1. Introduction
Autoimmune thyroid diseases (AITDs) are a group of organ-specific autoimmune diseases (AIDs) that involve a loss of self-tolerance towards thyroid antigens, mainly thyroglobulin (Tg), thyroid peroxidase (TPO), and the thyrotropin (TSH) receptor (TR) [
1,
2,
3].
The prevalence of AITDs varies according to variables such as the geographical area studied, age, and sex, inter alia, and they affect approximately 5% of the general population, with a significantly higher prevalence in women (5–15%) compared to men (1–5%) [
4,
5,
6].
AITDs have various phenotypic manifestations or expressions, e.g., ranging from the isolated positivity of autoantibodies (Abs) directed against thyroid antigens (TgAb, TPOAb, and TRAb) to the presence of subclinical or overt thyroid dysfunction in hypothyroidism (in Hashimoto’s thyroiditis—HT) or hyperthyroidism (in Graves–Basedow disease—GBD) [
6,
7,
8].
Despite their protean manifestations, AITDs have similar immunogenetic substrates and pathophysiological mechanisms, converging in terms of their genetic, epigenetic, environmental, and other aspects [
9,
10].
Among the environmental (nutritional) mechanisms, the population statuses of several micronutrients stand out [especially iodine, selenium, iron, zinc, magnesium (Mg), copper, vitamin A, vitamin B12, and vitamin D (Vit–D), inter alia], all of which influence the synthesis and regulation of thyroid hormones [thyroxine (T4) and triiodothyronine (T3)], as well as immune responses to the thyroid gland [
11,
12,
13].
Thus, excess or deficiency of these micronutrients have been linked to a higher frequency of AITDs and functional, morphological, and/or malignant thyroid alterations [
12,
13,
14].
Based on the above information, Mg is a cofactor for several enzymes and enzymatic reactions and also metabolizes thyroid hormones; furthermore, the findings of some in vitro studies suggest that free Mg ions at the intracellular level act as second messengers in T lymphocyte (TL) and B lymphocyte (BL) activation, and are also associated with greater oxidative stress and an amplified inflammatory response [
15,
16].
Moreover, TL, BL, and antigen-presenting cells (APCs) express the enzyme 1α–hydroxylase; therefore, they are able to generate the active compound of Vit–D. The above information, in addition to the Vit–D receptor (VDR) also being expressed in these cells, suggests local effects on the immune system (immunomodulatory effects); accordingly, Vit–D acts as a modulator of the innate and adaptive immune system [
15,
16,
17,
18].
The authors of some studies have evaluated the association between a population’s Mg and/or Vit–D status and the risk of AITDs. Several of these have documented a possible relationship between deficiency in one or both micronutrients and a higher frequency of AITDs; however, the authors of other studies have not demonstrated such an association [
11,
12,
13,
16].
We hypothesize that the Mg and/or Vit–D population status is associated with alterations in thyroid function and an increased risk of AITDs. Therefore, in this study, we aim to determine whether serum Mg and/or Vit–D concentrations are associated with alterations in thyroid function and/or the presence of AITDs.
2. Materials and Methods
2.1. Study Population
Between June 2018 and July 2025, a population-based study was conducted in southwestern Colombia to evaluate the distribution and behavior of AITDs, as well as the distribution and frequency of differing thyroid function statuses (euthyroidism, hypothyroidism, or hyperthyroidism—both subclinical and overt) and potential associated factors.
The study was performed in line with the principles of the Declaration of Helsinki and approved by the research ethics committee of the Universidad del Cauca–Colombia (ID: 4656, January 2018).
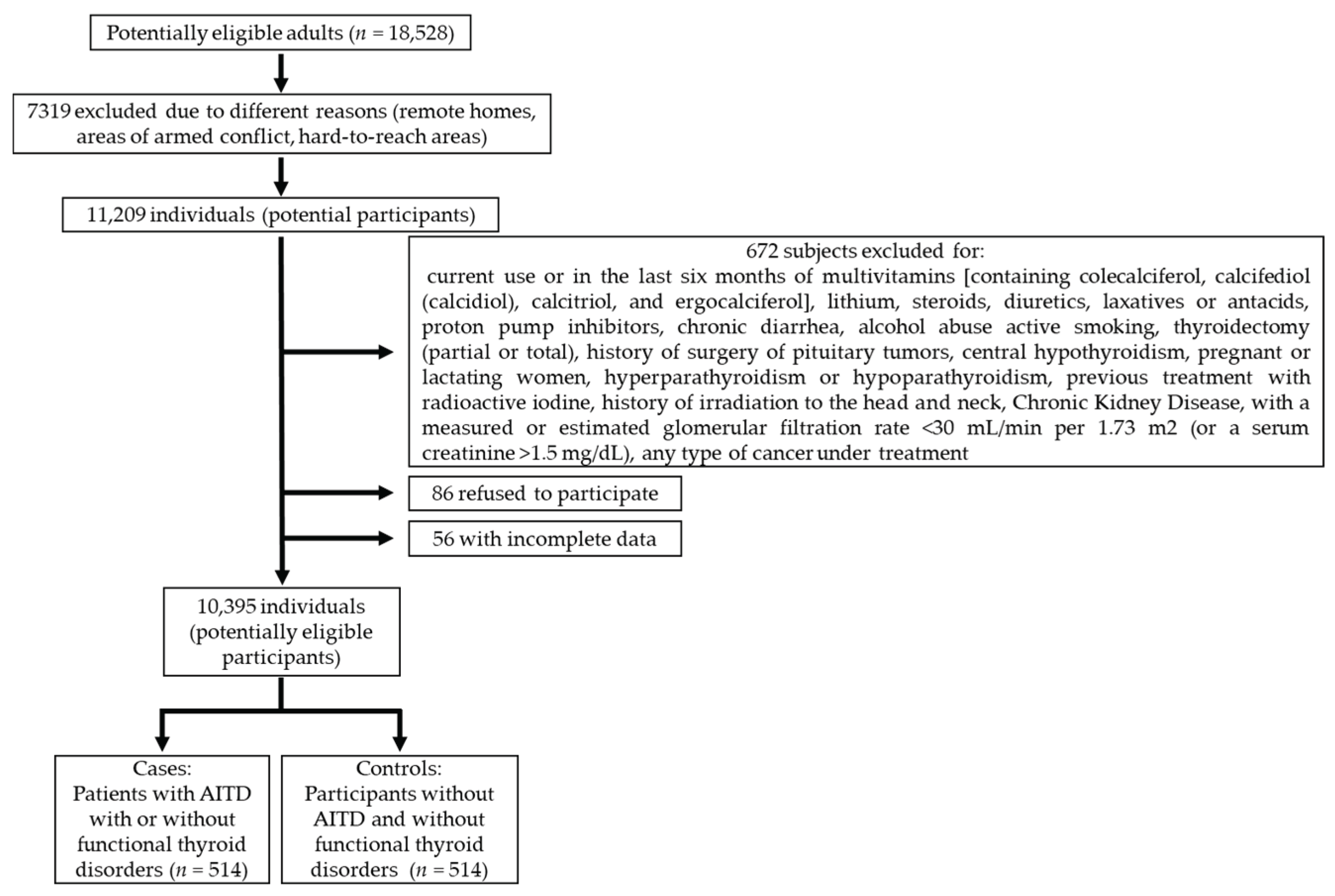
Adults (not institutionalized) residing in urban and rural areas of the study region were included. Participants were proactively invited through conducting direct visits to different neighborhoods and communities, ensuring representative coverage. Initially, 18,528 adults provided informed consent and completed a baseline health history questionnaire, of which 7,319 individuals were excluded based on predefined eligibility criteria, yielding an initial eligible cohort of 11,209 potential participants.
Subsequently, 672 individuals were excluded (for various reasons), 86 declined to participate, and incomplete data were obtained from 56, leaving a final pool of 10,395 potentially eligible participants, from whom cases (n=514) and controls (n=514) were selected (
Figure 1).
Each case was assigned a control in a 1:1 ratio and matched according to the following potential confounding variables: age, sex, socioeconomic status (SES), origin (urban or rural), and body mass index (BMI).
2.2. Study Design
This was a population-based case–control study.
2.3. Definition of AITDs and Normal or Abnormal Thyroid Function
The definition of an AITD was established by measuring the thyroid Abs (TPOAb, TgAb, and TRAb); positivity in at least one of the three thyroid Abs was defined as an AITD.
Thyroid function was assessed and classified by serum TSH and free T4 (FT4) concentrations as follows: normal TSH and FT4 (euthyroidism); elevated TSH with normal FT4 (subclinical hypothyroidism); elevated TSH with low FT4 (overt hypothyroidism); low TSH with elevated FT4 (overt hyperthyroidism); and low TSH with normal FT4 (subclinical hyperthyroidism). In the latter scenario, free T3 (FT3) was also measured to rule out (or confirm) the diagnosis of T3-related hyperthyroidism [
19,
20,
21].
2.4. Exclusion Criteria
To select both study groups, individuals who had been exposed to medications or health conditions that could potentially affect thyroid function in the last three months were excluded (
Figure 1). Each of these exclusion factors was identified through patient self-reporting.
2.5. Sociodemographic and Anthropometric Data
Participants (cases and controls) underwent a structured interview to determine their sociodemographic data and medical history. The following anthropometric data were obtained: age, height, weight, BMI (in kg/m²), and blood pressure (BP) values. Additionally, the presence of goiter was determined by inspection and palpation (according to the World Health Organization criteria) [
22].
2.6. Laboratory Methods
Baseline blood samples were collected by venipuncture between 7:00 and 9:00 a.m. after an overnight fast, centrifuged, frozen (at −80 °C), and stored.
The laboratory variables of interest were measured using a chemiluminescent immunoassay (IMMULITE® 2000 Systems Analyzers; Siemens, Munich, Germany).
In this way, TPOAb, TgAb, and TRAb positivity were determined when the reported values were ≥8.0 IU/mL, ≥18 IU/mL, and >1.75 IU/mL, respectively [
23].
Normal serum concentrations for TSH, FT4, and FT3 were as follows: 0.4–4.5 mIU/L; 0.89–1.76 ng/dL; and 3.5–8.3 pmol/L, respectively. The coefficients of variation for TPOAb, TgAb, and TRAb were 4.96%, 8.0% and 5.3%, respectively; while for TSH, FT4, and FT3 they were 4.6%, 6.4%, and 6.7%, respectively (manufacturer's cutoff values) [
23].
Serum Mg concentration was determined by spectrophotometry (normal reference range: 1.7–2.4 mg/dL). The coefficient of variation was 2.1%, with a detection limit of 0.1 mg/dL and a linearity limit of 4 mg/dL.
To evaluate Vit–D status, the serum concentration of 25–hydroxyvitamin D (25OH Vit–D) was determined by means of a competitive chemiluminescence immunoassay (coefficient of variation: 6.28%); the normal range was defined as 20–50 ng/mL [
24,
25].
2.7. Statistical Analysis
In this population-based case–control study, data from 1,028 records were collected using Windows Excel (Microsoft, 2020) and processed using SPSS version 25.0 software (IBM–SPSS Inc., Chicago, IL). Quality control was performed on 100% of the records included in the database, which included creating a descriptive analysis of each variable to cleanse and correct data; identify missing values, omissions, and blank cells; and determine which records met previously established operational definitions.
To describe the sociodemographic and clinical characteristics of the study population, descriptive statistics were used for numerical (range, mean ± standard deviation, quartiles) and categorical indicator variables (prevalence and percentage distributions). The 95% confidence intervals (CI) were calculated for the prevalence values obtained. Analysis of the statistical association between dichotomous categorical variables was performed using parametric and nonparametric statistical significance tests based on whether or not the statistical assumptions for their application were met (Chi-square and Fisher's exact probability tests). The relative inequality (odds ratio—OR) and its 95% CI were used to measure the strength of the association.
The bivariate statistical analysis for numerical variables was based on Student's t-test, one-way analysis of variance, and linear regression, depending on whether the statistical assumptions for their application were met.
A multivariate binary logistic regression model was used to determine which characteristics were significantly associated with functional alterations and thyroid autoimmunity.
Variables that were found to be significantly associated in the bivariate analysis (p-value <0.05) were introduced. Using clinical measurements of Mg and 25OH Vit–D, receiver operating characteristic (ROC) curves were obtained to analyze the cutoff points, their sensitivity and specificity, and the area under the curve (AUC) to assess their predictive value.
The a priori level of statistical significance was established at α= 0.05. The practical significance of the observed differences between cases and controls was assessed based on the magnitude of the effect.
3. Results
3.1. Anthropometric–sociodemographic Characteristics, Prevalence of Thyroid Functioning Status, and Positivity of Thyroid Abs
Among participants, the mean age was 47.95 years and there was a higher proportion of women (most belonged to the low-SES group and lived in urban areas). The mean BMI and BP were 27.7 kg/m
2 and 128.8/73.2 mmHg, respectively (
Table 1).
3.2. Prevalence of Goiter According to Age and Sex
The prevalence of goiter was significantly higher in the cases vs. the controls [64.6% vs. 11.5%, respectively; OR: 14,0; 95% CI: 10,2 – 19,6]; in patients <50 years of age, it was 69.8% in cases and 9.5% in controls (
p<0.001); in women, the prevalence was 75.6% (in cases) and 9.4% (in controls) (
p<0.001); whereas in men, it was 60% (in cases) and 9.5% (in controls) (
p<0.001) (
Table 2). The prevalence of goiter (in patients aged ≥50 years) was 58.1% in cases and 14% in controls (
p<0.001); in women, it was 57.1% and 10.9% (
p<0.001); while in men, it was 49.8% and 16.8% (
p<0.001). The ORs of goiter prevalence in women (vs. men) were 3.5 (95% CI: 2.9–4.3;
p<0.001) for those <50 years and 2.5 (95% CI: 2.0–2.9;
p<0.001) for those >50 years, respectively.
3.3. Prevalence of Thyroid Functional Disorders and Thyroid Abs Positivity
A significantly higher prevalence of subclinical hypothyroidism, primary hypothyroidism, and goiter was also documented in women (among cases). No differences were found in the prevalence of other thyroid disorders or in thyroid Abs positivity (among cases) based on sex or age (<50 vs. ≥50 years) (
Table 2).
3.4. Prevalence of Normal (Or Deficient) 25OH Vit–D and Mg Levels According to Age
The prevalence of a normal 25OH Vit–D level (20–50 ng/mL) was 62.5% vs. 93.6%, while the prevalence of 25OH Vit–D deficiency (<20 ng/mL and <12 ng/mL) was 37.5% vs. 6.4% and 5.1% vs. 0.6% in cases and controls, respectively.
The prevalence of 25OH Vit–D deficiency (<20 ng/mL) in participants aged <50 years was 36.5% (in cases) and 5.3% (in controls) [OR: 10.3 (95% CI: 5.8–18.3; p<0.001)], and in participants aged ≥50 years it was 38.9% (in cases) and 7.9% (in controls) [OR: 7.4 (95% CI: 4.3–12.9; p<0.001)].
Among cases, the OR of having a 25OH Vit–D level <20 ng/mL was 8.8 (95% CI: 5.9–13.0), while that of a level <12 ng/mL (compared to a level of 12–50 ng/mL) was 9.1 (95% CI: 5.7–30.2; p<0.001).
The prevalence of Mg deficiency (<1.7 mg/dL) in the total population was 13% (95% CI: 10.9–15.1), while that of normal Mg levels (1.7–2.4 mg/dL) was 78.9% (in cases) and 94.9% (in controls). However, the deficiency prevalence was 21.1% vs. 5.1% in cases and controls, respectively [OR: 5.0; 95% CI: 3.2–7.8; p<0.001 (in the total population); OR: 6.4; 95% CI: 3.0–13.2 and OR: 4.3; 95% CI: 2.4–7.7; p<0.001 (in participants aged <50 and ≥50 years, respectively)].
The prevalence of combined Mg and 25OH Vit–D deficiency (<20 ng/mL) was 9.9% (in the total population) and 18.5% and 1.4% [in cases and controls, respectively (OR: 4.9; 95% CI: 2.7.–7.6;
p<0.001)] (
Table 3).
3.5. Prevalence of Thyroid Abs Positivity According to 25OH Vit–D and Mg Levels
The prevalence of thyroid Abs positivity was significantly higher in participants with 25OH Vit–D levels from ≥12 to <20 ng/mL and in those <12 ng/mL (compared to 20–50 ng/mL) in relation to TPOAb (but not for TgAb or TRAb). The findings were similar in participants with Mg deficiency (compared to those with normal levels). Meanwhile, the prevalence of TPOAb and TgAb positivity was significantly higher in participants with combined Mg and 25OH Vit–D deficiency (
Table 4).
3.6. 25OH Vit–D and Mg Levels and Risk of Thyroid Dysfunction
Among the cases with 25OH Vit–D levels <20 ng/mL, the OR of presenting with hypothyroidism (subclinical or overt) was 3.98 (95% CI: 2.87–4.82; p<0.001); for those with Mg levels <1.7 mg/dL, it was 3.2 (95% CI: 1.96–5.41; p<0.001) for subclinical or overt hypothyroidism; and in those with combined Mg and 25OH Vit–D deficiency (<20 ng/mL), it was 3.81 (95% CI: 2.81–4.9; p<0.001) for subclinical or florid hypothyroidism.
Among the cases with 25OH Vit–D levels <20 ng/mL or with Mg levels <1.7 mg/dL, the ORs of presenting with hyperthyroidism (subclinical or overt) were 1.06 (95% CI: 0.87–2.02; p=0.32) and 1.1 (95% CI: 0.69–2.08; p=0.28), respectively.
Meanwhile, the OR for participants with combined Mg and 25OH Vit–D deficiency (<20 ng/mL) was 1.31 (95% CI: 0.91–2.46; p=0.09).
3.7. 25OH Vit–D and Mg Levels and AITD Risk
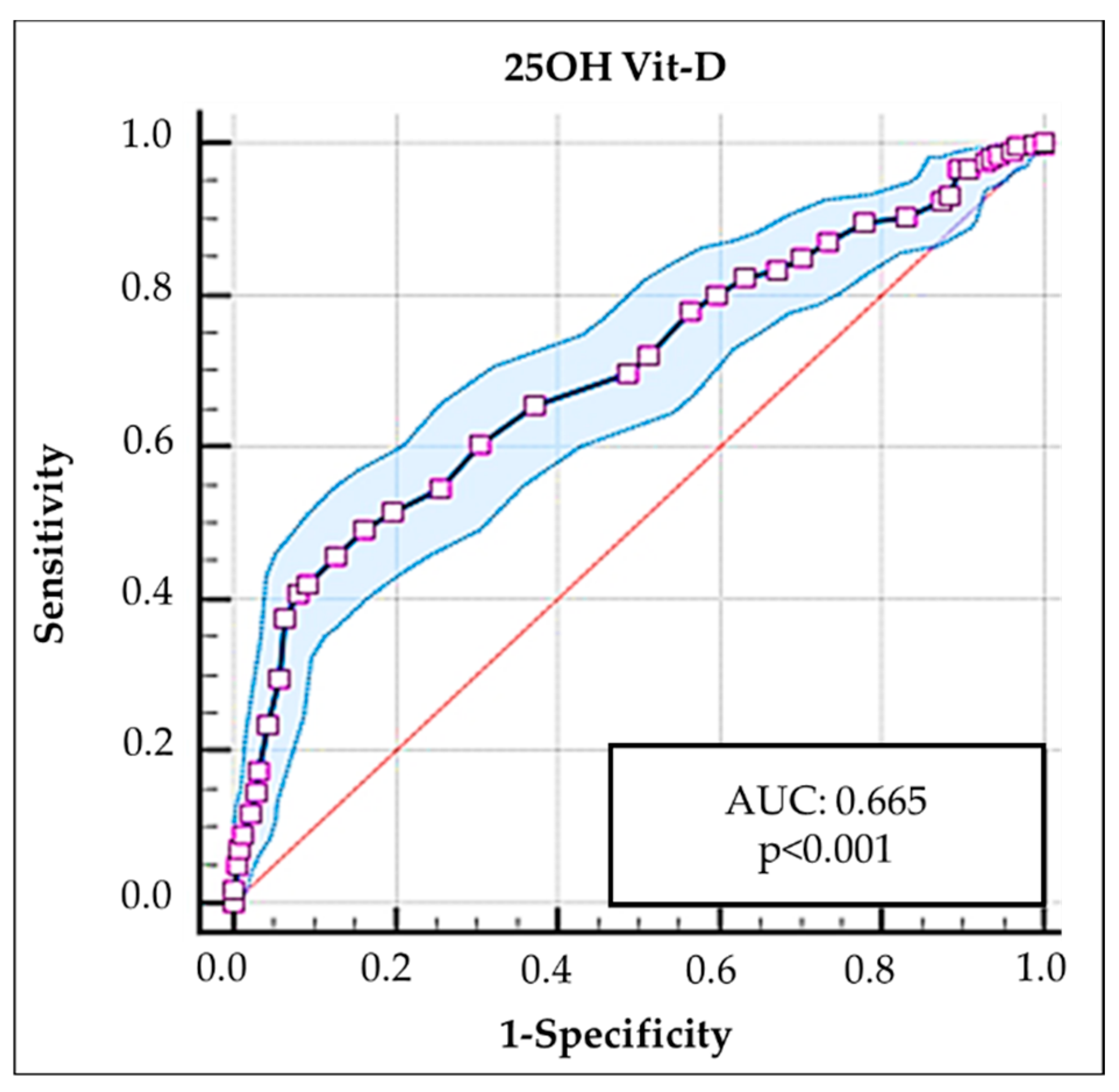
A complementary analysis was developed through an ROC curve analysis, which evaluated different cutoff points in the levels of 25OH Vit–D and Mg (AUC), with the purpose of determining which one best determined the highest frequency of AITDs.
In this sense, the 25OH Vit–D level that best discriminated the highest frequency of AITDs was 23.5 ng/mL [AUC: 0.665 (95% CI: 0.636–0.694;
p<0.001); sensitivity: 0.928, specificity: 0.300 (Youden index: 0.228)] (
Figure 2).
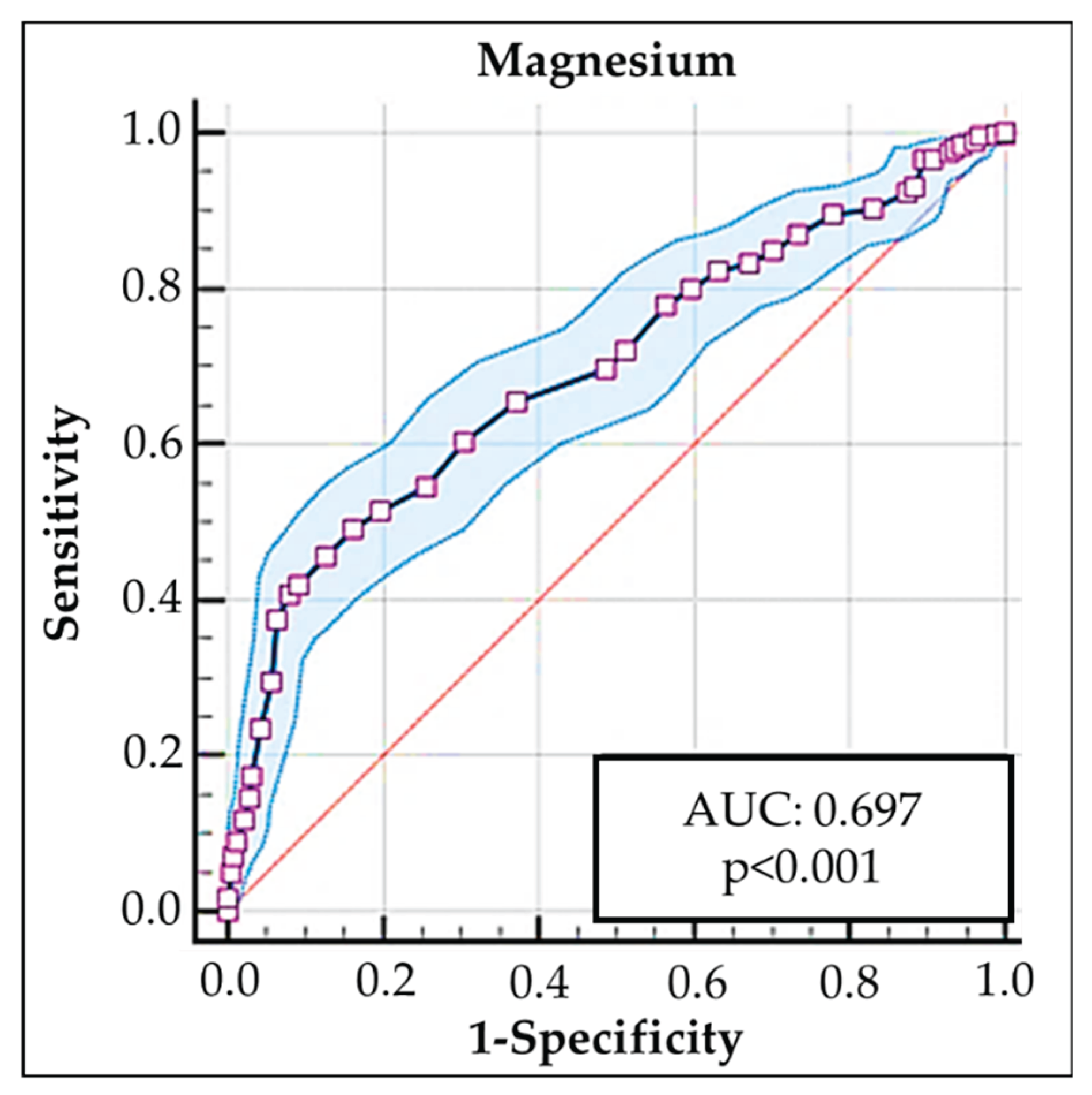
Meanwhile, the Mg level that best discriminated the highest frequency of AITDs was 1.8 mg/dL [AUC: 0.697 (95% CI: 0.668–0.725;
p<0.001); sensitivity: 0.874, specificity: 0.457 (Youden index: 0.332)] (
Figure 3).
In the logistic regression analysis (multivariate), it was found that the risk of presenting with an AITD was significantly higher in cases with 25OH Vit–D levels <23.5 ng/mL [OR: 8.7 (CI 95%: 5.9–13.0; p<0.001)], with magnesium levels <1.8 mg/dL [OR: 5.0 (CI 95%: 3.2–7.8; p<0.001)], and with combined magnesium (<1.7 mg/dL) and 25OH Vit–D deficiency (<20 ng/mL) [OR: 6.4 (95% CI: 4.1–7.7; p<0.001)].
4. Discussion
The authors of several studies have found conflicting results regarding the possible association between Vit–D deficiency and AITD risk; most of the studies with negative results were observational with small sample sizes (and involved participants with other AIDs), while in studies with larger sample sizes (and in intervention studies with Vit–D supplementation), an association was found between Vit–D deficiency and a higher risk of AITDs.
In fact, in intervention studies (with Vit–D supplementation), a significant reduction in thyroid antibody positivity (especially TPOAb and TgAb) has been documented, in addition to a reduction in TSH levels (and an increase in FT4 and FT3 values) in patients with established hypothyroidism under levothyroxine replacement therapy [
26,
27,
28,
29,
30,
31].
In our study, we found a high prevalence of 25OH Vit–D deficiency, which was significantly higher in cases (and independent of age), documenting a significant association between 25OH Vit–D deficiency (<20 ng/mL) and higher prevalence of hypothyroidism (subclinical or overt), as well as higher thyroid Abs positivity (specifically TPOAb); additionally, we found that a cutoff point of <23.5 ng/mL (25OH Vit–D value) best discriminates the risk of presenting with an AITD. Additionally, the higher prevalence of goiter and hypothyroidism in women has been documented in other population-based studies, linked to a higher frequency of deficiencies in other micronutrients, a feasibly greater exposure to environmental goitrogens, and a higher frequency of other autoimmune diseases in women.
In this regard, several systematic reviews and meta-analyses have been carried out evaluating the possible associations between Vit–D deficiency and AITDs and/or thyroid dysfunction, not only finding a high prevalence of Vit–D deficiency in patients with AITDs, but also an inverse relationship between this deficiency and thyroid dysfunction, HT and GBD [
32,
33,
34,
35].
Explaining the link between Vit–D deficiency and AITDs is complex; in this regard, it is well-known that due to the expression of 1α–hydroxylase (CYP27B1), BL, TL, and APCs are able to synthesize the active metabolite of Vit–D, which exhibits immunomodulatory properties. Moreover, the expression of the VDR in these cells suggests local action of Vit–D in the immune response. The relationship between polymorphisms of the Vit–D receptor or the CYP27B1 gene and the frequency of various AIDs supports these findings [
32,
33,
34,
35,
36].
The influence of Vit–D on immune tolerance has been extensively studied; e.g., in APCs, Vit–D is able to modulate and reduce the secretion of pro-inflammatory cytokines (and increase that of those with an anti-inflammatory pattern), consequently inducing a shift in phenotype toward a Th2 response (instead of a Th1 or Th17 response) [
36,
37,
38].
Vit–D also induces greater differentiation of naïve TLCD4+ into regulatory TL (Tregs), stimulating their proliferation and increasing the secretion of IL–10, TGF–β, granzymes, and perforins. Due to Vit–D deficiency, there is an increase in the secretion of TNF–α, which stimulates thyroid cells to release the cytokine CXCL10. As a result, a positive feedback loop occurs, initiating an autoimmune response that becomes permanent over time [
36,
37,
38].
Additionally, Vit–D inhibits NF–κB and p38 MAPK signaling in APCs (e.g., dendritic cells), further reducing the secretion of pro-inflammatory cytokines. It also limits the proliferation and differentiation of plasma cells, as well as the production of memory globulins, while inducing apoptosis in activated globulins. The decreased number of plasma cells results in reduced immunoglobulin production, primarily of the IgE and IgG classes. In cases of Vit–D deficiency, this process is not inhibited, leading to elevated immunoglobulin levels and ultimately causing cell damage (and, eventually, loss of self-tolerance to thyroid antigens and hypothyroidism) [
38,
39].
On the other hand, few studies have been conducted to assess the possible associations between Mg deficiency and the risk of AITDs; the limited evidence suggests that severely low serum Mg levels are associated with an increased rate of TgAb positivity, HT, and hypothyroidism [
40,
41].
In our study, we found a high prevalence of Mg deficiency among the cases and a significant association between this deficiency and a higher risk of AITDs, as well as a higher prevalence of hypothyroidism (subclinical or overt). Additionally, we found that a cutoff point of <1.8 mg/dL (in Mg) best discriminates the risk of presenting with AITDs.
In participants with combined Mg and 25OH Vit–D deficiency, we also found a higher prevalence of hypothyroidism (subclinical or florid) as well as a higher frequency of TPOAb and/or TgAb positivity; however, the magnitude of the association was smaller compared to that found with isolated Mg or 25OH Vit–D deficiency.
The findings of several studies suggest that Mg may play an important role in functional and/or autoimmune thyroid disorders; some findings suggest an inverse relationship with low serum Mg concentrations in individuals with positive TPOAb or TgAb (compared to those with negative TPOAbs) [
40,
41,
42].
In other studies, the authors have shown a negative correlation between serum Mg and lymphocyte activation in patients with GBD, suggesting that immune tolerance in the thyroid may be impaired by low Mg levels [
40,
41,
42,
43].
These findings suggest that Mg may act as an important regulator of immunity, as low serum Mg levels have been associated with increased secretion of pro-inflammatory cytokines. Additionally, mutations in MAGT1 (magnesium transporter 1), a specific Mg transporter, contribute to alterations in TL function and NK cell activation. Mg also promotes lymphocyte function-associated antigen 1 (LFA–1), a costimulatory molecule present on the membranes of TLCD8+ (which has antitumor properties).
It should also be noted that Mg is an enzymatic cofactor playing a fundamental role in mitochondrial oxidative phosphorylation and ATP synthesis; therefore, low levels of Mg can affect these functions and cause a decrease in iodine uptake at the thyroid level and, consequently, a decrease in the synthesis of thyroid hormones, causing increased TSH secretion [
40,
41,
42,
43,
44,
45].
Meanwhile, authors of other studies (in patients with GBD) have found low Mg levels (compared to healthy individuals), and that this low concentration was inversely related to the activation of TLCD3+, TLCD4+, TLCD8+, and BLCD19+; this decreased immune tolerance and caused an abnormal and amplified activation of immune cells. Additionally, low Mg levels can also reduce antioxidant responsiveness and allow the accumulation of reactive oxygen species, causing oxidative stress and tissue damage [
45,
46,
47,
48,
49,
50].
These findings may explain, at least in part, the association between low levels of Mg and the high prevalence of hypothyroidism and AITDs in our study.
The results of our study should be analyzed and considered in light of its potential limitations; e.g., due to the study design, we cannot establish a causal association between the deficiency of both micronutrients and the presence of thyroid dysfunction and/or AITDs; therefore, it is not possible to precisely establish a temporal sequence between the exposure (25OH Vit–D and/or Mg deficiency) and the observed outcomes.
Similarly, matching participants according to the specified potential confounding factors [age, SES, origin (urban or rural), and socioeconomic status] potentially reduces the possibility of assessing whether these variables differ between cases and controls (in addition to the potential risk of overmatching). The presence and effects of other potential residual confounding factors cannot be ruled out either, e.g., the nutritional habits of the study population in relation to the intake of foods or consumer products containing magnesium and/or Vit–D, which could have potentially influenced the differences found between the study groups. The population status of other micronutrients and the presence of endocrine disruptors, which can increase (or attenuate) the associations found, were also not taken into account.
5. Conclusions
In conclusion, in this study, we found that magnesium and Vit–D deficiency (isolated or combined) is associated with a significant increase in the risk of thyroid dysfunction (specifically hypothyroidism) and with increased positivity in thyroid antibodies.
Randomized controlled trials with a longer follow-up period are needed to confirm the causal relationship between Mg and/or Vit–D and thyroid outcomes (hypothyroidism, hyperthyroidism, and AITDs, inter alia) and to provide more reliable insights into the relevance of treatment effects with Mg and/or Vit–D (therapy or supplementation).
Author Contributions
Conceptualization, H.V.-U., A.C.-P., M.V.P.-F., K.U.-N. and H.V.-S.; methodology, H.V.-U., A.C.-P., M.V.P.-F., I.A.M.-C., and V.A.-D; investigation, H.V.-U., I.A.M.-C., A.C.-P., M.V.P.-F., and V.A.-D.; resources, H.V.-U., I.A.M.-C., K.U.-N. and M.V.P.-F.; data curation, H.V.-U., H.V.-S., V.A.-D., and M.V.P.-F.; writing—original draft preparation, H.V.-U., I.A.M.-C., K.U.-N., A.C.-P., and M.V.P.-F.; writing—review and editing, H.V.-U., A.C.-P., I.A.M.-C. and H.V.-S.; visualization, M.V.P.-F., K.U.-N., and H.V.-S.; supervision, H.V.-U.; project administration and funding acquisition, H.V.-U. and M.V.P.-F. All authors have read and agreed to the published version of the manuscript.
Funding
This study received funding from the Colombian Association of Endocrinology, Diabetes, and Metabolism (009-232025).
Institutional Review Board Statement
The study was performed in line with the principles of the Declaration of Helsinki and approved by the research ethics committee of the Universidad del Cauca–Colombia (ID: 4656, January 2018).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Vargas–Uricoechea, H. Molecular mechanisms in autoimmune thyroid disease. Cells 2023, 12, 918. [CrossRef]
- Martínez–Hernández, R.; Sánchez de la Blanca, N.; Sacristán–Gómez, P.; Serrano–Somavilla, A.; Muñoz De Nova, J.L.; Sánchez Cabo, F.; Heyn, H.; Sampedro–Núñez, M.; Marazuela, M. Unraveling the molecular architecture of autoimmune thyroid diseases at spatial resolution. Nat. Commun. 2024, 15, 5895. [CrossRef]
- Li, P.; Wang, Q.; Yang, Y.; Ding, Z. Autoimmune thyroid disease and human health: A systematic review of Mendelian randomization studies. Front. Immunol. 2025, 16, 1689498. [CrossRef]
- Conrad, N.; Misra, S.; Verbakel, J.Y.; Verbeke, G.; Molenberghs, G.; Taylor, P.N.; Mason, J.; Sattar, N.; McMurray, J.J.V.; McInnes, I.B.; et al. Incidence, prevalence, and co–occurrence of autoimmune disorders over time and by age, sex, and socioeconomic status: A population–based cohort study of 22 million individuals in the UK. Lancet 2023, 401, 1878–1890. [CrossRef]
- Petranović Ovčariček, P.; Görges, R.; Giovanella, L. Autoimmune thyroid diseases. Semin. Nucl. Med. 2024, 54, 219–236.
- Dwivedi, S.N.; Kalaria, T.; Buch, H. Thyroid autoantibodies. J. Clin. Pathol. 2023, 76, 19–28.
- Li, R.; He, T.; Xing, Z.; Mi, L.; Su, A.; Wu, W. The immune system in Hashimoto’s thyroiditis: Updating the current state of knowledge on potential therapies and animal model construction. Autoimmun. Rev. 2025, 24, 103783. [CrossRef]
- Kobayashi, G.; Okamura, T.; Hashimoto, Y.; Sakai, K.; Sumi, M.; Imai, D.; Kitagawa, N.; Hamaguchi, M.; Fukui, M. Pathogenesis of Graves’ disease determined using single–cell sequencing with thyroid autoantigen peptide stimulation in B cells. Cells 2025, 14, 1102. [CrossRef]
- Tywanek, E.; Michalak, A.; Świrska, J.; Zwolak, A. Autoimmunity, new potential biomarkers and the thyroid gland—The perspective of Hashimoto’s thyroiditis and its treatment. Int. J. Mol. Sci. 2024, 25, 4703. [CrossRef]
- Shrestha, A.; Adhikari, N.; Devkota, S.; Chowdhury, T.; Shiferaw–Deribe, Z.; Gousy, N.; Adhikari, S. Fluctuating hyperthyroidism and hypothyroidism in Graves’ disease. Cureus 2022, 14, e27715. [CrossRef]
- Shulhai, A.M.; Rotondo, R.; Petraroli, M.; Patianna, V.; Predieri, B.; Iughetti, L.; Esposito, S.; Street, M.E. The role of nutrition on thyroid function. Nutrients 2024, 16, 2496. [CrossRef]
- Mikulska, A.A.; Karaźniewicz–Łada, M.; Filipowicz, D.; Ruchała, M.; Główka, F.K. Metabolic characteristics of Hashimoto’s thyroiditis patients and the role of microelements and diet in disease management—An overview. Int. J. Mol. Sci. 2022, 23, 6580. [CrossRef]
- Yu, Y.; Tong, K.; Deng, J.; Wu, J.; Yu, R.; Xiang, Q. Unveiling the connection between micronutrients and autoimmune thyroiditis. Biol. Trace Elem. Res. 2025, 203, 4994–5005. [CrossRef]
- Barrea, L.; Gallo, M.; Ruggeri, R.M.; Giacinto, P.D.; Sesti, F.; Prinzi, N.; Adinolfi, V.; Barucca, V.; Renzelli, V.; Muscogiuri, G.; et al.; E.O.L.O. Group. Nutritional status and follicular–derived thyroid cancer: An update. Crit. Rev. Food Sci. Nutr. 2021, 61, 25–59. [CrossRef]
- Zhou, Q.; Xue, S.; Zhang, L.; Chen, G. Trace elements and the thyroid. Front. Endocrinol. (Lausanne) 2022, 13, 904889. [CrossRef]
- Li, S.; Xu, Q.; Wang, S.; Peng, H.; Liu, Y. Recent advances of trace elements in autoimmune thyroid disease. Front. Immunol. 2025, 16, 1662521. [CrossRef]
- Gorini, F.; Tonacci, A. Vitamin D: An essential nutrient in the dual relationship between autoimmune thyroid diseases and celiac disease—A comprehensive review. Nutrients 2024, 16, 1762. [CrossRef]
- Pleić, N.; Babić Leko, M.; Gunjača, I.; Zemunik, T. Vitamin D and thyroid function: A Mendelian randomization study. PLoS ONE 2024, 19, e0304253. [CrossRef]
- Jonklaas, J.; Bianco, A.C.; Bauer, A.J.; Burman, K.D.; Cappola, A.R.; Celi, F.S.; Cooper, D.S.; Kim, B.W.; Peeters, R.P.; Rosenthal, M.S.; et al.; American Thyroid Association Task Force on Thyroid Hormone Replacement. Guidelines for the treatment of hypothyroidism. Thyroid 2014, 24, 1670–1751. [CrossRef]
- Wang, Y.; Zhang, J.; Cheng, X.; Duan, X.; Liang, Y.; Sun, D. Quality of guidelines for hyperthyroidism. Postgrad. Med. 2025, 137, 139–147.
- National Institute for Health and Care Excellence (NICE). Thyroid disease: assessment and management. NICE Clinical Guidelines, No. 145; NICE: London, UK, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK550859/ (accessed on 18 November 2025).
- World Health Organization; UNICEF; ICCIDD. Indicators for assessing iodine deficiency disorders and their control through salt iodization. WHO/NUT/94.6; WHO: Geneva, Switzerland, 1994.
- Vargas–Uricoechea, H.; Urrego–Noguera, K.; Vargas–Sierra, H.; Pinzón–Fernández, M. Zinc and ferritin levels and their associations with functional disorders and/or thyroid autoimmunity: A population–based case–control study. Int. J. Mol. Sci. 2024, 25, 10217. [CrossRef]
- Laborteknic. Codigo 12797—Quimica Dedica A15–25. Available online: https://laborteknic.com/wp–content/uploads/2022/04/Codigo–12797–Quimica–Dedica–A15–25.pdf (accessed on 12 September 2024).
- DNA–NIK. OHVitaminD. Available online: https://dna–nik.com/pdf/OHVitaminD.pdf (accessed on 12 September 2024).
- Tang, J.; Shan, S.; Li, F.; Yun, P. Effects of vitamin D supplementation on autoantibodies and thyroid function in patients with Hashimoto's thyroiditis: A systematic review and meta–analysis. Medicine (Baltimore) 2023, 102, e36759. [CrossRef]
- Yu, Y.; Yang, X.; Wu, J.; Shangguan, X.; Bai, S.; Yu, R. A Mendelian randomization study of the effect of serum 25–hydroxyvitamin D levels on autoimmune thyroid disease. Front. Immunol. 2024, 14, 1298708. [CrossRef]
- Zhang, J.; Chen, Y.; Li, H.; Li, H. Effects of vitamin D on thyroid autoimmunity markers in Hashimoto’s thyroiditis: Systematic review and meta–analysis. J. Int. Med. Res. 2021, 49, 3000605211060675.
- Vahabi Anaraki, P.; Aminorroaya, A.; Amini, M.; Momeni, F.; Feizi, A.; Iraj, B.; Tabatabaei, A. Effect of vitamin D deficiency treatment on thyroid function and autoimmunity markers in Hashimoto’s thyroiditis: A double–blind randomized placebo–controlled clinical trial. J. Res. Med. Sci. 2017, 22, 103. [CrossRef]
- Chahardoli, R.; Saboor–Yaraghi, A.A.; Amouzegar, A.; Khalili, D.; Vakili, A.Z.; Azizi, F. Can supplementation with vitamin D modify thyroid autoantibodies and thyroid profile in Hashimoto's thyroiditis? A double blind, randomized clinical trial. Horm. Metab. Res. 2019, 51, 296–301.
- Luo, D.; Li, B.; Shan, Z.; Teng, W.; Liu, Q.; Li, J. The impacts of vitamin D supplementation on serum levels of thyroid autoantibodies in patients with autoimmune thyroid disease: A meta–analysis. PeerJ 2025, 13, e19541. [CrossRef]
- Khozam, S.A.; Sumaili, A.M.; Alflan, M.A.; Shawabkeh, R.A.S. Association between vitamin D deficiency and autoimmune thyroid disorder: A systematic review. Cureus 2022, 14, e25869. [CrossRef]
- Wang, J.; Lv, S.; Chen, G.; Gao, C.; He, J.; Zhong, H.; Xu, Y. Meta–analysis of the association between vitamin D and autoimmune thyroid disease. Nutrients 2015, 7, 2485–2498.
- Xu, M.Y.; Cao, B.; Yin, J.; Wang, D.F.; Chen, K.L.; Lu, Q.B. Vitamin D and Graves' disease: A meta–analysis update. Nutrients 2015, 7, 3813–3827. [CrossRef]
- Vargas–Uricoechea, H.; Castellanos–Pinedo, A.; Urrego–Noguera, K.; Pinzón–Fernández, M.V.; Meza–Cabrera, I.A.; Vargas–Sierra, H. A scoping review on the prevalence of Hashimoto's thyroiditis and the possible associated factors. Med. Sci. 2025, 13, 43. [CrossRef]
- Altieri, B.; Muscogiuri, G.; Barrea, L.; Mathieu, C.; Vallone, C.V.; Mascitelli, L.; Bizzaro, G.; Altieri, V.M.; Tirabassi, G.; Balercia, G.; et al. Does vitamin D play a role in autoimmune endocrine disorders? A proof of concept. Rev. Endocr. Metab. Disord. 2017, 18, 335–346. [CrossRef]
- Zhao, R.; Zhang, W.; Ma, C.; Zhao, Y.; Xiong, R.; Wang, H.; Chen, W.; Zheng, S.G. Immunomodulatory function of vitamin D and its role in autoimmune thyroid disease. Front. Immunol. 2021, 12, 574967. [CrossRef]
- Lebiedziński, F.; Lisowska, K.A. Impact of vitamin D on immunopathology of Hashimoto's thyroiditis: From theory to practice. Nutrients 2023, 15, 3174. [CrossRef]
- Czarnywojtek, A.; Florek, E.; Pietrończyk, K.; Sawicka–Gutaj, N.; Ruchała, M.; Ronen, O.; Nixon, I.J.; Shaha, A.R.; Rodrigo, J.P.; Tufano, R.P.; et al. The role of vitamin D in autoimmune thyroid diseases: A narrative review. J. Clin. Med. 2023, 12, 1452. [CrossRef]
- Gorini, F.; Tonacci, A. Vitamin D: An essential nutrient in the dual relationship between autoimmune thyroid diseases and celiac disease—A comprehensive review. Nutrients 2024, 16, 1762. [CrossRef]
- Wang, K.; Wei, H.; Zhang, W.; Li, Z.; Ding, L.; Yu, T.; Tan, L.; Liu, Y.; Liu, T.; Wang, H.; et al. Severely low serum magnesium is associated with increased risks of positive anti–thyroglobulin antibody and hypothyroidism: A cross–sectional study. Sci. Rep. 2018, 8, 9904. [CrossRef]
- Kolanu, B.R.; Vadakedath, S.; Boddula, V.; Kandi, V. Activities of serum magnesium and thyroid hormones in pre–, peri–, and post–menopausal women. Cureus 2020, 12, e6554. [CrossRef]
- Luo, Y.; Zeng, H.; Ye, Y.; Yu, G.; Song, C.; Liu, S.; Chen, X.; Jiang, Y.; Duan, H.; Li, Y.; et al. Associations of metal profiles in blood with thyroiditis: A cross–sectional study. Environ. Sci. Pollut. Res. Int. 2023, 30, 21072–21080. [CrossRef]
- Klatka, M.; Grywalska, E.; Partyka, M.; Charytanowicz, M.; Rolinski, J. Impact of methimazole treatment on magnesium concentration and lymphocytes activation in adolescents with Graves’ disease. Biol. Trace Elem. Res. 2013, 153, 155–170. [CrossRef]
- Jones, J.E.; Desper, P.C.; Shane, S.R.; Flink, E.B. Magnesium metabolism in hyperthyroidism and hypothyroidism. J. Clin. Investig. 1966, 45, 891–900. [CrossRef]
- Wang, K.; Wei, H.; Zhang, W.; Li, Z.; Ding, L.; Yu, T.; Tan, L.; Liu, Y.; Liu, T.; Wang, H.; et al. Severely low serum magnesium is associated with increased risks of positive anti–thyroglobulin antibody and hypothyroidism: A cross–sectional study. Sci. Rep. 2018, 8, 9904. [CrossRef]
- Klatka, M.; Grywalska, E.; Partyka, M.; Charytanowicz, M.; Rolinski, J. Impact of methimazole treatment on magnesium concentration and lymphocytes activation in adolescents with Graves’ disease. Biol. Trace Elem. Res. 2013, 153, 155–170. [CrossRef]
- Morais, J.B.; Severo, J.S.; Santos, L.R.; de Sousa Melo, S.R.; de Oliveira Santos, R.; de Oliveira, A.R.; Cruz, K.J.; do Nascimento Marreiro, D. Role of magnesium in oxidative stress in individuals with obesity. Biol. Trace Elem. Res. 2017, 176, 20–26. [CrossRef]
- Celik, E.; Celik, M.; Bulbul, B.Y.; Andac, B.; Okur, M.; Colak, S.Y.; Yekdes, A.C. Immunological harmony: The role of magnesium in the development of euthyroid Hashimoto’s thyroiditis. Journal of Elem. 2024, 29, 367–378. [CrossRef]
- Disashi, T.; Iwaoka, T.; Inoue, J.; Naomi, S.; Fujimoto, Y.; Umeda, T.; Tomita, K. Magnesium metabolism in hyperthyroidism. Endocr. J. 1996, 43, 397–402. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).