3.1. Electrochemical Characterization of the Metal Nanocomposites
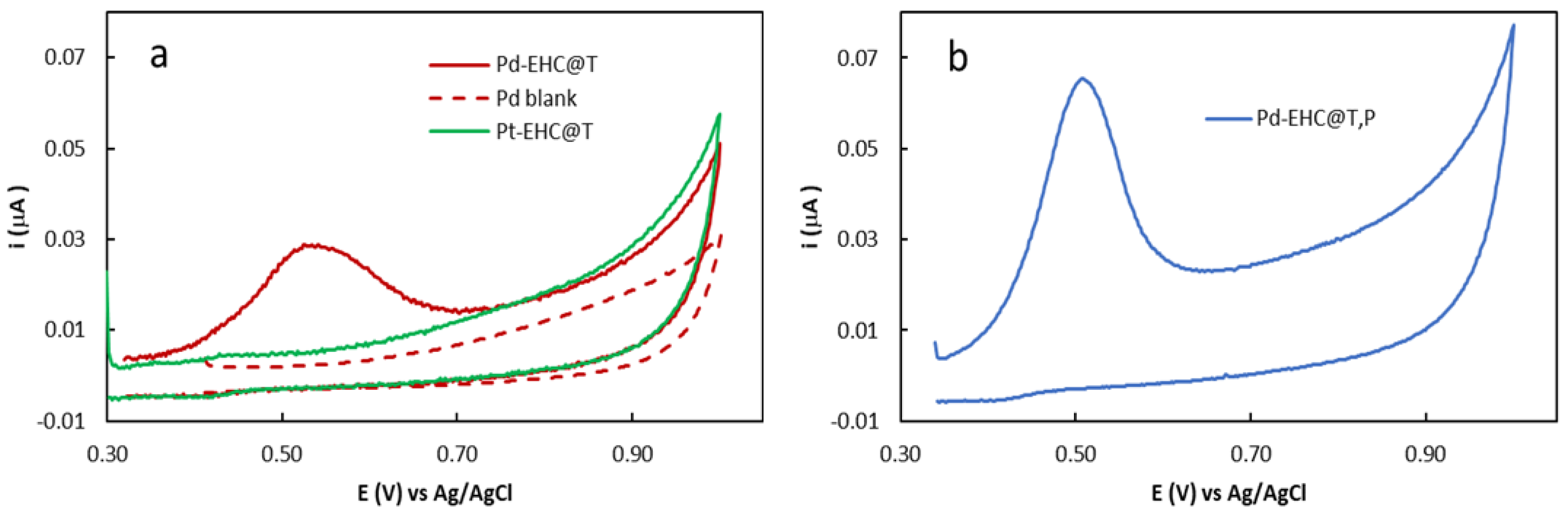
The initial assessment of whether Pd(II) and Pt(IV) species could be successfully reduced by the EHC@T solution was performed using anodic stripping voltammetry (ASV) on the modified glassy carbon electrodes (GCE) with the respective Pd-EHC@T and Pt-EHC@T solutions. As shown in
Figure 1a, a well-defined anodic stripping peak was observed for the Pd-based sample, providing clear evidence of metallic Pd formation via the electron-donating properties of EHC@T. To verify if Pd(II) ions were reduced by the tartaric buffer itself, rather than by the carbon nanomaterial, a control experiment was conducted in which the tartaric buffer was mixed with the palladium salt to form a blank solution, no Pd oxidation peaks were observed. Regarding the electrode modified with Pt-EHC@T, no oxidation signal was detected for platinum, suggesting that the Pt(IV) species from the [PtCl
6]
2- precursor were not reduced to Pt
0 by the EHC@T. This was further confirmed by TEM/EDS analysis, which showed no evidence of platinum nanoparticles or atomic clusters. Even though the standard reduction potential for the [PtCl
62-]/Pt couple (E
0=0.717V) is lower than that of Pd
2+/Pd (E
0= 0.915 V) [
17], making the reduction of Pd species thermodynamically more favourable, this selective reduction may also be attributed to the sluggish multi-electron transfer kinetics required for the Pt(IV) to Pt
0 transformation.
Interestingly, when Pd
0 is formed in the presence of EHC@P (sample Pd-EHC@T,P),
Figure 1b, a significant increase in the anodic peak height is observed compared to Pd–EHC@T. Integration of the ASV anodic peaks allowed for a comparative evaluation of the respective palladium loadings. The results indicate that the loading in the Pd–EHC@T,P nanocomposite is approximately twice that of Pd–EHC@T (91.6 ng cm⁻² and 51.5 ng cm⁻², respectively). This suggests that the inclusion of EHC@P plays a synergistic role in the immobilization and retention of Pd species, possibly through additional anchoring sites or oxygen-containing functional groups that stabilize the metal centers.
Notably, these values are several orders of magnitude lower than those typically categorized as 'ultra-low' in the literature, which often fall within the 1.4-50 μg cm
-2 range [
5,
18]. While achieving nanogram-scale loading is desirable for cost-efficiency, it raises serious concerns regarding long-term stability, a challenge that requires robust anchoring strategies.
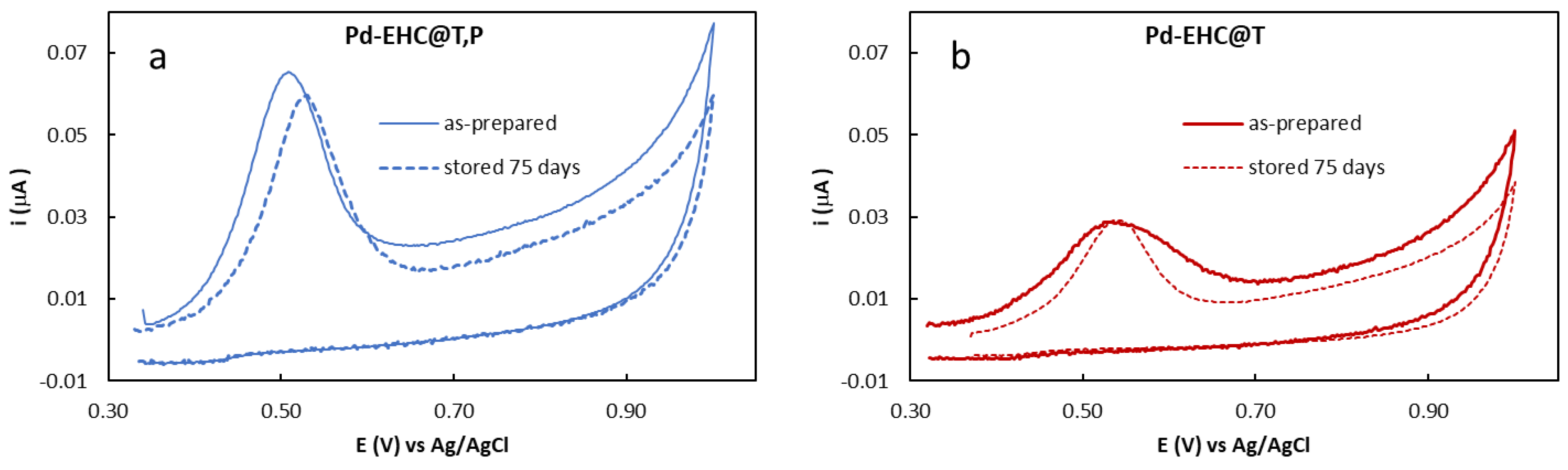
In this context, the stability of these nanocomposites was evaluated using different approaches. In one such approach, the Pd loading of the original stock solution of Pd–EHC@T,P and Pd–EHC@T solutions was monitored regularly over a period of 75 days, being defrosted for periodic electrochemical analysis and subsequently refrozen at −18 °C. For the Pd–EHC@T,P nanocomposite, the anodic stripping charge (measured via anodic oxidation in 1.0 M HCl) remained remarkably stable, changing from 11.8 μC to 10.3 μC (
Figure 2), which represents a minimal variation of 12.7%. This was accompanied by a minor anodic shift of only 15 mV.
In contrast, the Pd–EHC@T material exhibited a more pronounced decrease in the stripping charge, from 6.64 μC to 4.94 μC (a 25.6% reduction), alongside a noticeable decrease in the full width at half maximum (FWHM) of the stripping peak. Rather than a chemical loss of the metal, this decline may be attributed to a gradual loss of palladium accessibility. In the absence of the EHC@P stabilizer, structural changes or adsorption of residual species may hinder the electrolyte’s ability to reach and oxidize the palladium centers. This trend is consistent with our preliminary findings [
15], where storage at room temperature led to a near-total loss (~100%) of the stripping response. These results further highligh the critical role of both the EHC@P support and low-temperature storage in preserving the electrochemical availability of the metal under rigorous storage conditions.
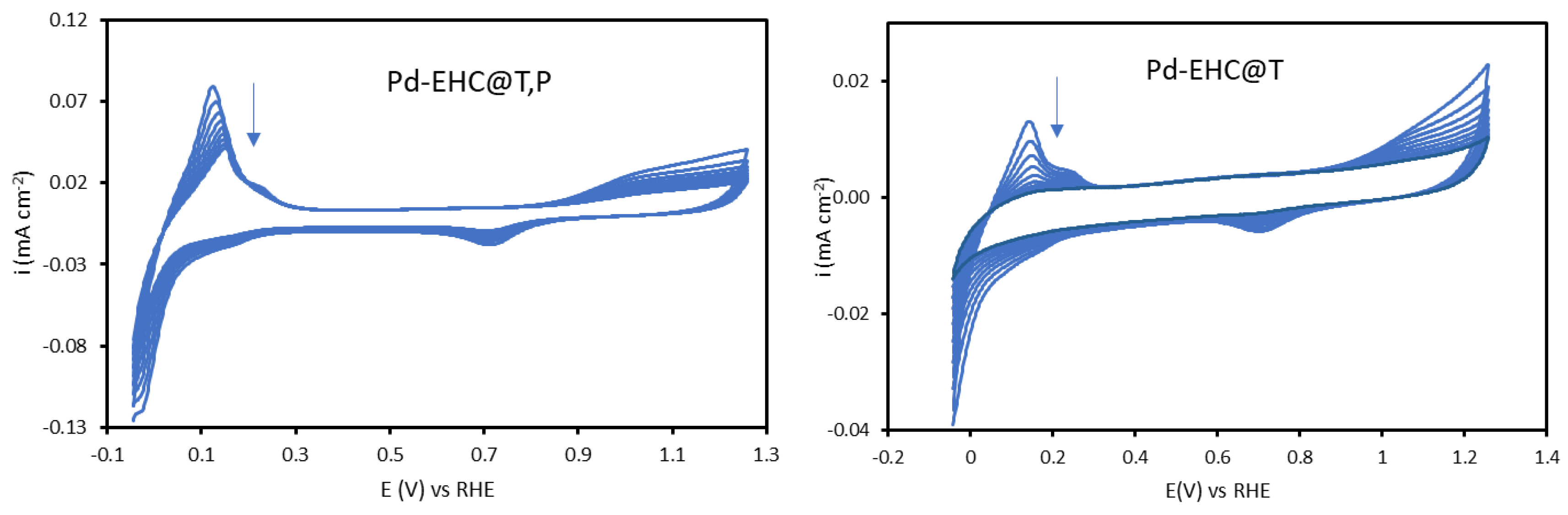
The synergistic effect of EHC@P and EHC@T on the stability of palladium was further demonstrated by recording consecutives cyclic voltammograms in 0.1 M H
2SO
4 using a glassy carbon electrode (GCE) modified with the nanocomposites,
Figure 3.
For the Pd–EHC@T modified electrode, the currents associated with the hydrogen adsorption/desorption peaks and the palladium oxide reduction peak decrease rapidly, becoming undetectable after few scans. In contrast, the Pd–EHC@T,P electrode also showed a decrease in current, but the decay was significantly less pronounced. This current reduction is likely a reflection of palladium dissolution at the highly anodic potentials of the scan. Consequently, these results reinforce the conclusion that EHC@P is essential for effectively anchoring the palladium nanoparticles, thereby mitigating metal dissolution and surface area loss during potential cycling. The enhanced stability of Pd–EHC@T,P suggests a stronger interaction between palladium species and the modified support, which will be further corroborated by morphological analysis.
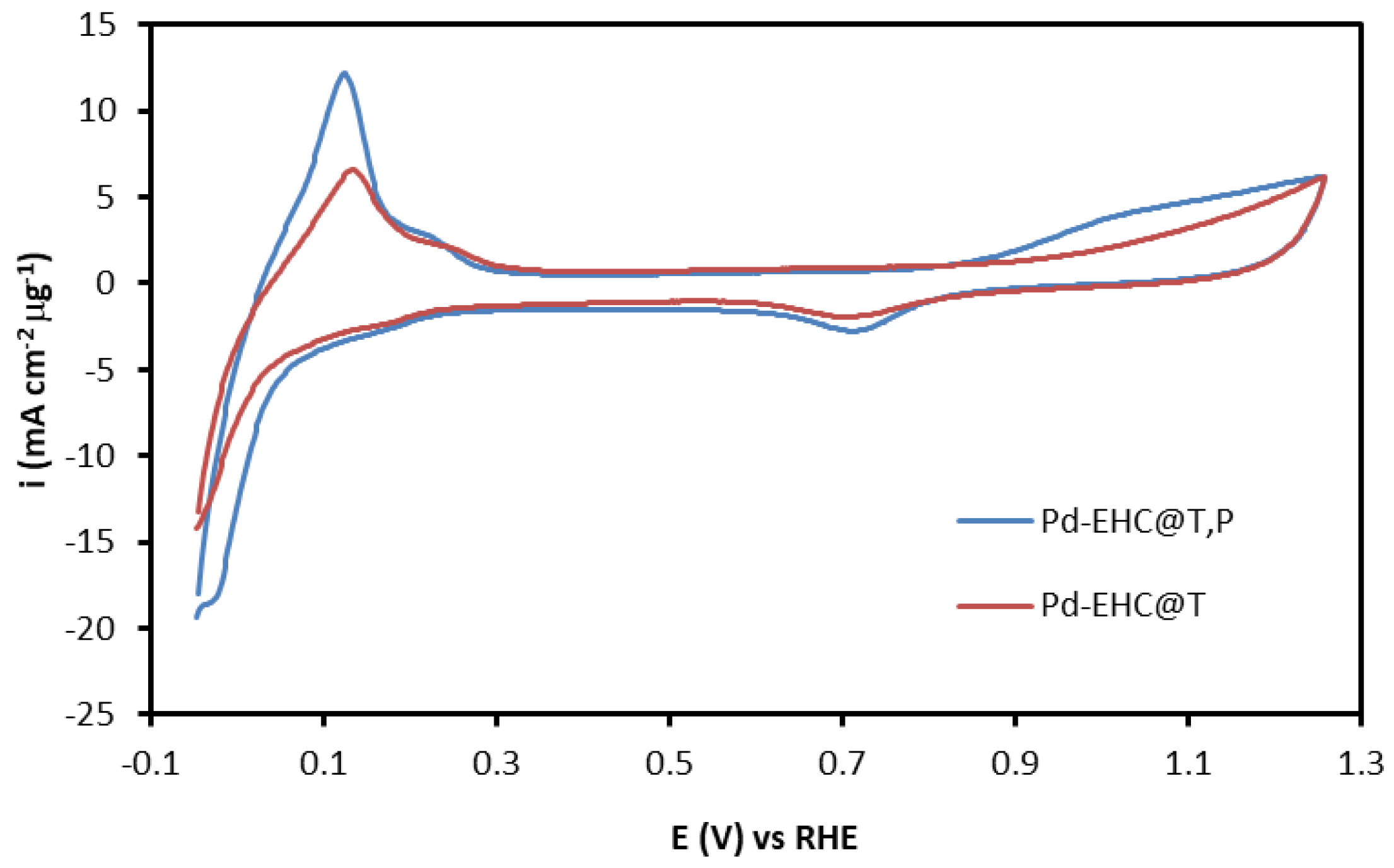
Given the different metal loadings of the immobilized materials, the cyclic voltammograms recorded in 0.1 M H
2SO
4 were normalized by both the electrode geometric area and the catalyst mass (
Figure 4). The mass-normalized voltammograms revealed that the electrochemical activity of Pd–EHC@T,P is superior to that of Pd–EHC@T, as evidenced by the enhanced voltammetric features for the hydrogen adsorption/desorption and palladium oxide formation/reduction regions. This indicates that the synergistic interaction between the two EHC supports not only increases the amount of retained palladium, but also ensures a higher dispersion of the metal, keeping the active sites more accessible for electrocatalytic reactions.
The electrochemical surface area (ECSA) was estimated from the charge associated with the PdO reduction peak. However, a rigorous ECSA determination was hindered by the inherent instability of the palladium surface at high anodic potentials. It was observed that the reduction charge increased with the anodic scan limit up to approximately 1.41 V vs. RHE (1.15 V vs. Ag/AgCl), but began to decrease at more positive potentials, suggesting the onset of Pd dissolution. Consequently, while the experimental charge for a complete PdO monolayer could not be precisely determined, the values obtained at a scan limit of 1.26 V vs. RHE (1.0 V vs. Ag/AgCl) were used for comparative purposes. The resulting apparent ECSA values were 74.2 m
2 g
-1 for Pd–EHC@T,P and 58.5 m
2 g
-1 for the Pd–EHC@T material. This ~27% increase in mass-specific area confirms that the addition of EHC@P effectively minimizes the formation of large clusters and maximizes the exposure of active sites, a crucial factor for achieving high catalytic efficiency at ultralow metal loadings. The calculated ECSA values are consistent with those reported in the literature for commercial Pd/C [
18,
19] and palladium nanoparticles supported on carbon-based materials such as graphene, reduced graphene oxide and carbon nanotubes [
20,
21], despite the significantly higher metal loadings typically employed in those studies.
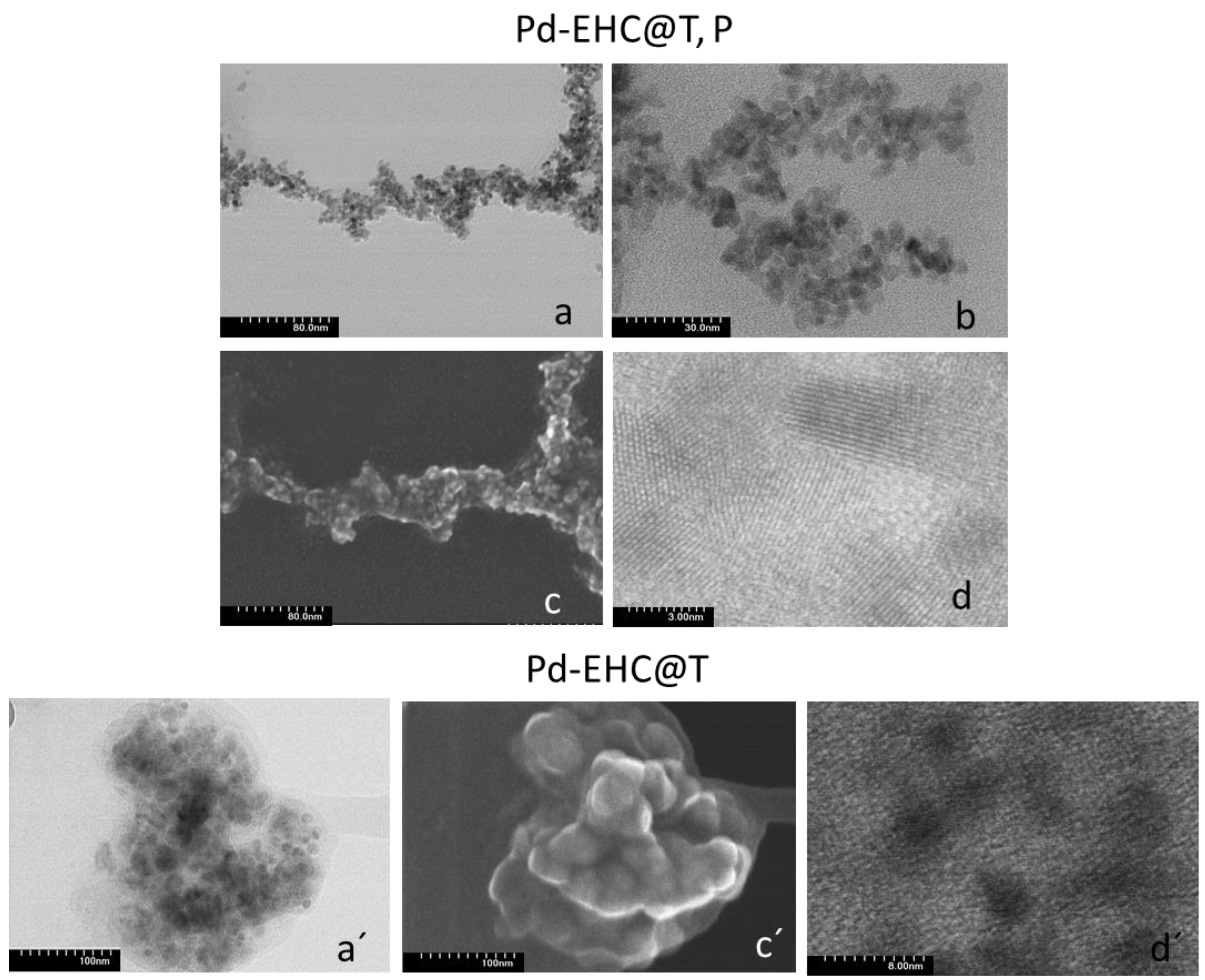
To evaluate the size, morphology, and dispersion of the palladium entities, TEM/HRTEM and STEM analyses were performed using different imaging modes (
Figure 5). The micrographs, obtained via transmitted and secondary electrons, show that in both samples the nanoparticles appear to be intimately embedded in or anchored to the carbon matrix. For the Pd–EHC@T sample, the images reveal the presence of relatively large aggregates of palladium nanoparticles (Pd NPs) that are heterogeneously distributed over the EHC support (
Figure 5a
´and 5c´), with individual particles diameters ranging between 5 and 22 nm. In contrast, the Pd–EHC@T,P nanocomposite exhibits a highly uniform distribution of smaller nanoparticles across the entire carbon framework, with a much narrower particle size distribution (3 to 5 nm). The crystalline nature of these nanoparticles was further confirmed by high-resolution TEM (
Figure 5d and
Figure 5d´), where lattice fringes are clearly visible. These morphological and structural observations are in excellent agreement with the electrochemical data. The superior dispersion and smaller particle size of the Pd–EHC@T,P effectively increase the available surface area, which explains its enhanced electrochemical activity. Furthermore, the smaller and well-anchored nanoparticles in Pd–EHC@T,P are less susceptible to the metal dissolution that led to the more pronounced current decay observed in consecutive voltammograms in Pd–EHC@T sample.
3.2. Electrochemical Activity for the Oxygen Reduction
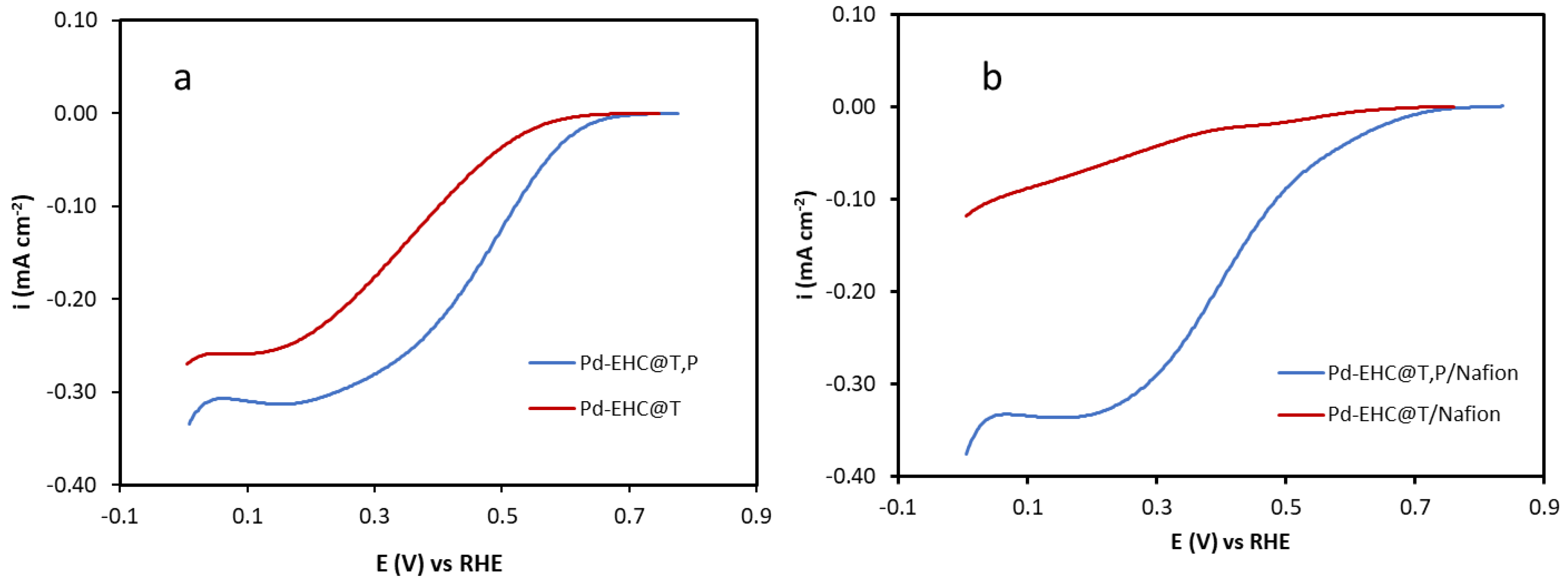
The electrocatalytic performance of the Pd–EHC@T and Pd–EHC@T,P nanocomposites toward the oxygen reduction reaction (ORR) was evaluated in acidic medium. Linear sweep voltammetry (LSV) measurements were carried out from the open-circuit potential (OCP) to −0.15 V vs. RHE in an oxygen-saturated 0.1 M H₂SO₄ solution. As shown in
Figure 6a and summarized in
Table 1, the Pd–EHC@T,P catalyst exhibits improved ORR performance relative to Pd–EHC@T, as reflected by a more positive onset potential (0.669 V vs. RHE versus 0.605 V for Pd–EHC@T) and higher mass and specific activities.
Following the initial ORR characterization (
Figure 6a), the influence of a Nafion overlayer, commonly employed in the fabrication of membrane electrode assemblies (MEAs), was subsequently investigated (
Figure 6b). For the Pd–EHC@T electrode, the presence of Nafion leads to a pronounced decrease in current density in the diffusion-limited region. Notably, this effect is not observed in the kinetic region. Since intrinsic catalytic descriptors are extracted from the kinetically controlled region, these results indicate that the addition of Nafion does not alter the intrinsic ORR activity of the Pd active sites in the Pd–EHC@T system. The reduction in current density observed at high overpotentials can therefore be ascribed to additional mass transport resistance introduced by the Nafion layer, most likely associated with hindered oxygen diffusion through the ionomer film. In contrast, the Pd–EHC@T,P electrode displays nearly identical current densities across the examined potential range. Although a detailed mechanistic interpretation is beyond the scope of this study, this behaviour suggests a more favourable interaction between the Pd–EHC@T,P catalyst layer and the ionomer, effectively mitigating mass transport limitations.
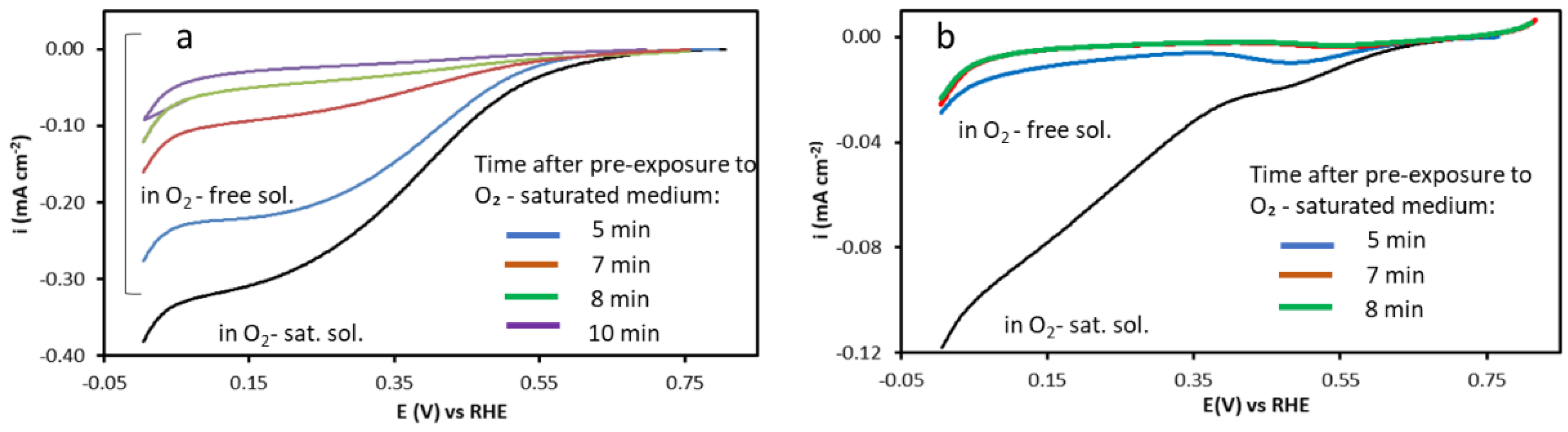
To investigate the oxygen-retention properties of the nanocomposites, the electrochemical response was monitored in an O
2-free 0.1 M H
2SO
4 solution after pre-exposing the electrodes to an O
2-saturated medium. It is important to note that the presence of the Nafion overlayer was found to be essential for observing any sustained current, without the ionomer, the ORR signal dropped immediately upon transfer, even for the modified electrode with Pd–EHC@T,P. For the Pd–EHC@T,P/Nafion system (
Figure 7a), a significant ORR current is observed, even 5 minutes after transfer, maintaining a significant limit current density of -0.22 mA cm⁻². This result provides direct evidence that the EHC@P support possesses an intrinsic ability to store oxygen within its porous structure. In this case, the Nafion film successfully traps the O
2 that was previously adsorbed or absorbed by the phosphorus-templated carbon matrix, allowing it to be gradually reduced at the Pd active sites. In contrast, the Pd–EHC@T/Nafion system (
Figure 7b) fails to show significant oxygen retention. Under identical conditions, the limit current density at 5 minutes is nearly negligible (~ -0.01 mA cm⁻²). Even with the Nafion 'seal' in place, the lack of a current signal confirms that the EHC@T support is unable to store oxygen. These findings demonstrate that the synthesis of the carbon support in phosphate buffer (EHC@P) creates a specialized architecture capable of oxygen sequestration. While the Nafion layer is necessary to reveal this effect by preventing rapid degassing, the experiment proves that only the Pd–EHC@T,P nanocomposite functions as a self-sustaining electrocatalytic system in oxygen-depleted conditions, whereas Pd–EHC@T lacks this storage functionality.