Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Reagents
2.2. Cell Treatments
2.2.1. PunA, α-ESA, and Cisplatin Treatment of OC and Normal Cell Lines
2.2.2. Combinational Chemo-Treatment
2.2.3. PunA Treatment on Cisplatin Resistant Cell Lines
2.2.4. PunA Treatment on OC Cells in High Versus Low Fat Medium
2.2.5. Ferrostatin-1 Treatment
2.3. Cell Viability Assay (MTS Assay)
2.4. Lipid Peroxidation Assay by Flow Cytometry
2.5. Fatty Acid Oxidation (FAO) Assay
2.6. Measurement of Oxygen Consumption Rate (OCR)
2.8. Peroxisome Proliferator-Activated Receptor Gamma (PPAR-γ) Assay
2.9. Tumor Development in C57BL/6J Mouse Model
2.10. PunA Preventive Treatment Using C57BL/6J Mouse Model
2.11. IVIS Bioluminescence Imaging
2.12. RNA Extraction and RT-qPCR
2.13. RNA Extraction and RNA Sequencing
2.14. Statistical Analysis
3. Results
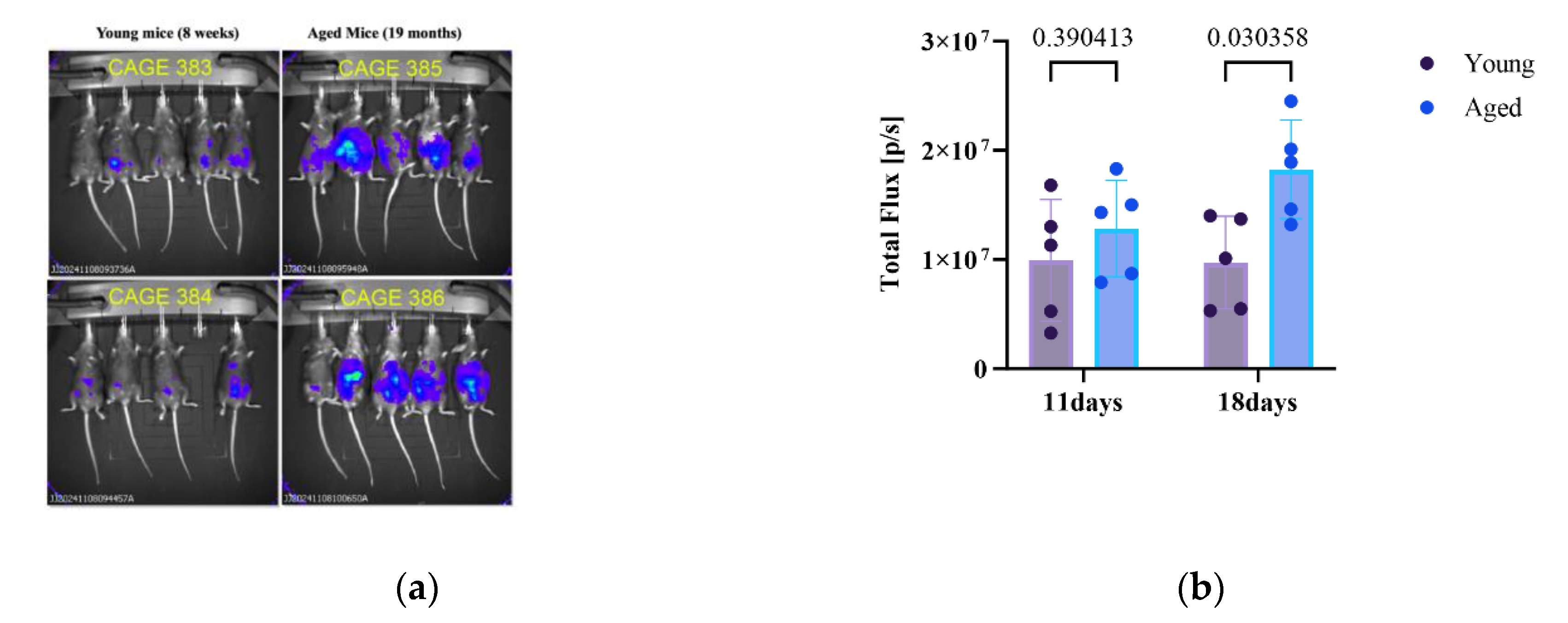
3.1. Aging Promotes Tumor Expansion in Immunocompetent Mice
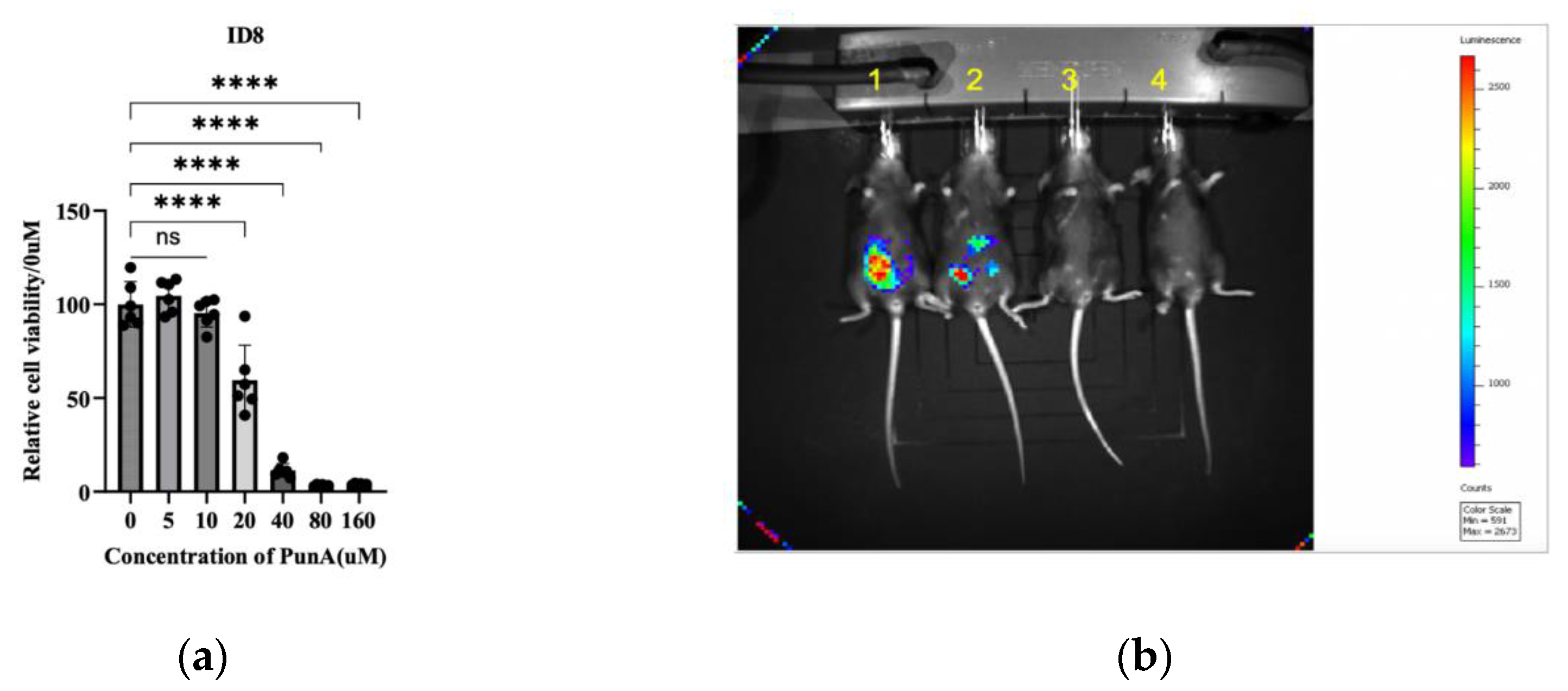
3.2. PunA Suppresses Cancer Progression in a Xenograft Mouse Model
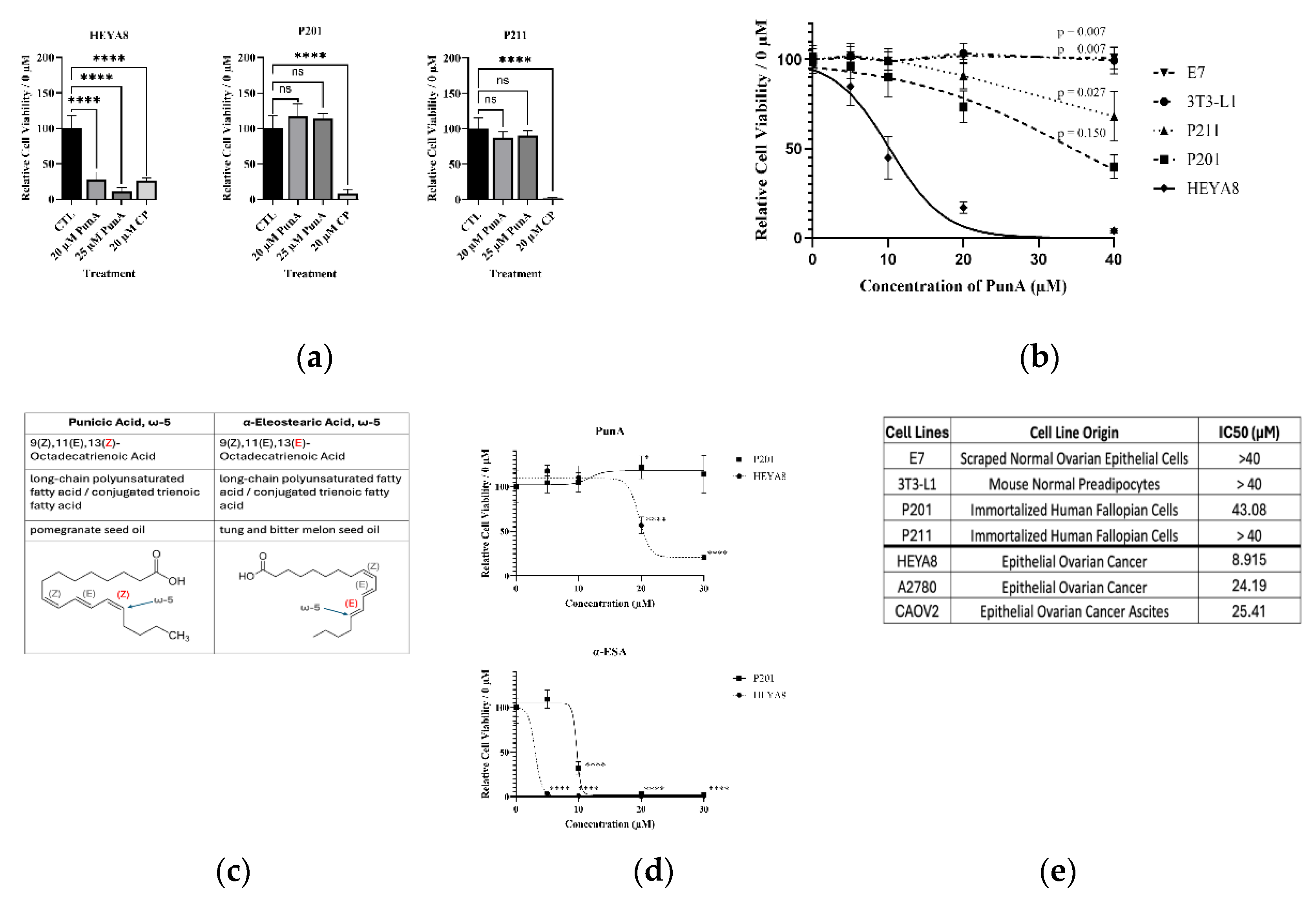
3.3. PunA Induces Cancer-Specific Cell Death
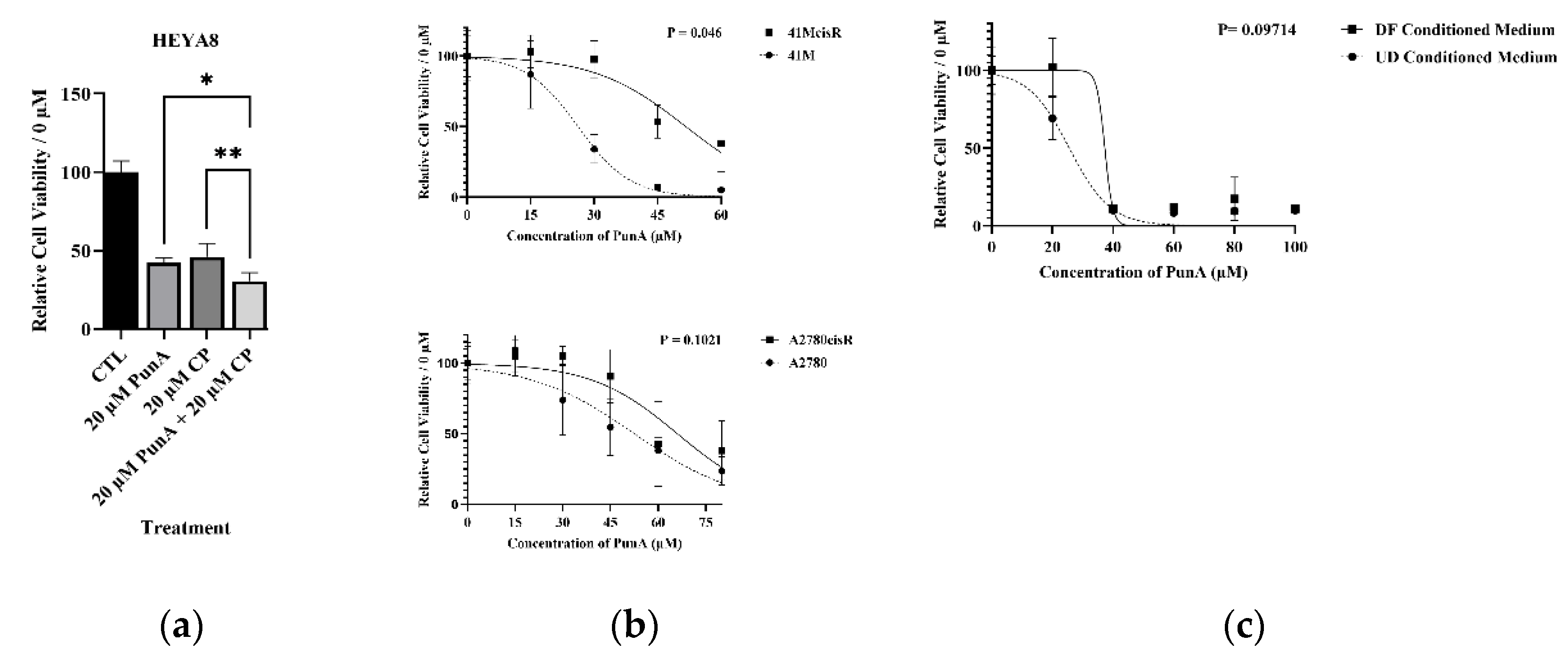
3.4. PunA Enhances Chemosensitivity of OC Cells
3.5. Chemo-Resistant OC Cells Exhibit Diminished Sensitivity to PunA
3.6. PunA Remains Effective in High Fat Environments
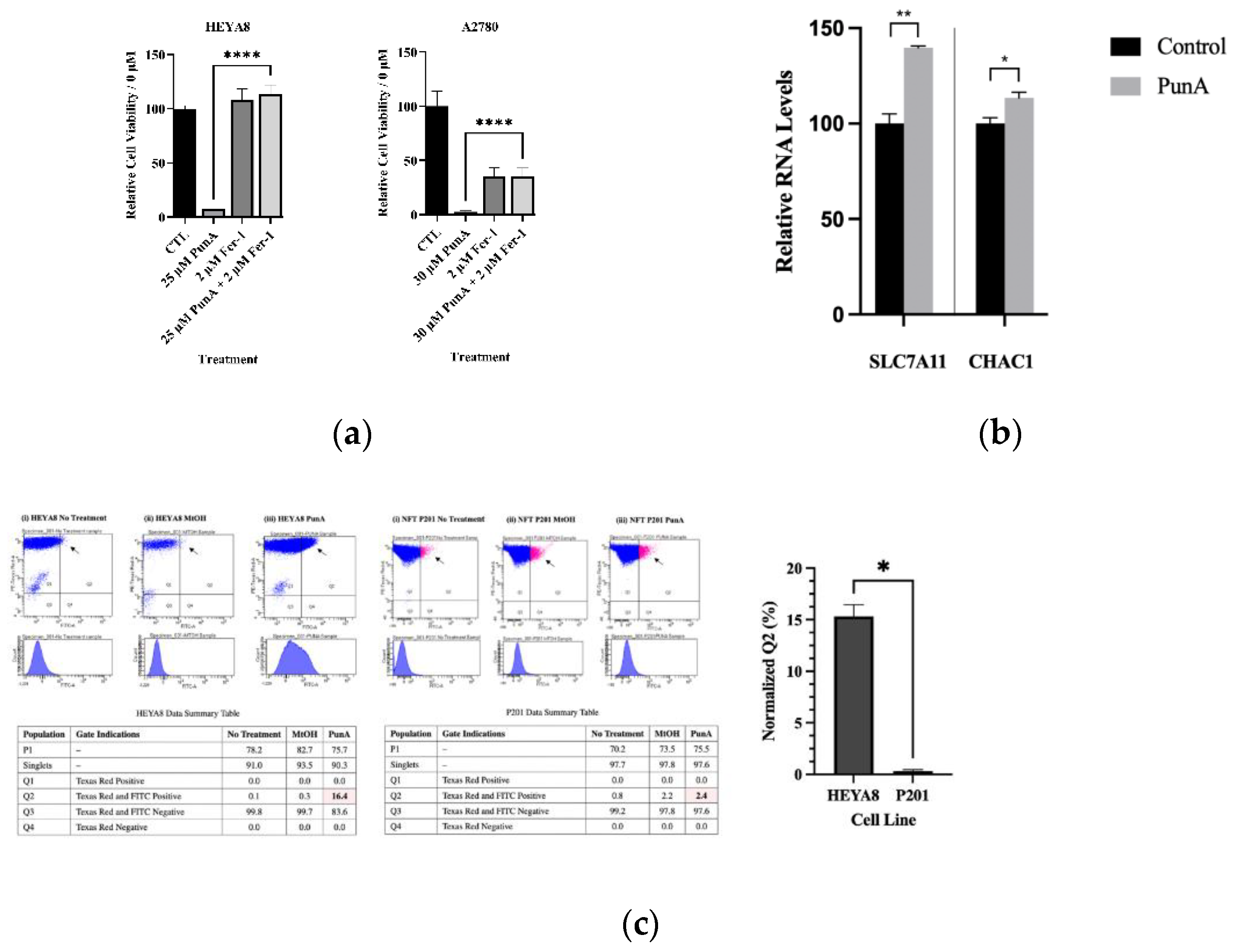
3.7. PunA Triggers Ferroptosis
3.8. PunA Selectively Increases Lipid Peroxidation in OC Cells
3.9. PunA Selectively Upregulates PPAR-γ Activity in OC Cells
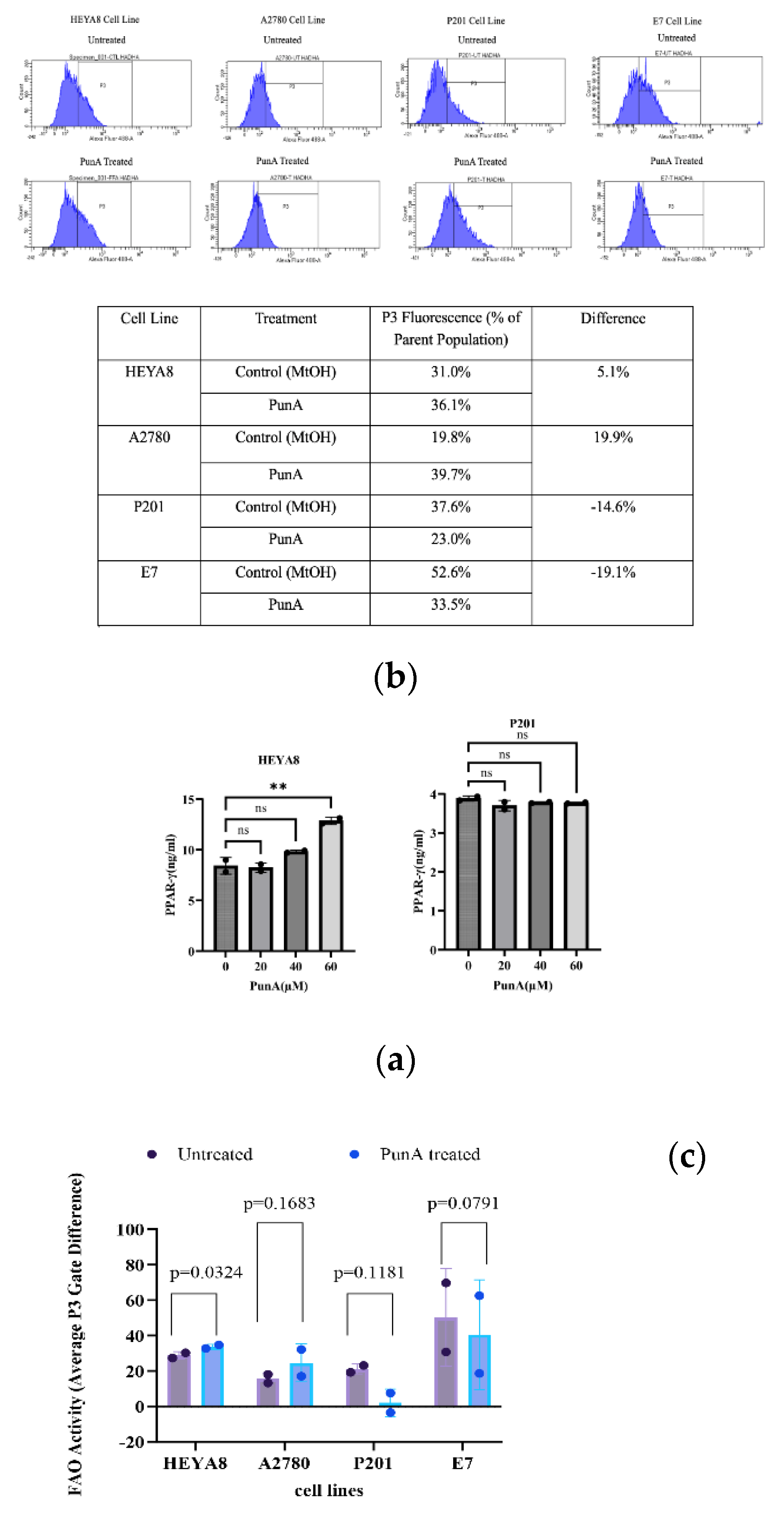
3.10. PunA Induces Differential Fatty Acid Oxidation Activity
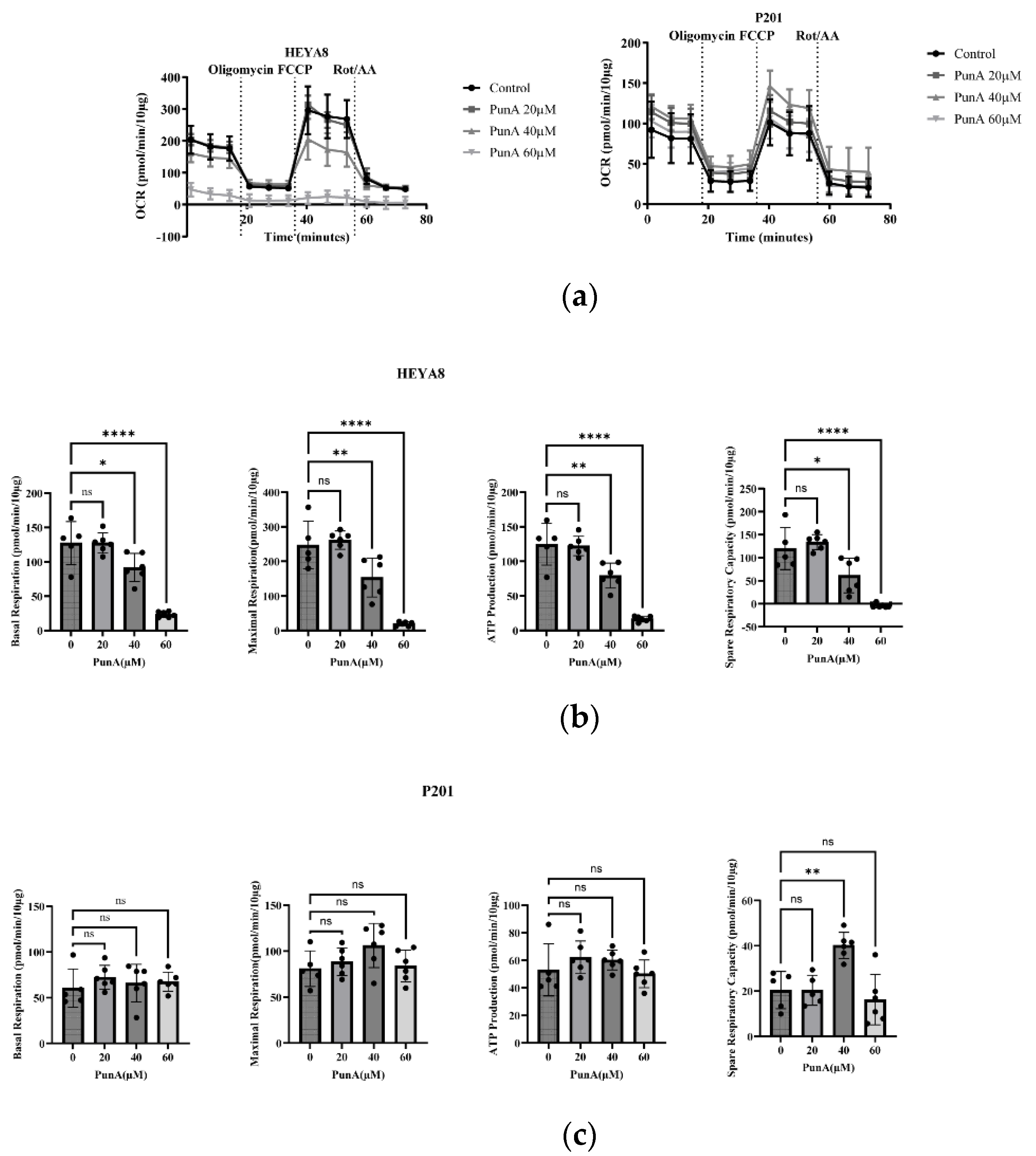
3.11. PunA Selectively Impairs Mitochondrial Respiration in OC Cells
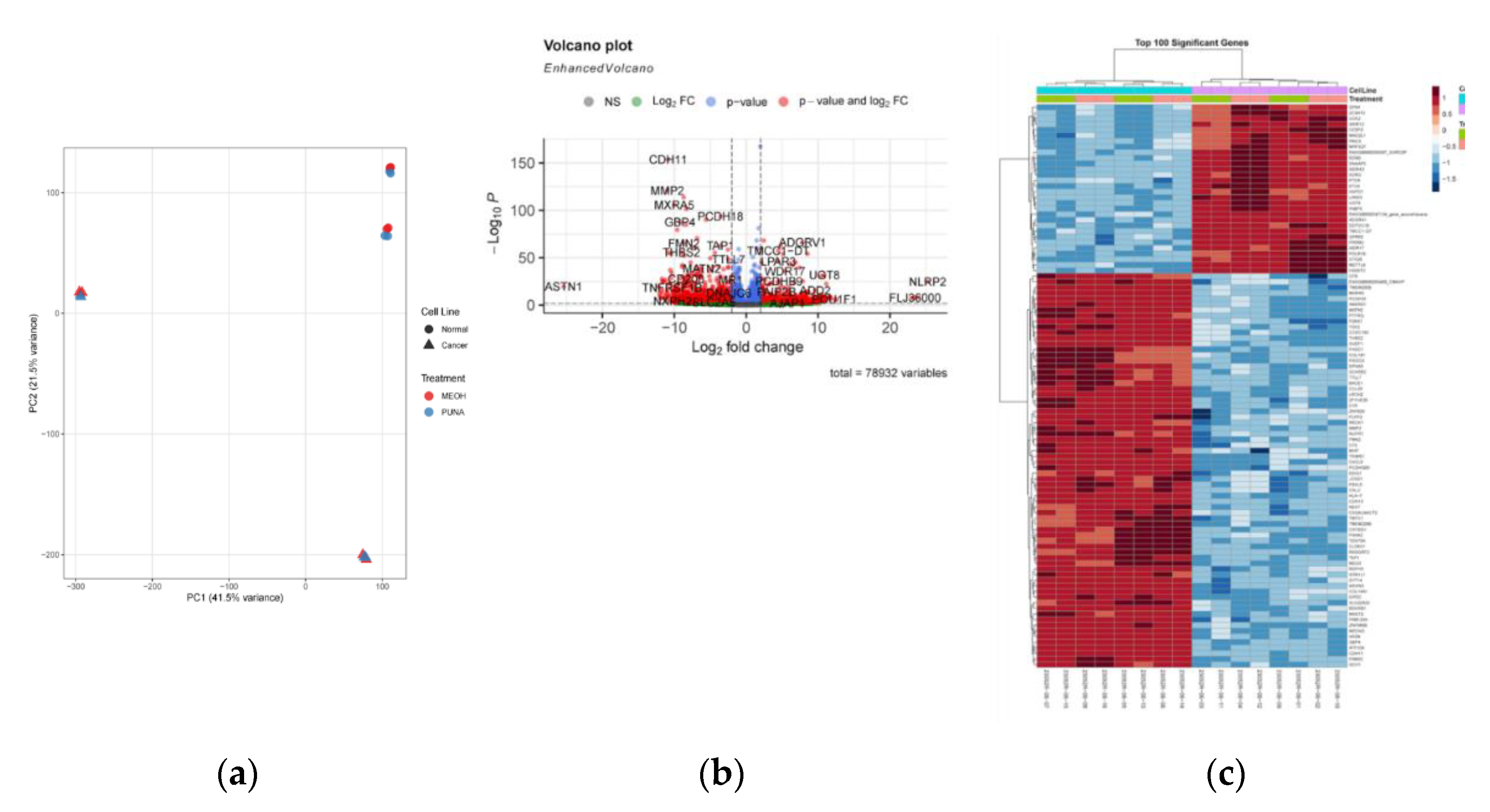
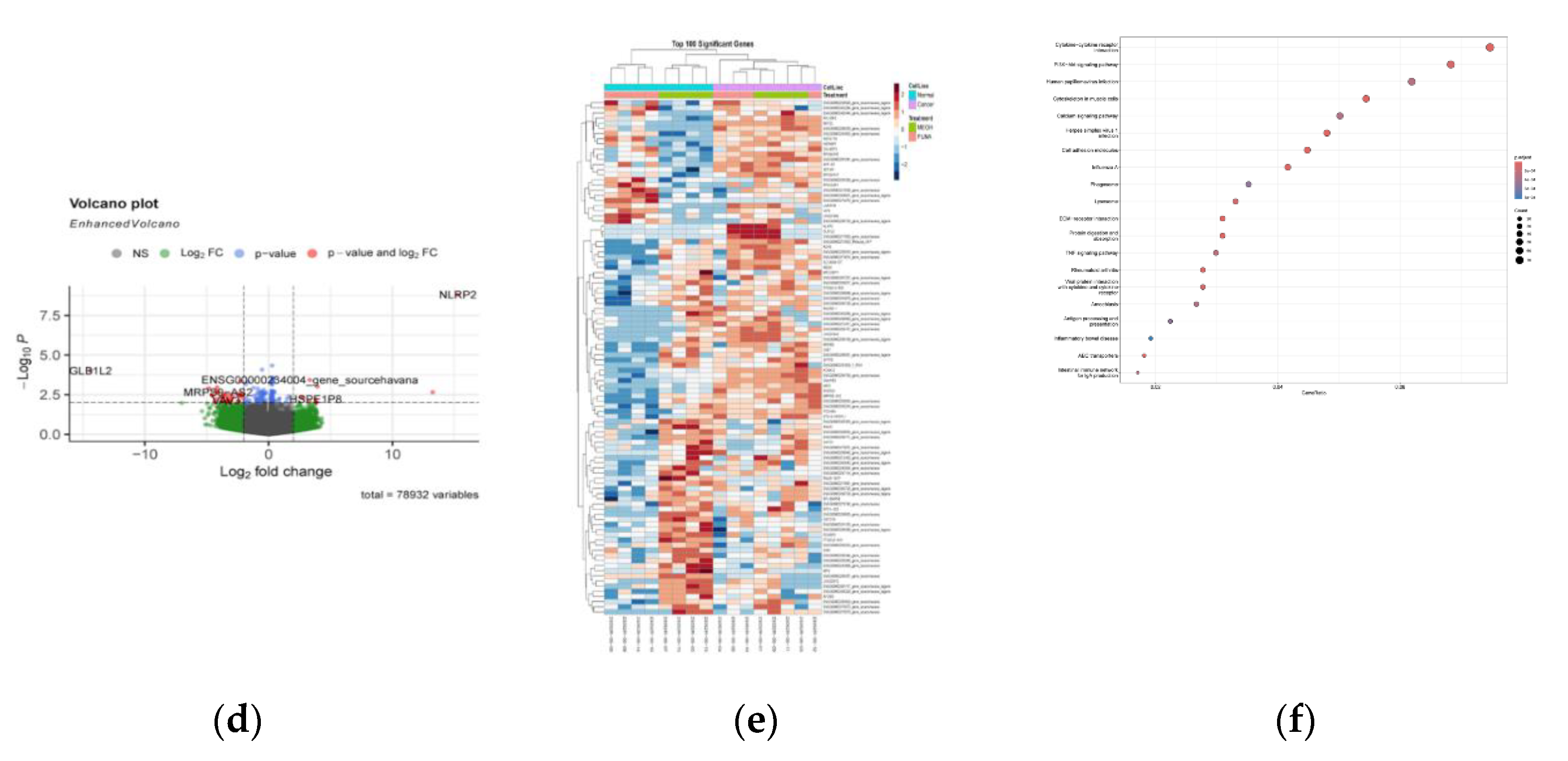
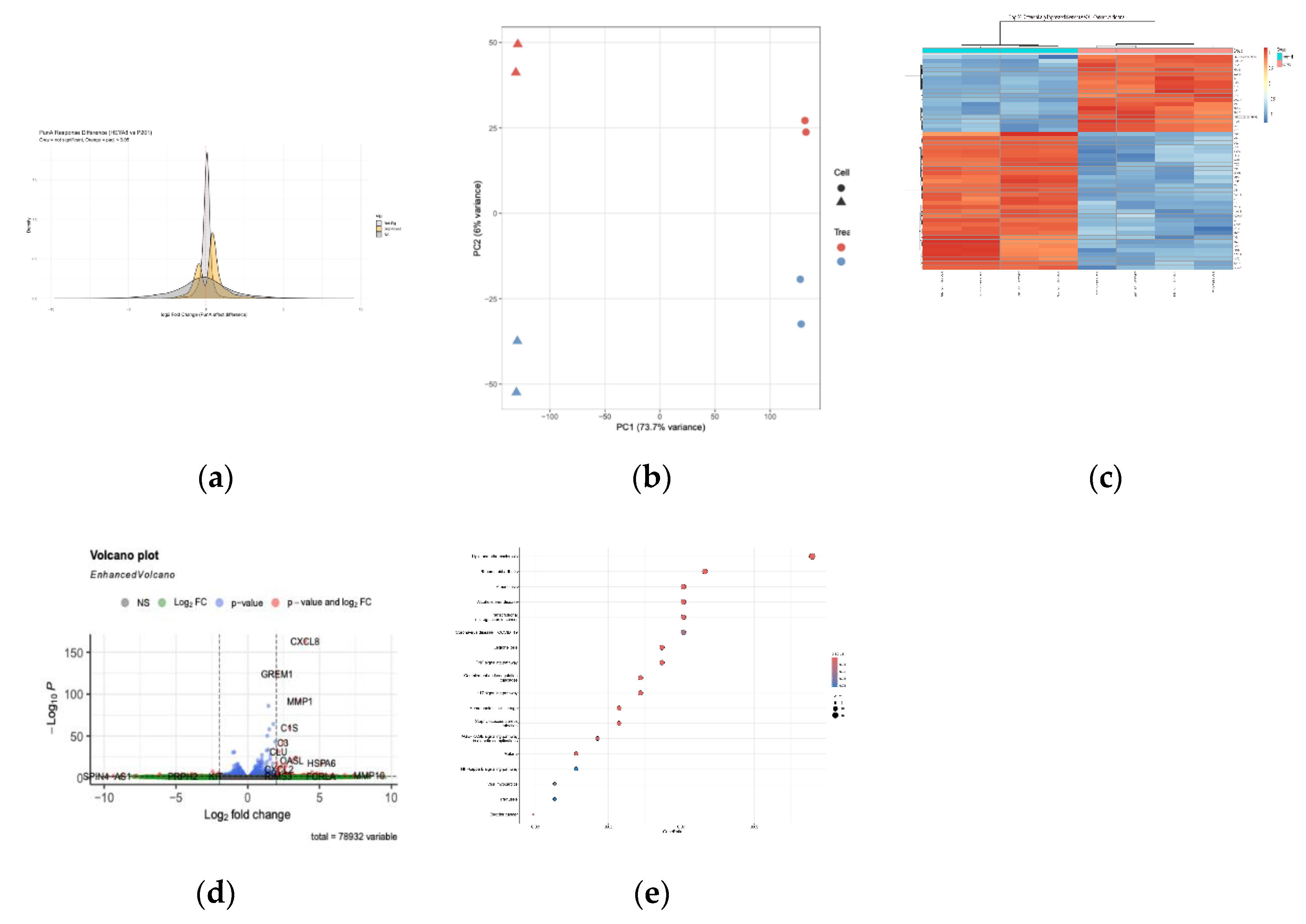
3.12. PunA Alters Gene Expression and Pathway Activation
3.13. PunA Enhances Inflammatory and Immune Gene Expression in OC Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| OC | Ovarian cancer |
| FFA | Free fatty acid |
| PunA | Punicic acid |
| α-ESA | α-Eleostearic acid |
| TME | Tumor microenvironment |
| ECM | Extracellular matrix |
| AT | Adipose tissue |
| AME | Adipose tumor microenvironment |
| CAAs | Cancer-associated adipocytes |
| NFT | Normal fallopian tube |
| PPAR-γ | Peroxisome proliferator-activated receptor gamma |
| IP | Intraperitoneal |
| HGSOC | High grade serous ovarian cancer |
| DF | Differentiated |
| UD | Undifferentiated |
| Fer-1 | Ferrostatin-1 |
| FAO | Fatty acid oxidation |
| HADHA | Hydroxyacyl-CoA dehydrogenase trifunctional multienzyme subunit alpha |
| PCA | Principle component analysis |
| DEGs | Differentially expressed genes |
| lncRNA | Long non-coding RNA |
| GO | Gene ontology |
| MHC | Major histocompatibility complex |
| ACSL4 | Acyl-CoA synthetase long-chain family member 4 |
| EMT | Epithelial-mesenchymal transition |
References
- Ovarian cancer. Available online: https://www.mayoclinic.org/diseases-conditions/ovarian-cancer/symptoms-causes/syc-20375941.
- Key Statistics for Ovarian Cancer. Accessed 19. Available online: https://www.cancer.org/cancer/types/ovarian-cancer/key-statistics.html#ovarian-cancer-estimates-for-2025.
- Ali, AT; Al-Ani, O; Al-Ani, F. Epidemiology and risk factors for ovarian cancer. Prz Menopauzalny 2023, 22(2), 93–104. [Google Scholar] [CrossRef] [PubMed]
- Loughran, EA; Leonard, AK; Hilliard, TS; et al. Aging Increases Susceptibility to Ovarian Cancer Metastasis in Murine Allograft Models and Alters Immune Composition of Peritoneal Adipose Tissue. Neoplasia 2018, 20(6), 621–631. [Google Scholar] [CrossRef]
- Mottini, C; Auciello, FR; Manni, I; et al. The cross-talk between the macro and micro-environment in precursor lesions of pancreatic cancer leads to new and promising circulating biomarkers. J Exp Clin Cancer Res. 2024, 43(1), 198. [Google Scholar] [CrossRef]
- Harper, EI; Sheedy, EF; Stack, MS. With Great Age Comes Great Metastatic Ability: Ovarian Cancer and the Appeal of the Aging Peritoneal Microenvironment. Cancers (Basel) 2018, 10(7). [Google Scholar] [CrossRef]
- Lengyel, E; Makowski, L; DiGiovanni, J; Kolonin, MG. Cancer as a Matter of Fat: The Crosstalk between Adipose Tissue and Tumors. Trends Cancer 2018, 4(5), 374–384. [Google Scholar] [CrossRef]
- Duarte Mendes, A; Freitas, AR; Vicente, R; et al. Adipocyte Microenvironment in Ovarian Cancer: A Critical Contributor? Int J Mol Sci 2023, 24(23). [Google Scholar] [CrossRef]
- Jiang, Y; Wang, C; Zhou, S. Targeting tumor microenvironment in ovarian cancer: Premise and promise. Biochim Biophys Acta Rev Cancer 2020, 1873(2), 188361. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q; Yang, J; Shen, H; Xu, W. Cancer-associated adipocytes in the ovarian cancer microenvironment. Am J Cancer Res. 2024, 14(7), 3259–3279. [Google Scholar] [CrossRef]
- Wu, Q; Li, B; Sun, S; Sun, S. Unraveling Adipocytes and Cancer Links: Is There a Role for Senescence? Front Cell Dev Biol. 2020, 8, 282. [Google Scholar] [CrossRef]
- Schaum, N; Lehallier, B; Hahn, O; et al. Ageing hallmarks exhibit organ-specific temporal signatures. Nature 2020, 583(7817), 596–602. [Google Scholar] [CrossRef] [PubMed]
- Feng, J; Rouse, CD; Taylor, L; et al. Regulation of Age-Related Lipid Metabolism in Ovarian Cancer. International Journal of Molecular Sciences 2025, 26(1), 320. [Google Scholar] [CrossRef]
- Dhar Dubey, KK; Sharma, G; Kumar, A. Conjugated Linolenic Acids: Implication in Cancer. J Agric Food Chem. 2019, 67(22), 6091–6101. [Google Scholar] [CrossRef]
- Yuan, G-f; Chen, X-E; Li, D. Conjugated Linolenic Acids and Their Bioactivities: A Review. Food & Function 2014, 5(7), 1360–1368. [Google Scholar] [CrossRef]
- Nugteren, DH; Christ-Hazelhof, E. Naturally occurring conjugated octadecatrienoic acids are strong inhibitors of prostaglandin biosynthesis. Prostaglandins 1987, 33(3), 403–17. [Google Scholar] [CrossRef] [PubMed]
- Gasmi, J; Sanderson, JT. Growth inhibitory, antiandrogenic, and pro-apoptotic effects of punicic acid in LNCaP human prostate cancer cells. J Agric Food Chem. 2010, 58(23), 12149–56. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, ME; Mizuno, NK; Schuster, T; Cleary, MP. Punicic acid is an omega-5 fatty acid capable of inhibiting breast cancer proliferation. Int J Oncol. 2010, 36(2), 421–6. [Google Scholar] [PubMed]
- Quitmeyer, B; Emelife, C; Klausner, H; Gbayisomore, O; Phelan, S. Differential Effects of Punicic Acid on Cytotoxicity and Peroxiredoxin Expression in MCF-7 Breast Cancer and MCF-10A Normal Cells. Anticancer Res. 2024, 44(11), 4751–4759. [Google Scholar] [CrossRef]
- Igarashi, M; Miyazawa, T. Newly recognized cytotoxic effect of conjugated trienoic fatty acids on cultured human tumor cells. Cancer Lett. 2000, 148(2), 173–9. [Google Scholar] [CrossRef]
- Zhang, T; Gao, Y; Mao, Y; et al. Growth inhibition and apoptotic effect of alpha-eleostearic acid on human breast cancer cells. J Nat Med. 2012, 66(1), 77–84. [Google Scholar] [CrossRef]
- Liu, H; Liu, S-L. Pharmacological Effects of Natural Components Against Ovarian Cancer and Mechanisms. In Ovarian Cancer: Molecular & Diagnostic Imaging and Treatment Strategies; Schatten, H, Ed.; Springer International Publishing, 2021; pp. 55–73. [Google Scholar]
- Chan, DW; Yung, MMH; Chan, Y-S; et al. MAP30 protein from Momordica charantia is therapeutic and has synergic activity with cisplatin against ovarian cancer in vivo by altering metabolism and inducing ferroptosis. Pharmacological Research 2020/11/01/ 2020, 161, 105157. [Google Scholar] [CrossRef]
- Pitchakarn, P; Umsumarng, S; Mapoung, S; et al. Kuguacin J isolated from bitter melon leaves modulates paclitaxel sensitivity in drug-resistant human ovarian cancer cells. Journal of Natural Medicines 2017/10/01 2017, 71(4), 693–702. [Google Scholar] [CrossRef]
- Yung, MMH; Ross, FA; Hardie, DG; et al. Bitter Melon (Momordica charantia) Extract Inhibits Tumorigenicity and Overcomes Cisplatin-Resistance in Ovarian Cancer Cells Through Targeting AMPK Signaling Cascade. Integrative Cancer Therapies 2016, 15(3), 376–389. [Google Scholar] [CrossRef] [PubMed]
- Song, HK; Hwang, DY. Use of C57BL/6N mice on the variety of immunological researches. Lab Anim Res. 2017, 33(2), 119–123. [Google Scholar] [CrossRef] [PubMed]
- Rose, PG. First-Line Chemotherapy for Ovarian Cancer: Inferences From Recent Studies. Oncologist 2016, 21(11), 1286–1290. [Google Scholar] [CrossRef]
- Prat, J; D'Angelo, E; Espinosa, I. Ovarian carcinomas: at least five different diseases with distinct histological features and molecular genetics. Human Pathology 2018/10/01/ 2018, 80, 11–27. [Google Scholar] [CrossRef]
- Hobbs, C; Huang, Z; Murphy, S; Berchuck, A. The function of matrix metalloproteinase 1 in ovarian cancer . Gynecologic Oncology 2019, 154, 80. [Google Scholar] [CrossRef]
- Romani, AMP. Cisplatin in cancer treatment. Biochemical Pharmacology 2022/12/01/ 2022, 206, 115323. [Google Scholar] [CrossRef] [PubMed]
- Abu Samaan, TM; Samec, M; Liskova, A; Kubatka, P; Büsselberg, D. Paclitaxel's Mechanistic and Clinical Effects on Breast Cancer. Biomolecules 2019, 9(12). [Google Scholar] [CrossRef]
- Baumann, KE; Siamakpour-Reihani, S; Dottino, J; et al. High-fat diet and obesity are associated with differential angiogenic gene expression in epithelial ovarian cancer. Gynecologic Oncology 2023, 179, 97–105. [Google Scholar] [CrossRef]
- Martiná Strnadová DT, and Isabell Kaczmarek. Utilizing the property of differentiated 3T3-L1 cells to produce fats in the cell culture medium, we induced cell differentiation by insulin induction. Protocols. Star Protocols. June 6, 2024;doi:https://star-protocols.cell.com/protocols/3504.
- Kim, JW; Lee, J-Y; Oh, M; Lee, E-W. An integrated view of lipid metabolism in ferroptosis revisited via lipidomic analysis. Experimental & Molecular Medicine 2023/08/01 2023, 55(8), 1620–1631. [Google Scholar] [CrossRef]
- Sun, D; Wang, L; Wu, Y; et al. Lipid metabolism in ferroptosis: mechanistic insights and therapeutic potential. Front Immunol. 2025, 16, 1545339. [Google Scholar] [CrossRef]
- Do, Q; Xu, L. How do different lipid peroxidation mechanisms contribute to ferroptosis? Cell Rep Phys Sci 2023, 4(12). [Google Scholar] [CrossRef] [PubMed]
- Cheng, HS; Yip, YS; Lim, EKY; Wahli, W; Tan, NS. PPARs and Tumor Microenvironment: The Emerging Roles of the Metabolic Master Regulators in Tumor Stromal-Epithelial Crosstalk and Carcinogenesis. Cancers (Basel) 2021, 13(9). [Google Scholar] [CrossRef] [PubMed]
- Ma, J; Wang, S; Zhang, P; et al. Emerging roles for fatty acid oxidation in cancer. Genes & Diseases 2025/07/01/ 2025, 12(4), 101491. [Google Scholar] [CrossRef]
- Wang, X; Song, H; Liang, J; Jia, Y; Zhang, Y. Abnormal expression of HADH, an enzyme of fatty acid oxidation, affects tumor development and prognosis (Review). Mol Med Rep. 2022, 26(6). [Google Scholar] [CrossRef]
- Peng, Y; Zhang, L; Zhang, M. Comprehensive Analysis of Genomic Instability Derived lncRNAs Prognostic Signature and the Associated Tumor Microenvironment in Glioma. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics 2026, 201(1), 64–88. [Google Scholar] [CrossRef]
- Zhang, X; Li, M; Sun, G; Bai, Y; Lv, D; Liu, C. MiR-563 restrains cell proliferation via targeting LIN28B in human lung cancer. Thoracic Cancer 2020, 11(1), 55–61. [Google Scholar] [CrossRef]
- Antonarakis, SE. Small nuclear RNA genes in Mendelian disorders. Nature Genetics 2025/12/04 2025. [Google Scholar] [CrossRef]
- Labidi-Galy, SI; Papp, E; Hallberg, D; et al. High grade serous ovarian carcinomas originate in the fallopian tube. Nat Commun 2017, 8(1), 1093. [Google Scholar] [CrossRef]
- Lin, A; Giuliano, CJ; Palladino, A; et al. Off-target toxicity is a common mechanism of action of cancer drugs undergoing clinical trials. Science Translational Medicine 2019, 11(509), eaaw8412. [Google Scholar] [CrossRef] [PubMed]
- Ma, LL; Liang, L; Zhou, D; Wang, SW. Tumor suppressor miR-424-5p abrogates ferroptosis in ovarian cancer through targeting ACSL4. Neoplasma 2021, 68(1), 165–173. [Google Scholar] [CrossRef]
- Jiang, X; Stockwell, BR; Conrad, M. Ferroptosis: mechanisms, biology and role in disease. Nature Reviews Molecular Cell Biology 2021/04/01 2021, 22(4), 266–282. [Google Scholar] [CrossRef]
- Vermonden, P; Martin, M; Glowacka, K; et al. Phospholipase PLA2G7 is complementary to GPX4 in mitigating punicic-acid-induced ferroptosis in prostate cancer cells. iScience 2024, 27(5). [Google Scholar] [CrossRef]
- Vermonden, P; Vancoppenolle, M; Dierge, E; et al. Punicic Acid Triggers Ferroptotic Cell Death in Carcinoma Cells. Nutrients 2021, 13(8). [Google Scholar] [CrossRef]
- Dasari, S; Bernard Tchounwou, P. Cisplatin in cancer therapy: Molecular mechanisms of action. European Journal of Pharmacology 2014/10/05/ 2014, 740, 364–378. [Google Scholar] [CrossRef] [PubMed]
- Duarte, D; Vale, N. Evaluation of synergism in drug combinations and reference models for future orientations in oncology. Current Research in Pharmacology and Drug Discovery 2022/01/01/ 2022, 3, 100110. [Google Scholar] [CrossRef]
- Alderden, RA; Hall, MD; Hambley, TW. The Discovery and Development of Cisplatin. Journal of Chemical Education 2006/05/01 2006, 83(5), 728. [Google Scholar] [CrossRef]
- Chehade, H; Tedja, R; Ramos, H; et al. Regulatory Role of the Adipose Microenvironment on Ovarian Cancer Progression. Cancers 2022, 14(9), 2267. [Google Scholar] [CrossRef] [PubMed]
- Li, Z; Fang, X; Wang, S. Omentum provides a special cell microenvironment for ovarian cancer. Cancer Rep (Hoboken) 2023, 6(10), e1858. [Google Scholar] [CrossRef] [PubMed]
- Li, J; Cao, F; Yin, H-l; et al. Ferroptosis: past, present and future. Cell Death & Disease 2020/02/03 2020, 11(2), 88. [Google Scholar] [CrossRef]
- Janani, C; Ranjitha Kumari, BD. PPAR gamma gene – A review. Diabetes & Metabolic Syndrome: Clinical Research & Reviews 2015/01/01/ 2015, 9(1), 46–50. [Google Scholar] [CrossRef]
- Krishnan, A; Nair, SA; Pillai, MR. Biology of PPAR gamma in cancer: a critical review on existing lacunae. Curr Mol Med. 2007, 7(6), 532–40. [Google Scholar] [CrossRef] [PubMed]
- Yang, L; Yuan, J; Liu, L; et al. α-linolenic acid inhibits human renal cell carcinoma cell proliferation through PPAR-γ activation and COX-2 inhibition. Oncol Lett. 2013/07/01 2013, 6(1), 197–202. [Google Scholar] [CrossRef] [PubMed]
- Gupta, RA; Brockman, JA; Sarraf, P; Willson, TM; DuBois, RN. Target Genes of Peroxisome Proliferator-activated Receptor γ in Colorectal Cancer Cells *. Journal of Biological Chemistry 2001, 276(32), 29681–29687. [Google Scholar] [CrossRef] [PubMed]
- Gou, Q; Gong, X; Jin, J; Shi, J; Hou, Y. Peroxisome proliferator-activated receptors (PPARs) are potential drug targets for cancer therapy. Oncotarget 2017, 8(36), 60704–60709. [Google Scholar] [CrossRef]
- He, Y; Sun, MM; Zhang, GG; et al. Targeting PI3K/Akt signal transduction for cancer therapy. Signal Transduct Target Ther 2021, 6(1), 425. [Google Scholar] [CrossRef]
- Janku, F; Wheler, JJ; Westin, SN; et al. PI3K/AKT/mTOR inhibitors in patients with breast and gynecologic malignancies harboring PIK3CA mutations. J Clin Oncol. 2012, 30(8), 777–82. [Google Scholar] [CrossRef]
- Fan, L; Lei, H; Lin, Y; et al. Identification of a Gene Set Correlated With Immune Status in Ovarian Cancer by Transcriptome-Wide Data Mining. Front Mol Biosci. 2021, 8, 670666. [Google Scholar] [CrossRef]
- Zhang, T; Ren, Y; Yang, P; Wang, J; Zhou, H. Cancer-associated fibroblasts in pancreatic ductal adenocarcinoma. Cell Death Dis. 2022, 13(10), 897. [Google Scholar] [CrossRef]
- Li, J; Sun, J; Zeng, Z; et al. Tumour-associated macrophages in gastric cancer: From function and mechanism to application. Clin Transl Med 2023, 13(8), e1386. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



