Submitted:
10 February 2026
Posted:
11 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Surgical Technique
2.2. Postoperative Rehabilitation
2.3. Clinical and Radiologic Outcomes Evaluation
3. Results
3.1. Demographics
3.2. Clinical Outcomes
3.3. Range of Motion
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Yamamoto, A.; Takagishi, K.; Osawa, T.; Yanagawa, T.; Nakajima, D.; Shitara, H.; Kobayashi, T. Prevalence and risk factors of a rotator cuff tear in the general population. J. Shoulder Elbow Surg. 2010, 19, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Galatz, L.M.; Ball, C.M.; Teefey, S.A.; Middleton, W.D.; Yamaguchi, K. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J. Bone Joint Surg. Am. 2004, 86, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Sugaya, H.; Maeda, K.; Matsuki, K.; Moriishi, J. Repair integrity and functional outcome after arthroscopic double-row rotator cuff repair: A prospective outcome study. J. Bone Joint Surg. Am. 2007, 89, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Matthewson, G.; Beach, C.J.; Nelson, A.A.; Woodmass, J.M.; Ono, Y.; Boorman, R.S.; Thornton, G.M.; Lo, I.K. Partial thickness rotator cuff tears: Current concepts. Bone Joint J. 2015, 97-B, 28–35. [Google Scholar] [CrossRef]
- Ting, R.S.; Wajswelner, H.; Scadding, C.; Cook, J.; Cass, B.; Krishnan, J. Revision rotator cuff repair with versus without an arthroscopically inserted onlay bioinductive implant. Orthop. J. Sports Med. 2023, 11, 23259671221148105. [Google Scholar] [CrossRef]
- Thangarajah, T.; Pendegrass, C.J.; Shahbazi, S.; Lambert, S.; Alexander, S.; Blunn, G.W.; Carr, A.J. Augmentation of rotator cuff repair with soft tissue scaffolds. JBJS Essent. Surg. Tech. 2022, 12, e21.00050. [Google Scholar] [CrossRef]
- Camacho-Chacon, S.; Sanz-Herrera, J.A.; Mora, G.; Cuéllar, R.; Vaquero, J.; Vidal, J.; Vaquero-Martín, J. Histologic evidence of neotendon formation after bioinductive implant application in rotator cuff tears. J. Exp. Orthop. 2022, 9, 20. [Google Scholar] [CrossRef]
- Bushnell, B.D.; Creighton, R.A.; Hovis, W.D.; Tokish, J.M. Retear rates and clinical outcomes after repair of full-thickness rotator cuff tears augmented with a bioinductive collagen implant: A multicenter study. JSES Int. 2021, 5, 381–388. [Google Scholar] [CrossRef]
- McIntyre, L.F.; Bishai, S.K.; Brown, P.B.; Bushnell, B.D.; Trenhaile, S.W. Patient-reported outcomes after use of a bioinductive collagen implant to facilitate healing of partial- and full-thickness rotator cuff tears. Arthrosc. Sports Med. Rehabil. 2021, 3, e1137–e1148. [Google Scholar] [CrossRef]
- Yoo, J.S.; Lee, J.H. Clinical outcomes of a bioinductive collagen implant in bursal-sided partial-thickness rotator cuff tears. Medicina 2025, 61, 988. [Google Scholar] [CrossRef]
- Lawrence, C.; Gulotta, L.; Dines, D.; Rodeo, S.; Warren, R.; Dines, J. Tendon elongation with continuity after rotator cuff repair: An important predictor of structural failure. Orthop. J. Sports Med. 2022, 10, 23259671221083905. [Google Scholar]
- Lawrence, C.; et al. Failure with continuity: Dynamic elongation after rotator cuff repair. Orthop. J. Sports Med. 2022, 10, 23259671221083915. [Google Scholar]
- Ridley, T.J.; et al. Regeneten bioinductive implant rehabilitation protocol. In Univ. Minnesota Orthopaedics; (White Paper/Protocol); 2021. [Google Scholar]
- Micheloni, G.M.; et al. Bio-inductive implant in rotator cuff tears: Our experience and technical notes. Acta Biomed. 2020, 91, 153–160. [Google Scholar] [CrossRef]
- Chona, D.V.; et al. Clinical and structural outcomes of revision rotator cuff repair. J. Shoulder Elbow Surg. 2017, 26, 1889–1895. [Google Scholar] [CrossRef]
- McElvany, M.D.; McGoldrick, E.; Gee, A.O.; Neradilek, M.B.; Matsen, F.A. Rotator cuff repair: Published evidence on factors associated with repair integrity and clinical outcome. Am. J. Sports Med. 2015, 43, 491–500. [Google Scholar] [CrossRef]
- Petri, M.; Greenspoon, J.A.; Millett, P.J. Surgical management of rotator cuff tears: An evidence-based review of the literature. Arthroscopy 2016, 32, 1866–1877. [Google Scholar]
- Sclamberg, S.G.; Tibone, J.E.; Itamura, J.M.; Kasraeian, S. Six-month magnetic resonance imaging follow-up of arthroscopic rotator cuff repairs: Initial results and correlation with clinical outcome. J. Shoulder Elbow Surg. 2004, 13, 570–574. [Google Scholar] [CrossRef]
- Bokor, D.J.; Sonnabend, D.; Deady, L.; Cass, B.; Young, A.; van Kampen, C.; van der Merwe, W.; Visser, J.D.; van den Hoorn, B. Evidence of healing of partial-thickness rotator cuff tears following arthroscopic augmentation with a bioinductive collagen implant: 2-year MRI and clinical outcomes. J. Shoulder Elbow Surg. 2019, 28, 2427–2436. [Google Scholar] [CrossRef]
- Barfod, K.W.; et al. The influence of smoking and diabetes on rotator cuff repair outcomes. J. Shoulder Elbow Surg. 2018, 27, 580–587. [Google Scholar] [CrossRef]
- Abrams, G.D.; et al. Arthroscopic repair of full-thickness rotator cuff tears with and without acromioplasty: Randomized prospective trial. Am. J. Sports Med. 2014, 42, 1296–1303. [Google Scholar] [CrossRef]
- Park, J.Y.; et al. Revision rotator cuff repair: Risk factors for retear and clinical outcomes. Am. J. Sports Med. 2018, 46, 157–165. [Google Scholar]
- Holtby, R.; Razmjou, H. Relationship between clinical and radiologic outcomes after arthroscopic rotator cuff repair: A prospective study. J. Shoulder Elbow Surg. 2010, 19, 573–579. [Google Scholar] [CrossRef]
- Lo, I.K.; Burkhart, S.S. Arthroscopic revision of failed rotator cuff repairs: Technique and results. Arthroscopy 2004, 20, 250–267. [Google Scholar] [CrossRef] [PubMed]
- Mall, N.A.; Lee, A.S.; Chahal, J.; Sherman, S.L.; Romeo, A.A.; Verma, N.N.; Cole, B.J. An evidenced-based examination of the epidemiology and outcomes of traumatic rotator cuff tears. Arthroscopy 2013, 29, 366–376. [Google Scholar] [CrossRef] [PubMed]
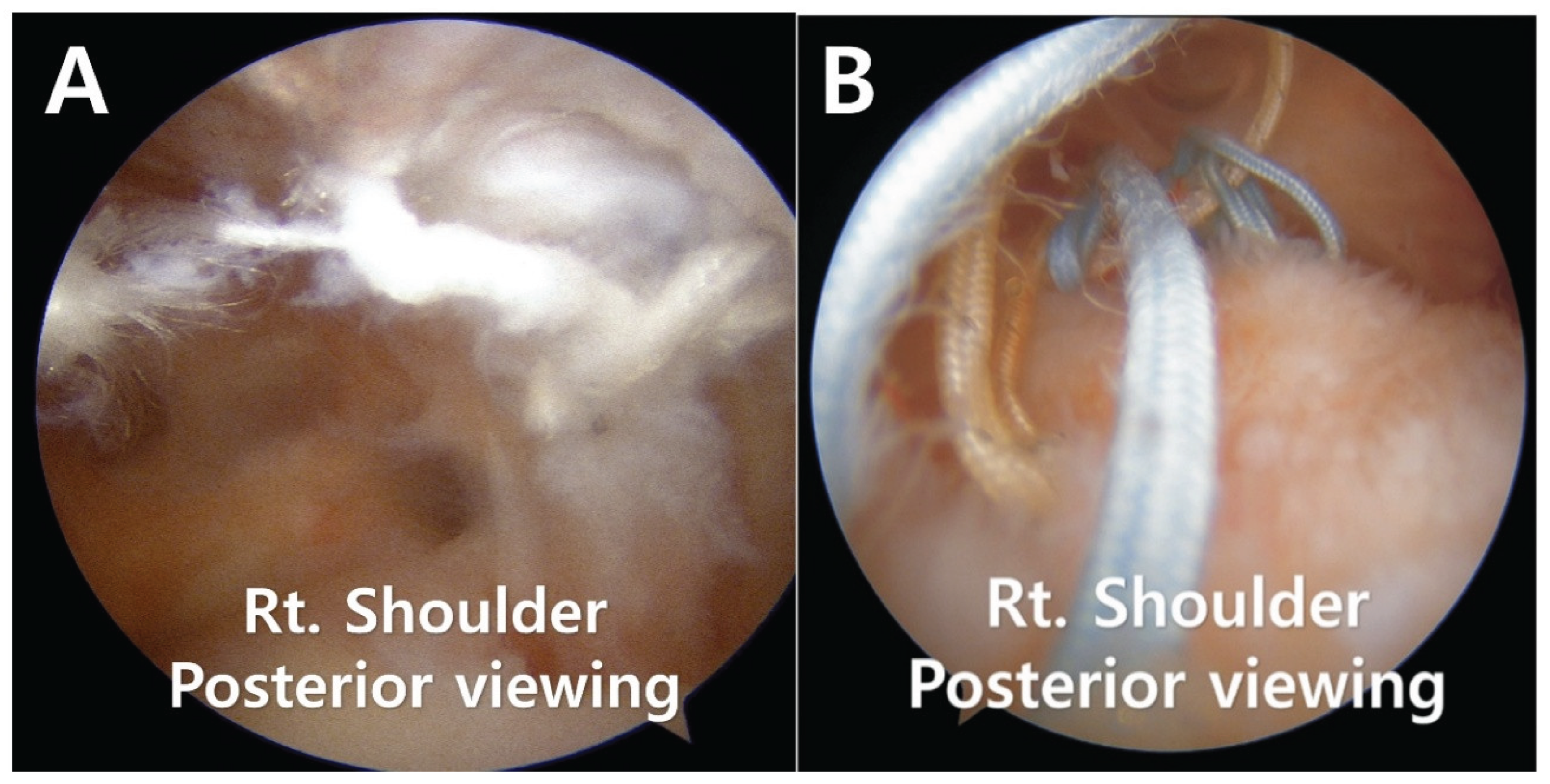
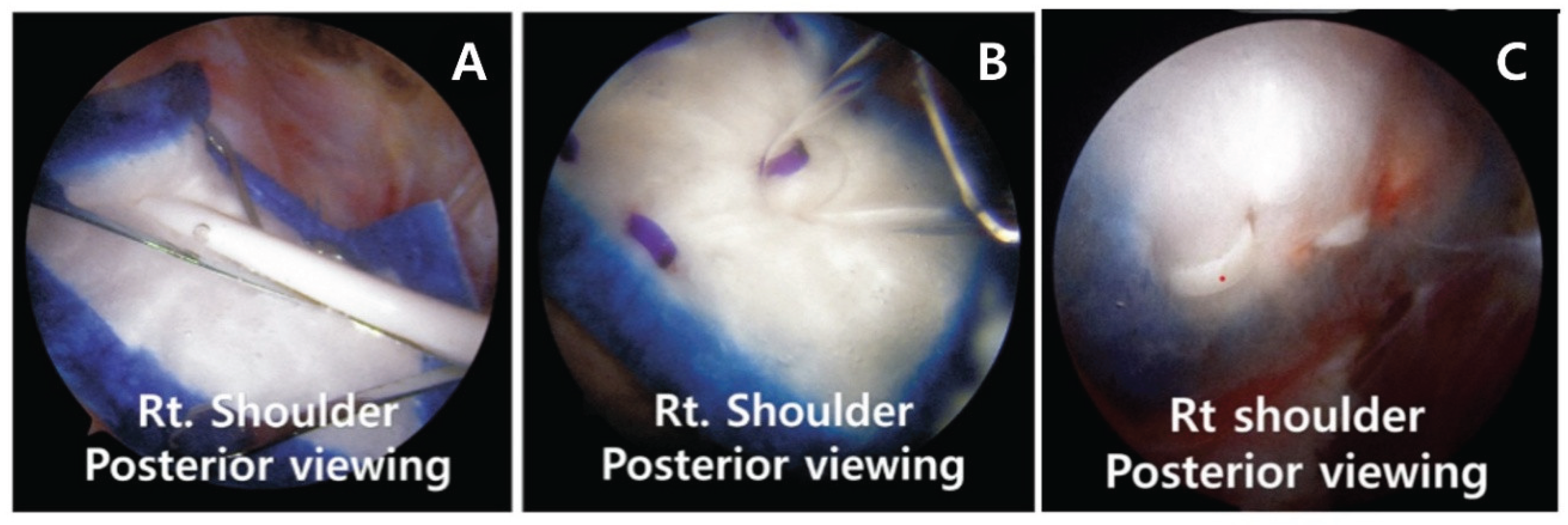
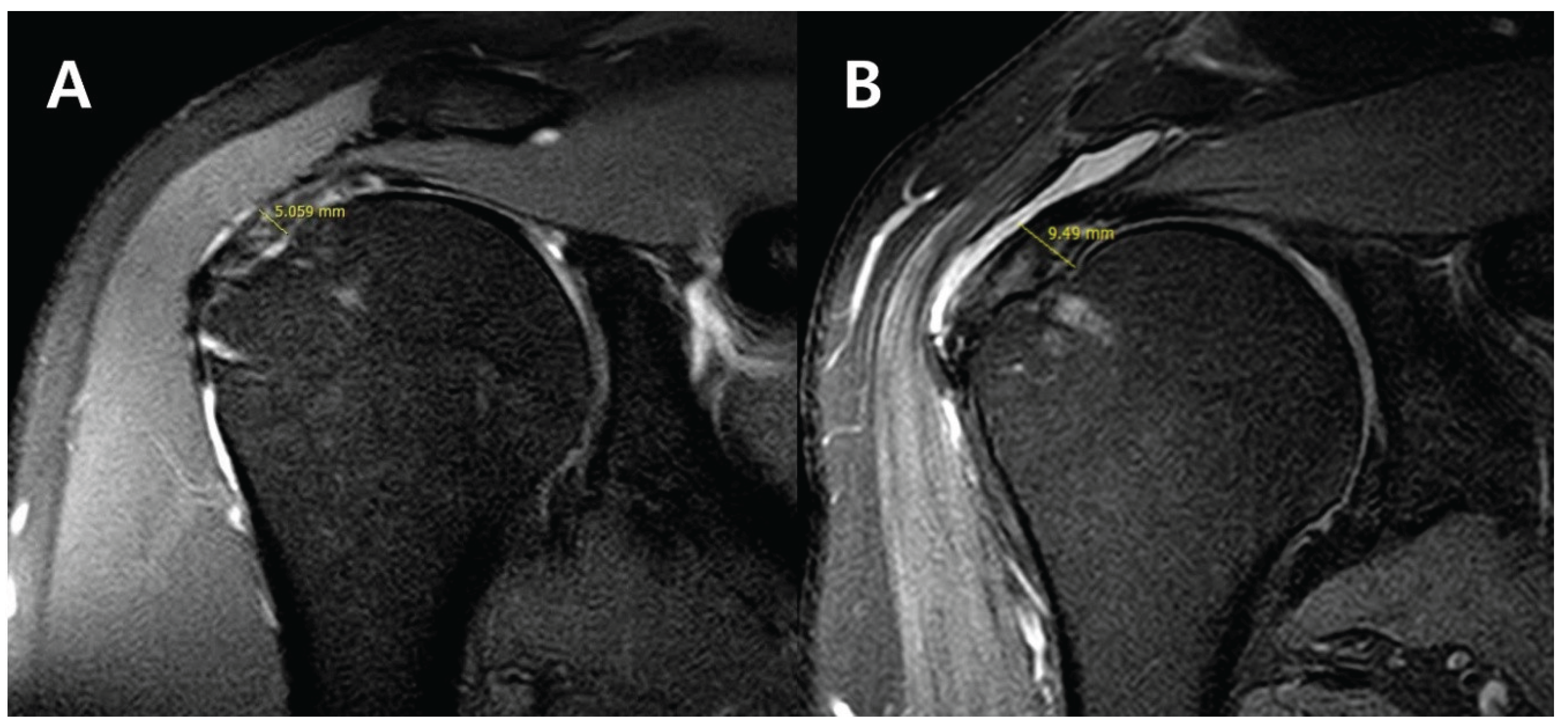
| Case | Age (years) | Sex | BMI (kg/m2) | Dominant side involved | Symptom duration (years) | Diabetes | Smoking | Supraspinatus thickness (mm, MRI) |
| 1 | 62 | M | 24.8 | Yes | 2.5 | No | No | 2.1 |
| 2 | 58 | F | 22.5 | No | 2.1 | No | No | 2.8 |
| 3 | 67 | M | 26.3 | Yes | 3.0 | Yes | Yes | 3.0 |
| 4 | 55 | F | 23.4 | Yes | 2.3 | No | No | 3.7 |
| 5 | 63 | M | 25.6 | No | 2.6 | No | Yes | 4.2 |
| 6 | 60 | F | 21.8 | Yes | 2.4 | No | No | 4.3 |
| 7 | 70 | M | 27.1 | No | 3.2 | Yes | No | 4.8 |
| 8 | 61 | M | 24.2 | Yes | 2.8 | No | Yes | 5.1 |
| 9 | 57 | F | 23.7 | Yes | 2.2 | No | No | 5.2 |
| 10 | 65 | M | 26.9 | No | 3.1 | Yes | Yes | 5.5 |
| 11 | 59 | F | 22.1 | Yes | 2.5 | No | No | 5.7 |
| 12 | 62 | M | 25.0 | Yes | 2.7 | No | Yes | 5.9 |
| 13 | 64 | F | 23.5 | No | 2.9 | No | No | 5.8 |
| 14 | 68 | M | 27.3 | Yes | 3.3 | Yes | Yes | 5.9 |
| 15 | 56 | F | 22.9 | No | 2.4 | No | No | 4.4 |
| Outcome measure | Preoperative |
Postoperative (6 months) |
Postoperative (12 months) |
p-value (preop vs POD 1Y) |
| VAS pain score | 6.5 ± 1.0 | 2.8 ± 0.9 | 2.1 ± 0.8 | <0.001 |
| ASES score | 45.2 ± 8.5 | 71.8 ± 9.3 | 78.5 ± 8.9 | <0.001 |
| SANE score (%) | 41.0 ± 12.1 | 70.2 ± 11.5 | 81.4 ± 9.8 | <0.001 |
| WORC index (%) | 39.6 ± 10.7 | 72.1 ± 12.3 | 83.3 ± 10.1 | <0.001 |
| Supraspinatus thickness (mm, MRI) |
4.5 ± 2.2 | 6.9 ± 2.5 | <0.001 | |
| Forward flexion ROM (°) | 162.0 ± 12.3 | 172.5 ± 8.4 | 176.0 ± 6.5 | n.s. |
| External rotation at side (°) | 60.5 ± 8.1 | 68.2 ± 6.5 | 71.4 ± 6.2 | 0.041 |
| Internal rotation (spinal level) | L2–L3 (T12–L4) |
T12–L1 (T11–L2) |
T11–T12 (T10–L1) |
0.049 |
| Graft failure rate (MRI) | – | 0% | – |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




