Preprint Disclaimer
This manuscript is a preprint that has not undergone peer review. The content should not be used to guide clinical practice.
The author is a “spetspatient” [
1] (expert patient engaged in coproduction of knowledge and care) and a patient-researcher. The patient/author has provided written informed consent for publication of clinical details and images.
Introduction
Early in the pandemic, thromboembolic complications were widely reported in acutely ill patients [
2,
3]. Mechanistic studies proposed platelet activation by SARS-CoV-2 and amyloidogenic spike protein effects yielding fibrin(ogen) resistant to fibrinolysis [
4,
5,
6,
7]. To facilitate communication, the term “microclots” or fibrinaloid microclot complexes (FMCs) has been used to denote insoluble fibrin–amyloid aggregates in plasma (approximately 8–200 µm). Subsequent work reported hyperactivated platelets, increased viscosity, thrombotic endotheliitis with debris, extracellular viral vesicles, and neutrophil extracellular traps (NETs) in PCC, and post-translational modifications within fibrinaloid microclot complexes, potentially inducing persistent inflammation and capillary obstruction [
8,
9,
10].
These phenomena may underpin microvascular dysfunction and autonomic phenotypes such as POTS and plausibly contribute to post-exertional malaise via perfusion impairment, tissue hypoxia, lactate accumulation, and disturbances of ATP-dependent processes in neural and muscle cells [
11,
12,
13], (
Figure 1).
Methods
Design and participants: This is a case report. The author/patient had persistent symptoms following SARS-CoV-2 infection consistent with PCC. Clinical evaluations were conducted in Sweden, with additional biomarker assessments performed through a biorepository program in Stellenbosch, South Africa.
Microclot assessment: Plasma fluorescence microscopy was used to quantify fibrinaloid microclot burden on a 0–16 semi-quantitative scale, with reference values <8.
Intervention: A repurposed triple antithrombotic regimen was initiated targeting coagulation cascade and platelet aggregation: acetylsalicylic acid 75 mg once daily, clopidogrel 75 mg once daily, apixaban 5 mg twice daily, plus pantoprazole 40 mg once daily for gastroprotection was initiated by the author/patient . Recommended duration in the observational protocol was 2–6 months with monthly clinical follow-up [
14].
Outcomes and follow-up: Symptom reports were collected longitudinally. Standardized walking tests and chair-stand tests were performed serially; wearable data were used to estimate exertional intensity. Repeat microclot microscopy was performed.
Results
A 52-year-old previously healthy woman (physician) developed exertional chest pain and reduced exercise capacity after COVID-19 (2020). Baseline ECG, troponin T, and CT pulmonary angiography were unremarkable. Recurrent bradycardia occurred. Echocardiography, cardiac MRI, and Holter monitoring were normal. A six-minute walk test (6MWT) was pathological with marked tachycardia and angina.
High-resolution CT with inspiratory/expiratory imaging showed peripheral mosaic attenuation (air trapping). Adenosine stress cardiac MRI demonstrated, and invasive coronary angiography confirmed, microvascular dysfunction: coronary flow reserve 1.1 (reference >2.0) and index of microcirculatory resistance 118 (reference <25), with normal epicardial coronaries.
Anti-anginal therapy (beta-blocker, long-acting nitrate) yielded partial benefit; physical capacity and work ability remained limited (~50%).
In 2025, plasma fluorescence microscopy demonstrated markedly elevated microclot burden: 15/16 (reference <8).
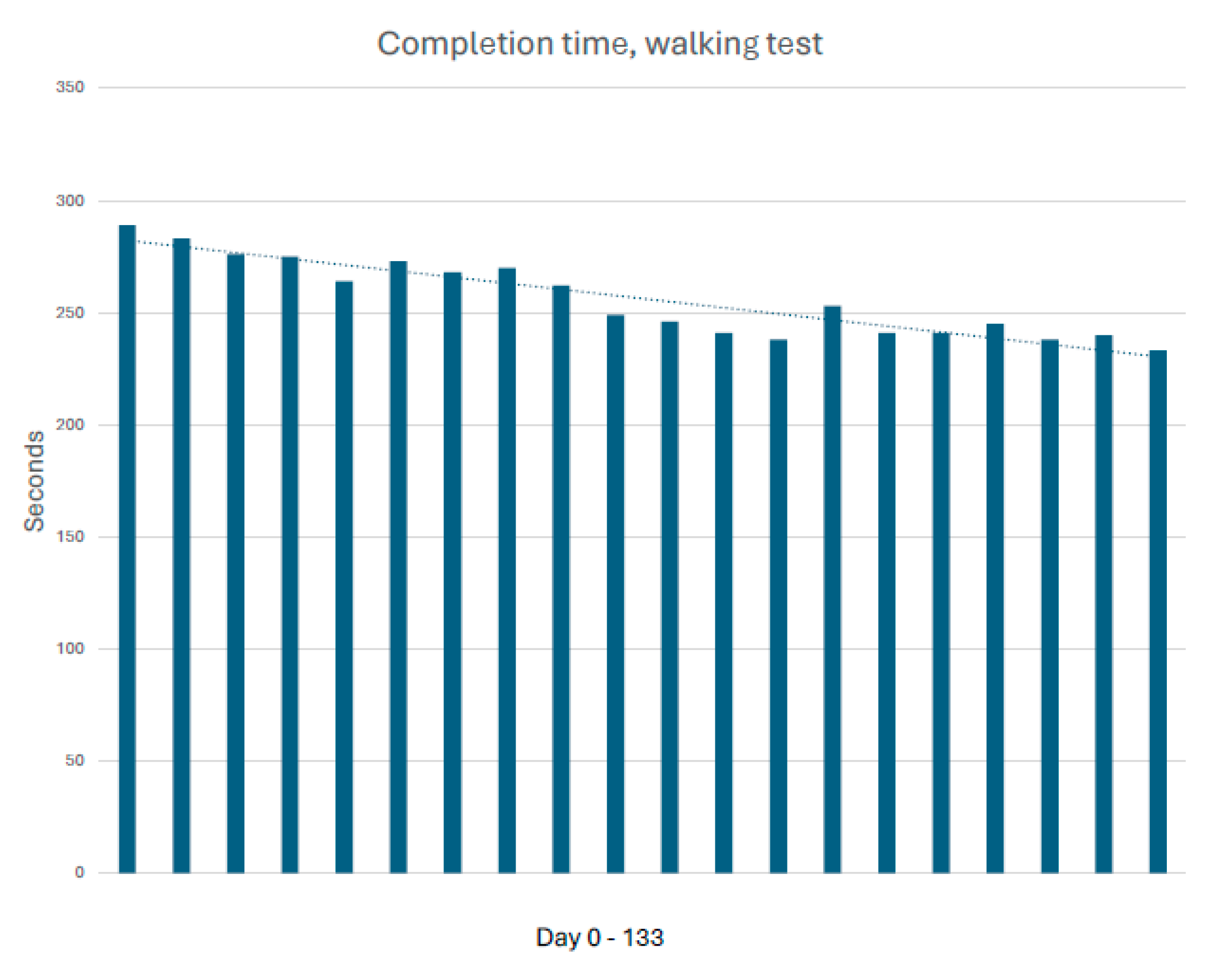
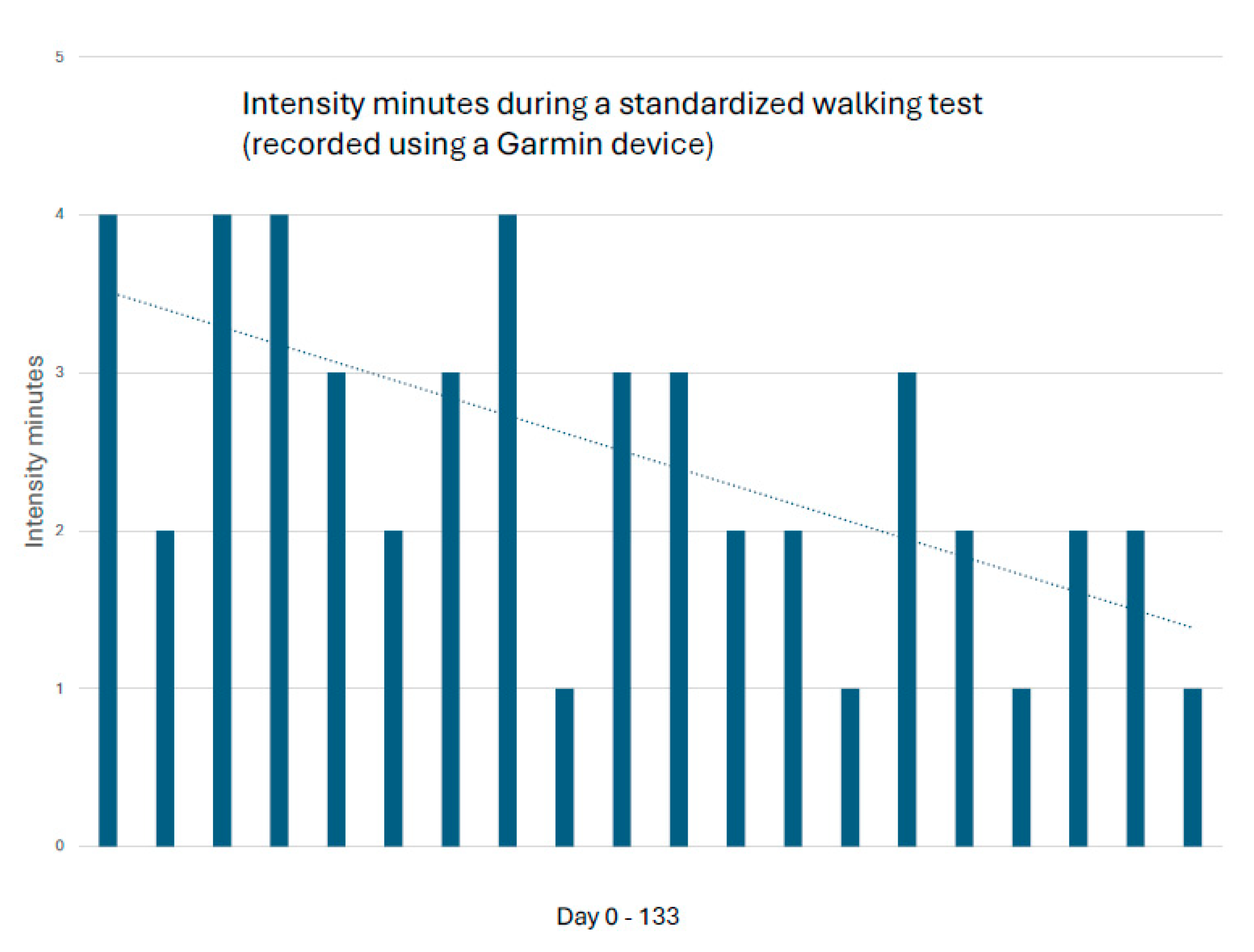
Following initiation of triple antithrombotic therapy, standardized 0.4-km walking tests showed improved completion time (from 289 s to 233 s) and reduced exertional intensity (intensity minutes decreased from 4 to 1) (
Figure 2 and
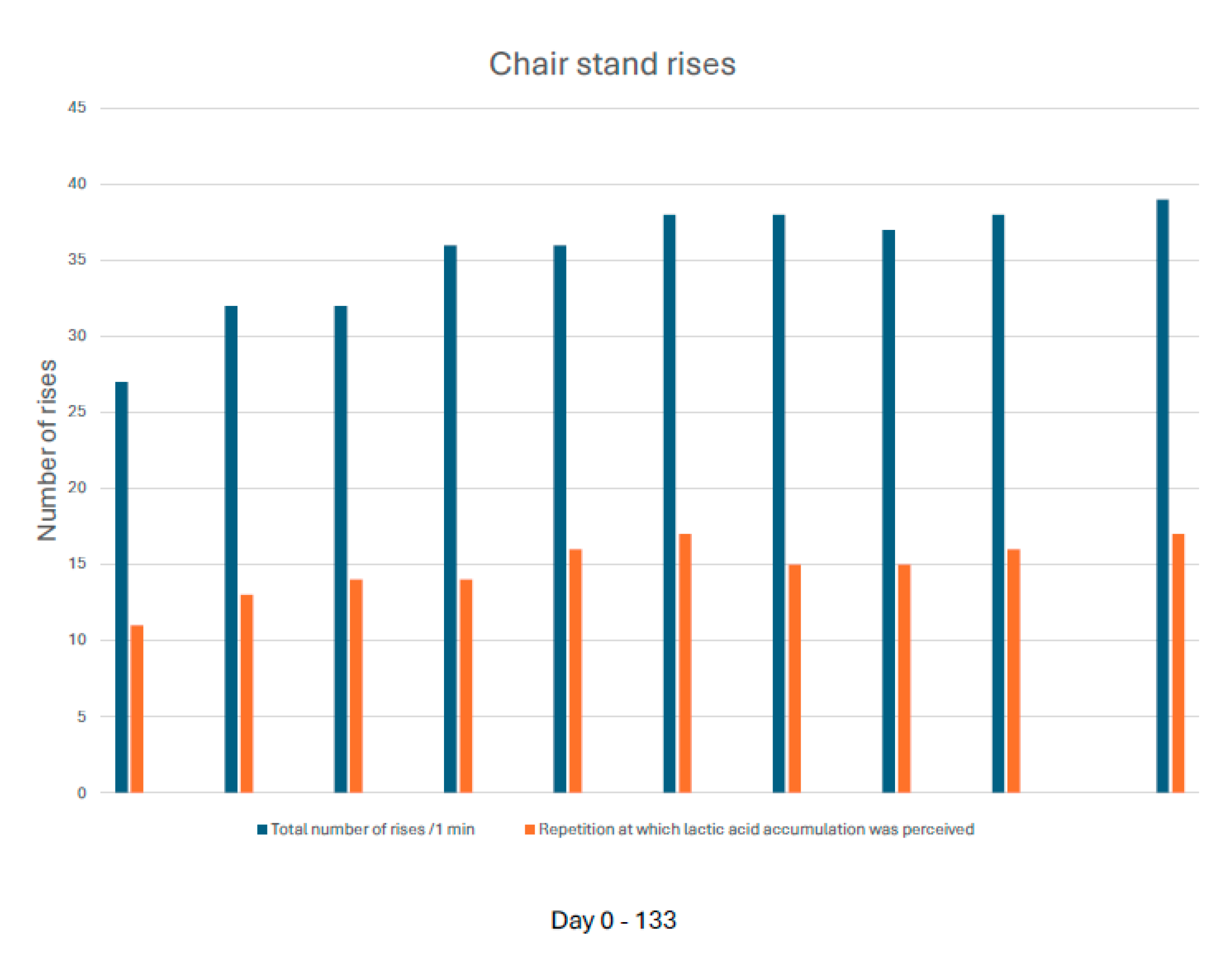
Figure 3). Chair-stand capacity improved (total rises from 27 to 39; onset of perceived lactate accumulation from repetition 11 to 17) [
Figure 4]. Within months, longer walking distances were tolerated without angina or post-exertional malaise; anti-anginal medication requirements decreased. At 5.5 months, microclot burden had decreased from 15/16 to 3/16; FUNCAP-27 had improved from 138 to 152 and PGIC was 6/7.
Discussion
This case align with a microvascular explanatory model for specific PCC phenotypes in which microclots and endothelial or microvascular dysfunction may contribute to heterogeneous systemic symptoms, including angina, exercise intolerance, POTS [
15], and post-exertional malaise. Repurposed triple antithrombotic therapy addresses dysregulated clotting physiology by preventing formation of new microclots and potentially facilitating fibrinolytic breakdown of those already formed, which may promote endothelial repair and symptomatic improvement (
Figure 1).
Alternative explanations—including spontaneous recovery and placebo effects—cannot be excluded. The temporal sequence of early clinical improvement followed by biomarker change suggests (without proving) a therapeutic signal, warranting double-blind randomized controlled trials and standardized, blinded microclot analytics to assess causality and clinical utility. Access to specialized diagnostics remains challenging because microclots are not detectable by conventional thrombosis tests in many countries.
Conclusions
PCC may involve microvascular pathology with microclots and endothelial dysregulation. In this case report, repurposed triple antithrombotic therapy was associated with symptom improvement and reduction in microclot burden. Robust randomized trials and standardized biomarker platforms are needed to define efficacy, safety, patient selection, and care pathways.
Ethics and Safety
This case series does not constitute human subjects research and is therefore exempt from formal ethical review. Written informed consent for publication of clinical details and images was obtained from the patient/author.
The regimen (“triple therapy”) comprises dual antiplatelet therapy plus a direct oral anticoagulant with known bleeding risks; use should be specialist-led with individualized risk–benefit assessment, gastroprotection, and monitoring. The protocol is off-label/repurposed and not standard of care; randomized evidence is pending.
In this case, prescription and clinical monitoring was made by the author/patient, weighing benefits versus watchful waiting in the presence of confirmed microclot pathology.
The study Biorepository and database program for blood coagulation, where the tests were performed, has obtained the required ethical approvals.
Funding
No specific funding was received for this work.
Data Availability
All data supporting the findings are reported within the article; additional de‑identified data may be available upon reasonable request.
Acknowledgments
I thank the team at Stellenbosch University Hospital for methodological and clinical insights. I also acknowledge my cardiologist at Karolinska University Hospital, Stockholm, for recommending that the case series be written in English, and my general practitioner in Enebyberg, Stockholm, for additional clinical advice. Finally, I acknowledge several physician-researchers with whom I have a professional relationship for their constructive feedback on the manuscript. All individuals acknowledged have provided written permission and confirmed the nature of their contribution.
Competing Interests
The author is the same person as the patient. She serves as a board member of the Swedish Covid Association (a patient organization for PCC).
Use of AI
M365 Copilot, an AI language model, was used to support linguistic clarity by providing alternative phrasings, improving grammar, and assisting with structural refinements of the manuscript text. No clinical reasoning, diagnostic judgment, data generation, or scientific interpretation was performed by the AI. The author reviewed, verified, and approved all AI‑assisted text.
References
- Riggare, S; Blease, C. Spetspatients are helping to reshape healthcare. BMJ 2025, 390, r1634. [Google Scholar] [CrossRef] [PubMed]
- Grobler, C; Maphumulo, SC; Grobbelaar, LM; et al. Covid-19: The Rollercoaster of Fibrin(ogen), D-Dimer, Von Willebrand Factor, P-Selectin and Their Interactions with Endothelial Cells, Platelets and Erythrocytes. Int J Mol Sci. 2020, 21(14), 5168. [Google Scholar] [CrossRef]
- Katsoularis, I; Fonseca-Rodríguez, O; Farrington, P; et al. Risks of deep vein thrombosis, pulmonary embolism, and bleeding after COVID-19: nationwide self-controlled case series and matched cohort study. BMJ 2022, 377, e069590. [Google Scholar] [CrossRef]
- Pretorius, E; Vlok, M; Venter, C; et al. Persistent clotting protein pathology in Long COVID/PASC is accompanied by increased levels of antiplasmin. Cardiovasc Diabetol. 2021, 20(1), 172. [Google Scholar] [CrossRef] [PubMed]
- Grobbelaar, LM; Venter, C; Vlok, M; et al. SARS-CoV-2 spike protein S1 induces fibrin(ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19. Biosci Rep. 2021, 41(8), BSR20210611. [Google Scholar] [CrossRef] [PubMed]
- Kell, DB; Laubscher, GJ; Pretorius, E. A central role for amyloid fibrin microclots in Long COVID/PASC: origins and therapeutic implications. Biochem J. 2022, 479(4), 537–559. [Google Scholar] [CrossRef] [PubMed]
- Westman, H; Hammarström, P; Nyström, S. SARS-CoV-2 spike protein amyloid fibrils impair fibrin formation and fibrinolysis. Biochemistry 2025. [Google Scholar] [CrossRef] [PubMed]
- Kell, DB; Pretorius, E. The potential role of ischaemia-reperfusion injury in chronic, relapsing diseases such as rheumatoid arthritis, Long COVID, and ME/CFS: evidence, mechanisms, and therapeutic implications. Biochem J. 2022, 479(16), 1653–1708. [Google Scholar] [CrossRef] [PubMed]
- Thierry, AR; Usher, T; Sanchez, C; et al. Circulating microclots are structurally associated with neutrophil extracellular traps and are elevated in Long COVID patients. J Med Virol. 2025, 97(10), e70613. [Google Scholar] [CrossRef] [PubMed]
- Booyens, RN; Vlok, M; Bester, C; et al. Post-translational modifications within fibrinaloid microclot complexes distinguish Pre-COVID-19 POTS, Long COVID, and Long COVID-POTS and reveal disease-specific molecular pathways. Pre-print 2025. [Google Scholar] [CrossRef]
- Mahdi, A; Zhao, A; Fredengren, E; et al. Dysregulations in hemostasis, metabolism, immune response, and angiogenesis in post-acute COVID-19 syndrome with and without POTS: a multi-omic profiling study. Sci Rep. 2023, 13, 20230. [Google Scholar] [CrossRef] [PubMed]
- Kell, DB; Khan, MA; Pretorius, E. Fibrinaloid microclots in Long COVID: assessing the actual evidence properly. Res Pract Thromb Haemost. 2024, 8(7), 102566. [Google Scholar] [CrossRef] [PubMed]
- Scheibenbogen, C; Wirth, KJ. Key pathophysiological role of skeletal muscle disturbance in Post-COVID and ME/CFS: accumulated evidence. J Cachexia Sarcopenia Muscle 2025, 16(1), e13669. [Google Scholar] [CrossRef] [PubMed]
- Laubscher, GJ; Khan, MA; Venter, C; et al. Treatment of Long COVID symptoms with triple anticoagulant therapy; Pre-print, 2023. [Google Scholar] [CrossRef]
- Björnson, M; Wijnbladh, K; Törnberg, A; et al. Prevalence and clinical impact of POTS in highly symptomatic Long COVID. Circ Arrhythm Electrophysiol. 2025, 18(10), e013629. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |