cui, Y.; Coulter, D.A.; Zhou, Z. Loss of CDKL5 in Glutamatergic Neurons Disrupts Hippocampal Microcircuitry and Leads to Memory Impairment in Mice. J Neurosci 2017, 37, 7420–7437. [Google Scholar] [CrossRef]
- Loi, M.; Trazzi, S.; Fuchs, C.; Galvani, G.; Medici, G.; Gennaccaro, L.; Tassinari, M.; Ciani, E. Increased DNA Damage and Apoptosis in CDKL5-Deficient Neurons. Mol Neurobiol 2020, 57, 2244–2262. [Google Scholar] [CrossRef]
- Stella, N. Endocannabinoid signaling in microglial cells. Neuropharmacology 2009, 56 Suppl 1, 244–253. [Google Scholar] [CrossRef]
- Kasatkina, L.A.; Rittchen, S.; Sturm, E.M. Neuroprotective and Immunomodulatory Action of the Endocannabinoid System under Neuroinflammation. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Lysenko, L.V.; Kim, J.; Henry, C.; Tyrtyshnaia, A.; Kohnz, R.A.; Madamba, F.; Simon, G.M.; Kleschevnikova, N.E.; Nomura, D.K.; Ezekowitz, R.A.; et al. Monoacylglycerol lipase inhibitor JZL184 improves behavior and neural properties in Ts65Dn mice, a model of down syndrome. PLoS One 2014, 9, e114521. [Google Scholar] [CrossRef]
- Zou, M.; Zhang, Y.; Du, C.; Yang, B.; Guo, P.; Liang, H.; Zhang, Y.; Tian, W.; Yang, L.; Liu, D.; et al. Augmentation of Endogenous 2-Arachidonoylglycerol Mitigates Autistic Behaviors of BTBR Mice. Mol Neurobiol 2025, 62, 5022–5038. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Moraga, A.; Beckers, T.; Luyten, L. The effects of stress on avoidance in rodents: An unresolved matter. Front Behav Neurosci 2022, 16, 983026. [Google Scholar] [CrossRef] [PubMed]
- Lutz, B.; Marsicano, G.; Maldonado, R.; Hillard, C.J. The endocannabinoid system in guarding against fear, anxiety and stress. Nat Rev Neurosci 2015, 16, 705–718. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Fukaya, M.; Uchigashima, M.; Miura, E.; Kamiya, H.; Kano, M.; Watanabe, M. Localization of diacylglycerol lipase-alpha around postsynaptic spine suggests close proximity between production site of an endocannabinoid, 2-arachidonoyl-glycerol, and presynaptic cannabinoid CB1 receptor. J Neurosci 2006, 26, 4740–4751. [Google Scholar] [CrossRef]
- Baggelaar, M.P.; Maccarrone, M.; van der Stelt, M. 2-Arachidonoylglycerol: A signaling lipid with manifold actions in the brain. Prog Lipid Res 2018, 71, 1–17. [Google Scholar] [CrossRef]
- Bietar, B.; Tanner, S.; Lehmann, C. Neuroprotection and Beyond: The Central Role of CB1 and CB2 Receptors in Stroke Recovery. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Molina-Holgado, E.; Vela, J.M.; Arevalo-Martin, A.; Almazan, G.; Molina-Holgado, F.; Borrell, J.; Guaza, C. Cannabinoids promote oligodendrocyte progenitor survival: involvement of cannabinoid receptors and phosphatidylinositol-3 kinase/Akt signaling. J Neurosci 2002, 22, 9742–9753. [Google Scholar] [CrossRef]
- Sanchez-Alegria, K.; Flores-Leon, M.; Avila-Munoz, E.; Rodriguez-Corona, N.; Arias, C. PI3K Signaling in Neurons: A Central Node for the Control of Multiple Functions. Int J Mol Sci 2018, 19. [Google Scholar] [CrossRef]
- Li, Z.H.; Cheng, L.; Wen, C.; Ding, L.; You, Q.Y.; Zhang, S.B. Activation of CNR1/PI3K/AKT Pathway by Tanshinone IIA Protects Hippocampal Neurons and Ameliorates Sleep Deprivation-Induced Cognitive Dysfunction in Rats. Front Pharmacol 2022, 13, 823732. [Google Scholar] [CrossRef]
- Galvani, G.; Mottolese, N.; Gennaccaro, L.; Loi, M.; Medici, G.; Tassinari, M.; Fuchs, C.; Ciani, E.; Trazzi, S. Inhibition of microglia overactivation restores neuronal survival in a mouse model of CDKL5 deficiency disorder. J Neuroinflammation 2021, 18, 155. [Google Scholar] [CrossRef]
- Tassinari, M.; Mottolese, N.; Galvani, G.; Ferrara, D.; Gennaccaro, L.; Loi, M.; Medici, G.; Candini, G.; Rimondini, R.; Ciani, E.; et al. Luteolin Treatment Ameliorates Brain Development and Behavioral Performance in a Mouse Model of CDKL5 Deficiency Disorder. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Paraiso-Luna, J.; Aguareles, J.; Martin, R.; Ayo-Martin, A.C.; Simon-Sanchez, S.; Garcia-Rincon, D.; Costas-Insua, C.; Garcia-Taboada, E.; de Salas-Quiroga, A.; Diaz-Alonso, J.; et al. Endocannabinoid signalling in stem cells and cerebral organoids drives differentiation to deep layer projection neurons via CB(1) receptors. Development 2020, 147. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, J.M.; Thomas, B.F.; Budimirovic, D.; Siegel, S.; Tassone, F.; Hagerman, R.; Faulk, C.; O’Quinn, S.; Sebree, T. Role of the endocannabinoid system in fragile X syndrome: potential mechanisms for benefit from cannabidiol treatment. J Neurodev Disord 2023, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Busquets-Garcia, A.; Gomis-Gonzalez, M.; Guegan, T.; Agustin-Pavon, C.; Pastor, A.; Mato, S.; Perez-Samartin, A.; Matute, C.; de la Torre, R.; Dierssen, M.; et al. Targeting the endocannabinoid system in the treatment of fragile X syndrome. Nat Med 2013, 19, 603–607. [Google Scholar] [CrossRef]
- Bach, S.; Shovlin, S.; Moriarty, M.; Bardoni, B.; Tropea, D. Rett Syndrome and Fragile X Syndrome: Different Etiology With Common Molecular Dysfunctions. Front Cell Neurosci 2021, 15, 764761. [Google Scholar] [CrossRef]
- Devinsky, O.; Verducci, C.; Thiele, E.A.; Laux, L.C.; Patel, A.D.; Filloux, F.; Szaflarski, J.P.; Wilfong, A.; Clark, G.D.; Park, Y.D.; et al. Open-label use of highly purified CBD (Epidiolex(R)) in patients with CDKL5 deficiency disorder and Aicardi, Dup15q, and Doose syndromes. Epilepsy Behav 2018, 86, 131–137. [Google Scholar] [CrossRef]
- Vignoli, A.; Prato, G.; Alfei, E.; Bagnasco, I.; Danieli, A.; Celario, M.; Favaro, J.; Matricardi, S.; Operto, F.F.; Orsini, A.; et al. Is highly purified cannabidiol a treatment opportunity for drug-resistant epilepsy in subjects with typical Rett syndrome and CDKL5 deficiency disorder? Epilepsia Open 2025, 10, 1111–1119. [Google Scholar] [CrossRef]
- Silva, G.D.; Del Guerra, F.B.; de Oliveira Lelis, M.; Pinto, L.F. Cannabidiol in the Treatment of Epilepsy: A Focused Review of Evidence and Gaps. Front Neurol 2020, 11, 531939. [Google Scholar] [CrossRef]
- Liu, S.; He, Z.; Li, J. Long-term efficacy and adverse effects of cannabidiol in adjuvant treatment of drug-resistant epilepsy: a systematic review and meta-analysis. Ther Adv Neurol Disord 2023, 16, 17562864231207755. [Google Scholar] [CrossRef]
- Qian, X.; Liu, W.; Chen, Y.; Zhang, J.; Jiang, Y.; Pan, L.; Hu, C. A UPLC-MS/MS method for simultaneous determination of arachidonic acid, stearic acid, and related endocannabinoids in human plasma. Heliyon 2024, 10, e28467. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976, 72, 248–254. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



