Submitted:
11 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
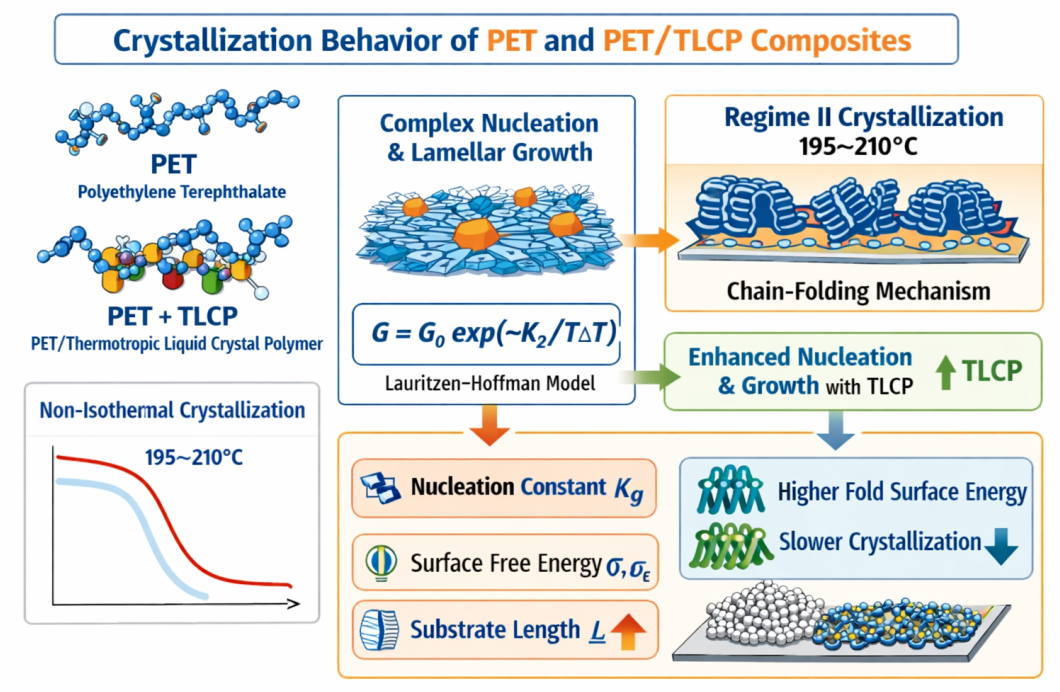
1. Introduction
2. Materials and Methods
Materials
Composite Preparation
Differential Scanning Calorimetry Measurements:
Results & Discussion
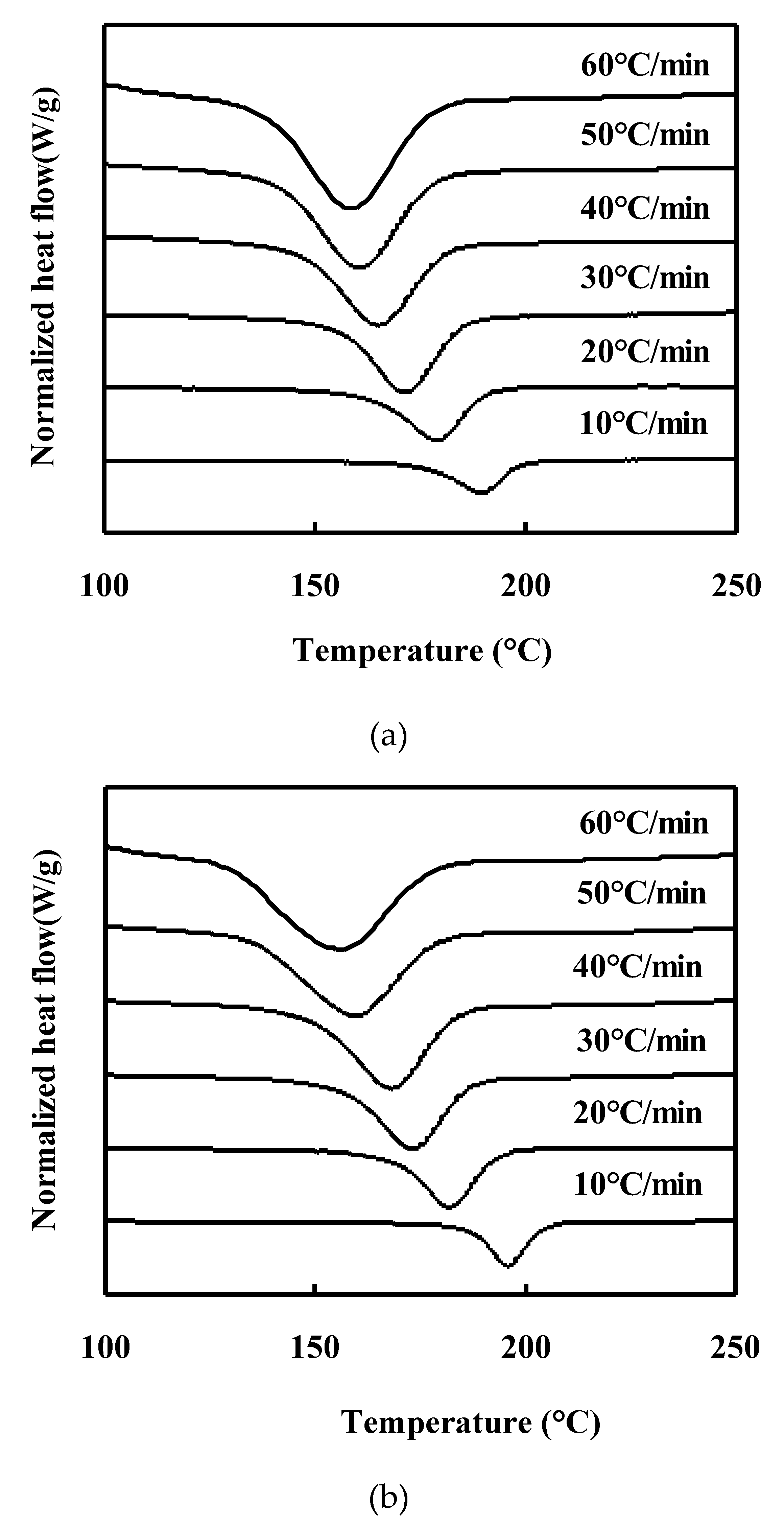
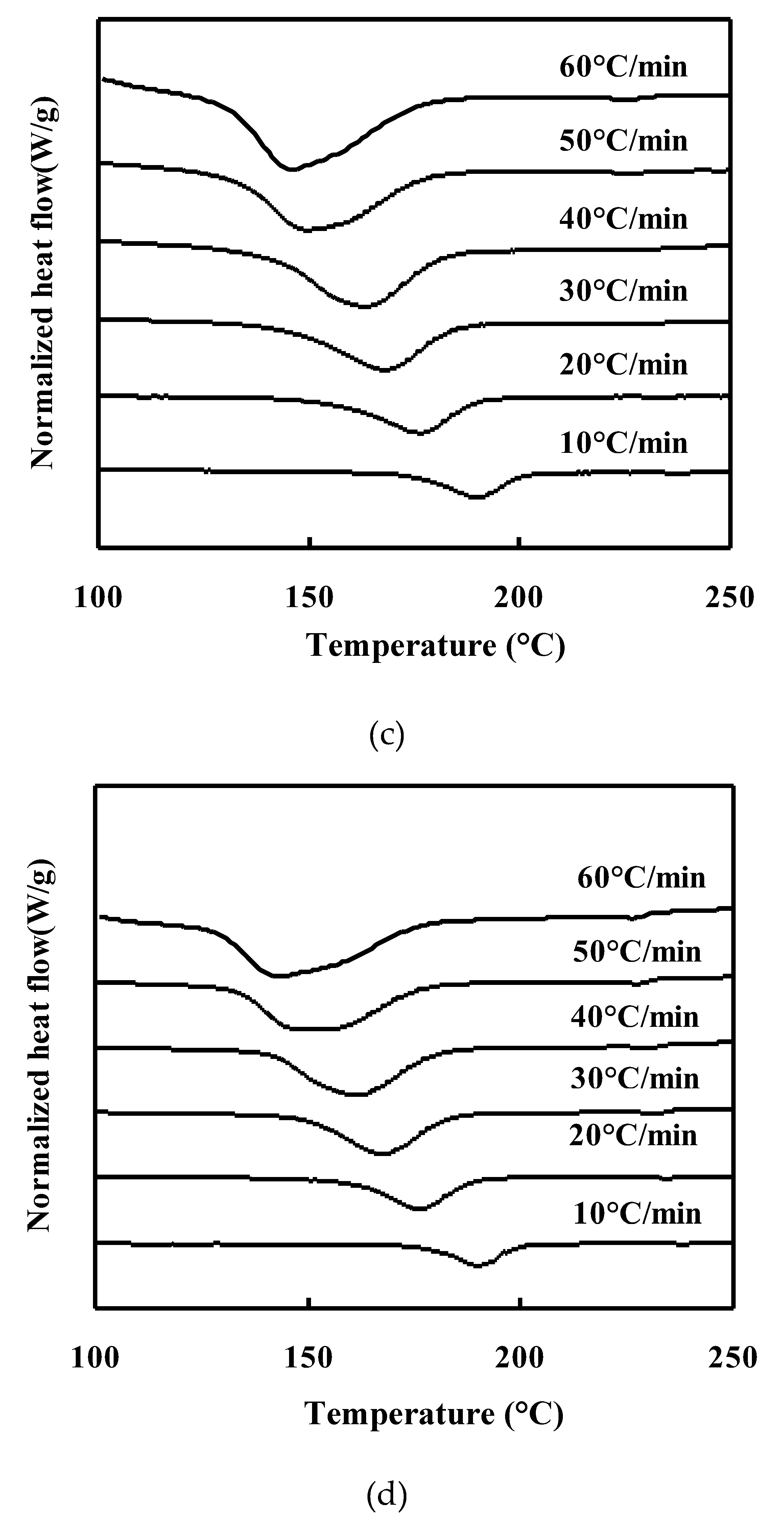
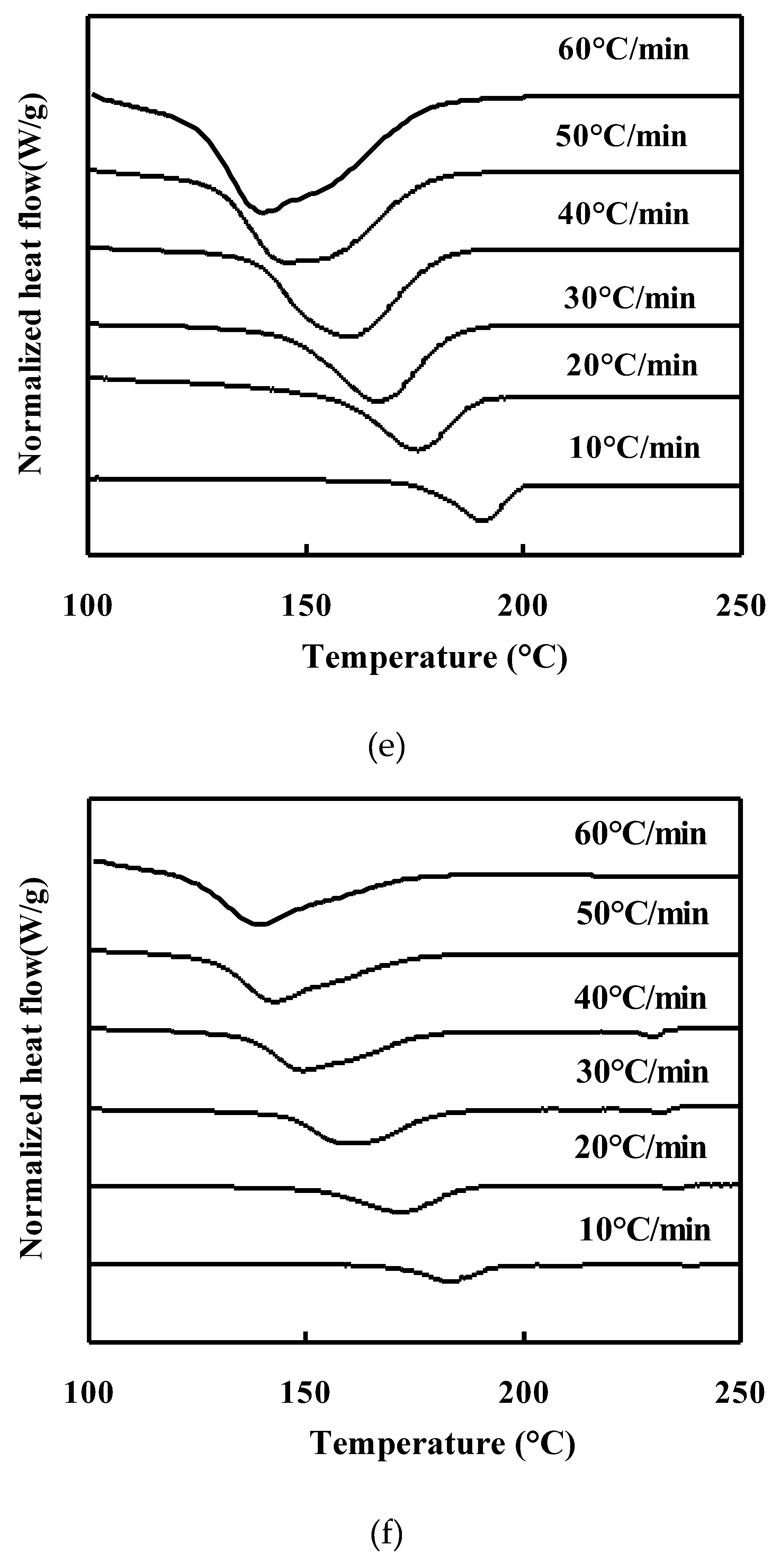
Non-isothermal Crystallization behavior of neat PET and PET/TLCP Composites:
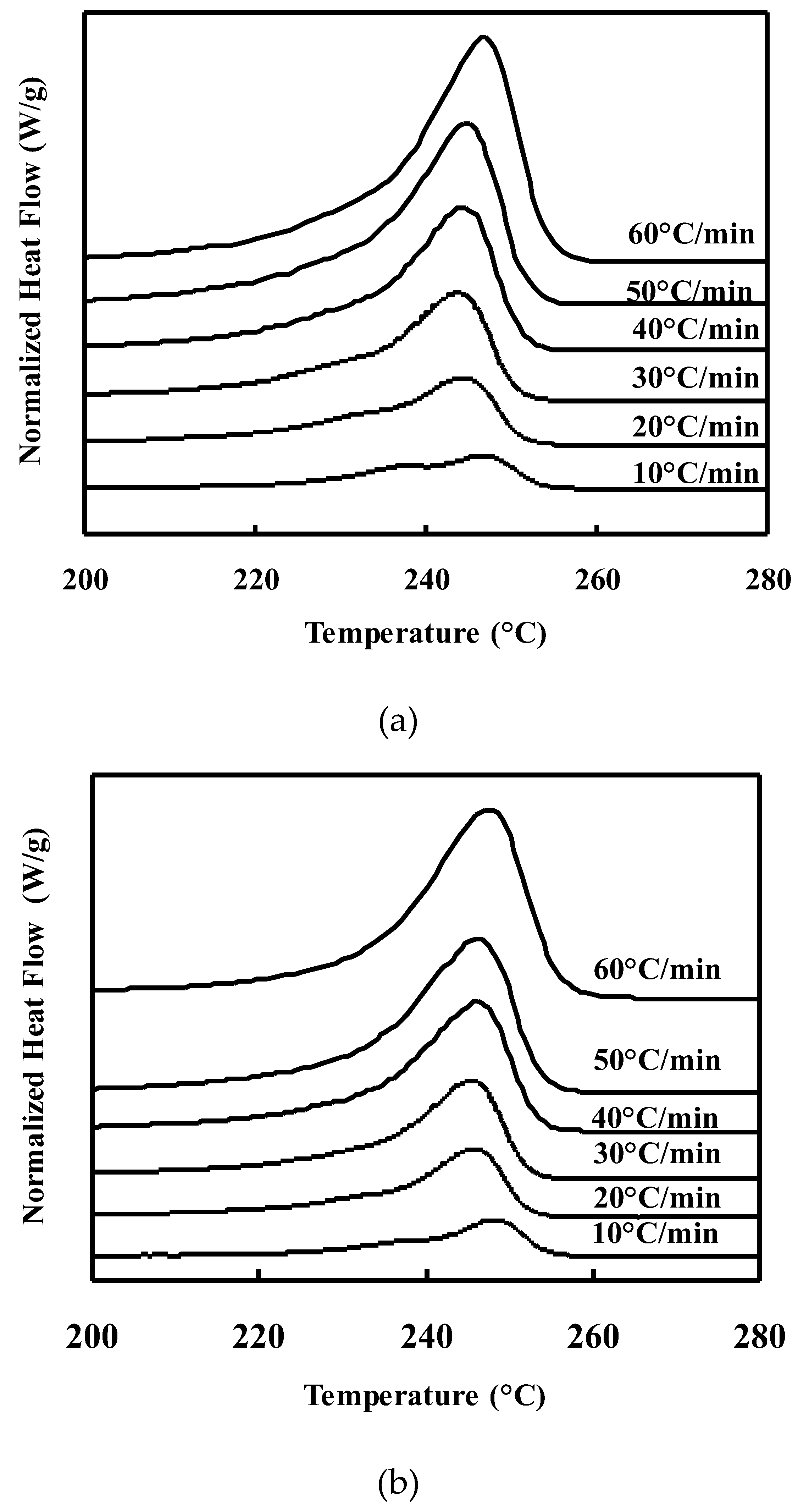
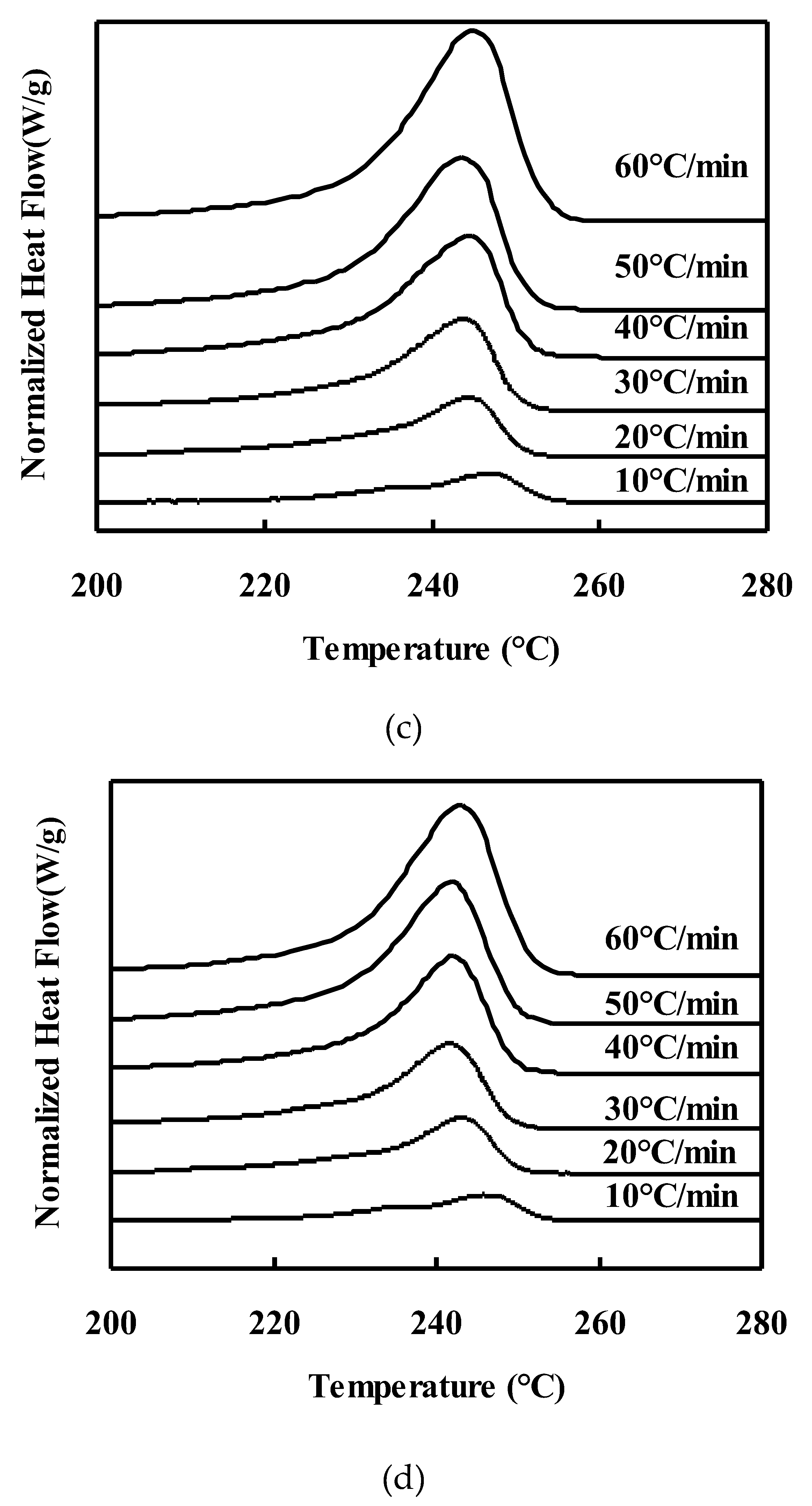
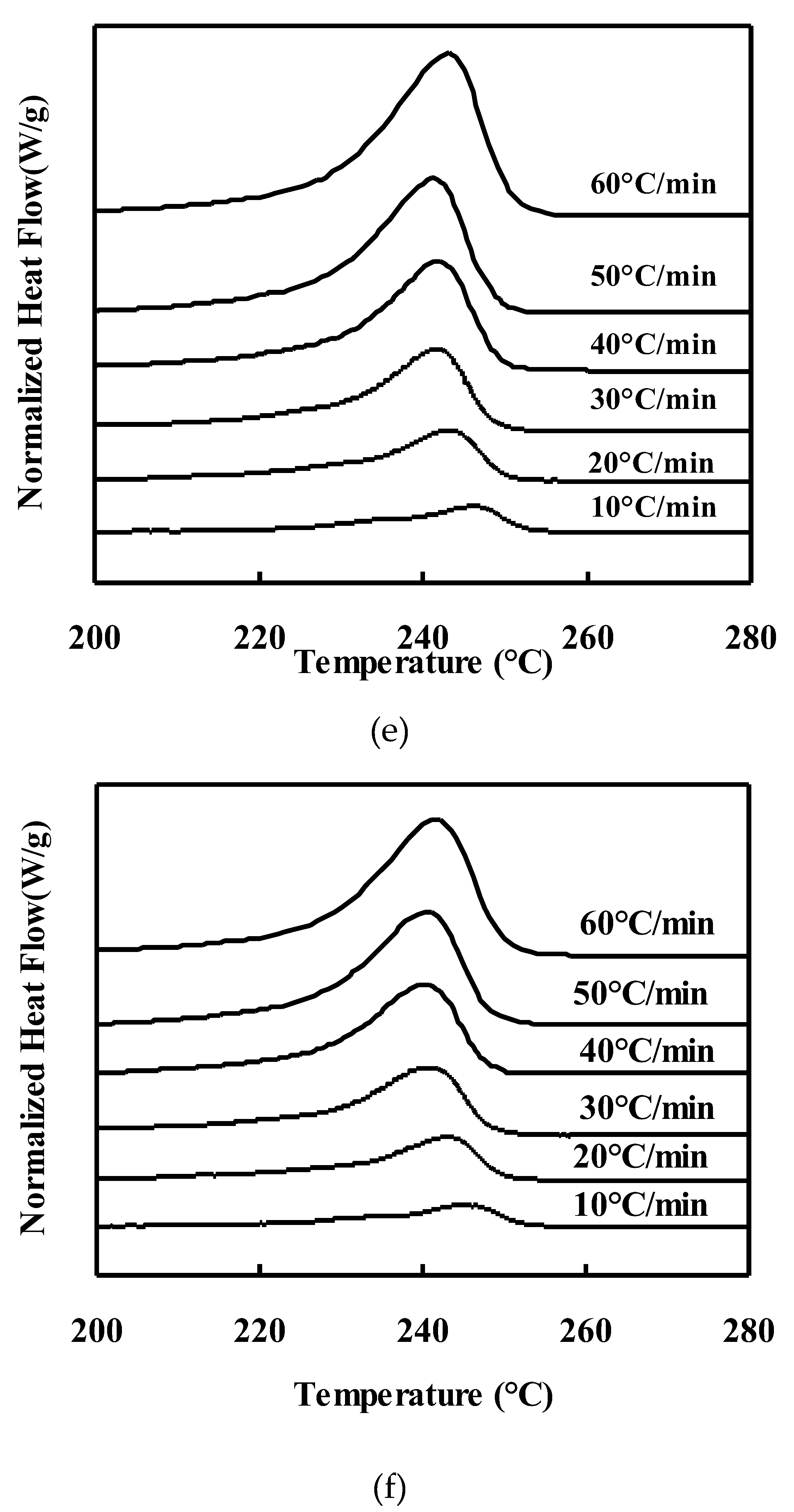
Melting behavior of neat PET and PET/TLCP composites after nonisothermal crystallization :
| Rate of heating (°C/min) | PET/TLCP | |||||||||||
| 100/00 | 90/10 | 80/20 | 70/30 | 60/40 | 50/50 | |||||||
| I | II | I | II | I | II | I | II | I | II | I | II | |
| 2 | 244 | 254 | 243 | 251 | 243 | 253 | 238 | 250 | 238 | 250 | 238 | 250 |
| 5 | 239 | 251 | 238 | 250 | 237 | 249 | 234 | 248 | 236 | 248 | 232 | 246 |
| 10 | 237 | 248 | 237 | 249 | 235 | 248 | 234 | 247 | 235 | 247 | 234 | 246 |
| 20 | 232 | 245 | 232 | 246 | - | 245 | - | 243 | - | 243 | - | 243 |
| 30 | 229 | 244 | - | 246 | - | 244 | - | 242 | - | 242 | - | 241 |
| 40 | - | 246 | - | 246 | - | 245 | - | 242 | - | 242 | - | 240 |
| 50 | - | 245 | - | 246 | - | 244 | - | 242 | - | 241 | - | 240 |
| 60 | - | 247 | - | 247 | - | 245 | - | 243 | - | 243 | - | 242 |
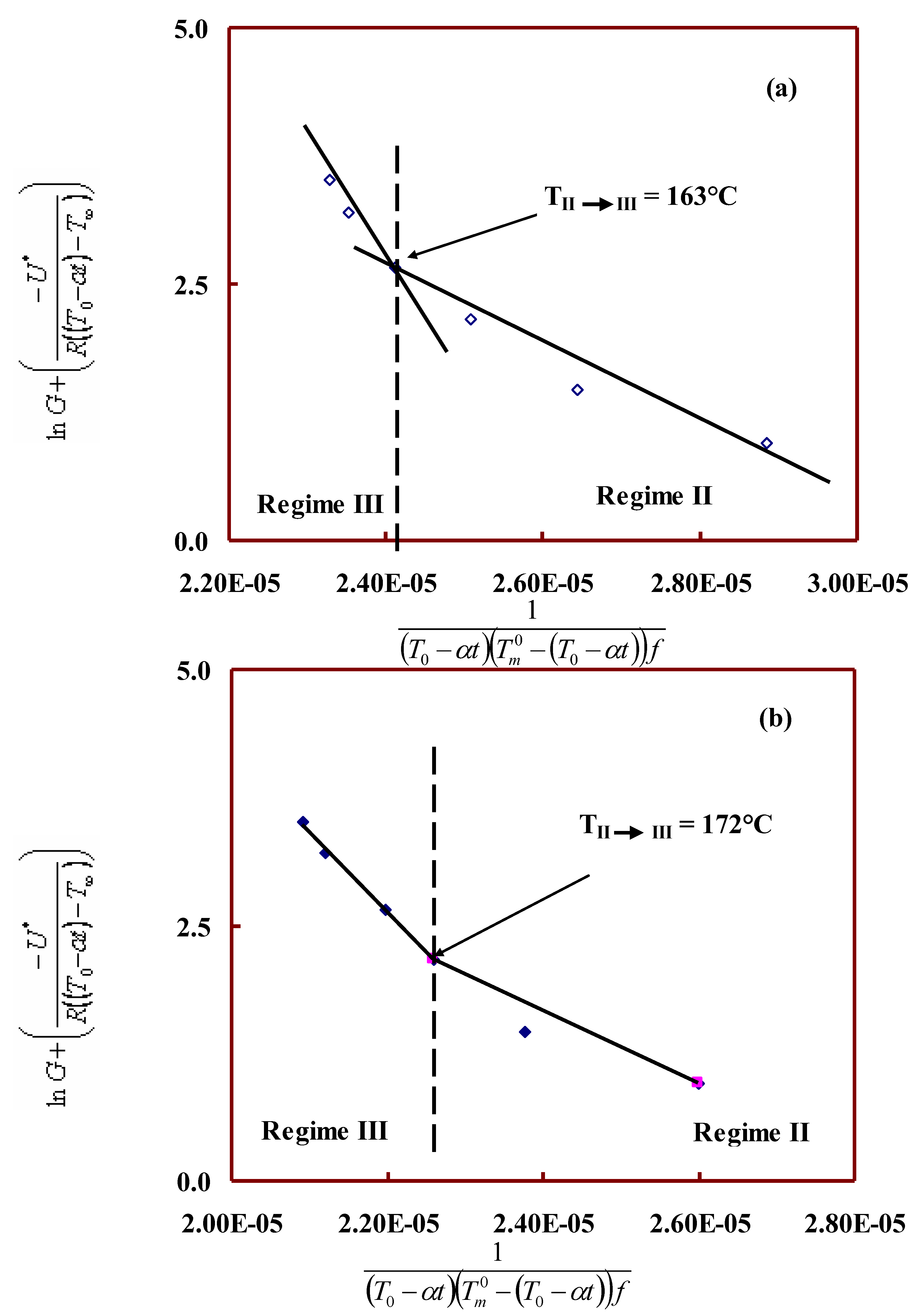
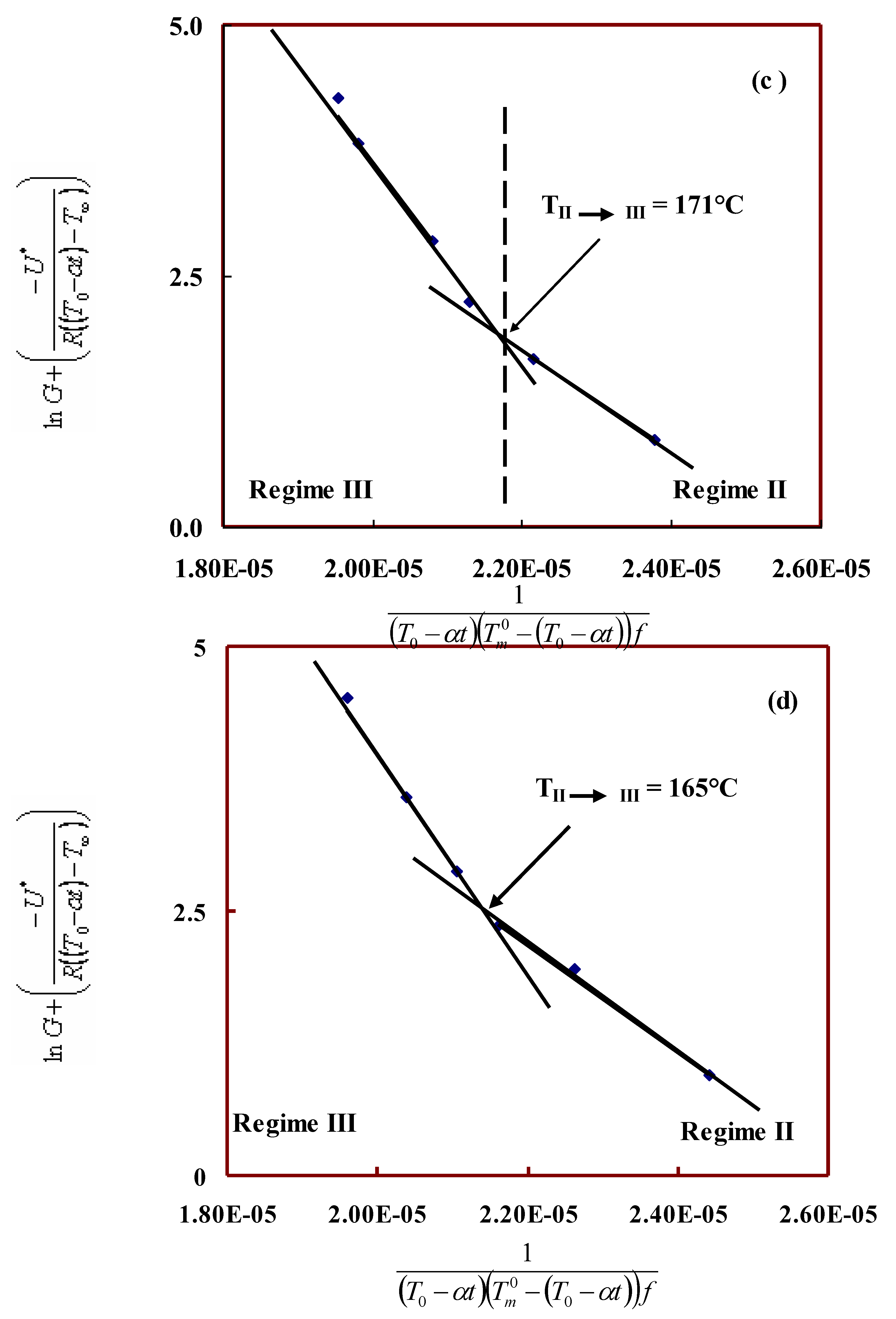
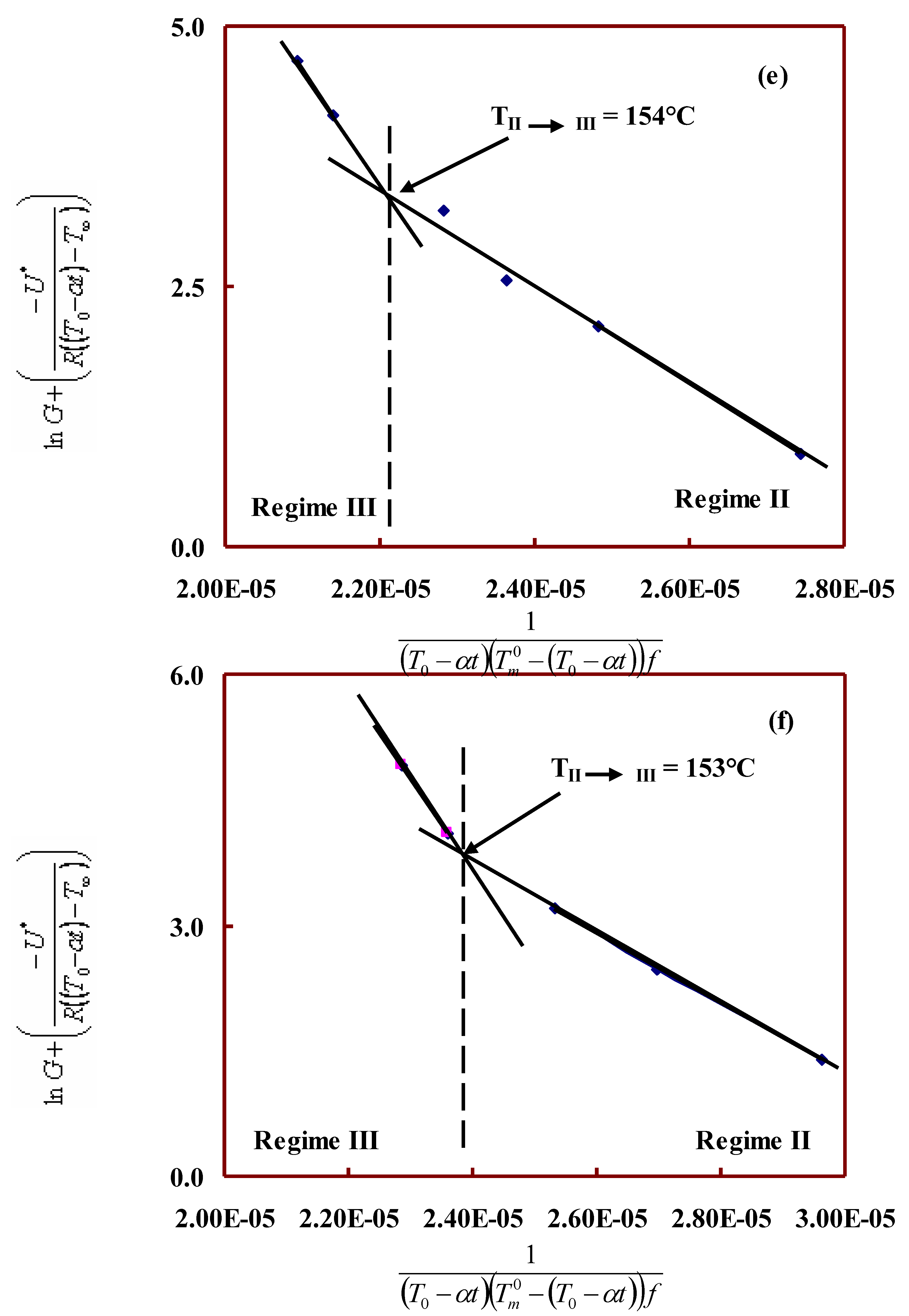
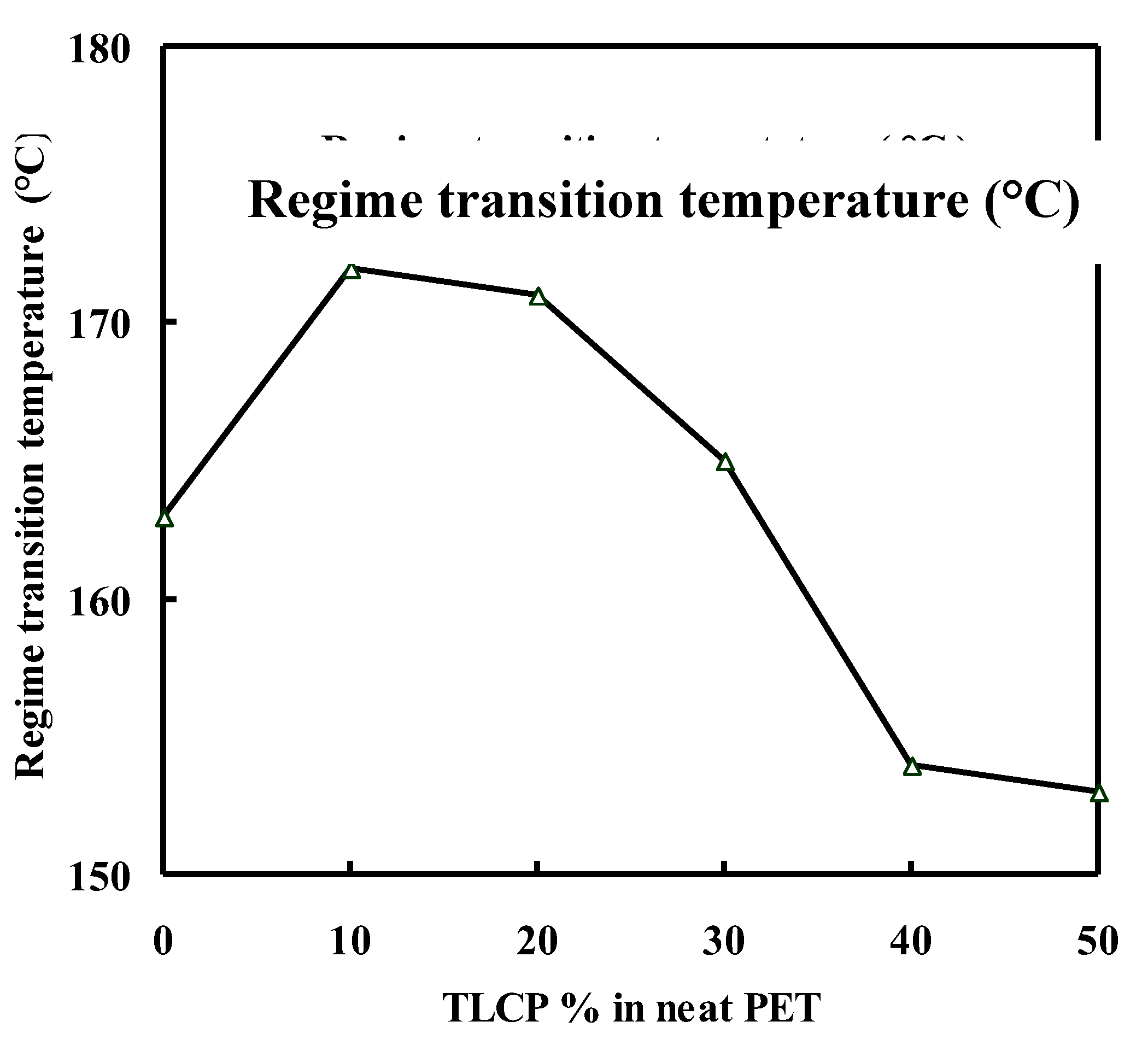
Interfacial free energy and regime kinetics:
Conclusion
Conflicts of Interest
References
- Ozawa T. Polymer 12:150, 1971.
- Wu, J.; Schultz, J. M.; Samon, J. M.; Pangelinan, A. B.; Chuah, H. H. Polymer 42, 7141, 2001.
- Grande, J. A. Mod Plast 12, 97, 1997.
- Jabarin SA. J Appl Polym Sci 34:97–102,1987.
- Fann DM, Huang SK, Lee JY. Polym Engng Sci 38:265–273., 1998.
- Incarnato L, Motta O, Acierno D. Polymer 39:5085–5091, 1998.
- C. C. Lin, Polym. Eng. Sci., 23, 113,1983.
- S. A. Jabarin, J. Appl. Polym. Sci., 34, 85,1987..
- W. H. Cobbs and R L. Burton, J. Polym Sci, 10, 275,1953.
- F. D. Hartley, F. W. Lord, and L. B. Morgan, PhiLfans. Roy. Soc. London, 7.23, 1954.
- B. Gunther and H. G. Zachmann, Polymer, 24, 85, 1983.
- R. Bianchi, P. Chivacci, R Vosa, and G. Guerra, J. AppL Polym Sci, 43, 1087 1991.
- T. Asano, A. Dzeick-Picuth. and H. G. Zachmann, J. Mater. Sci, 24. 1967,1989.
- S. A. Jabarin, J. AppL Pobm Sci, 34.85 ,1987.
- S. Vyazovkin, N. Sbirrazzuoli, J. Phys. Chem. B 107,882, 2003.
- S.C.Tjong,Mater Sci Eng.: R: Reports 41,1,2003.
- M.Lai,H.Yang ,Y.Luan,Y.Yang and J.Lin , J. Appl. Pol.y Sci., 86, 436,2002.
- Hoffman, J.D.; Miller, R.L, Polymer 38, 3151-3212, 1997..
- Cheng, S.Z.D., Janimak, J. J.; Zhang, A. Q. Cheng H. N. Macromolecules 23,298-303, 1990.
- J. Runt, D.M. Miley, X.Zhang, K.P.Gallangher, K.M.Featers, and J. Fishburn, Macromolecules, 25, 1929, 1992.
- S.C.Tjong, Mater Sci Eng.: R: Reports 41, 1, 2003.
| PET/TLCP wt/wt % | 100/00 | 90/10 | 80/20 | 70/30 | 60/40 | 50/50 |
| Nozzle Temperature (°C) | 290 | 285 | 285 | 270 | 270 | 270 |
| Barrel Temperature (°C) | 285 | 275 | 275 | 265 | 265 | 265 |
| Barrel Temperature (°C) | 275 | 275 | 275 | 255 | 255 | 255 |
| Barrel Temperature (°C) | 230 | 230 | 230 | 225 | 225 | 225 |
| Pressure (MPa) | 100 | 100 | 100 | 70 | 70 | 70 |
| Speed (mm/sec) | 91 | 91 | 91 | 91 | 91 | 91 |
| Plasticization (%) | 80 | 80 | 80 | 80 | 80 | 80 |
| Stroke (mm) | 30 | 30 | 30 | 30 | 30 | 30 |
| PET/ TLCP |
Rate of cooling | Onset of cooling temp. | Crystallization Peak temp. | Heat of crystallization |
| (°C/min) | (Tonset)°C | Tc(°C) | ΔHc(J/g) | |
| 100/00 | 10 | 197.76 | 189.73 | -41.721 |
| 20 | 190.36 | 179.07 | -39.295 | |
| 30 | 184.54 | 171.48 | -37.235 | |
| 40 | 181.03 | 165.20 | -35.495 | |
| 50 | 177.63 | 160.68 | -32.644 | |
| 60 | 176.74 | 158.86 | -32.713 | |
| 90/10 | 10 | 201.53 | 193.53 | -37.954 |
| 20 | 191.61 | 180.60 | -37.838 | |
| 30 | 185.56 | 171.98 | -36.532 | |
| 40 | 182.38 | 166.54 | -34.483 | |
| 50 | 177.75 | 159.00 | -33.667 | |
| 60 | 176.72 | 155.86 | -32.839 | |
| 80/20 | 10 | 196.81 | 186.90 | -27.632 |
| 20 | 186.76 | 175.00 | -29.165 | |
| 30 | 182.64 | 167.08 | -29.397 | |
| 40 | 181.20 | 161.87 | -29.318 | |
| 50 | 175.26 | 149.01 | -22.116 | |
| 60 | 175.63 | 144.86 | -27.545 | |
| 70/30 | 10 | 197.70 | 188.57 | -25.875 |
| 20 | 186.94 | 176.27 | -24.404 | |
| 30 | 182.47 | 167.58 | -25.124 | |
| 40 | 179.38 | 161.87 | -22.938 | |
| 50 | 176.71 | 154.01 | -23.331 | |
| 60 | 175.11 | 142.86 | -23.900 | |
| 60/40 | 10 | 195.91 | 188.67 | -18.464 |
| 20 | 186.50 | 175.00 | -18.596 | |
| 30 | 181.52 | 167.08 | -18.394 | |
| 40 | 179.28 | 160.54 | -18.710 | |
| 50 | 175.82 | 146.51 | -19.156 | |
| 60 | 175.15 | 140.86 | -20.529 | |
| 50/50 | 10 | 185.28 | 182.97 | -17.176 |
| 20 | 184.34 | 171.47 | -17.904 | |
| 30 | 179.65 | 162.28 | -17.202 | |
| 40 | 174.74 | 149.87 | -14.357 | |
| 50 | 166.93 | 143.17 | -16.478 | |
| 60 | 159.86 | 138.86 | -14.375 |
| PET/ TLCP |
Rate of cooling (°C/min) |
Tc (°C) |
× 105 |
|
| 100/00 | 10 | 190 | 0.9548 | 2.88 |
| 20 | 179 | 1.4727 | 2.64 | |
| 30 | 171 | 2.1726 | 2.51 | |
| 40 | 165 | 2.6675 | 2.41 | |
| 50 | 161 | 3.2083 | 2.35 | |
| 60 | 159 | 3.5164 | 2.33 | |
| 90/10 | 10 | 194 | 0.9548 | 2.60 |
| 20 | 181 | 1.4727 | 2.38 | |
| 30 | 172 | 2.1726 | 2.26 | |
| 40 | 167 | 2.6675 | 2.20 | |
| 50 | 159 | 3.2083 | 2.12 | |
| 60 | 156 | 3.5164 | 2.09 | |
| 80/20 | 10 | 187 | 0.8650 | 2.38 |
| 20 | 175 | 1.6793 | 2.22 | |
| 30 | 167 | 2.2378 | 2.13 | |
| 40 | 162 | 2.8453 | 2.08 | |
| 50 | 149 | 3.8285 | 1.98 | |
| 60 | 145 | 4.2764 | 1.95 | |
| 70/30 | 10 | 189 | 0.9498 | 2.44 |
| 20 | 176 | 1.9521 | 2.26 | |
| 30 | 168 | 2.3711 | 2.16 | |
| 40 | 162 | 2.8815 | 2.11 | |
| 50 | 154 | 3.5830 | 2.04 | |
| 60 | 143 | 4.5248 | 1.96 | |
| 60/40 | 10 | 189 | 0.9033 | 2.74 |
| 20 | 175 | 2.1291 | 2.48 | |
| 30 | 167 | 2.5618 | 2.36 | |
| 40 | 161 | 3.2341 | 2.28 | |
| 50 | 147 | 4.1428 | 2.14 | |
| 60 | 141 | 4.6789 | 2.09 | |
| 50/50 | 10 | 183 | 1.4088 | 2.96 |
| 20 | 171 | 2.4792 | 2.70 | |
| 30 | 162 | 3.2134 | 2.53 | |
| 40 | 150 | 4.1100 | 2.36 | |
| 50 | 143 | 4.9273 | 2.28 | |
| 60 | 139 | 5.4913 | 2.24 |
| PET/ TLCP |
(C) | Regime transition temp. (°C) |
Nucleation constant Kg x 105 (K2) |
Kg(III) / Kg (II) |
Fold surface free energy (ergs/cm2) |
Work of chain folding q (kcal/mol) |
|||
| II | III | II | III | II | III | ||||
| 100/00 | 271 | 163 | 5.2 | 13.5 | 2.6 | 208.2 | 272.3 | 15.1 | 19.8 |
| 90/10 | 284 | 172 | 3.6 | 7.8 | 2.2 | 141.3 | 154.7 | 10.3 | 11.2 |
| 80/20 | 289 | 171 | 5.1 | 9.8 | 1.9 | 198.4 | 191.1 | 14.4 | 13.9 |
| 70/30 | 287 | 165 | 4.7 | 10.4 | 2.2 | 200.2 | 204.9 | 14.5 | 14.9 |
| 60/40 | 275 | 154 | 4.4 | 11.6 | 2.6 | 177.2 | 233.4 | 12.9 | 17.0 |
| 50/50 | 264 | 153 | 4.5 | 11.6 | 2.6 | 183.7 | 238.5 | 13.3 | 17.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




